Valorization of Wine-Making By-Products’ Extracts in Cosmetics
Abstract
:1. Introduction
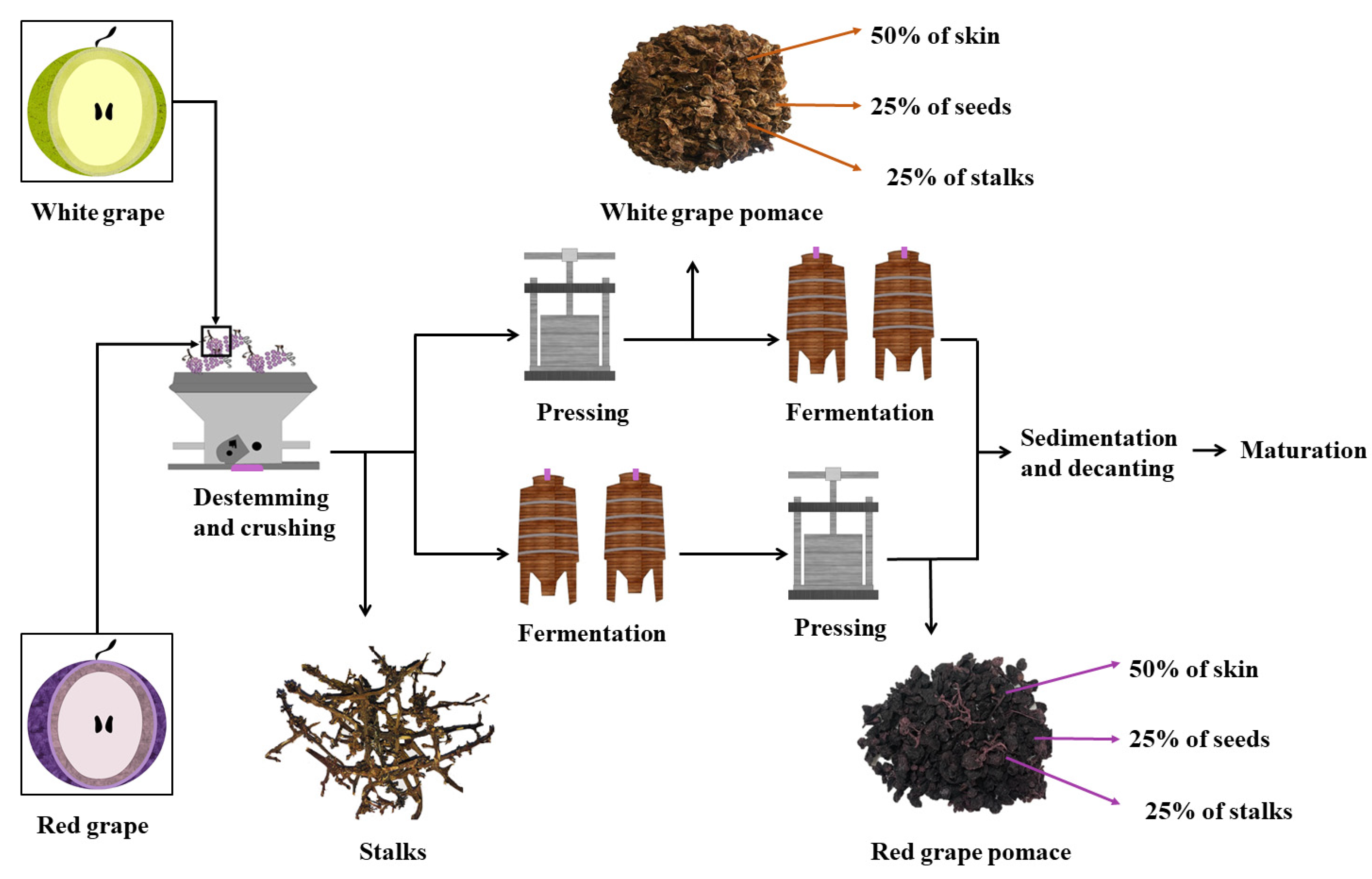
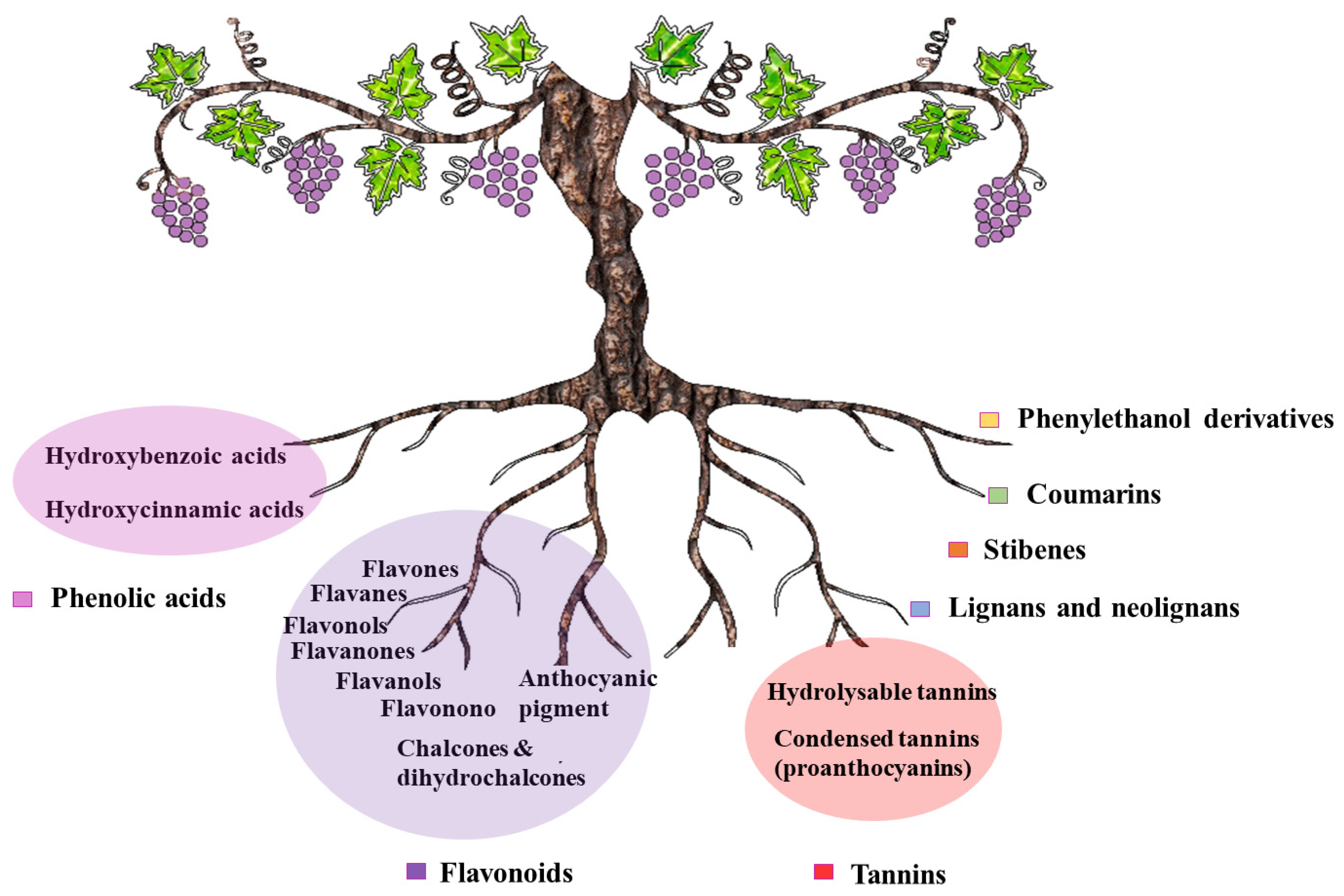
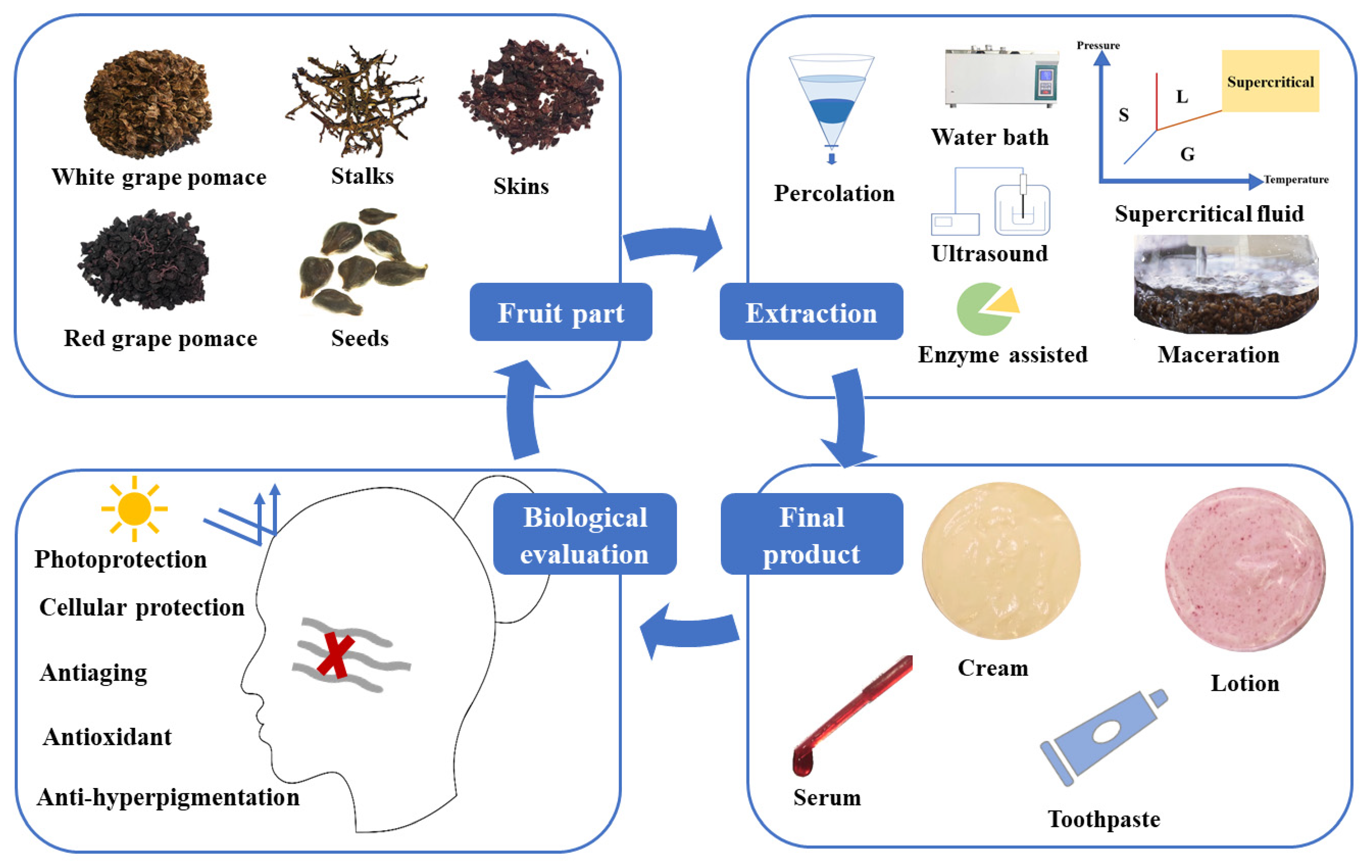
2. Extraction of Phenolic Compounds from Grape By-Products
3. Potential In Vitro Cosmetic Applications of Grape Pomace Extracts
3.1. Antioxidant Activity
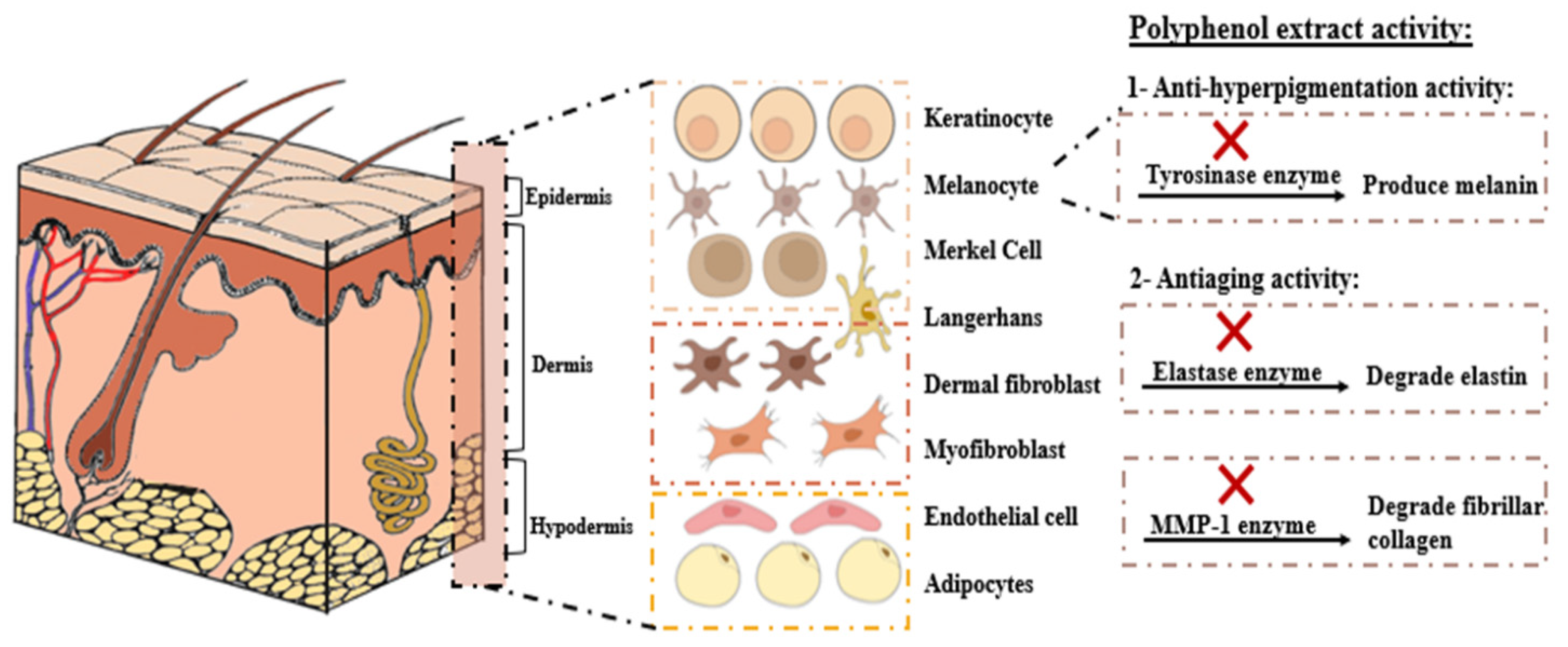
3.2. Anti-Hyperpigmentation Activity
3.3. Antiaging Activity
4. Incorporation of the Grape By-Products Extracts in Cosmetic Products and Their In Vitro and/or In Vivo Testing
4.1. Sunscreen Application
4.2. Skin Penetration
4.3. Antiaging and Skin Depigmenting Activities
4.4. Oral Care Application
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- El Kantar, S.; Rajha, H.N.; Maroun, R.G.; Louka, N. Intensification of polyphenols extraction from orange peels using infrared as a novel and energy saving pretreatment. J. Food Sci. 2020, 85, 414–420. [Google Scholar] [CrossRef]
- El Kantar, S.; Boussetta, N.; Rajha, H.N.; Maroun, R.G.; Louka, N.; Vorobiev, E. High voltage electrical discharges combined with enzymatic hydrolysis for extraction of polyphenols and fermentable sugars from orange peels. Food Res. Int. 2018, 107, 755–762. [Google Scholar] [CrossRef]
- El Kantar, S.; Boussetta, N.; Lebovka, N.; Foucart, F.; Rajha, H.N.; Maroun, R.G.; Louka, N.; Vorobiev, E. Pulsed electric field treatment of citrus fruits: Improvement of juice and polyphenols extraction. Innov. Food Sci. Emerg. Technol. 2018, 46, 153–161. [Google Scholar] [CrossRef]
- El Kantar, S.; Rajha, H.N.; Boussetta, N.; Vorobiev, E.; Maroun, R.G.; Louka, N. Green extraction of polyphenols from grapefruit peels using high voltage electrical discharges, deep eutectic solvents and aqueous glycerol. Food Chem. 2019, 295, 165–171. [Google Scholar] [CrossRef] [PubMed]
- Abi-Khattar, A.M.; Rajha, H.N.; Abdel-Massih, R.M.; Habchi, R.; Maroun, R.G.; Debs, E.; Louka, N. “Intensification of Vaporization by Decompression to the Vacuum” (IVDV), a novel technology applied as a pretreatment to improve polyphenols extraction from olive leaves. Food Chem. 2021, 342, 128236. [Google Scholar] [CrossRef] [PubMed]
- Abi-Khattar, A.M.; Rajha, H.N.; Abdel-Massih, R.M.; Maroun, R.G.; Louka, N.; Debs, E. Intensification of polyphenol extraction from olive leaves using ired-irrad®, an environmentally-friendly innovative technology. Antioxidants 2019, 8, 227. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Abi-Khattar, A.M.; Rajha, H.N.; Maroun, R.G.; Abdel-Massih, R.M.; Louka, N.; Debs, E. Green extraction of polyphenols from olive leaves using ired-irrad® as a pretreatment. In Proceedings of the 5th International Conference on Renewable Energy in Developing Countries REDEC 2020, Marrakech, Morocco, 29–30 June 2020; pp. 1–5. [Google Scholar] [CrossRef]
