Compatibility of Personalized Formulations in Cleoderm™, A Skin Rebalancing Cream Base for Oily and Sensitive Skin
Abstract
1. Introduction
2. Materials and Methods
2.1. Reagents, Reference Standards, and Materials
2.2. Equipment
2.3. Chromatographic Determinations
2.4. Forced Degradation Studies: Characteristics Indicating Stability
- Dilution in acid (0.1 M HCl);
- Dilution in base (0.1 M NaOH);
- Dilution in hydrogen peroxide (H2O2);
- Exposure to ultraviolet light at 365 nm (for 24 h);
- Heating at 70 °C (for 24 h).
2.5. Validation of the HPLC Methods
2.6. Preparation of API Cream Samples for the Compatibility Study
- The required quantity of each ingredient for the total amount to be prepared was calculated.
- Each ingredient was accurately weighed.
- The API was placed in an adequate EMP jar, and combined with a small amount of Levigant, according to each API’s properties.
- The Cleoderm™ was further added into the mixture, and the formulation was mixed using an electronic mixing device (FagronLab™ EMP, Scheßlitz, Germany) for 4 min at a medium mixing speed.
- The product was then passed through a roll mill (FagronLab™ TRM Ointment Mill, Saint Paul, MN, USA) thrice.
- The final product was packaged in airless precise-dose, light-resistant bottles and labeled.
- The creams were then immediately assayed at T = 0 and stored at room temperature (15–30 °C) for the duration of the study.
2.7. Compatibility Study: Physico-Chemical Evaluation
2.8. Compatibility Study: Microbiological Evaluation
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Tan, A.U.; Schlosser, B.J.; Paller, A.S. A Review of Diagnosis and Treatment of Acne in Adult Female Patients. Int. J. Women’s Dermatol. 2018, 4, 56–71. [Google Scholar] [CrossRef]
- Bhate, K.; Williams, H.C. Epidemiology of Acne Vulgaris. Br. J. Dermatol. 2013, 168, 474–485. [Google Scholar] [CrossRef]
- Abokwidir, M.; Feldman, S.R. Rosacea Management. Ski. Appendage Disord. 2016, 2, 26–34. [Google Scholar] [CrossRef]
- Rivero, A.L.; Whitfeld, M. An Update on the Treatment of Rosacea. Aust. Prescr. 2018, 41, 20–24. [Google Scholar] [CrossRef]
- Ganceviciene, R.; Liakou, A.I.; Theodoridis, A.; Makrantonaki, E.; Zouboulis, C.C. Skin Anti-Aging Strategies. Derm. Endocrinol. 2012, 4, 308–319. [Google Scholar] [CrossRef]
- Mukherjee, S.; Date, A.; Patravale, V.; Korting, H.C.; Roeder, A.; Weindl, G. Retinoids in the Treatment of Skin Aging: An Overview of Clinical Efficacy and Safety. Clin. Interv. Aging 2006, 1, 327–348. [Google Scholar] [CrossRef]
- Polonini, H.; Zander, C.; Radke, J. CleodermTM Clarifying Cream: A Novel, Topical Vehicle Using Plant-Based Excipients and Actives Targeting Acne and Oily Skin. J. Cosmet. Dermatol. Sci. Appl. 2021, 11, 381–399. [Google Scholar] [CrossRef]
- Papakonstantinou, E.; Roth, M.; Karakiulakis, G. Hyaluronic Acid: A Key Molecule in Skin Aging. Dermatoendocrinol. 2012, 4, 253–258. [Google Scholar] [CrossRef]
- Resende, D.I.S.P.; Ferreira, M.S.; Sousa-Lobo, J.M.; Sousa, E.; Almeida, I.F. Usage of Synthetic Peptides in Cosmetics for Sensitive Skin. Pharmaceuticals 2021, 14, 702. [Google Scholar] [CrossRef]
- Zaenglein, A.L.; Pathy, A.L.; Schlosser, B.J.; Alikhan, A.; Baldwin, H.E.; Berson, D.S.; Bowe, W.P.; Graber, E.M.; Harper, J.C.; Kang, S.; et al. Guidelines of Care for the Management of Acne Vulgaris. J. Am. Acad. Dermatol. 2016, 74, 945–973.e33. [Google Scholar] [CrossRef]
- Afzali, B.M.; Yaghoobi, E.; Yaghoobi, R.; Bagherani, N.; Dabbagh, M.A. Comparison of the Efficacy of 5% Topical Spironolactone Gel and Placebo in the Treatment of Mild and Moderate Acne Vulgaris: A Randomized Controlled Trial. J. Dermatolog. Treat. 2012, 23, 21–25. [Google Scholar] [CrossRef] [PubMed]
- Gruber, D.M.; Sator, M.O.; Joura, E.A.; Kokoschka, E.M.; Heinze, G.; Huber, J.C. Topical Cyproterone Acetate Treatment in Women with Acne: A Placebo- Controlled Trial. Arch. Dermatol. 1998, 134, 459–463. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Önder, M. An Investigation of Efficacy of Topical Niacinamide for the Treatment of Mild and Moderate Acne Vulgaris. J. Turk. Acad. Dermatol. 2008, 2, 4–7. [Google Scholar]
- Bissett, D.L.; Oblong, J.E.; Berge, C.A. Niacinamide: A B Vitamin That Improves Aging Facial Skin Appearance. Dermatol. Surg. 2005, 31, 860–866. [Google Scholar] [CrossRef] [PubMed]
- Holzer, G.; Riegler, E.; Hönigsmann, H.; Farokhnia, S.; Schmidt, B. Effects and Side-Effects of 2% Progesterone Cream on the Skin of Peri- and Postmenopausal Women: Results from a Double-Blind, Vehicle-Controlled, Randomized Study. Br. J. Dermatol. 2005, 153, 626–634. [Google Scholar] [CrossRef]
- Schmidt, J.B.; Binder, M.; Macheiner, W.; Kainz, C.; Gitsch, G.; Bieglmayer, C. Treatment of Skin Ageing Symptoms in Perimenopausal Females with Estrogen Compounds. A Pilot Study. Maturitas 1994, 20, 25–30. [Google Scholar] [CrossRef]
- Atefi, N.; Dalvand, B.; Ghassemi, M.; Mehran, G.; Heydarian, A. Therapeutic Effects of Topical Tranexamic Acid in Comparison with Hydroquinone in Treatment of Women with Melasma. Dermatol. Ther. 2017, 7, 417–424. [Google Scholar] [CrossRef]
- Drugs.Com. Available online: https://www.drugs.com/ (accessed on 25 August 2022).
