Mechanistic Understanding of Contact Allergy
Abstract
:1. Contact Dermatitis
2. Diagnosis of ACD
3. Chemical Reactivity and Immune System Activation
4. The Innate Molecular Immune Response to Contact Allergens
5. The Innate Cellular Immune Response to Contact Allergens
6. Identification of Contact Allergens by in Vitro Assays: Alternatives to Animal Testing
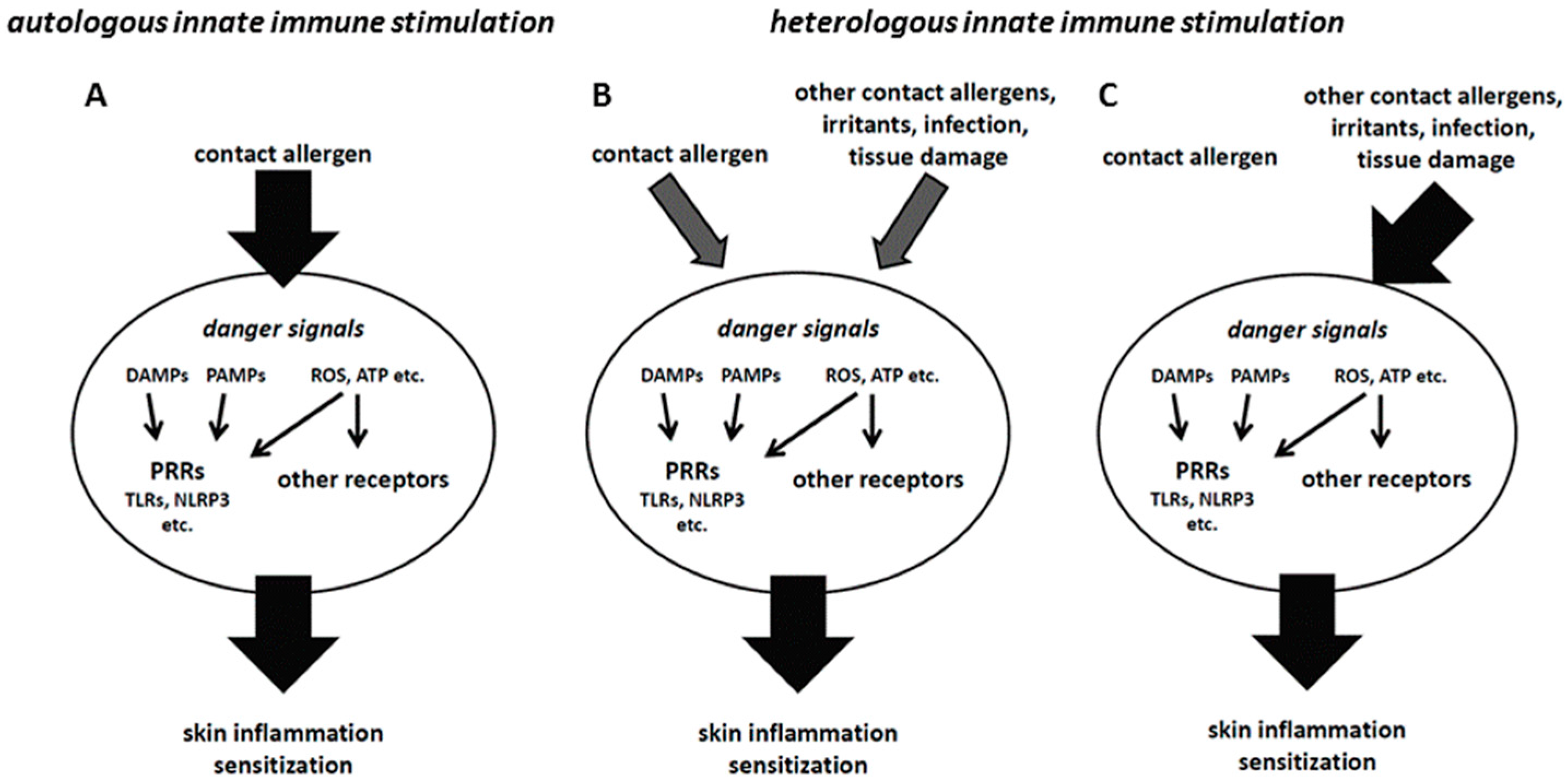
7. Heterologous Innate Immune Stimulation
8. Conclusions
Acknowledgments
Conflicts of Interest
Abbreviations
| ACD | allergic contact dermatitis |
| CHS | contact hypersensitivity |
| DC | dendritic cell |
| ICD | irritant contact dermatitis |
| NLR | NOD-like receptor |
| PAMP | pathogen associated molecular pattern |
| PRR | pattern recognition receptor |
| ROS | reactive oxygen species |
| TLR | Toll-like receptor |
References
- Karlberg, A.T.; Borje, A.; Duus Johansen, J.; Liden, C.; Rastogi, S.; Roberts, D.; Uter, W.; White, I.R. Activation of non-sensitizing or low-sensitizing fragrance substances into potent sensitizers—Prehaptens and prohaptens. Contact Dermat. 2013, 69, 323–334. [Google Scholar] [CrossRef] [PubMed]
- Peiser, M.; Tralau, T.; Heidler, J.; Api, A.M.; Arts, J.H.; Basketter, D.A.; English, J.; Diepgen, T.L.; Fuhlbrigge, R.C.; Gaspari, A.A.; et al. Allergic contact dermatitis: epidemiology, molecular mechanisms, in vitro methods and regulatory aspects. Current knowledge assembled at an international workshop at BfR, Germany. Cell. Mol. Life Sci. 2012, 69, 763–781. [Google Scholar] [CrossRef] [PubMed]
- Brasch, J.; Becker, D.; Aberer, W.; Bircher, A.; Kranke, B.; Jung, K.; Przybilla, B.; Biedermann, T.; Werfel, T.; John, S.M.; et al. Guideline contact dermatitis: S1—Guidelines of the German Contact Allergy Group (DKG) of the German Dermatology Society (DDG), the Information Network of Dermatological Clinics (IVDK), the German Society for Allergology and Clinical Immunology (DGAKI), the Working Group for Occupational and Environmental Dermatology (ABD) of the DDG, the Medical Association of German Allergologists (AeDA), the Professional Association of German Dermatologists (BVDD) and the DDG. Allergo J. Int. 2014, 23, 126–138. [Google Scholar] [PubMed]
- Holness, D.L. Occupational skin allergies: Testing and treatment (the case of occupational allergic contact dermatitis). Curr. Allergy Asthma Rep. 2014, 14. [Google Scholar] [CrossRef] [PubMed]
- Wiszniewska, M.; Walusiak-Skorupa, J. Recent Trends in Occupational Contact Dermatitis. Curr. Allergy Asthma Rep. 2015, 15. [Google Scholar] [CrossRef] [PubMed]
