Melanogenesis and Melasma Treatment
Abstract
:1. Introduction
2. Main Signalling Pathways in Melanogenesis
3. Key Pathways in Melasma
3.1. Ultraviolet Light
3.2. Genetics
3.3. Sex Hormones
3.4. Wnt Signalling Pathway
4. Treatments and Future Perspectives
4.1. Phenolic and Non-Phenolic Compounds
4.2. Other Therapies
4.3. Future Treatments
5. Conclusions and Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Nordlund, J.J. The melanocyte and the epidermal melanin unit: An expanded concept. Dermatol. Clin. 2007, 25, 271–281. [Google Scholar] [CrossRef]
- Wu, X.; Hammer, J.A. Melanosome transfer: It is best to give and receive. Curr. Opin. Cell. Biol. 2014, 29, 1–7. [Google Scholar] [CrossRef] [Green Version]
- Costin, G.E.; Hearing, V.J. Human skin pigmentation: Melanocytes modulate skin color in response to stress. FASEB J. 2007, 21, 976–994. [Google Scholar] [CrossRef] [PubMed]
- Videira, I.F.; Moura, D.F.; Magina, S. Mechanisms regulating melanogenesis. An. Bras. Dermatol. 2013, 88, 76–83. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ebanks, J.P.; Wickett, R.R.; Boissy, R.E. Mechanisms Regulating Skin Pigmentation: The Rise and Fall of Complexion Coloration. Int. J. Mol. Sci. 2009, 10, 4066–4087. [Google Scholar] [CrossRef] [Green Version]
- Halaban, R.; Pomerantz, S.H.; Marshall, S.; Lambert, D.T.; Lerner, A.B. Regulation of Tyrosinase in Human Melanocytes Grown in Culture. J. Cell. Biol. 1983, 97, 480–488. [Google Scholar] [CrossRef]
- Kawaguchi, M.; Suzuki, T. Melanogenesis and New Signaling Regulators for the Treatment of Melasma. In Melasma and Vitiligo in Brown Skin; Handog, E., Enriquez-Macarayo, M., Eds.; Springer: New Delhi, India, 2017; pp. 92–95. [Google Scholar]
- Li, P.; Liu, L.; Chang, C.; Gao, R.; Leung, C. Silencing Stem Cell Factor Gene in Fibroblasts to Regulate Paracrine Factor Productions and Enhance c-Kit Expression in Melanocytes on Melanogenesis. Int. J. Mol. Sci. 2018, 19, 1475. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bae-Harboe, Y.C.; Park, H. Tyrosinase: A Central Regulatory Protein for Cutaneous Pigmentation. J. Investig. Dermatol. 2012, 132, 2678–2680. [Google Scholar] [CrossRef] [Green Version]
- Park, H.; Wu, C.; Yonemoto, L.; Murphy-Smith, M.; Wu, H.; Stachur, C.M.; Gilchrest, B.A. MITF mediates cAMP-induced protein kinase C- β expression in human melanocytes. Biochem. J. 2006, 578, 571–578. [Google Scholar] [CrossRef] [Green Version]
- Imokawa, G.; Yada, Y.; Kimura, M. Signalling mechanisms of endothelin-induced mitogenesis and melanogenesis in human melanocytes. Biochem. J. 1996, 312, 305–312. [Google Scholar] [CrossRef] [Green Version]
