Treatment of Head Louse Infestation with a Novel Mixture Made of Semi-Crystalline Polymers and Plant Extracts: Blind, Randomized, Controlled, Superiority Trial
Abstract
1. Introduction
2. Materials and Methods
2.1. Treatments Used in This Study
2.2. Ethics
2.3. Design
2.4. Statistical Analysis
- Processing: A: 0, B: 1
- Gender: F: 0, M: 1
- Hair length: short: 0, shoulders: 1, below shoulders: 2
- Hair type: frizzy: 0, curly: 1, straight: 2, flexible: 3
- Scalp at inclusion: normal: 1
- Antibiotic treatment before: no: 0
- Infestation: low: 1, moderate: 2, high: 3
3. Results
3.1. Cure rate of the Two Treatments for the In Vivo Part of the Study
3.1.1. Results for the Criterion “Absence of Live Lice on D8”
3.1.2. Results for the Criterion “Absence of Living Immature on D8”
3.1.3. Results for the Criteria “Absence of Live Lice and Live Immature on D2” and “Absence of Live Lice and Immature Lice on D1”
3.1.4. Criterion “Absence of Live Lice on D1 and D2 and Absence of Live Immatures on D8”
3.1.5. Additional Analyses
3.2. Ovicidal Efficacy of the Two Treatments, in Vitro Part of the Study
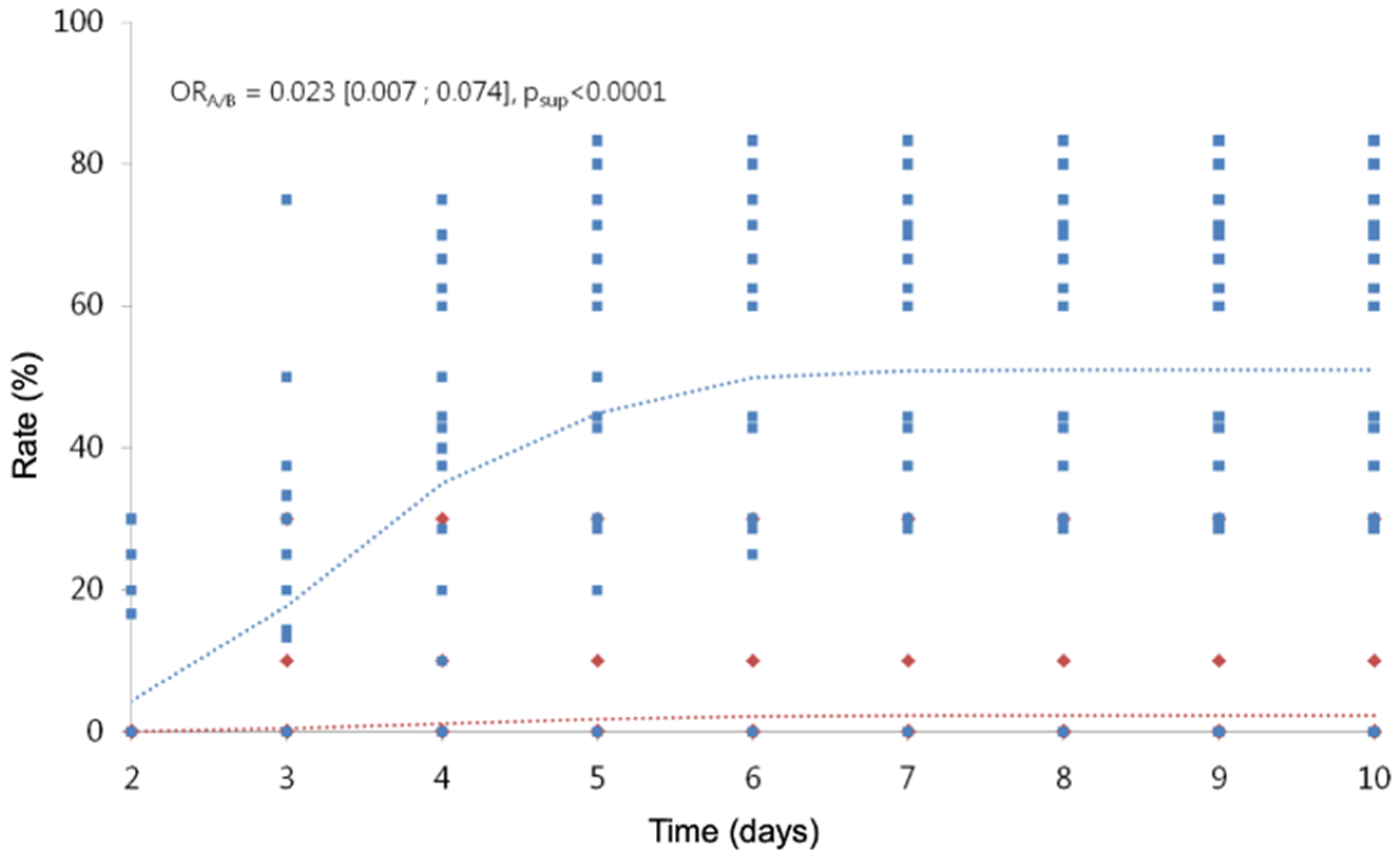
3.2.1. Comparison of Cumulative Hatching Rates on D10
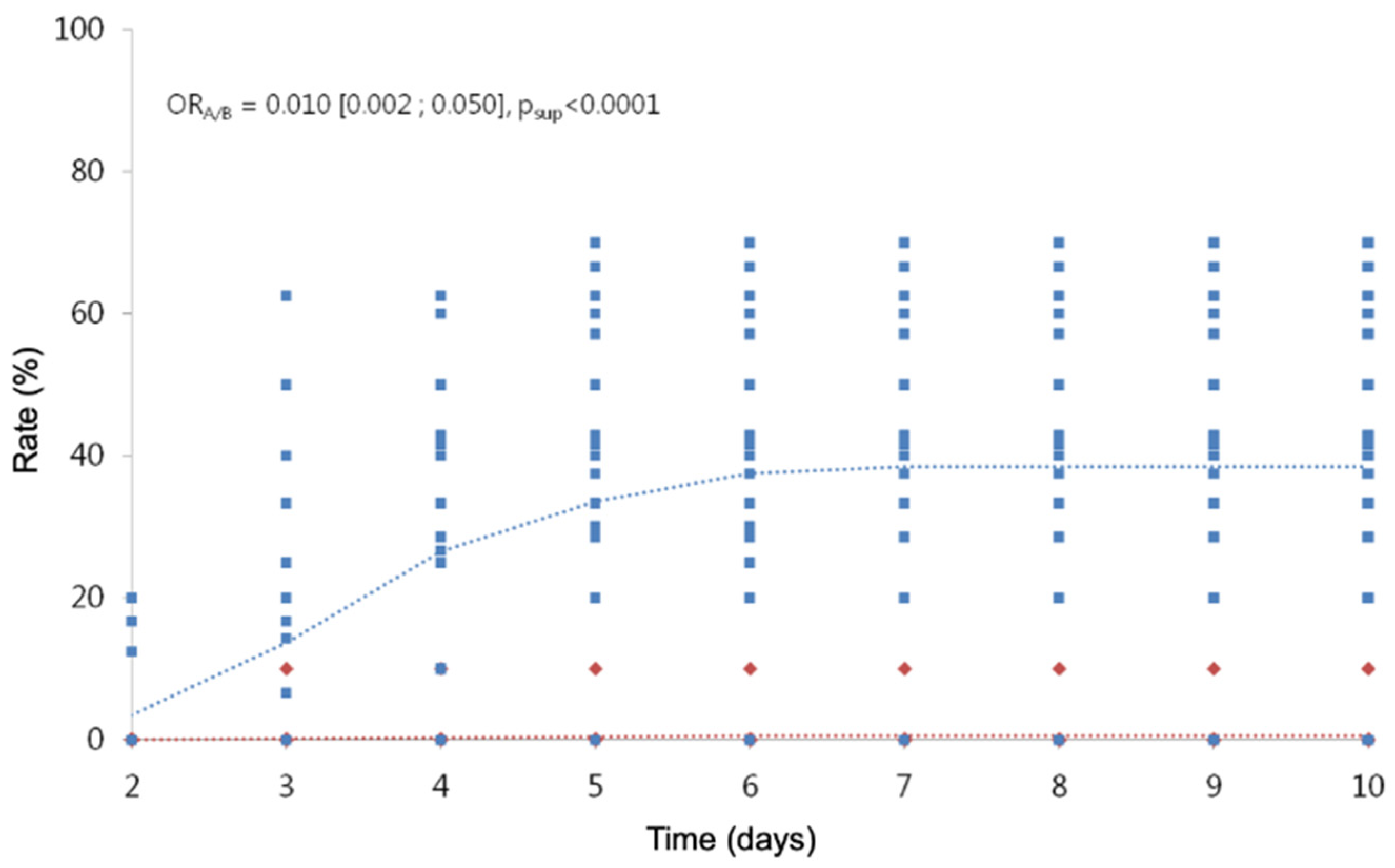
3.2.2. Comparison of Cumulative Hatching Rates from D2 to D10
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Chosidow, O. Scabies and pediculosis. Lancet 2000, 355, 819–826. [Google Scholar] [CrossRef]
- Frankowski, B.L.; Bocchini, J.A. Head lice. Pediatrics 2010, 126, 392–403. [Google Scholar] [CrossRef] [PubMed]
- Durand, R.; Bouvresse, S.; Berdjane, Z.; Izri, A.; Chosidow, O.; Clark, J.M. Insecticide resistance in head lice: Clinical, parasitological and genetic aspects. Clin. Microbiol. Infect. 2012, 18, 338–344. [Google Scholar] [CrossRef] [PubMed]
- Pearlman, D.L. A simple treatment for head lice: Dry-on, suffocation-based pediculicide. Pediatrics 2004, 114, 275–279. [Google Scholar] [CrossRef]
- Richling, I.; Böckeler, W. Lethal Effects of Treatment with a Special Dimeticone Formula on Head Lice and House Crickets (Orthoptera, Ensifera: Acheta domestica and Anoplura, Phthiraptera: Pediculus humanus). Arzneimittelforschung 2008, 58, 248–254. [Google Scholar] [CrossRef]
