3.7. Clinical Test Protection and Soothing
Both extracts were proven to be clinically effective soothing and protecting agents with significant reduction in erythema and TEWL as compared to placebo and control. In Study Scheme 1, the products were applied before UV exposure and then again, after 24 h (
Table 2). The relative protective and soothing effects are expressed in
Table 8 and
Table 9 for cream preparations of the
Fucus vesiculosus and
Undaria pinnatifida extracts. Out of the three study schemes, this first scheme of application generated the most protection; however, the other two schemes (application before and soon after exposure, or only after exposure) also markedly decreased TEWL and erythema.
Table 8.
Transepithelial water loss after application of placebo or active cream, as compared to control (Study Scheme 1); n = 25 (percent change of mean vs. control).
Table 8.
Transepithelial water loss after application of placebo or active cream, as compared to control (Study Scheme 1); n = 25 (percent change of mean vs. control).
| Product | T0 (%) | T1h (%) | T2h (%) | T24h (%) |
|---|
| Fucus vesiculosus extract | −7.9 | −21.9 | −22.1 | −18.3 |
| Undaria pinnatifida extract | −7.3 | −19.8 | −19.6 | −17.1 |
| Placebo | −1.6 | −8.0 | −7.6 | −2.6 |
Table 9.
Erythema after application of placebo or active cream, as compared to control (Study Scheme 1); n = 25 (percent change of mean vs. control).
Table 9.
Erythema after application of placebo or active cream, as compared to control (Study Scheme 1); n = 25 (percent change of mean vs. control).
| Product | T0 (%) | T1h (%) | T2h (%) | T24h (%) |
|---|
| Fucus vesiculosus extract | −10.6 | −21.6 | −22.6 | −14.7 |
| Undaria pinnatifida extract | −9.0 | −18.5 | −19.6 | −12.1 |
| Placebo | −1.4 | −2.2 | −2.0 | −0.2 |
Statistical analysis was performed by way of
t-testing for paired samples, to compare both
Undaria pinnatifida and
Fucus vesiculosus extracts to the placebo and control data sets in Study Scheme 1, for both TEWL and erythema. Significant differences were found between the fucoidan extracts and the placebo against control tests in all cases, as summarized in
Table 10.
Table 10.
Significant differences between data sets for Study Scheme 1 (Student’s t-test for paired data).
Table 10.
Significant differences between data sets for Study Scheme 1 (Student’s t-test for paired data).
| Extract | TEWL % | Erythema % |
|---|
| Control | Placebo | Control | Placebo |
|---|
| Fucus vesiculosus extract | p < 0.001 | p < 0.002 | p < 0.001 | p < 0.001 |
| Undaria pinnatifida extract | p < 0.001 | p < 0.002 | p < 0.001 | p < 0.001 |
The data from Study Schemes 2 and 3 showed that both extracts were protective and soothing as summarized in
Table 11 and
Table 12, respectively. These observations were statistically significant with regard to both placebo and control (
t-test for paired data,
p < 0.05). The relative protective and soothing effects on erythema and TEWL were slightly greater for the
Fucus vesiculosus extract than for the
Undaria pinnatifida extract; however, both extracts demonstrated significant protective and soothing effects.
Table 11.
Erythema and TEWL Study Scheme 2, as compared to control (n = 25) (percent change of mean vs. control).
Table 11.
Erythema and TEWL Study Scheme 2, as compared to control (n = 25) (percent change of mean vs. control).
| Extract | TEWL % | Erythema % |
|---|
| 24 h | 48 h | 24 h | 48 h |
|---|
| Fucus vesiculosus | −8.4 | −16.6 | −10.4 | −8.9 |
| Undaria pinnatifida | −7.7 | −14.4 | −10.3 | −8.1 |
| Placebo | −1.2 | −2.3 | −1.2 | −2.5 |
Table 12.
Erythema and TEWL Study Scheme 3, as compared to control (n = 25) (percent change of mean vs. control).
Table 12.
