1. Introduction
In the last few decades “green” has become increasingly relevant in various aspects of everyday life, and personal care (PC) products and cosmetics are no exception. Aware of the impact of increased consumption and highly concerned with ingredients’ provenance and sustainability, growing practices, supply chain policies, etc., end consumers are looking more toward ethically conceived PC and cosmetics products.
With over-reliance on resources and pollution becoming major concerns, the overall waste management must be profoundly revamped, and a growing number of businesses are taking the U-turn toward sustainability and integrating the green values of a circular economy into their processes where residues and by-products are consequently converted into value added products.
Due to the global demographic expansion over the last decades and the subsequent substantial demand for edible processed goods, agricultural practices and industrial food manufacturing industries are among the activities that generate enormous quantities of highly environmentally impacting wastes (seeds, pulp, pomace, peels, etc.) [
1]. Urged to decrease its environmental impact, the agri-food sector is putting significant efforts into the identification and development of new uses for their activities’ by-products [
2]. Properly managed, these wastes become main streams of original raw material sources for the cosmetics sector [
3,
4]. These new environmentally-friendly habits offer valuable advantages: (a) reusing such “waste” material to get as much value as possible from every resource, which offers a unprecedented degree of traceability on the materials’ origin as sourcing channels already exist and are secured [
5]; (b) developing the potential of waste to be used as raw material enables the diversification of value chains for one single market segment and may subsequently imply the growth of farmers/producers’ incomes, hence strengthening those sourcing channels; and (c) they constitute furthermore fantastic communication opportunities and with the appropriate marketing strategy, cosmetic brands can really connect with customers through their mutual responsibility toward the environment preservation and gain new market shares.
This article presents a procedure to design efficient cosmetic ingredients while meeting the challenging consumer’s demands for sustainability, naturality, transparency and traceability.
In order to integrate those sustainability values into our process of ingredients’ development and to take advantage of the “green” phenomenon, we accessed a wide range of by-products that were extracted using several solvents. Given the promising results already shown by our research team regarding leaves of
Prunus domestica L. (Rosaceae) [
6], special attention was brought to the potentiality to create valorisation channels for by-products of the plum industry (food usages mainly: fresh, canned or dried fruits, more processed products, etc.).
Prunus domestica L. is a species of flowering plant that includes many varieties of plum trees. P. domestica is the most commonly grown plum in Europe over a wide range of climatic conditions: the fruits constitute a first-rate dessert, and are appreciated to produce tarts, eau de vie, as well as dried plums, known as prunes.
The Blue Perdrigon (also known as Violet Perdrigon, Perdrigon Viollette or Brignoles Violette) is a very old plum variety cultivated in the Provence region and in the Alps, characterized by downy branches and middle-sized oval fruits ripening from mid-August till late September. The variety name stems from the colour of the fruit skin: first reddish, it rapidly turns purple. The flesh, rather firm, sugary and adhering to the stone, presents a greenish to yellowish colour.
In Provence, the dried Brignoles prunes, known as Pistoles, are obtained as follows: the fresh fruit is peeled (the fruit can be slightly warmed up to ease the skin removal), dried, de-stoned, hand-flattened and further dried before consumption [
7]. A resurgence of interest was observed over the last few decades for this culinary specialty, the production of which generates quantities of unused skins that could be valorised somehow rather than simply discarded as they are nowadays.
Young plum trees are immature and do not produce fruits; according to varieties, it sometimes takes four to six years to bear fruits. As many other stone fruits, plum trees are alternate bearing ones, meaning the plant will produce more fruits every other year. Even if this behaviour is internally regulated by the plant (under the control of plant hormones, e.g., gibberellins, among others), it can be controlled by appropriate crop management including regular tree thinning and pruning [
8]. The huge quantities of leaves and thin branches, usually burned, might, if properly valued, constitute a new source of revenue for the plum producers, generating incomes even during the immaturity period before the exploitation of fruits themselves.
If P. domestica fruits and seeds have already been valorised in PC products, no cosmetic ingredients based on leaves or skin exist to our knowledge. Hence, both leaves and skins of Brignoles prunes have therefore been included in our screening of the cosmetic potential of agricultural by-products: organic solvent extracts obtained from some 30 plants and plants by-products were investigated for their anti-aging properties. From this survey, the ethanolic crude extract of P. domestica leaves displays the best unprecedented anti-aging activities and was then selected for further investigations leading to the development of an innovative anti-aging active.
2. Materials and Methods
All chemicals were obtained from Sigma-Aldrich (Saint-Louis, Missouri, MI, USA) unless otherwise stated.
2.1. Plant Material
Leaves and branches of P. domestica were collected in Brignoles, in Provence-Alpes-Côte d’Azur, France. Skins were recovered from Brignoles plums warmed up to produce pistoles. A voucher specimen of P. domestica leafy branches has been deposited in the botanical collection at the Natural History Museum, Nice, under the reference number NICE-D-4432.
2.2. Plant Extraction
Both leaves on one side and skins of P. domestica on the other side were air dried and stored in a ventilated area. The plant material was crushed into a fine powder directly before extraction. For the initial activities’ screening, extracts of plant by-products were obtained via accelerated solvent extraction (ASE), using the Dionex ASE 350 system from Thermo Scientific (Waltham, Massachusetts, MA, USA). These solid–liquid extractions were performed at 70 °C, at a pressure of 100 bar, using ethanol. Powdered plant samples mixed with diatomaceous earth were placed in stainless steel extraction cells with volumes of 66 mL. After a 5 min heating step of the extraction cell at 70 °C, three successive static extraction stages of 5 min were performed by pumping fresh solvent through the system to rinse the sample and the tubing. All the solvent present in the system was then purged with compressed nitrogen, and the resulting extracts (total solvent volume, e.g., extractant and rinsing) were collected in septum-topped vials.
In the second phase of the study, extraction of plant material was performed via direct maceration in ethanol (extraction ratio 1/5) at room temperature (RT) using a magnetic stirrer (500 rpm) for 2 h. The resulting extracts were then filtered over 8–12 µm filter paper.
In the final development phase, extraction of the plant material was performed via direct maceration in propylene glycol (in fact, transposition of the initial ASE extraction is not possible using propylene glycol (PG), as this solvent might be far too viscous to be used for such assays). About 1 g of plant material was extracted with approximately 10 g of solvent (extraction ratio 1/10) at RT or at 70 °C using a magnetic stirrer (500 rpm) for 4 h, 7 h or 48 h. The resulting extracts were then filtered over 8–12 µm filter paper.
PG extraction yields are calculated as follow: the amount of PG added to the dried raw material was weighted and compared to the weight of the PG extract collected after maceration and removal of the plant material by filtration. A further squeezing step of the filter paper was initially performed to gain access to the residual PG soaking the plant material; as no significant increase of the resulting extraction yield was found, this step was dismissed in further experiments at the lab scale.
2.3. Fractionation of P. domestica Leaves Extract
To identify the active fractions, the ethanolic extract of P. domestica leaves was then fractionated over silica gel (normal phase). The fractionation of the bulk extract led to the recovery of six distinct fractions: F1 (300 mL cyclohexane/diethyl ether 50/50 v/v), F2 (300 mL diethyl ether/methanol 80/20 v/v), F3 (300 mL diethyl ether/methanol 50/50 v/v), F4 (300 mL diethyl ether/methanol 20/80 v/v), F5 (300 mL methanol) and F6 (300 mL methanol/water 50/50 v/v). The resulting fractions were further evaluated for their bioactivities, and their respective compositions were addressed by HPLC (high performance liquid chromatography).
2.4. Bioassays
The bioassays were performed as presented previously [
9].
2.4.1. Materials
Untreated 96-well plates were purchased from Thermo Nunc (Villebon-sur-Yvette, Ile-de-France, France), whereas the UV-transparent ones were obtained from Costar, Sigma-Aldrich (Saint-Quentin Fallavier, Auvergne-Rhône-Alpes, France). Adhesive films (Greiner Bio-One, Courtaboeuf, Île-de-France, France) were used to seal the 96-well plates during incubation. Samples (extracts, fractions, standards and controls) were prepared at a concentration of 3.433 mg/mL (to reach a final sample’s concentration of 100 µg/mL in the well once all the reagents had been added) in dimethyl sulfoxide (DMSO) in 1.5 mL Eppendorf tubes, appropriate for the use of the automated pipetting system epMotion® 5075 (Eppendorf, Montesson, Île-de-France, France).
A hydroglycerinated active containing, among others, some
Prunus persica leaves extract, commercialized for its claimed lightening and anti-aging activities, was tested alongside our samples to perform direct comparison between
Prunus species-based ingredients. DMSO tested alone constitutes the negative control (OD
control, with OD stating for optical density) in each plate; it appeared to have no activity on its own. Two positive controls were added in each plate (
Table 1).
2.4.2. Instrumentation
An automated pipetting system Eppendorf epMotion® 5075 was used to carry out the bioassays. A microplate reader (Spectramax Plus 384, Molecular Device, Wokingham, Berkshire, UK) was used to measure absorbance values. Data were acquired with the SoftMaxPro software (Molecular Devices, Wokingham, Berkshire, UK) and the Prism software (GraphPad Software, La Jolla, California, CA, USA) was used to calculate inhibition percentages.
The results are presented as inhibition percentages (I%) calculated as follows (for the DPPH radical scavenging assay, and tyrosinase, lipoxygenase and elastase assays):
or as follows (for the hyaluronidase and collagenase assays):
Similarly, all OD (apart the hyaluronidase ones) were corrected with the blank measurement corresponding to the absorbance of the sample before addition of the substrate.