- Polonini, H.C.; Soldati, P.P.; Oliveira, M.A.L.D.; Brandão, M.A.F.; Chaves, M.d.G.M.; Raposo, N.R.B. Transdermal formulations containing human sexual steroids: Development and validation of methods and in vitro drug release. Quim. Nova 2014, 37, 720–727. [Google Scholar] [CrossRef]
- Ferreira, A.O.; Polonini, H.C.; Silva, S.L.; Patrício, F.B.; Brandão, M.A.F.; Raposo, N.R.B. Feasibility of Amlodipine Besylate, Chloroquine Phosphate, Dapsone, Phenytoin, Pyridoxine Hydrochloride, Sulfadiazine, Sulfasalazine, Tetracycline Hydrochloride, Trimethoprim and Zonisamide in SyrSpend® SF PH4 Oral Suspensions. J. Pharm. Biomed. Anal. 2016, 118, 105–112. [Google Scholar] [CrossRef]
- De Almeida, P.A.; Alves, M.C.; Polonini, H.C.; Dutra, L.S.; Leite, M.N.; Raposo, N.R.B.; Ferreira, A.D.O.; BrandÃo, M.A.F. New HPLC Method for Quality Control of β-Escin in Aesculus Hippocastanum L. Hydroalcoholic Extract. Lat. Am. J. Pharm. 2013, 32, 1082–1087. [Google Scholar]
- Polonini, H.C.; Bastos, C.D.A.; Oliveira, M.A.L.D.; Silva, C.G.A.D.; Collins, C.H.; Brandão, M.A.Ô.F.; Raposo, N.R.B. In Vitro Drug Release and Ex Vivo Percutaneous Absorption of Resveratrol Cream Using HPLC with Zirconized Silica Stationary Phase. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2014, 947–948, 23–31. [Google Scholar] [CrossRef]
- Guy, R.C. International Conference on Harmonisation. Encycl. Toxicol. Third Ed. 2014, 2, 1070–1072. [Google Scholar] [CrossRef]
- United States Pharmacopeial Convention <51> Antimicrobial Effectiveness Testing. In United States Pharmacopeia 40—National Formulary 35; 2020.
- Pong, A.; Raghavarao, D. Comparison of Bracketing and Matrixing Designs for a Two-Year Stability Study. J. Biopharm. Stat. 2000, 10, 217–228. [Google Scholar] [CrossRef]
- Conference, I.; Harmonisation, O.N.; Technical, O.F.; For, R.; Of, R.; For, P. International Conference on Harmonisation: Guidance on Q1D Bracketing and Matrixing Designs for Stability Testing of New Drug Substances and Products; Availability. Notice. Fed. Regist. 2003, 68, 2339–2340. [Google Scholar]
- United States Pharmacopeial Convention <795> Pharmaceutical Compounding—Nonsterile Preparations. In United States Pharmacopeia 43—National Formulary 38; 2020.
