Diffmap: Enhancement Difference Map for Peripheral Prostate Zone Cancer Localization Based on Functional Data Analysis and Dynamic Contrast Enhancement MRI
Abstract
1. Introduction
2. Materials and Methods
2.1. Time Point Calculation
- “peak-drop”: the time point corresponding to the maximum value of the derivative curve is selected as and the first preceding local minimum (“valley”) is selected as ;
- “peak-start”: the time points corresponding to the maximum derivative value are selected as and the earliest available DCE time moment is selected as .
2.2. Image Subtraction
- “clipping”: all negative values in are substituted with a zero as follows:
- “non-clipping”: all values remain as-is as follows:
2.3. Distribution Normalization
- Min-max normalization: the distribution is rescaled to a range . Intermediate values are linearly adjusted as follows:
- Percentile ranking: all values are ranked based on their value within the distribution. Ranks are then normalized by dividing by the total number of values as follows:
2.4. Evaluation
3. Results
4. Discussion and Conclusions
- Spatial context. The current approach treats individual voxels independently, without explicitly modeling spatial relationships between neighboring voxels or adjacent slices. Since adjacent voxels are often spatially and functionally connected, incorporating spatial context, such as neighborhood-based features or slice-wise dependencies, may improve robustness to noise and enhance the accuracy of PCa localization.
- Pharmacokinetic analysis. Other types of information can be derived from MRI-DCE, which may provide additional diagnostic value. For instance, pharmacokinetic parametric maps: (volume transfer between blood plasma and extracellular-extravascular space), (transfer rate between extracellular-extravascular space and blood plasma), (volume fraction of extracellular-extravascular space), (volume fraction of plasma in the tissue).
- Other MRI data. The present model utilizes a single input of the dynamic contrast-enhanced MRI sequence. Augmenting the model with additional MRI modalities, such as T2-weighted or diffusion-weighted imaging (DWI), may increase the diagnostic power.
- Image registration. Patient motion during MRI acquisition can lead to misalignment and distortion between image data, particularly in DCE-MRI. As the proposed method relies on voxel-level precision, such discrepancies may reduce its predictive performance. Implementing image registration techniques to spatially align the images across time points could mitigate this issue.
- Sample size. Although the full dataset included 144 patients, expert-validated peripheral zone annotations were available for only 20 cases at the time of analysis. This study, therefore, represents a methodological validation on a curated subset, which limits statistical power and generalizability.