- Regazzetti, C.; De Donatis, G.M.; Ghorbel, H.H.; Cardot-leccia, N.; Ambrosetti, D.; Bahadoran, P.; Chignon-Sicard, B.; Lacour, J.; Ballotti, R.; Mahns, A.; et al. Endothelial Cells Promote Pigmentation through Endothelin Receptor B Activation. J. Investig. Dermatol. 2015, 135, 3096–3104. [Google Scholar] [CrossRef] [Green Version]
- Lee, A.Y.; Noh, M. The regulation of epidermal melanogenesis via cAMP and/or PKC signaling pathways: Insights for the development of hypopigmenting agents. Arch. Pharm. Res. 2013, 36, 792–801. [Google Scholar] [CrossRef] [PubMed]
- Reynolds, J.L.; Akhter, J.; Morris, D.L. In vitro effect of histamine and histamine H1 and H2 receptor antagonists on cellular proliferation of human malignant melanoma cell lines. Melanoma Res. 1996, 6, 95–99. [Google Scholar] [CrossRef] [PubMed]
- Kim, N.H.; Lee, A.Y. Histamine effect on melanocyte proliferation and vitiliginous keratinocyte survival. Exp. Dermatol. 2010, 19, 1073–1079. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.J.; Park, M.K.; Lee, E.J.; Kim, Y.L.; Kim, H.J.; Kang, J.H.; Kim, H.M.; Lee, A.Y.; Lee, C.H. Histamine receptor 2-mediated growth-differentiation factor-15 expression is involved in histamine-induced melanogenesis. Int. J. Biochem. Cell Biol. 2012, 44, 2124–2128. [Google Scholar] [CrossRef]
- Fu, C.; Chen, J.; Lu, J.; Yi, L.U.; Tong, X. Roles of inflammation factors in melanogenesis (Review). Mol. Med. Rep. 2020, 21, 1421–1430. [Google Scholar] [CrossRef] [Green Version]
- Montaudié, H.; Bertolotto, C.; Ballotti, R.; Passeron, T. Fisiología del sistema pigmentario. Melanogénesis. EMC Dermatol. 2014, 48, 1–11. [Google Scholar] [CrossRef]
- Lee, A.Y. An updated review of melasma pathogenesis. Dermatol. Sin. 2014, 32, 233–239. [Google Scholar] [CrossRef]
- Rajanala, S.; Maymone, M.B.C.; Vashi, N.A. Melasma pathogenesis: A review of the latest research, pathological findings, and investigational therapies. Dermatol. Online J. 2019, 25. [Google Scholar] [CrossRef]
- Zhou, L.L.; Baibergenova, A. Melasma: Systematic review of the systemic treatments. Int. J. Dermatol. 2017, 56, 902–908. [Google Scholar] [CrossRef] [PubMed]
- Khlgatian, M.K.; Hadshiew, I.M.; Asawanonda, P.; Yaar, M.; Eller, M.S.; Fujita, M.; Norris, D.A.; Gilchrest, B.A. Tyrosinase gene expression is regulated by p53. J. Investig. Dermatol. 2002, 118, 126–132. [Google Scholar] [CrossRef] [Green Version]