- Majekodunmi, B.D.; Lau-Cam, C.A.; Nash, R.A. Stability of Benzoyl Peroxide in Aromatic Ester-Containing Topical Formulations. Pharm. Dev. Technol. 2007, 12, 609–620. [Google Scholar] [CrossRef] [PubMed]
- Shatalebi, M.A.; Roostaei, M. Preparation and Physicochemical Evaluation of Benzoyl Peroxide 5% Foamable Emu Oil Emulsion. Jundishapur J. Nat. Pharm. Prod. 2015, 10, 3–8. [Google Scholar] [CrossRef]
- Langlah, N.; Pinsuwan, S.; Amnuaikit, T. Preparation and Physicochemical Study of Liposomes Containing Nicotinamide. In Proceedings of the 2012 7th IEEE International Conference on Nano/Micro Engineered and Molecular Systems (NEMS), Kyoto, Japan, 5–8 March 2012; pp. 537–541. [Google Scholar] [CrossRef]
- Jeon, J.S.; Lee, M.J.; Yoon, M.H.; Park, J.A.; Yi, H.; Cho, H.J.; Shin, H.C. Determination of Arbutin, Niacinamide, and Adenosine in Functional Cosmetic Products by High-Performance Liquid Chromatography. Anal. Lett. 2014, 47, 1650–1660. [Google Scholar] [CrossRef]
- Ilic, D.; Cvetkovic, M.; Tasic-Kostov, M. Emulsions with Alkyl Polyglucosides as Carriers for Off-Label Topical Spironolactone–Safety and Stability Evaluation. Pharm. Dev. Technol. 2021, 26, 373–379. [Google Scholar] [CrossRef]
- Allen, L.V.; Erickson, M.A. Stability of Ketoconazole, Metolazone, Metronidazole, Procainamide Hydrochloride, and Spironolactone in Extemporaneously Compounded Oral Liquids. Am. J. Health Pharm. 1996, 53, 2073–2078. [Google Scholar] [CrossRef]
- Tirnaksiz, F.; Kayiş, A.; Çelebi, N.; Adişen, E.; Erel, A. Preparation and Evaluation of Topical Microemulsion System Containing Metronidazole for Remission in Rosacea. Chem. Pharm. Bull. 2012, 60, 583–592. [Google Scholar] [CrossRef][Green Version]
- Basu Sarkar, A.; Kandimalla, A. Chemical Stability of Progesterone in Compounded Topical Preparations Using PLO Transdermal Cream and HRT Cream Base over a 90-Day Period at Two Controlled Temperatures. J. Steroids Horm. Sci. 2013, 04, 2–4. [Google Scholar] [CrossRef]
- Gatti, R.; Gioia, M.G.; Cavrini, V. Analysis and Stability Study of Retinoids in Pharmaceuticals by LC with Fluorescence Detection. J. Pharm. Biomed. Anal. 2000, 23, 147–159. [Google Scholar] [CrossRef]