- Rajha, H.N.; Abi-Khattar, A.M.; El Kantar, S.; Boussetta, N.; Lebovka, N.; Maroun, R.G.; Louka, N.; Vorobiev, E. Comparison of aqueous extraction efficiency and biological activities of polyphenols from pomegranate peels assisted by infrared, ultrasound, pulsed electric fields and high-voltage electrical discharges. Innov. Food Sci. Emerg. Technol. 2019, 58, 102212. [Google Scholar] [CrossRef]
- Rajha, H.N.; Mhanna, T.; El Kantar, S.; El Khoury, A.; Louka, N.; Maroun, R.G. Innovative process of polyphenol recovery from pomegranate peels by combining green deep eutectic solvents and a new infrared technology. LWT 2019, 111, 138–146. [Google Scholar] [CrossRef]
- Rajha, H.N.; Koubaa, M.; Boussetta, N.; Maroun, R.G.; Louka, N.; Lebovka, N.; Vorobiev, E. Selective ultrasound-assisted aqueous extraction of polyphenols from pomegranate peels and seeds. J. Food Process. Preserv. 2020, 44, e14545. [Google Scholar] [CrossRef]
- El Darra, N.; Rajha, H.N.; Debs, E.; Saleh, F.; El-Ghazzawi, I.; Louka, N.; Maroun, R.G. Comparative Study between Ethanolic and β -Cyclodextrin Assisted Extraction of Polyphenols from Peach Pomace. Int. J. Food Sci. 2018, 2018, 9491681. [Google Scholar] [CrossRef] [Green Version]
- Rajha, H.N.; Boussetta, N.; Louka, N.; Maroun, R.G.; Vorobiev, E. Pulsed Electric Fields and High Voltage Electrical Discharge Assisted Extraction of Biocompounds from Vine Shoots. In Handbook of Electroporation; Springer: Cham, Switzerland, 2016; pp. 1–16. ISBN 9783319267791. [Google Scholar]
- Rajha, H.N.; El Kantar, S.; Afif, C.; Boussetta, N.; Louka, N.; Maroun, R.G.; Vorobiev, E. Selective multistage extraction process of biomolecules from vine shoots by a combination of biological, chemical, and physical treatments. Comptes Rendus Chim. 2018, 21, 581–589. [Google Scholar] [CrossRef]
- Rajha, H.N.; Boussetta, N.; Louka, N.; Maroun, R.G.; Vorobiev, E. Electrical, mechanical, and chemical effects of high-voltage electrical discharges on the polyphenol extraction from vine shoots. Innov. Food Sci. Emerg. Technol. 2015, 31, 60–66. [Google Scholar] [CrossRef]
- Rajha, H.N.; Boussetta, N.; Louka, N.; Maroun, R.G.; Vorobiev, E. Effect of alternative physical pretreatments (pulsed electric field, high voltage electrical discharges and ultrasound) on the dead-end ultrafiltration of vine-shoot extracts. Sep. Purif. Technol. 2015, 146, 243–251. [Google Scholar] [CrossRef]
- Rajha, H.N.; Chacar, S.; Afif, C.; Vorobiev, E.; Louka, N.; Maroun, R.G. β-Cyclodextrin-Assisted Extraction of Polyphenols from Vine Shoot Cultivars. J. Agric. Food Chem. 2015, 63, 3387–3393. [Google Scholar] [CrossRef]