- Galibert, M.D.; Carreira, S.; Goding, C.R. The Usf-1 transcription factor is a novel target for the stress-responsive p38 kinase and mediates UV-induced Tyrosinase expression. EMBO J. 2001, 20, 5022–5031. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hunt, G.; Kyne, S.; Wakamatsu, K.; Ito, S.; Thody, A.J. Nle4DPhe7α-melanocyte-stimulating hormone increases the eumelanin:phaeomelanin ratio in cultured human melanocytes. J. Investig. Dermatol. 1995, 104, 83–85. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Im, S.; Moro, O.; Peng, F.; Medrano, E.E.; Cornelius, J.; Babcock, G.; Nordlund, J.J.; Abdel-Malek, Z.A. Activation of the cyclic AMP pathway by α-melanotropin mediates the response of human melanocytes to ultraviolet B radiation. Cancer Res. 1998, 58, 47–54. [Google Scholar]
- Ryter, S.W.; Hong, P.K.; Hoetzel, A.; Park, J.W.; Nakahira, K.; Wang, X.; Choi, A.M.K. Mechanisms of cell death in oxidative stress. Antiox. Redox Signal. 2007, 9, 49–89. [Google Scholar] [CrossRef]
- Premi, S.; Wallisch, S.; Mano, C.M.; Weiner, A.B.; Bacchiocchi, A.; Wakamatsu, K.; Bechara, E.J.; Halaban, R.; Douki, T.; Brash, D.E. Photochemistry. Chemiexcitation of melanin derivatives induces DNA photoproducts long after UV exposure. Science 2015, 347, 842–847. [Google Scholar] [CrossRef]
- Miyamura, Y.; Coelho, S.G.; Schlenz, K.; Batzer, J.; Smuda, C.; Choi, W.; Brenner, M.; Passeron, T.; Zhang, G.; Kolbe, L.; et al. The deceptive nature of UVA tanning versus the modest protective effects of UVB tanning on human skin. Pigment. Cell. Melanoma Res. 2011, 24, 136–147. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Maddodi, N.; Jayanthy, A.; Setaluri, V. Shining light on skin pigmentation: The darker and the brighter side of effects of UV radiation. Photochem. Photobiol. 2012, 88, 1075–1082. [Google Scholar] [CrossRef] [Green Version]
- Holmo, N.F.; Ramos, G.B.; Salomão, H.; Werneck, R.I.; Mira, M.T.; Miot, L.D.B.; Miot, H.A. Complex segregation analysis of facial melasma in Brazil: Evidence for a genetic susceptibility with a dominant pattern of segregation. Arch. Dermatol. Res. 2018, 310, 827–831. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Passeron, T.; Picardo, M. Melasma, a photoaging disorder. Pigment. Cell. Melanoma Res. 2018, 31, 461–465. [Google Scholar] [CrossRef] [Green Version]
- Kang, H.Y.; Suzuki, I.; Lee, D.J.; Ha, J.; Reiniche, P.; Aubert, J.; Deret, S.; Zugaj, D.; Voegel, J.J.; Ortonne, J.P. Transcriptional profiling shows altered expression of wnt pathway- and lipid metabolism-related genes as well as melanogenesis-related genes in melasma. J. Investig. Dermatol. 2011, 131, 1692–1700. [Google Scholar] [CrossRef] [Green Version]
- Lieberman, R.; Moy, L. Estrogen receptor expression in melasma: Results from facial skin of affected patients. J. Drugs Dermatol. 2008, 7, 463–465. [Google Scholar] [PubMed]