- Burgess, I.F.; Brown, C.M.; Lee, P.N. Treatment of head louse infestation with 4% dimeticone lotion: Randomised controlled equivalence trial. BMJ 2005, 330, 1423. [Google Scholar] [CrossRef]
- Lebwohl, M.; Clark, L.; Levitt, J. Therapy for head lice based on life cycle, resistance, and safety considerations. Pediatrics 2007, 119, 965–974. [Google Scholar] [CrossRef]
- Chosidow, O.; Giraudeau, B.; Cottrell, J.; Izri, A.; Hofmann, R.; Mann, S.G.; Burgess, I. Oral ivermectin versus malathion lotion for difficult-to-treat head lice. N. Engl. J. Med. 2010, 362, 896–905. [Google Scholar] [CrossRef]
- Heukelbach, J.; Oliveira, F.A.; Richter, J.; Häussinger, D. Dimeticone-based pediculicides: A physical approach to eradicate head lice. Open Dermatol. J. 2010, 4, 77–81. [Google Scholar] [CrossRef]
- Mehlhorn, H.; Abdel-Ghaffar, F.; Al-Rasheid, K.A.; Schmidt, J.; Semmler, M. Ovicidal effects of a neem seed extract preparation on eggs of body and head lice. Parasitol. Res. 2011, 109, 1299–1302. [Google Scholar] [CrossRef]
- Combescot-Lang, C.; Vander Stichele, R.H.; Toubate, B.; Veirron, E.; Mumcuoglu, K.Y. Ex vivo effectiveness of French over-the-counter products against head lice (Pediculus humanus capitis De Geer, 1778). Parasitol. Res. 2015, 114, 1779–1792. [Google Scholar] [CrossRef]
- Mumcuoglu, K.Y.; Miller, J.; Zamir, C.; Zentner, G.; Helbin, V.; Ingber, A. The in vivo pediculicidal efficacy of a natural remedy. Isr. Med. Assoc. J. IMAJ 2002, 4, 790–793. [Google Scholar] [PubMed]
- Kurt, O.; Balcioglu, I.C.; Burgess, I.F.; Limoncu, M.E.; Girginkardeşler, N.; Tabak, T.; Muslu, H.; Ermiş, O.; Sahin, M.T.; Bilac, C.; et al. Treatment of head lice with dimeticone 4% lotion: Comparison of two formulations in a randomised controlled trial in rural Turkey. BMC Public Health 2009, 9, 441. [Google Scholar] [CrossRef] [PubMed]
- Burgess, I.F.; Burgess, N.A. Dimeticone 4% liquid gel found to kill all lice and eggs with a single 15 minute application. BMC Res. Notes 2011, 4, 15. [Google Scholar] [CrossRef] [PubMed]
- Heukelbach, J.; Pilger, D.; Oliveira, F.A.; Khakban, A.; Ariza, L.; Feldmeier, H. A highly efficacious pediculicide based on dimeticone: Randomized observer blinded comparative trial. BMC Infect. Dis. 2008, 8, 115. [Google Scholar] [CrossRef] [PubMed]
