Assessing Gait in Parkinson’s Disease Using Wearable Motion Sensors: A Systematic Review
Abstract
:1. Introduction
1.1. Inertial Measuring Units
1.2. Clinical Application
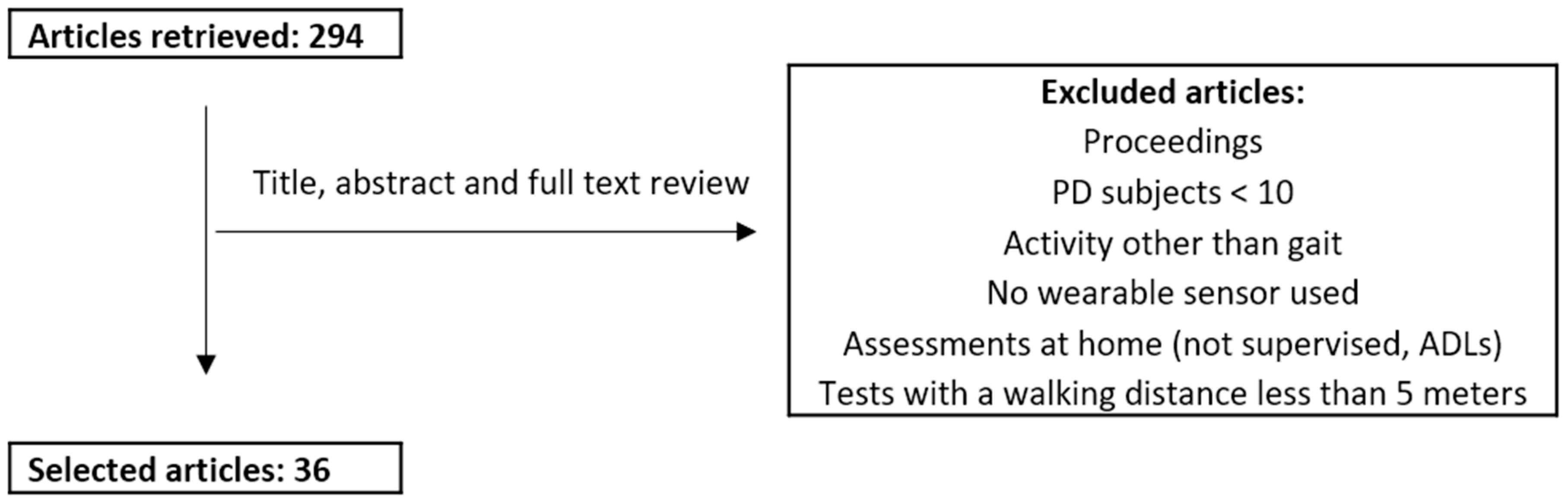
2. Materials and Methods
Review Process: Search Strategy and Selection Criteria
3. Results
3.1. Format and Disambiguation
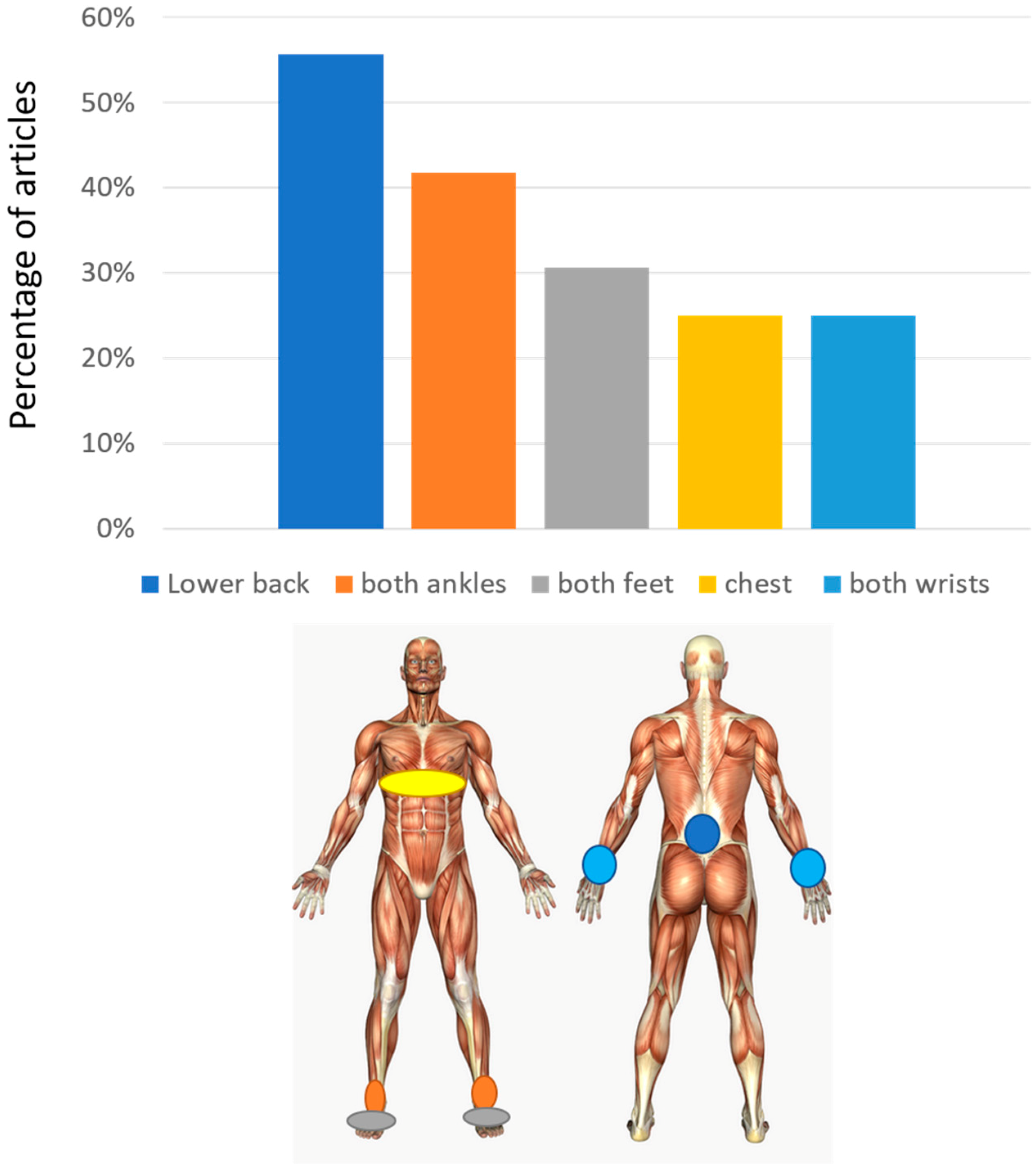
3.2. Sensor Number and Placement
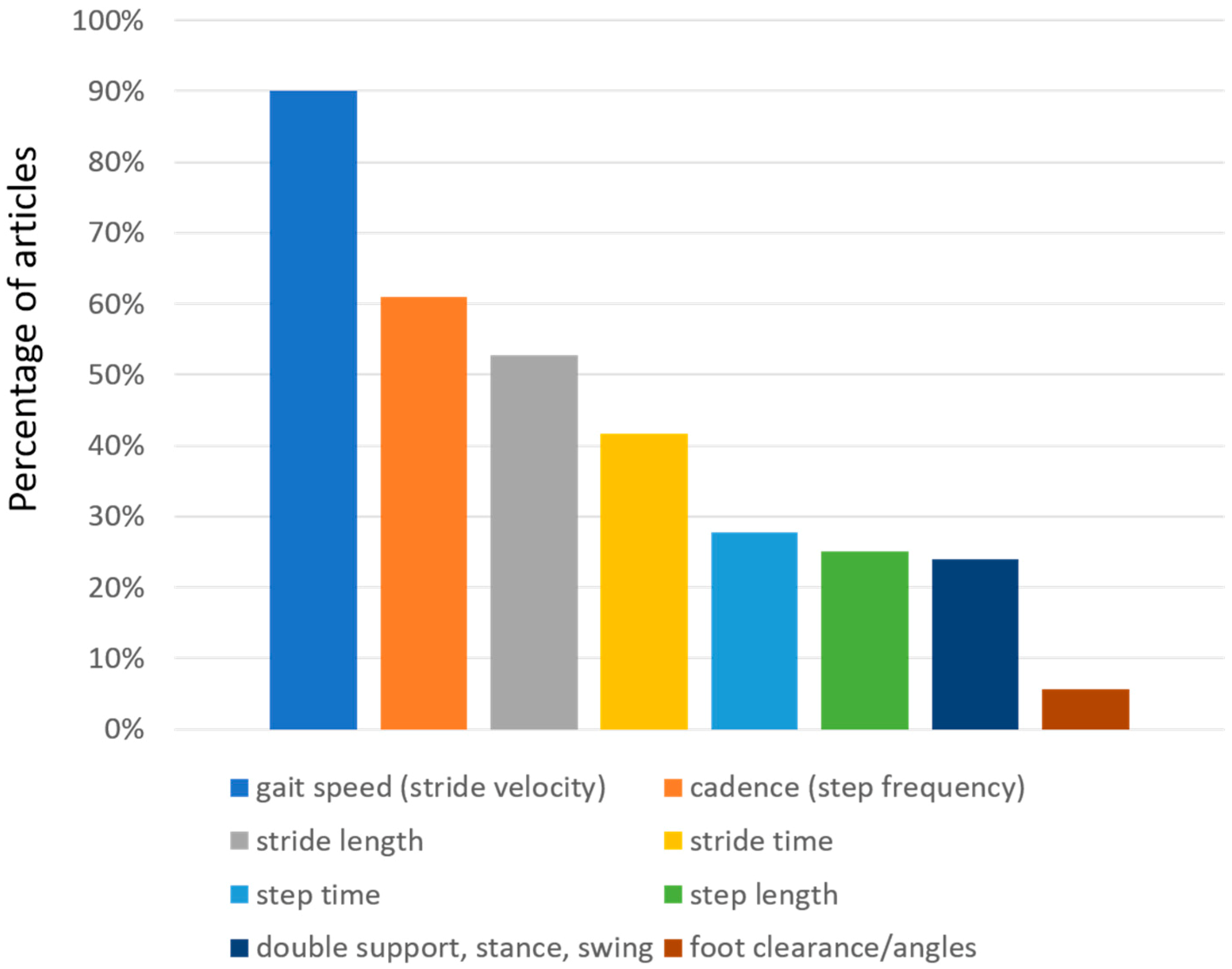
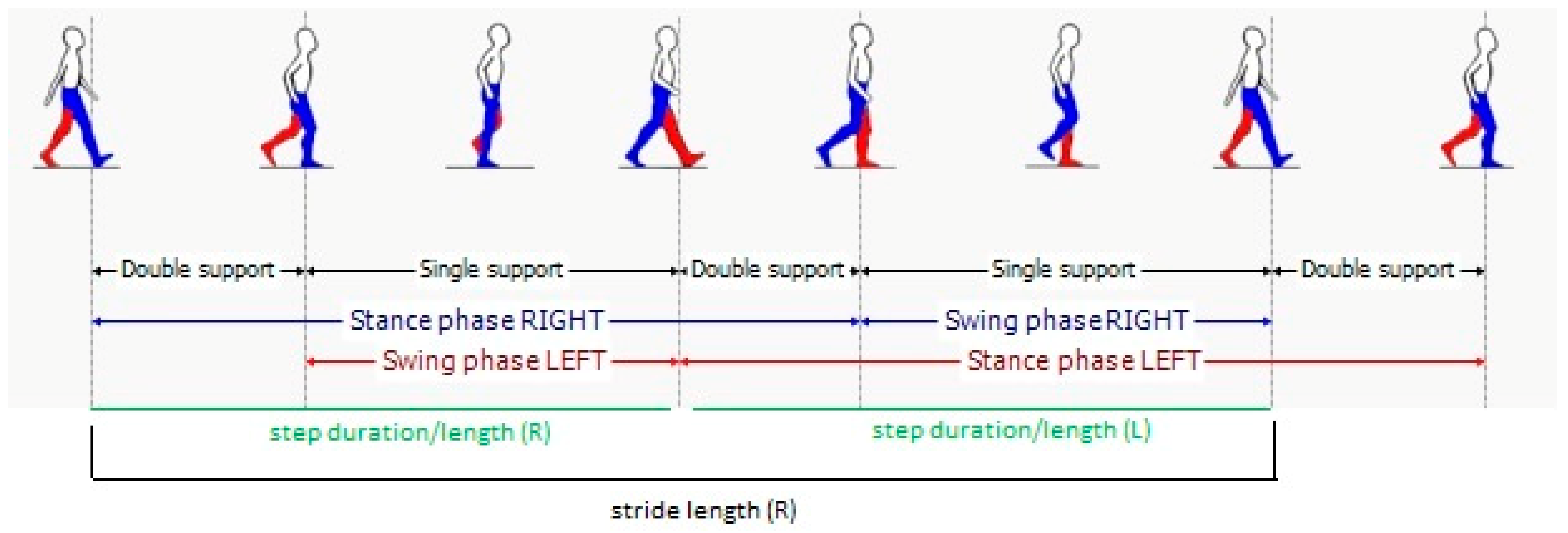
3.3. Spatio-Temporal Parameters
3.4. Characteristics of Patients
4. Discussion
5. Conclusions
Funding
Conflicts of Interest
References
- Hausdorff, J.M. Gait dynamics in Parkinson’s disease: Common and distinct behavior among stride length, gait variability, and fractal-like scaling. Chaos An Interdiscip. J. Nonlinear Sci. 2009, 19, 026113. [Google Scholar] [CrossRef] [PubMed]
- Zampieri, C.; Salarian, A.; Carlson-Kuhta, P.; Aminian, K.; Nutt, J.G.; Horak, F.B. The instrumented timed up and go test: potential outcome measure for disease modifying therapies in Parkinson’s disease. J. Neurol. Neurosurg. Psychiatry 2010, 81, 171–176. [Google Scholar] [CrossRef] [PubMed]
- Gómez-Esteban, J.C.; Zarranz, J.J.; Lezcano, E.; Tijero, B.; Luna, A.; Velasco, F.; Rouco, I.; Garamendi, I. Influence of Motor Symptoms upon the Quality of Life of Patients with Parkinson’s Disease. Eur. Neurol. 2007, 57, 161–165. [Google Scholar] [CrossRef] [PubMed]
- Soh, S.-E.; Morris, M.E.; McGinley, J.L. Determinants of health-related quality of life in Parkinson’s disease: A systematic review. Parkinsonism Relat. Disord. 2011, 17, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Patel, S.; Park, H.; Bonato, P.; Chan, L.; Rodgers, M. A review of wearable sensors and systems with application in rehabilitation. J. Neuroeng. Rehabil. 2012, 9, 21. [Google Scholar] [CrossRef] [PubMed]
- Schlachetzki, J.C.M.; Barth, J.; Marxreiter, F.; Gossler, J.; Kohl, Z.; Reinfelder, S.; Gassner, H.; Aminian, K.; Eskofier, B.M.; Winkler, J.; et al. Wearable sensors objectively measure gait parameters in Parkinson’s disease. PLoS ONE 2017, 12, e0183989. [Google Scholar] [CrossRef] [PubMed]