2.4.3. DPPH Radical Scavenging Assay
The antioxidant activity of extracts was evaluated based on the scavenging activity of the stable 1,1-diphenyl-2-picrylhydrazyl radical (DPPH) [
10,
11]: 150 µL of a solution of ethanol/acetate buffer 0.1 M (50/50) was distributed in each well, together with 7.5 µL of the samples evaluated. A first OD reading was performed at 517 nm (OD
blank). Then, 100 µL of a DPPH solution (386.25 µM in ethanol) were distributed in each well. The sealed plate was incubated in the dark at RT for 30 min before the final OD reading.
2.4.4. Tyrosinase Assays
Tyrosinase is a copper-containing enzyme that plays a key role in melanogenesis; it is mainly involved in the hydroxylation of L-tyrosine into L-DOPA (l-3,4-dihydroxyphenylalanine) and its further oxidation to dopaquinone [
12]. The assays were performed
via distribution of 150 µL of a solution of mushroom tyrosinase (171.66 U/mL in phosphate buffer) in each well, together with 7.5 µL of the extracts evaluated. The filmed plate was incubated at RT for 20 min. Then, 100 µL of a solution of substrate (either L-tyrosine or L-DOPA, 1 mM in phosphate buffer) were distributed in each well, and the final OD reading was performed at 480 nm after 20 min of incubation.
2.4.5. Lipoxygenase Assay
Lipoxygenase is an iron-containing enzyme known to play a key role in inflammation [
13]. The assays were performed as follows: 150 µL of a solution of soybean lipoxygenase (686.66 U/mL in phosphate buffer) was distributed in each well, together with 7.5 µL of the extracts evaluated. The filmed plate was incubated in the dark for 10 min. Then, 100 µL of a solution of linoleic acid in phosphate buffer were distributed in each well. After incubation for 2 min in the dark, a first OD reading was performed at 235 nm; the final OD reading was performed after a further incubation of 50 min.
2.4.6. Elastase Assay
Elastase is an endopeptidase that preferentially digests elastin and is the highly elastic protein responsible for cutaneous firmness, together with collagen [
14]. The assays were performed as follows: 150 µL of a solution of porcine pancreatic elastase (0.171 U/mL in Tris buffer) was distributed in each well, together with 7.5 µL of the extracts. The filmed plate was incubated at RT for 20 min. A first OD reading was performed at 410 nm before the addition of 100 µL of a solution of N-succinyl-Ala-Ala-Ala-p-nitroanilide (2.06 mM in Tris buffer). The final OD reading was performed after a 40-min incubation.
2.4.7. Hyaluronidase Assay
Hyaluronidases are enzymes that degrade hyaluronic acids, which are widely distributed in the body and notably at the periphery of collagen and elastin fibres, that play therefore a major role in skin aging [
15,
16]. The assays were performed as follows: 150 µL of a solution of hyaluronidase (13.3 U/mL in hyaluronidase buffer) was distributed in each well, together with 7.5 µL of the extracts evaluated. The filmed plate was incubated at 37 °C for 20 min, and a first OD reading was performed at 405 nm. Then, 100 µL of a solution of hyaluronic acid (150 µg/mL in pH 5.35 buffer) was distributed in each well. After 30 min incubation at 37 °C, 50 µL of cetyltrimethylammonium bromide (40 mM in a 2 % NaOH solution) was added and a final OD reading was performed.
2.4.8. Collagenase Assay
Collagenases constitute a family of enzymes that cleave collagen and that are more generally involved in the degradation of the extracellular matrix components, thus leading to sagging skin [
17]. The assays were performed as follows: 150 µL of a solution of collagenase (53 U/mL in tricine buffer) was distributed in each well, together with 7.5 µL of the extracts. The filmed plate was incubated at RT for 15 min before the first OD reading was performed at 345 nm. Then, 100 µL of a solution of 2-furanacryloyl-l-leucylglycyl-l-prolyl-l-alanine (5.15 mM in tricine buffer) was distributed in each well, and a final OD reading was performed after a 30 min-incubation.
2.5. Solid Phase Extraction
SPE cartridges (Waters, Guyancourt, Ile-de-France, France; Oasis HLB6cc, 500 mg) were selected as they are filled with polymeric reversed-phase sorbent adequate for the extraction of a wide range of acidic, basic, and neutral compounds from various matrices. The cartridges were fitted into stopcocks and connected to a vacuum manifold. The sorbent was conditioned with 10 mL of methanol (MeOH) and equilibrated with 10 mL water as recommended by the supplier. The propylene glycol extract of P. domestica leaves to analyse was diluted in water (PG extract/water 1/3) to lower its viscosity, hence facilitating its further loading onto the cartridge with the stopcocks opened and the vacuum turned on. The presence of extracted molecules in the effluent was monitored by regular HPLC analyses. Once the cartridge was saturated and the PG was eliminated, elution of the sample was achieved with 4 mL MeOH/dichloromethane (DCM) 1/1 (v/v) to elute all the molecules retained on the cartridge.
2.6. HPLC Analysis
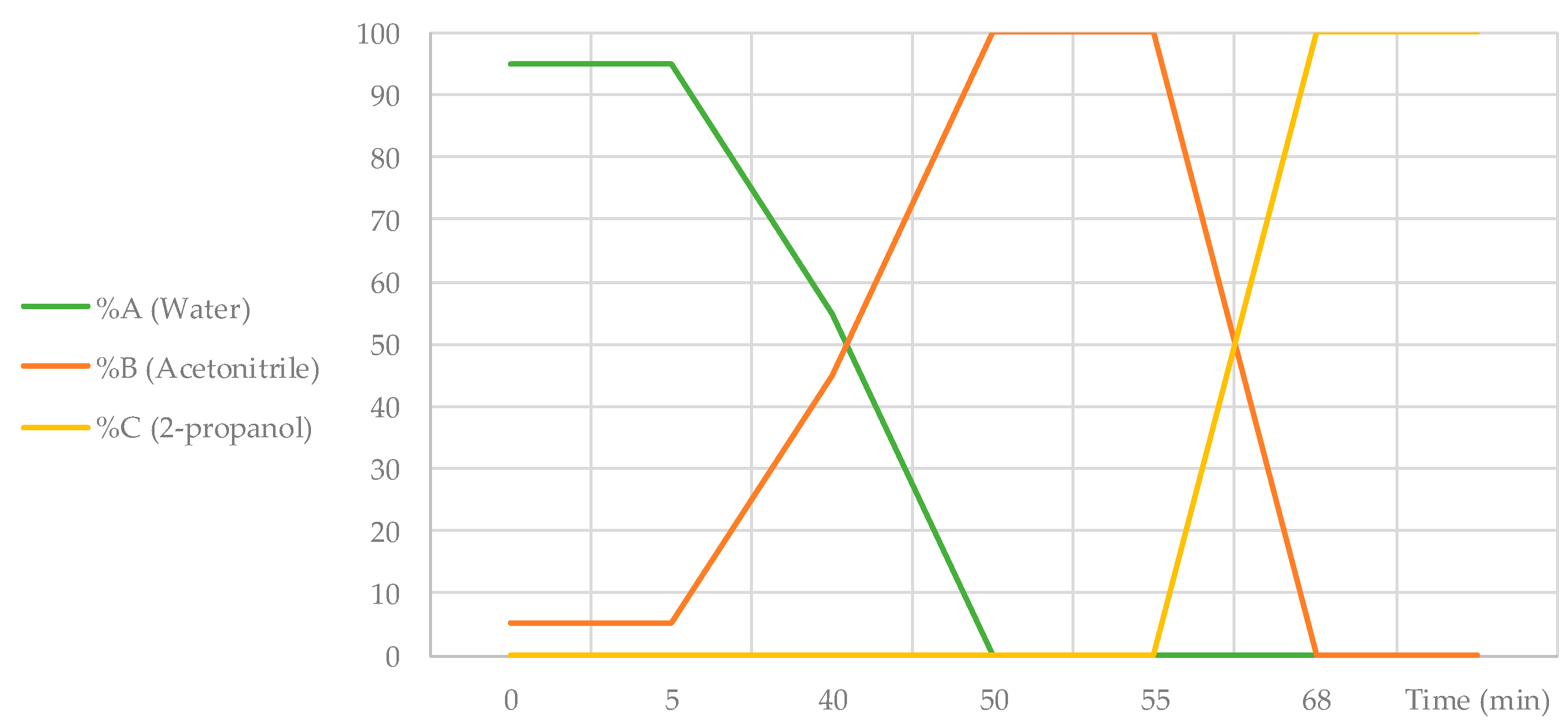
P. domestica extracts and SPE effluent samples were analysed using an HPLC Agilent 1200 system (Courtaboeuf, Ile-de-France, France) equipped with a DAD (diode array detector) and an ELSD (evaporative light scattering detector) operating under the following conditions: injection volume of 20 µL and flow rate was set at 1.0 mL/min. Separations were performed on a C18 column (Phenomenex, Torrance, CA, USA, Luna® 5 μm, 150 mm × 4.6 mm i.d.). The mobile phase consisted of a multistep gradient of water (A), acetonitrile (B) and 2-propanol (C), all acidified with 0.1% formic acid (
Figure 1): 0–5 min, 5% B; 5–40 min, 5–45% B; 40–50 min, 100% B; 50–55 min, 100% B; 55–68 min, 100% C, 68–70 min, 100% C. The DAD was set at 366 nm, and ELSD conditions were set as follows: nebulizer gas pressure 3.7 bars, evaporative tube temperature 40 °C and gain 4.
