From Mice to Men: An Evolutionary Conserved Breakdown of the Epidermal Calcium Gradient and Its Impact on the Cornified Envelope
Abstract
1. Introduction
2. Materials and Methods
3. Results
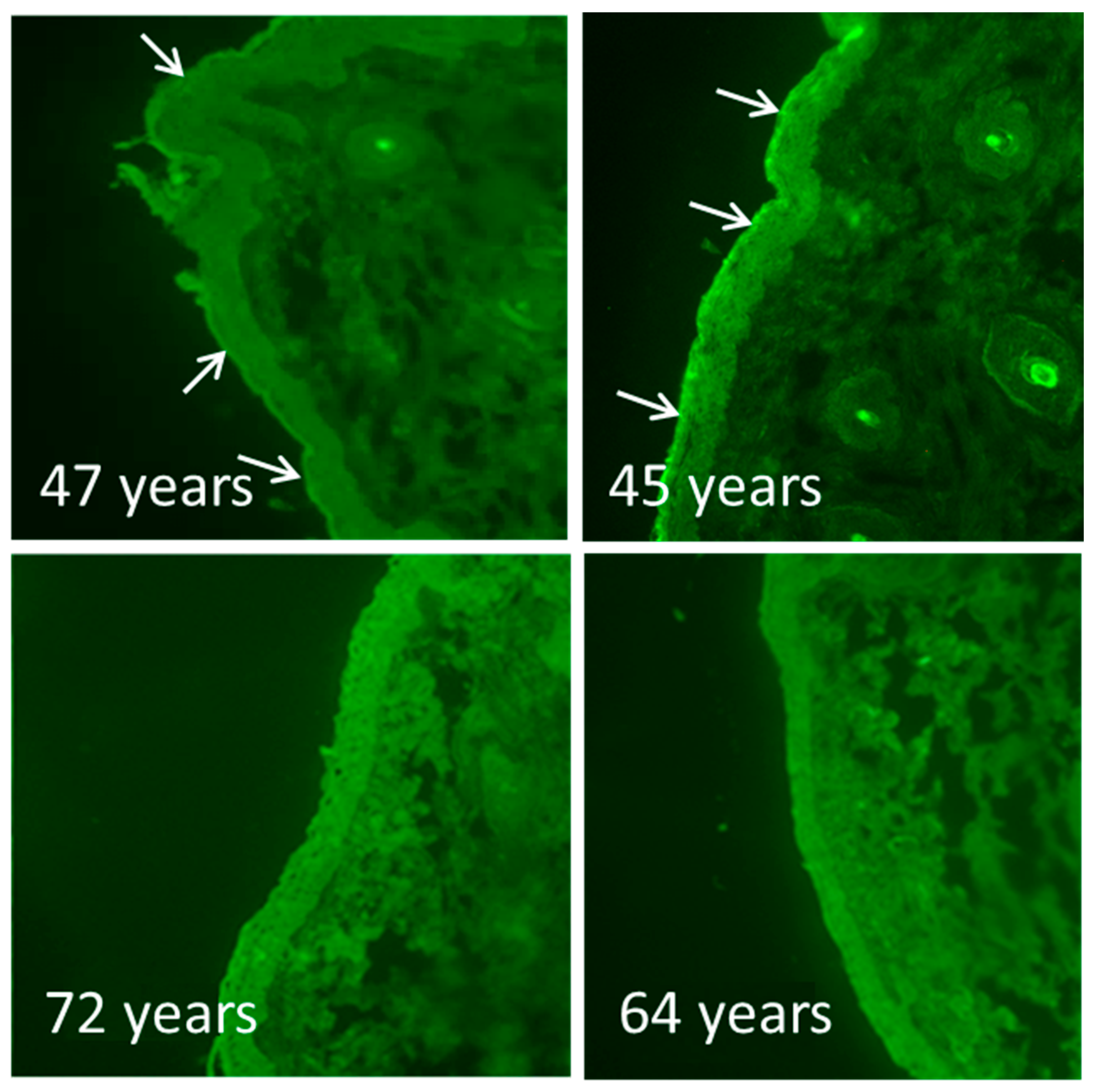
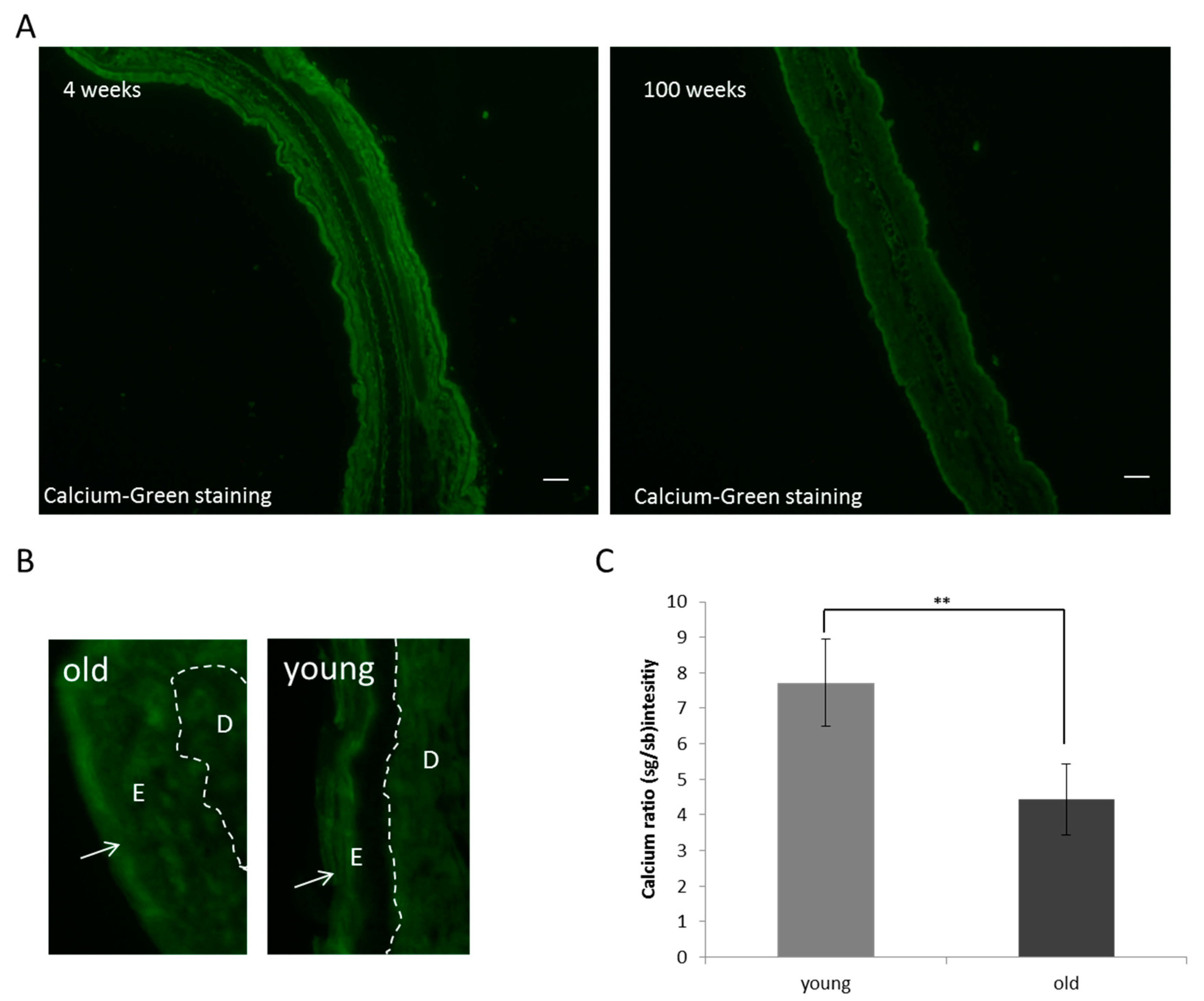
3.1. Breakdown of the Epidermal Calcium Gradient
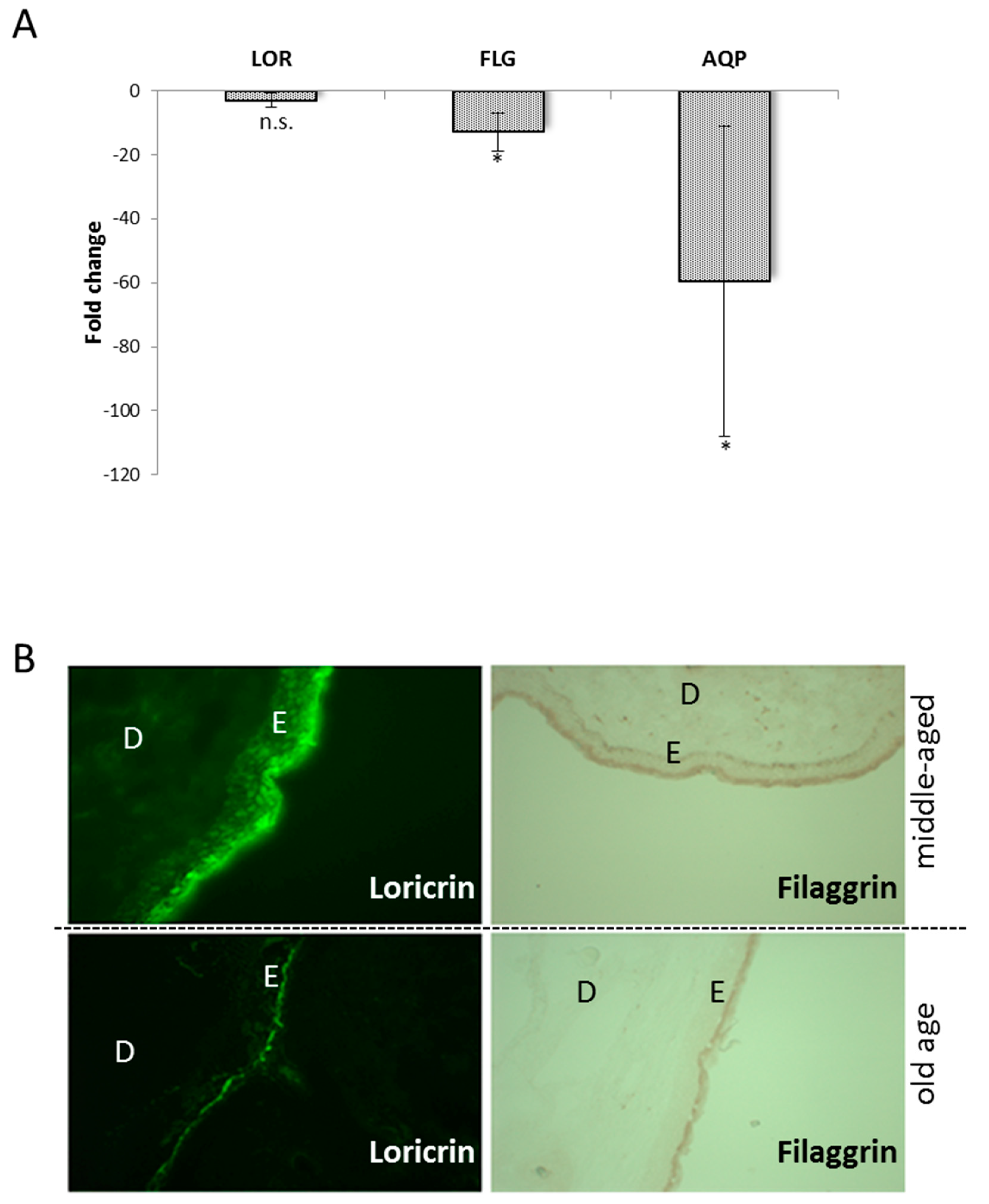
3.2. The Impact of the Collapsing Calcium Gradient on the Cornified Envelope Conposition
3.3. Breakdown of the Epidermal Calcium Gradient in Mice
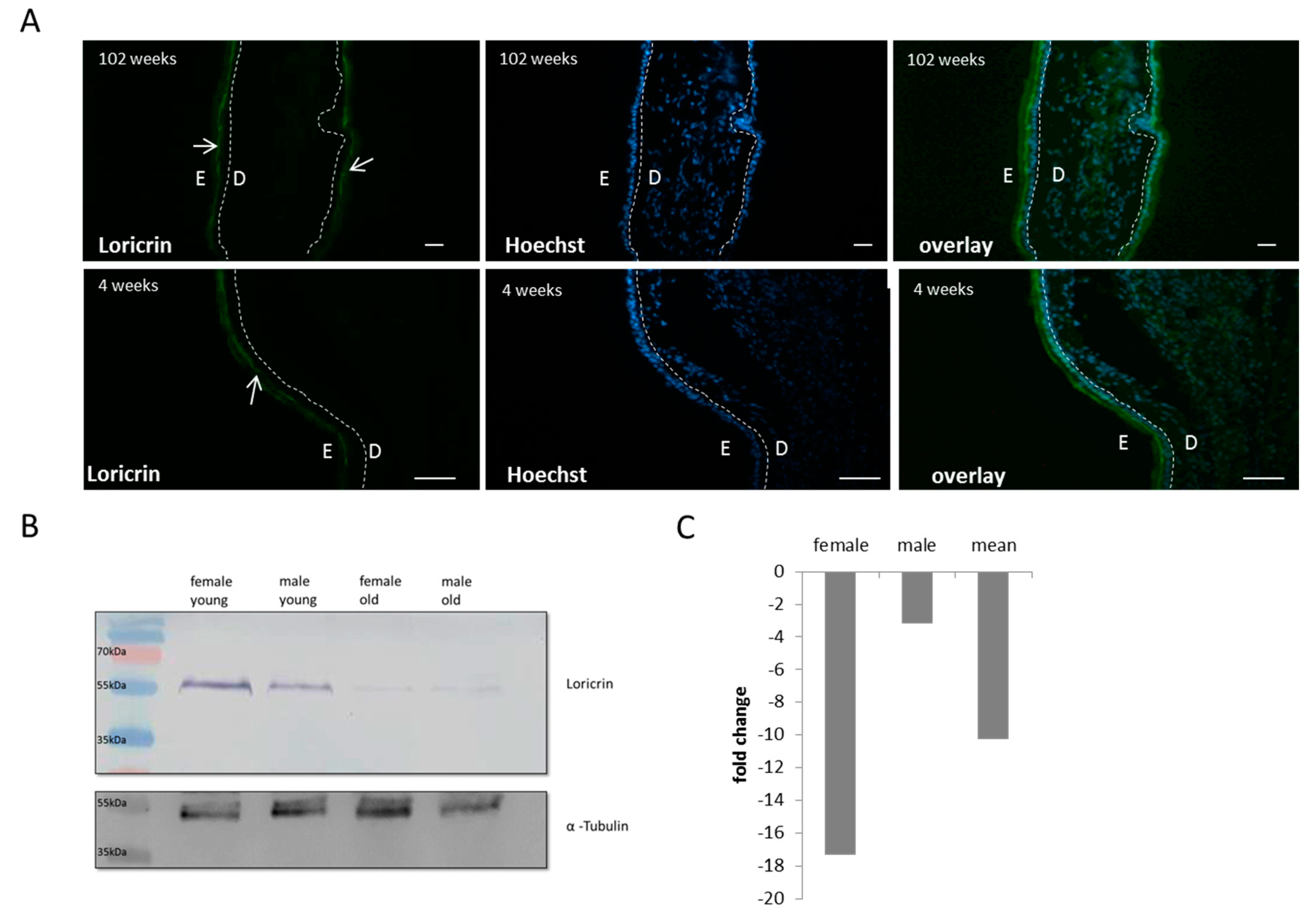
3.4. Altered Cornified Envelope Composition
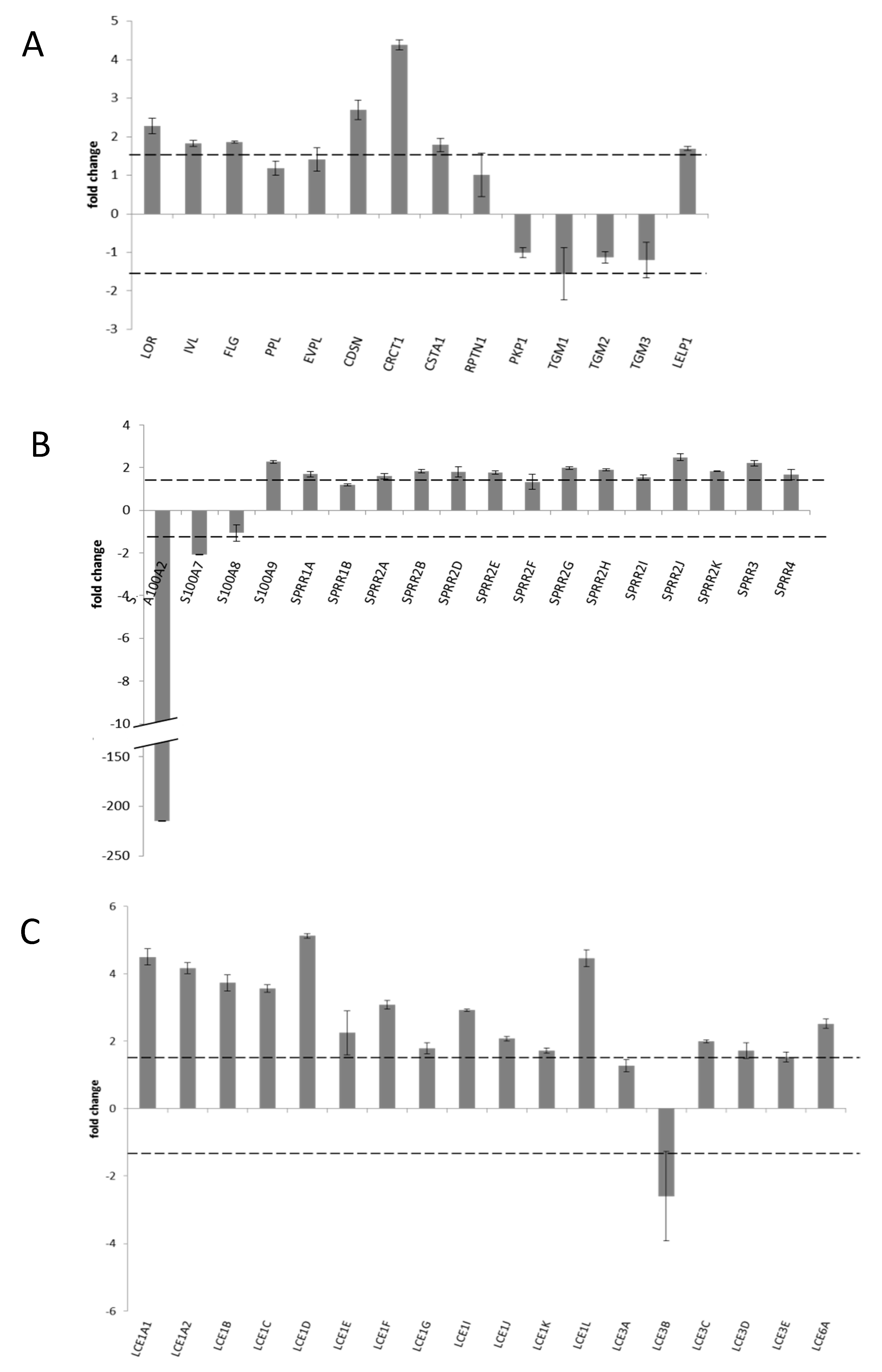
3.5. Transcriptional Profile of CE Specific Genes
4. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Nemes, Z.; Steinert, P.M. Bricks and mortar of the epidermal barrier. Exp. Mol. Med. 1999, 31, 5–19. [Google Scholar] [CrossRef] [PubMed]
- Marekov, L.N.; Steinert, P.M. Ceramides are bound to structural proteins of the human foreskin epidermal cornified cell envelope. J. Biol. Chem. 1998, 273, 17763–17770. [Google Scholar] [CrossRef] [PubMed]
- Streubel, M.K.; Rinnerthaler, M.; Bischof, J.; Richter, K. Changes in the Composition of the Cornified Envelope during Skin Aging: A Calcium Centric Point of View. In Textbook of Aging Skin; Farage, M.A., Miller, K.W., Maibach, H.I., Eds.; Springer: Berlin/Heidelberg, Germany, 2017; pp. 265–284. [Google Scholar]
- Rinnerthaler, M.; Streubel, M.K.; Bischof, J.; Richter, K. Skin aging, gene expression and calcium. Exp. Gerontol. 2015, 68, 59–65. [Google Scholar] [CrossRef] [PubMed]
