Synthesis and Characterization of Polyaniline-Based Composites for Electromagnetic Compatibility of Electronic Devices
Abstract
1. Introduction
2. Materials and Methods
2.1. Synthesis of PANI Films
2.2. Synthesis of Gelatin–PANI Composites
2.3. Characterization Techniques
3. Results and Discussion
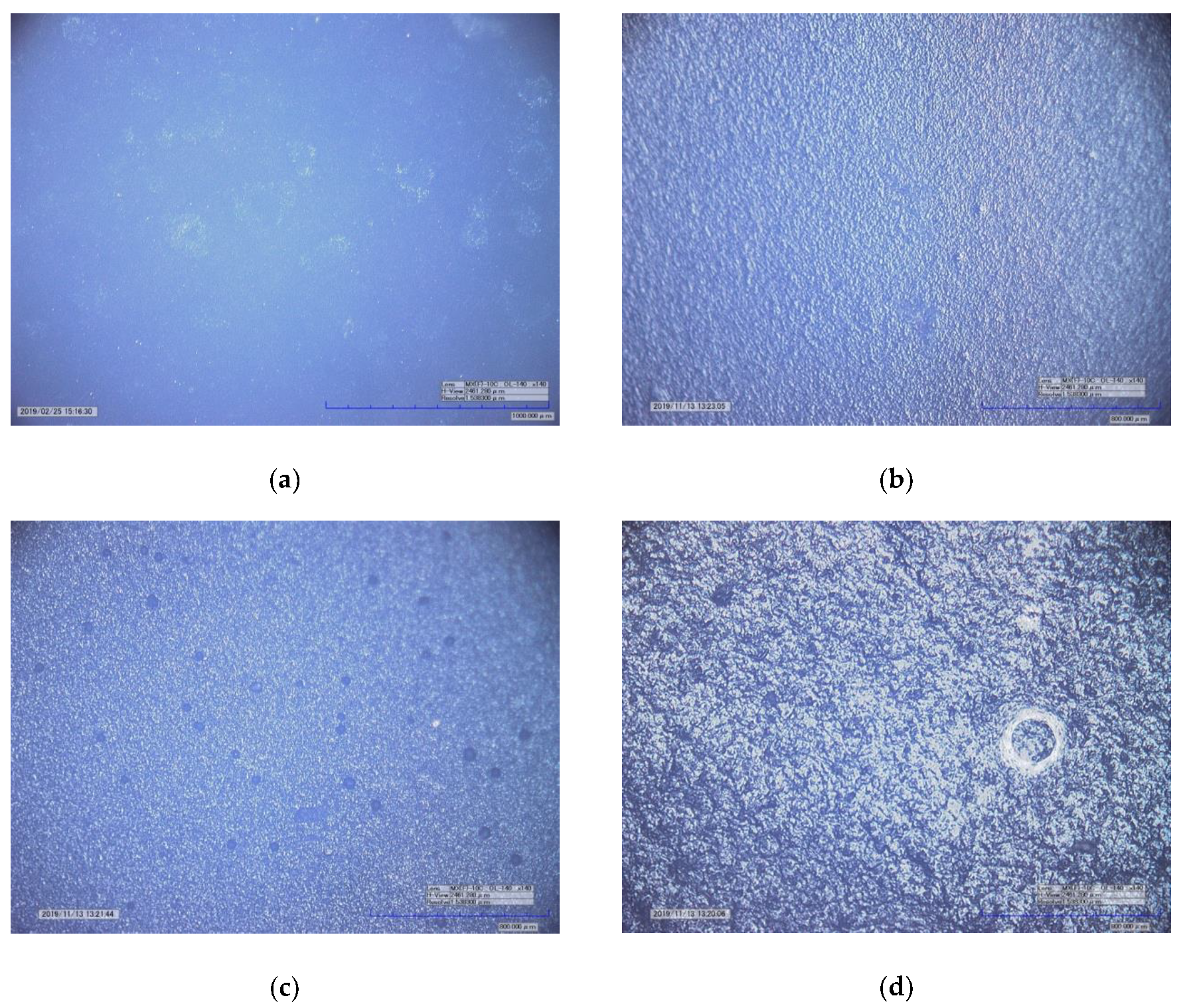
3.1. Surface Morphology of the Samples
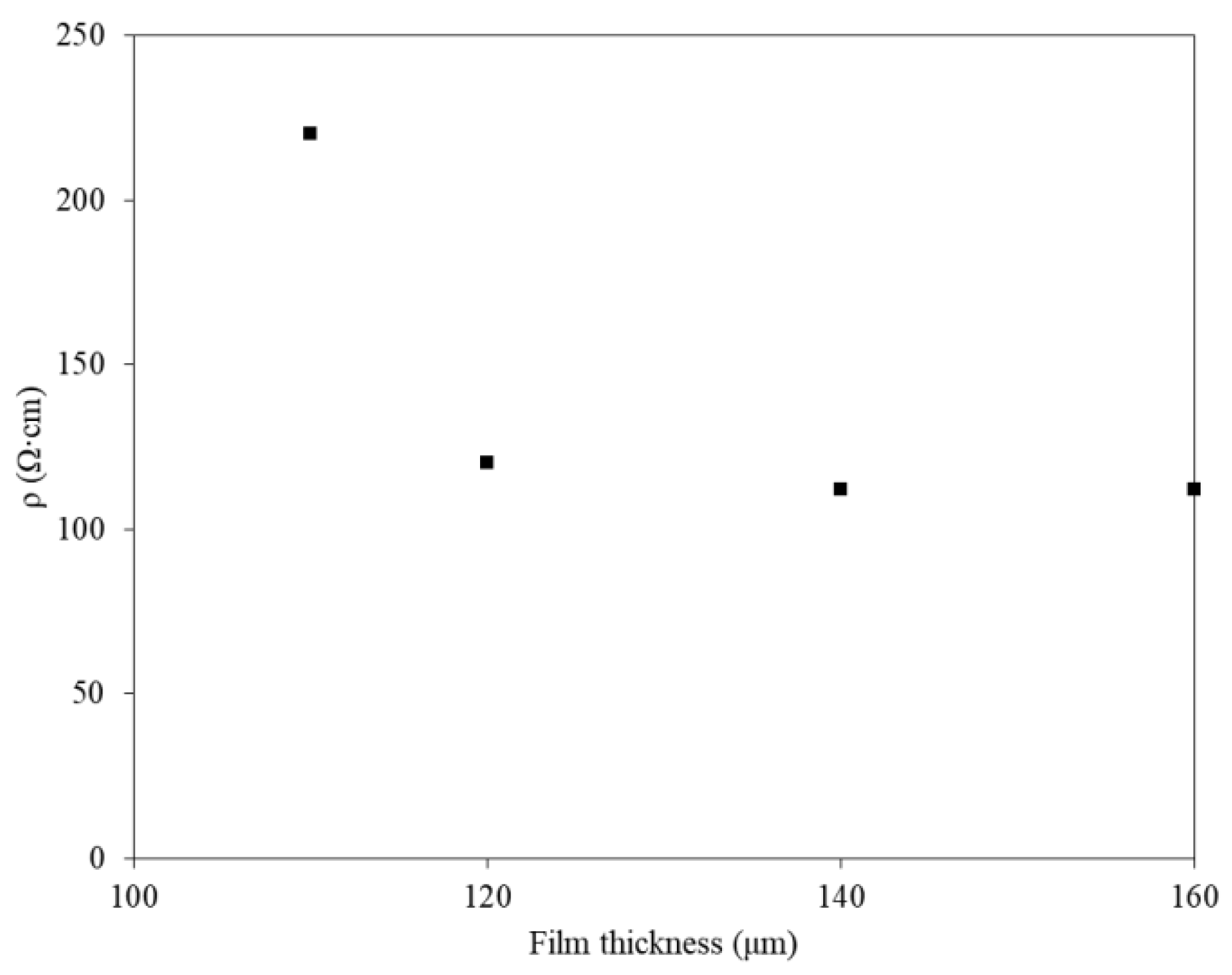
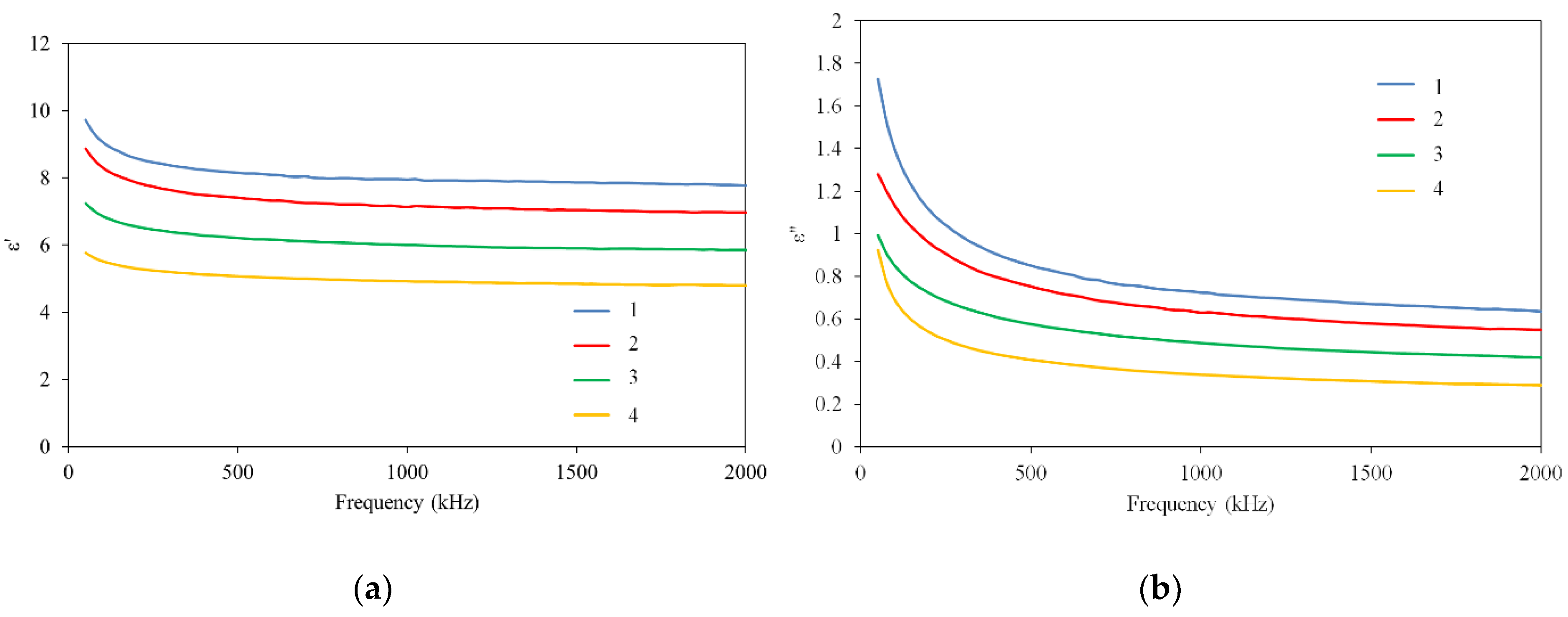
3.2. Electrophysical Characteristics
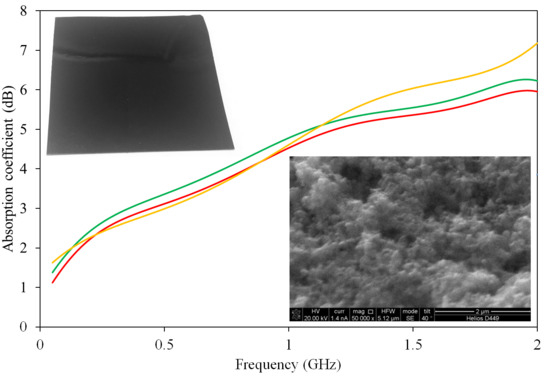
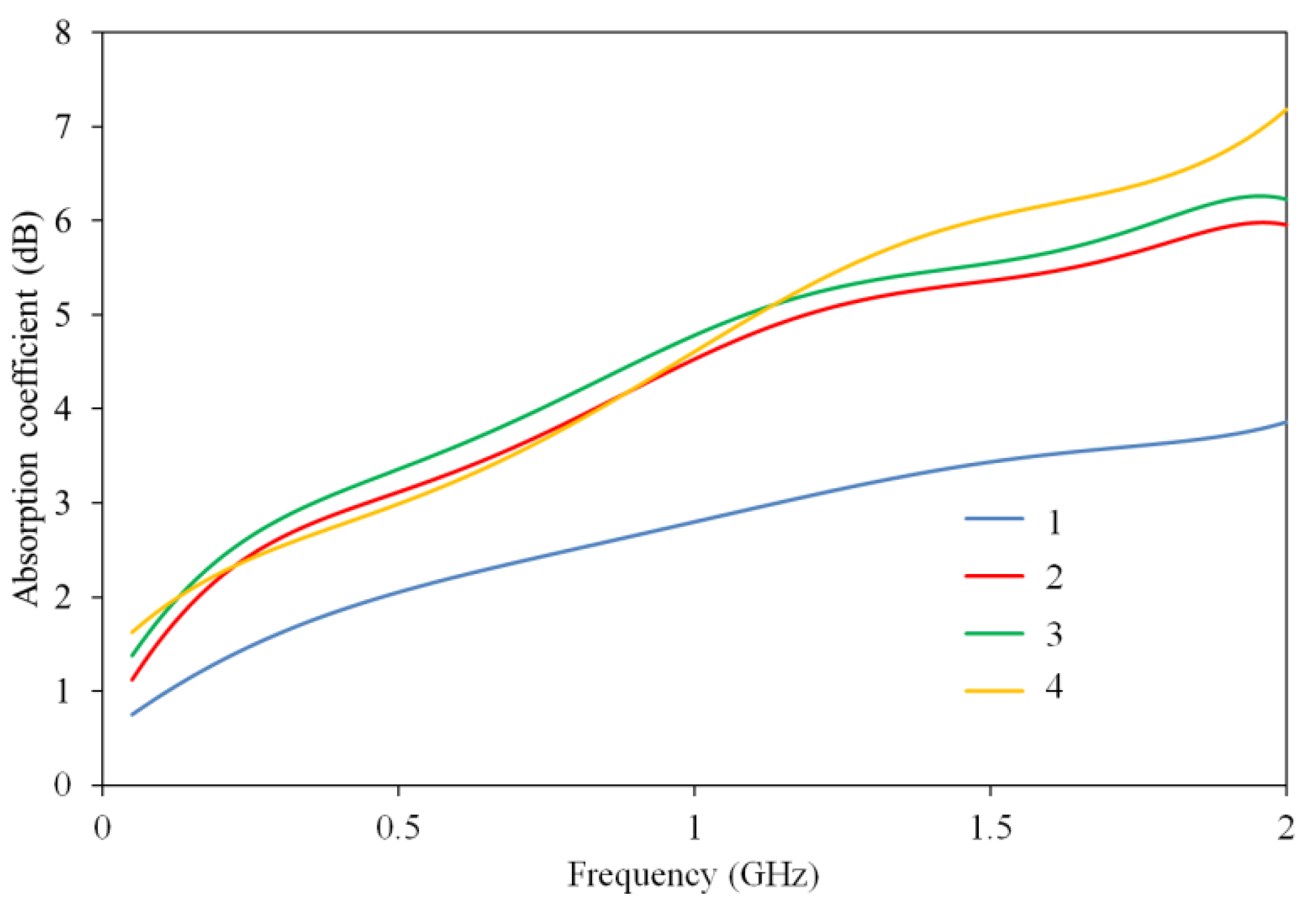
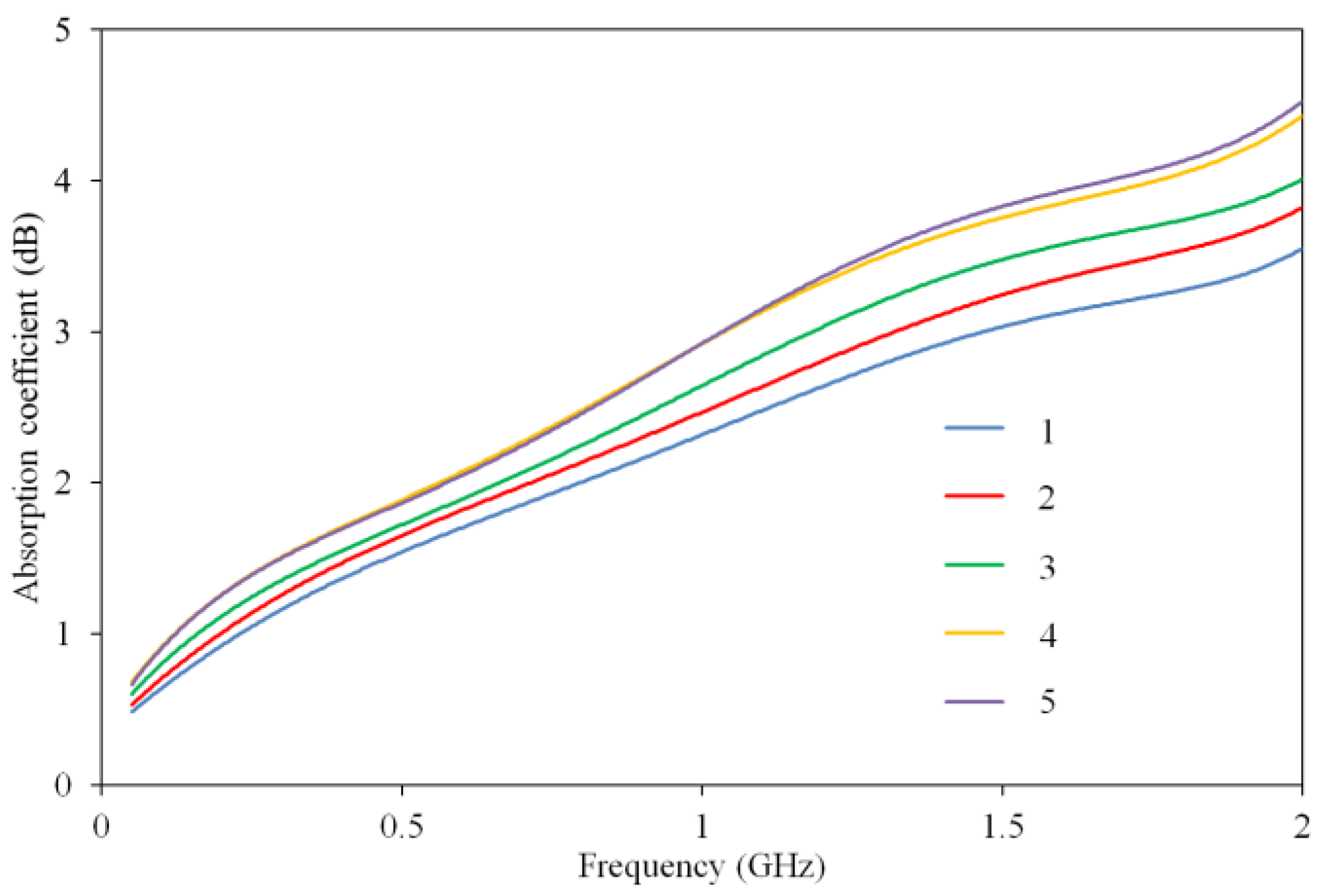
3.3. EMR Absorption Measurement
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Ott, H.W. Electromagnetic Compatibility Engineering; John Wiley & Sons: Hoboken, NJ, USA, 2009; pp. 243–245. ISBN 978-0-470-18930-6. [Google Scholar]
- Mouritz, A.P. Introduction to Aerospace Materials; Woodhead Publishing: Oxford, UK, 2012; pp. 296–298. ISBN 978-0-85709-515-2. [Google Scholar]
- Fang, F.; Li, Y.-Q.; Xiao, H.-M.; Hu, H.; Fu, S.-Y. Layer-structured silver nanowire/polyaniline composite film as high performance X-band EMI shielding material. J. Mater. Chem. C 2016, 19, 4193–4203. [Google Scholar] [CrossRef]