- Fyhrquist, N.; Lehto, E.; Lauerma, A. New findings in allergic contact dermatitis. Curr. Opin. Allergy Clin. Immunol. 2014, 14, 430–435. [Google Scholar] [CrossRef] [PubMed]
- De Groot, A.C. New Contact Allergens: 2008 to 2015. Dermatitis 2015, 26, 199–215. [Google Scholar] [CrossRef] [PubMed]
- Martin, S.F. Immunological mechanisms in allergic contact dermatitis. Curr. Opin. Allergy Clin. Immunol. 2015, 15, 124–130. [Google Scholar] [CrossRef] [PubMed]
- Vocanson, M.; Hennino, A.; Rozieres, A.; Poyet, G.; Nicolas, J.F. Effector and regulatory mechanisms in allergic contact dermatitis. Allergy 2009, 64, 1699–1714. [Google Scholar] [CrossRef] [PubMed]
- Martin, S.F.; Esser, P.R.; Schmucker, S.; Dietz, L.; Naisbitt, D.J.; Park, B.K.; Vocanson, M.; Nicolas, J.F.; Keller, M.; Pichler, W.J.; et al. T-cell recognition of chemicals, protein allergens and drugs: Towards the development of in vitro assays. Cell. Mol. Life Sci. 2010, 67, 4171–4184. [Google Scholar] [CrossRef] [PubMed]
- Goubier, A.; Vocanson, M.; Macari, C.; Poyet, G.; Herbelin, A.; Nicolas, J.F.; Dubois, B.; Kaiserlian, D. Invariant NKT cells suppress CD8+ T-cell-mediated allergic contact dermatitis independently of regulatory CD4+ T cells. J. Investig. Dermatol. 2013, 133, 980–987. [Google Scholar] [CrossRef] [PubMed]
- Dhingra, N.; Shemer, A.; Correa da Rosa, J.; Rozenblit, M.; Fuentes-Duculan, J.; Gittler, J.K.; Finney, R.; Czarnowicki, T.; Zheng, X.; Xu, H.; et al. Molecular profiling of contact dermatitis skin identifies allergen-dependent differences in immune response. J. Allergy Clin. Immunol. 2014, 134, 362–372. [Google Scholar] [CrossRef] [PubMed]
- Quaranta, M.; Knapp, B.; Garzorz, N.; Mattii, M.; Pullabhatla, V.; Pennino, D.; Andres, C.; Traidl-Hoffmann, C.; Cavani, A.; Theis, F.J.; et al. Intraindividual genome expression analysis reveals a specific molecular signature of psoriasis and eczema. Sci. Transl. Med. 2014, 6. [Google Scholar] [CrossRef] [PubMed]
- Martin, S.F. Allergic contact dermatitis: Xenoinflammation of the skin. Curr. Opin. Immunol. 2012, 24, 720–729. [Google Scholar] [CrossRef] [PubMed]
- Kaplan, D.H.; Igyarto, B.Z.; Gaspari, A.A. Early immune events in the induction of allergic contact dermatitis. Nat. Rev. Immunol. 2012, 12, 114–124. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, M.; Raghavan, B.; Muller, V.; Vogl, T.; Fejer, G.; Tchaptchet, S.; Keck, S.; Kalis, C.; Nielsen, P.J.; Galanos, C.; et al. Crucial role for human Toll-like receptor 4 in the development of contact allergy to nickel. Nat. Immunol. 2010, 11, 814–819. [Google Scholar] [CrossRef] [PubMed]
