Electrophysiological Correlates of Virtual-Reality Applications in the Rehabilitation Setting: New Perspectives for Stroke Patients
Abstract
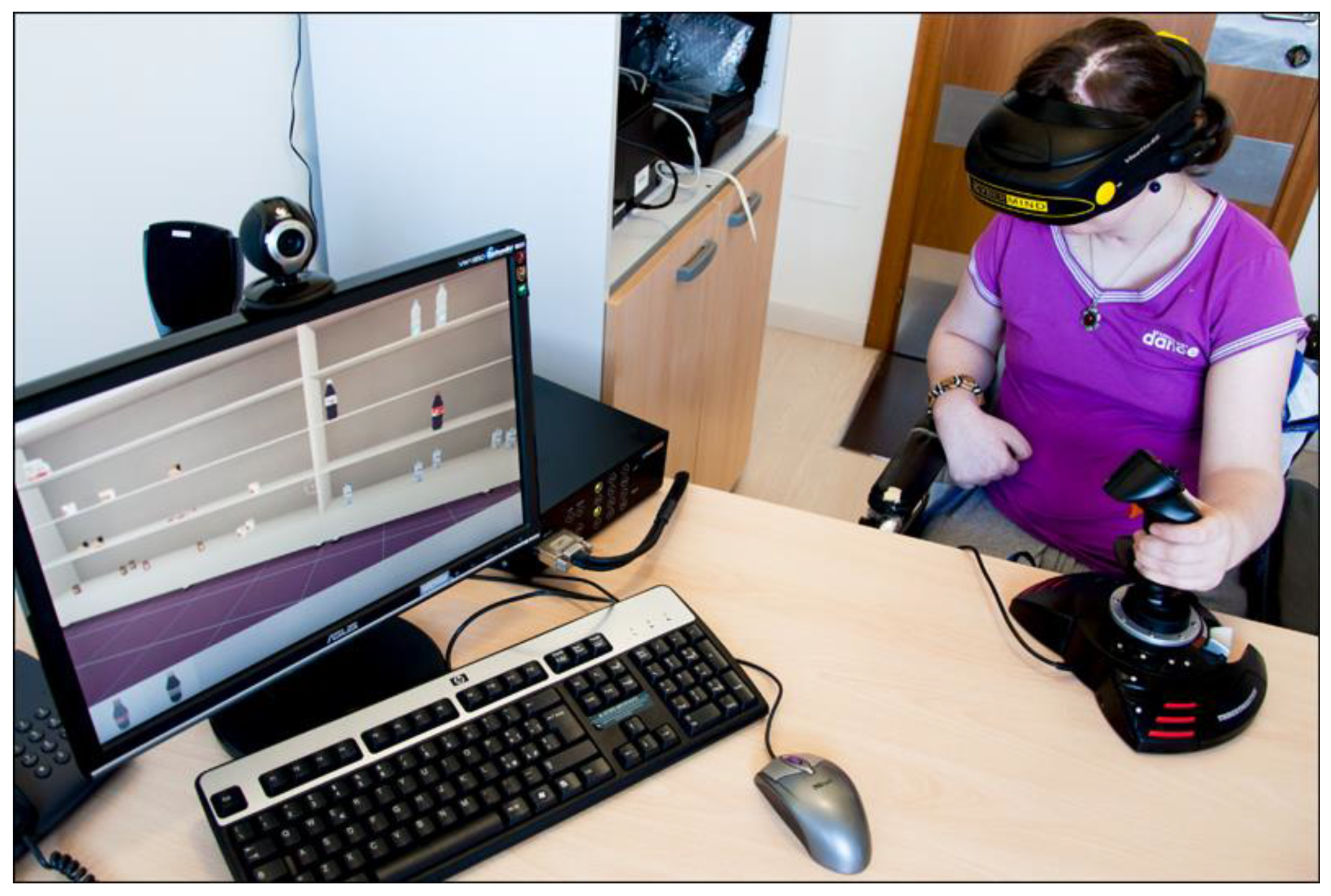
1. Advanced Neurorehabilitation Systems for Recovering Patients with Stroke
- (a)
- The sense of presence is the most important component differentiating the VR from the other device [12,13]. VR allows the users to be in a virtual environment (VE) rather than in the place in which is located. Sanchez-Vives & Slater [14] claims that to be “present” means to transfer the consciousness in the VEs. It depends on different converging characteristics: the modality in which the user is represented in the VE, the number and quality of feedback, user’s characteristics (i.e., age, gender), and VE characteristics (how much realistic it is). In the simulation of a sensory-motor experience, the VEs recreate as close as possible a real situation, inducing the brain system to reactivate the underlying neural networks for the expected effect as in a real environment.