- Raymond, A.A.; Gonzalez de Peredo, A.; Stella, A.; Ishida-Yamamoto, A.; Bouyssie, D.; Serre, G.; Monsarrat, B.; Simon, M. Lamellar bodies of human epidermis: Proteomics characterization by high throughput mass spectrometry and possible involvement of clip-170 in their trafficking/secretion. Mol. Cell. Proteom. 2008, 7, 2151–2175. [Google Scholar] [CrossRef] [PubMed]
- Kalinin, A.; Marekov, L.N.; Steinert, P.M. Assembly of the epidermal cornified cell envelope. J. Cell. Sci. 2001, 114, 3069–3070. [Google Scholar] [PubMed]
- Rinnerthaler, M.; Bischof, J.; Streubel, M.K.; Trost, A.; Richter, K. Oxidative stress in aging human skin. Biomolecules 2015, 5, 545–589. [Google Scholar] [CrossRef] [PubMed]
- Rinnerthaler, M.; Duschl, J.; Steinbacher, P.; Salzmann, M.; Bischof, J.; Schuller, M.; Wimmer, H.; Peer, T.; Bauer, J.W.; Richter, K. Age-related changes in the composition of the cornified envelope in human skin. Exp. Dermatol. 2013, 22, 329–335. [Google Scholar] [CrossRef] [PubMed]
- Lener, T.; Moll, P.R.; Rinnerthaler, M.; Bauer, J.; Aberger, F.; Richter, K. Expression profiling of aging in the human skin. Exp. Gerontol. 2006, 41, 387–397. [Google Scholar] [CrossRef] [PubMed]
- Untergasser, A.; Cutcutache, I.; Koressaar, T.; Ye, J.; Faircloth, B.C.; Remm, M.; Rozen, S.G. Primer3-new capabilities and interfaces. Nucleic Acids Res. 2012, 40. [Google Scholar] [CrossRef] [PubMed]
- Yoo, J.; Omori, M.; Gyarmati, D.; Zhou, B.H.; Aye, T.; Brewer, A.; Comeau, M.R.; Campbell, D.J.; Ziegler, S.F. Spontaneous atopic dermatitis in mice expressing an inducible thymic stromal lymphopoietin transgene specifically in the skin. J. Exp. Med. 2005, 202, 541–549. [Google Scholar] [CrossRef] [PubMed]
- Boury-Jamot, M.; Daraspe, J.; Bonte, F.; Perrier, E.; Schnebert, S.; Dumas, M.; Verbavatz, J.M. Skin aquaporins: Function in hydration, wound healing, and skin epidermis homeostasis. Handb. Exp. Pharmacol. 2009, 205–217. [Google Scholar] [CrossRef]