2.7. Gas Chromatography-Mass Spectrometry (GC-MS)
GC-MS analyses were performed using an Agilent 6890 gas chromatograph (Palo Alto, CA, USA) equipped with an Agilent MSD5973N mass selective detector, a multifunction automatic sampler (Combi-Pal, CTC Analytics, Zwingen, Switzerland), both on a DB-5HT capillary column ((5%-phenyl)-methylpolysiloxane; 0.25 mm × 30 m; film thickness, 0.10 μm) and on an HP-1 MS capillary column (100% polydimethylpolysiloxane; 0.2 mm × 50 m; film thickness, 0.33 μm). Samples (1 µL) were injected in splitless mode (split vent: 50 mL/min, 30 s) and the injector was set at a temperature of 250 °C. The carrier gas was helium in constant flow mode at 1 mL/min. The oven temperature was programmed to rise from 60 °C to 180 °C at 2 °C/min, then from 180 °C to 300 °C at 6 °C/min and kept isothermally at 300 °C for 5 min. Acquisition was performed in scan mode (35–500 a.m.u. (atomic mass unit)/s; scan rate: 3.15 scans/s) and mass spectra were generated at 70 eV.
Compound identifications were based on the comparison of mass spectra with literature, commercial libraries (NIST, Wiley, Indianapolis, USA) and laboratory MS libraries built up from pure substances, combined with comparison of the GC linear retention index (LRI) [
18,
19]. Retention indices were determined with a series of linear alkanes C8–C24 used as a reference.
2.8. Stability Assays
Stability assays were undertaken to ensure that the ingredient developed meets aesthetic and quality standards when stored under specific conditions. Some accelerated stability assays were carried out as follows: the liquid ingredient directly developed in propylene glycol was stored in glass vials at 42 °C, and weekly monitoring was undertaken to assess eventual aesthetic changes in colour and odour, or modification of its chemical composition using HPLC-DAD-ELSD after separation from the cosmetic support
via SPE using the methodology presented in
Section 2.5.
3. Results
3.1. Bioactivities of Crude Extracts
As already stated, a series of by-products of Mediterranean origin were initially selected based on their accessibility (easy recovery after the initial industrial processing or during agricultural activity, considerable volumes recovered that allow to picture promising revalorization, etc.) and their originality regarding the cosmetic activity, and more particularly the anti-aging one. The initial plant screening was performed using ASE, an appropriate technique to extract metabolites covering a large range of structures and polarities, while enabling considerable reduction of extraction time and solvent consumption, which is ideal for the large-scale screening of numerous samples [
20]. Some thirty extracts were recovered, and their anti-aging properties were evaluated
in vitro.
Skin aging is a complex mechanism that is driven by both intrinsic (natural age-dependent/chronological aging, genetic, etc.) and extrinsic factors (premature environmentally-driven aging induced by exposure to ultraviolet light, weather changes and environmental pollution, which aggravates the natural aging process) [
21]. Skin exposure to UV-light, stress, etc., leads to an increased generation of ROS (reactive oxygen species) and subsequent induction of oxidative stress, which constitutes one of the greatest cutaneous threats. High levels of ROS might, among others, activate hyaluronidase, collagenase and elastase, enzymes that specifically degrade structural building blocks of the skin, namely the hyaluronic acid in charge of retaining the moisture of the skin, as well as collagen and elastin, responsible for its elasticity and strength, hence contributing to skin aging [
21,
22].
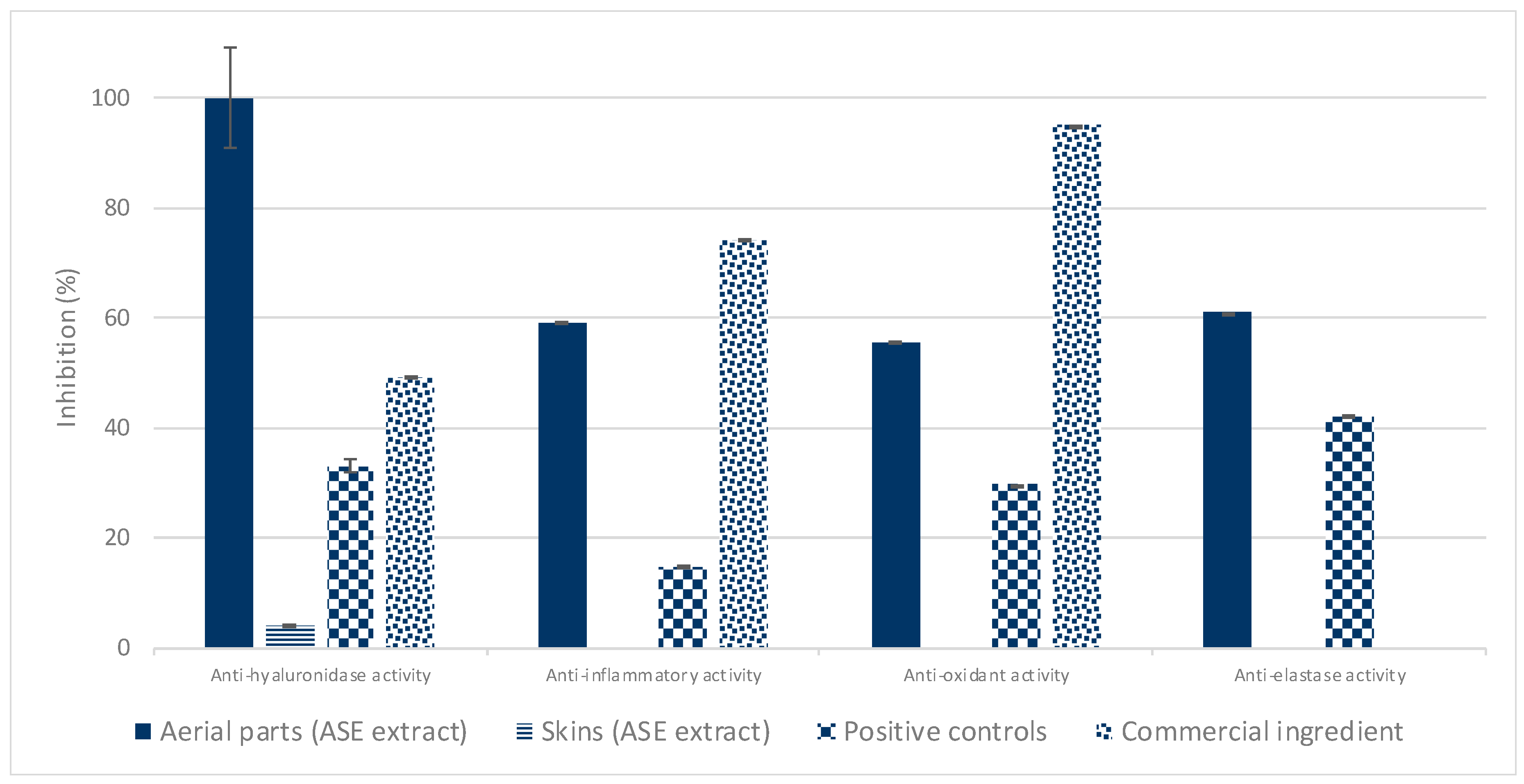
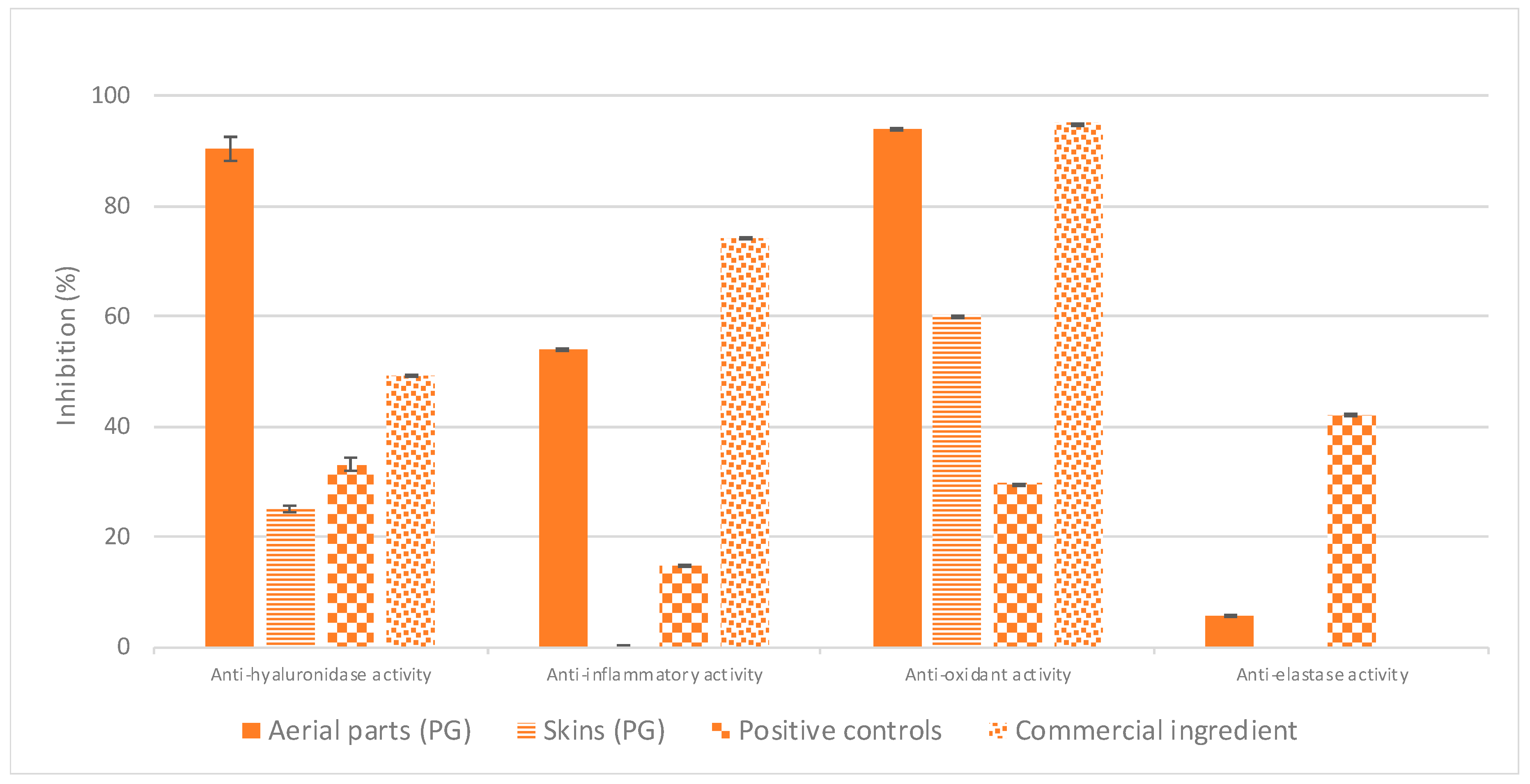
The free radical scavenging and anti-inflammatory activities, as well as specific enzymes’ inhibitory activities of natural extracts were investigated to assess their potentiality to slow down the skin aging process. From this survey, a total of six plants were identified as potentially interesting for the development of anti-aging ingredients. Furthermore, it appears that the crude extract of
P. domestica leaves displays the best unprecedented anti-aging activities, while no remarkable bioactivity was observed for
P. domestica dried skins (
Figure 2).
