The Scalp Has a Lower Stratum Corneum Function with a Lower Sensory Input than Other Areas of the Skin Evaluated by the Electrical Current Perception Threshold
Abstract
:1. Introduction
2. Experimental Section
2.1. Study Design
2.2. Participants
2.3. Test Procedure
2.4. Statistical Analysis
3. Results
3.1. Participants with Skin Symptoms
| Score | Scaling | Erythema | Papule | Eschar | Pustule | Nodule |
|---|---|---|---|---|---|---|
| 0 | 15 | 3 | 28 | 35 | 31 | 43 |
| 1 | 18 | 26 | 11 | 8 | 12 | 3 |
| 2 | 11 | 14 | 8 | 4 | 1 | 1 |
| 3 | 3 | 4 | 0 | 0 | 3 | 0 |
| 4 | 0 | 0 | 0 | 0 | 0 | 0 |
| SUM | 47 | 47 | 47 | 47 | 47 | 47 |
3.2. Comparison of Measured Data between the Scalp and the Forehead
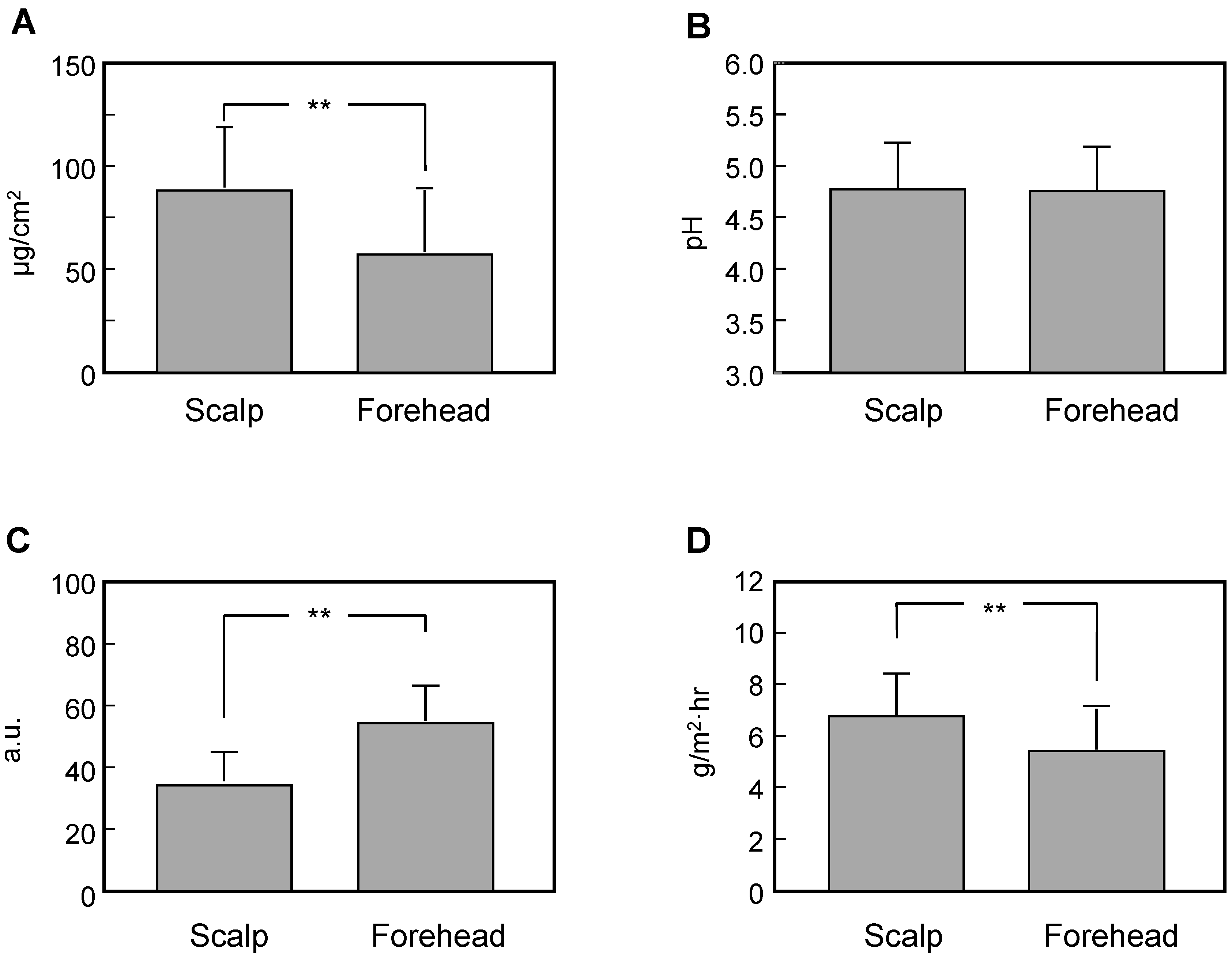
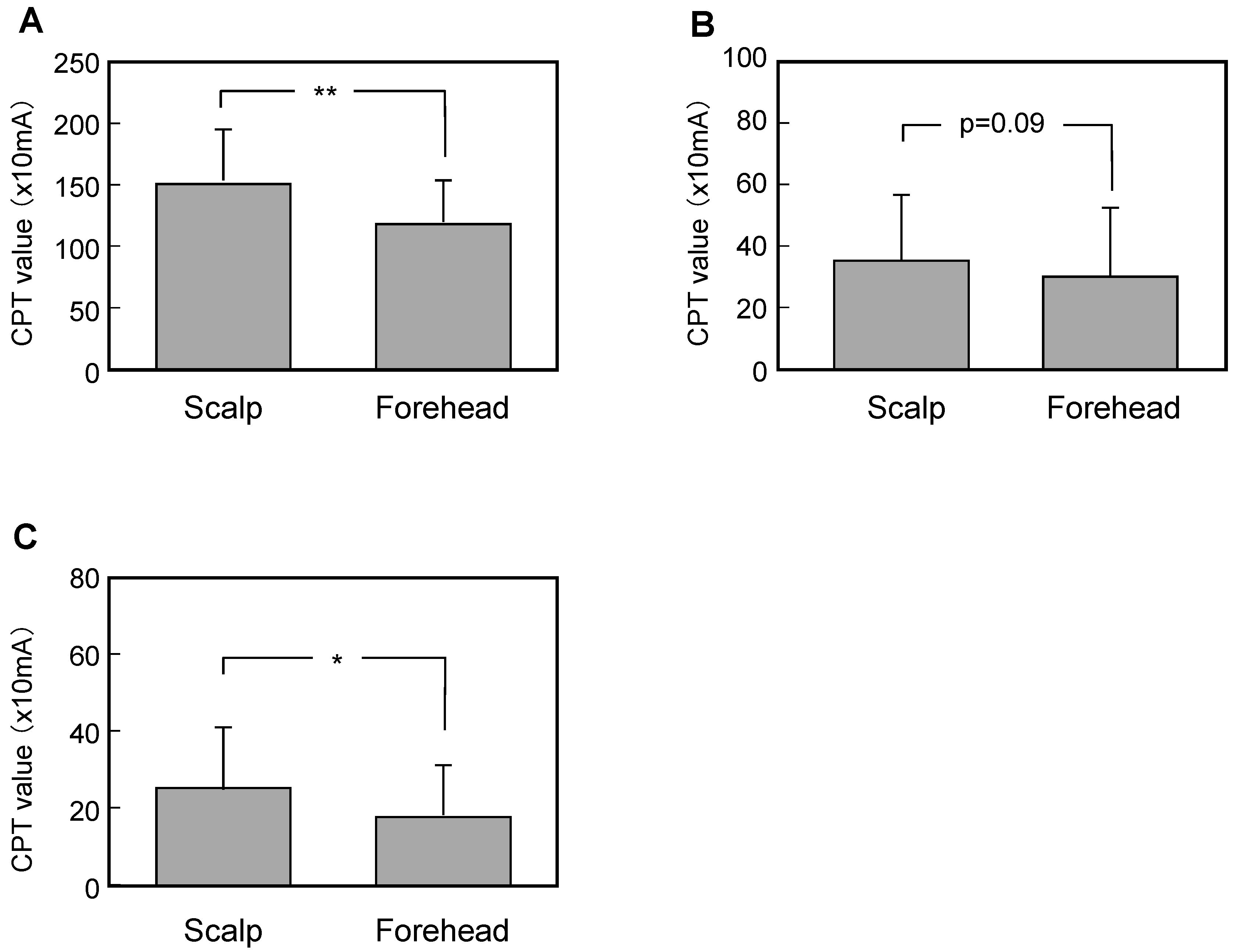
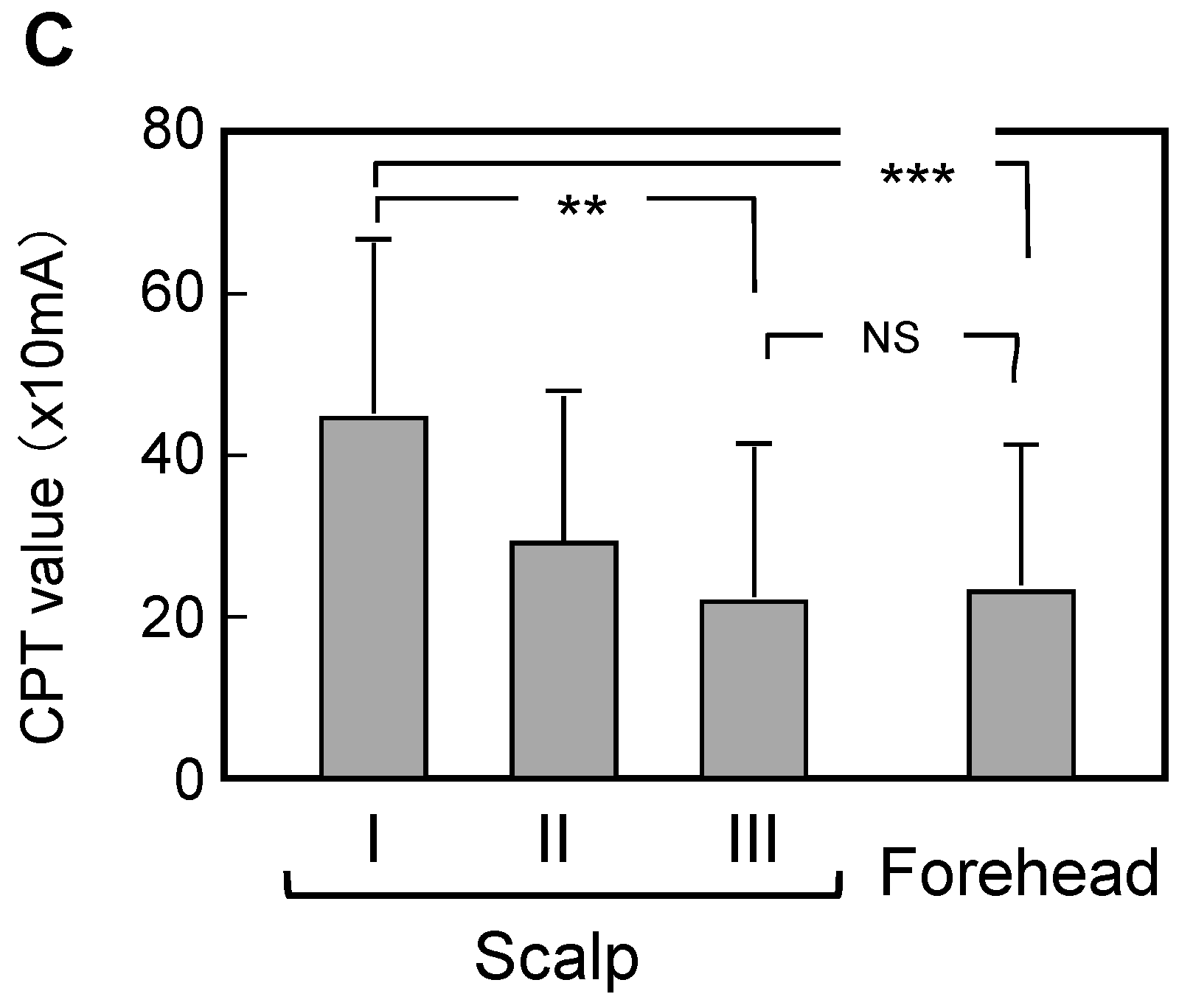
3.3. Comparison of Measured Data between Scalps with Skin Symptoms of Different Severity and the Forehead
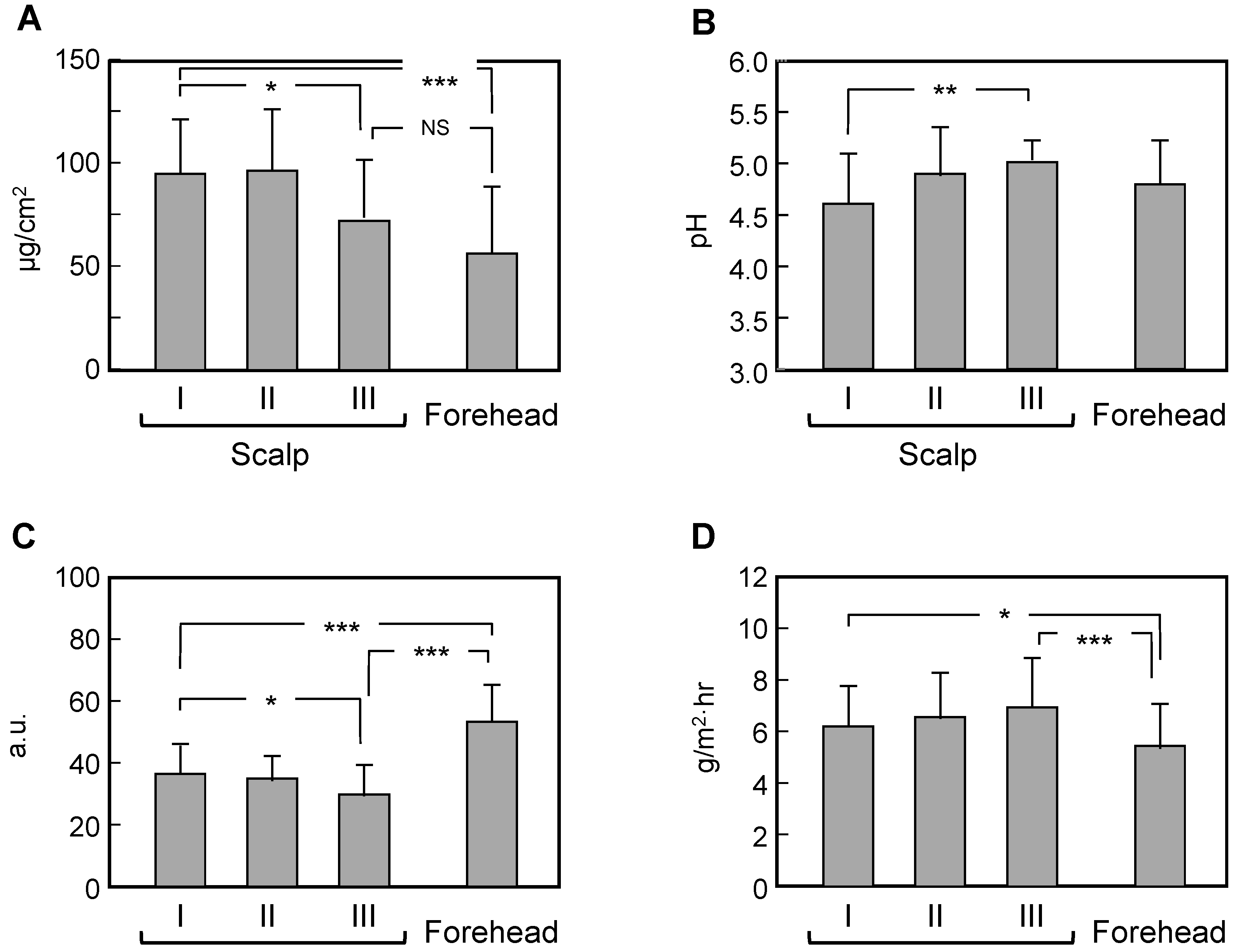
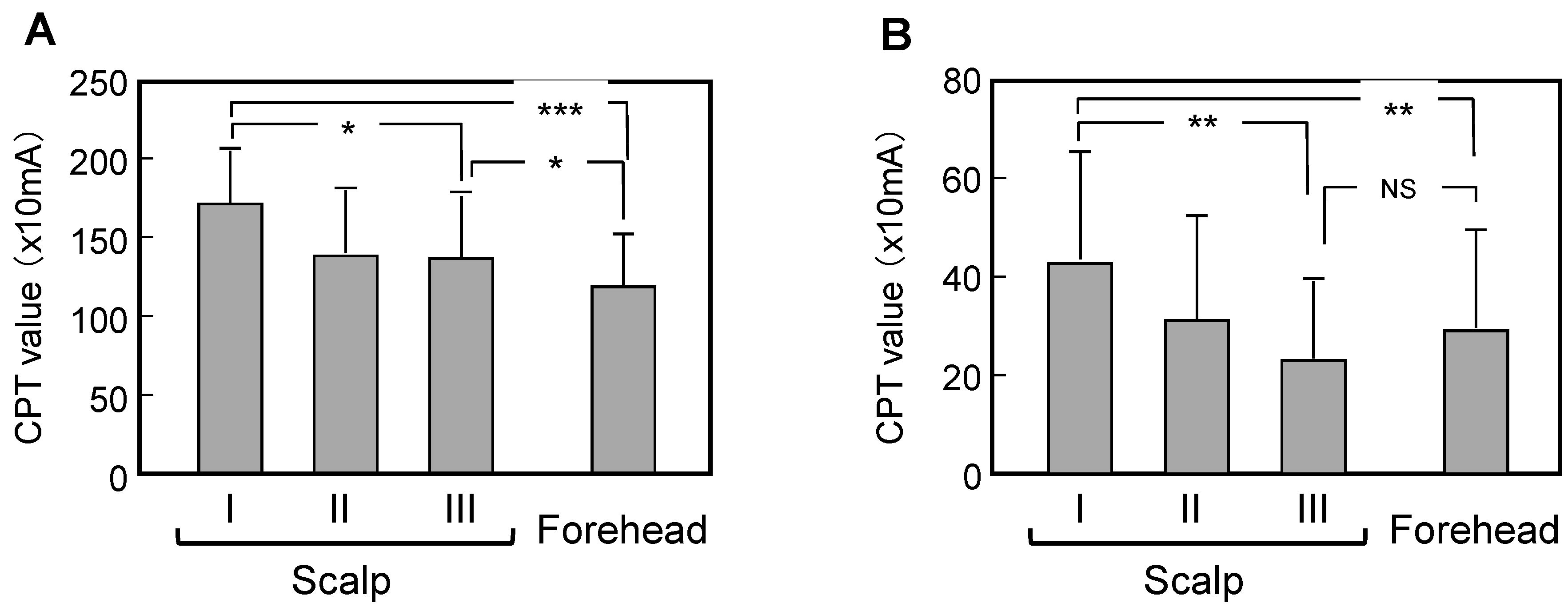
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Hay, R.J. Malassezia, dandruff and seborrhoeic dermatitis: An overview. Br. J. Dermatol. 2011, 165, 2–8. [Google Scholar] [CrossRef] [PubMed]