- Weeks, A.; Daly, D.C.; Simpson, B.B. The phylogenetic history and historical biogeography of the frankincense and myrrh family (Burseraceae) based on nuclear and chloroplast sequence data. Mol. Phylogenet. Evol. 2005, 35, 85–101. [Google Scholar] [CrossRef]
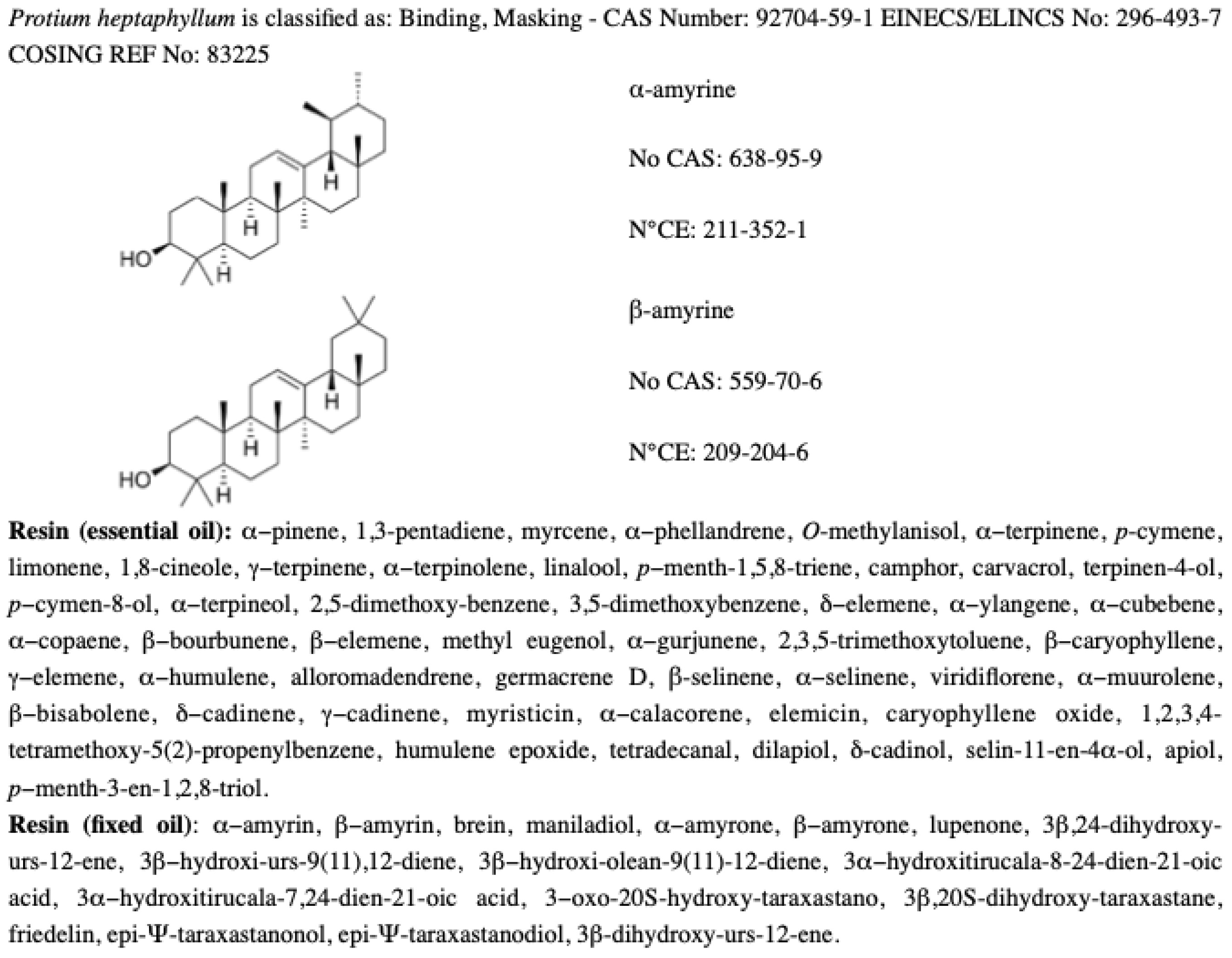
- Lima, E.M.; Nascimento, A.M.; Lenz, D.; Scherer, R.; Meyrelles, S.S.; Boëchat, G.A.P.; Andrade, T.U.; Endringer, D.C. Triterpenes from the Protium heptaphyllum resin–chemical composition and cytotoxicity. Rev. Bras. Farmacogn. 2014, 24, 399–407. [Google Scholar] [CrossRef]
- Lorenzi, H. Árvores Brasileiras: Manual de Identificação e Cultivo de Plantas Arbóreas Nativas do Brasil, 3rd ed.; Instituto Plantarum: Nova Odessa, Brazil, 2008. [Google Scholar]
- Lorenzi, H.; Matos, F.J.A. Medicinal Plants in Brazil: Native and Exotic; Instituto Plantarum: Nova Odessa, Brazil, 2008. [Google Scholar]
- Corrêia, M. Dictionary of Useful Plants of Brazil; National Press: Rio de Janeiro, Brazil, 1926; Volume 1. [Google Scholar]
- Ndinga, A.M.E. Inventaire et Analyse Chimique des Exsudats des Plantes d’Utilisation Courante au Congo-Brazzaville. Ph.D. Thesis, Chimie analytique. Université Paris Sud - Paris XI. Université MarienNgouabi, Brazzaville, Congo, 2015. [Google Scholar]
- Langenheim, J.H. Plant Resins: Chemistry, Evolution, Ecology and Ethnobotany; Timber Press: Portland, OR, USA, 2003. [Google Scholar]
- Oliveira, F.A.; Vieira-Júnior, G.M.; Chaves, M.H.; Almeida, F.R.; Florêncio, M.G.; Lima, R.C., Jr.; Rao, V.S. Gastroprotective and anti-inflammatory effects of resin from Protium heptaphyllum in mice and rats. Pharmacol. Res. 2004, 49, 105–111. [Google Scholar] [CrossRef]
- Susunaga, G.S. Estudo Químico e Biológico da Resina Produzida pela Espécie Protium heptaphyllum March. (Burseraceae). Master’s Thesis, Universidade Federal do Amazonas, Manaus, Brazil, 1996; pp. 1–163. [Google Scholar]
- Siani, A.C.; Ramos, M.F.S.; Menezes, O.; Ribeiro, R.; Fernandez, E.; Soares, R.O.; Rosas, E.C.; Susunaga, G.S.; Guimarães, A.C.; Zoghbi, M.G.; et al. Evaluation of antiinflamatory-related activity of essential oils from the leaves and resin of species of Protium. J. Ethnopharmacol. 1999, 66, 57–69. [Google Scholar] [CrossRef]
- Bandeira, N.; Fonseca, A.M.; Costa, S.M.O.; Lins, M.U.D.S.; Pessoa, O.D.L.; Monte, F.J.Q.; Lemos, T.L.G. Antimicrobial and antioxidant activities of the essential oil of resin of Protium heptaphyllum. Nat. Prod. Commun. 2006, 1, 1934578X0600100207. [Google Scholar] [CrossRef]