The easiness of the raw material supply (plant growing in the region, processing activities generating both by-products), and the attractive marketing potential of the resulting cosmetic active conveying the circular economy concepts, induced the selection of P. domestica for further investigations.
A larger
P. domestica leaves extract was obtained
via maceration in ethanol (plant/solvent ratio 1/5
w/
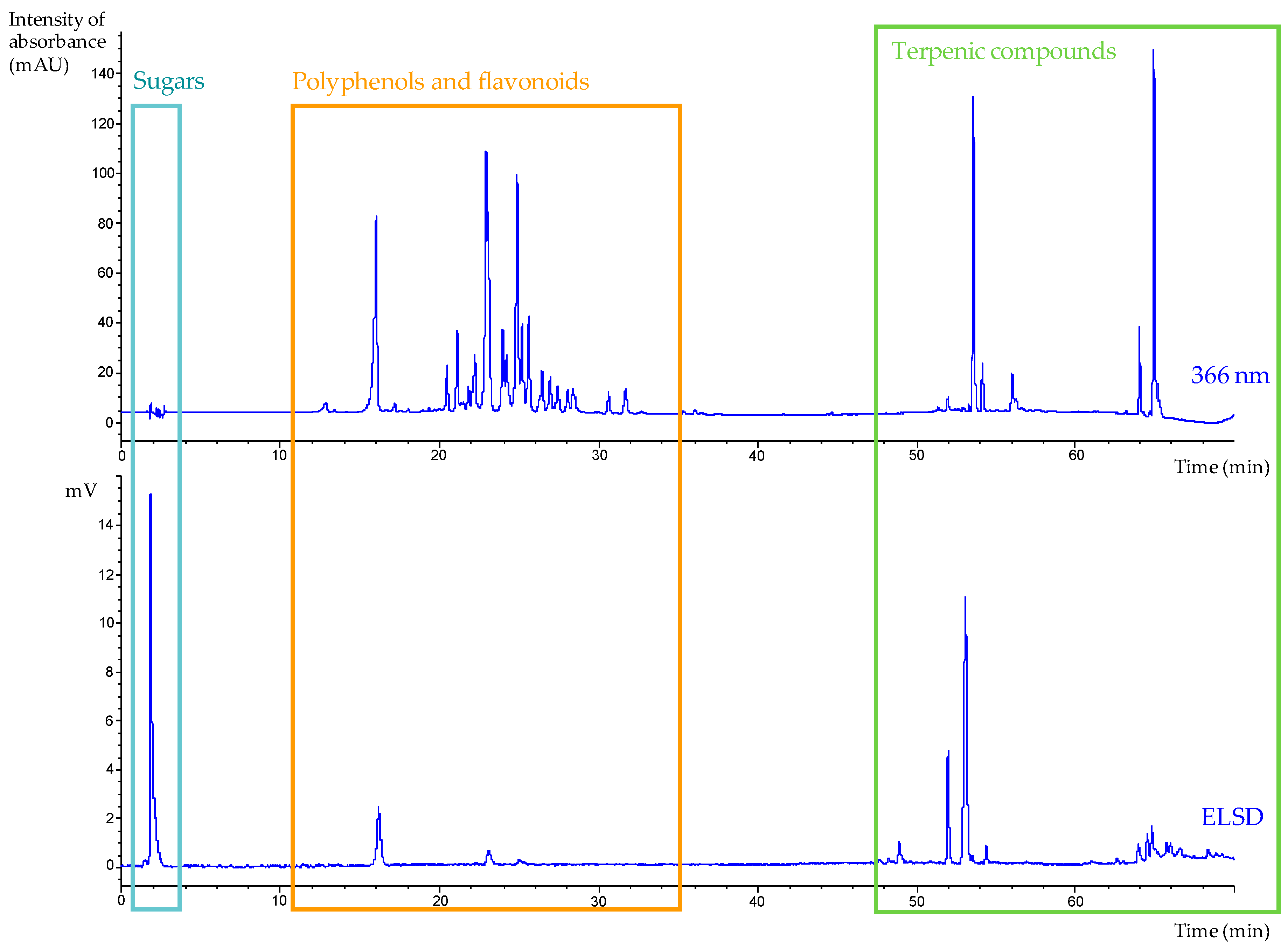
w) at RT (extraction yield 12.3 ± 0.9%). Its HPLC profile (
Figure 3) revealed the presence of some highly polar compounds (e.g., sugars) eluted at the beginning of the analysis, as well as a large group of polyphenols and flavonoids eluted between 12 and 35 min and a series of terpenic compounds.
This extract was then fractionated over silica gel. The fractionation led to the recovery of six distinct fractions: F1 (cyclohexane/diethyl ether 50/50
v/
v), F2 (diethyl ether/methanol 80/20
v/
v), F3 (diethyl ether/methanol 50/50
v/
v), F4 (diethyl ether/methanol 20/80
v/
v), F5 (methanol) and F6 (methanol/water 50/50
v/
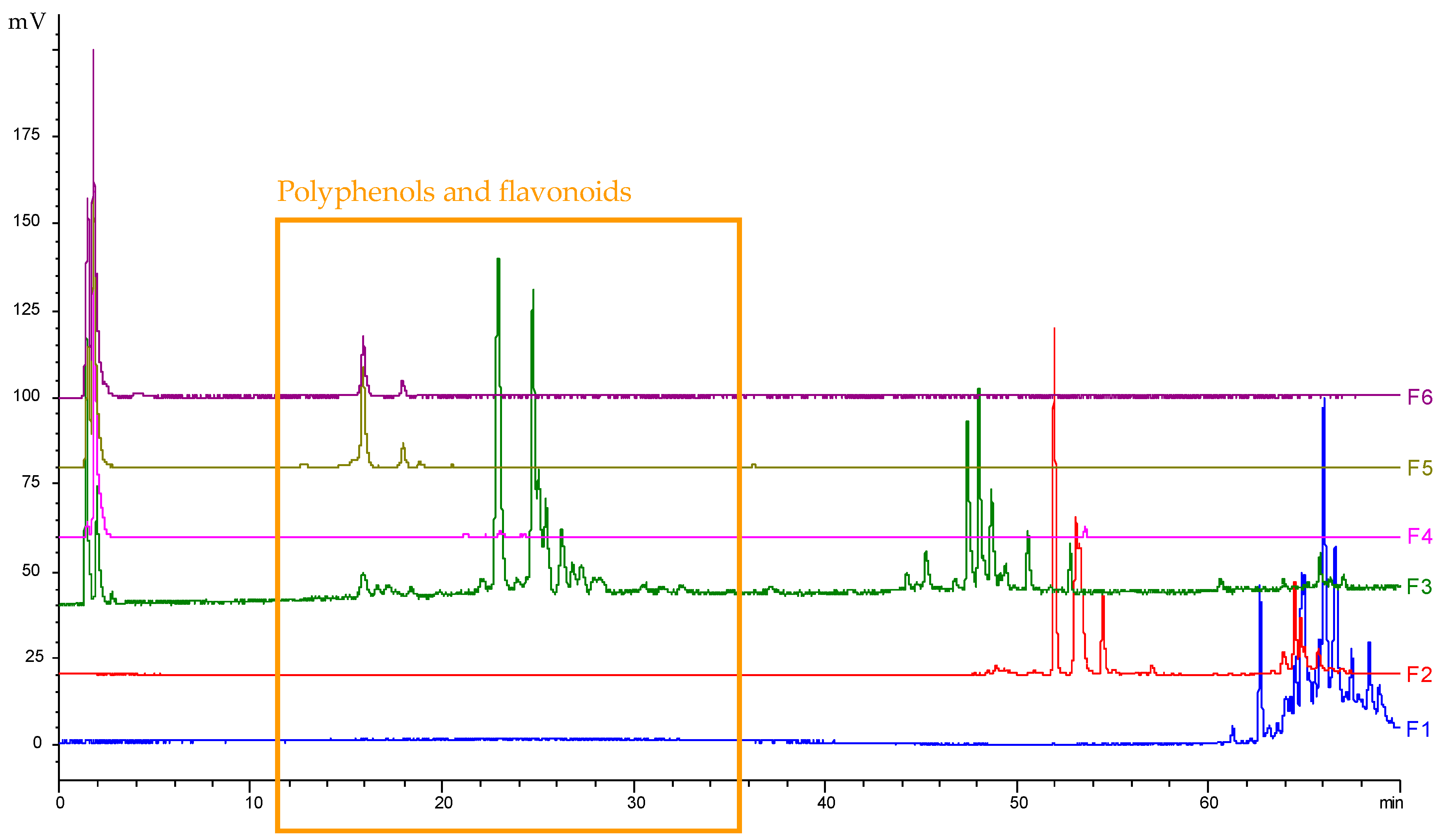
v). The resulting fractions F1 to F6 were analysed using HPLC (
Figure 4) and further evaluated for their bioactivities. To do so, fraction solutions were concentrated at 3.433 mg/mL in DMSO and their activities were assessed the same way as the one of the crude extract. Results of these bioassays are presented in
Table 2. The bulk extract obtained
via ethanolic maceration presents slightly less interesting activities compared to the one obtained using ASE; these differences might be due to the short maceration time (only 2 h), or due to the lower plant/solvent ratio used for the maceration. However, one can note that F3 displayed some very interesting anti-oxidant potency, as well as strong anti-hyaluronidase, anti-inflammatory and anti-elastase activities.