- Turner, G.A.; Hoptroff, M.; Harding, C.R. Stratum corneum dysfunction in dandruff. Int. J. Cosmet. Sci. 2012, 34, 298–306. [Google Scholar] [CrossRef] [PubMed]
- Gaitanis, G.; Magiatis, P.; Hantschke, M.; Bassukas, I.D.; Velegraki, A. The Malassezia genus in skin and systemic diseases. Clin. Microbiol. Rev. 2012, 25, 106–141. [Google Scholar] [CrossRef] [PubMed]
- Warner, R.R.; Schwartz, J.R.; Boissy, Y.; Dawson, T.L., Jr. Dandruff has an altered stratum corneum ultrastructure that is improved with zinc pyrithione shampoo. J. Am. Acad. Dermatol. 2001, 45, 897–903. [Google Scholar] [CrossRef] [PubMed]
- Bin Saif, G.A.; Ericson, M.E.; Yosipovitch, G. The itchy scalp—Scratching for an explanation. J. Eur. Exp. Dermatol. 2011, 20, 959–968. [Google Scholar] [CrossRef] [PubMed]
- Ya-Xian, Z.; Suetake, T.; Tagami, H. Number of cell layers of the stratum corneum in normal skin-relationship to the anatomical location on the body, age, sex and physical parameters. Arch. Dermatol. Res. 1999, 291, 555–559. [Google Scholar] [CrossRef] [PubMed]