- Tamega Ade, A.; Miot, H.A.; Moço, N.P.; Silva, M.G.; Marques, M.E.; Miot, L.D. Gene and protein expression of oestrogen-β and progesterone receptors in facial melasma and adjacent healthy skin in women. Int. J. Cosmet. Sci. 2015, 37, 222–228. [Google Scholar] [CrossRef] [PubMed]
- Cario, M. How hormones may modulate human skin pigmentation in melasma: An in vitro perspective. Exp. Dermatol. 2019, 28, 709–718. [Google Scholar] [CrossRef] [Green Version]
- Kippenberger, S.; Loitsch, S.; Solano, F.; Bernd, A.; Kaufmann, R. Quantification of tyrosinase, TRP-1, and TRP-2 transcripts in human melanocytes by reverse transcriptase-competitive multiplex PCR—Regulation by steroid hormones. J. Investig. Dermatol. 1998, 110, 364–367. [Google Scholar]
- Cohen, P.R. Melasma treatment: A novel approach using a topical agent that contains an anti-estrogen and a vascular endothelial growth factor inhibitor. Med. Hypotheses 2017, 101, 1–5. [Google Scholar] [CrossRef]
- Kim, N.H.; Cheong, K.A.; Lee, T.R.; Lee, A.Y. PDZK1 upregulation in estrogen-related hyperpigmentation in Melasma. J. Investig. Dermatol. 2012, 132, 2622–2631. [Google Scholar] [CrossRef] [Green Version]
- Handa, S.; De, D.; Khullar, G.; Radotra, B.D.; Sachdeva, N. The clinicoaetiological, hormonal and histopathological characteristics of melasma in men. Clin. Exp. Dermatol. 2018, 43, 36–41. [Google Scholar] [CrossRef]
- Famenini, S.; Gharavi, N.M.; Beynet, D.P. Finasteride associated melasma in a Caucasian male. J. Drugs Dermatol. 2014, 13, 484–486. [Google Scholar]
- Kim, J.Y.; Lee, T.R.; Lee, A.Y. Reduced WIF-1 expression stimulates skin hyperpigmentation in patients with melasma. J. Investig. Derm. 2013, 133, 191–200. [Google Scholar] [CrossRef] [Green Version]
- Yamaguchi, Y.; Passeron, T.; Watabe, H.; Yasumoto, K.I.; Rouzaud, F.; Hoashi, T.; Hearing, V.J. The effects of dickkopf 1 on gene expression and wnt signaling by melanocytes: Mechanisms underlying its suppression of melanocyte function and proliferation. J. Investig. Dermatol. 2007, 127, 1217–1225. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yamaguchi, Y.; Passeron, T.; Hoashi, T.; Watabe, H.; Rouzaud, F.; Yasumoto, K.; Hara, T.; Tohyama, C.; Katayama, I.; Miki, T.; et al. Dickkopf 1 (DKK1) regulates skin pigmentation and thickness by affecting Wnt/ β-catenin signaling in keratinocytes. FASEB J. 2008, 22, 1009–1020. [Google Scholar] [CrossRef]
- Bandyopadhyay, D. Topical treatment of melasma. Indian J. Dermatol. 2009, 54, 303–309. [Google Scholar] [CrossRef] [PubMed]
- Pillaiyar, T.; Manickam, M.; Namasivayam, V. Skin whitening agents: Medicinal chemistry perspective of tyrosinase inhibitors. J. Enzym. Inhib. Med. Chem. 2017, 32, 403–425. [Google Scholar] [CrossRef] [Green Version]