A positive relationship between the anti-oxidant activity of fractions F3, F5 and F6 and the occurrence of compounds eluting in the phenolic/flavonoid region was found (
Figure 4). Even though some further phytochemical analysis is needed to properly characterise the compounds constituting these fractions, one can mention the identification of kaempferol, quercetin and rutin, as well as some of their respective glycosylated derivatives in this eluting region from several
P. domestica extracts previously studied by our research team [
6]. Such observations are also consistent with correlations between chemical composition and bioactivities reported in the literature [
9,
23,
24,
25,
26,
27].
The bulk extract, as well as fractions F1–F6, were further analysed using GC-MS. The compounds identified are listed in
Table 3; no compound was identified with certainty in F2. One must keep in mind that this volatile fraction only represents a small proportion of the molecular content of the respective samples analysed.
3.2. Liquid Cosmetic Ingredient
Botanical extracts that display interesting bioactivities are usually not directly incorporable into cosmetic formulations as they may display undesirable qualities (colour, odour, viscosity, etc.) [
35,
36]. The addition of an appropriate cosmetic support, either liquid or solid, can somehow ease their incorporation into a cosmetic formulation. By being willing to develop liquid ingredients as they are often preferred when formulating cosmetics, propylene glycol was hence used to directly extract the active metabolites of
P. domestica by-products to free the researchers from the time-consuming steps of solvent evaporation and resolubilisation of the dry extract in PG.
P. domestica skins were also incorporated in this experimentation as PG, owing to its lower polarity compared to ethanol, might extract other components that might present some cosmetic activities.
To prepare such extracts, maceration parameters defined earlier for the development of liquid ingredients were used in the first instance [
9]: dried
P. domestica leaves and skin macerate in PG (ratio dried by-product/PG 1/10
w/
w) under stirring for 7 h at RT. The resulting extracts were then filtered over filter paper, and their activities, as well as the ones of propylene glycol alone (no activity reported; data not shown) were assessed using
in vitro bioassays (
Figure 5).
As shown in
Figure 5, the
P. domestica skins PG extract again displayed no significant activities; this bio-inactivity could be explained either by the absence of active molecules in plum skins or by the heating applied to ease the fruit peeling during the preparation of Pistoles [
7], which may lead to degradation of thermolabile active compounds (for example, loss of anti-oxidant activity linked to thermal treatment and to the subsequent decrease of phenolic content was already reported in the literature [
37,
38]). Some exhaustive phytochemical characterisation could be considered to understand its bio-inactivity (and to potentially rule out one of these hypothesis), but given the absence of anti-aging properties,
P. domestica skins were subsequently dropped from this study without further experimentation.
Focus was placed on the development of a liquid ingredient based on
P. domestica leaves as the relevant PG extract displays even better activities (particularly better antioxidant and anti-inflammatory activities were evidenced) than the corresponding EtOH extract obtained using ASE (see
Section 3.1). Several assumptions can be made to explain these differences: PG may be a more appropriate solvent than EtOH to extract active compounds contained in
P. domestica leaves, or on the contrary, it could also be less efficient as EtOH to extract antagonist compounds and thwart the active ones.
3.2.1. Optimization of Extraction Parameters
Attempts were then undertaken to optimize the PG extraction procedure to potentialize the bioactivity of the resulting P. domestica leaves extract (subsequently named PDL) while keeping in mind further industrial scale-up and formulation: the effects of variations of several extraction parameters on the bioactivities of the resultant extract were monitored.
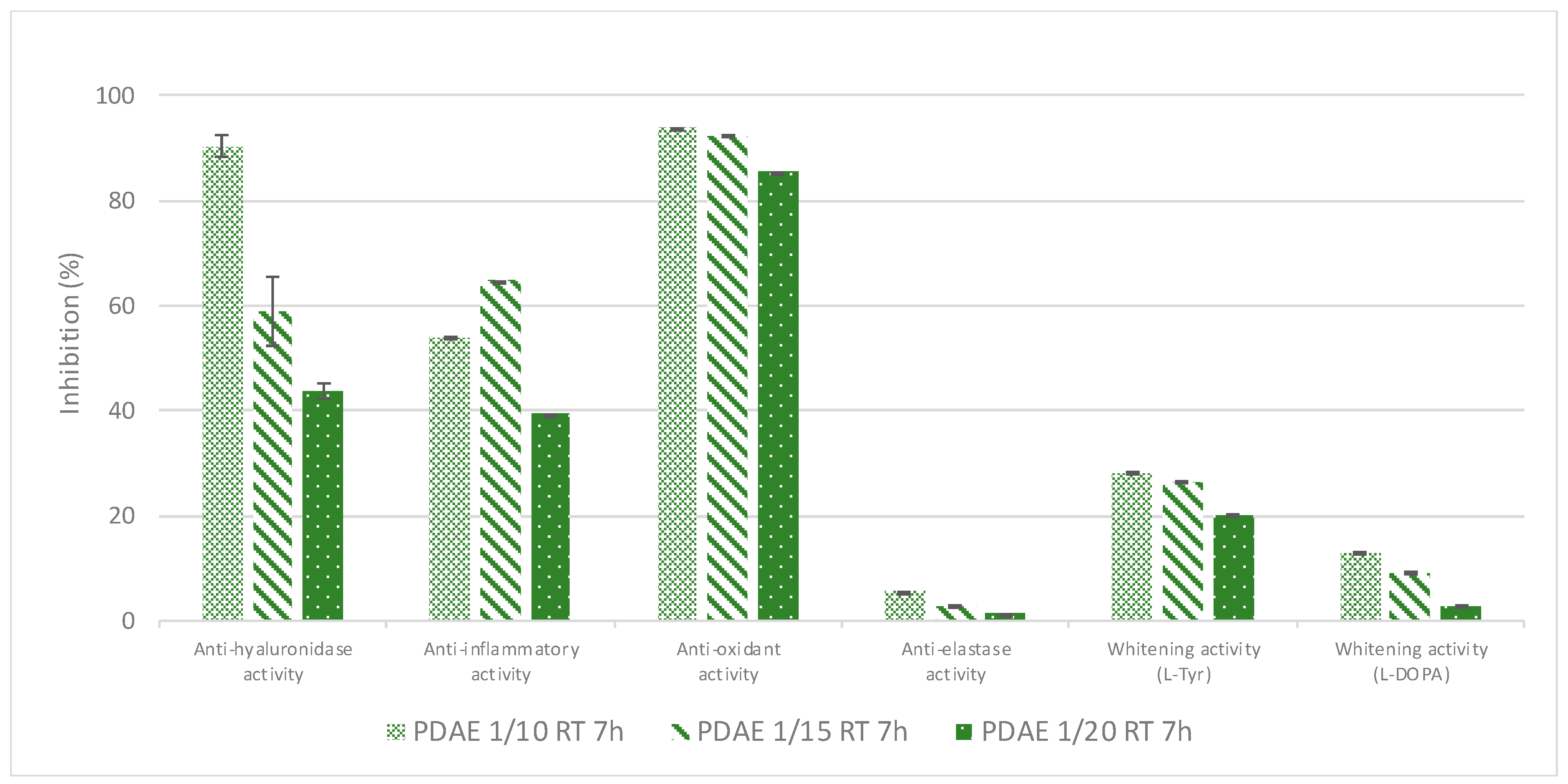
First, the effect of the plant/PG ratio on the resultant extracts’ bioactivities was evaluated: the plant/PG ratio of 1/10 (
w/
w) was set as the minimal convenient ratio to properly extract the plant material and to further manipulate the resulting extract (using a lower plant/PG ratio means that not all the plant material was soaked in PG, so the extraction would be incomplete). The bioactivities of the resultant extract obtained after a 7h-maceration performed at RT were compared to those obtained respectively with plant/PG ratios of 1/15 (
w/
w) and 1/20 (
w/
w) (
Figure 6).
