Topical Sunscreen Application Preventing Skin Cancer: Systematic Review
Abstract
1. Introduction
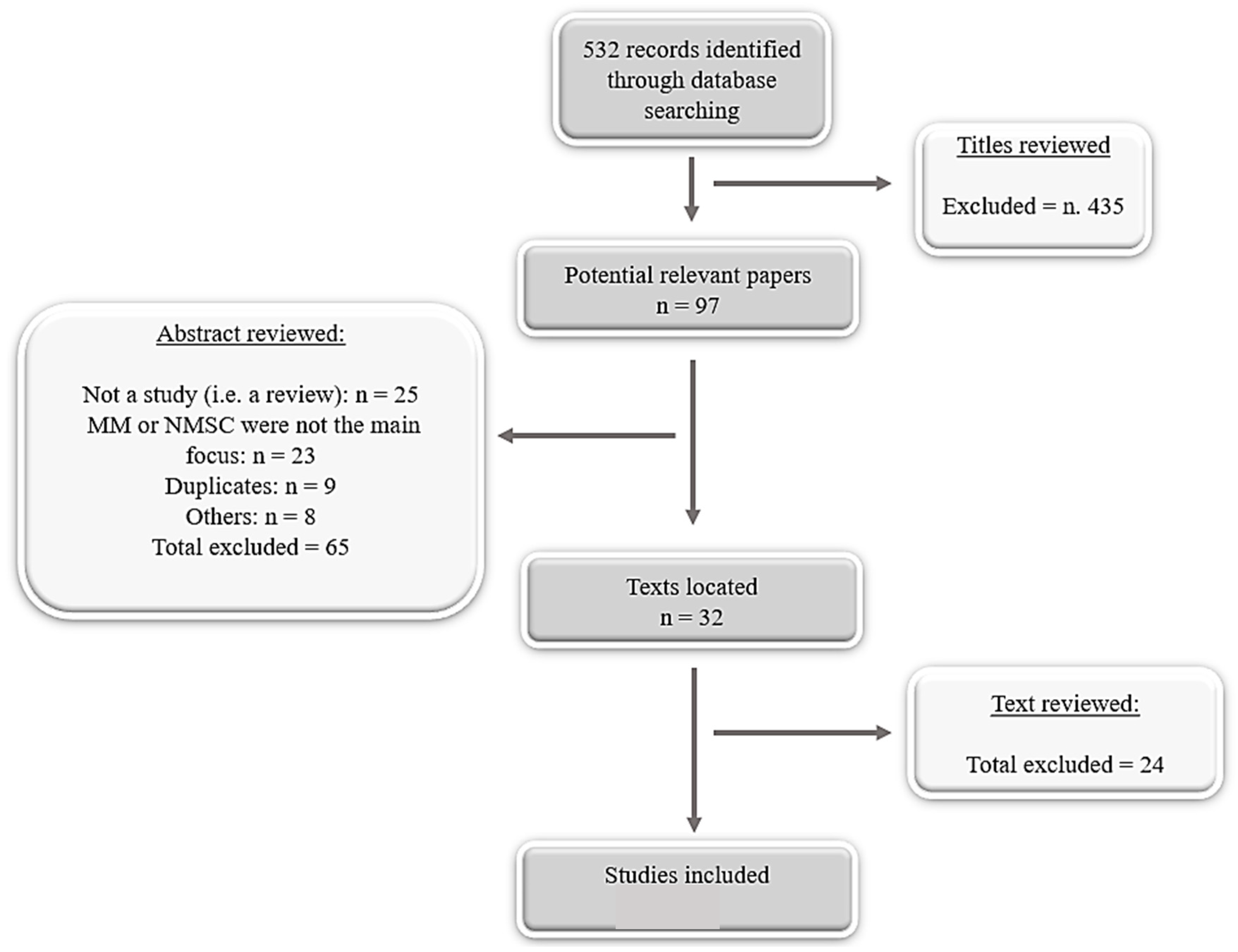
2. Materials and Methods
2.1. Criteria for Considering Studies for this Review. The Specification of Types of Studies, of Participants, of Interventions (and Comparisons), and the Types of Outcomes that Are of Interest Were Considered the Basis of the Pre-Specified Eligibility Criteria for this Work
- Types of studies
- Merge search results using reference management software, and remove duplicate records of the same report.