- (b)
- Immersion is an objective property of the system that aims at generating a sense of presence as human subjective response. The degree of perceived immersion is dependent on technology. Finally, a high level of immersion does not guarantee a corresponding level of presence, while a higher sense of presence generates a deeper emotional response [15].
2. Looking for Endophenotypes of Recovery: The Electroencephalography
3. EEG Reveals Neurophysiological Correlates of Neurorehabilitation with VR
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Katan, M.; Luft, A. Global Burden of Stroke. Semin. Neurol. 2018, 38, 208–211. [Google Scholar] [CrossRef]
- Massetti, T.; da Silva, T.D.; Crocetta, T.B.; Guarnieri, R.; de Freitas, B.L.; Bianchi Lopes, P.; Watson, S.; Tonks, J.; de Mello Monteiro, C.B. The Clinical Utility of Virtual Reality in Neurorehabilitation: A Systematic Review. J. Cent. Nerv. Syst. Dis. 2018, 27, 1179573518813541. [Google Scholar] [CrossRef]
- Palma, G.C.; Freitas, T.B.; Bonuzzi, G.M.; Soares, M.A.; Leite, P.H.; Mazzini, N.A.; Almeida, M.R.; Pompeu, J.E.; Torriani-Pasin, C. Effects of virtual reality for stroke individuals based on the International Classification of Functioning and Health: A systematic review. Top. Stroke Rehabil. 2017, 24, 269–278. [Google Scholar] [CrossRef] [PubMed]
- Merians, A.S.; Fluet, G.G. Rehabilitation Applications Using Virtual Reality for Persons with Residual Impairments Following Stroke. In Virtual Reality for Physical and Motor Rehabilitation. Virtual Reality Technologies for Health and Clinical Applications; Weiss, P., Keshner, E., Levin, M., Eds.; Springer: New York, NY, USA, 2014. [Google Scholar] [CrossRef]
- Barker, R.N.; Brauer, S.G. Upper limb recovery after stroke: The stroke survivors’ perspective. Disabil. Rehabil. 2005, 27, 1213–1223. [Google Scholar] [CrossRef] [PubMed]
- Kleim, J.A.; Jones, T.A. Principles of experience-dependent neural plasticity: Implications for rehabilitation after brain damage. J. Speech Lang. Hear. Res. 2008, 51, S225–S239. [Google Scholar] [CrossRef]
- Proffitt, R.; Lange, B. Considerations in the efficacy and effectiveness of virtual reality interventions for stroke rehabilitation: Moving the field forward. Phys. Ther. 2015, 95, 441–448. [Google Scholar] [CrossRef]
- Laver, K.E.; Lange, B.; George, S. Virtual reality for stroke rehabilitation. Cochrane Database Syst. Rev. 2017, 11, CD008349. [Google Scholar] [CrossRef] [PubMed]
- Bowman, D.A.; McMahan, R.P. Virtual Reality: How much immersion is enough? Computer 2007, 40, 36–43. [Google Scholar] [CrossRef]
- Bowman, D.A.; Kruijff, E.; LaViola, J.J.; Poupyrev, I. 3D User Interfaces: Theory and Practice, Course Smart eTextbook; Addison-Wesley: Boston, MA, USA, 2004; Available online: https://www.amazon.co.uk/3D-User-Interfaces-CourseSmart-eTextbook-ebook/dp/B00AU3JU9O (accessed on 2 March 2021).