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Bray, F.; Laversanne, M.; Sung, H.; Ferlay, J.; Siegel, R.L.; Soerjomataram, I.; Jemal, A. Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2024, 74, 229–263. [Google Scholar]
- Weinreb, J.C.; Barentsz, J.O.; Choyke, P.L.; Cornud, F.; Haider, M.A.; Macura, K.J.; Margolis, D.; Schnall, M.D.; Shtern, F.; Tempany, C.M.; et al. PI-RADS prostate imaging—Reporting and data system: 2015, version 2. Eur. Urol. 2016, 69, 16–40. [Google Scholar]
- Turkbey, B.; Rosenkrantz, A.B.; Haider, M.A.; Padhani, A.R.; Villeirs, G.; Macura, K.J.; Tempany, C.M.; Choyke, P.L.; Cornud, F.; Margolis, D.J.; et al. Prostate imaging reporting and data system version 2.1: 2019 update of prostate imaging reporting and data system version 2. Eur. Urol. 2019, 76, 340–351. [Google Scholar] [CrossRef] [PubMed]
- Launer, B.M.; Ellis, T.A.; Scarpato, K.R. A contemporary review: mpMRI in prostate cancer screening and diagnosis. Urol. Oncol. Semin. Orig. Investig. 2025, 43, 15–22. [Google Scholar]
- Kuhl, C.K.; Bruhn, R.; Krämer, N.; Nebelung, S.; Heidenreich, A.; Schrading, S. Abbreviated biparametric prostate MR imaging in men with elevated prostate-specific antigen. Radiology 2017, 285, 493–505. [Google Scholar] [CrossRef]
- Woo, S.; Suh, C.H.; Kim, S.Y.; Cho, J.Y.; Kim, S.H.; Moon, M.H. Head-to-head comparison between biparametric and multiparametric MRI for the diagnosis of prostate cancer: A systematic review and meta-analysis. Am. J. Roentgenol. 2018, 211, W226–W241. [Google Scholar]
- Belue, M.J.; Yilmaz, E.C.; Daryanani, A.; Turkbey, B. Current status of biparametric MRI in prostate cancer diagnosis: Literature analysis. Life 2022, 12, 804. [Google Scholar] [CrossRef]
- Greenberg, J.W.; Koller, C.R.; Casado, C.; Triche, B.L.; Krane, L.S. A narrative review of biparametric MRI (bpMRI) implementation on screening, detection, and the overall accuracy for prostate cancer. Ther. Adv. Urol. 2022, 14, 17562872221096377. [Google Scholar] [CrossRef]
- Yu, X.; Liu, R.; Song, L.; Gao, W.; Wang, X.; Zhang, Y. Differences in the pathogenetic characteristics of prostate cancer in the transitional and peripheral zones and the possible molecular biological mechanisms. Front. Oncol. 2023, 13, 1165732. [Google Scholar] [CrossRef]
- McNeal, J.E.; Redwine, E.A.; Freiha, F.S.; Stamey, T.A. Zonal distribution of prostatic adenocarcinoma: Correlation with histologic pattern and direction of spread. Am. J. Surg. Pathol. 1988, 12, 897–906. [Google Scholar] [CrossRef]
- Sato, S.; Kimura, T.; Onuma, H.; Egawa, S.; Takahashi, H. Transition zone prostate cancer is associated with better clinical outcomes than peripheral zone cancer. BJUI Compass 2021, 2, 169–177. [Google Scholar] [CrossRef]
- Wang, J.-L.; Chiou, J.-M.; Müller, H.-G. Functional data analysis. Annu. Rev. Stat. Appl. 2016, 3, 257–295. [Google Scholar] [CrossRef]