- Examine titles and abstracts to remove obviously irrelevant reports
- Examine full-text reports for compliance of studies with eligibility criteria.
- Make final decisions on study inclusion and proceed to data collection.
- Types of participants
- Types of interventions
- Experimental studied measuring UVR induced damages in humane skin using sunscreen with Sun Protection Factor (SPF) at least 15 or more versus placebo or other interventions were considered in this review. The Sun Protection Factor refers to the product’s ability to screen or block out the UVB sun’s rays. Following the European guidelines, the sunscreen products must assure also at least 1/3 of protection against UVA in relation to the UVB protection. Thus, values lower than 15 were not considered, because they cannot guarantee a minimum of UVA protection.
- Type of outcomes measures
- -
- Primary outcomes: melanoma confirmed clinically or histopathologically at any follow-up, basal-cell carcinomas (BCC) confirmed clinically or histopathologically at any follow-up, squamous-cell carcinomas (SCC) confirmed clinically or histopathologically at any follow-up.
- -
- Secondary outcomes: actinic keratoses (AK) clinically or histopathologically confirmed at any follow-up. Studies that were conducted on animals, animal models, and cell lines were excluded.
2.2. Search Strategy for Identification of Studies
2.3. Extraction and Unification Data
- -
- General data: author and year of publication, study design, and characteristic of selected population.
- -
- Treatment strategy, in term of application of sunscreen alone or with oral supplementation.
- -
- Sunscreen information: SPF, brand, UV filters and their percentage, spectrum, and type of formulation.
- -
- Additional information: phototype, intentional exposure, sunscreen amount, latitude, reapplication.
- -
- Statistical information: statistical method used, adjusting for factors.
3. Result
3.1. Included Studies Description
3.1.1. Melanoma (MM)
3.1.2. Non-Melanoma Skin Cancer (NMSC)
3.1.3. Precancerous Skin Lesions (PSL)
3.2. General Data Results and Statistical Evaluation
4. Discussion
Comments on Included Studies
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Rogers, H.W.; Weinstock, M.A.; Harris, A.R.; Hinckley, M.R.; Feldman, S.R.; Fleischer, A.B.; Coldiron, B.M. Incidence estimate of nonmelanoma skin cancer in the United States 2006. Arch. Dermatol. 2010, 146, 283–287. [Google Scholar] [CrossRef] [PubMed]
- Apalla, Z.; Lallas, A.; Sotiriou, E.; Lazaridou, E.; Ioannides, D. Epidemiological trends in skin cancer. Dermatol. Pract. Concept. 2017, 7, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Scherer, D.; Kumar, R. Genetics of pigmentation in skin cancer—A review. Mutat. Res. 2010, 705, 141–153. [Google Scholar] [CrossRef] [PubMed]
- Erb, P.; Jingmin, J.; Kump, E.; Mielgo, A.; Wernli, M. Apoptosis and pathogenesis of melanoma and non-melanoma skin cancer. Adv. Exp. Med. Biol. 2008, 624, 283–295. [Google Scholar] [PubMed]
- Salasche, S.J. Epidemiology of actinic keratoses and squamous cell carcinoma. J. Am. Acad. Dermatol. 2000, 42, 4–7. [Google Scholar] [CrossRef] [PubMed]
- Madan, V.; Lear, J.T.; Szeimies, R.M. Non-melanoma skin cancer. Lancet 2010, 375, 673–685. [Google Scholar] [CrossRef]
- Schalka, S.; Manoel, V.; Dos Reis, S., II. Sun protection factor: Meaning and controversies. An. Bras. Dermatol. 2011, 86, 507–515. [Google Scholar] [CrossRef]
- European Union. Regulation (EC) No 1223/2009 of the European Parliament and of the Council of the on Cosmetic Products. Off. J. Eur. Union L 2009, 342, 1. [Google Scholar]