- Korge, B.P.; IshidaYamamoto, A.; Punter, C.; DoppingHepenstal, P.J.C.; Iizuka, H.; Stephenson, A.; Eady, R.A.J.; Munro, C.S. Loricrin mutation in vohwinkel’s keratoderma is unique to the variant with ichthyosis. J. Investig. Dermatol. 1997, 109, 604–610. [Google Scholar] [CrossRef] [PubMed]
- Ishida-Yamamoto, A.; Takahashi, H.; Iizuka, H. Loricrin and human skin diseases: Molecular basis of loricrin keratodermas. Histol. Histopathol. 1998, 13, 819–826. [Google Scholar] [PubMed]
- Koch, P.J.; de Viragh, P.A.; Scharer, E.; Bundman, D.; Longley, M.A.; Bickenbach, J.; Kawachi, Y.; Suga, Y.; Zhou, Z.J.; Huber, M.; et al. Lessons from loricrin-deficient mice: Compensatory mechanisms maintaining skin barrier function in the absence of a major cornified envelope protein. J. Cell. Biol. 2000, 151, 389–400. [Google Scholar] [CrossRef] [PubMed]
- Denda, M.; Tomitaka, A.; Akamatsu, H.; Matsunaga, K. Altered distribution of calcium in facial epidermis of aged adults. J. Investig. Dermatol. 2003, 121, 1557–1558. [Google Scholar] [PubMed]
- LaCelle, P.T.; Lambert, A.; Ekambaram, M.C.; Robinson, N.A.; Eckert, R.L. In vitro cross-linking of recombinant human involucrin. Skin Pharmacol. Appl. 1998, 11, 214–226. [Google Scholar] [CrossRef] [PubMed]
- Kezic, S.; Jakasa, I. Filaggrin and skin barrier function. Curr. Probl. Dermatol. 2016, 49, 1–7. [Google Scholar] [PubMed]
- Choi, E.H.; Man, M.Q.; Xu, P.; Xin, S.J.; Liu, Z.L.; Crumrine, D.A.; Jiang, Y.J.; Fluhr, J.W.; Feingold, K.R.; Elias, P.M.; et al. Stratum corneum acidification is impaired in moderately aged human and murine skin. J. Investig. Dermatol. 2007, 127, 2847–2856. [Google Scholar] [CrossRef] [PubMed]
