Sources of Extraterrestrial Rare Earth Elements: To the Moon and Beyond
Abstract
:1. Introduction
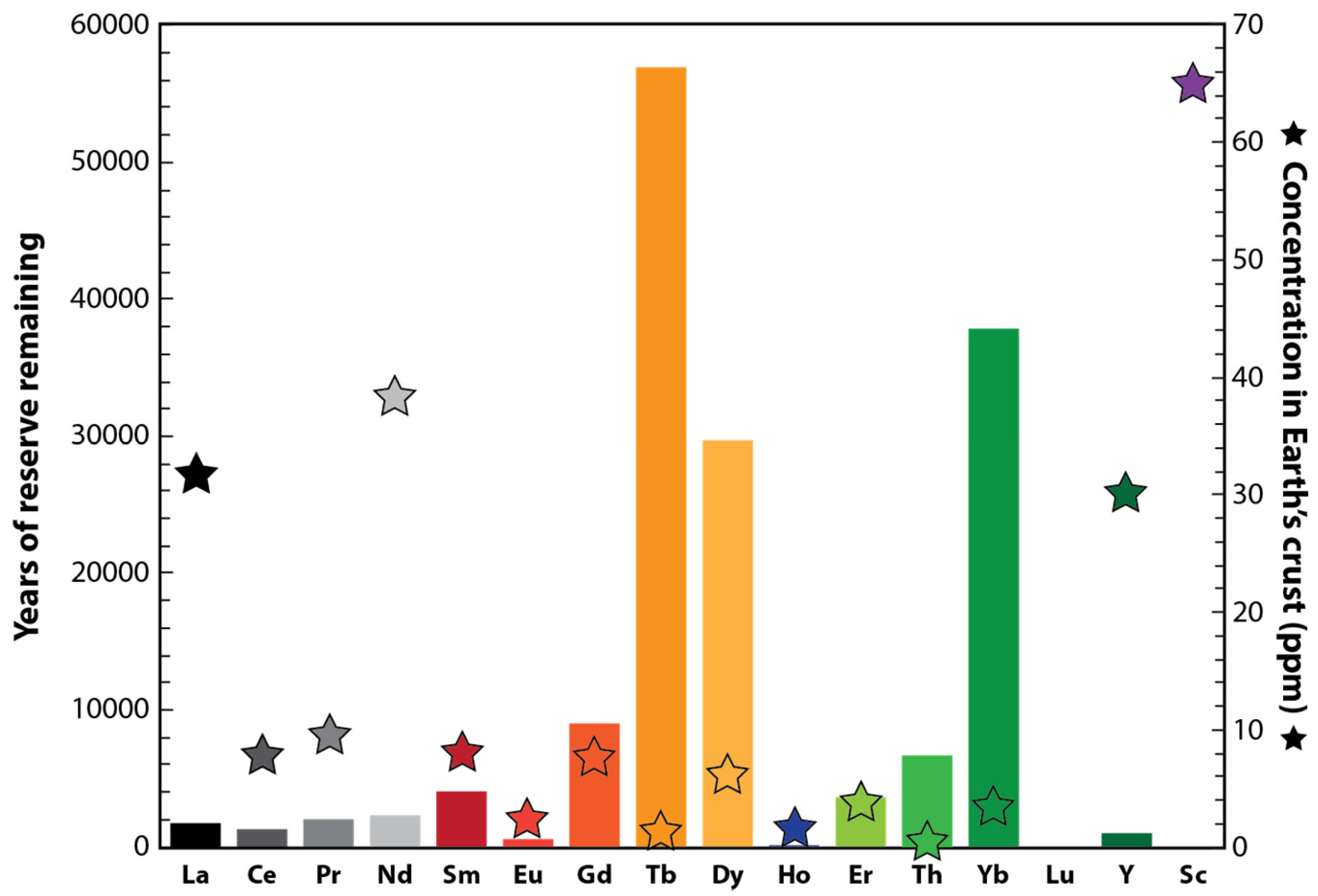
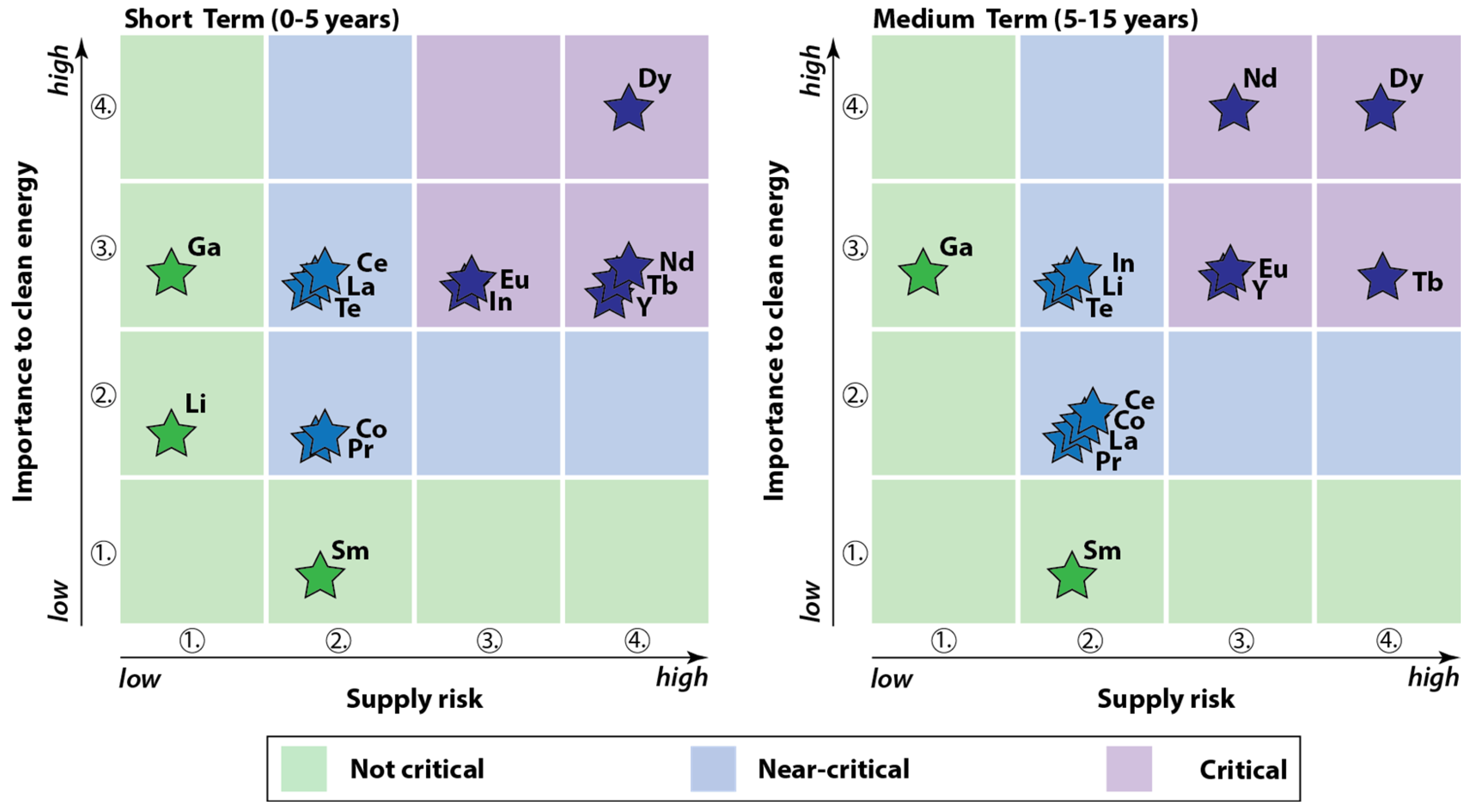
2. The Criticality of REEs in Our Society
3. Earth’s Moon: Missions to Our Nearest Neighbor
4. Lunar Differentiation
4.1. The Lunar Magma Ocean
4.2. Lunar Mineralogy
5. Lunar Resources
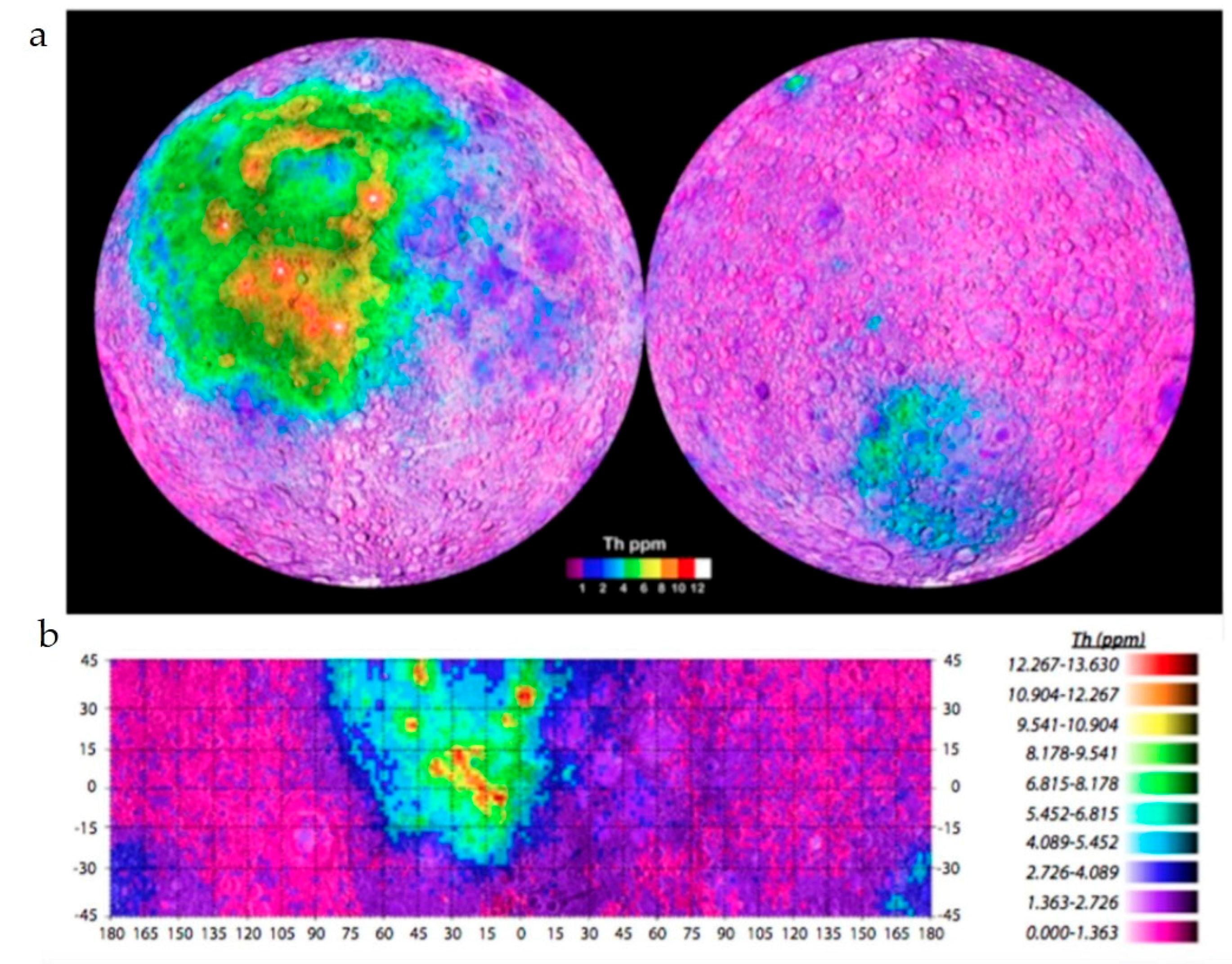
5.1. The KREEP Source
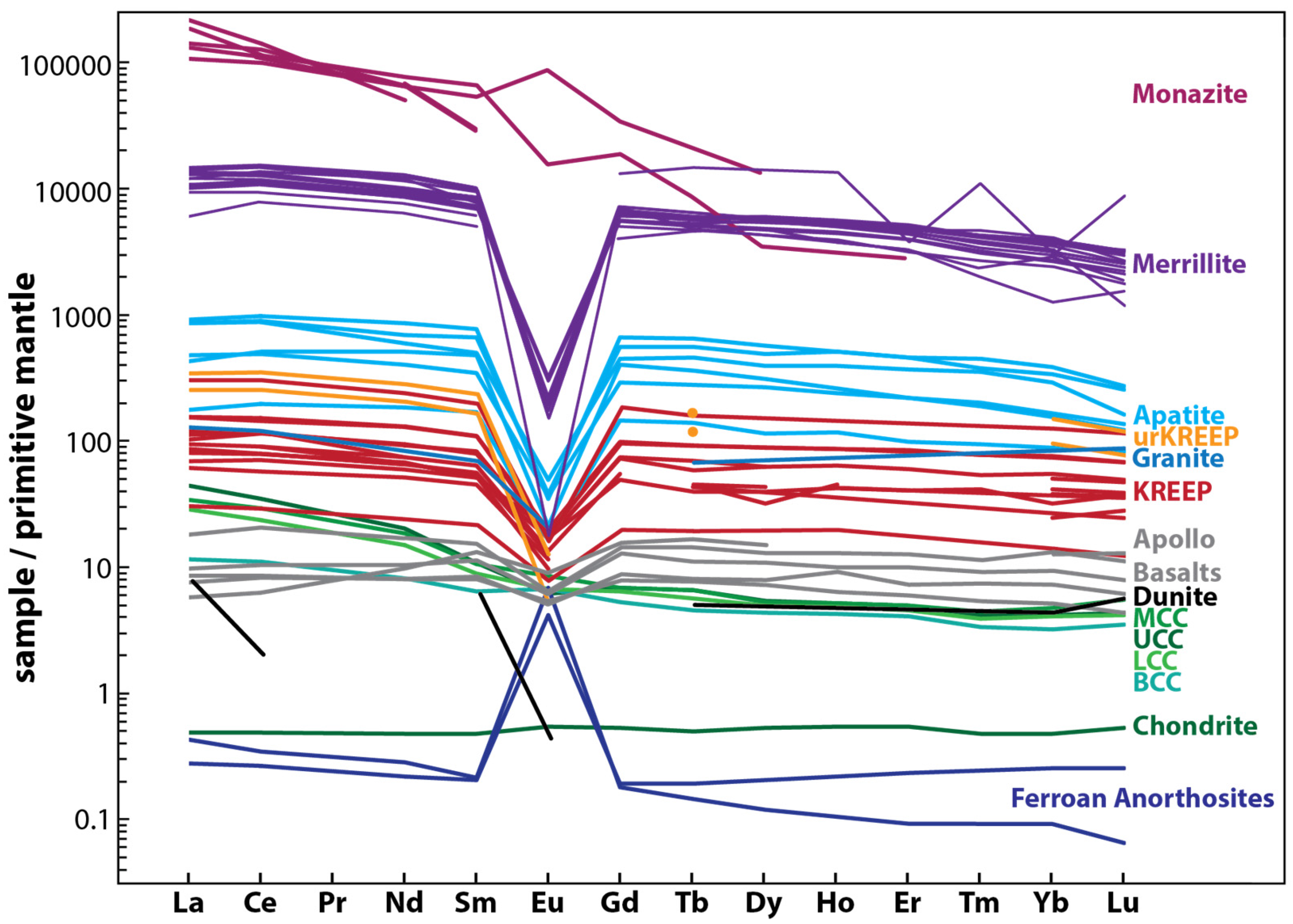