- Rajha, H.N.; Boussetta, N.; Louka, N.; Maroun, R.G.; Vorobiev, E. A comparative study of physical pretreatments for the extraction of polyphenols and proteins from vine shoots. Food Res. Int. 2014, 65, 462–468. [Google Scholar] [CrossRef]
- Rajha, H.N.; Ziegler, W.; Louka, N.; Hobaika, Z.; Vorobiev, E.; Boechzelt, H.G.; Maroun, R.G. Effect of the drying process on the intensification of phenolic compounds recovery from grape pomace using accelerated solvent extraction. Int. J. Mol. Sci. 2014, 15, 18640–18658. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rajha, H.N.; El Darra, N.; Hobaika, Z.; Boussetta, N.; Vorobiev, E.; Maroun, R.G.; Louka, N. Extraction of Total Phenolic Compounds, Flavonoids, Anthocyanins and Tannins from Grape Byproducts by Response Surface Methodology. Influence of Solid-Liquid Ratio, Particle Size, Time, Temperature and Solvent Mixtures on the Optimization Process. Food Nutr. Sci. 2014, 5, 397–409. [Google Scholar] [CrossRef] [Green Version]
- Rajha, H.N.; Louka, N.; El Darra, N.; Hobaika, Z.; Boussetta, N.; Vorobiev, E.; Maroun, R.G. Multiple Response Optimization of High Temperature, Low Time Aqueous Extraction Process of Phenolic Compounds from Grape Byproducts. Food Nutr. Sci. 2014, 5, 351–360. [Google Scholar] [CrossRef] [Green Version]
- Rajha, H.N.; El Darra, N.; Vorobiev, E.; Louka, N.; Maroun, R.G. An Environment Friendly, Low-Cost Extraction Process of Phenolic Compounds from Grape Byproducts. Optimization by Multi-Response Surface Methodology. Food Nutr. Sci. 2013, 4, 650–659. [Google Scholar] [CrossRef] [Green Version]
- Manca, M.L.; Firoznezhad, M.; Caddeo, C.; Marongiu, F.; Escribano-Ferrer, E.; Sarais, G.; Peris, J.E.; Usach, I.; Zaru, M.; Manconi, M.; et al. Phytocomplexes extracted from grape seeds and stalks delivered in phospholipid vesicles tailored for the treatment of skin damages. Ind. Crops Prod. 2019, 128, 471–478. [Google Scholar] [CrossRef]
- Spinei, M.; Oroian, M. The potential of grape pomace varieties as a dietary source of pectic substances. Foods 2021, 10, 867. [Google Scholar] [CrossRef] [PubMed]
- Nigam, P.S.N.; Pandey, A. (Eds.) Biotechnology for Agro-Industrial Residues Utilisation: Utilisation of Agro-Residues; Springer Science and Business Media: New York, NY, USA, 2009; pp. 293–296. ISBN 9781402099410. [Google Scholar]
- Muhlack, R.A.; Potumarthi, R.; Jeffery, D.W. Sustainable wineries through waste valorisation: A review of grape marc utilisation for value-added products. Waste Manag. 2018, 72, 99–118. [Google Scholar] [CrossRef] [PubMed]
- FAO-OIV Focus 2016 Table and Dried Grapes; FAO: Rome, Italy, 2016.