- Vermeij, W.P.; Alia, A.; Backendorf, C. Ros quenching potential of the epidermal cornified cell envelope. J. Investig. Dermatol. 2011, 131, 1435–1441. [Google Scholar] [CrossRef] [PubMed]
- Harman, D. Aging—A theory based on free-radical and radiation-chemistry. J. Gerontol. 1956, 11, 298–300. [Google Scholar] [CrossRef] [PubMed]
- Matsubara, D.; Niki, T.; Ishikawa, S.; Goto, A.; Ohara, E.; Yokomizo, T.; Heizmann, C.W.; Aburatani, H.; Moriyama, S.; Moriyama, H.; et al. Differential expression of s100a2 and s100a4 in lung adenocarcinomas: Clinicopathological significance, relationship to p53 and identification of their target genes. Cancer Sci. 2005, 96, 844–857. [Google Scholar] [CrossRef] [PubMed]
- Sudbrak, R.; Brown, J.; Dobson-Stone, C.; Carter, S.; Ramser, J.; White, J.; Healy, E.; Dissanayake, M.; Larregue, M.; Perrussel, M.; et al. Hailey-hailey disease is caused by mutations in ATP2C1 encoding a novel Ca2+ pump. Hum. Mol. Genet. 2000, 9, 1131–1140. [Google Scholar] [CrossRef] [PubMed]
| Gene | Orientation | Primer sequence 5′-3′ | Gene | Orientation | Primer sequence 5′-3′ |
|---|---|---|---|---|---|
| Loricrin | fwd rev | GTGGAAAGACCTCTGGTGGA TGGAACCACCTCCATAGGAA | SPRR2G | fwd rev | GGGAAGGCATTTTTCTGAGAC GGAACATCCGTGACACACAG |
| Filaggrin | fwd rev | TCAAACAGGAGGGACAGACC TATCCTCCCTGACCACTTGC | TGM2 | fwd rev | GCCAGAGAACTGGGAGTCAG GGGTACAGGTCTGGTGCAGT |
| Involucrin | fwd rev | CCAGTGAAAGGAGACGTGCT TATGGGTGAGTAGGCCAGCT | CSTA1 | fwd rev | ACATTTGAGGCAACCCCATA GATGGTCTTCAAAAGCAACAAA |
| SPRR2Jps | fwd rev | TTTGCCAGCCTCCTCTTGTG TTCTGCTGGCATGACTGAGG | RPTN | fwd rev | GGGATTTTGGACTTTGGTGA CCATCAATGGAGGAAGAGGA |
| LCE1D | fwd rev | AAAGTGGTGCAGAAAGAACACA GATGCAAAGAGGCATTGTCAT | PKP1 | fwd rev | AGGGGAGCACTTAAGGCAAT GCCTACAGCAGTCTCCCTTG |