- Baranska, A.; Shawket, A.; Jouve, M.; Baratin, M.; Malosse, C.; Voluzan, O.; Manh, T.P.V.; Fiore, F.; Bajénoff, M.; Benaroch, P.; et al. Unveiling skin macrophage dynamics explains both tattoo persistence and strenuous removal. J. Exp. Med. 2018, 215, 1115–1133. [Google Scholar] [CrossRef] [Green Version]
- Jimbow, K.; Obata, H.; Pathak, M.A.; Fitzpatrick, T.B. Mechanism of depigmentation by hydroquinone. J. Investig. Dermatol. 1974, 62, 436–449. [Google Scholar] [CrossRef] [Green Version]
- Mazurek, K.; Pierzchała, E. Comparison of efficacy of products containing azelaic acid in melasma treatment. J Cosmet. Dermatol. 2016, 15, 269–282. [Google Scholar] [CrossRef]
- Ertam, I.; Mutlu, B.; Unal, I.; Alper, S.; Kivçak, B.; Ozer, O. Efficiency of ellagic acid and arbutin in melasma: A randomized, prospective, open-label study. J. Dermatol. 2008, 35, 570–574. [Google Scholar] [CrossRef] [PubMed]
- Han, T.Y.; Son, I.P.; Jang, W.S.; Chang, H.S.; Kim, J.H.; Sim, Y.H.; Kim, D.H.; Kim, B.J.; Kim, M.N.; Lee, H.K.; et al. Efficacy of hydrogel mask with 2% arbutin for melasma. Korean J. Dermatol. 2011, 49, 210–216. [Google Scholar]
- Maeda, K.; Fukuda, M. Arbutin: Mechanism of its depigmenting action in human melanocyte culture. J. Pharmacol. Exp. Ther. 1996, 276, 765–769. [Google Scholar] [PubMed]
- Grimes, P.E. Melasma: Etiologic and Therapeutic Considerations. Arch. Dermatol. 1995, 131, 1453–1457. [Google Scholar] [CrossRef] [PubMed]
- Parvez, S.; Kang, M.; Chung, H.S.; Cho, C.; Hong, M.C.; Shin, M.K.; Bae, H. Survey and mechanism of skin depigmenting and lightening agents. Phytother. Res. 2006, 20, 921–934. [Google Scholar] [CrossRef]
- Picardo, M.; Carrera, M. New and Experimental Treatments of Cloasma and Other Hypermelanoses. Dermatol. Clin. 2007, 25, 353–362. [Google Scholar] [CrossRef]
- Deo, K.S.; Dash, K.N.; Sharma, Y.K.; Virmani, N.C.; Oberai, C. Kojic acid vis-a-vis its combinations with hydroquinone and betamethasone valerate in melasma: A randomized, single blind, comparative study of efficacy and safety. Indian J. Dermatol. 2013, 58, 281–285. [Google Scholar] [CrossRef] [PubMed]
- Küçük, Ö.S. Current treatment approaches for melasma. Bezm. Sci. 2018, 6, 54–62. [Google Scholar] [CrossRef]
- Monteiro, R.C.; Kishore, N.; Bhat, R.M.; Sukumar, D.; Martis, J.; Ganesh, K. A comparative study of the efficacy of 4% hydroquinone vs 0.75% Kojic acid cream in the treatment of facial melasma. Indian J. Dermatol. 2013, 58, 157. [Google Scholar] [CrossRef] [PubMed]
- Berardesca, E.; Rigoni, C.; Cantù, A.; Cameli, N.; Tedeschi, A.; Donne Dermatologhe Italia; Laureti, T. Effectiveness of a new cosmetic treatment for melasma. J. Cosmet. Dermatol. 2020, 19, 1684–1690. [Google Scholar] [CrossRef] [PubMed]