Erythema and TEWL Study Scheme 3, as compared to control (n = 25) (percent change of mean vs. control).
| Extract | TEWL % | Erythema % |
|---|
| 1 h | 24 h | 1 h | 24 h |
|---|
| Fucus vesiculosus | −14.9 | −11.3 | −11.7 | −20.7 |
| Undaria pinnatifida | −14.0 | −8.4 | −9.8 | −18.2 |
| Placebo | 0.4 | −1.2 | −0.6 | −7.9 |
3.8. Clinical Test of Fucus vesiculosus Extract for Age Spots, Brightness and Wrinkles
Results from the 20 subjects in the double-blind, placebo-controlled, hemi-face study showed that
Fucus vesiculosus extract at 0.3%
w/
v was an effective cosmetic ingredient for reducing the melanin index of age spots, increasing brightness and decreasing wrinkles (
Figure 2,
Figure 3 and
Figure 4). The clinical evaluation was performed by a dermatologist as described in
Section 2.8, with clinical scores assigned from the following: no variation, slight improvement, moderate improvement, or remarkable improvement. The percentage of subjects that experienced an improvement, as evaluated by the dermatologist, is noted in
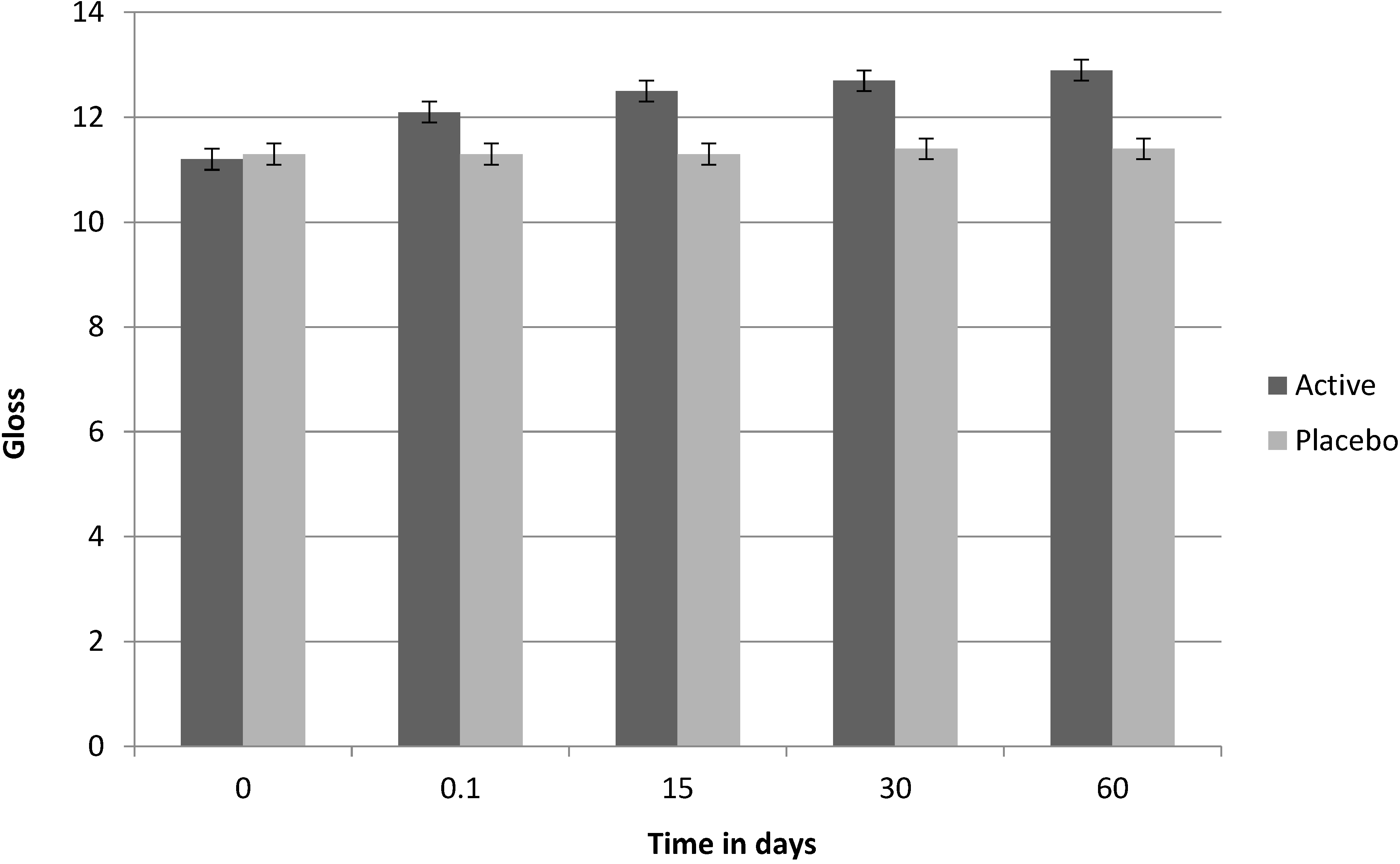
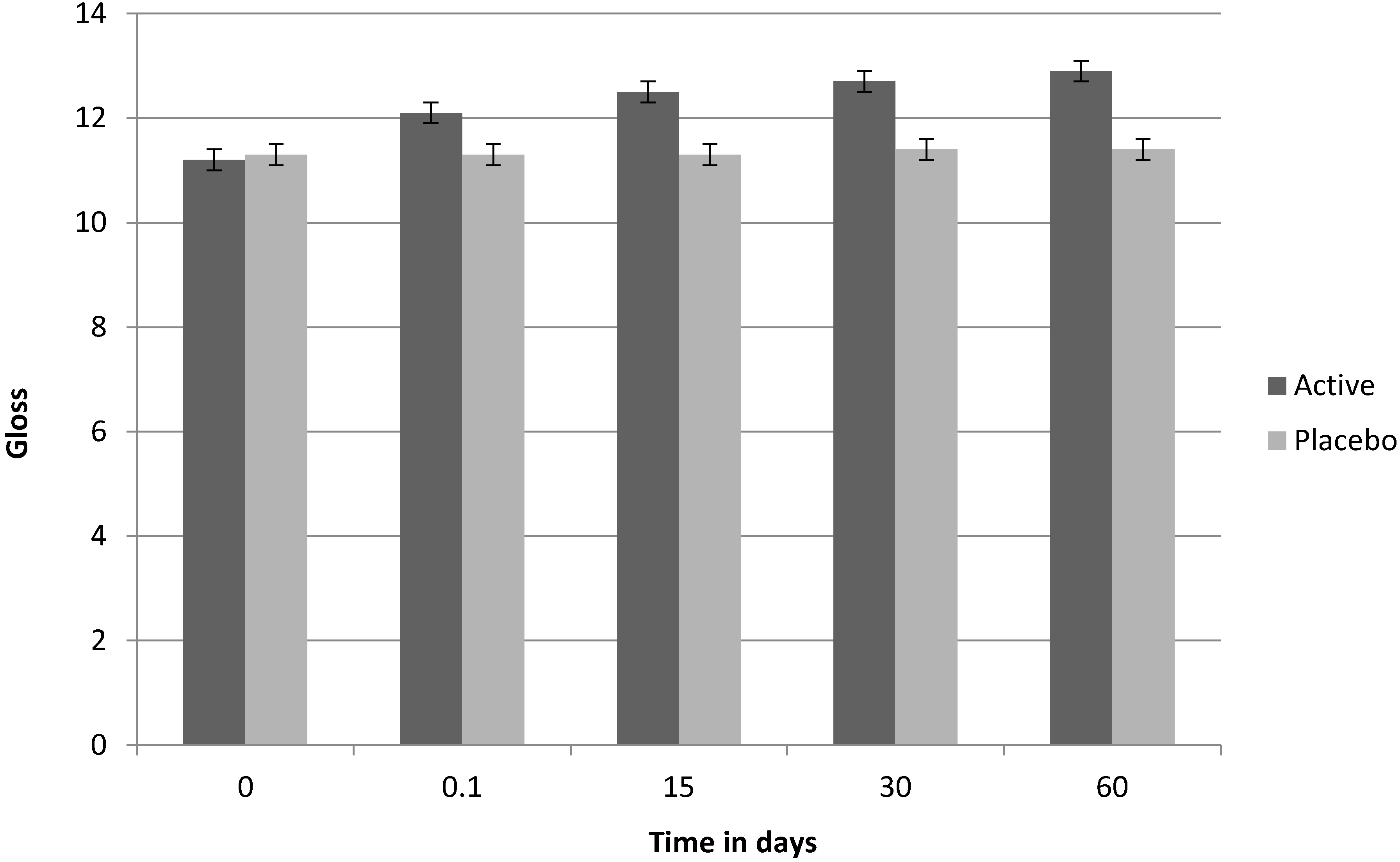
Table 13. According to the clinical analysis, after 60 days of use, 50% of the volunteers showed an improvement in skin brightness, 65% showed a reduction in skin spot appearance and 45% showed an improvement in the appearance of wrinkles. Whilst the trial was terminated at 60 days, the rate of reduction in age spot index and increase in brightness indicates marked trends. Intergroup analysis was performed by way of the Wilcoxon signed rank test for non-parametric data (
Figure 2 and
Figure 3).