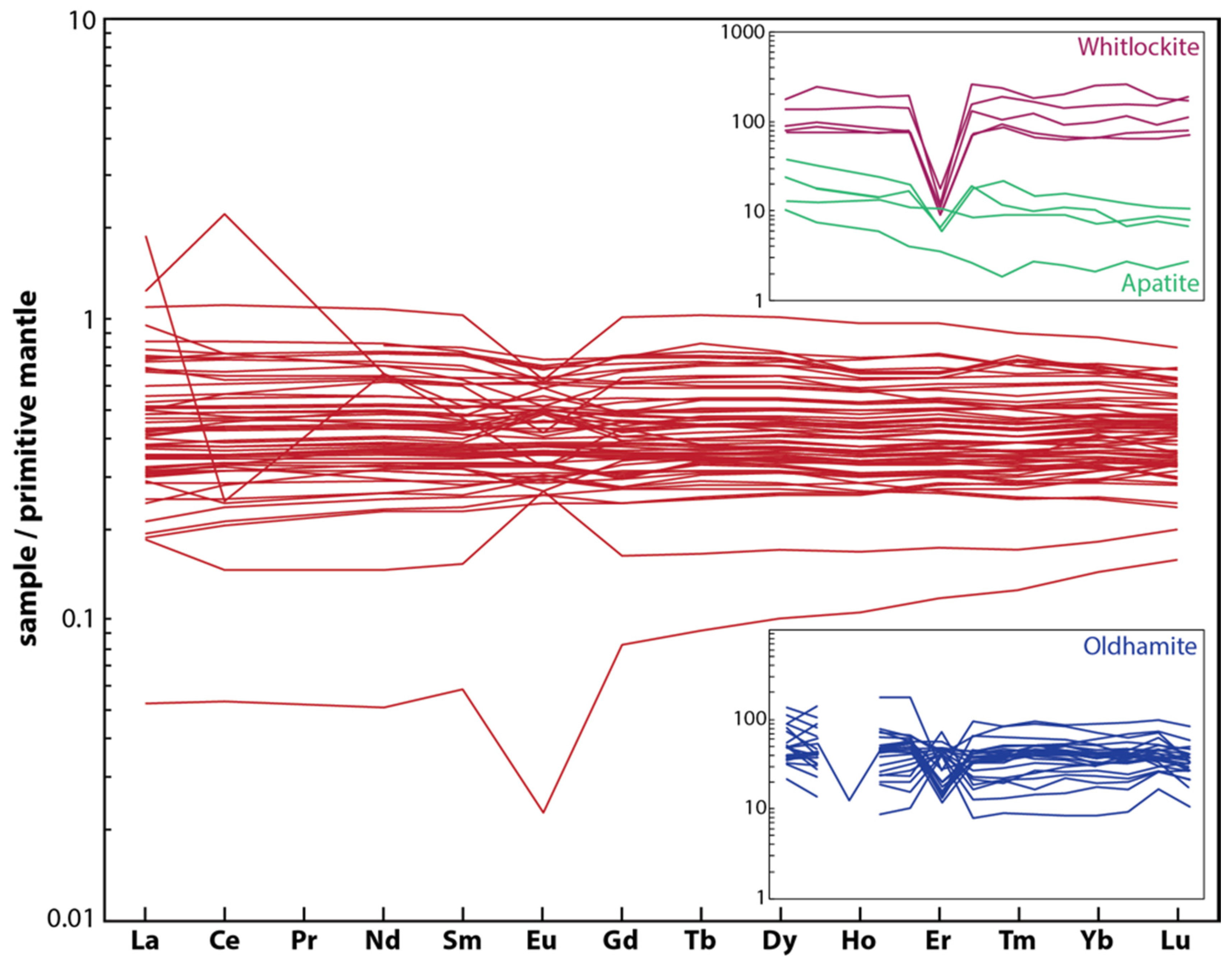
5.2. Lunar REE-Bearing Minerals
5.3. Other Potential Lunar Resources
5.3.1. Helium-3
5.3.2. Water
5.3.3. Oxygen
5.3.4. Aluminum
5.3.5. Magnesium
5.3.6. Iron
5.3.7. Basaltic Glass
5.3.8. Vacuum
5.3.9. Lunar Regolith
5.4. REEs beyond the Moon
5.5. Missions beyond the Moon
6. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Reinstein, E.R. Owning Outer Space. Northwest. J. Int. Law Bus. 1999, 20, 1. [Google Scholar]