Independent of the plant/PG ratio, the corresponding extraction yield was roughly similar (≈80%); however, the extract obtained using the plant/PG ratio of 1/10 (w/w) displays the best bioactivities. Therefore, the plant/PG ratio of 1/10 (w/w) was used for further optimisation of the extraction parameters.
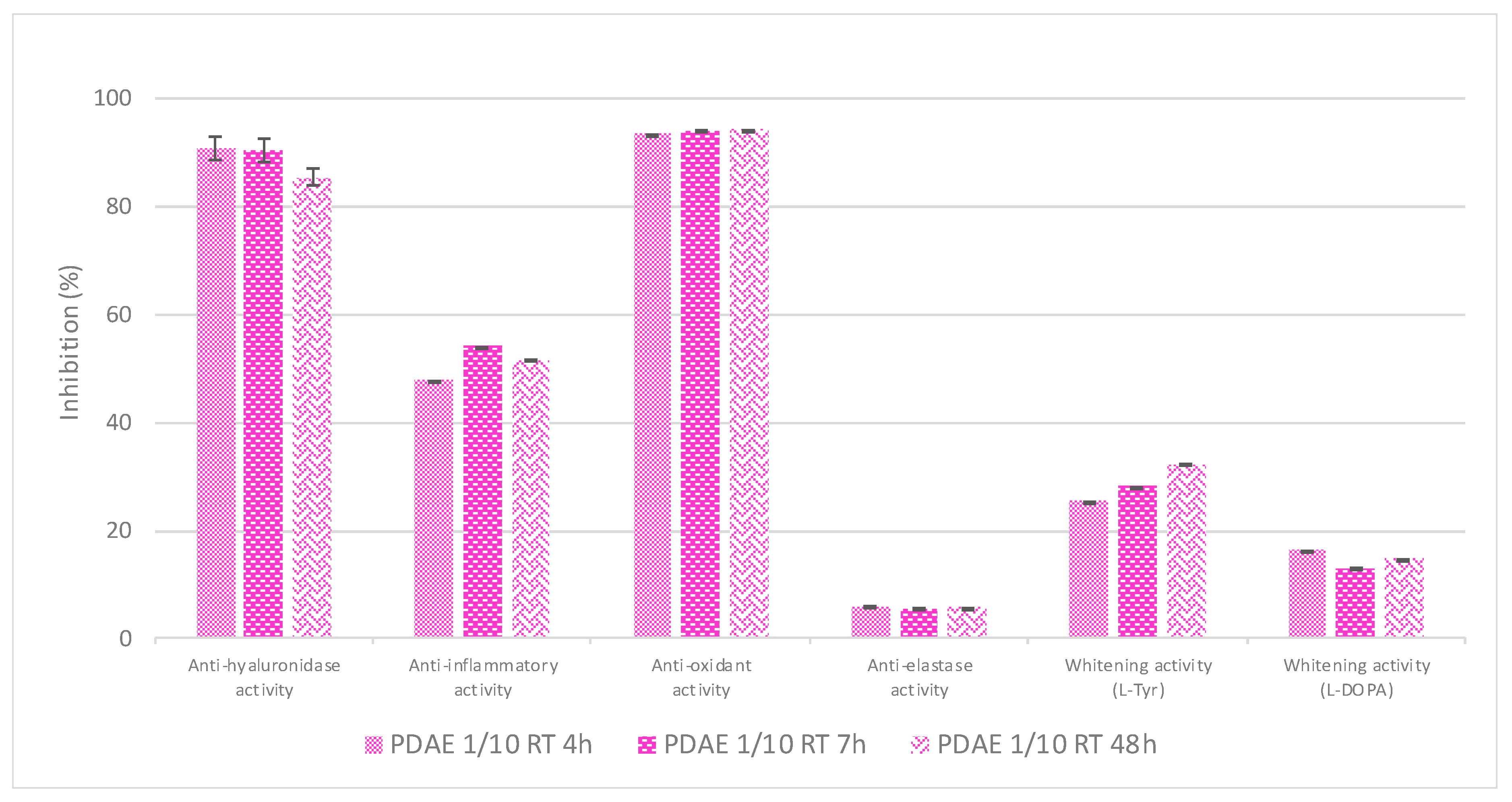
Then, the effect of the duration of the contact between the dried plant material and the PG on the resultant extracts’ bioactivities was evaluated (
Figure 7). No matter this duration, the extraction yields were similar (≈73–74%) and all three extracts displayed very interesting antioxidant and anti-hyaluronidase activities. Some promising anti-inflammatory activity was also found. However, given the fact that a long extraction process would be less interesting from an economical point of view, one can conclude that maceration times of between 4 h and 7 h are appropriate.
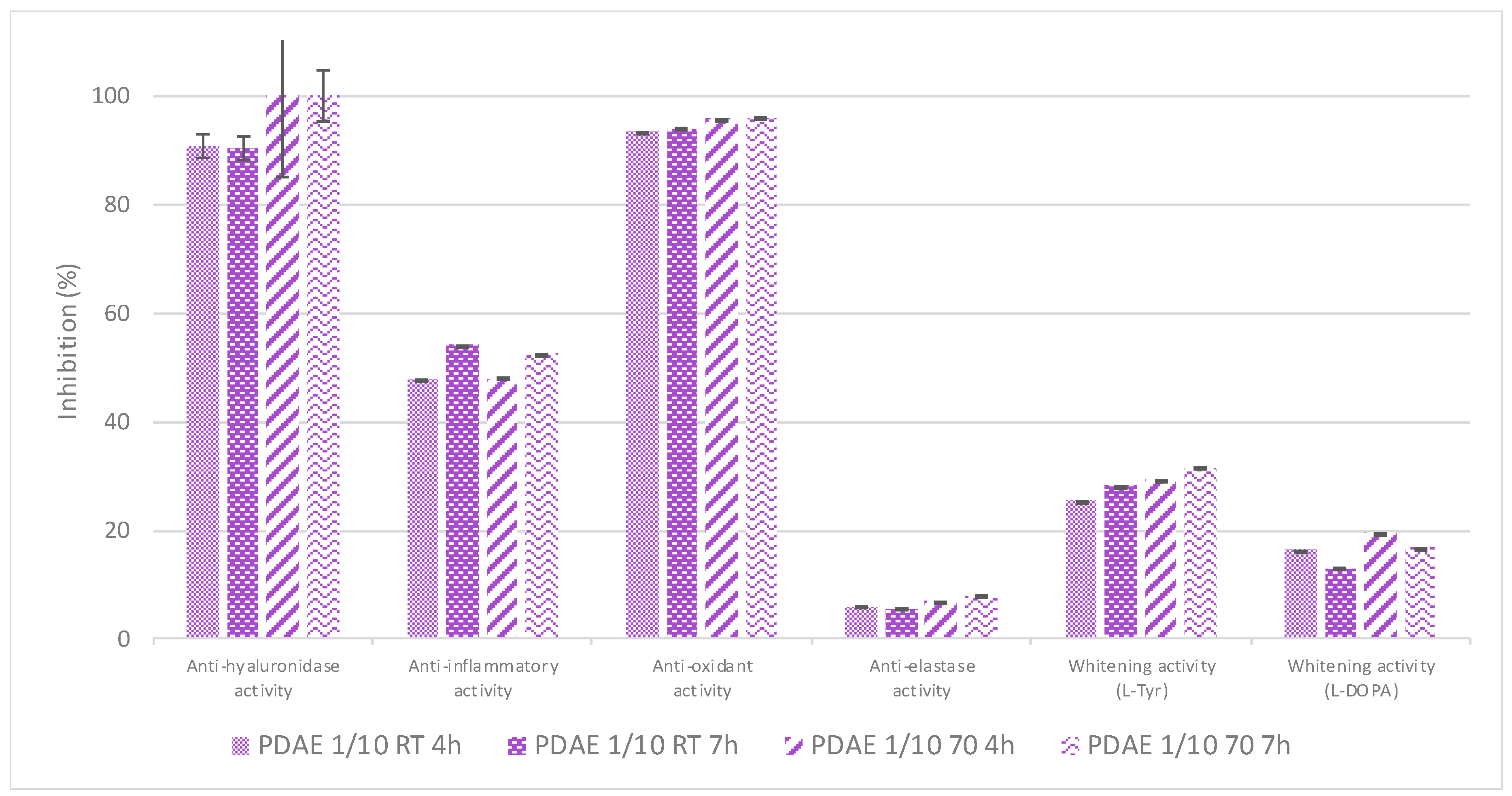
Finally, the effect of the temperature at which the maceration was performed was evaluated: to do so, both 4 h- and 7 h-macerations were performed either at RT or at 70 °C, respectively, using a plant/PG ratio of 1/10 (
w/
w) and the resultant extracts’ bioactivities were evaluated using
in vitro assays (
Figure 8).