- Raghavan, B.; Martin, S.F.; Esser, P.R.; Goebeler, M.; Schmidt, M. Metal allergens nickel and cobalt facilitate TLR4 homodimerization independently of MD2. EMBO Rep. 2012, 13, 1109–1115. [Google Scholar] [CrossRef] [PubMed]
- Martin, S.F.; Dudda, J.C.; Bachtanian, E.; Lembo, A.; Liller, S.; Durr, C.; Heimesaat, M.M.; Bereswill, S.; Fejer, G.; Vassileva, R.; et al. Toll-like receptor and IL-12 signaling control susceptibility to contact hypersensitivity. J. Exp. Med. 2008, 205, 2151–2162. [Google Scholar] [CrossRef] [PubMed]
- Esser, P.R.; Wolfle, U.; Durr, C.; von Loewenich, F.D.; Schempp, C.M.; Freudenberg, M.A.; Jakob, T.; Martin, S.F. Contact sensitizers induce skin inflammation via ROS production and hyaluronic acid degradation. PLoS ONE 2012, 7, e41340. [Google Scholar] [CrossRef] [PubMed]
- El Ali, Z.; Gerbeix, C.; Hemon, P.; Esser, P.R.; Martin, S.F.; Pallardy, M.; Kerdine-Romer, S. Allergic skin inflammation induced by chemical sensitizers is controlled by the transcription factor Nrf2. Toxicol. Sci. 2013, 134, 39–48. [Google Scholar] [CrossRef] [PubMed]
- Weber, F.C.; Esser, P.R.; Muller, T.; Ganesan, J.; Pellegatti, P.; Simon, M.M.; Zeiser, R.; Idzko, M.; Jakob, T.; Martin, S.F. Lack of the purinergic receptor P2X7 results in resistance to contact hypersensitivity. J. Exp. Med. 2010, 207, 2609–2619. [Google Scholar] [CrossRef] [PubMed]
- Yasukawa, S.; Miyazaki, Y.; Yoshii, C.; Nakaya, M.; Ozaki, N.; Toda, S.; Kuroda, E.; Ishibashi, K.; Yasuda, T.; Natsuaki, Y.; et al. An ITAM-Syk-CARD9 signalling axis triggers contact hypersensitivity by stimulating IL-1 production in dendritic cells. Nat. Commun. 2014, 5. [Google Scholar] [CrossRef] [PubMed]
- Dudeck, A.; Dudeck, J.; Scholten, J.; Petzold, A.; Surianarayanan, S.; Kohler, A.; Peschke, K.; Vohringer, D.; Waskow, C.; Krieg, T.; et al. Mast Cells Are Key Promoters of Contact Allergy that Mediate the Adjuvant Effects of Haptens. Immunity 2011, 34, 973–984. [Google Scholar] [CrossRef] [PubMed]
- Weber, F.C.; Nemeth, T.; Csepregi, J.Z.; Dudeck, A.; Roers, A.; Ozsvari, B.; Oswald, E.; Puskas, L.G.; Jakob, T.; Mocsai, A.; et al. Neutrophils are required for both the sensitization and elicitation phase of contact hypersensitivity. J. Exp. Med. 2015, 212, 15–22. [Google Scholar] [CrossRef] [PubMed]