- O’goshi, K.; Iguchi, M.; Tagami, H. Functional analysis of the stratum corneum of scalp skin: Studies in patients with alopecia areata and androgenetic alopecia. Arch. Dermatol. Res. 2000, 292, 605–611. [Google Scholar] [CrossRef] [PubMed]
- Myles, K.; Kalb, J.T.; Lowery, J.; Kattel, B.P. The effect of hair density on the coupling between the tactor and the skin of the human head. Appl. Ergon. 2015, 48, 177–185. [Google Scholar] [CrossRef] [PubMed]
- Zhai, H.; Fautz, R.; Fuchs, A.; Bhandarkar, S.; Maibach, H.I. Human scalp irritation compared to that of the arm and back. Contact Dermatitis 2004, 51, 196–200. [Google Scholar] [CrossRef] [PubMed]
- Essick, G.; Guest, S.; Martinez, E.; Chen, C.; McGlone, F. Site-dependent and subject-related variations in perioral thermal sensitivity. Somatosens. Mot. Res. 2004, 21, 159–175. [Google Scholar] [CrossRef] [PubMed]
- Mehrabyan, A.; Guest, S.; Essick, G.; McGlone, F. Tactile and thermal detection thresholds of the scalp skin. Somatosens. Mot. Res. 2011, 28, 31–47. [Google Scholar] [CrossRef] [PubMed]