- Temova Rakuša, Ž.; Škufca, P.; Kristl, A.; Roškar, R. Retinoid Stability and Degradation Kinetics in Commercial Cosmetic Products. J. Cosmet. Dermatol. 2021, 20, 2350–2358. [Google Scholar] [CrossRef] [PubMed]
| Active Pharmaceutical Ingredient | Concentrations Tested (mg/g) | Pharmaceutical Indication * |
|---|---|---|
| Benzoyl peroxide | 2.5% and 10.0% | Antibacterial drug, commonly used to treat acne |
| Cyproterone acetate | 0.5% and 2.0% | Steroid hormone used (in combination or not with other substances) to treat women with severe acne and symptoms of androgenization |
| Estriol | 0.1% and 1.0% | Estrogenic hormone used to improve general skin condition |
| Metronidazole | 0.75% and 5.0% | Antibiotic drug, used to treat a wide variety of skin infections |
| Niacinamide | 1.0% and 5.0% | Form of Vitamin B3, which can improve general skin condition and hydration when used topically |
| Progesterone | 0.5% and 2.0% | A steroid hormone used to improve general skin condition |
| Retinoic acid | 0.025% and 0.1% | Morphogen derived from retinol (Vitamin A), commonly used for treating severe acne |
| Spironolactone | 1.0% and 5.0% | Anti-androgen drug, used topically for treating hormonal acne |
| Tranexamic acid | 1.0% and 5.0% | A synthetic amino acid lysine derivate, which can act as a brightening agent to reduce dark spots and improve hyperpigmentation |
| Active Pharmaceutical Ingredient | Mobile Phase Composition | Working Concentration (µg/mL) | Column | Flux (mL/min) | Ultraviolet Detection Wavelength (nm) |
|---|---|---|---|---|---|
| Benzoyl peroxide | Acetonitrile and water (750:250, v/v) | 250.0, in acetonitrile; 20 µL injection | C18(L1), 4.6 mm × 250 mm; at 45 °C | 1.0 | 254 |
| Cyproterone acetate | Water, methanol, and acetonitrile (40:40:20, v/v/v) | 100.0, in methanol; 20 µL injection | C18(L1), 4.6 mm × 125 mm; at 40 °C | 1.5 | 282 |
| Estriol | Ethanol and water (60:40, v/v) | 40.0, in ethanol; 10 µL injection | C18(L1), 4.6 mm × 250 mm; at 30 °C | 0.3 | 205 |
| Metronidazole | Acetonitrile and Solution A (glacial acetic acid and water, 40:60, v/v) (40:60, v/v) | 20.0, in methanol; 10 µL injection | C18(L1), 4.6 mm × 250 mm; at 30 °C | 1.0 | 316 |