- Weiss, A.; Herman, T.; Giladi, N.; Hausdorff, J.M. Objective Assessment of Fall Risk in Parkinson’s Disease Using a Body-Fixed Sensor Worn for 3 Days. PLoS ONE 2014, 9, e96675. [Google Scholar] [CrossRef] [PubMed]
- Casamassima, F.; Ferrari, A.; Milosevic, B.; Ginis, P.; Farella, E.; Rocchi, L. A wearable system for gait training in subjects with Parkinson’s disease. Sensors (Basel). 2014, 14, 6229–6246. [Google Scholar] [CrossRef] [PubMed]
- Aich, S.; Pradhan, P.; Park, J.; Sethi, N.; Vathsa, V.; Kim, H.-C. A Validation Study of Freezing of Gait (FoG) Detection and Machine-Learning-Based FoG Prediction Using Estimated Gait Characteristics with a Wearable Accelerometer. Sensors 2018, 18, 3287. [Google Scholar] [CrossRef] [PubMed]
- Bernhard, F.P.; Sartor, J.; Bettecken, K.; Hobert, M.A.; Arnold, C.; Weber, Y.G.; Poli, S.; Margraf, N.G.; Schlenstedt, C.; Hansen, C.; et al. Wearables for gait and balance assessment in the neurological ward - study design and first results of a prospective cross-sectional feasibility study with 384 inpatients. BMC Neurol. 2018, 18, 114. [Google Scholar] [CrossRef] [PubMed]
- Caramia, C.; Torricelli, D.; Schmid, M.; Munoz-Gonzalez, A.; Gonzalez-Vargas, J.; Grandas, F.; Pons, J.L. IMU-Based Classification of Parkinson’s Disease From Gait: A Sensitivity Analysis on Sensor Location and Feature Selection. IEEE J. Biomed. Heal. Informatics 2018, 22, 1765–1774. [Google Scholar] [CrossRef] [PubMed]
- Pau, M.; Corona, F.; Pili, R.; Casula, C.; Guicciardi, M.; Cossu, G.; Murgia, M. Quantitative assessment of gait parameters in people with Parkinson’s disease in laboratory and clinical setting: Are the measures interchangeable? Neurol. Int. 2018, 10, 7729. [Google Scholar] [CrossRef] [PubMed]
- Zago, M.; Sforza, C.; Pacifici, I.; Cimolin, V.; Camerota, F.; Celletti, C.; Condoluci, C.; De Pandis, M.F.; Galli, M. Gait evaluation using inertial measurement units in subjects with Parkinson’s disease. J. Electromyogr. Kinesiol. 2018, 42, 44–48. [Google Scholar] [CrossRef] [PubMed]