- Kullavanijaya, P.; Lim, H.W. Photoprotection. J. Am. Acad. Dermatol. 2005, 52, 937–958. [Google Scholar] [CrossRef]
- Gil, E.M.; Kim, T.H. UV-induced immune suppression and sunscreen. Photodermatol. Photoimmunol. Photomed. 2000, 16, 101–110. [Google Scholar] [CrossRef]
- Stiefel, C.; Schwack, W. Photoprotection in changing times UV filter efficacy and safety, sensitization processes and regulatory aspects. Int. J. Cosmet. Sci. 2015, 37, 2–30. [Google Scholar] [CrossRef]
- Sambandan, D.R.; Ratner, D. Sunscreens: An overview and update. J. Am. Acad. Dermatol. 2011, 64, 748–758. [Google Scholar] [CrossRef] [PubMed]
- John, J.; Prue, F.J.; Hart, H. Photoprotection: Sunscreens and the immunomodulatory effects of UV irradiation. Mutat. Res. 1998, 422, 155–159. [Google Scholar]
- Nadim, A. Shaath, Ultraviolet filters. Photochem. Photobiol. Sci. 2010, 9, 464–469. [Google Scholar]
- Green, A.C.; Gail, W.M.; Logan, V.; Strutton, G.M. Reduced Melanoma after Regular Sunscreen Use: Randomized Trial Follow-Up. J. Clin. Oncol. 2011, 29, 257–263. [Google Scholar] [CrossRef] [PubMed]
- Ghiasvand, R.; Weiderpass, E.; Green, A.C.; Lund, E.; Veierød, M.B. Sunscreen Use and Subsequent Melanoma Risk: A Population-Based Cohort Study. J. Clin. Oncol. 2016, 34, 3976–3982. [Google Scholar] [CrossRef] [PubMed]
- Green, A.C.; Gail, W.M.; Neale, R.; Hart, V.; Leslie, D.; Parsons, P.; Marks, G.; Gaffney, P.; Battistuta, D.; Frost, C.; et al. Daily sunscreen application and betacarotene supplementation in prevention of basal-cell and squamous-cell carcinomas of the skin: A randomized controlled trial. Lancet 1999, 354, 723–729. [Google Scholar] [CrossRef]
- Van Der Pols, J.C.; Williams, G.M.; Pandeya, N.; Logan, V.; Green, A.C. Prolonged Prevention of Squamous Cell Carcinoma of the Skin by Regular Sunscreen Use. Cancer Epidemiol. Biomark. Prev. 2006, 15, 2546–2548. [Google Scholar] [CrossRef]
- Ulrich, C.; Jurgensen, J.S.; Degen, A.; Hackethal, M.; Ulrich, M.; Patel, M.J.; Eberle, J.; Terhorst, D.; Sterry, W.; Stockfleth, E. Prevention of non-melanoma skin cancer in organ transplant patients by regular use of a sunscreen: A 24 month, prospective, case-control study. Br. J. Dermatol. 2009, 161, 78–84. [Google Scholar] [CrossRef]
- Thompson, S.C. Reduction of solar keratoses by regular sunscreen use. N. Engl. J. Med. 1993, 329, 1147–1151. [Google Scholar] [CrossRef]
- Naylor, M.F.; Boyd, A.; Smith, D.W.; Cameron, G.S.; Hubbard, D.; Neldner, K.H. High Sun Protection Factor Sunscreens in the Suppression of Actinic Neoplasia. Arch. Dermatol. 1995, 131, 170–175. [Google Scholar] [CrossRef]
- Darlington, S.; Williams, G.; Neale, R.; Frost, C.; Green, A. A randomized controlled trial to assess sunscreen application and betacarotene supplementation in the prevent solar keratosis. Arch. Dermatol. 2003, 139, 451–455. [Google Scholar] [CrossRef] [PubMed]
- Food and Drug Administration. Labelling and effectiveness testing; sunscreen drug products for over-the-counter human use. Fed. Regist. 2010, 76, 35620–35665. [Google Scholar]
- Standardization, T.I. Cosmetics—Sun Protection Test Methods—In Vivo Determination of the Sun Protection Factor (SPF) 2010; ISO; European Committee for Standardization (CEN): Brussels, Belgium, 2010; p. 4444. [Google Scholar]
- Scotto, J.; Fears, T.R.; Fraumeni, J.F. Incidence of Nonmelanoma Skin Cancer in the United States; US Department of Health and Human Services, NIH Publication: Rockville Pike, Bethesda, Maryland, April 1983; No. 83-2433.