- Chakhmouradiani, A.R.; Wall, F. Rare Earth Elements: Minerals, Mines, Magnets (and More). Elements 2012, 8, 333–340. [Google Scholar] [CrossRef]
- USGS. Rare Earths Statistics and Information. 2017. Available online: https://minerals.usgs.gov/minerals/pubs/commodity/rare_earths/ (accessed on 18 March 2017).
- Haque, N.; Hughes, A.; Lim, S.; Vernon, C. Rare Earth Elements: Overview of Mining, Mineralogy, Uses, Sustainability and Environmental Impact. Resources 2014, 3, 614–635. [Google Scholar] [CrossRef]
- Haskin, L.A.; Frey, F.A.; Schmitt, R.A.; Smith, R.H. Meteoritic, Solar and Terrestrial Rare-Earth Distributions. Phys. Chem. Earth 1966, 7, 167–321. [Google Scholar] [CrossRef]
- Kynicky, J.; Smith, M.P.; Xu, C. Diversity of Rare Earth Deposits: The Key Example of China. Elements 2012, 8, 361–367. [Google Scholar] [CrossRef]
- USGS. Mineral Commodities Survey, U.S. Department of the Interior. 2014. Available online: https://minerals.usgs.gov/minerals/pubs/mcs/2014/mcs2014.pdf (accessed on 31 March 2017).
- Gray, T. The Elements, a Visual Exploration of Every Known Atom in the Universe; Black Dog and Leventhal Publishers: New York, NY, USA, 2012; 240p. [Google Scholar]
- DOE. U.S. Department of Energy Critical Materials Strategy. 2011. Available online: https://energy.gov/sites/prod/files/DOE_CMS2011_FINAL_Full.pdf (accessed on 6 August 2017).