- Schwab, L.; Goroncy, L.; Palaniyandi, S.; Gautam, S.; Triantafyllopoulou, A.; Mocsai, A.; Reichardt, W.; Karlsson, F.J.; Radhakrishnan, S.V.; Hanke, K.; et al. Neutrophil granulocytes recruited upon translocation of intestinal bacteria enhance graft-versus-host disease via tissue damage. Nat. Med. 2014, 20, 648–654. [Google Scholar] [CrossRef] [PubMed]
- Lim, K.; Hyun, Y.M.; Lambert-Emo, K.; Capece, T.; Bae, S.; Miller, R.; Topham, D.J.; Kim, M. Neutrophil trails guide influenza-specific CD8+ T cells in the airways. Science 2015, 349. [Google Scholar] [CrossRef] [PubMed]
- Hampton, H.R.; Bailey, J.; Tomura, M.; Brink, R.; Chtanova, T. Microbe-dependent lymphatic migration of neutrophils modulates lymphocyte proliferation in lymph nodes. Nat. Commun. 2015, 6. [Google Scholar] [CrossRef] [PubMed]
- MacKay, C.; Davies, M.; Summerfield, V.; Maxwell, G. From pathways to people: Applying the adverse outcome pathway (AOP) for skin sensitization to risk assessment. ALTEX 2013, 30, 473–486. [Google Scholar] [CrossRef] [PubMed]
- Maxwell, G.; MacKay, C.; Cubberley, R.; Davies, M.; Gellatly, N.; Glavin, S.; Gouin, T.; Jacquoilleot, S.; Moore, C.; Pendlington, R.; et al. Applying the skin sensitisation adverse outcome pathway (AOP) to quantitative risk assessment. Toxicol. Vitr. 2014, 28, 8–12. [Google Scholar] [CrossRef] [PubMed]
- Leist, M.; Hasiwa, N.; Rovida, C.; Daneshian, M.; Basketter, D.; Kimber, I.; Clewell, H.; Gocht, T.; Goldberg, A.; Busquet, F.; et al. Consensus report on the future of animal-free systemic toxicity testing. ALTEX 2014, 31, 341–356. [Google Scholar] [CrossRef] [PubMed]
- van der Veen, J.W.; Soeteman-Hernandez, L.G.; Ezendam, J.; Stierum, R.; Kuper, F.C.; van Loveren, H. Anchoring molecular mechanisms to the adverse outcome pathway for skin sensitization: Analysis of existing data. Crit. Rev. Toxicol. 2014, 44, 590–599. [Google Scholar] [CrossRef] [PubMed]
- Wong, C.L.; Ghassabian, S.; Smith, M.T.; Lam, A.L. In vitro methods for hazard assessment of industrial chemicals—Opportunities and challenges. Front. Pharmacol. 2015, 6. [Google Scholar] [CrossRef] [PubMed]
- Reisinger, K.; Hoffmann, S.; Alepee, N.; Ashikaga, T.; Barroso, J.; Elcombe, C.; Gellatly, N.; Galbiati, V.; Gibbs, S.; Groux, H.; et al. Systematic evaluation of non-animal test methods for skin sensitisation safety assessment. Toxicol. Vitr. 2015, 29, 259–270. [Google Scholar] [CrossRef] [PubMed]