- De Fabo, E.C. Artic stratospheric ozone depletion and increased UVB radiation: Potential impacts to human health. Int J. Circumpolar Health 2005, 64, 509–522. [Google Scholar] [CrossRef] [PubMed]
- Schaart, F.; Garbe, C.; Orfanos, C. Disappearance of the ozone layer and skin cancer: Attempt at risk assessment. Hautarzt 1993, 44, 63–68. [Google Scholar] [PubMed]
- Autier, P.; Boniol, M.; Doré, J.F. Sunscreen use and increased duration of intentional sun exposure: Still a burning issue. Int. J. Cancer 2007, 121, 1–5. [Google Scholar] [CrossRef] [PubMed]
| Study, Year | Population | Findings |
|---|---|---|
| MELANOMA | ||
| [15] Green A.C., 2011, RTC | Australia (Nambour, Queensland), n. 1339, general population, age 20–69 | Risk of melanoma reduced in daily sunscreen application compared with discretionary use, HR 0.50; 95% CI, 0.24 to 1.02. Invasive melanoma was reduced by 73% in the daily sunscreen group HR 0.27; 95% CI, 0.08 to 0.97 |
| [16] Ghiasvand R., 2016, P-BCS | Norway, n. 171,725, general population, age 30–75 | Risk of melanoma reduced in sunscreen SPF > 15 group compared with sunscreen SPF < 15, HR 0.67; 95% CI, 0.53 to 0.83 |
| NMSC | ||
| [17] Green A.C., 1999, RTC [18] Van Der Pols J.C., RTC follow-up 2006 | Australia (Nambour, Queensland), n. 1621, general population, age 20–69 | The incidence of SCC reduces in sunscreen group compared with control group, RR 0·61, 95% IC, 0·46–0·81. |
| [19] Ulrich C., 2009, C-C | Berlin, n. 120, immunocompromised organ transplant, age 40–77 | 8 new cases of SCC were developed in control group compared 0 diagnosticated in the intervention group (P < 0.01) and 2 new BCC cases in intervention groups compared to 3 cases in control group (n.s.). 11 BCC (2 vs. 9; ns). |
| AK | ||
| [20] Thompson SC., 1993, RCT | Australia, n. 588, general population, age over 40 | The incidence of AK reduces in sunscreen group compared with placebo group, MD 1.53, 95% CI, 0.8 to 2.25 |
| [21] Naylor M.F., 1995, PCT | USA, n. 90, high-risk population, age 39–70 | Reduction of 51% in appearance rate of AK in intervention group compared to the control group |
| [22] Darlington S., 2003, RTC | Australia (Nambour, Queensland), n. 1621, general population, age 20–69 | The AK incidence decreases in sunscreen group compared to no-sunscreen-group, 1992–1994: RR 0.78, 95% CI, 0.64–0.96; 1994–1996: RR 0.94, 95% CI, 0.75–1.19 |
| Study, Year | Approaches | Sunscreen | Application | Additional Info | ||||
|---|---|---|---|---|---|---|---|---|
| Phototype | Intentional Exposure | Sunscreen Amount | Latitude | Re-application | ||||
| MM | ||||||||
| [15] Green A.C., 2011 | (1) sunscreen SPF 16 plus 30mg betacarotene (2) sunscreen SPF 16 plus placebo tables (3) betacarotente only (4) placebo only | SPF: 16, Sunscreen brand: Auscreen Ultrablock Lotion SPF 15-plus, Ross Cosmetics, Melbourne, Australia, Type of formulation: lotion water-resistant, UV filters percentage: 8% Octinoxate and 2% Avobenzone, Spectrum: broad-spectrum rated according to Australian Standard 2604.1. | Daily group: Self-application of a layer to all exposed sites every morning (suggestion: reapplication for heavy sweating or long sun exposure), Discretional group: continue application of sunscreen at their usual discretionary rate | Measured weights of returned bottles | Suggestion | |||
| [16] Ghiasvand R., 2016 | (1) sunscreen SPF < 15 (2) sunscreen SPF > 15 | Participants usual routine | Participants usual routine | Excluded: Very dark skin | High or low latitude | |||
| NMSC | ||||||||