- Faisal, M.; Khasim, S. Polyaniline–antimony oxide composites for effective broadband EMI shielding. Iran Polym. J. 2013, 22, 473–480. [Google Scholar] [CrossRef]
- Khasim, S. Polyaniline-Graphene nanoplatelet composite films with improved conductivity for high performance X-band microwave shielding applications. Res. Phys. 2019, 12, 1073–1081. [Google Scholar] [CrossRef]
- Shakir, M.F.; Khan, A.N.; Khan, R.; Javed, S.; Tariq, A.; Azeem, M.; Riaz, A.; Shafqat, A.; Cheema, H.M.; Akram, M.A.; et al. EMI shielding properties of polymer blends with inclusion of graphene nano platelets. Res. Phys. 2019, 14, 102365. [Google Scholar] [CrossRef]
- Cheng, K.; Li, H.; Zhu, M.; Qiu, H.; Yang, J. In situ polymerization of graphene-polyaniline@polyimide composite films with high EMI shielding and electrical properties. RSC Adv. 2020, 10, 2368–2377. [Google Scholar] [CrossRef]
- Kumar, A.; Kumar, V.; Kumar, M.; Awasthi, K. Synthesis and Characterization of Hybrid PANI/MWCNT Nanocomposites for EMI Applications. Polym. Compos. 2018, 39, 3858–3868. [Google Scholar] [CrossRef]
- Li, H.; Lu, X.; Yuan, D.; Sun, J.; Erden, F.; Wang, F.; He, C. Lightweight flexible carbon nanotube/polyaniline films with outstanding EMI shielding properties. J. Mater. Chem. C 2017, 5, 8694–8698. [Google Scholar] [CrossRef]