- Schallreuter, K.; Wood, J. A possible mechanism of action for azelaic acid in the human epidermis. Arch. Derm. Res. 1990, 282, 168–171. [Google Scholar] [CrossRef] [PubMed]
- Miao, F.; Su, M.Y.; Jiang, S.; Luo, L.F.; Shi, Y.; Lei, T.C. Intramelanocytic Acidification Plays a Role in the Antimelanogenic and Antioxidative Properties of Vitamin C and Its Derivatives. Oxid. Med. Cell Longev. 2019, 2019, 2084805. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Huh, C.H.; Seo, K.I.; Park, J.Y.; Lim, J.G.; Eun, H.C.; Park, K.C. A randomized, double-blind, placebo-controlled trial of vitamin C iontophoresis in melasma. Dermatology 2003, 206, 316–320. [Google Scholar] [CrossRef]
- Jiménez-Cervantes, C.; Solano, F.; Kobayashi, T.; Urabe, K.; Hearing, V.; Lozano, J. A new enzymatic function in the melanogenic pathway. The 5,6-dihydroxyindole-2-carboxylic acid oxidase activity of tyrosinase-related protein-1 (TRP1). J. Biol. Chem. 1994, 269, 17993–18000. [Google Scholar] [CrossRef]
- Espinal-Perez, L.E.; Moncada, B.; Castanedo-Cazares, J.P. A double-blind randomized trial of 5% ascorbic acid vs. 4% hydroquinone in melasma. Int. J. Dermatol. 2004, 43, 604–607. [Google Scholar] [CrossRef] [PubMed]
- Fu, B.; Li, H.; Wang, X.; Lee, F.S.C.; Cui, S. Isolation and identification of flavonoids in licorice and a study of their inhibitory effects on tyrosinase. J. Agric. Food Chem. 2005, 53, 7408–7414. [Google Scholar] [CrossRef]
- Choi, Y.K.; Rho, Y.K.; Yoo, K.H.; Lim, Y.Y.; Li, K.; Kim, B.J.; Seo, S.J.; Kim, M.N.; Hong, C.K.; Kim, D.S. Effects of vitamin C vs. multivitamin on melanogenesis: Comparative study in vitro and in vivo. Pharmacol. Ther. 2019, 49, 218–226. [Google Scholar] [CrossRef]
- Amer, M.; Metwalli, M. Topical liquiritin improves melasma. Int. J. Dermatol. 2000, 39, 299–301. [Google Scholar] [CrossRef] [PubMed]
- Toossi, P.; Esmaili-Azad, M.; Saeedi, M. Evaluation of Licorice efficacy on melasma. Iran. J. Dermatol. 2013, 16, 118–119. [Google Scholar]
- Goenka, S.; Nagabhushanam, K.; Majeed, M.; Simon, S.R. Calebin-A, a Curcuminoid Analog Inhibits α-MSH-Induced Melanogenesis in B16F10 Mouse Melanoma Cells. Cosmetics 2019, 6, 51. [Google Scholar] [CrossRef] [Green Version]
- Romero, C.; Aberdam, E.; Larnier, C.; Ortonne, J.P. Retinoic acid as modulator of UVB-induced melanocyte differentiation. Involvement of the melanogenic enzymes expression. J. Cell. Sci. 1994, 107, 1095–1103. [Google Scholar] [CrossRef]
- Wang, J.V.; Schoenberg, E.; Saedi, N. Bakuchiol as a Trendy Ingredient in Skincare: Recent Evidence. Skinmed 2019, 17, 188–189. [Google Scholar] [PubMed]