| LCE3C | fwd rev | AGGAGCTCCAGTTCCTGTGA AGAAGACTCTCCAGGATCAGAT | SPRR1A | fwd rev | GCTGAGGCTGCTGTCTATCC GGGCAATGTTAAGAGGCTCA |
| S100A9 | fwd rev | ACCAGGACAATCAGCTGAGC TAGACTTGGTTGGGCAGCTG | SPRR1B | fwd rev | GCTGAGCCTCAATTTGCATT GCTACCAAGTTCTCGGGTGA |
| TGM1 | fwd rev | TCAGATGCTGGAGGTGACAG CCCAGTCTTCCTGTCTGAGC | SPRR2A | fwd rev | GGATCCACCTGTCTCTGCTC TATTACTGCCTCCCCTCCTG |
| TGM3 | fwd rev | CAAAACTCAAACCCCCTCAA CAGGTGTGTGCTCAGTTGCT | SPRR2B | fwd rev | CTGTCTCACTGGCTCACCAA AGGAAAGCAGCAGGAGATCA |
| CDSN | fwd rev | CACACCCTACCAGACCCATT ATCCAGAGGGAACTGGGAGT | SPRR2D | fwd rev | AAGTGCCCACCTAAGAGCAA GGGGAAACATCTTCCCTGTT |
| PPL | fwd rev | GCTGCTGACCTTCCTTCTTG TGTTGGTGCCTGTCTTTCTG | SPRR2E | fwd rev | AAGGCTGTCATGGGTGAATC CTGAGCATTTGGGACACTGA |
| EVPL | fwd rev | GCCATGCTAGAAGGCTATCG GTGGACACTCAGGAGGAGGA | SPRR2F | fwd rev | TCAGGACAGGGGAAAGAGAA CCCCTTTACAGCAGCAAGAT |
| SPRR3 | fwd rev | CAGACCCTTGAGGAGCTGAC AGAGGCTCTGGAGACACCTT | SPRR4 | fwd rev | TCCCATCAGCATCAGAATCA TGCTGTGCAGGACACTTCTC |
| LCE1A1 | fwd rev | GCTGCTGACCTGAGCAACTA TGTCCCCAAGAAGACAAACC | S100A8 | fwd rev | GGAAATCACCATGCCCTCTA TGGCTGTCTTTGTGAGATGC |
| LCE1A2 | fwd rev | GCTGCTGACCTGAGCAACTA GGTCCTTCAAAAGGGGACAT | S100A7 | fwd vrev | CAGGCAGTCTCTCATCACCA ACCCAGAACCATGACCTGAG |
| LCE1B | fwd rev | CCCTTGCACTTCAGACAAGC TGTTTGCAAGGGGAGACTTT | CRCT1 | fwd rev | GGCCTAGGAGTTTGGTTTCC ACACTGCTGTGCACAACCTC |
| LCE1C | fwd rev | GCTGACAAGTGCCATGAAGA GGGATTAGGTCATTGGGACA | S100A2 | fwd rev | CCAGTCAAGAGGACGAGAGG ACCTGCTGGTCACTGTCCTT |
| LCE3E | fwd rev | GCGGAGACAAGGAATTTCAG ACCAGGGAATGAGGACTGTG | LELP1 | fwd rev | TGTCCTTCTCCATGCCTACC CCACCACAAAAGCAAATCCT |
| LCE3D | fwd rev | GACCTGGAACATGATGCTGA TCCACAGGAAAGATGGGAAC | SPRR2H | fwd rev | ATCTTCCCTCCAAAGCCATT CTGAGCATCTGGGACAGTGA |