- Shi, S.; Zhang, L.; Li, J. Complex permittivity and electromagnetic interference shielding properties of Ti3SiC2/polyaniline composites. J. Mater. Sci. 2009, 44, 945–948. [Google Scholar] [CrossRef]
- Saboor, A.; Khalid, S.M.; Jan, R.; Khan, A.N.; Zia, T.; Farooq, M.U.; Afridi, S.; Sadiq, M.; Arif, M. PS/PANI/MoS2 Hybrid Polymer Composites with High Dielectric Behavior and Electrical Conductivity for EMI Shielding Effectiveness. Materials 2019, 12, 2690. [Google Scholar] [CrossRef]
- Rahul, D.S.; Pais, T.P.M.; Sharath, N.; Ali, S.A.; Faisal, M. Polyaniline-copper oxide composite: A high performance shield against electromagnetic pollution. AIP Conf. Proc. 2015, 1665, 140021. [Google Scholar] [CrossRef]
- Yang, Z.; Zhag, Y.; Wen, B. Enhanced electromagnetic interference shielding capability in bamboo fiber@polyaniline composites through microwave reflection cavity design. Compos. Sci. Technol. 2019, 178, 41–49. [Google Scholar] [CrossRef]
- Zhang, Y.; Qiu, M.; Yu, Y.; Wen, B.; Cheng, L. A novel polyaniline-coated bagasse fiber composite with core-shell heterostructure provides effective electromagnetic shielding performance. ACS Appl. Mater. Interfaces 2017, 9, 809–818. [Google Scholar] [CrossRef]
- Wanasinghe, D.; Aslani, F.; Ma, G.; Habibi, D. Review of polymer composites with diverse nanofillers for electromagnetic interference shielding. Nanomaterials 2020, 10, 541. [Google Scholar] [CrossRef] [PubMed]