- Weiss, P.; Kizony, R.; Feintuch, U.; Rand, D.; Katz, N. Virtual Reality Applications in Neurorehabilitation. In Textbook of Neural Repair and Rehabilitation; Selzer, M., Clarke, S., Cohen, L., Kwakkel, G., Miller, R., Eds.; Cambridge University Press: Cambridge, UK, 2014; pp. 198–218. [Google Scholar] [CrossRef]
- Schultheis, M.T.; Himelstein, J.; Rizzo, A.A. Virtual reality and neuropsychology: Upgrading the current tools. J. Head Trauma Rehabil. 2002, 17, 378–394. [Google Scholar] [CrossRef] [PubMed]
- Riva, G. The neuroscience of body memory: From the self through the space to the others. Cortex 2018, 104, 241–260. [Google Scholar] [CrossRef] [PubMed]
- Sanchez-Vives, M.V.; Slater, M. From presence to consciousness through virtual reality. Nat. Rev. Neurosci. 2005, 6, 332–339. [Google Scholar] [CrossRef] [PubMed]
- Perez-Marcos, D.; Slater, M.; Sanchez-Vives, M.V. Inducing a virtual hand ownership illusion through a brain-computer interface. Neuroreport 2009, 20, 589–594. [Google Scholar] [CrossRef]
- Turolla, A.; Dam, M.; Ventura, L.; Tonin, P.; Agostini, M.; Zucconi, C.; Kiper, P.; Cagnin, A.; Piron, L. Virtual Reality for the rehabilitation of the upper limb motor function after stroke: A prospective controlled trial. J. Neuroeng. Rehabil. 2013, 10, 85. [Google Scholar] [CrossRef]
- Schiza, E.; Matsangidou, M.; Neokleous, K.; Pattichis, C.S. Virtual Reality Applications for Neurological Disease: A Review. Front. Robot AI 2019, 6, 100. [Google Scholar] [CrossRef]
- Lang, C.E.; Macdonald, J.R.; Reisman, D.S.; Boyd, L.; Jacobson Kimberley, T.; Schindler-Ivens, S.M.; Hornby, T.G.; Ross, S.A.; Scheets, P.L. Observation of amounts of movement practice provided during stroke rehabilitation. Arch. Phys. Med. Rehabil. 2009, 90, 1692–1698. [Google Scholar] [CrossRef] [PubMed]
- Lewis, G.N.; Rosie, J.A. Virtual reality games for movement rehabilitation in neurological conditions: How do we meet the needs and expectations of the users? Disabil. Rehabil. 2012, 34, 1880–1886. [Google Scholar] [CrossRef] [PubMed]
- Sarfo, F.S.; Ulasavets, U.; Opare-Sem, O.K.; Ovbiagele, B. Tele-Rehabilitation after Stroke: An Updated Systematic Review of the Literature. J. Stroke Cerebrovasc. Dis. 2018, 27, 2306–2318. [Google Scholar] [CrossRef]
- Agostini, M.; Garzon, M.; Benavides-Varela, S.; de Pellegrin, S.; Bencini, G.; Rossi, G.; Rosadoni, S.; Mancuso, M.; Turolla, A.; Meneghello, F.; et al. Telerehabilitation in poststroke anomia. Biomed. Res. Int. 2014, 2014, 706909. [Google Scholar] [CrossRef]
- Piron, L.; Turolla, A.; Tonin, P.; Piccione, F.; Lain, L.; Dam, M. Satisfaction with care in post-stroke patients undergoing a telerehabilitation programme at home. J. Telemed. Telecare 2008, 14, 257–260. [Google Scholar] [CrossRef] [PubMed]
- Tieri, G.; Morone, G.; Paolucci, S.; Iosa, M. Virtual reality in cognitive and motor rehabilitation: Facts, fiction and fallacies. Expert Rev. Med. Devices 2018, 15, 107–117. [Google Scholar] [CrossRef]
- Bernardoni, F.; Ozen, O.; Buetler, K.; Marchal-Crespo, L. Virtual Reality Environments and Haptic Strategies to Enhance Implicit Learning and Motivation in Robot-Assisted Training. In Proceedings of the 2019 IEEE 16th International Conference on Rehabilitation Robotics (ICORR), Toronto, ON, Canada, 24–28 June 2019; pp. 760–765. [Google Scholar] [CrossRef]