- Ullah, S.; Finch, C.F. Applications of functional data analysis: A systematic review. BMC Med. Res. Methodol. 2013, 13, 43. [Google Scholar] [CrossRef]
- Erbas, B.; Akram, M.; Gertig, D.M.; English, D.; Hopper, J.L.; Kavanagh, A.M.; Hyndman, R. Using functional data analysis models to estimate future time trends in age-specific breast cancer mortality for the United States and England—Wales. J. Epidemiol. 2010, 20, 159–165. [Google Scholar] [CrossRef]
- Locantore, N.; Marron, J.S.; Simpson, D.G.; Tripoli, N.; Zhang, J.T.; Cohen, K.L.; Boente, G.; Fraiman, R.; Brumback, B.; Croux, C.; et al. Robust principal component analysis for functional data. Test 1999, 8, 1–73. [Google Scholar] [CrossRef]
- Ratcliffe, S.J.; Leader, L.R.; Heller, G.Z. Functional data analysis with application to periodically stimulated foetal heart rate data. I: Functional regression. Stat. Med. 2002, 21, 1103–1114. [Google Scholar] [CrossRef] [PubMed]
- Vakorin, V.A.; Borowsky, R.; Sarty, G.E. Characterizing the functional MRI response using Tikhonov regularization. Stat. Med. 2007, 26, 3830–3844. [Google Scholar] [CrossRef] [PubMed]
- Wu, P.-S.; Müller, H.-G. Functional embedding for the classification of gene expression profiles. Bioinformatics 2010, 26, 509–517. [Google Scholar] [CrossRef] [PubMed]
- Hansford, B.G.; Peng, Y.; Jiang, Y.; Vannier, M.W.; Antic, T.; Thomas, S.; McCann, S.; Oto, A. Dynamic contrast-enhanced MR imaging curve-type analysis: Is it helpful in the differentiation of prostate cancer from healthy peripheral zone? Radiology 2015, 275, 448–457. [Google Scholar] [CrossRef]
- Chen, C.; Yang, Z.; Sweeney, E.; Hectors, S.J.; Hu, J.C.; Margolis, D.J. Prostate heterogeneity correlates with clinical features on multiparametric MRI. Abdom. Radiol. 2021, 46, 5369–5376. [Google Scholar] [CrossRef]
- Tolkach, Y.; Kristiansen, G. The heterogeneity of prostate cancer: A practical approach. Pathobiology 2018, 85, 108–116. [Google Scholar] [CrossRef]
- Midiri, F.; Vernuccio, F.; Purpura, P.; Alongi, P.; Bartolotta, T.V. Multiparametric MRI and radiomics in prostate cancer: A review of the current literature. Diagnostics 2021, 11, 1829. [Google Scholar] [CrossRef]
- Huang, T.B.; Shi, R.J.; Shang, J.W.; Zhao, R.Z.; Wang, Y.M.; Xia, W.; Wang, S.Q.; Tan, R.Y.; Hua, L.X. Challenges in magnetic resonance imaging-based detection of clinically significant prostate cancer in young patients: Two alternative approaches. Quant. Imaging Med. Surg. 2024, 14, 9419. [Google Scholar] [CrossRef] [PubMed]
- Wallström, J.; Thimansson, E.; Andersson, J.; Karlsson, M.; Zackrisson, S.; Bratt, O.; Jäderling, F. An online national quality assessment survey of prostate MRI reading: Interreader variability in prostate volume measurement and PI-RADS classification. Eur. J. Radiol. Open 2025, 14, 100625. [Google Scholar] [CrossRef]