- Kamchi, N.E.; Belaabed, B.; Wojkiewicz, J.L.; Lamouri, S.; Lasri, T. Hybrid polyaniline/nanomagnetic particles composites: High performance materials for EMI shielding. J. Appl. Polym. Sci. 2013, 127, 4426–4432. [Google Scholar] [CrossRef]
- Zahari, M.H.B.; Guan, B.H.; Meng, C.E. Development and evaluation of BaFe12O19-PANI-MWCNT composite for electromagnetic interference (EMI) shielding. Prog. Electromagn. Res. C 2018, 80, 55–64. [Google Scholar] [CrossRef]
- Niu, Y. Preparation of polyaniline/polyacrylate composites and their application for electromagnetic interference shielding. Polym. Compos. 2006, 27, 627–632. [Google Scholar] [CrossRef]
- Niu, Y. Electromagnetic interference shielding with polyaniline nanofibers composite coatings. Polym. Eng. Sci. 2008, 48, 355–359. [Google Scholar] [CrossRef]
- Shakir, M.F.; Rashid, I.A.; Tariq, A.; Nawab, Y.; Afzal, A.; Nabeel, M.; Naseem, A.; Hamid, U. EMI shielding characteristics of electrically conductive polymer blends of PS/PANI in microwave and IR region. J. Electronic Mat. 2020, 49, 1660–1665. [Google Scholar] [CrossRef]
- Sobhaa, A.P.; Sreekalac, P.S.; Narayanankuttya, S.K. Electrical, thermal, mechanical and electromagnetic interference shielding properties of PANI/FMWCNT/TPU composites. Prog. Org. Coat. 2017, 113, 168–174. [Google Scholar] [CrossRef]