- Gottesman, I.I.; Shields, J.J. A polygenic theory of schizophrenia. Proc. Natl. Acad. Sci. USA 1967, 58, 199–205. [Google Scholar] [CrossRef]
- Cottone, C.; Cancelli, A.; Pasqualetti, P.; Porcaro, C.; Salustri, C.; Tecchio, F. A New, High-Efficacy, Noninvasive Transcranial Electric Stimulation Tuned to Local Neurodynamics. J. Neurosci. 2018, 38, 586–594. [Google Scholar] [CrossRef] [PubMed]
- Porcaro, C.; Cottone, C.; Cancelli, A.; Rossini, P.M.; Zito, G.; Tecchio, F. Cortical neurodynamics changes mediate the efficacy of a personalized neuromodulation against multiple sclerosis fatigue. Sci. Rep. 2019, 3, 18213. [Google Scholar] [CrossRef] [PubMed]
- Ansado, J.; Chasen, C.; Bouchard, S.; Northoff, G. How brain imaging provides predictive biomarkers for therapeutic success in the context of virtual reality cognitive training. Neurosci. Biobehav. Rev. 2021, 120, 583–594. [Google Scholar] [CrossRef] [PubMed]
- Ayaz, H.; Onaral, B.; Izzetoglu, K.; Shewokis, P.A.; Mckendrick, R.; Parasuraman, R. Continuous monitoring of brain dynamics with functional near infrared spectroscopy as a tool for neuroergonomic research: Empirical examples and a technological development. Front. Hum. Neurosci. 2013, 7, 871. [Google Scholar] [CrossRef]
- Shih, J.J.; Krusienski, D.J.; Wolpaw, J.R. Brain-computer interfaces in medicine. Mayo Clin. Proc. 2012, 87, 268–279. [Google Scholar] [CrossRef]
- Pfurtscheller, G.; da Silva, F.H.L. Event-related EEG/MEG synchronization and desynchronization: Basic principles. Clin. Neurophysiol. 1999, 110, 1842–1857. [Google Scholar] [CrossRef]
- Neuper, C.; Schlögl, A.; Pfurtscheller, G. Enhancement of left-right sensorimotor EEG differences during feedback-regulated motor imagery. J. Clin. Neurophysiol. 1999, 16, 373–382. [Google Scholar] [CrossRef]
- Porcaro, C.; Mayhew, S.D.; Bagshaw, A.P. Role of the Ipsilateral Primary Motor Cortex in the Visuo-Motor Network During Fine Contractions and Accurate Performance. Int. J. Neural Syst. 2021, 2150011. [Google Scholar] [CrossRef]
- Mayhew, S.D.; Porcaro, C.; Tecchio, F.; Bagshaw, A.P. fMRI characterization of widespread brain networks relevant for behavioural variability in fine hand motor control with and without visual feedback. Neuroimage 2017, 148, 330–342. [Google Scholar] [CrossRef] [PubMed]
- Pichiorri, F.; Mattia, D. Brain-computer interfaces in neurologic rehabilitation practice. Handb. Clin. Neurol. 2020, 168, 101–116. [Google Scholar] [CrossRef] [PubMed]
- Liu, M.; Wang, J.; Yan, N.; Yang, Q. Development of EEG Biofeedback System Based on Virtual Reality Environment. In Proceedings of the 27th Annual Conference on IEEE Engineering in Medicine and Biology, Shanghai, China, 1–4 September 2005; pp. 5362–5364. [Google Scholar] [CrossRef]
- Teo, W.P.; Muthalib, M.; Yamin, S.; Hendy, A.M.; Bramstedt, K.; Kotsopoulos, E.; Perrey, S.; Ayaz, H. Does a combination of virtual reality, neuromodulation and neuroimaging provide a comprehensive platform for neurorehabilitation?—A narrative review of the literature. Front. Hum. Neurosci. 2016, 10, 284. [Google Scholar] [CrossRef]