- Bertoli, M.; Cereatti, A.; Trojaniello, D.; Avanzino, L.; Pelosin, E.; Del Din, S.; Rochester, L.; Ginis, P.; Bekkers, E.M.J.; Mirelman, A.; et al. Estimation of spatio-temporal parameters of gait from magneto-inertial measurement units: multicenter validation among Parkinson, mildly cognitively impaired and healthy older adults. Biomed. Eng. Online 2018, 17, 58. [Google Scholar] [CrossRef] [PubMed]
- Raccagni, C.; Gaßner, H.; Eschlboeck, S.; Boesch, S.; Krismer, F.; Seppi, K.; Poewe, W.; Eskofier, B.M.; Winkler, J.; Wenning, G.; et al. Sensor-based gait analysis in atypical parkinsonian disorders. Brain Behav. 2018, 8, e00977. [Google Scholar] [CrossRef] [PubMed]
- Montero-Odasso, M.; Pieruccini-Faria, F.; Bartha, R.; Black, S.E.; Finger, E.; Freedman, M.; Greenberg, B.; Grimes, D.A.; Hegele, R.A.; Hudson, C.; et al. Motor Phenotype in Neurodegenerative Disorders: Gait and Balance Platform Study Design Protocol for the Ontario Neurodegenerative Research Initiative (ONDRI). J. Alzheimer’s Dis. 2017, 59, 707–721. [Google Scholar] [CrossRef] [PubMed]
- Bettecken, K.; Bernhard, F.; Sartor, J.; Hobert, M.A.; Hofmann, M.; Gladow, T.; van Uem, J.M.T.; Liepelt-Scarfone, I.; Maetzler, W. No relevant association of kinematic gait parameters with Health-related Quality of Life in Parkinson’s disease. PLoS ONE 2017, 12, e0176816. [Google Scholar] [CrossRef] [PubMed]
- Gougeon, M.A.; Zhou, L.; Nantel, J. Nordic Walking improves trunk stability and gait spatial-temporal characteristics in people with Parkinson disease. NeuroRehabilitation 2017, 41, 205–210. [Google Scholar] [CrossRef] [PubMed]
- de Souza Fortaleza, A.C.; Mancini, M.; Carlson-Kuhta, P.; King, L.A.; Nutt, J.G.; Chagas, E.F.; Freitas, I.F.; Horak, F.B. Dual task interference on postural sway, postural transitions and gait in people with Parkinson’s disease and freezing of gait. Gait Posture 2017, 56, 76–81. [Google Scholar] [CrossRef] [PubMed]
- Ginis, P.; Heremans, E.; Ferrari, A.; Dockx, K.; Canning, C.G.; Nieuwboer, A. Prolonged Walking with a Wearable System Providing Intelligent Auditory Input in People with Parkinson’s Disease. Front. Neurol. 2017, 8, 128. [Google Scholar] [CrossRef] [PubMed]