- Franz, T.; Sicker, T.; Lueke, J.; Dinh, B.; Ho, T.P.; Spinos, T.; Horn, L.C.; Schaudinn, A.; Liatsikos, E.; Stolzenburg, J.U. To biopsy or not biopsy, that is the question-PI-RADS 3 prostate lesions–validation of clinical and radiological parameters for biopsy decision-making. BMC Urol. 2025, 25, 274. [Google Scholar] [CrossRef] [PubMed]
- Brancato, V.; Aiello, M.; Basso, L.; Monti, S.; Palumbo, L.; Di Costanzo, G.; Salvatore, M.; Ragozzino, A.; Cavaliere, C. Evaluation of a multiparametric MRI radiomic-based approach for stratification of equivocal PI-RADS 3 and upgraded PI-RADS 4 prostatic lesions. Sci. Rep. 2021, 11, 643. [Google Scholar] [CrossRef] [PubMed]
- Tang, J.; Zheng, X.; Wang, X.; Mao, Q.; Xie, L.; Wang, R. Computer-aided detection of prostate cancer in early stages using multi-parameter MRI: A promising approach for early diagnosis. Technol. Health Care 2024, 32, 125–133. [Google Scholar] [CrossRef]
- Zhang, G.M.; Han, Y.Q.; Wei, J.W.; Qi, Y.F.; Gu, D.S.; Lei, J.; Yan, W.G.; Xiao, Y.; Xue, H.D.; Feng, F.; et al. Radiomics based on MRI as a biomarker to guide therapy by predicting upgrading of prostate cancer from biopsy to radical prostatectomy. J. Magn. Reson. Imaging 2020, 52, 1239–1248. [Google Scholar] [CrossRef]
- Winkel, D.J.; Breit, H.C.; Shi, B.; Boll, D.T.; Seifert, H.H.; Wetterauer, C. Predicting clinically significant prostate cancer from quantitative image features including compressed sensing radial MRI of prostate perfusion using machine learning: Comparison with PI-RADS v2 assessment scores. Quant. Imaging Med. Surg. 2020, 10, 808–817. [Google Scholar] [CrossRef]
- Li, M.; Yang, L.; Yue, Y.; Xu, J.; Huang, C.; Song, B. Use of radiomics to improve diagnostic performance of PI-RADS v2.1 in prostate cancer. Front. Oncol. 2021, 10, 631831. [Google Scholar] [CrossRef]
- Winkel, D.J.; Breit, H.C.; Block, T.K.; Boll, D.T.; Heye, T.J. High spatiotemporal resolution dynamic contrast-enhanced MRI improves the image-based discrimination of histopathology risk groups of peripheral zone prostate cancer: A supervised machine learning approach. Eur. Radiol. 2020, 30, 4828–4837. [Google Scholar] [CrossRef] [PubMed]
- Afshari Mirak, S.; Mohammadian Bajgiran, A.; Sung, K.; Asvadi, N.H.; Markovic, D.; Felker, E.R.; Lu, D.; Sisk, A.; Reiter, R.E.; Raman, S.S. Dynamic contrast-enhanced (DCE) MR imaging: The role of qualitative and quantitative parameters for evaluating prostate tumors stratified by Gleason score and PI-RADS v2. Abdom. Radiol. 2020, 45, 2225–2234. [Google Scholar] [CrossRef] [PubMed]
- Delongchamps, N.B.; Rouanne, M.; Flam, T.; Beuvon, F.; Liberatore, M.; Zerbib, M.; Cornud, F. Multiparametric magnetic resonance imaging for the detection and localization of prostate cancer: Combination of T2-weighted, dynamic contrast-enhanced and diffusion-weighted imaging. BJU Int. 2011, 107, 1411–1418. [Google Scholar] [CrossRef] [PubMed]