- Golev, A.; Scott, M.; Erskine, P.D.; Ali, S.H.; Ballantyne, G.R. Rare earth supply chains: Current status, constraints and opportunities. Resour. Policy 2014, 41, 52–59. [Google Scholar] [CrossRef]
- Goonan, T.G. Rare Earth Elements—End Use and Recyclability; Scientific Investigations Report 2011–5094; United States Geologic Survey. Available online: https://pubs.usgs.gov/sir/2011/5094/pdf/sir2011-5094.pdf (accessed on 6 August 2017).
- Lewis, L.H.; Jimenez-Villacorta, F. Perspectives on permanent magnet materials for energy conversion and power generation. Metall. Mater. Trans. A 2013, 44A, 2–20. [Google Scholar] [CrossRef]
- Graso, V.B. Rare Earth Elements in National Defense: Background, Oversight Issues and Options for Congress; Congressional Research Service Report 7-5700, R41744; Congressional Research Service, 2013; Available online: https://fas.org/sgp/crs/natsec/R41744.pdf (accessed on 21 May 2017).
- Carrettin, S.; Concepcion, P.; Corma, A.; Nieto, J.M.L.; Puntes, V.F. Nanocrystalline CeO2 increases the activity of Au for CO oxidation by two orders of magnitude. Angew. Chem. Int. Ed. 2004, 43, 2538–2540. [Google Scholar] [CrossRef] [PubMed]
- Trovarelli, A.; de Leitenburg, C.; Boaro, M.; Dolcetti, G. The utilization of ceria in industrial catalysis. Catal. Today 1999, 50, 353–367. [Google Scholar] [CrossRef]
- Gester, S.; Metz, P.; Zierau, O.; Vollmet, G. An efficient synthesis of the potent phytoestrogens 8-prenylnaringenin and 6-(1,1-dimethlyallyl)naringenin by europium(III)-catlyzed Claisen rearrangement. Tetrahedron 2001, 57, 1015–1018. [Google Scholar] [CrossRef]
- Amin, S.; Voss, D.A.; Horrocks, W.D.; Lake, C.H.; Churchill, M.R.; Morrow, J.R. Laser-induced luminescence studies and crystal structure of the Europium(III) complex of 1,4,7,10-tetrakis(carbamoylmethyl)-1,4,7,10-tetraazacyclododecane—The link between phosphate diester binding and catalysis by lanthanide(III) macrocyclic complexes. Inorg. Chem. 1995, 34, 3294–3300. [Google Scholar] [CrossRef]
- Gai, S.; Li, C.; Yang, P.; Lin, J. Recent progress in rare earth micro/nanocrystals: Soft chemical synthesis, luminescent properties, and biomedical applications. Chem. Rev. 2014, 114, 2343–2389. [Google Scholar] [CrossRef] [PubMed]
- Gambogi, J. Rare Earths in Mineral Commodity summaries USGS. 2016. Available online: https://minerals.usgs.gov/minerals/pubs/commodity/rare_earths/mcs-2016-raree.pdf (accessed on 12 May 2017).
- United Nations. UNFPA Revision of Population Prospects; United Nations, 2015; Available online: https://esa.un.org/unpd/wpp/publications/files/key_findings_wpp_2015.pdf (accessed on 8 May 2017).
- Ali, S.H.; Giurco, D.; Arndt, N.; Nickless, E.; Borwn, G.; Demetriades, A.; Durrheim, R.; Enriquez, M.A.; Kinnaird, J.; Littleboy, A.; et al. Mineral supply for sustainable development requires resource governance. Nature 2017, 543, 367–372. [Google Scholar] [CrossRef] [PubMed]
- Badescu, V. Moon: Prospective Energy and Material Resources; Springer: New York, NY, USA, 2012; 771p. [Google Scholar]
- Crawford, I.A.; Joy, K.H. Lunar exploration: opening a window into the history and evolution of the inner Solar System. Philos. Trans. R. Soc. A 2014, 372, 20130315. [Google Scholar] [CrossRef] [PubMed]
- Crawford, I.A. Lunar resources: A Review. Prog. Phys. Geogr. 2015, 39, 137–167. [Google Scholar] [CrossRef]
- Crawford, I.A.; Anand, M.; Cockell, C.S.; Falcke, H.; Green, D.A.; Jaumann, R.; Wieczorek, M.A. Back to the Moon: The scientific rationale for resuming lunar surface exploration. Planet. Space Sci. 2012, 74, 3–14. [Google Scholar] [CrossRef]
- Duke, M.B.; Gaddis, L.R.; Taylor, G.J.; Schmitt, H.H. Development of the Moon. In New Views on the Moon; Jolliff, B., Wieczorek, M., Shearer, C., Neal, C., Eds.; Mineralogical Society of America: Chantilly, VA, USA, 2006; Volume 60. [Google Scholar]
- Aldridge, E.C.; Fiorina, C.S.; Jackson, M.P.; Leshin, L.A.; Lyles, L.L.; Spudis, P.D.; Tyson, N.D.; Walker, R.S.; Zuber, M.T. A Journey to Inspire, Innovate and Discover, Report of the President’s Commission on Implementation of United States Space Exploration Policy; U.S. Government Printing Office: Washington, DC, USA, 2004.