| [17] Green A.C., 1999 [18] Van Der Pols J.C., 2006 | (1) sunscreen SPF 16 plus 30mg betacarotene (2) sunscreen SPF 16 plus placebo tables (3) betacarotente only (4) placebo only | SPF: 16, Sunscreen brand: Auscreen Ultrablock Lotion SPF 15-plus, Ross Cosmetics, Melbourne, Australia, Type of formulation: lotion water-resistant, UV filters percentage: 8% Octinoxate and 2% Avobenzone, Spectrum: broad-spectrum rated according to Australian Standard 2604.1. | Daily group: Self-application of a layer to all exposed sites every morning (suggestion: reapplication for heavy sweating or long sun exposure), discretional group: continue application of sunscreen at their usual discretionary rate | Measured weights of returned bottles | Suggestion | |||
| [19] Ulrich C., 2009 | (1) sunscreen SPF 50 plus education (2) sunscreen SPF 50 self-responsible application | SPF: 60+, Sunscreen brand: Daylong actinica; Spirig Pharma Ltd. Switzerland, Type of formulation: water-resistant cream lotion, UV filters: Bis-ethylhexyloxyphenol methoxyphenyl triazine, ethylhexyl triazone, isoamyl p-tetramethylbutylphenol, butyl methoxydibenzoylmethanemethoxycinnamate, ethylhexyl methoxycinnamate and methylene bis-benzotriazolyl, Spectrum: SPF over 60 for UVB, good UVA protection according to the Australian Standards. | Both groups: Written and oral information on sun protection, Sunscreen group: trained 2 mg cm2 to the head, neck, forearms, and hands. | Included: only phenotype II and III | 2mg/cm2 | |||
| AK | ||||||||
| [20] Thmpson SC., 1993 | (1) sunscreen SPF 17, (2) placebo. | SPF: 16, Sunscreen brand: Auscreen Ultrablock Lotion SPF 15-plus, Ross Cosmetics, Melbourne, Australia, Type of formulation: lotion water-resistant, UV filters percentage: 8% Octinoxate and 2% Avobenzone, Spectrum: broad-spectrum rated according to Australian Standard 2604.1. | apply 1.5 mL of sunscreen on exposed spots of the skin every day and it was suggest reapplying it if necessary. | 1.5 mL per day | Suggestion | |||
| [21] Naylor M.F., 1995 | (1) sunscreen SPF 29, (2) placebo. | SPF: 29, Sunscreen brand: n.d., Type of formulation: n.d., UV filters presented: methoxycinnamate, benzophenone-3 and octyl salicylate, Spectrum absorption 280–320 nm (UVB). | participants usual routine. | Estimation n. battles ordered per month | Participants usual routine | |||
| [22] Darlington S., 2003 | (1) sunscreen broad-spectrum SPF 16 plus 30 mg betacarotene, (2) sunscreen broad-spectrum SPF 16 plus placebo tables, (3) betacarotente only, (4) placebo only. | SPF: 16, Sunscreen brand: Auscreen Ultrablock Lotion SPF 15-plus, Ross Cosmetics, Melbourne, Australia, Type of formulation: lotion water-resistant, UV filters percentage: 8% Octinoxate and 2% Avobenzone, Spectrum: broad-spectrum rated according to Australian Standard 2604.1. | Daily group: Self-application of a layer to all exposed sites every morning (suggestion: reapplication for heavy sweating or long sun exposure), discretional group: continue application of sunscreen at their usual discretionary rate. | Measured weights of returned bottles | Suggestion | |||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Perugini, P.; Bonetti, M.; Cozzi, A.C.; Colombo, G.L. Topical Sunscreen Application Preventing Skin Cancer: Systematic Review. Cosmetics 2019, 6, 42. https://doi.org/10.3390/cosmetics6030042
Perugini P, Bonetti M, Cozzi AC, Colombo GL. Topical Sunscreen Application Preventing Skin Cancer: Systematic Review. Cosmetics. 2019; 6(3):42. https://doi.org/10.3390/cosmetics6030042
Chicago/Turabian StylePerugini, Paola, Margherita Bonetti, Arianna Cecilia Cozzi, and Giorgio Lorenzo Colombo. 2019. "Topical Sunscreen Application Preventing Skin Cancer: Systematic Review" Cosmetics 6, no. 3: 42. https://doi.org/10.3390/cosmetics6030042
APA StylePerugini, P., Bonetti, M., Cozzi, A. C., & Colombo, G. L. (2019). Topical Sunscreen Application Preventing Skin Cancer: Systematic Review. Cosmetics, 6(3), 42. https://doi.org/10.3390/cosmetics6030042