Table 13.
Dermatologist clinical analysis of Fucus vesiculosus extract (percentage of subjects showing an improvement).
Table 13.
Dermatologist clinical analysis of Fucus vesiculosus extract (percentage of subjects showing an improvement).
| Metric | Treatment | Placebo | Treatment | Placebo | Treatment | Placebo |
|---|
| Variation vs. T0 | T15 days | T15 days | T30 days | T30 days | T60 days | T60 days |
| Wrinkle evaluation | 10% | 0 | 30% | 0 | 45% | 5% |
| Reduction of skin spot appearance | 0 | 0 | 40% | 0 | 65% | 0 |
| Skin brightness | 10% | 0 | 40% | 0 | 50% | 0 |
Figure 2.
Clinical test on Fucus vesiculosus extract: age spot intensity over time.
Figure 2.
Clinical test on Fucus vesiculosus extract: age spot intensity over time.
Figure 3.
Clinical test on Fucus vesiculosus extract: brightness (gloss) factor, over time.
Figure 3.
Clinical test on Fucus vesiculosus extract: brightness (gloss) factor, over time.
Figure 4.
Clinical test on Fucus vesiculosus extract: wrinkle depth over time.
Figure 4.
Clinical test on Fucus vesiculosus extract: wrinkle depth over time.
3.9. Discussion
Despite the popularity of marine algal extracts in cosmetic preparations, little has been reported to date regarding the specific bioactivity of these extracts. Over the past 20 years, there has been a growing body of in vitro research on the bioactive properties of fucoidans, and many of these studies have focused on the potential for fucoidan as a cosmetic ingredient. The key focus of research to date (including this study) has been to investigate the inhibitory effects of topically applied fucoidan on aging and photo-damaged skin. In this study, two extracts from two different marine algal sources were examined. The extracts were highly purified, extensively characterized fucoidan and polyphenol extracts, with purity accounting for upwards of 85% of the whole extracts. The purities were verified analytically, confirming the removal of many undesired components, such as salts and iodine.
The stimulation of skin matrix enzymes, including collagenase and elastase, is a key aspect of skin aging [
11]. It is thought that the accumulation of degraded collagen fibrils prevents new tissue formation and causes further skin degradation by inducing further enzyme activity in a “positive feedback loop”. Lessening such enzyme activity may assist in the reduction of skin degradation and promote the formation of new matrix. Skin care applications for fucoidan as an immune regulator and as a soothing ingredient have been established in this study, with the
Undaria pinnatifida extract. In this study, inhibition of collagenase (bacterial) and elastase (human neutrophil) by the
Undaria pinnatifida extract
in vitro was noted. Although it is not possible to directly infer effects from
in vitro studies, early clinical observations demonstrated improvements in skin.