- Rukwied, R.; Zeck, S.; Schmelz, M.; McGlone, F. Sensitivity of human scalp skin to pruritic stimuli investigated by intradermal microdialysis in vivo. J. Am. Acad. Dermatol. 2002, 47, 245–250. [Google Scholar] [CrossRef] [PubMed]
- Masson, E.A.; Veves, A.; Fernando, D.; Boulton, A.J.M. Current perception thresholds: A new, quick, and reproducible method for the assessment of peripheral neuropathy in diabetes mellitus. Diabetologia 1989, 32, 724–728. [Google Scholar] [CrossRef] [PubMed]
- Baquis, G.D.; Brown, W.F.; Capell, J.T.; Chaudhry, V.; Cros, D.; Drexinger, B.; Gelblum, J.B.; Gilchrist, J.M.; Gitter, A.J.; Haig, A.J.; et al. Technology review: The Neurometer® Current Perception Threshold (CPT). Muscle Nerve 1999, 22, 523–531. [Google Scholar]
- Kobayashi, H.; Kikuchi, K.; Tsubono, Y.; Tagami, H. Measurement of electrical current perception threshold of sensory nerves for pruritus in atopic dermatitis patients and normal individuals with various degrees of mild damage to the stratum corneum. Dermatology 2003, 206, 204–211. [Google Scholar] [CrossRef] [PubMed]