- Raagulan, K.; Braveenth, R.; Kim, B.M.; Lim, K.J.; Lee, S.B.; Kim, M.; Chai, K.Y. An effective utilization of MXene and its effect on electromagnetic interference shielding: Flexible, free-standing and thermally conductive composite from MXene–PAT–poly(p-aminophenol)–polyaniline co-polymer. RSC Adv. 2020, 10, 1613–1633. [Google Scholar] [CrossRef]
- Bhowmick, B.; Mollick, M.R.; Mondal, D.; Maity, D.; Bain, M.K.; Bera, N.K.; Rana, D.; Chattopadhyay, S.; Chakraborty, M.; Chattopadhyay, D. Poloxamer and gelatin gel guided polyaniline nanofibers: Synthesis and characterization. Polym. Int. 2014, 63, 1505–1512. [Google Scholar] [CrossRef]
- Li, M.; Guo, Y.; Wei, Y.; MacDiarmid, A.G.; Lelkes, P.I. Electrospinning polyaniline-contained gelatin nanofibers for tissue engineering applications. Biomaterials 2006, 27, 2705–2715. [Google Scholar] [CrossRef] [PubMed]
- Ostrovidov, S.; Ebrahimi, M.; Bae, H.; Nguyen, H.K.; Salehi, S.; Kim, S.B.; Kumatani, A.; Matsue, T.; Shi, X.; Nakajima, K.; et al. Gelatin-polyaniline composite nanofibers enhanced excitation-contraction coupling system maturation in myotubes. ACS Appl. Mater. Interfaces 2017, 49, 42444–42458. [Google Scholar] [CrossRef] [PubMed]