| Niacinamide | Methanol, acetic acid, and sulfonate buffer (27:1:73, v/v/v) | 100.0, in water; 20 µL injection | C18(L1), 4.6 mm × 150 mm; at 25 °C | 1.0 | 280 |
| Progesterone | Ethanol and water (65:35, v/v) | 100.0, in ethanol; 20 µL injection | C18(L1), 4.6 mm × 250 mm; at 45 °C | 1.2 | 254 |
| Retinoic acid | Methanol, water, and glacial acetic acid (80:20:0.5, v/v/v) | 10.0, in methanol; 50 µL injection | C18(L1), 4.6 mm × 150 mm; at 25 °C | 2.0 | 353 |
| Spironolactone | Water, phosphoric acid, methanol, and acetonitrile (435:2.7:50:515, v/v/v/v) | 100.0, in water; 20 µL injection | C18(L1), 4.6 mm × 250 mm; at 25 °C | 1.0 | 254 |
| Tranexamic acid | Phosphate buffer pH 2.5 and methanol (60:40, v/v) | 100.0, in ultra-purified water; 100 µL injection | C18(L1), 4.6 mm × 250 mm; at 35 °C | 1.0 | 220 |
| Active Pharmaceutical Ingredient | Linearity | Specificity | Precision | Accuracy | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Range (µg/mL) | Analytical Curve | R2 | ANOVA Significance of Regression (F) | LOD (µg/mL) | LOQ (µg/mL) | Discrepancy (%) | Repeatability (CV, %) | Intermediate Precision (CV, %) | Recovery (%) | |
| Benzoyl peroxide | 180.60–335.40 | y = 369,637.54x − 2,191,586.24 | 0.9988 | 5507.09 | 0.01 | 0.02 | 1.82 | 1.76 | 1.69 | 99.75 |
| Cyproterone acetate | 72.80–135.20 | y = 481,222.28x + 5,470,613.44 | 0.9917 | 774.07 | 0.01 | 0.02 | 0.86 | 0.99 | 0.98 | 98.04 |
| Estriol | 28.28–52.52 | y = 3,451,831.25x − 8,500,128.91 | 0.9951 | 1315.26 | 0.01 | 0.03 | 1.78 | 0.65 | 2.08 | 99.91 |
| Metronidazole | 14.01–26.03 | y = 95.83x − 29.50 | 0.9904 | 660.16 | 0.06 | 0.17 | 0.60 | 3.32 | 4.84 | 100.85 |
| Niacinamide | 72.24–134.16 | y = 58,009.28x + 197,874.53 | 0.9988 | 5547.81 | 0.03 | 0.09 | 1.17 | 0.15 | 0.59 | 99.52 |
| Progesterone | 70.56–131.04 | y = 27.36x + 40.39 | 0.9983 | 3845.49 | 0.97 | 0.32 | 0.85 | 0.56 | 0.81 | 99.74 |
| Retinoic acid | 7.49–13.91 | y = 330.11x + 191.91 | 0.9961 | 1641.15 | 0.12 | 0.37 | 1.81 | 1.17 | 2.57 | 101.09 |
| Spironolactone | 70.14–130.26 | y = 41.21x − 35.69 | 0.9993 | 9420.07 | 0.13 | 0.39 | 0.44 | 0.72 | 1.38 | 99.50 |
| Tranexamic acid | 145.60–270.40 | y = 31,315.34x − 82,589.51 | 0.9988 | 5499.49 | 20.01 | 6.60 | 1.52 | 1.87 | 2.91 | 99.85 |