- Lee, S.Y.; Kim, B.N.; Han, J.H.; Chang, S.T.; Choi, Y.W.; Kim, Y.H.; Min, J. Treatment of phenol-contaminated soil by Corynebacterium glutamicum and toxicity removal evaluation. J. Hazard. Mater. 2010, 182, 937–940. [Google Scholar] [CrossRef]
- Soceanu, A.; Dobrinas, S.; Sirbu, A.; Manea, N.; Popescu, V. Economic aspects of waste recovery in the wine industry. A multidisciplinary approach. Sci. Total Environ. 2021, 759, 143543. [Google Scholar] [CrossRef]
- Lin, D.; Xiao, M.; Zhao, J.; Li, Z.; Xing, B.; Li, X.; Kong, M.; Li, L.; Zhang, Q.; Liu, Y.; et al. An overview of plant phenolic compounds and their importance in human nutrition and management of type 2 diabetes. Molecules 2016, 21, 1374. [Google Scholar] [CrossRef]
- Garrido, J.; Borges, F. Wine and grape polyphenols—A chemical perspective. Food Res. Int. 2013, 54, 1844–1858. [Google Scholar] [CrossRef] [Green Version]
- Daayf, F.; El Hadrami, A.; El-Bebany, A.F.; Henriquez, M.A.; Yao, Z.; Derksen, H.; El-Hadrami, I.; Adam, L.R. Phenolic Compounds in Plant Defense and Pathogen Counter-Defense Mechanisms. In Recent Advances in Polyphenol Research; Wiley: Hoboken, NJ, USA, 2012; Volume 3, pp. 191–208. ISBN 9781444337464. [Google Scholar]
- Nunes, M.A.; Rodrigues, F.; Oliveira, M.B.P.P. Grape Processing By-Products as Active Ingredients for Cosmetic Proposes. In Handbook of Grape Processing By-Products; Galanakis, C.M., Ed.; Elsevier Inc.: Amsterdam, The Netherlands, 2017; pp. 267–292. ISBN 9780128098714. [Google Scholar]
- De Lima Cherubim, D.J.; Buzanello Martins, C.V.; Oliveira Fariña, L.; da Silva de Lucca, R.A. Polyphenols as natural antioxidants in cosmetics applications. J. Cosmet. Dermatol. 2020, 19, 33–37. [Google Scholar] [CrossRef]
- Morocho-Jácome, A.L.; Freire, T.B.; de Oliveira, A.C.; de Almeida, T.S.; Rosado, C.; Velasco, M.V.R.; Baby, A.R. In vivo SPF from multifunctional sunscreen systems developed with natural compounds—A review. J. Cosmet. Dermatol. 2020, 20, 729–737. [Google Scholar] [CrossRef]
- Richard, G.; Maroun, H.N.R.; Eugene Vorobiev, N.L. Emerging Technologies for the Recovery of Valuable Compounds From Grape Processing By-Products. In Handbook of Grape Processing By-Products; Galanakis, C.M., Ed.; Elsevier Inc.: Amsterdam, The Netherlands, 2017; pp. 155–181. ISBN 9780128098707. [Google Scholar]
- Simonetti, G.; Brasili, E.; Pasqua, G. Antifungal Activity of Phenolic and Polyphenolic Compounds from Different Matrices of Vitis vinifera L. Against Human Pathogens. Molecules 2020, 25, 3748. [Google Scholar] [CrossRef]
- Matos, M.S.; Romero-Díez, R.; Álvarez, A.; Bronze, M.R.; Rodríguez-Rojo, S.; Mato, R.B.; Cocero, M.J.; Matias, A.A. Polyphenol-rich extracts obtained from winemaking waste streams as natural ingredients with cosmeceutical potential. Antioxidants 2019, 8, 355. [Google Scholar] [CrossRef] [Green Version]
- Perra, M.; Lozano-Sánchez, J.; Leyva-Jiménez, F.J.; Segura-Carretero, A.; Pedraz, J.L.; Bacchetta, G.; Muntoni, A.; De Gioannis, G.; Manca, M.L.; Manconi, M. Extraction of the antioxidant phytocomplex from wine-making by-products and sustainable loading in phospholipid vesicles specifically tailored for skin protection. Biomed. Pharmacother. 2021, 142, 111959. [Google Scholar] [CrossRef] [PubMed]
- Emmulo, E.; Ceccantoni, B.; Bellincontro, A.; Mencarelli, F. Use of water and ethanol extracts from wine grape seed pomace to prepare an antioxidant toothpaste. J. Sci. Food Agric. 2021, 101, 5813–5818. [Google Scholar] [CrossRef]
- Michailidis, D.; Angelis, A.; Nikolaou, P.E.; Mitakou, S.; Skaltsounis, A.L. Exploitation of vitis vinifera, foeniculum vulgare, cannabis sativa and punica granatum by-product seeds as dermo-cosmetic agents. Molecules 2021, 26, 731. [Google Scholar] [CrossRef] [PubMed]
- Yarovaya, L.; Waranuch, N.; Wisuitiprot, W.; Khunkitti, W. Effect of grape seed extract on skin fibroblasts exposed to UVA light and its photostability in sunscreen formulation. J. Cosmet. Dermatol. 2020, 20, 1271–1282. [Google Scholar] [CrossRef] [PubMed]
- Khunkitti, L.Y. and W. The Effect of Grape Seed Extract as a Sunscreen Booster. Songklanakarin J. Sci. Technol. 2019, 4, 708–715. [Google Scholar] [CrossRef]
- Waqas, M.K.; Akhtar, N.; Rasul, A.; Sethi, A.; Abbas, K.; Hussain, T. Non-invasive in vivo evaluations of cosmetic emulsion containing phytoextract of grape seeds in the treatment of skin aging by using non-invasive bio-engineering techniques. Acta Pol. Pharm. Drug Res. 2018, 75, 97–105. [Google Scholar]
- Ferri, M.; Rondini, G.; Calabretta, M.M.; Michelini, E.; Vallini, V.; Fava, F.; Roda, A.; Minnucci, G.; Tassoni, A. White grape pomace extracts, obtained by a sequential enzymatic plus ethanol-based extraction, exert antioxidant, anti-tyrosinase and anti-inflammatory activities. N. Biotechnol. 2017, 39, 51–58. [Google Scholar] [CrossRef]
- Hübner, A.A.; Sarruf, F.D.; Oliveira, C.A.; Neto, A.V.; Fischer, D.C.H.; Kato, E.T.M.; Lourenço, F.R.; Baby, A.R.; Bacchi, E.M. Safety and photoprotective efficacy of a sunscreen system based on grape pomace (Vitis vinifera L.) phenolics from winemaking. Pharmaceutics 2020, 12, 1148. [Google Scholar] [CrossRef]
- Leal, C.; Gouvinhas, I.; Santos, R.A.; Rosa, E.; Silva, A.M.; Saavedra, M.J.; Barros, A.I.R.N.A. Potential application of grape (Vitis vinifera L.) stem extracts in the cosmetic and pharmaceutical industries: Valorization of a by-product. Ind. Crops Prod. 2020, 154, 112675. [Google Scholar] [CrossRef]
- Maluf, D.F.; Gonçalves, M.M.; D’Angelo, R.W.O.; Girassol, A.B.; Tulio, A.P.; Pupo, Y.M.; Farago, P.V. Cytoprotection of antioxidant biocompounds from grape pomace: Further exfoliant phytoactive ingredients for cosmetic products. Cosmetics 2018, 5, 46. [Google Scholar] [CrossRef] [Green Version]
- Panić, M.; Gunjević, V.; Radošević, K.; Bubalo, M.C.; Ganić, K.K.; Redovniković, I.R. Cosmotherm as an effective tool for selection of deep eutectic solvents based ready-to-use extracts from Graševina grape pomace. Molecules 2021, 26, 4722. [Google Scholar] [CrossRef] [PubMed]
- Rafique, M.; Nisar, S.; Shah, H.; Hussain, I. Development of grape seed extract based formulations by using non-invasive biophysical technique and its impact on skin aging Development of grape seed extract based formulations by using non- invasive biophysical technique and its impact on skin aging. Pak. J. Pharm. Sci. 2021, 34, 1621–1628. [Google Scholar] [CrossRef]
- Limsuwan, T.; Amnuikit, T. Effect of grape seed extract in sunscreen lotion on sun protection factor (SPF) determined by in vitro method. ACM Int. Conf. Proceeding Ser. 2017, F1309, 109–112. [Google Scholar] [CrossRef]