- Xue, J.; Liu, Y.; Darabi, M.A.; Tu, G.; Huang, L.; Ying, L.; Xiao, B.; Wu, Y.; Xing, M.; Zhang, L.; et al. An injectable conductive Gelatin-PANI hydrogel system serves as a promising carrier to deliver BMSCs for Parkinson’s disease treatment. Mater. Sci. Eng. C 2019, 100, 584–597. [Google Scholar] [CrossRef]
- Blinova, N.V.; Trchová, M.; Stejskal, J. The polymerization of aniline at a solution–gelatin gel interface. Eur. Polym. J. 2009, 45, 668–673. [Google Scholar] [CrossRef]
- Long, Y.; Huang, K.; Yuan, J.; Han, D.; Niu, L.; Chen, Z.; Gu, C.; Jin, A.; Duvail, J.L. Electrical conductivity of a single Au/polyaniline microfiber. Appl. Phys. Lett. 2006, 88, 162113. [Google Scholar] [CrossRef]
- Gareev, K.G.; Bagrets, V.S.; Khmelnitskiy, I.K.; Ivanitsa, M.G.; Testov, D.O. Research and Development of “Gelatin–Conductive Polymer” Composites for Electromagnetic Compatibility. In Proceedings of the 2020 IEEE Conference of Russian Young Researchers in Electrical and Electronic Engineering (EIConRus), St. Petersburg/Moscow, Russia, 27–30 January 2020; pp. 1069–1072. [Google Scholar] [CrossRef]
- Gareev, K.G.; Luchinin, V.V.; Sevost’yanov, E.N.; Testov, I.O.; Testov, O.A. Frequency dependence of an electromagnetic absorption coefficient in magnetic fluid. Tech. Phys. 2019, 89, 954–957. [Google Scholar] [CrossRef]