| LCE3C | fwd rev | AGGAGCTCCAGTTCCTGTGA AGAAGACTCTCCAGGATCAGAT | SPRR2I | fwd rev | TGCCTCCATAGCAACAACTG CATTCTCTGCAGGCCCTTTA |
| LCE3B | fwd rev | AGCATCCTCAGACACGGACT CACTGTGCTGTGCTCTTCCT | SPRR2K | fwd rev | CCCTTCACAACCTCTCATGG CAAAGGAAGGACCATTTCCA |
| LCE3A | fwd rev | TTTGAGACAAAAGGGTTTGGA ACATGGTTGGACACAGGTGA | LCE1I | fwd rev | TGACCTGGGTGAGGAAGACT AATGGGCATTGTCGTTTCAT |
| LCE1E | fwd rev | GACAAATGGGACAGGAGGAA TGTAACAAAGGGGAAACATGAA | LCE1J | fwd rev | CAAAGTGTCCCCCAAAATGT GCGATGGAGAGATCTGTGGT |
| LCE6A | fwd rev | TGCTAAGAAACTGGGCACAA TGTTGAACTGAAAGGGTAGGC | LCE1K | fwd rev | TGTGGCAGTAGCCAACAGTC GGGTAGCAACATGGGAACAA |
| LCE1F | fwd rev | TGCTGCTGACCTGTATGAGG TGCAGAACATTTCACAGGAGA | LCE1L | fwd rev | GCTGACCTGGGTCATGAAG TTGGAATCACAGAAGGAGATGA |
| LCE1G | fwd rev | TCCTCATGAAATGACAATGACC GGCACAGTGTGTTTGAAGGA | Beta Actin | fwd rev | GTCCCTCACCCTCCCAAAAG CTCAGACCTGGGCCATTCAG |
| AQP3 | fwd rev | CCA CAG CTT AGG TTT GGA GC TCC CGG ATC CCT AAG ACT GT | - | - | - |
| Gene | Ø ∆Ct ys (n = 6) | SEM Ø ∆Ct ys (n = 6) | Ø ∆Ct os (n = 6) | SEM Ø ∆Ct os (n = 6) | p-Value (ys-os) |
|---|---|---|---|---|---|
| LOR | 5.03 | 0.3612882 | 3.8425 | 0.19493945 | 0.0133 |
| IVL | 2.74833333 | 0.22913182 | 1.8825 | 0.07959097 | 0.0040 |
| FLG | 3.765 | 0.12894336 | 2.8625 | 0.02568506 | 0.0037 |
| CDSN | 2.91333333 | 0.14708746 | 1.4825 | 0.25663149 | 0.0109 |
| CRCT1 | 3.20333333 | 0.22338619 | 1.0725 | 0.12823048 | 0.0009 |
| CSTA1 | 3.56666667 | 0.32272925 | 2.7275 | 0.17311284 | 0.0689 |
| TGM1 | 4.93333333 | 0.19329382 | 5.57416667 | 0.6776563 | 0.285 |
| LELP1 | 3.315 | 0.30997984 | 2.5525 | 0.04826144 | 0.0295 |
| A100A2 | 5.297 | 0.20829066 | 13.0441667 | 0.22215172 | 0.0001 |