No significant changes were observed in terms of bioactivities between extracts obtained after 4h- and 7h-macerations (the 7 h-ones were only slightly more active, particularly against lipoxygenase, e.g., anti-inflammatory activity), which were either performed at RT or at 70 °C. However, the application of heating induced a significant energetic consumption that brought no added value to the resultant extract in terms of bioactivity; further experimentation will therefore be performed at RT, which is also more ecologically relevant for a future process scale-up.
3.2.2. Impact of the Ingredient’s Colour
Solvents used to extract metabolites from a plant or a by-product also extract the molecules responsible for the latter’s colour, while odourlessness and colourlessness are preferable for cosmetic purposes [
35,
36,
39] and need therefore to be submitted to further processing, including notably a discoloration procedure. Such a discoloration can be achieved
via adsorption of compounds responsible for the colour on activated carbon: the powdered activated carbon is easily added to the liquid extract and removed by settling and filtration. However, such adsorption is not selective and active molecules can be retained on the activated carbon, implying a consequent loss of activity of the resulting ingredient [
9]. Some loss of ingredient mass was also reported in some cases after activated carbon’s elimination [
40]. Many time-consuming trials are often necessary to determine the appropriate discoloration parameters (varying activated carbon/ingredient (
w/
w) ratio, duration of the contact between the activated carbon and the ingredient, etc.). Discoloration attempts using molecular distillation could also be performed but imply the cost afferent to this equipment.
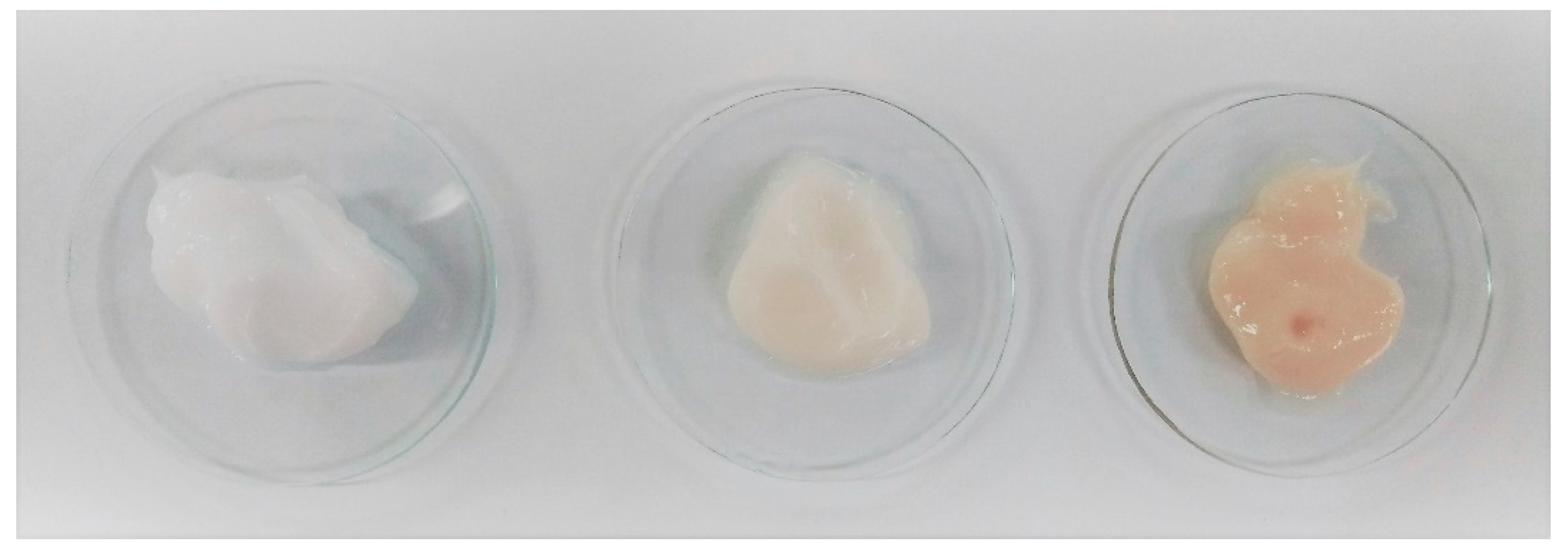
To avoid such losses of ingredient’s mass and activity, some preliminary formulation trials have been performed to establish whether this discoloration procedure is mandatory in the precise case of the PDL ingredient; this ingredient was incorporated at 1% and 4.5% in a typical oil-in-water (O/W) emulsion to observe its impact on the finished products’ aspect (
Figure 9). The use of the non-discoloured ingredient only resulted in a slight beige coloration, which is convenient for cosmetic formulations.
3.3. Green Liquid Cosmetic Ingredient
To develop an even greener ingredient, consistent with the notions of naturality and circular economy mentioned earlier, these optimized maceration conditions (7 h, RT, plant/PG ratio 1/10
w/
w) were then transposed to maceration assays using several green glycols issued from the biomass obtained from different suppliers. The bioactivities of the resultant extracts (subsequently named gPDL standing for green PDL extract) were assessed using
in vitro assays and compared to the ones with the corresponding PDL extract obtained using conventional PG (
Table 4). Apart from green glycol 2 that presented some anti-hyaluronidase activity, none of the green glycols tested shared responsibility in the bioactivities found.
Extraction trials with green glycols were obtained with extraction yields similar to the ones calculated for PDL obtained with conventional PG (≈73–74 %). All the seven gPDL displayed anti-hyaluronidase and anti-oxidant activities like the ones of conventional PDL; gPDL6 displayed some additional interesting anti-inflammatory activity.
All the green glycols tested here are therefore convenient solvents to obtain a greener version of this ingredient based on P. domestica leaves extract. The final decision about the green PG that will be used at the industrial scale will correspond to the best compromise between the bioactivities of the resultant ingredient and more trivially, the green glycols’ prices per kilogram.
3.4. HPLC Analysis of Liquid Ingredients
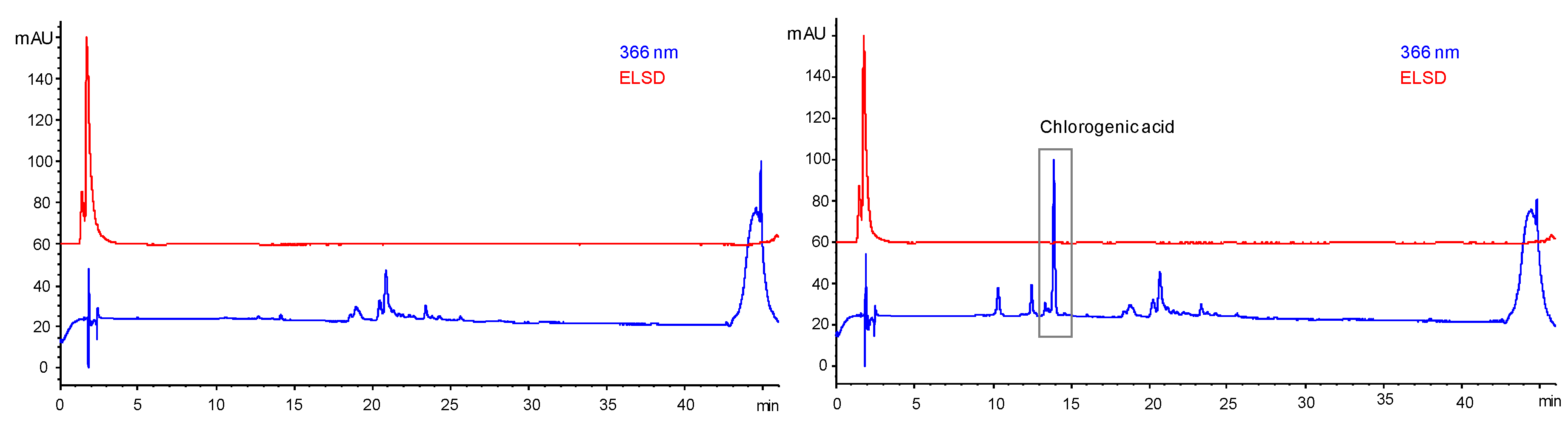
HPLC analysis of such liquid ingredients for compositional or stability purposes revealed themselves to be useless given the prevalence of the propylene glycol signal masking the extracted molecules’ signals. No experimental methodology developed to remove the glycol and to gain access to the extracted molecules could be found in the literature, so we had to develop such a protocol. SPE was selected to achieve this PG removal and several parameters had to be settled on to obtain a convenient PG elimination (sorbent type, saturation volume, etc.). The closest access we had to the chemistry of the plum leaves’ extract was the HPLC profile obtained for the ethanolic P. domestica leaves’ extract, which is not completely representative of the chemical profile of the PG extract as the different polarities of both solvents enable the extraction of various families of molecules, a fact evidenced by the bioactivity discrepancies observed between both extracts. By extrapolation, given the nature of the families of compounds identified in this extract, two cartridges were tested (Oasis HLB6cc, Waters, Guyancourt, Ile-de-France, France and Strata XL, Phenomenex, Torrance, California, USA). Both were composed of a reversed phase polymeric sorbent that strongly retained neutral, acidic, and basic compounds and gave similar results. For convenience, further optimisation of our SPE methodology was then achieved using Oasis HLB6cc cartridges available at the laboratory.