- Lin, C.T.; Chung, I.F.; Ko, L.W.; Chen, Y.C.; Liang, S.F.; Duann, J.R. EEG-based assessment of driver cognitive responses in a dynamic virtual-reality driving environment. IEEE Trans. Biomed. Eng. 2007, 54, 1349–1352. [Google Scholar] [PubMed]
- Jäncke, L.; Cheetham, M.; Baumgartner, T. Virtual reality and the role of the prefrontal cortex in adults and children. Front. Neurosci. 2009, 3, 52–59. [Google Scholar] [CrossRef]
- Calabrò, R.S.; Naro, A.; Russo, M.; Leo, A.; de Luca, R.; Balletta, T.; Buda, A.; La Rosa, G.; Bramanti, A.; Bramanti, P. The role of virtual reality in improving motor performance as revealed by EEG: A randomized clinical trial. J. Neuroeng. Rehabil. 2017, 14, 53. [Google Scholar] [CrossRef]
- Castermans, T.; Duvinage, M.; Cheron, G.; Dutoit, T. Towards effective noninvasive brain-computer interfaces dedicated to gait rehabilitation systems. Brain Sci. 2014, 4, 1–48. [Google Scholar] [CrossRef]
- Comani, S.; Velluto, L.; Schinaia, L. Monitoring neuro-motor recovery from stroke with high-resolution EEG, robotics and virtual reality: A proof of concept. IEEE Trans. Neural. Syst. Rehabil. Eng. 2015, 23, 1106–1116. [Google Scholar] [CrossRef]
- Comani, S.; Schinaia, L.; Tamburro, G.; Velluto, L.; Sorbi, S.; Conforto, S.; Guarnieri, B. Assessing neuro-motor recovery in a stroke survivor with high-resolution EEG, robotics and Virtual Reality. Annu. Int. Conf. IEEE Eng. Med. Biol. Soc. 2015, 2015, 3925–3928. [Google Scholar]
- Steinisch, M.; Tana, M.G.; Comani, S. A post-stroke rehabilitation system integrating robotics, VR and high-resolution EEG imaging. IEEE Trans. Neural. Syst. Rehabil. Eng. 2013, 21, 849–859. [Google Scholar] [CrossRef] [PubMed]
- Saposnik, G.; Levin, M. Outcome Research Canada (SORCan) Working Group.Virtual reality in stroke rehabilitation: A meta-analysis and implications for clinicians. Stroke 2011, 42, 1380–1386. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Arcuri, F.; Porcaro, C.; Ciancarelli, I.; Tonin, P.; Cerasa, A. Electrophysiological Correlates of Virtual-Reality Applications in the Rehabilitation Setting: New Perspectives for Stroke Patients. Electronics 2021, 10, 836. https://doi.org/10.3390/electronics10070836
Arcuri F, Porcaro C, Ciancarelli I, Tonin P, Cerasa A. Electrophysiological Correlates of Virtual-Reality Applications in the Rehabilitation Setting: New Perspectives for Stroke Patients. Electronics. 2021; 10(7):836. https://doi.org/10.3390/electronics10070836
Chicago/Turabian StyleArcuri, Francesco, Camillo Porcaro, Irene Ciancarelli, Paolo Tonin, and Antonio Cerasa. 2021. "Electrophysiological Correlates of Virtual-Reality Applications in the Rehabilitation Setting: New Perspectives for Stroke Patients" Electronics 10, no. 7: 836. https://doi.org/10.3390/electronics10070836
APA StyleArcuri, F., Porcaro, C., Ciancarelli, I., Tonin, P., & Cerasa, A. (2021). Electrophysiological Correlates of Virtual-Reality Applications in the Rehabilitation Setting: New Perspectives for Stroke Patients. Electronics, 10(7), 836. https://doi.org/10.3390/electronics10070836