- Rafique, M.; Hussain Shah, S.N. Anti-Ageing Potential of a Cream (W/O Emulsion) Containing Grape Seed Extract (GSE): Formulation and in vivo Evaluation of Effectiveness Using Non-Invasive Biophysical Technique. J. Clin. Exp. Dermatol. Res. 2019, 10, 2155–9554. [Google Scholar] [CrossRef] [Green Version]
- Surini, S.; Mubarak, H.; Ramadon, D. Cosmetic serum containing grape (Vitis vinifera L.) seed extract phytosome: Formulation and in vitro penetration study. J. Young Pharm. 2018, 10, s51–s55. [Google Scholar] [CrossRef] [Green Version]
- Sharif, A.; Akhtar, N.; Khan, M.S.; Menaa, A.; Menaa, B.; Khan, B.A.; Menaa, F. Formulation and evaluation on human skin of a water-in-oil emulsion containing Muscat hamburg black grape seed extract. Int. J. Cosmet. Sci. 2014, 37, 253–258. [Google Scholar] [CrossRef]
- Soto, M.L.; Falqué, E.; Domínguez, H. Relevance of natural phenolics from grape and derivative products in the formulation of cosmetics. Cosmetics 2015, 2, 259–276. [Google Scholar] [CrossRef] [Green Version]
- Caddeo, C.; Manca, M.L.; Peris, J.E.; Usach, I.; Diez-Sales, O.; Matos, M.; Fernàndez-Busquets, X.; Fadda, A.M.; Manconi, M. Tocopherol-loaded transfersomes: In vitro antioxidant activity and efficacy in skin regeneration. Int. J. Pharm. 2018, 551, 34–41. [Google Scholar] [CrossRef]
- Anastasiadi, M.; Pratsinis, H.; Kletsas, D.; Skaltsounis, A.L.; Haroutounian, S.A. Grape stem extracts: Polyphenolic content and assessment of their in vitro antioxidant properties. LWT Food Sci. Technol. 2012, 48, 316–322. [Google Scholar] [CrossRef]
- Franco, D.; Sineiro, J.; Rubilar, M.; Sánchez, M.; Jerez, M.; Pinelo, M.; Costoya, N.; Núñez, M.J. Polyphenols from plant materials: Extraction and antioxidant power. Electron. J. Environ. Agric. Food Chem. 2008, 7, 3210–3216. [Google Scholar]
- Wittenauer, J.; MäcKle, S.; Sußmann, D.; Schweiggert-Weisz, U.; Carle, R. Inhibitory effects of polyphenols from grape pomace extract on collagenase and elastase activity. Fitoterapia 2015, 101, 179–187. [Google Scholar] [CrossRef] [PubMed]
| Grape Variety/Fruit Part | Pre-Treatment | Extraction Parameters (Method; Solvent; Solid to Liquid Ratio; Time; Temperature) | Extract Treatment | Spectrophotometric Methods | Chromatographic Methods | Identified Phenolic Compounds | Analysis on Extract In Vitro | Incorporation in Product | Studied Activity | Ref | |
|---|---|---|---|---|---|---|---|---|---|---|---|
| In Vitro | In Vivo | ||||||||||
| Vitis viniferaL./seeds | Drying | Solid liquid extraction EtOH: H2O (95:5 v/v) 7 days | Concentration under rotavapor (40 °C) Filtration | DPPH and FRAP assays | HPLC | Catechin, epicatechin, gallic acid, epicatechin gallate, and procyanidin dimers (B-1, B-2 and B-3) | Yes | No | Yes | Antiaging | [49] |
| Vitis viniferaL. Grechetto/skin and seeds | - | Homogenizing by UltraTurrax T25 (3 min, 30,618 g) or ultrasound bath (5 min) EtOH: H2O (60:40 v/v) or H2O (100%) 1:2 | Centrifugation (21,074× g, 15 min, 4 °C) | Proanthocyanidins, Folin-Ciocalteu and DPPH assays | Reversed phase—HPLC | Quercetin glucoside | No | Yes | No | Antioxidant | [39] |
| Freezing under liquid nitrogen Grinding | Solid liquid extraction EtOH (100%) 1:3 1 h 20 °C | Centrifugation (21,074× g, 15 min, 4 °C) Concentration under rotavapor (4 °C) Freeze drying | |||||||||
| Vitis viniferaL. Cabernet Sauvignon/seeds | Freezing under liquid nitrogen Grinding | Solid liquid extraction EtOH: H2O (60:40 v/v) or H2O (100%) 1:10 1 h 20 °C | Centrifugation (21,074× g, 15 min, 4 °C) Concentration under rotavapor (4 °C) Freeze drying | Folin-Ciocalteu assay | - | - | No | Yes | Yes | ||
| Carignano/skins | Freeze drying Grinding | Solid liquid extraction and sonication at scheduled times by high-intensity ultrasonic disintegrator (1000 s, 200 cycles, 5 on, 5 off, 15 μm of probe amplitude) EtOH: H2O (70:30 v/v) 1:32 48 h 25 °C | Centrifugation (8000 rpm, 30 min, ×2) Concentration under rotavapor Freeze drying Loading into vesicles (liposomes, montanov-glycerosomes, glycerosomes and montanov-liposomes) | Folin-Ciocalteu and DPPH assays | HPLC-ESI-TOF-MS | Both samples contained: Catechin isomers, fisetin, quercetin, myricetin, quercetin hexoside, syringetin hexoside, quercetin-3-methyletere, apigenin-6,8-di-C-arabinoside and apigenin-6,8-di-C-arabinoside Sample not pre-treated: Syringic acid and myricetin hexoside Sample pre-treated: Epicatechin gallate | Yes | No | No | Antioxidant Cellular protection | [38] |
| Suspension in distilled water (constant stirring, 24 h, 25 °C, 1:40) Filtration Freeze drying Grinding | |||||||||||
| Vitis vinifera/seeds paste | Cold pressing of seeds Defatting of the seed pastes | Ultrasound extraction EtOH and EtOH: H2O (1:1 v/v); 1:3 20 min | Evaporation to dryness | Folin-Ciocalteu and DPPH assays | HPLC-DAD and LC-HRMS | Gluconic acid, tartaric acid, citric acid, gallic acid, glucogalli, catechin, epicatechin, epicatechin gallate, dimer and trimer and tetramer proanthocyanidins, dimer and trimer proanthocyanidins gallate | Yes | No | No | Antioxidant Inhibition activity against collagenase, elastase and tyrosinase enzymes | [40] |
| Supercritical fluid extraction (300 bar) CO2 at 15 g/min with 10% EtOH and CO2 at 15 g/min with 20% EtOH w/w | - | Gluconic acid, tartaric acid, citric acid, gallic acid, glucogalli, catechin and epicatechin | |||||||||
| Grape/seeds | Grinding | Maceration extraction EtOH: H2O (95:5 v/v) 1 week | Filtration (Whatman grade 1 size) Concentration under rotavapor (40 °C) | - | HPLC | Catechin and epicatechin | Yes | Yes | No | Photoprotection | [41] |
| Vitis viniferaL. Cabernet Sauvignon/pomace | Drying Pulverizing | Percolation extraction EtOH: H2O (70:30 v/v) | Concentration (ascending film evaporator) Homogenization Freeze drying | - | - | - | No | Yes | Yes | Photoprotection | [45] |
| Vitis viniferaL. Red varieties: Tinta Roriz, Touriga Nacional, Castelão and Syrah/stems White varieties: Arinto, and Fernão Pires/stems | Grinding | Solid liquid extraction MeOH: H2O (70:30 v/v) 0.4:15 30 min Room temperature | Centrifugation (10,000 rpm, 15 min, at 4 °C) Filtration (0.45 µm PVDF filter) | ABTS, DPPH, and FRAP assays | Reversed phase- HPLC-DAD | Gallic acid, protocatechuic acid, catechin, epicatechin, trans-cinnamic acid, caftaric acid, quercetin-3-O-rutinoside, resveratrol, ε-viniferin, malvidin-3-O-galactoside, malvidin-3-O- glucoside | Yes | No | No | Antioxidant and inhibition activity against elastase and tyrosinase enzymes | [46] |