| S100A7 | 1.41333333 | 0.14077662 | 2.46416667 | 0.01767767 | 0.0195 |
| S100A9 | 2.70666667 | 0.25035808 | 1.52083333 | 0.06124858 | 0.0106 |
| SPRR1A | 2.28666667 | 0.30113904 | 1.53083333 | 0.12780736 | 0.0421 |
| SPRR2A | 0.19833333 | 0.05606296 | −0.48083333 | 0.1255488 | 0.0710 |
| SPRR2B | 2.63833333 | 0.06172295 | 1.76416667 | 0.08603133 | 0.0060 |
| SPRR2D | 10.2283333 | 0.79166053 | 9.37583333 | 0.24859885 | 0.4486 |
| SPRR2E | 3.425 | 0.26437295 | 2.61083333 | 0.08327498 | 0.0763 |
| SPRR2G | 2.47666667 | 0.20021169 | 1.49083333 | 0.07315319 | 0.0550 |
| SPRR2H | 3.03 | 0.30980056 | 2.1175 | 0.04891887 | 0.0827 |
| SPRR2I | 2.41166667 | 0.2943708 | 1.77916667 | 0.10552119 | 0.1783 |
| SPRR2J | 2.975 | 0.5721997 | 1.66583333 | 0.16571813 | 0.0262 |
| SPRR2K | 5.63833333 | 0.50090335 | 4.75916667 | 0.01504623 | 0.2473 |
| SPRR3 | 2.89166667 | 0.48133062 | 1.75416667 | 0.12045631 | 0.0454 |
| SPRR4 | 5.84666667 | 0.67670259 | 5.10416667 | 0.24363851 | 0.3486 |
| LCE1A1 | 2.11666667 | 0.05635946 | −0.05083333 | 0.24534273 | 0.0001 |
| LCE1A2 | 2.36166667 | 0.09449427 | 0.3025 | 0.16478521 | 0.0004 |
| LCE1B | 2.725 | 0.12471634 | 0.82916667 | 0.24769773 | 0.0071 |
| LCE1C | 2.565 | 0.27731601 | 0.73416667 | 0.11885449 | 0.0005 |
| LCE1D | 2.35666667 | 0.32636253 | 0.00083333 | 0.06795628 | 0.0004 |
| LCE1E | 4.21666667 | 0.61893838 | 3.0525 | 0.65375943 | 0.2335 |
| LCE1F | 2.49833333 | 0.39387145 | 0.88083333 | 0.12817631 | 0.0085 |
| LCE1G | 3.31333333 | 0.60679829 | 2.4775 | 0.16788306 | 0.186 |
| LCE1I | 2.53 | 0.20341187 | 0.9875 | 0.0274621 | 0.0054 |
| LCE1J | 3.25 | 0.36124976 | 2.20083333 | 0.06133922 | 0.0268 |
| LCE1K | 3.42166667 | 0.3042226 | 2.64416667 | 0.0803724 | 0.1721 |
| LCE1L | 3.00666667 | 0.2834828 | 0.8525 | 0.25470844 | 0.0007 |