| Active Pharmaceutical Ingredient | HCl (%d) | NaOH (%d) | UV (%d) | Heat (%d) | H2O2 (%d) |
|---|---|---|---|---|---|
| Benzoyl peroxide | 29.99 | −99.54 | 6.09 | −93.40 | 22.10 |
| Cyproterone acetate | −6.38 | −99.34 | 8.11 | 6.61 | 5.58 |
| Estriol | 147.94 | 163.85 | 3.23 | 609.02 | 12.57 |
| Metronidazole | 11.57 | −99.97 | 16.86 | −6.67 | 12.29 |
| Niacinamide | −92.17 | −86.23 | 25.70 | −6.11 | −3.23 |
| Progesterone | 8.91 | −68.25 | −0.89 | 11.36 | 4.29 |
| Retinoic acid | −31.76 | 0.99 | −47.85 | −27.78 | −7.05 |
| Spironolactone | −1.22 | −96.74 | −9.42 | −2.79 | 6.98 |
| Tranexamic acid | −5.54 | −78.56 | −8.78 | −7.17 | 3.54 |
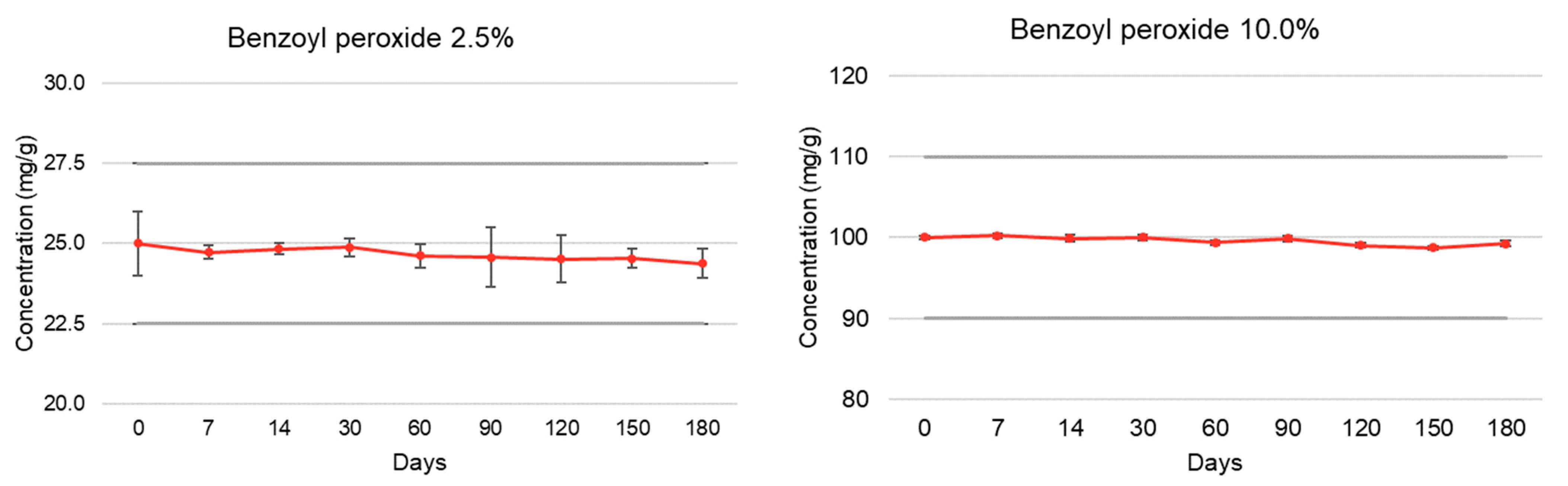
| Active Pharmaceutical Ingredient | Elapsed Time (Days) | % Recovery (Room Temperature, 15–30 °C) | |||
|---|---|---|---|---|---|
| Low Concentration | pH | High Concentration | pH | ||
| Benzoyl peroxide | T = 0 | 100.00 ± 1.00 | 4.82 | 100.00 ± 0.19 | 4.72 |
| (2.5% and 10.0%) | T = 7 | 98.92 ± 0.20 | 4.64 | 100.22 ± 0.26 | 4.67 |
| T = 14 | 99.33 ± 0.17 | 4.63 | 99.90 ± 0.43 | 4.70 | |
| T = 30 | 99.50 ± 0.28 | 4.65 | 100.00 ± 0.30 | 4.69 | |
| T = 60 | 98.48 ± 0.37 | 4.68 | 99.36 ± 0.28 | 4.52 | |
| T = 90 | 98.30 ± 0.94 | 4.67 | 99.88 ± 0.35 | 4.48 | |
| T = 120 | 98.06 ± 0.74 | 4.63 | 99.03 ± 0.30 | 4.51 | |
| T = 150 | 98.15 ± 0.30 | 4.66 | 98.72 ± 0.19 | 4.50 | |
| T = 180 | 97.52 ± 0.46 | 4.65 | 99.23 ± 0.37 | 4.51 | |
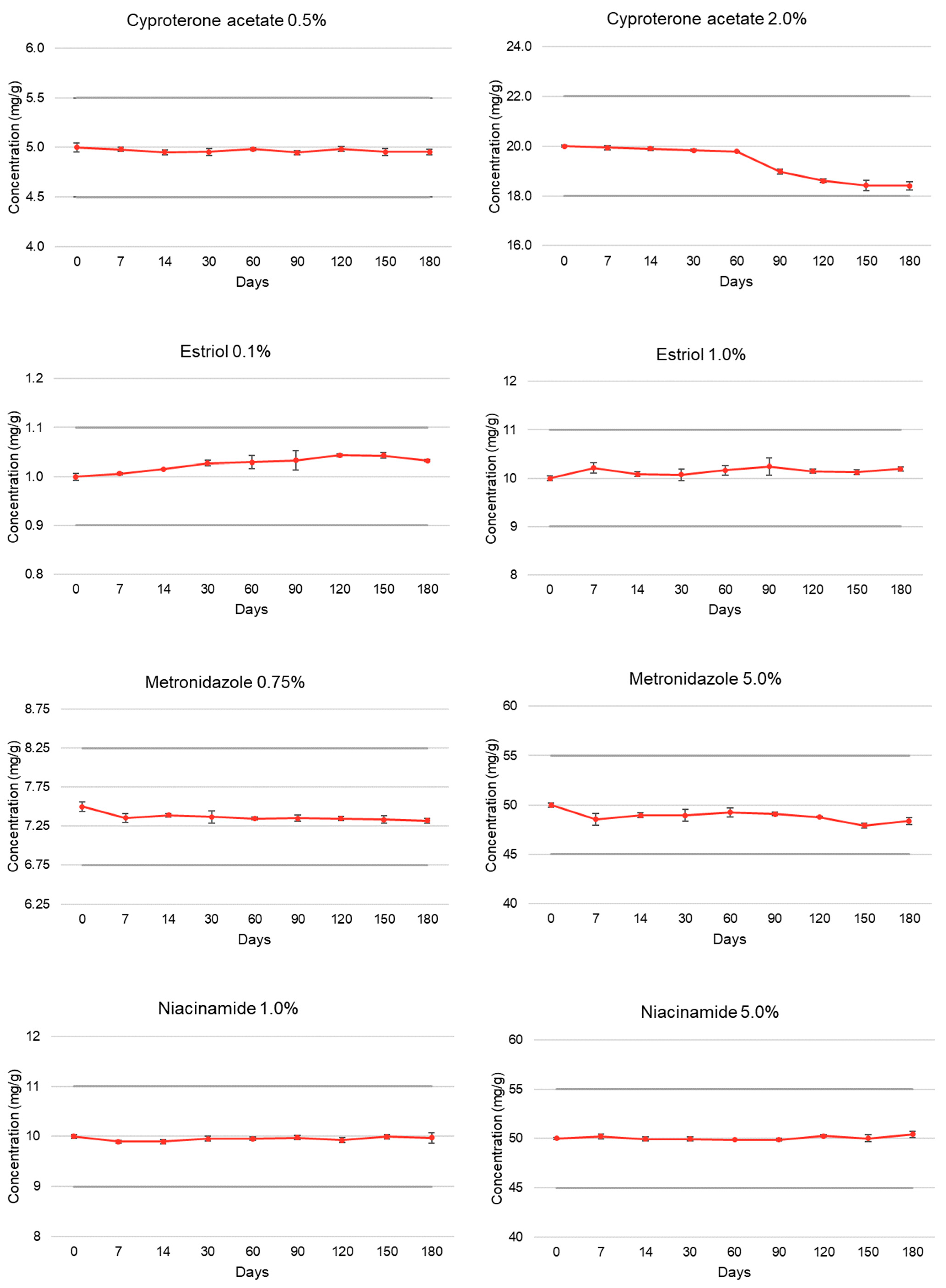
| Cyproterone acetate | T = 0 | 100.00 ± 0.85 | 5.01 | 100.00 ± 0.22 | 5.01 |
| (0.5% and 2.0%) | T = 7 | 99.60 ± 0.44 | 5.06 | 99.73 ± 0.44 | 5.03 |
| T = 14 | 99.06 ± 0.52 | 5.04 | 99.51 ± 0.36 | 5.01 | |
| T = 30 | 99.11 ± 0.67 | 5.07 | 99.19 ± 0.24 | 5.06 | |
| T = 60 | 99.65 ± 0.26 | 4.98 | 98.94 ± 0.07 | 4.92 | |
| T = 90 | 98.95 ± 0.40 | 4.44 | 94.91 ± 0.52 | 4.94 | |
| T = 120 | 99.69 ± 0.47 | 4.99 | 93.06 ± 0.41 | 4.94 | |
| T = 150 | 99.09 ± 0.77 | 4.99 | 92.13 ± 1.13 | 4.94 | |
| T = 180 | 99.10 ± 0.52 | 4.99 | 92.09 ± 0.90 | 4.94 | |
| Estriol | T = 0 | 100.00 ± 0.70 | 5.15 | 100.00 ± 0.53 | 5.13 |
| (0.1% and 1.0%) | T = 7 | 100.63 ± 0.21 | 5.09 | 102.13 ± 1.09 | 5.05 |
| T = 14 | 101.53 ± 0.06 | 5.15 | 100.84 ± 0.44 | 5.20 | |
| T = 30 | 102.74 ± 0.59 | 5.07 | 100.72 ± 1.18 | 5.11 | |