- Oliveira, F.A. Estudo das Atividades Farmacológicas da Resina de Protium hetaphyllum (Aubl.) March. e de seus Principais Constituintes, Mistura de α e β Amirina. Ph.D. Thesis, Universidade Federal do Ceará, Fortaleza, Brazil, 2005; pp. 1–242. [Google Scholar]
- Maia, R.M.; Barbosa, P.R.; Cruz, F.G.; Roque, N.F.; Fascio, M. Triterpenos da resina de Protium heptaphyllum March (Bourseraceae): Caracterização em misturas binárias. Quim. Nova 2000, 23, 623–626. [Google Scholar] [CrossRef]
- Rüdiger, A.L.; Siani, A.C.; Junior, V.V. The chemistry and pharmacology of the South America genus Protium Burm. f.(Burseraceae). Pharmacogn. Rev. 2007, 1, 93–104. [Google Scholar]
- Feldmeier, H. Pediculosis capitis: New insights into epidemiology, diagnosis and treatment. Eur. J. Clin. Microbiol. Infect. Dis. 2012, 31, 2105–2110. [Google Scholar] [CrossRef] [PubMed]
- Balcioglu, C.; Burgess, I.F.; Limoncu, M.E.; Şahin, M.T.; Ozbel, Y.; Bilac, C.; Larsen, K.S. Plastic detection comb better than visual screening for diagnosis of head louse infestation. Epidemiol. Infest. 2008, 136, 1425–1431. [Google Scholar] [CrossRef]
- Ash, M. Handbook of Preservatives; Synapse Info Resources: Endicott, NY, USA, 2004; p. 474. [Google Scholar]
- Meinking, T.L.; Clineschmidt, C.M.; Chen, C.; Kolber, M.A.; Tipping, R.W.; Furtek, C.I.; Guzzo, C.A. An observer-blinded study of 1% permethrin creme rinse with and without adjunctive combing in patients with head lice. J. Pediatrics 2002, 141, 665–670. [Google Scholar] [CrossRef]
- Meinking, T.L.; Vicaria, M.; Eyerdam, D.H.; Villar, M.E.; Reyna, S.; Suarez, G. Efficacy of a reduced application time of Ovide lotion (0.5% malathion) compared to Nix creme rinse (1% permethrin) for the treatment of head lice. Pediatric Dermatol. 2004, 21, 670–674. [Google Scholar] [CrossRef]
- Hipolito, R.B.; Mallorca, F.G.; Zuniga-Macaraig, Z.O.; Apolinario, C.; Wheeler-Sherman, J. Head louse infestation: Single drug versus combination therapy with one percent permethrin and trimethoprim/sulfamethoxazole. Pediatrics 2001, 107, 30. [Google Scholar] [CrossRef]
- Carson, D.S.; Tribble, W.; Weart, C.W. Pyrethrins combined with piperonyl butoxide (RID) vs 1% permethrin (NIX) in the treatment of head lice. Am. J. Dis. Child. 1988, 142, 768–769. [Google Scholar] [CrossRef]