- Banavase Channakeshavaiah, R.; Andanooru Chandrappa, N.K. Topical metformin in the treatment of melasma: A preliminary clinical trial. J. Cosmet. Derm. 2020, 19, 1161–1164. [Google Scholar] [CrossRef]
- Pekmezci, E. A novel triple combination in treatment of melasma: Significant outcome with far less actives. J. Cosmet. Derm. 2019, 18, 1700–1704. [Google Scholar] [CrossRef] [PubMed]
- Sarkar, R.; Garg, V.; Bansal, S.; Sethi, S.; Gupta, C. Comparative evaluation of efficacy and tolerability of glycolic acid, salicylic Mandelic acid, and Phytic acid combination peels in Melasma. Dermatol. Surg. 2016, 42, 384–391. [Google Scholar] [CrossRef] [PubMed]
- Sadick, N.; Edison, B.L.; John, G.; Bohnert, K.L.; Green, B. An Advanced, Physician-Strength Retinol Peel Improves Signs of Aging and Acne Across a Range of Skin Types Including Melasma and Skin of Color. J. Drugs Dermatol. 2019, 18, 918–923. [Google Scholar] [PubMed]
- McKesey, J.; Tovar-Garza, A.; Pandya, A.G. Melasma Treatment: An Evidence-Based Review. Am. J. Clin. Dermatol. 2020, 21, 173–225. [Google Scholar] [CrossRef] [PubMed]
- Shah, S.D.; Aurangabadkar, S.J. Laser Toning in Melasma. J. Cutan. Aesthet. Surg. 2019, 12, 76–84. [Google Scholar] [CrossRef]
- Del Rosario, E.; Florez-Pollack, S.; Zapata, L., Jr.; Hernandez, K.; Tovar-Garza, A.; Rodrigues, M.; Hynan, L.S.; Pandya, A.G. Randomized, placebo-controlled, double-blind study of oral tranexamic acid in the treatment of moderate-to-severe melasma. J. Am. Acad. Dermatol. 2018, 78, 363–369. [Google Scholar] [CrossRef]
- Cho, H.H.; Choi, M.; Cho, S.; Lee, J.H. Role of oral tranexamic acid in melasma patients treated with IPL and low fluence QS Nd:YAG laser. J. Dermatol. Treat. 2013, 24, 292–296. [Google Scholar] [CrossRef]
- Beyzaee, A.M.; Patil, A.; Goldust, M.; Moslemi, M.; Kazeminejad, A.; Rokni, G.R. Comparative Efficacy of Fractional CO2 Laser and Q-Switched Nd:YAG Laser in Combination Therapy with Tranexamic Acid in Refractory Melasma: Results of a Prospective Clinical Trial. Cosmetics 2021, 8, 37. [Google Scholar] [CrossRef]
- Sarkar, R.; Bansal, A.; Ailawadi, P. Future therapies in melasma: What lies ahead? Indian J. Dermatol. Venereol. Leprol. 2020, 86, 8–17. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.; Huh, Y.; Lim, K.M. Anti-Pigmentary Natural Compounds and Their Mode of Action. Int. J. Mol. Sci. 2021, 22, 6206. [Google Scholar] [CrossRef]