After proper conditioning and equilibration steps, saturation of the Oasis HLB6cc cartridge was assessed
via sequential addition on the cartridge of the liquid ingredient obtained
via maceration of
P. domestica leaves in PG, properly diluted in water to lower its viscosity, 0.5 mL at a time. Regular HPLC analysis was performed to monitor the saturation of the cartridge that was supposed to be reached when
P. domestica extracted molecules appeared in the subsequent HPLC chromatogram. HPLC analysis of the effluent’s composition enabled the identification of
P. domestica extracted molecules (the major one being identified as chlorogenic acid) in the effluent as soon as 3 mL of the ingredient were loaded on the cartridge, indicating its saturation (
Figure 10).
Once the volume of the ingredient necessary to saturate the cartridge was determined, we had to establish the volume necessary to elute all the molecules retained on the cartridge. To do so, elution of the sample was achieved by adding MeOH/DCM (1/1 v/v), 1 mL at a time; we found that the addition of 4 mL of MeOH/DCM (1/1 v/v) ensured the complete elution of the compounds retained on the cartridge.
Hence the appropriate SPE conditions to gain access to the extracted molecules using Oasis HLB6cc cartridges consisted of a load of 2.5 mL of the ingredient on the cartridge, and after removal of the glycol, in the successive use of 4 mL of MeOH/DCM (1/1 v/v) to collect the molecules retained on the cartridge.
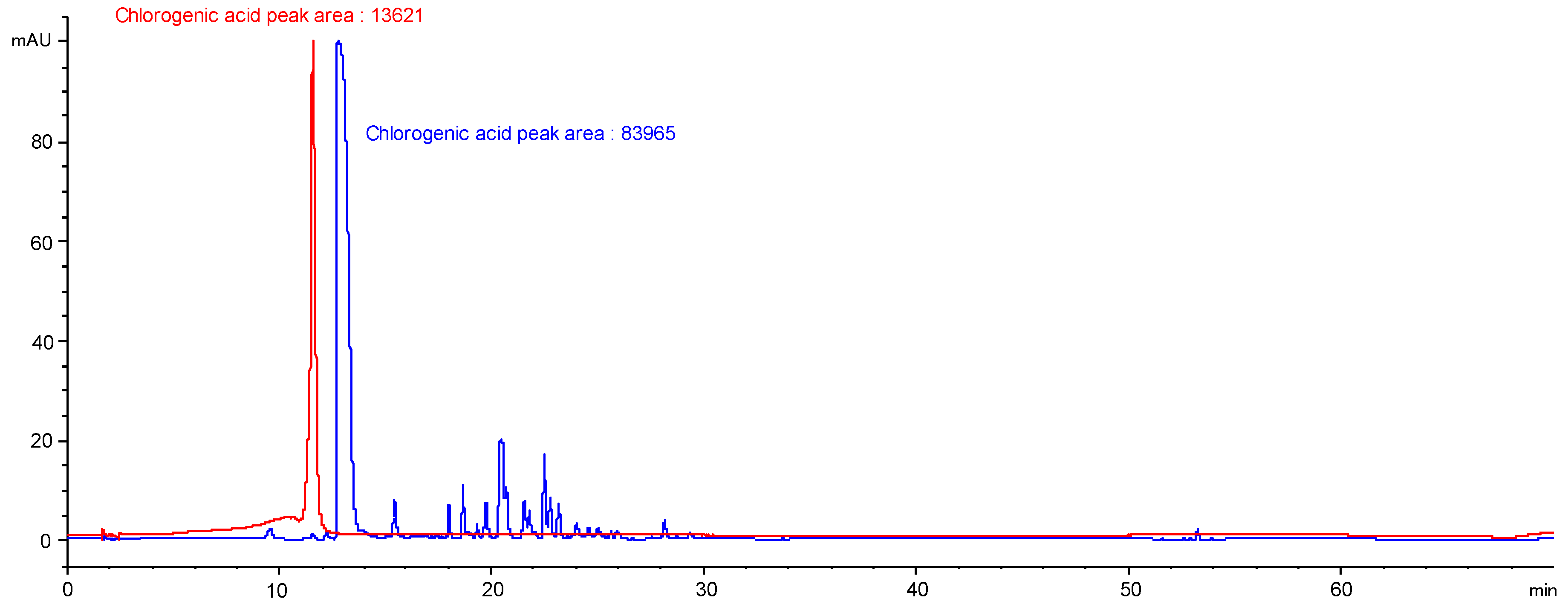
To validate this methodology, a solution of chlorogenic acid (5 mg/mL in MeOH) was analysed using HPLC and the area of the chlorogenic acid peak was determined. In parallel, the PG ingredient diluted in water (1/3) was supplemented with 12.5 mg of chlorogenic acid to reach the final concentration of 5 mg/mL and loaded on a SPE cartridge. Elution was performed as determined and the resulting extract was concentrated and analysed using HPLC in the same analytical conditions. The area of the chlorogenic acid peak was determined and compared to the one determined previously (
Figure 11); one can conclude that this methodology enabled a signal amplification by a factor of 6. Such a procedure enabling the removal of glycol and the access to the extracted molecules is essential to (a) get a glimpse on the molecular composition of the ingredient obtained and identify the molecule responsible for the bioactivities evidenced; and to (b) ensure the stability monitoring of the ingredient’s composition over time, which is a mandatory pre-requisite before its commercialisation. In fact, it is absolutely necessary to certify the post-manufacture stability of a cosmetic ingredient and to establish its appropriate conditions of storage and use (type of container in which it may be stored, maximum duration of storage, ideal storage temperature, etc.) to share with customers, namely the finished cosmetic products’ formulators.
3.5. Stability Testing
Accelerated stability testing was then undertaken to ensure that the liquid ingredient developed meets the intended quality standards when stored under specific conditions. To do so, the P. domestica ingredient was stored in a glass vial at 42 °C, and weekly monitoring will be undertaken to evaluate critical aesthetic properties (changes in colour and odour), and to track any change in its chemical composition by HPLC-DAD-ELSD. Those stability assays are not over, but so far, i.e., three weeks after their beginning, no alteration of the ingredient was noticed.
Once the definite formulation of the ingredient has been determined, further stability assays will be performed by putting the finished product in an aging chamber to monitor the potential changes that could take place from the initial formulation to the final consumer’s everyday use of the product.
4. Discussion and Conclusions
This article presents an integrated strategy to develop innovative natural cosmetic actives to fully satisfy green-minded consumers by using agricultural by-products as starting raw material, hence placing notions of naturality and circular economy at the very centre of this kind of design. Such a conversion of by-products, originally heading for destruction, into high value raw materials presents benefits for various actors of the integrated supply chain generated. In fact, it represents a waste management solution for the agri-food sector and brings more sourcing transparency and sustainability to the cosmetic sector, while constituting a fantastic opportunity for both actors to communicate about their environmentally-friendly actions.
However, before a formulator will consider using such a natural cosmetic active in an actual finished-product formulation, the ingredient must still pass several crucial steps: its efficacy, quality, shelf life and safety/tolerability must be controlled prior to being launched on the market. In fact, the safety of finished cosmetic products is based on the safety of the individual ingredients it contains. Even if not mandatory, it is highly recommended to assess the stability of ingredients over time to establish that no chemical changes generating undesired molecules (harmful molecules, compounds responsible for an unwanted colour or odour, etc.) takes place. The objective is to ensure that the challenged ingredient preserves its physical and chemical integrities, its microbiological qualities and functionalities under appropriate storage conditions [
41]. No method enabling such an assessment exists for liquid cosmetic ingredients directly developed in glycolic solvents. To determine that the liquid ingredients developed during this project meets the intended quality standards, a simple SPE method was developed to eliminate their glycol content to gain access to their molecular composition using HPLC analysis. This method can easily be transferred with some eventual adjustments (type of sorbent, washing solvent, etc.) to any type of natural cosmetic ingredients developed using glycolic solvent, whether the raw material consists of plant or an agri-food by-product.
The toxicological assessment of the cosmetic ingredient is mandatory before its commercial launch. Regularly revised guidelines regarding all aspects of safety evaluation of cosmetic substances are published by the Scientific Committee on Consumer Safety (SCCS) at the European level, while the U.S. Food and Drug Administration (FDA) guarantees the vigilance about worldwide regulation compliance [
42]. A thorough examination of the literature available about the species studied usually already brings some clues and might lead to its abandonment at the very beginning of the development if too many toxicological risks are identified. If nothing is reported in the literature, the ingredient development is undertaken, and the proper toxicology assessment is usually performed at the very end of the process for cost reasons. However, this assessment is more and more integrated directly into the cosmetic ingredient R&D process to avoid the unpleasant discovery of some toxicity issue at the end of the process, which could lead to the restriction or even the abandonment of the ingredient’s use due to safety concerns [
43]. It is generally recommended to at least assess the potency of the ingredient to induce some skin or ocular irritability [
44,
45], skin sensitization [
46] and phototoxicity [
47]; some additional mutagenicity, carcinogenicity, reprotoxicity assays, etc., might complete the safety assessment of a cosmetic active.
Even if no pesticides were used on the crop from where the starting material comes from, the presence of pesticides, and more widely, of any contaminant (herbicides, pollutants, etc.) in the final cosmetic ingredient cannot be ruled out. Analysis of these residues must be undertaken using GC-MS/MS and LC-MS/MS screening via multi-residue analyses before commercialisation of the finished cosmetic ingredient.
Therefore, the development of new natural ingredients for cosmetics is a long-term and not always rewarding procedure but using agri-food by-products as a starting material opens new innovation pathways.