- Warlop, T.; Detrembleur, C.; Buxes Lopez, M.; Stoquart, G.; Lejeune, T.; Jeanjean, A. Does Nordic Walking restore the temporal organization of gait variability in Parkinson’s disease? J. Neuroeng. Rehabil. 2017, 14, 17. [Google Scholar] [CrossRef] [PubMed]
- Micó-Amigo, M.E.; Kingma, I.; Faber, G.S.; Kunikoshi, A.; van Uem, J.M.T.; van Lummel, R.C.; Maetzler, W.; van Dieën, J.H. Is the Assessment of 5 Meters of Gait with a Single Body-Fixed-Sensor Enough to Recognize Idiopathic Parkinson’s Disease-Associated Gait? Ann. Biomed. Eng. 2017, 45, 1266–1278. [Google Scholar] [CrossRef] [PubMed]
- Curtze, C.; Nutt, J.G.; Carlson-Kuhta, P.; Mancini, M.; Horak, F.B. Objective Gait and Balance Impairments Relate to Balance Confidence and Perceived Mobility in People With Parkinson Disease. Phys. Ther. 2016, 96, 1734–1743. [Google Scholar] [CrossRef] [PubMed]
- Ireland, D.; Wang, Z.; Lamont, R.; Liddle, J. Classification of Movement of People with Parkinsons Disease Using Wearable Inertial Movement Units and Machine Learning. Stud. Health Technol. Inform. 2016, 227, 61–66. [Google Scholar] [PubMed]
- Hatanaka, N.; Sato, K.; Hishikawa, N.; Takemoto, M.; Ohta, Y.; Yamashita, T.; Abe, K. Comparative Gait Analysis in Progressive Supranuclear Palsy and Parkinson’s Disease. Eur. Neurol. 2016, 75, 282–289. [Google Scholar] [CrossRef] [PubMed]
- Horak, F.B.; Mancini, M.; Carlson-Kuhta, P.; Nutt, J.G.; Salarian, A. Balance and Gait Represent Independent Domains of Mobility in Parkinson Disease. Phys. Ther. 2016, 96, 1364–1371. [Google Scholar] [CrossRef] [PubMed]
- Elshehabi, M.; Maier, K.S.; Hasmann, S.E.; Nussbaum, S.; Herbst, H.; Heger, T.; Berg, D.; Hobert, M.A.; Maetzler, W. Limited Effect of Dopaminergic Medication on Straight Walking and Turning in Early-to-Moderate Parkinson’s Disease during Single and Dual Tasking. Front. Aging Neurosci. 2016, 8, 4. [Google Scholar] [CrossRef] [PubMed]
- Nero, H.; Benka Wallén, M.; Franzén, E.; Ståhle, A.; Hagströmer, M. Accelerometer Cut Points for Physical Activity Assessment of Older Adults with Parkinson’s Disease. PLoS ONE 2015, 10, e0135899. [Google Scholar] [CrossRef] [PubMed]