- Serrano, L.; Decesar, L.S.; Pham, L. Evaluation of the efficacy and safety of 1% sodium chloride (LiceFreee Spray) against 1% permethrin crème rinse on head lice infested individuals. Pharmacol. Pharm. 2013, 4, 266–273. [Google Scholar] [CrossRef]
- Downs, A.M.R.; Stafford, K.A.; Harvey, I.; Coles, G.C. Evidence for double resistance to permethrin and malathion in head lice. Br. J. Dermatol. 1999, 141, 508–511. [Google Scholar] [CrossRef]
- Meinking, T.L.; Vicaria, M.; Eyerdam, D.H.; Villar, M.E.; Reyna, S.; Suarez, G. A Randomized, Investigator-Blinded, Time-Ranging Study of the Comparative Efficacy of 0.5% Malathion Gel Versus Ovide® Lotion (0.5% Malathion) or Nix® Crème Rinse (1% Permethrin) Used as Labeled, for the Treatment of Head Lice. Pediatric Dermatol. 2007, 24, 405–411. [Google Scholar] [CrossRef] [PubMed]
- Burgess, I.F.; Brunton, E.R.; Burgess, N.A. Single application of 4% dimeticone liquid gel versus two applications of 1% permethrin creme rinse for treatment of head louse infestation: A randomised controlled trial. BMC Dermatol. 2013, 13, 5. [Google Scholar] [CrossRef] [PubMed]
- Burgess, I.F.; Burgess, N.A.; Brunton, E.R. Tocopheryl acetate 20% spray for elimination of head louse infestation: A randomised controlled trial comparing with 1% permethrin creme rinse. BMC Pharmacol. Toxicol. 2013, 14, 43. [Google Scholar] [CrossRef] [PubMed]
- Brandenburg, K.; Deinard, A.S.; DiNapoli, J.; Englender, S.J.; Orthoefer, J.; Wagner, D. 1% permethrin cream rinse vs 1% lindane shampoo in treating pediculosis capitis. Am. J. Dis. Child. 1986, 140, 894–896. [Google Scholar] [CrossRef] [PubMed]
- Taplin, D.; Meinking, T.L.; Castiliero, M.; Sanchez, R. Permethrin 1% creme rinse for the treatment of Pediculus humanus var capitis infestation. Pediatric Dermatol. 1986, 3, 344–348. [Google Scholar] [CrossRef]
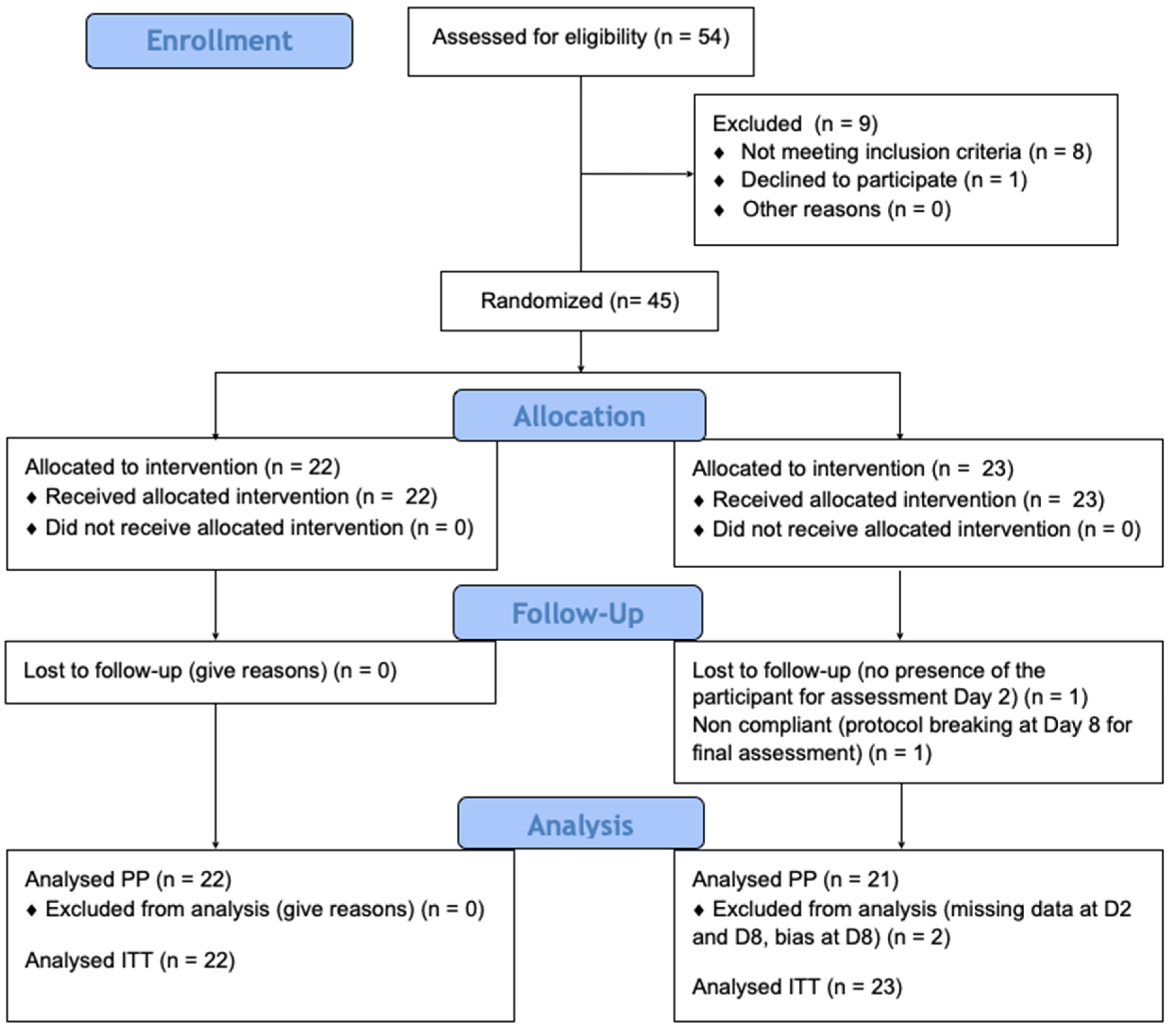


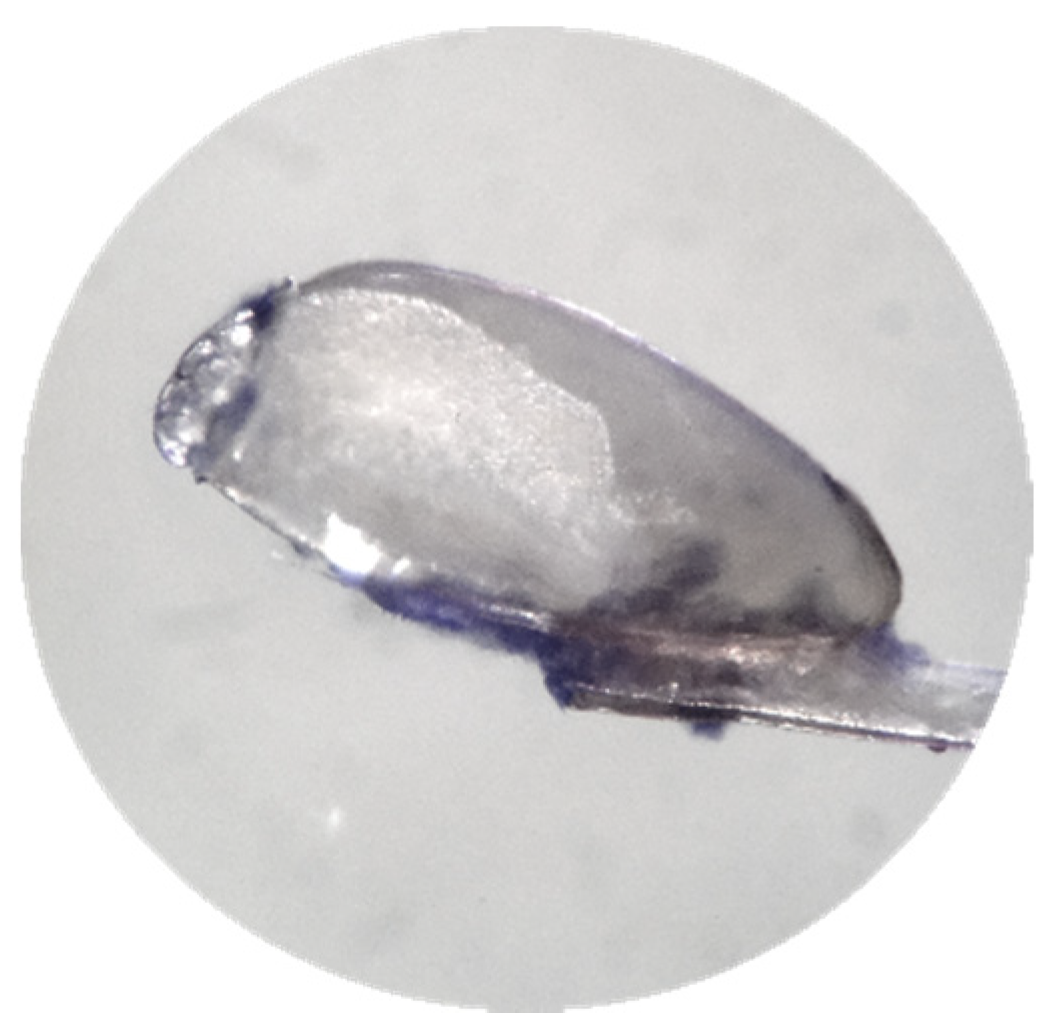
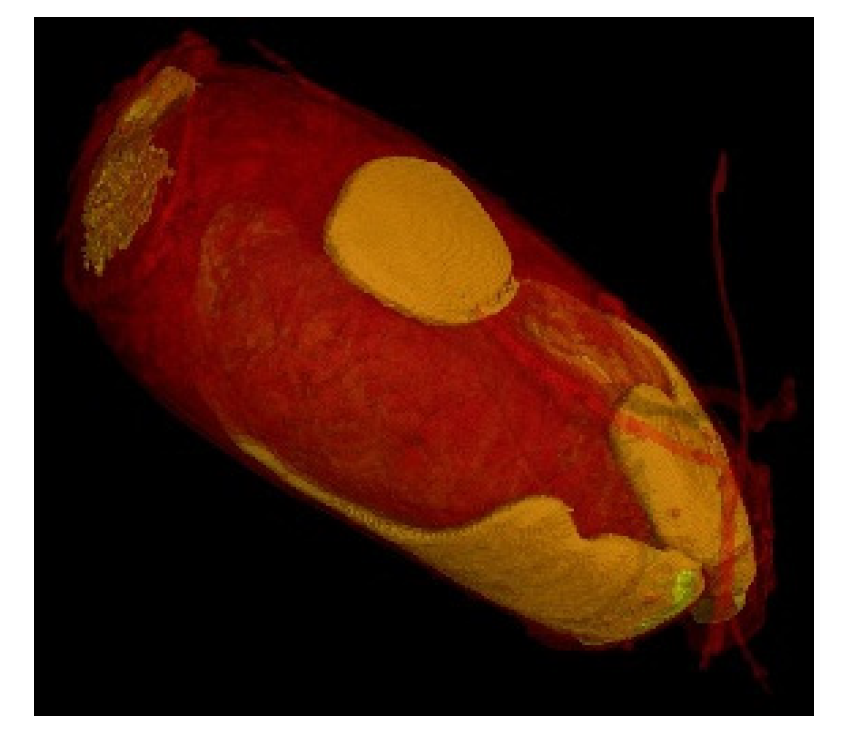
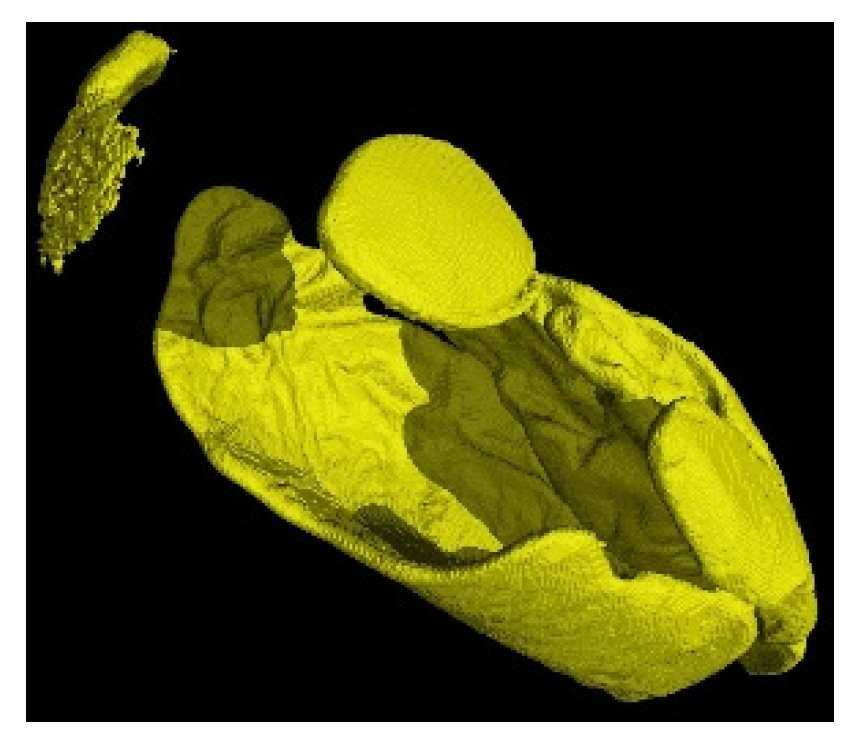
| Total Sample (n = 45) | Treatment A (n = 22) | Treatment B (n = 23) | ||
|---|---|---|---|---|
| Gender F | 38 (84%) | 18 (82%) | 20 (87%) | |
| Age (years) | 8 (7–9) | 8 (6–9) | 8 (7–10) | |
| Sibling | 1 (1–2) | 2 (1–2) | 2 (1–3) | |