| T = 60 | 102.94 ± 1.33 | 5.13 | 101.67 ± 0.94 | 5.17 | |
| T = 90 | 103.31 ± 1.88 | 5.14 | 102.43 ± 1.73 | 5.15 | |
| T = 120 | 104.32 ± 0.27 | 5.17 | 101.48 ± 0.40 | 5.09 | |
| T = 150 | 104.30 ± 0.53 | 5.06 | 101.24 ± 0.49 | 5.06 | |
| T = 180 | 103.23 ± 0.20 | 5.09 | 101.95 ± 0.43 | 5.05 | |
| Metronidazole | T = 0 | 100.00 ± 0.82 | 5.13 | 100.00 ± 0.41 | 5.14 |
| (0.75% and 5.0%) | T = 7 | 98.07 ± 0.75 | 5.16 | 97.08 ± 1.26 | 5.15 |
| T = 14 | 98.57 ± 0.31 | 5.21 | 97.89 ± 0.49 | 5.23 | |
| T = 30 | 98.25 ± 1.08 | 5.18 | 97.88 ± 1.24 | 5.20 | |
| T = 60 | 98.00 ± 0.19 | 5.12 | 98.49 ± 0.92 | 5.11 | |
| T = 90 | 98.06 ± 0.56 | 5.08 | 98.18 ± 0.37 | 5.11 | |
| T = 120 | 98.00 ± 0.34 | 5.09 | 97.51 ± 0.14 | 5.11 | |
| T = 150 | 97.80 ± 0.64 | 5.10 | 95.80 ± 0.56 | 5.13 | |
| T = 180 | 97.62 ± 0.45 | 5.12 | 96.72 ± 0.76 | 5.12 | |
| Niacinamide | T = 0 | 100.00 ± 0.37 | 5.38 | 100.00 ± 0.21 | 5.39 |
| (1.0% and 5.0%) | T = 7 | 98.99 ± 0.27 | 5.22 | 100.42 ± 0.53 | 5.50 |
| T = 14 | 98.95 ± 0.48 | 5.24 | 99.90 ± 0.39 | 5.51 | |
| T = 30 | 99.57 ± 0.43 | 5.26 | 99.91 ± 0.42 | 5.52 | |
| T = 60 | 99.55 ± 0.34 | 5.22 | 99.72 ± 0.12 | 5.42 | |
| T = 90 | 99.76 ± 0.46 | 5.23 | 99.76 ± 0.32 | 5.50 | |
| T = 120 | 99.26 ± 0.52 | 5.25 | 100.54 ± 0.21 | 5.51 | |
| T = 150 | 99.94 ± 0.45 | 5.22 | 100.05 ± 0.67 | 5.53 | |
| T = 180 | 99.72 ± 1.05 | 5.25 | 100.84 ± 0.57 | 5.50 | |
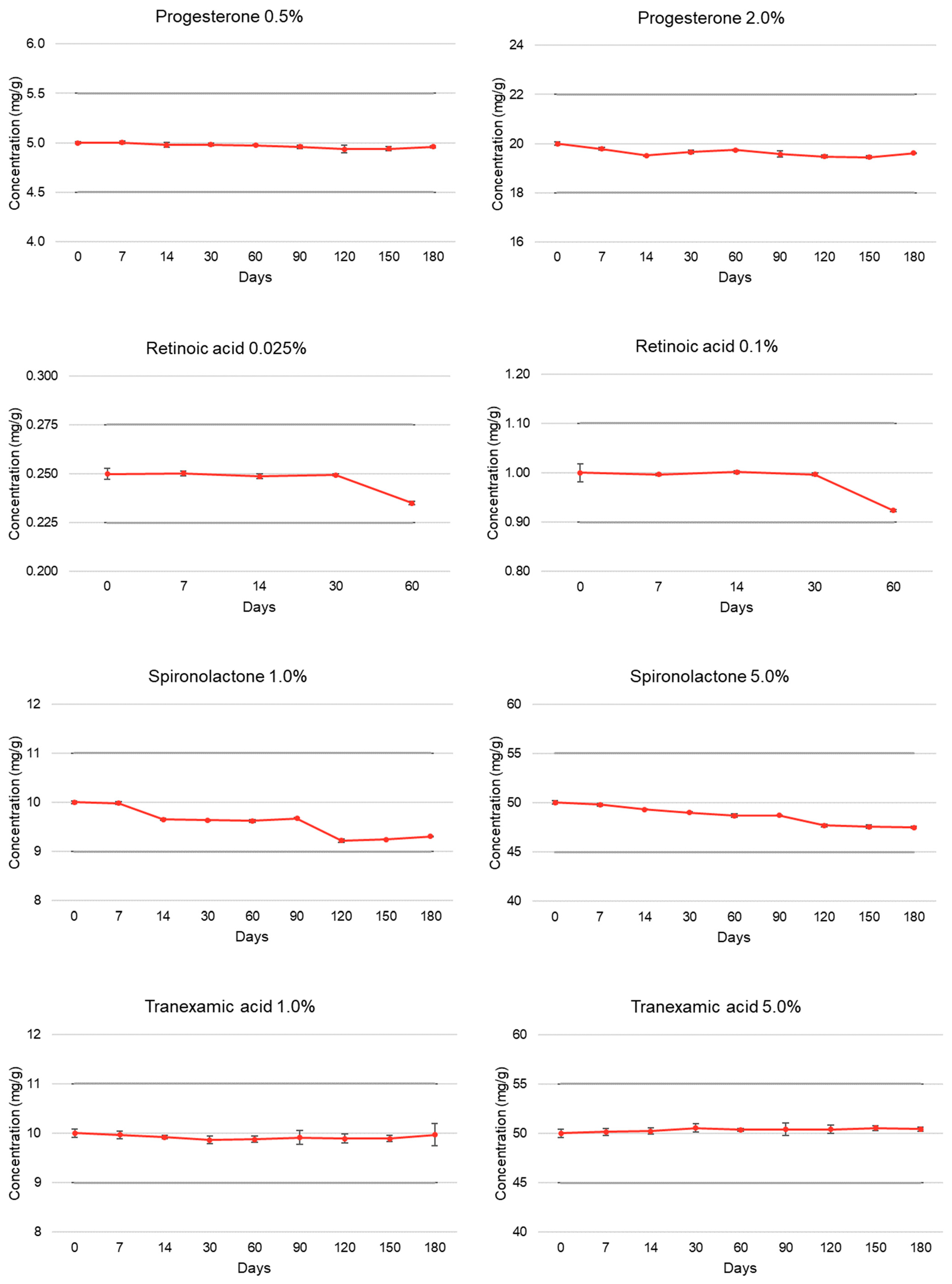
| Progesterone | T = 0 | 100.00 ± 0.26 | 5.18 | 100.00 ± 0.32 | 4.98 |
| (0.5% and 2.0%) | T = 7 | 100.05 ± 0.27 | 5.16 | 98.91 ± 0.30 | 4.97 |
| T = 14 | 99.58 ± 0.49 | 5.18 | 97.58 ± 0.19 | 4.97 | |
| T = 30 | 99.64 ± 0.31 | 5.23 | 98.31 ± 0.37 | 4.55 | |
| T = 60 | 99.48 ± 0.13 | 5.21 | 98.71 ± 0.11 | 4.94 | |
| T = 90 | 99.22 ± 0.34 | 5.13 | 97.87 ± 0.64 | 4.95 | |
| T = 120 | 98.73 ± 0.76 | 5.14 | 97.41 ± 0.31 | 4.91 | |
| T = 150 | 98.82 ± 0.38 | 5.12 | 97.24 ± 0.32 | 4.93 | |
| T = 180 | 99.18 ± 0.20 | 4.96 | 98.09 ± 0.06 | 5.06 | |
| Retinoic acid | T = 0 | 100.00 ± 1.11 | 5.09 | 100.00 ± 1.81 | 5.10 |
| (0.025% and 0.1%) | T = 7 | 100.08 ± 0.44 | 4.94 | 99.64 ± 0.19 | 5.05 |
| T = 14 | 99.49 ± 0.52 | 4.91 | 100.16 ± 0.31 | 5.03 | |
| T = 30 | 99.78 ± 0.24 | 4.90 | 99.70 ± 0.27 | 5.04 | |