- Ferrari, A.; Ginis, P.; Hardegger, M.; Casamassima, F.; Rocchi, L.; Chiari, L. A Mobile Kalman-Filter Based Solution for the Real-Time Estimation of Spatio-Temporal Gait Parameters. IEEE Trans. Neural Syst. Rehabil. Eng. 2016, 24, 764–773. [Google Scholar] [CrossRef] [PubMed]
- Trojaniello, D.; Ravaschio, A.; Hausdorff, J.M.; Cereatti, A. Comparative assessment of different methods for the estimation of gait temporal parameters using a single inertial sensor: application to elderly, post-stroke, Parkinson’s disease and Huntington’s disease subjects. Gait Posture 2015, 42, 310–316. [Google Scholar] [CrossRef] [PubMed]
- Curtze, C.; Nutt, J.G.; Carlson-Kuhta, P.; Mancini, M.; Horak, F.B. Levodopa Is a Double-Edged Sword for Balance and Gait in People With Parkinson’s Disease. Mov. Disord. 2015, 30, 1361–1370. [Google Scholar] [CrossRef] [PubMed]
- Del Din, S.; Godfrey, A.; Rochester, L. Validation of an Accelerometer to Quantify a Comprehensive Battery of Gait Characteristics in Healthy Older Adults and Parkinson’s Disease: Toward Clinical and at Home Use. IEEE J. Biomed. Heal. Informatics 2016, 20, 838–847. [Google Scholar] [CrossRef] [PubMed]
- Brodie, M.A.D.; Canning, C.G.; Beijer, T.R.; Lord, S.R. Uncontrolled head oscillations in people with Parkinson’s disease may reflect an inability to respond to perturbations while walking. Physiol. Meas. 2015, 36, 873–881. [Google Scholar] [CrossRef] [PubMed]
- Djurić-Jovicić, M.D.; Jovicić, N.S.; Radovanović, S.M.; Kresojević, N.D.; Kostić, V.S.; Popović, M.B. Quantitative and qualitative gait assessments in Parkinson’s disease patients. Vojnosanit. Pregl. 2014, 71, 809–816. [Google Scholar] [CrossRef] [PubMed]
- Herman, T.; Weiss, A.; Brozgol, M.; Giladi, N.; Hausdorff, J.M. Gait and balance in Parkinson’s disease subtypes: objective measures and classification considerations. J. Neurol. 2014, 261, 2401–2410. [Google Scholar] [CrossRef] [PubMed]
- Esser, P.; Dawes, H.; Collett, J.; Howells, K. Insights into gait disorders: Walking variability using phase plot analysis, Parkinson’s disease. Gait Posture 2013, 38, 648–652. [Google Scholar] [CrossRef] [PubMed]