| Hair length | ||||
| Short On the shoulders Under the shoulders | 8 (18%) 3 (7%) 34 (76%) | 4 (18%) 1 (5%) 17 (77%) | 4 (17%) 2 (9%) 17 (74%) | |
| Hair type | Crinkly Curly Straight Flexible | 22 (49%) 8 (18%) 14 (31%) 1 (2%) | 10 (45%) 6 (27%) 6 (27%) 0 (0%) | 12 (52%) 2 (9%) 8 (35%) 1 (4%) |
| Normal scalp | 45 (100%) | 22 (100%) | 23 (100%) | |
| Antibiotic treatment before inclusion | 0 (0%) | 0 (0%) | 0 (0%) | |
| Lice infection degree | Low Moderate Strong | 20 (44%) 12 (27%) 13 (29%) | 8 (36%) 6 (27%) 8 (36%) | 12 (52%) 6 (26%) 5 (22%) |
| Treatment A | Treatment B | Difference (IC95%) | p Sup A vs. B | p Sup B vs. A | |
|---|---|---|---|---|---|
| Without considering exogenous re-infestations, which are then considered as failures | |||||
| ITT, worst-case for A | 18/22 (81.8%) | 11/23 (47.8%) | 34.0 (5.8; 60.0) | 0.009 | 0.991 |
| ITT, worst-case for B | 18/22 (81.8%) | 9/23 (39.1%) | 42.7 (14.2; 67.2) | 0.002 | 0.998 |
| PP | 18/22 (81.8%) | 9/21 (42.9%) | 39.0 (7.8; 63.8) | 0.004 | 0.996 |
| Excluding children with exogenous re-infestations | |||||
| ITT, worst-case for A | 18/19 (94.7%) | 11/22 (50.0%) | 44.7 (14.1; 68.6) | 0.001 | 0.999 |
| ITT, worst-case for B | 18/19 (94.7%) | 9/22 (40.9%) | 53.8 (24.2; 75.8) | 0.0002 | 0.9998 |
| PP | 18/19 (94.7%) | 9/20 (45.0%) | 49.7 (21.2; 74.0) | 0.0004 | 0.9996 |
| By including children with exogenous re-infestations and considering them as successful | |||||
| ITT, worst-case for A | 21/22 (95.5%) | 12/23 (52.2%) | 43.3 (16.0; 67.3) | 0.0005 | 0.9995 |
| ITT, worst-case for B | 21/22 (95.5%) | 10/23 (43.5%) | 52.0 (25.7; 74.3) | 0.0001 | 0.9999 |
| PP | 21/22 (95.5%) | 10/21 (47.6%) | 47.8 (17.6; 70.4) | 0.0003 | 0.9997 |
| OR | IC95% | p | |
|---|---|---|---|
| Without considering exogenous re-infestations, which are then considered as failures | |||
| Worst-case analysis for A | |||
| Treatment treatment B treatment A | Reference 11.039 | (1.887; 64.577) | 0.008 |
| Infestation Low Moderate High | Reference 0.214 0.077 | (0.034; 1.350) (0.010; 0.598) | 0.10 0.01 |
| Excluding children with exogenous re-infestations | |||
| Worst-case analysis for A | |||
| Treatment treatment B treatment A | Reference 41.857 | (2.985; 587.019) | 0.006 |
| Infestation Low Moderate High | Reference 0.210 0.062 | (0.027; 1.636) (0.005; 0.774) | 0.14 0.03 |