- Carpenter, J.; Fisackerly, R.; Houdou, B. Establishing lunar resource viability. Space Policy 2016, 37, 52–57. [Google Scholar] [CrossRef]
- Chamberlain, P.G.; Taylor, L.A.; Podnieks, E.R.; Miller, R.J. A review of possible mining applications in space. In Resources of Near-Earth Space; Lewis, J.S., Matthews, M.S., Guerrieri, M.L., Eds.; University of Arizona Press: Tucson, AZ, USA, 1992; pp. 51–68. [Google Scholar]
- Shishko, R.; Fradet, R.; Saydam, S.; Tapia-Cortez, C.; Dempster, A.; Coulton, J.; Do, S. An Integrated Economics Model for ISRU in Support of a Mars Colony—Initial Results Report. In Proceedings of the 10th Symposium on Space Resource Utilization, AIAA SciTech Forum (AIAA 2017-0422), Grapevine, TX, USA; 2015. Available online: https://arc.aiaa.org/doi/pdf/10.2514/6.2017-0422 (accessed on 6 August 2017).
- Wang, K.-T.; Lemouhna, P.N.; Tang, Q.; Li, W.; Cui, X.-M. Lunar regolith can allow the synthesis of cement materials with near-zero water consumption. Gondwana Res. 2017, 44, 1–6. [Google Scholar] [CrossRef]
- ASI; BNSC; CNES; CNSA; CSA; CSIRO; DLR; ESA; ISRO; JAXA; KARL; NASA; NSAU; Roscosmos. The Global Exploration Strategy: The Framework for Coordination, 2007. Available online: https://www.nasa.gov/pdf/296751main_GES_framework.pdf (accessed on 1 May 2017).
- Wood, J.A.; Dickey, J.S.; Marvin, U.B.; Powell, B.M. Lunar anorthosites and a geophysical model of the Moon. In Proceedings of the Apollo 11 Lunar Science Conference, Houston, TX, USA, 5–8 January 1970; Pergamon: New York, NY, USA, 1970; pp. 965–988. [Google Scholar]
- Smith, J.V.; Andersen, A.T.; Newton, R.C.; Olsen, E.J.; Crewe, A.V.; Isaacson, M.S.; Johnson, D.; Wyllie, P.J. Petrologic history of the Moon inferred from petrography, mineralogy, and petrogenesis of the Apollo 11 rocks. In Proceedings of the Apollo 11 Lunar Science Conference, Houston, TX, USA, 5–8 January 1970; Pergamon Press: New York, NY, USA, 1970; pp. 897–926. [Google Scholar]
- Taylor, S.R.; Jakes, P. The geochemical evolution of the moon. Proceedings of 5th Lunar Science Conference, Houston, TX, USA, 18–22 March 1974; pp. 1287–1305. [Google Scholar]
- Taylor, S.R. Lunar Science: A Post-Apollo View; Pergamon Press: New York, NY, USA, 1975; p. 372. [Google Scholar]
- Taylor, S.R. Structure and evolution of the moon. Nature 1979, 281, 105–110. [Google Scholar] [CrossRef]
- Shirley, D.N. A partially molten magma ocean model. J. Geophys. Res. 1983, 88, A519–A527. [Google Scholar] [CrossRef]
- Warren, P.W. The lunar magma ocean concept and lunar evolution. Ann. Rev. Earth Planet. Sci. 1985, 13, 201–240. [Google Scholar] [CrossRef]
- Heiken, G.H.; Vaniman, D.T.; French, B.M. (Eds.) Lunar Sourebook a User’s Guide to the Moon; Cambridge University Press and the Lunar Planetary Institute: Cambridge, UK, 1991; 736p. [Google Scholar]
- Snyder, G.A.; Taylor, L.A.; Neal, C.R. A chemical model for generating the sources of mare basalts: combined equilibrium and fractional crystallization of the lunar magmasphere. Geochim. Cosmochim. Acta 1992, 56, 3809–3823. [Google Scholar] [CrossRef]