| T = 60 | 94.00 ± 0.36 * | 5.01 | 92.35 ± 0.28 * | 4.90 | |
| Spironolactone | T = 0 | 100.00 ± 0.28 | 5.12 | 100.00 ± 0.36 | 5.08 |
| (1.0% and 5.0%) | T = 7 | 99.79 ± 0.29 | 5.15 | 99.60 ± 0.32 | 5.04 |
| T = 14 | 96.52 ± 0.26 | 5.12 | 98.63 ± 0.09 | 5.09 | |
| T = 30 | 96.39 ± 0.13 | 5.13 | 98.02 ± 0.10 | 5.07 | |
| T = 60 | 96.21 ± 0.31 | 5.06 | 97.34 ± 0.36 | 5.09 | |
| T = 90 | 96.74 ± 0.10 | 5.07 | 97.39 ± 0.12 | 5.05 | |
| T = 120 | 92.19 ± 0.35 | 5.09 | 95.39 ± 0.30 | 5.03 | |
| T = 150 | 92.46 ± 0.12 | 5.07 | 95.13 ± 0.36 | 5.05 | |
| T = 180 | 93.10 ± 0.06 | 5.09 | 94.96 ± 0.21 | 5.06 | |
| Tranexamic acid | T = 0 | 100.00 ± 0.87 | 5.91 | 100.00 ± 0.85 | 6.38 |
| (1.0% and 5.0%) | T = 7 | 99.68 ± 0.77 | 5.83 | 100.31 ± 0.68 | 6.52 |
| T = 14 | 99.20 ± 0.38 | 5.79 | 100.45 ± 0.64 | 6.45 | |
| T = 30 | 98.61 ± 0.76 | 5.69 | 101.11 ± 0.79 | 6.43 | |
| T = 60 | 98.78 ± 0.64 | 5.59 | 100.75 ± 0.28 | 6.43 | |
| T = 90 | 99.14 ± 1.38 | 5.51 | 100.83 ± 1.31 | 6.41 | |
| T = 120 | 98.92 ± 0.97 | 5.53 | 100.82 ± 0.88 | 6.46 | |
| T = 150 | 98.90 ± 0.65 | 5.49 | 101.05 ± 0.53 | 6.44 | |
| T = 180 | 99.69 ± 2.25 | 5.55 | 100.87 ± 0.40 | 6.41 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Polonini, H.; Marianni, B.; Taylor, S.; Zander, C. Compatibility of Personalized Formulations in Cleoderm™, A Skin Rebalancing Cream Base for Oily and Sensitive Skin. Cosmetics 2022, 9, 92. https://doi.org/10.3390/cosmetics9050092
Polonini H, Marianni B, Taylor S, Zander C. Compatibility of Personalized Formulations in Cleoderm™, A Skin Rebalancing Cream Base for Oily and Sensitive Skin. Cosmetics. 2022; 9(5):92. https://doi.org/10.3390/cosmetics9050092
Chicago/Turabian StylePolonini, Hudson, Bruna Marianni, Sarah Taylor, and Clark Zander. 2022. "Compatibility of Personalized Formulations in Cleoderm™, A Skin Rebalancing Cream Base for Oily and Sensitive Skin" Cosmetics 9, no. 5: 92. https://doi.org/10.3390/cosmetics9050092
APA StylePolonini, H., Marianni, B., Taylor, S., & Zander, C. (2022). Compatibility of Personalized Formulations in Cleoderm™, A Skin Rebalancing Cream Base for Oily and Sensitive Skin. Cosmetics, 9(5), 92. https://doi.org/10.3390/cosmetics9050092