- Boo, Y.C. Human skin lightening efficacy of resveratrol and its analogs: From in vitro studies to cosmetic applications. Antioxidants 2019, 8, 332. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Boo, Y.C. p-coumaric acid as an active ingredient in cosmetics: A review focusing on its antimelanogenic effects. Antioxidants 2019, 8, 275. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Moon, H.R.; Jo, S.Y.; Kim, H.T.; Lee, W.J.; Won, C.H.; Lee, M.W.; Choi, J.H.; Chang, S.E. Loratadine, an H 1 antihistamine, inhibits melanogenesis in human melanocytes. BioMed Res. Int. 2019, 2019, 5971546. [Google Scholar] [CrossRef] [PubMed] [Green Version]
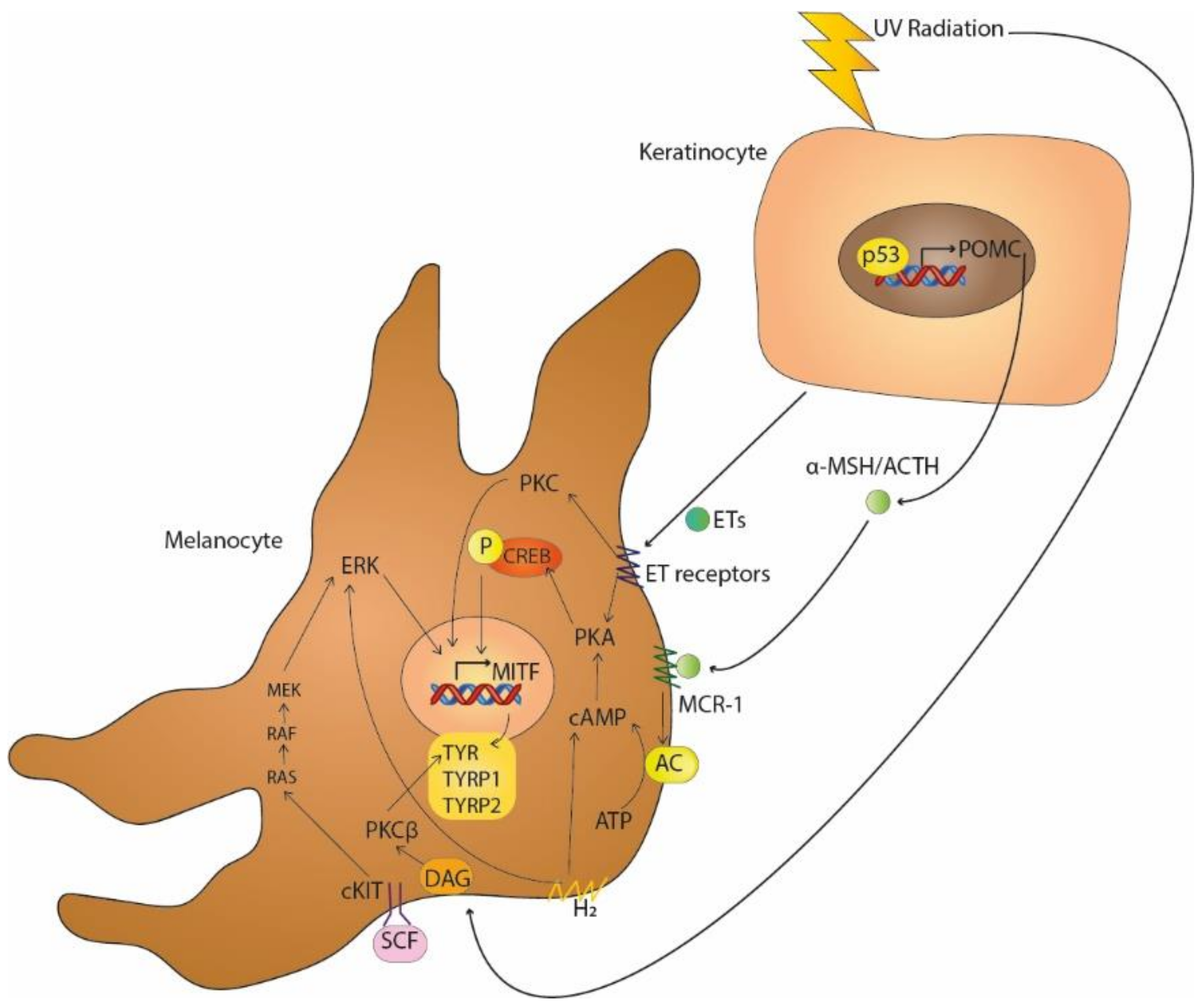
| Key Signalling Pathways in Melanogenesis | Activators | References |
|---|---|---|
| MC1-R | α-MSH, ACTH | [4,5,6] |
| SCF-c-KIT receptor tyrosine kinase | SCF | [3,7,8] |
| Protein kinase C-dependent signalling | DAG | [9,10] |
| Endothelin pathway | ETs | [11,12,13] |
| H1 and H2 receptors | H1, H2 | [14,15,16] |
| Keratinocyte-derived paracrine factors | IL-18, IL-33, GM-CSF, PGE2, PGF2α | [17] |
| Fibroblast-derived paracrine factors | IL-33, PGE2, PGF2α | [17] |
| Current Treatments | Targets | References | |
|---|---|---|---|
| Phenolic compounds | Hydroquinone | Tyrosinase inhibitor | [44,45,46,47,48] |
| Glabridin | [17] | ||
| Non-phenolic compounds | Arbutin | Tyrosinase inhibitor | [49,50,51] |
| Deoxyarbutin | [52,53,54] | ||
| Kojic acid | [55,56,57,58] | ||
| Azelaic acid | [48,59] | ||
| Vitamin C | Tyrosinase inhibitor | [60,61,62,63,64,65] | |
| Liquorice compounds | Melanin dispersion and epidermal melanin removal | [64,66,67] | |
| Curcumoid | αMSH-stimulated melanogenesis suppression and inhibition of melanosomes maturation | [68] | |
| Retinoids | Multiple targets (e.g., reduction in TYR transcription) | [69,70] | |
| Triple combination cream | Multiple targets | [71,72] | |
| Chemical peelings | Melanin removal | [73] | |
| Laser and light therapies | Melanin removal | [54,74,75,76] | |
| Oral treatments | Unknown | [77,78,79] | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Maddaleno, A.S.; Camargo, J.; Mitjans, M.; Vinardell, M.P. Melanogenesis and Melasma Treatment. Cosmetics 2021, 8, 82. https://doi.org/10.3390/cosmetics8030082
Maddaleno AS, Camargo J, Mitjans M, Vinardell MP. Melanogenesis and Melasma Treatment. Cosmetics. 2021; 8(3):82. https://doi.org/10.3390/cosmetics8030082
Chicago/Turabian StyleMaddaleno, Adriana Solange, Judit Camargo, Montserrat Mitjans, and Maria Pilar Vinardell. 2021. "Melanogenesis and Melasma Treatment" Cosmetics 8, no. 3: 82. https://doi.org/10.3390/cosmetics8030082
APA StyleMaddaleno, A. S., Camargo, J., Mitjans, M., & Vinardell, M. P. (2021). Melanogenesis and Melasma Treatment. Cosmetics, 8(3), 82. https://doi.org/10.3390/cosmetics8030082