- Thulasi Seetha, S.; Garanzini, E.; Tenconi, C.; Marenghi, C.; Avuzzi, B.; Catanzaro, M.; Stagni, S.; Villa, S.; Chiorda, B.N.; Badenchini, F.; et al. Stability of multi-parametric prostate MRI radiomic features to variations in segmentation. J. Pers. Med. 2023, 13, 1172. [Google Scholar] [CrossRef]
- Vaitulevičius, A.; Bernatavičienė, J.; Markevičiutė, J.; Naruševičiūtė, I.; Trakymas, M.; Treigys, P. Advancements in prostate zone segmentation: Integrating attention mechanisms into the nnU-Net framework. Mach. Learn. Sci. Technol. 2024, 5, 045003. [Google Scholar] [CrossRef]
- Pineda, F.D.; Medved, M.; Wang, S.; Fan, X.; Schacht, D.V.; Sennett, C.; Oto, A.; Newstead, G.M.; Abe, H.; Karczmar, G.S. Ultrafast bilateral DCE-MRI of the breast with conventional Fourier sampling: Preliminary evaluation of semi-quantitative analysis. Acad. Radiol. 2016, 23, 1137–1144. [Google Scholar] [CrossRef]
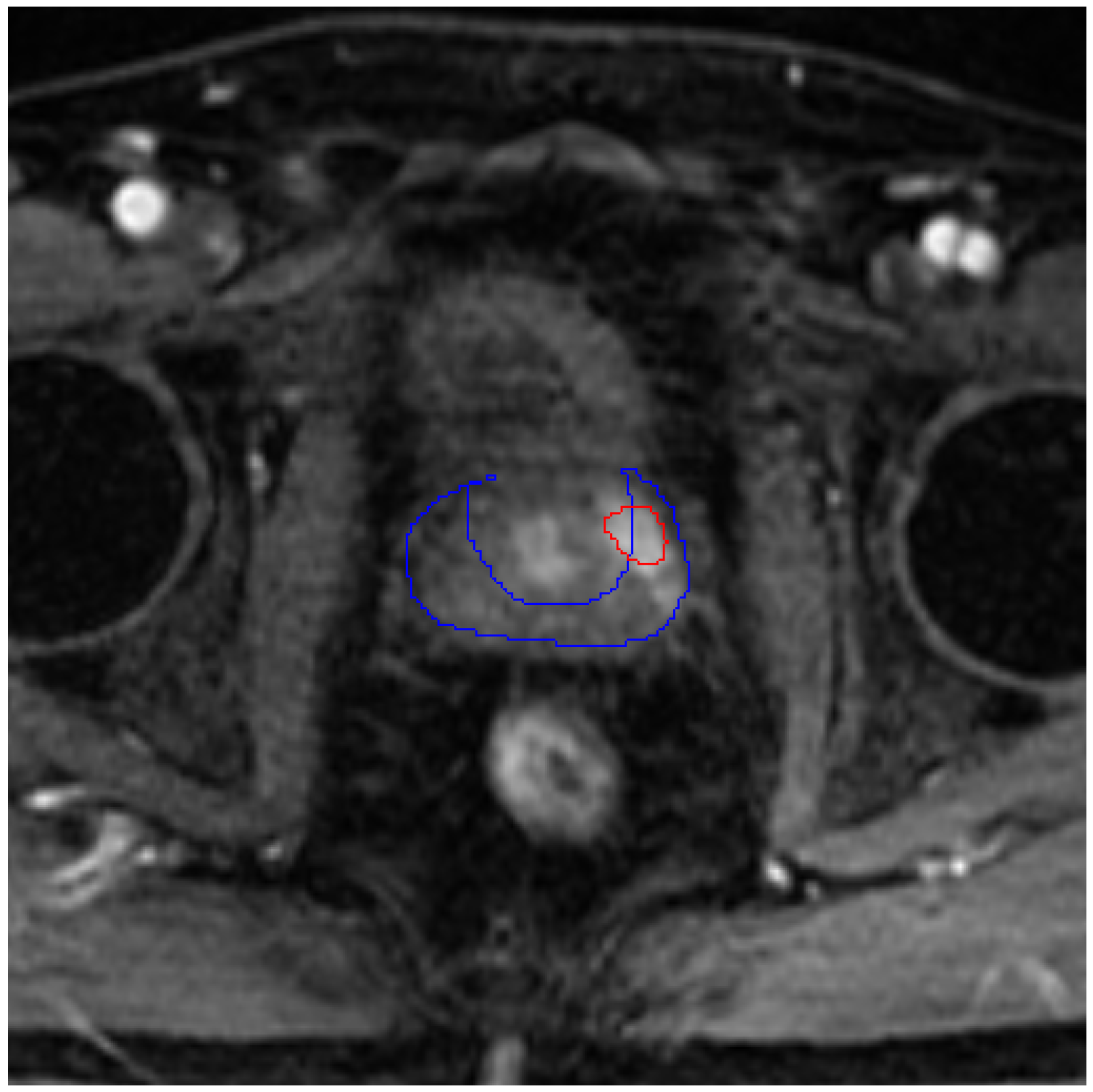








| Min | Mean | Max | |
|---|---|---|---|
| Cancer voxels | 80 | 1987 | 10,131 |
| Non-cancer voxels | 7507 | 45,995 | 145,360 |
| Cancer voxel ratio, % | 0.2 | 4.7 | 26.3 |
| Time Point Selection Variant | Image Subtraction Variant | Distribution Normalization Variant | Log Loss | Log Loss |
|---|---|---|---|---|
| Peak-drop | Clipping | Min-max norm. | 3.504 | 5.136 |
| Percentile rank. | 0.713 | 0.240 | ||
| Non-clipping | Min-max norm. | 0.609 | 0.143 | |
| Percentile rank. | 0.767 | 0.353 | ||
| Peak-start | Clipping | Min-max norm. | 1.600 | 2.697 |
| Percentile rank. | 0.641 | 0.165 | ||
| Non-clipping | Min-max norm. | 0.578 | 0.112 | |
| Percentile rank. | 0.647 | 0.180 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Surkant, R.; Markevičiūtė, J.; Naruševičiūtė, I.; Trakymas, M.; Treigys, P.; Bernatavičienė, J. Diffmap: Enhancement Difference Map for Peripheral Prostate Zone Cancer Localization Based on Functional Data Analysis and Dynamic Contrast Enhancement MRI. Electronics 2026, 15, 507. https://doi.org/10.3390/electronics15030507
Surkant R, Markevičiūtė J, Naruševičiūtė I, Trakymas M, Treigys P, Bernatavičienė J. Diffmap: Enhancement Difference Map for Peripheral Prostate Zone Cancer Localization Based on Functional Data Analysis and Dynamic Contrast Enhancement MRI. Electronics. 2026; 15(3):507. https://doi.org/10.3390/electronics15030507
Chicago/Turabian StyleSurkant, Roman, Jurgita Markevičiūtė, Ieva Naruševičiūtė, Mantas Trakymas, Povilas Treigys, and Jolita Bernatavičienė. 2026. "Diffmap: Enhancement Difference Map for Peripheral Prostate Zone Cancer Localization Based on Functional Data Analysis and Dynamic Contrast Enhancement MRI" Electronics 15, no. 3: 507. https://doi.org/10.3390/electronics15030507
APA StyleSurkant, R., Markevičiūtė, J., Naruševičiūtė, I., Trakymas, M., Treigys, P., & Bernatavičienė, J. (2026). Diffmap: Enhancement Difference Map for Peripheral Prostate Zone Cancer Localization Based on Functional Data Analysis and Dynamic Contrast Enhancement MRI. Electronics, 15(3), 507. https://doi.org/10.3390/electronics15030507