| Tempranillo/pomace | With and without microwaves irradiation (80 °C for 60 s) | Solid liquid extraction EtOH: H2O (50:50 v/v) 60 °C | Concentration under rotavapor | Folin-Ciocalteu, total monomeric anthocyanin pigment content, ORAC, HOSC and HORAC assays | HPLC-DAD-MS/MS | Gallic acid, 2-S-glutathionylcaftaric acid, procyanidin dimer and trimer, caftaric acid, catechin, epicatechin, delphinidin-3-O-glucoside, quercetin-3-O-glucoside, coutaric acid, cyanidin-3-O-glucoside, visitin A, petunidin-3-O-glucoside, myricetin-3-O-glucoside, malvidin-3-O-glucoside, peonidin-3-O-glucoside, quercetin-3-O-glucuronide, delphinidin-3-O-6″-p-acetylglucoside, 10-carboxypyranomalvidin-3-6″-p-coumaroylglucoside, syringetin-3-O-glucoside, myricetin, cyanidin-3-O-6″-p-coumaroylglucoside, petunidin-3-O-6″-p-coumaroylglucoside, quercetin, malvidin-3-O-6″-p-coumaroylglucoside, peonidin-3-O-6″-p-coumaroylglucoside, kaempferol | Yes | No | No | Inhibition activity against elastase, tyrosinase and MMP-1 enzymes Antioxidant activity | [37] |
| Grape/seeds | Crushing | Maceration extraction EtOH: H2O (95:5 v/v) 1:3 1 week | Filtration Concentration under rotavapor (40 °C) | Folin-Ciocalteu, total monomeric anthocyanin content, DPPH and FRAP assays | - | - | Yes | Yes | No | Photoprotection | [42] |
| Red grape/seeds Red grape/stalks | Freeze drying Grinding | Maceration extraction EtOH 1:5 48 h Room temperature (25 °C) | Centrifugation (8000 rpm, 30 min, ×2) Freeze drying Loading into vesicles (liposomes, hyalurosomes and transfersomes) | DPPH assay | HPLC-DAD | Seeds: catechin, epicatechin, epicatechin gallate Stalks: gallic acid, epigallocatechin gallate, quercetin, quercetin 3-glucoside and malvidin-3-glucoside | Yes | No | No | Antioxidant Cellular protection | [22] |
| Vitis vinifera/seed | Grinding | Maceration extraction MeOH: H2O (70:30) 1:5 72 h Room temperature | Filtration (16 folds of muslin cloth and Whatman grade 1 size) Concentration under rotavapor (40 °C) | - | - | - | No | No | Yes | Antiaging Skin depigmenting | [43] |
| Vitis labruscaL./pomace | Drying (60 °C, 2 h) Grinding | Ace: H2O (75:25), MeOH: H2O (75:25), Ace (100%), EtOH (100%), EtOH: H2O (75:25) and MeOH (100%) | - | - | - | - | - | - | - | - | [47] |
| Solid liquid extraction Ace: H2O (75:25) 1:5 120 min <60 °C | Filtration (pore filtre membrane 0.22 µm) Freeze drying | Folin-Ciocalteu and DPPH assays | HPLC-PAD | Ellagic acid | Yes | No | No | Antioxidant Cell protection and cytotoxicity | |||
| Mix of Vitis vinifera L. Trebbiano & Verdicchio (60:40)/pomace | Wet pomace: Grinding and addition of distilled water (1:5 g/mL) | Enzyme-assisted extraction H2O (100%) and EtOH: H2O (95:5 v/v) Overnight 24 °C | - | Folin-Ciocalteu, flavonoid and flavanol content assays | HPLC-DAD | Catechin, epicatechin, epigallocatechin gallate, epicatechin gallate, epigallocatechin, gallic acid, cis-piceid, cis- resveratroloside. | Yes | No | No | Anti-tyrosinase Anti-elastase | [44] |
| Dried pomace: Drying at 60 °C for 24 h; grinding then hydration (1:5 g/mL for 1 h) | Catechin, epicatechin, epigallocatechin gallate, epicatechin gallate, epigallocatechin, gallic acid, protocatechuic acid, chlogenic acid, cis-piceid, cis- resveratroloside, quercetin | ||||||||||
| Grape/seeds | - | Purchased extract | - | - | - | - | Yes | Yes | No | Photoprotection | [50] |
| Grape/seeds | - | - | - | DPPH assay | - | - | Yes | No | Yes | Antiaging | [51] |
| Vitis viniferaL./seeds | - | Purchased extract | - | Folin-Ciocalteu assay | - | - | No | Yes | No | Skin penetration | [52] |
| Muscat Hamburg/seeds | Grinding | Sonication MetOH: H2O: 1 M HCl (70:29.5:0.5) 1:5 15 to 20 min | Filtration (Whatman grade 1) Concentration under rotavapor Filtration | - | - | - | No | No | Yes | Skin depigmenting Moisturizing Antiaging | [53] |
| Grape Variety/Fruit Part | Studied Activity | Type of Assay | Sample | Studied Concentrations | Effective Concentration | Observed Effect | Ref |
|---|---|---|---|---|---|---|---|
| Carignano/skins | Antioxidant activity | DPPH assay | Extract in dispersion | - | 30 mg/mL | 84% reduction of DPPH radicals | [38] |
| Extract loaded vesicles (montanov-glycerosomes) | - | 30 mg/mL | 86% reduction of DPPH radicals | ||||
| Extract-loaded vesicles (glycerosomes) | - | 30 mg/mL | 88% reduction of DPPH radicals | ||||
| Extract-loaded vesicles (montanov-liposomes) | - | 30 mg/mL | 89.5% reduction of DPPH radicals | ||||
| Extract-loaded vesicles (liposomes) | - | 30 mg/mL | 90% reduction of DPPH radicals | ||||
| Cellular protection in the presence of H2O2 | Fibroblast cell culture | Extract in dispersion | 0.3, 3 and 30 and μg/mL | 0.3 μg/mL | Increased viability of fibroblasts to 70% | ||
| Extract-loaded vesicles (liposomes, montanov-liposomes, montanov-glycerosomes and glycerosomes) | 0.3 μg/mL | Increased viability of fibroblasts ≥83% | |||||
| Positive control (treated cells with H2O2) | - | - | Reduction of the viability of fibroblasts to 50% | ||||
| Vitis vinifera/seeds paste | Anti-tyrosinase activity | Enzymatic assay | Ultrasound extract in EtOH | 20, 150 and 500 μg/mL | 500 μg/mL | Up to 75% decrease of tyrosinase activity | [40] |
| Ultrasound extract in EtOH: H2O (1:1) | 20, 150 and 500 μg/mL | 500 μg/mL | Up to 72.4% decrease of tyrosinase activity | ||||
| Supercritical fluid extract (10% EtOH) | 20, 150 and 500 μg/mL | 500 μg/mL | Up to 10% decrease of tyrosinase activity | ||||
| Supercritical fluid extract (20% EtOH) | 20, 150 and 500 μg/mL | 500 μg/mL | Up to 15% decrease of tyrosinase activity | ||||
| Positive control: kojic acid | - | 7.1 μg/mL | Up to 52% decrease of tyrosinase activity | ||||
| Anti-elastase activity | Enzymatic assay | Ultrasound extract in EtOH | 30, 150 and 300 μg/mL | 300 μg/mL | Up to 91.3% decrease of elastase activity | ||
| Ultrasound extract in EtOH: H2O (1:1) | 30, 150 and 300 μg/mL | 300 μg/mL | Up to 83.2% decreasing of elastase activity | ||||
| Supercritical fluid extract (10% EtOH) | 30, 150 and 300 μg/mL | 300 μg/mL | Up to 30% decrease of elastase activity | ||||
| Supercritical fluid extract (20% EtOH) | 30, 150 and 300 μg/mL | 300 μg/mL | Up to 35% decrease of elastase activity | ||||
| Positive control: Elastatinal | - | 0.5 μg/mL | Up to 51.3% decrease of elastase activity | ||||
| Anti-collagenase activity | Enzymatic assay | Ultrasound extract in EtOH and EtOH: H2O (1:1) | 50, 200 and 600 μg/mL | 200 μg/mL | Up to 100% decrease of collagenase activity | ||
| Supercritical fluid extract (20% EtOH) | 50, 200 and 600 μg/mL | 600 μg/mL | Up to 90% decrease of collagenase activity | ||||
| Supercritical fluid extract (10% EtOH) | 50, 200 and 600 μg/mL | 600 μg/mL | Up to 80% decrease of collagenase activity | ||||