| By including children with exogenous re-infestations and considering them as successful | |||
| Worst-case analysis for A | |||
| Treatment treatment B treatment A | Reference 53.044 | (3.714; 757.547) | 0.003 |
| Infestation Low Moderate High | Reference 0.188 0.059 | (0.024; 1.453) (0.005; 0.720) | 0.11 0.03 |
| Treatment A | Treatment B | Difference (IC95%) | p Sup A vs. B | |
|---|---|---|---|---|
| ITT, worst-case for A | 22/22 (100%) | 9/23 (39.1%) | 60.9 (35.8; 80.9) | < 0.0001 |
| PP | 22/22 (100%) | 7/21 (33.3%) | 66.7 (38.5; 85.4) | < 0.0001 |
| At Day 2 | Treatment A | Treatment B | Difference (IC95%) | p Sup A vs. B |
|---|---|---|---|---|
| ITT, worst-case for A | 21/22 (95.5%) | 12/23 (52.2%) | 43.3 (16.0; 67.3) | 0.0005 |
| PP | 21/22 (95.5%) | 11/22 (50/0%) | 45.5 (14.4; 70.1) | 0.0004 |
| At Day 1 | Treatment A | Treatment B | Difference (IC95%) | p sup A vs. B |
| ITT, worst-case for A / PP | 21/22 (95.5%) | 12/23 (52.2%) | 43.3 (16.0; 67.3) | 0.0005 |
| Treatment A | Treatment B | Difference (IC95%) | p Sup A. vs. B | |
|---|---|---|---|---|
| ITT, worst-case for A | 21/22 (95.5%) | 9/23 (39.1%) | 56.3 (30.7; 77.6) | <0.0001 |
| PP | 21/22 (95.5%) | 7/21 (33.3%) | 62.1 (33.1; 82.0) | <0.0001 |
| Treatment A | Treatment B | Difference (IC95%) | P-Value ¶ | |
|---|---|---|---|---|
| Intent-to-treat * | ||||
| Treatment success at D8 † | 21/22 (95.5%) | 9/23 (39.1%) | 56.3 (30.7; 77.6) | <0.0001 |
| No alive lice at D8 ‡ | 21/22 (95.5%) | 12/23 (52.2%) | 43.3 (16.0; 67.3) | 0.0005 |
| No alive nymphs at D8 | 22/22 (100%) | 9/23 (39.1%) | 60.9 (35.8; 80.9) | <0.0001 |
| No alive lice and no alive nymph at D1 | 21/22 (95.5%) | 12/23 (52.2%) | 43.3 (16.0; 67.3) | 0.0005 |
| No alive lice and no alive nymph at D2 | 21/22 (95.5%) | 12/23 (52.2%) | 43.3 (16.0; 67.3) | 0.0005 |
| Per protocol | ||||
| Treatment success at D8 † | 21/22 (95.5%) | 7/21 (33.3%) | 62.1 (33.1; 82.0) | <0.0001 |
| No alive lice at D8 ‡ | 21/22 (95.5%) | 10/21 (47.6%) | 47.8 (17.6; 70.4) | 0.0003 |
| No alive nymphs at D8 | 22/22 (100%) | 7/21 (33.3%) | 66.7 (38.5; 85.4) | <0.0001 |
| No alive lice and no alive nymph at D1 | 21/22 (95.5%) | 12/23 (52.2%) | 43.3 (16.0; 67.3) | 0.0005 |
| No alive lice and no alive nymph at D2 | 21/22 (95.5%) | 11/22 (50.0%) | 45.5 (14.4; 70.1) | 0.0004 |