Further
in vitro data for anti-aging applications for
Undaria pinnatifida extract reported here includes
SIRT1 protein expression; changes in the gene expression profile of UV irradiated reconstructed human epidermis and inhibition of glycation, a marker for aging skin. Increasing the levels of
SIRT1 can mimic the benefits of caloric restriction, enhancing sugar and lipid metabolism, and maintaining a younger physiology [
19,
20]. Sirtuins are expressed in human skin, and play complex roles in cellular metabolism [
19]. In the skin,
SIRT1 levels are depressed by UV irradiation and oxidative damage and, in other studies,
SIRT1 stimulating extracts have also been effective in addressing skin aging [
21]. Although it is not possible to directly extrapolate from
in vitro studies, increasing the levels of
SIRT1 may assist in maintaining skin function, by reversing the effects of external factors. Whilst we demonstrated that
SIRT1 protein levels were increased
in vitro by both extracts, the gene expression of
SIRT1 was not assessed in the reconstructed epidermis analysis described in this paper. This would clearly be a valuable marker in future studies of either
Undaria pinnatifida extract or
Fucus vesiculosus extract.
The role of fucoidan in UV protection may be explained by
in vitro observations of the activation of Toll-like receptors (genes associated with the expression of antimicrobial peptides that are critical to innate immunity). Genes for Toll-like receptors 2 and 3 were strongly activated, at 387% and 229%, respectively. There was a large increase in the expression of wound-healing genes for the main matrix metalloproteases: at four hours, gene expression in skin treated with the
Undaria pinnatifida extract was more than doubled as compared to control, showing an enhanced wound-healing signal. The stimulation of Toll-like receptors by other types of fucoidan has been demonstrated in cell culture [
22]. The increased expression of extracellular matrix enzyme genes
in vitro by the
Undaria pinnatifida extract may be indicative of the ability of the extract to enhance the early wound-healing response, and then modulate that response. In addition, the decreased level of gene expression for
IL6 in vitro is noteworthy, with potential relevance to psoriasis [
23]. The “quick response” defense activity may be cosmetically useful for enhancing dermal protection.
The polyphenol-rich
Fucus vesiculosus extract was assessed in the ORAC5.0 assay, and demonstrated a marked total antioxidant value, with particular emphasis on inhibition of the superoxide free radical. Superoxide can be considered to be a precursor of the other free radicals, and this activity is useful in terms of topical cosmetic use as it can prevent environmental direct oxidative damage at the skin surface. Superoxide is not the strongest oxidant, as the hydroxyl radical is much more reactive [
24]; however, superoxide is highly toxic to cells, and contributes to lipid and DNA damage. Antioxidants that scavenge superoxide ions help to prevent the formation of radicals such as hydrogen peroxide and the highly reactive hydroxyl species, thus preventing further tissue damage [
11]. The polyphenol component of this extract may also be responsible for a higher expression of the
SIRT1 protein
in vitro, in comparison to the
Undaria pinnatifida extract.
In clinical applications, the
Fucus vesiculosus extract exerted slightly superior soothing and protection results against UV damage when compared to the
Undaria pinnatifida extract.