- Palmerini, L.; Mellone, S.; Avanzolini, G.; Valzania, F.; Chiari, L. Quantification of Motor Impairment in Parkinson’s Disease Using an Instrumented Timed Up and Go Test. IEEE Trans. Neural Syst. Rehabil. Eng. 2013, 21, 664–673. [Google Scholar] [CrossRef] [PubMed]
- Esser, P.; Dawes, H.; Collett, J.; Feltham, M.G.; Howells, K. Assessment of spatio-temporal gait parameters using inertial measurement units in neurological populations. Gait Posture 2011, 34, 558–560. [Google Scholar] [CrossRef] [PubMed]
- Espay, A.J.; Baram, Y.; Dwivedi, A.K.; Shukla, R.; Gartner, M.; Gaines, L.; Duker, A.P.; Revilla, F.J. At-home training with closed-loop augmented-reality cueing device for improving gait in patients with Parkinson disease. J. Rehabil. Res. Dev. 2010, 47, 573–581. [Google Scholar] [CrossRef] [PubMed]
- Salarian, A.; Horak, F.B.; Zampieri, C.; Carlson-Kuhta, P.; Nutt, J.G.; Aminian, K. iTUG, a sensitive and reliable measure of mobility. IEEE Trans. Neural Syst. Rehabil. Eng. 2010, 18, 303–310. [Google Scholar] [CrossRef] [PubMed]
- Rochester, L.; Baker, K.; Hetherington, V.; Jones, D.; Willems, A.-M.; Kwakkel, G.; Van Wegen, E.; Lim, I.; Nieuwboer, A. Evidence for motor learning in Parkinson’s disease: Acquisition, automaticity and retention of cued gait performance after training with external rhythmical cues. Brain Res. 2010, 1319, 103–111. [Google Scholar] [CrossRef] [PubMed]
- Lowry, K.A.; Smiley-Oyen, A.L.; Carrel, A.J.; Kerr, J.P. Walking stability using harmonic ratios in Parkinson’s disease. Mov. Disord. 2009, 24, 261–267. [Google Scholar] [CrossRef] [PubMed]
- Warlop, T.; Detrembleur, C.; Stoquart, G.; Lejeune, T.; Jeanjean, A. Gait Complexity and Regularity Are Differently Modulated by Treadmill Walking in Parkinson’s Disease and Healthy Population. Front. Physiol. 2018, 9, 68. [Google Scholar] [CrossRef] [PubMed]
- Hafer, J.F.; Boyer, K.A. Age related differences in segment coordination and its variability during gait. Gait Posture 2018, 62, 92–98. [Google Scholar] [CrossRef] [PubMed]
- Djaldetti, R.; Ziv, I.; Melamed, E. The mystery of motor asymmetry in Parkinson’s disease. Lancet Neurol. 2006, 5, 796–802. [Google Scholar] [CrossRef]