- Lee, E.; An, S.; Lee, T.R.; Kim, H.K. Development of a novel method for quantitative evaluation of sensory skin irritation inhibitors. Skin Res. Technol. 2009, 15, 464–469. [Google Scholar] [CrossRef] [PubMed]
- Parra, J.L.; Paye, M. EEMCO guidance for the in vivo assessment of skin surface pH. Skin Pharmacol. Appl. Skin Physiol. 2003, 16, 188–202. [Google Scholar] [CrossRef] [PubMed]
- Tagami, H. Location-related differences in structure and function of the stratum corneum with special emphasis on those of the facial skin. Int. J. Cosmet. Sci. 2008, 30, 413–434. [Google Scholar] [CrossRef] [PubMed]
- Mori, T.; Ishida, K.; Mukumoto, S.; Yamada, Y.; Imokawa, G.; Kabashima, K.; Kobayashi, M.; Bito, T.; Nakamura, M.; Ogasawara, K.; et al. Comparison of skin barrier function and sensory nerve electric current perception threshold between IgE-high extrinsic and IgE-normal intrinsic types of atopic dermatitis. Br. J. Dermatol. 2010, 162, 83–90. [Google Scholar] [CrossRef] [PubMed]
- Tominaga, M.; Ozawa, S.; Tengara, S.; Ogawa, H.; Takamori, K. Intraepidermal nerve fibers increase in dry skin of acetone-treated mice. J. Dermatol. Sci. 2007, 48, 103–111. [Google Scholar] [CrossRef] [PubMed]