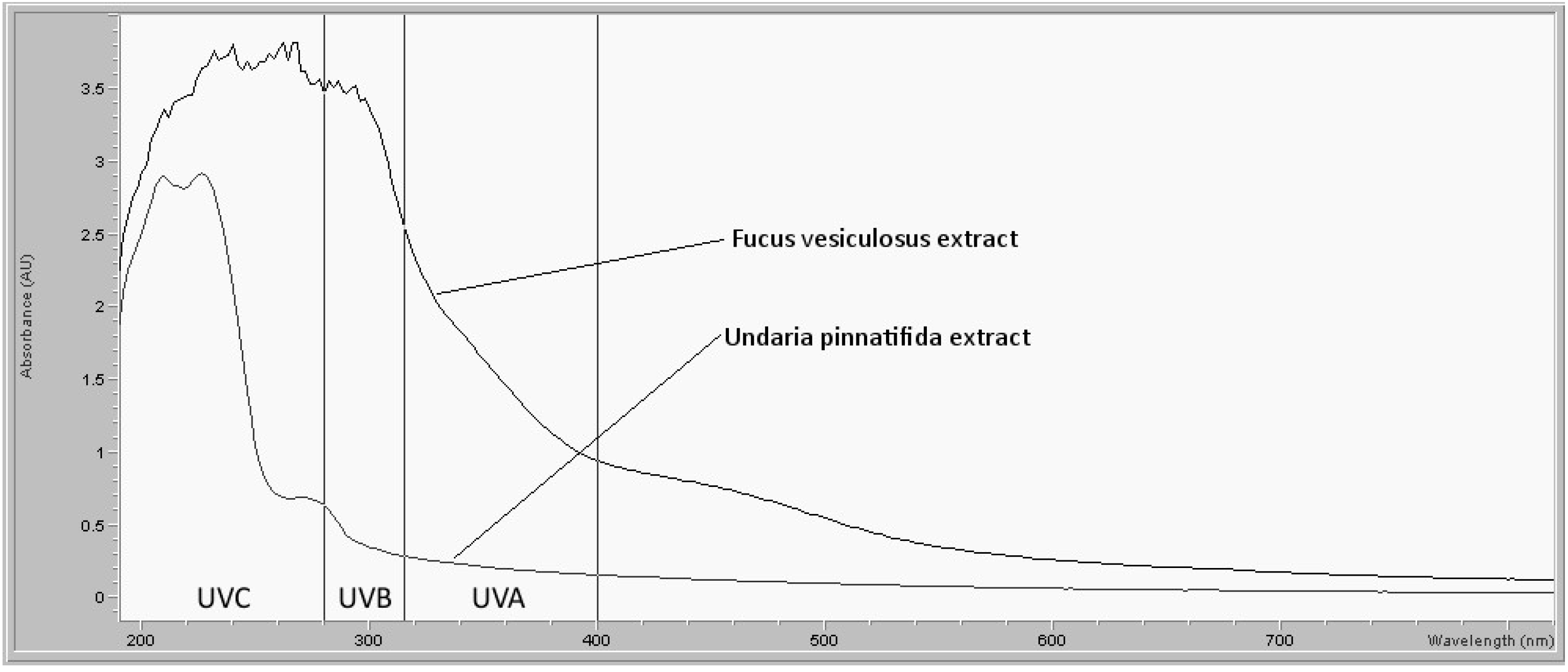
In vitro measurements showed that the
Fucus vesiculosus extract absorbed UV radiation in the skin-damaging UVA and UVB ranges, whereas the
Undaria pinnatifida extract did not.
In vitro testing demonstrated similar inhibition of the skin matrix enzyme human neutrophil elastase by both extracts; however, unlike the
Undaria pinnatifida extract,
Fucus vesiculosus extract was a highly effective inhibitor of mushroom tyrosinase as well as a highly effective antioxidant. As the tyrosinase examined in this study was mushroom-derived, it is not a directly comparable reflection of mammalian enzyme activity [
25], but, nonetheless, provides indicative data.
Recently, a tyrosinase inhibitory fucoidan was isolated from kelp [
26], with an apparent half-maximal inhibitory concentration of about 1 mg/mL. This is considerably higher than the inhibitory concentrations observed here, which is likely attributable to the polyphenol content of the
Fucus vesiculosus extract used in this study. Kang
et al. isolated polyphloroglucinols from
Ecklonia stolonifera [
9] with half-maximal inhibitory values in the μg/mL range, closer to the values observed in this study. Another research group noted fucoidan-reversible inhibition of tyrosinase, which was related to copper binding sites [
27]. Clinical examination of the
Fucus vesiculosus extract used here verified the skin brightening potential and age spot pigmentation reduction. It is not possible to directly infer that tyrosinase inhibition is taking place, as the other activities of the extract, such as antioxidative,
SIRT1 increasing, or other unknown effects may have resulted in the clinical observations. The small reduction in wrinkle depth of 6% compared to a 2% decrease for the placebo may also be a result of inhibition of oxidative damage. It is possible that absorbance of the fucoidan into the surface of the skin occurred and that this contributed to the small anti-wrinkle effect; however, absorbance measurement was not within the scope of this study.