- Haberko, J.; Raczkowska, J.; Bernasik, A.; Rysz, J.; Nocuń, M.; Nizioł, J.; Łużny, W.; Budkowski, A. Conductivity of thin polymer films containing polyaniline. Mol. Cryst. Liq. Cryst. 2008, 485, 48–55. [Google Scholar] [CrossRef]
- Vanga Bouanga, C.; Fatyeyeva, K.; Baillif, P.-Y.; Bardeau, J.-F.; Khaokong, C.; Pilard, J.-F.; Tabellout, M. Study of dielectric relaxation phenomena and electrical properties of conductive polyaniline based composite films. J. NonCrystal. Sol. 2010, 356, 611–615. [Google Scholar] [CrossRef]
- Altarawneh, M.M.; Alharazneh, G.A.; Al-Madanat, O.Y. Dielectric properties of single wall carbon nanotubes-based gelatin phantoms. J. Adv. Dielectr. 2018, 8, 1850010. [Google Scholar] [CrossRef]
- Zhang, W.; Ye, H.-Y.; Graf, R.; Spiess, H.W.; Yao, Y.-F.; Zhu, R.-Q.; Xiong, R.-G. Tunable and Switchable Dielectric Constant in an Amphidynamic Crystal. J. Am. Chem. Soc. 2013, 135, 5230–5233. [Google Scholar] [CrossRef] [PubMed]
- Ye, H.-Y.; Zhang, Y.; Fu, D.-W.; Xiong, R.-G. An above-room-temperature ferroelectric organo–metal halide perovskite: (3-Pyrrolinium)(CdCl3). Angew. Chem. Int. Ed. 2014, 53, 1–7. [Google Scholar] [CrossRef] [PubMed]



© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gareev, K.G.; Bagrets, V.S.; Golubkov, V.A.; Ivanitsa, M.G.; Khmelnitskiy, I.K.; Luchinin, V.V.; Mikhailova, O.N.; Testov, D.O. Synthesis and Characterization of Polyaniline-Based Composites for Electromagnetic Compatibility of Electronic Devices. Electronics 2020, 9, 734. https://doi.org/10.3390/electronics9050734
Gareev KG, Bagrets VS, Golubkov VA, Ivanitsa MG, Khmelnitskiy IK, Luchinin VV, Mikhailova ON, Testov DO. Synthesis and Characterization of Polyaniline-Based Composites for Electromagnetic Compatibility of Electronic Devices. Electronics. 2020; 9(5):734. https://doi.org/10.3390/electronics9050734
Chicago/Turabian StyleGareev, Kamil G., Vladislava S. Bagrets, Vladimir A. Golubkov, Maria G. Ivanitsa, Ivan K. Khmelnitskiy, Victor V. Luchinin, Olga N. Mikhailova, and Dmitriy O. Testov. 2020. "Synthesis and Characterization of Polyaniline-Based Composites for Electromagnetic Compatibility of Electronic Devices" Electronics 9, no. 5: 734. https://doi.org/10.3390/electronics9050734
APA StyleGareev, K. G., Bagrets, V. S., Golubkov, V. A., Ivanitsa, M. G., Khmelnitskiy, I. K., Luchinin, V. V., Mikhailova, O. N., & Testov, D. O. (2020). Synthesis and Characterization of Polyaniline-Based Composites for Electromagnetic Compatibility of Electronic Devices. Electronics, 9(5), 734. https://doi.org/10.3390/electronics9050734