- Florence, P.; Cornillon, C.; D’arras, M.F.; Flament, F.; Panhard, S.; Diridollou, S.; Loussouarn, G. Functional and structural age-related changes in the scalp skin of Caucasian women. Skin Res. Technol. 2013, 19, 384–393. [Google Scholar] [PubMed]
- Saint-Martory, C.; Roguedas-Contios, A.M.; Sibaud, V.; Degouy, A.; Schmitt, A.M.; Misery, L. Sensitive skin is not limited to the face. Br. J. Dermatol. 2008, 158, 130–133. [Google Scholar] [CrossRef] [PubMed]
- Misery, L.; Sibaud, V.; Ambronati, M.; Macy, G.; Boussetta, S.; Taieb, C. Sensitive scalp: Does this condition exist? An epidemiological study. Contact Dermatitis 2008, 58, 234–238. [Google Scholar] [CrossRef] [PubMed]
- Misery, L.; Rahhali, N.; Ambonati, M.; Black, D.; Saint-Martory, C.; Schmitt, A.M.; Taieb, C. Evaluation of sensitive scalp severity and symptomatology by using a new score. J. Eur. Acad. Dermatol. Venereol. 2011, 25, 1295–1298. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Takagi, Y.; Takatoku, H.; Terazaki, H.; Nakamura, T.; Ishida, K.; Kitahara, T. The Scalp Has a Lower Stratum Corneum Function with a Lower Sensory Input than Other Areas of the Skin Evaluated by the Electrical Current Perception Threshold. Cosmetics 2015, 2, 384-393. https://doi.org/10.3390/cosmetics2040384
Takagi Y, Takatoku H, Terazaki H, Nakamura T, Ishida K, Kitahara T. The Scalp Has a Lower Stratum Corneum Function with a Lower Sensory Input than Other Areas of the Skin Evaluated by the Electrical Current Perception Threshold. Cosmetics. 2015; 2(4):384-393. https://doi.org/10.3390/cosmetics2040384
Chicago/Turabian StyleTakagi, Yutaka, Hiroko Takatoku, Hiroyuki Terazaki, Tadashi Nakamura, Koichi Ishida, and Takashi Kitahara. 2015. "The Scalp Has a Lower Stratum Corneum Function with a Lower Sensory Input than Other Areas of the Skin Evaluated by the Electrical Current Perception Threshold" Cosmetics 2, no. 4: 384-393. https://doi.org/10.3390/cosmetics2040384
APA StyleTakagi, Y., Takatoku, H., Terazaki, H., Nakamura, T., Ishida, K., & Kitahara, T. (2015). The Scalp Has a Lower Stratum Corneum Function with a Lower Sensory Input than Other Areas of the Skin Evaluated by the Electrical Current Perception Threshold. Cosmetics, 2(4), 384-393. https://doi.org/10.3390/cosmetics2040384