| Positive control: Phosphoramidon | - | 16 μM | Up to 46.4% decrease of collagenase activity | ||||
| Grape/seeds | Cytotoxicity | MTT assay on fibroblast cell culture | Extract | 0.1 to 31.25 μg/mL | 31.25 μg/mL | Dose-dependent and significant increased fibroblast viability by up to 132% | [41] |
| UV protection | MTT assay on irradiated fibroblasts with UVA (20 J.cm−2) | Extract | 0.1 to 25 μg/mL | 25 μg/mL | Improved cell viability by up to 68% | ||
| Vitis viniferaL. Red varieties: Tinta Roriz, Touriga Nacional, Castelão, Syrah/stems White varieties: Arinto, and Fernão Pires/stems | Anti-tyrosinase activity | Enzymatic assay | Extract | - | 1 mg/mL | Inhibition ranging from 41.47% to 53.83% | [46] |
| Anti-elastase activity | Enzymatic assay | Extract | - | 1 mg/mL | Inhibition ranging from 67.98% to 98.02% | ||
| Antioxidant activity | DPPH assay | Extract | - | Capacity ranging from 0.15 to 0.64 T/g dw | |||
| ABTS assay | Extract | - | Capacity ranging from 0.35 to 0.84 T/g dw | ||||
| FRAP assay | Extract | - | Capacity ranging from 0.35 to 1.03 mmol T/g dw | ||||
| Grape/seeds | Antioxidant activity | DPPH assay | Extract | 1 to 100 μg/mL | 33.17 µg/mL | 50% reduction of DPPH radicals | [42] |
| Vitamin E acetate | 1 to 1000 μg/mL | 12.23 µg/mL | |||||
| Trolox | 6.08 µg/mL | ||||||
| BHT | 206.81 µg/mL | ||||||
| FRAP assay | Extract | 1 to 100 μg/mL | 1 mg/mL equivalent to: 4.17 mM of vitamin C and 0.73 of Trolox | Reduction of Fe+3 | |||
| Photoprotection | UV absorption spectrum | Extract | 1%, 3%, and 5% (w/v) | 3% and 5% w/v More effective than 1% w/v of extract | UVA (320–400 nm) and blue light (400–495 nm) absorption | ||
| Tempranillo/pomace | Anti-tyrosinase activity | Enzymatic assay | Extract without irradiation | Increasing concentration | 4.03 ± 0.14 mg/mL | 50% reduction of tyrosinase activity | [37] |
| MW irradiated extract | 4.00 ± 0.14 mg/mL | ||||||
| Anti-elastase activity | Extract without irradiation | 0.87 ± 0.03 mg/mL | 50% reduction of elastase activity | ||||
| MW irradiated extract | 3.43 ± 0.11 mg/mL | ||||||
| Anti- MMP-1 activity | Extract without irradiation | 1.08 ± 0.08 mg/mL | 50% reduction of MMP-1 activity | ||||
| MW irradiated extract | 1.16 ± 0.06 mg/mL | ||||||
| Antioxidant activity | ORAC assay | Extract without irradiation | - | - | 481 ± 30 µmol TE/g Extract | ||
| MW irradiated extract | 448 ± 31 µmol TE/g Extract | ||||||
| HOSC assay | Extract without irradiation | 746 ± 49 µmol TE/g Extract | |||||
| MW irradiated extract | 441 ± 34 µmol TE/g Extract | ||||||
| HORAC assay | Extract without irradiation | 305 ± 28 µmol CAE/g Extract | |||||
| MW irradiated extract | 198 ± 19 µmol CAE/g Extract | ||||||
| Cellular protective effects against oxidative damage (TBHP) | keratinocyte cell culture | Extract without irradiation | 0.25 mg/mL to 2 mg/mL | 0.25 mg extract/mL | Cell viability increased to 60% | ||
| MW irradiated extract | Cell viability increased to 45% | ||||||
| Fibroblast cell culture | Extract without irradiation | Cell viability increased to 50% | |||||
| MW irradiated extract | Cell viability increased to 20% | ||||||
| Red grape/seeds Red grape/stalks | Antioxidant | DPPH assay | Extract in dispersion | - | 40 μg/mL | 78% reduction of DPPH radicals | [22] |
| Extract loaded vesicles (liposomes, transfersomes, hyalurosomes, hyalo-transfersomes) | - | 40 μg/mL | 88% reduction of DPPH radicals | ||||
| Protection in the presence of H2O2 | Fibroblast and keratinocyte cell culture | Extract in dispersion | - | 2 μg/mL | Viability of keratinocytes and fibroblasts increased to 88 and 92% | ||
| Extract loaded vesicles (liposomes, transfersomes, hyalurosomes, hyalo-transfersomes) | - | 2 μg/mL | Viability increased >100% in both cell lines | ||||
| Positive control (exposed to H2O2 and untreated with extract) | - | - | Viability of keratinocytes and fibroblasts reduced to ∼65 and 70% | ||||
| Vitis labruscaL./pomace | Antioxidant | DPPH assay | Extract | 0.5 to 25 µg/mL | 6.9 µg/mL | 50% reduction of DPPH radicals | [47] |
| BHT | 0.11 to 19 µg/mL | 7.6 µg/mL | 50% reduction of DPPH radicals | ||||
| Quercetin | 0.75 to 15 µg/mL | 4.5 µg/mL | 50% reduction of DPPH radicals | ||||
| Cytotoxicity | Fibroblast cell culture | Extract | 0.5 to 200 mg/mL | No statically significant difference | Maintained cell viability | ||
| Control (without extract) | - | ||||||
| Cytoprotecting in the presence of H2O2 (600 μM) | Fibroblast cell culture | Freeze dried extract | 0.73 to 3.65 mg/mL | 0.73 mg/mL | Maintained cell viability and protecting against H2O2 damage | ||
| Positive control (without extract) | - | - | Cell viability reduced to 60% | ||||
| Mix of Vitis vinifera L. Trebbiano & Verdicchio (60:40)/pomace | Anti-tyrosinase | Enzymatic assay | Ethanol extract | - | Aliquots of 0.3 mL | 79% decrease of tyrosinase activity | [44] |
| Water extract | Up to 71% decrease of tyrosinase activity | ||||||
| Anti-elastase | Enzymatic assay | Extract | 1 mg/mL | - | From 67.98% to 98.02 inhibition | ||
| Grape/seeds | Antioxidant | DPPH assay | Extract | - | 6.87 µg/mL | 50% inhibition of DPPH radicals | [50] |
| Positive control: Ascorbic acid | - | 4.40 µg/mL | 50% inhibition of DPPH radicals |
| Grape Variety/Fruit Part | Product | Product Evaluation | Effect | Ref | ||
|---|---|---|---|---|---|---|
| Stability | In Vitro | In Vivo | ||||
| Vitis viniferaL./seeds | Emulsion: Propylene, paraben, paraffin oil, Abil-EM 90, distilled water, methylparaben, olive oil, lemon oil, grape seed extract (5%) | Rheological studies Macroscopic observations | - | Skin evaluation Parameters: skin moisture content, Sebum content, elasticity, and SELs (scaliness, wrinkles, roughness) 40 females 12 weeks Cheeks | Reduce: roughness (14%), scaliness (13%), winkles (21%) and sebum content (26.13%) Enhance: elasticity (45.3%) and hydration (29.85%) | [49] |
| Emulgel: Propylene, paraben, paraffin oil, Abil-EM 90, distilled water, methylparaben, olive oil, Carbapol 940, Triethanolamine, lemon oil, grape seed extract (5%) | Reduce: roughness (55%), scaliness (26%), winkles (23.9%), and sebum (30.3%) Enhance: elasticity (50%) and hydration (32.2%) | |||||
| VitisviniferaL. Grechetto/skin and seeds | Toothpaste: 2% or 10% ethanol skins extract added into commercial toothpaste. | - | Total polyphenols content | - | No significant difference of polyphenol content between 2% and 10% samples. | [39] |
| Toothpaste: 2% or 10% ethanol seeds extract added into commercial toothpaste. | Significant increase of polyphenol content in the 10% sample | |||||