| Ref | # PD subjects | H&Y stage | IMUs on both Ankles or on both Tibias | IMUs on both Feet | IMU on Lower Back | Other Locations (#IMUs) | # IMUs | Gait speed (stride velocity) | Cadence (or Step Frequency) | Stride Length | Stride Length Variability | Stride Time (Gait Cycle Time) | Stride Time Variability (Gait Cycle Time Variability) | Step Length | Step Length Variability | Step Time | Step Time Variability | Asymmetry Right-Left | Double Support (Time or %) | Stance (Time or %) | Swing (Time or %) | Foot Clearance | Heel-Strike and Toe-Off Angles |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| [9] | 51 | 2–4 | knees (2) | 2 | x | x | x | x | x | ||||||||||||||
| [10] | 51 | x | x | 3 | x | x | x | ||||||||||||||||
| [11] | 27 | 1–3 | x | x | x | thighs (2), chest (1) | 8 | x | x | x | x | x | x | x | x | ||||||||
| [12] | 50 | 1–3 | x | 2 | x | x | x | x | x | x | |||||||||||||
| [13] | 22 | 2.5–3.5 | x | 2 | x | x | x | x | x | x | x | x | |||||||||||
| [14] | 125 | x | 2 | x | x | x | x | x | x | x | |||||||||||||
| [15] | 50 | 2–3 | x | 1 | x | x | x | x | x | ||||||||||||||
| [6] | 190 | 2.12 ± 0.06 | x | 2 | x | x | x | x | x | x | x | x | |||||||||||
| [16] | 140 | x | hip (1) | 3 | x | x | |||||||||||||||||
| [17] | 43 | x | x | 3 | x | x | x | x | |||||||||||||||
| [18] | 12 | 1–3 | x | x | wrists (2), chest (1) | 6 | x | x | x | x | |||||||||||||
| [19] | 56 | x | x | x | wrist (2), chest (1) | 8 | x | x | |||||||||||||||
| [20] | 28 | 2.35 ± 0.5 | x | x | x | wrist (2) | 7 | x | x | x | x | ||||||||||||
| [21] | 14 | 1–3 | ankle (1) | 1 | x | x | x | x | |||||||||||||||
| [23] | 104 | 2.5 ± 0.6 | x | x | wrists (2), chest (1) | 6 | x | x | x | x | x | x | x | ||||||||||
| [24] | 14 | 1.77 ± 0.44 | x | x | wrists (2) | 5 | x | x | x | ||||||||||||||
| [25] | 124 | 1–4: 1 (13), 2 (31), 3 (68), 4 (12). | waist (1) | 1 | x | x | x | x | x | ||||||||||||||
| [26] | 100 | ON 2.33 ± 0.53, OFF 2.51 ± 0.57 | x | x | wrists (2), chest (1). | 6 | x | x | |||||||||||||||
| [27] | 39 | 2–3 | x | x | 3 | x | x | x | x | x | |||||||||||||
| [28] | 30 | 2–3: 2 (15), 3 (15) | hip (1) | 1 | x | ||||||||||||||||||