- Urbisch, D.; Mehling, A.; Guth, K.; Ramirez, T.; Honarvar, N.; Kolle, S.; Landsiedel, R.; Jaworska, J.; Kern, P.S.; Gerberick, F.; et al. Assessing skin sensitization hazard in mice and men using non-animal test methods. Regul. Toxicol. Pharmacol. 2015, 71, 337–351. [Google Scholar] [CrossRef] [PubMed]
- Hayes, J.D.; Dinkova-Kostova, A.T. The Nrf2 regulatory network provides an interface between redox and intermediary metabolism. Trends Biochem. Sci. 2014, 39, 199–218. [Google Scholar] [CrossRef] [PubMed]
- Tebay, L.E.; Robertson, H.; Durant, S.T.; Vitale, S.R.; Penning, T.M.; Dinkova-Kostova, A.T.; Hayes, J.D. Mechanisms of activation of the transcription factor Nrf2 by redox stressors, nutrient cues, and energy status and the pathways through which it attenuates degenerative disease. Free Radic. Biol. Med. 2015, 88, 108–146. [Google Scholar] [CrossRef] [PubMed]
- Bauch, C.; Kolle, S.N.; Ramirez, T.; Eltze, T.; Fabian, E.; Mehling, A.; Teubner, W.; van Ravenzwaay, B.; Landsiedel, R. Putting the parts together: Combining in vitro methods to test for skin sensitizing potentials. Regul. Toxicol. Pharmacol. 2012, 63, 489–504. [Google Scholar] [CrossRef] [PubMed]
- Natsch, A.; Ryan, C.A.; Foertsch, L.; Emter, R.; Jaworska, J.; Gerberick, F.; Kern, P. A dataset on 145 chemicals tested in alternative assays for skin sensitization undergoing prevalidation. J. Appl. Toxicol. 2013, 33, 1337–1352. [Google Scholar] [CrossRef] [PubMed]
- van der Veen, J.W.; Rorije, E.; Emter, R.; Natsch, A.; van Loveren, H.; Ezendam, J. Evaluating the performance of integrated approaches for hazard identification of skin sensitizing chemicals. Regul. Toxicol. Pharmacol. 2014, 69, 371–379. [Google Scholar] [CrossRef] [PubMed]
- Vandebriel, R.J.; Pennings, J.L.; Baken, K.A.; Pronk, T.E.; Boorsma, A.; Gottschalk, R.; Van Loveren, H. Keratinocyte gene expression profiles discriminate sensitizing and irritating compounds. Toxicol. Sci. 2010, 117, 81–89. [Google Scholar] [CrossRef] [PubMed]
- van der Veen, J.W.; Pronk, T.E.; van Loveren, H.; Ezendam, J. Applicability of a keratinocyte gene signature to predict skin sensitizing potential. Toxicol. Vitr. 2013, 27, 314–322. [Google Scholar] [CrossRef] [PubMed]
- Lambrechts, N.; Nelissen, I.; Van Tendeloo, V.; Witters, H.; Van Den Heuvel, R.; Hooyberghs, J.; Schoeters, G. Functionality and specificity of gene markers for skin sensitization in dendritic cells. Toxicol. Lett. 2011, 203, 106–110. [Google Scholar] [CrossRef] [PubMed]