| VitisviniferaL. Cabernet Sauvignon/seeds | Toothpaste: 2.5% or 5% of ethanol extract added into commercial toothpaste. | - | Shelf life evaluation (after 2 and 4 months, at ambient temperature) by evaluating: Total polyphenols content and antioxidant activity | Acceptability of the toothpaste was evaluated by 10 consumers | Increase in total polyphenol content after 2 months, and persisted after 4 months. Better preservation action in ethanol than in water. | |
| Toothpaste: 5% or 10% of water extract added into commercial toothpaste. | Decrease in in total polyphenol content after 2 months and 4 months. Highest antioxidant activity in 10% sample. Most appreciated clinically in 5% sample. | |||||
| Grape/seeds | Cream: Mineral oil (5%), cetomacrogol 1000 (7%), cetyl alcohol (2%), octyl methoxycinnamate OMC (7%), grape seeds extract (3%), xanthan (1%), glycerine (5%), phenoxyethanol (0.5%), purified water q.s. to 100% * | - | Photoprotection efficacy: measurement of UV emissions before and after exposure to UV light using PMMA plates and Transpore tapes | - | SPF 9.92 on PMMA plates after UV exposure SPF 13.64 on Transpore tapes after UV exposure | [41] |
| VitisviniferaL. Cabernet Sauvignon/pomace | Cream: Ammonium acryloyldimethyltaurate/VP copolymer, trilaureth-4 phosphate, rapeseed oil sorbitol esters, mineral oil and isopropyl palmitate (4%), ammonium acryloyldimethyltaurate vinylpyrrolidone (0.5%), propylene glycol (5%), ethyl alcohol (2.5%), disodium EDTA (0.1%), grape pomace extract (10%), water purified q.s. to 100%, butylmethoxydibenzoyl methane (2.5%), ethylhexyl methoxycinnamate (5%), ethylhexyl dimethyl PABA (4%), butylated hydroxy toluene (0.1%), mixture of phenoxyethanol and parabens others (0.75%) * | Evaluation of organoleptic properties by aspect, color, and odor and the pH value. | Design of Experiment DPPH assay SPF measurement using PMMA plates | Primary and cumulative cutaneous irritability and sensitization tests 60 males and females 6 weeks Application on the back skin | Antioxidant activity (40.10%) Absence of irritation and dermal sensitization In vitro SPF: 16.33 In vivo SPF: 12.30 | [45] |
| Phototoxicity and Photosensitization test 30 males and females 5 weeks Applications on the back skin | ||||||
| Grape/seeds | Cream: Mineral oil (16.07%), span 80 (3.93%), Tween 80 (3.07), triethanolamine (0.4%), Carbopol 940 (0.5%), glycerin (5%), propylparaben (1%), q.s. water to 100%, grape seeds extract (3%) * | Evaluation for texture profiles, viscosity and pH: at 25 °C & 4 °C before and after heat cool cycling (6 cycles) for 12 h | SPF and PA measurement | - | UV protection booster: SPF 1.29 PA 1.19 | [42] |
| Grape/seeds | Lotion: Anisotriazine (8%), titanium dioxide (12%), silicone DC 5562 (5%), sorbitan monostearate (1%), cremophore A6 (2%), cremophore A25 (1.5%), cetyl alcohol (0.8%), beeswax (0.8%), isopropyl myristate (2.4%), disodium EDTA (0.2%), glycerin (1%), propylene glycol (2%), methyl paraben (0.1%), propyl paraben (0.02%), polysorbate 60 (1%), q.s. to water 100%, grape seeds extract (1%) * | Evaluation for the physical characteristic of pH and viscosity after freeze-thaw condition (4 °C, 45 °C, 24 h) for each condition (6 cycles) | SPF measurements DPPH assay | - | SPF booster up to 53.58 Antioxidant activity (84.04%) | [50] |
| Grape/seeds | Cream: ABIL-EM 90 (cetyl dimethicone copolyol), paraffin oil, distilled water, grape seeds extract (3%),methyl paraben, glycerin, lemon oil, triethanolamine solution | Evaluation at 8 °C, 25 °C, 40 °C for 12 weeks for stability, color and pH | - | Study of the moisture content, pH value, sebum content, elasticity and average pore size of skin Questionnaire subjective evaluation 20 females 12 weeks Application on cheeks | Stable product Antiaging effects: Reduction of size pores (56.8%) Reduction in roughness (18.98%) Increase in skin elasticity (47.95%) Increase in sebum content (93.85%) Increase in hydration (47.56%). | [51] |
| Vitisvinifera/seed | Cream: Paraffin oil (16%), abil-EM 90 (4%), distilled water q.s. to 100%, grape seeds extract (4%), rose oil (2 to 3 drops) | Evaluation at 8 °C, 25 °C,40 °C for 12 weeks for pH, centrifugation, electrical conductivity, phase separation, organoleptic and physical characteristics | - | Study of skin microrelief parameters, elasticity, moisture contents and melanin 11 males 12 weeks Application on cheeks | Stable product Antiaging effects Skin depigmenting effects | [43] |
| Vitis viniferaL./seeds | Liquid gel-based serum: Grape seeds extract phytosome (10%), carbopol ultrez 30 (0.5%), triethanolamine (TEA) (0.4%), propylene glycol (10%), methylparabene (0.18), propylparaben (0.02%), sodium metabisulfite (0.075%), demineralized water q.s. to 100% * | Physicochemical evaluations: Organoleptic test (color, odor, syneresis), homogeneity, pH, viscosity and rheology | Penetration test (18 h) | - | Increasing the penetration of the phytosome into membrane | [52] |
| Muscat Hamburg/seeds | Cream: Liquid paraffin (16%), abil EM 90 (4%), grape seed extract (2%), distilled water (98%) | - | - | Patch test Study the effects of the formulations (containing or not the extract) on melanin content, erythema, moisture, elasticity and sebum 110 Males 8 weeks Application on cheeks | Absence of skin sensitive cases Safe application Decrease of skin melanin content (~18%) Increase in skin elasticity (~13%) Decrease in skin sebum content (15%) Increase skin moisture (data not indicated) | [53] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hoss, I.; Rajha, H.N.; El Khoury, R.; Youssef, S.; Manca, M.L.; Manconi, M.; Louka, N.; Maroun, R.G. Valorization of Wine-Making By-Products’ Extracts in Cosmetics. Cosmetics 2021, 8, 109. https://doi.org/10.3390/cosmetics8040109
Hoss I, Rajha HN, El Khoury R, Youssef S, Manca ML, Manconi M, Louka N, Maroun RG. Valorization of Wine-Making By-Products’ Extracts in Cosmetics. Cosmetics. 2021; 8(4):109. https://doi.org/10.3390/cosmetics8040109
Chicago/Turabian StyleHoss, Israa, Hiba N. Rajha, Rindala El Khoury, Sahar Youssef, Maria Letizia Manca, Maria Manconi, Nicolas Louka, and Richard G. Maroun. 2021. "Valorization of Wine-Making By-Products’ Extracts in Cosmetics" Cosmetics 8, no. 4: 109. https://doi.org/10.3390/cosmetics8040109
APA StyleHoss, I., Rajha, H. N., El Khoury, R., Youssef, S., Manca, M. L., Manconi, M., Louka, N., & Maroun, R. G. (2021). Valorization of Wine-Making By-Products’ Extracts in Cosmetics. Cosmetics, 8(4), 109. https://doi.org/10.3390/cosmetics8040109