| Absence of Alive Lice on D1 and D2 and Absence of Living Immatures on D8 | |||
|---|---|---|---|
| No | Yes | ||
| Absence of alive lice and living immatures on D1 | No | 1 | 0 |
| Yes | 0 | 21 | |
| Absence of Alive Lice on D1 and D2 and Absence of Living Immatures on D8 | |||
|---|---|---|---|
| No | Yes | ||
| Absence of alive lice and living immatures on D1 | No | 11 | 0 |
| Yes | 3 | 9 | |
| Treatment A | Treatment B | Odds-Ratio | IC95% | p | |
|---|---|---|---|---|---|
| Hatching rate (%) | 1.8 (6.6) | 53.7 (26.5) | 0.017 | (0.006; 0.049) | <0.0001 |
| Hatching rate of viable nymphs (%) | 0.5 (2.1) | 40.8 (20.4) | 0.007 | (<0.001; 0.055) | <0.0001 |
| Odds-Ratio A vs. B | IC95% | p | |
|---|---|---|---|
| Hatching rate (%) | 0.023 | (0.007; 0.074) | <0.0001 |
| Hatching rate of viable nymphs (%) | 0.010 | (0.002; 0.050) | <0.0001 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Leal Cardoso, J.H.; Noronha Coelho de Souza, A.; Militão de Souza, F.; Sa Preire, S.; Pinçon, C. Treatment of Head Louse Infestation with a Novel Mixture Made of Semi-Crystalline Polymers and Plant Extracts: Blind, Randomized, Controlled, Superiority Trial. Cosmetics 2020, 7, 25. https://doi.org/10.3390/cosmetics7020025
Leal Cardoso JH, Noronha Coelho de Souza A, Militão de Souza F, Sa Preire S, Pinçon C. Treatment of Head Louse Infestation with a Novel Mixture Made of Semi-Crystalline Polymers and Plant Extracts: Blind, Randomized, Controlled, Superiority Trial. Cosmetics. 2020; 7(2):25. https://doi.org/10.3390/cosmetics7020025
Chicago/Turabian StyleLeal Cardoso, José Henrique, Andrelina Noronha Coelho de Souza, Francisco Militão de Souza, Samia Sa Preire, and Claire Pinçon. 2020. "Treatment of Head Louse Infestation with a Novel Mixture Made of Semi-Crystalline Polymers and Plant Extracts: Blind, Randomized, Controlled, Superiority Trial" Cosmetics 7, no. 2: 25. https://doi.org/10.3390/cosmetics7020025
APA StyleLeal Cardoso, J. H., Noronha Coelho de Souza, A., Militão de Souza, F., Sa Preire, S., & Pinçon, C. (2020). Treatment of Head Louse Infestation with a Novel Mixture Made of Semi-Crystalline Polymers and Plant Extracts: Blind, Randomized, Controlled, Superiority Trial. Cosmetics, 7(2), 25. https://doi.org/10.3390/cosmetics7020025