| LCE3B | 7.88333333 | 0.67345193 | 9.26083333 | 1.3296496 | 0.3363 |
| LCE3C | 3.23166667 | 0.28404078 | 2.23916667 | 0.04677072 | 0.0086 |
| LCE3D | 2.82 | 0.32581309 | 2.04083333 | 0.22607459 | 0.0744 |
| LCE3E | 2.67833333 | 0.28519243 | 2.06916667 | 0.14301806 | 0.1548 |
| LCE6A | 2.42 | 0.23960906 | 1.08916667 | 0.13181953 | 0.0018 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Streubel, M.K.; Neuhofer, C.; Bischof, J.; Steinbacher, P.; Russe, E.; Wechselberger, G.; Richter, K.; Rinnerthaler, M. From Mice to Men: An Evolutionary Conserved Breakdown of the Epidermal Calcium Gradient and Its Impact on the Cornified Envelope. Cosmetics 2018, 5, 35. https://doi.org/10.3390/cosmetics5020035
Streubel MK, Neuhofer C, Bischof J, Steinbacher P, Russe E, Wechselberger G, Richter K, Rinnerthaler M. From Mice to Men: An Evolutionary Conserved Breakdown of the Epidermal Calcium Gradient and Its Impact on the Cornified Envelope. Cosmetics. 2018; 5(2):35. https://doi.org/10.3390/cosmetics5020035
Chicago/Turabian StyleStreubel, Maria Karolin, Claudia Neuhofer, Johannes Bischof, Peter Steinbacher, Elisabeth Russe, Gottfried Wechselberger, Klaus Richter, and Mark Rinnerthaler. 2018. "From Mice to Men: An Evolutionary Conserved Breakdown of the Epidermal Calcium Gradient and Its Impact on the Cornified Envelope" Cosmetics 5, no. 2: 35. https://doi.org/10.3390/cosmetics5020035
APA StyleStreubel, M. K., Neuhofer, C., Bischof, J., Steinbacher, P., Russe, E., Wechselberger, G., Richter, K., & Rinnerthaler, M. (2018). From Mice to Men: An Evolutionary Conserved Breakdown of the Epidermal Calcium Gradient and Its Impact on the Cornified Envelope. Cosmetics, 5(2), 35. https://doi.org/10.3390/cosmetics5020035