| [29] | 16 | 1–3: 1 (2), 2(8), 3 (6) | x | 2 | x | x | x | ||||||||||||||||
| [30] | 10 | x | 1 | x | x | x | x | x | |||||||||||||||
| [31] | 104 | 2–4: 2 (52), 3–4 (52) | x | x | wrists (2), chest (1) | 6 | x | x | x | x | |||||||||||||
| [32] | 30 | 1–3: 1 (8), 2(20), 3 (2) | x | 1 | x | x | x | x | x | x | x | ||||||||||||
| [33] | 10 | head (1) | 1 | x | x | x | |||||||||||||||||
| [34] | 12 | 2–4 | x | x | thighs (2) | 6 | x | x | x | x | x | x | x | x | |||||||||
| [35] | 110 | 1–4 | x | 1 | x | x | x | x | |||||||||||||||
| [36] | 14 | x | 1 | x | x | x | |||||||||||||||||
| [37] | 20 | 1.5–2.5: 1.5 (1), 2 (1), 2.5 (18) | x | 1 | x | x | |||||||||||||||||
| [38] | 24 | x | 1 | x | x | x | |||||||||||||||||
| [39] | 13 | x | 1 | x | x | x | |||||||||||||||||
| [40] | 12 | 1–2.5 | x | wrists (2), thighs (2), chest (1) | 7 | x | x | x | |||||||||||||||
| [41] | 153 | 2–4: 2 (71), 3 (64), 4 (18) | legs (2), chest (3) | 5 | x | x | x | ||||||||||||||||
| [2] | 12 | 1–2.5 | x | wrists (2), chest (1) | 5 | x | x | x | x | x | x | x | |||||||||||
| [42] | 11 | 1–3 | x | 1 | x | x | x | x | x | x | |||||||||||||
| Sum | 15 | 11 | 20 | 32 | 22 | 19 | 2 | 15 | 8 | 9 | 3 | 10 | 4 | 6 | 9 | 9 | 8 | 2 | 2 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Brognara, L.; Palumbo, P.; Grimm, B.; Palmerini, L. Assessing Gait in Parkinson’s Disease Using Wearable Motion Sensors: A Systematic Review. Diseases 2019, 7, 18. https://doi.org/10.3390/diseases7010018
Brognara L, Palumbo P, Grimm B, Palmerini L. Assessing Gait in Parkinson’s Disease Using Wearable Motion Sensors: A Systematic Review. Diseases. 2019; 7(1):18. https://doi.org/10.3390/diseases7010018
Chicago/Turabian StyleBrognara, Lorenzo, Pierpaolo Palumbo, Bernd Grimm, and Luca Palmerini. 2019. "Assessing Gait in Parkinson’s Disease Using Wearable Motion Sensors: A Systematic Review" Diseases 7, no. 1: 18. https://doi.org/10.3390/diseases7010018
APA StyleBrognara, L., Palumbo, P., Grimm, B., & Palmerini, L. (2019). Assessing Gait in Parkinson’s Disease Using Wearable Motion Sensors: A Systematic Review. Diseases, 7(1), 18. https://doi.org/10.3390/diseases7010018