- Johansson, H.; Albrekt, A.S.; Borrebaeck, C.A.; Lindstedt, M. The GARD assay for assessment of chemical skin sensitizers. Toxicol. Vitr. 2013, 27, 1163–1169. [Google Scholar] [CrossRef] [PubMed]
- Cottrez, F.; Boitel, E.; Auriault, C.; Aeby, P.; Groux, H. Genes specifically modulated in sensitized skins allow the detection of sensitizers in a reconstructed human skin model. Development of the SENS-IS assay. Toxicol. Vitr. 2015, 29, 787–802. [Google Scholar] [CrossRef] [PubMed]
- Agner, T.; Johansen, J.D.; Overgaard, L.; Volund, A.; Basketter, D.; Menne, T. Combined effects of irritants and allergens. Synergistic effects of nickel and sodium lauryl sulfate in nickel—Sensitized individuals. Contact Dermat. 2002, 47, 21–26. [Google Scholar] [CrossRef]
- Pedersen, L.K.; Johansen, J.D.; Held, E.; Agner, T. Augmentation of skin response by exposure to a combination of allergens and irritants—A review. Contact Dermat. 2004, 50, 265–273. [Google Scholar] [CrossRef] [PubMed]
- Bonefeld, C.M.; Nielsen, M.M.; Rubin, I.M.; Vennegaard, M.T.; Dabelsteen, S.; Gimenez-Arnau, E.; Lepoittevin, J.P.; Geisler, C.; Johansen, J.D. Enhanced sensitization and elicitation responses caused by mixtures of common fragrance allergens. Contact Dermat. 2011, 65, 336–342. [Google Scholar] [CrossRef] [PubMed]
- Martin, S.F. Adaptation in the innate immune system and heterologous innate immunity. Cell. Mol. Life Sci. 2014, 71, 4115–4130. [Google Scholar] [CrossRef] [PubMed]
- Martin, S.F. New concepts in cutaneous allergy. Contact Dermat. 2015, 72, 2–10. [Google Scholar] [CrossRef] [PubMed]
- Kienhuis, A.S.; Slob, W.; Gremmer, E.R.; Vermeulen, J.P.; Ezendam, J. A Dose-Response Modeling Approach Shows That Effects From Mixture Exposure to the Skin Sensitizers Isoeugenol and Cinnamal Are in Line With Dose Addition and Not With Synergism. Toxicol. Sci. 2015, 147, 68–74. [Google Scholar] [CrossRef] [PubMed]
© 2016 by the author; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Martin, S.F. Mechanistic Understanding of Contact Allergy. Cosmetics 2016, 3, 8. https://doi.org/10.3390/cosmetics3010008
Martin SF. Mechanistic Understanding of Contact Allergy. Cosmetics. 2016; 3(1):8. https://doi.org/10.3390/cosmetics3010008
Chicago/Turabian StyleMartin, Stefan F. 2016. "Mechanistic Understanding of Contact Allergy" Cosmetics 3, no. 1: 8. https://doi.org/10.3390/cosmetics3010008
APA StyleMartin, S. F. (2016). Mechanistic Understanding of Contact Allergy. Cosmetics, 3(1), 8. https://doi.org/10.3390/cosmetics3010008