- Papike, J.J.; Ryder, G.; Shearer, C.K. Lunar Materials. In Planetary Materials, Reviews in Mineralogy; Papike, J.J., Ed.; Mineralogical Society of America: Washington, DC, USA, 1998; Volume 36, pp. 5.1–5.23. [Google Scholar]
- Jolliff, B.L.; Wieczorek, M.A.; Shearer, C.K.; Neal, C.R. (Eds.) New Views of the Moon; Reviews in Mineralogy and Geochemistry, Mineralogical Society of America: Chantilly, VA, USA, 2006; Volume 60, p. 721. [Google Scholar]
- Longhi, J. Petrogenesis of the picritic mare magmas: on constraints on the extent of early lunar differentiation. Geochim. Cosmochim. Acta 2006, 70, 5919–5934. [Google Scholar] [CrossRef]
- Touboul, M.; Kleine, T.; Bourdon, B.; Palme, H.; Wieler, R. Late formation and prolonged differentiation of the Moon inferred from W isotopes in lunar metals. Nature 2007, 450, 1206–1209. [Google Scholar] [CrossRef] [PubMed]
- Halliday, A.N. A young Moon-forming giant impact 70–110 million years ago accompanied by late-stage mixing, core formation and degassing of the Earth. Philos. Trans. R. Soc. 2008, 366, 4163–4181. [Google Scholar] [CrossRef] [PubMed]
- Brandon, A.D.; Lapen, T.J.; Debaille, V.; Beard, B.K.; Rankenburg, K.; Neal, C. Re-evaluating 142Nd/144Nd in lunar mare basalts with the implications for the early evolution and bulk Sm/Nd of the Moon. Geochim. Cosmochim. Acta 2009, 73, 6421–6445. [Google Scholar] [CrossRef]
- Kleine, T.; Touboul, M.; Bourdon, B.; Nimmo, F.; Mezger, K.; Palme, H.; Jacobsen, S.B.; Yin, Q.-Z.; Halliday, A.N. Hf-W chronology of the accretion and early evolution of asteroids and terrestrial planets. Geochim. Cosmochim. Acta 2009, 73, 5150–5188. [Google Scholar] [CrossRef]
- Borg, L.E.; Connelly, J.N.; Boyet, M.; Carlson, R.W. Chronological evidence that the Moon is either young or did not have a global magma ocean. Science 2011, 477, 70–72. [Google Scholar] [CrossRef] [PubMed]
- Elardo, S.M.; Draper, D.S.; Shearer, C.K. Lunar magma ocean crystallization revisited: bulk composition, early cumulate mineralogy and the source regions of the Mg-suite. Geochim. Cosmochim. Acta 2011, 75, 3024–3045. [Google Scholar] [CrossRef]
- Elkins-Tanton, L.T. Magma oceans in the inner solar system. Ann. Rev. Earth Planet. Sci. 2012, 40, 113–139. [Google Scholar] [CrossRef]
- Jacobson, S.A.; Mobirdelli, A.; Rayond, S.N.; O’Brien, D.P.; Walsh, K.J.; Rubie, D.C. Highly siderophile elements in Earth’s mantle as a clock for the Moon-forming impact. Nature 2014, 508, 84–87. [Google Scholar] [CrossRef] [PubMed]
- McLeod, C.L.; Brandon, A.D.; Armytage, R.M.G. Constraints on the formation age and evolution of the Moon from 142Nd-143Nd systematics of Apollo 12 basalts. Earth Planet. Sci. Lett. 2014, 396, 179–189. [Google Scholar] [CrossRef]
- McLeod, C.L.; Brandon, A.D.; Fernandes, V.A.; Peslier, A.H.; Fritz, J.; Lapen, T.; Shafer, J.T.; Butcher, A.R.; Irving, A.J. Constraints on formation and evolution of the lunar crust from feldspathic granultic breccias NWA 3163 and 4881. Geochim. Cosmochim. Acta 2016, 187, 350–374. [Google Scholar] [CrossRef]
- Borg, L.E.; Gaffney, A.M.; Shearer, C.K. A review of lunar chronology revealing a preponderance of 4.34–4.37 Ga ages. Meteorit. Planet. Sci. 2015, 50, 715–732. [Google Scholar] [CrossRef]
- Boyce, J.W.; Treiman, A.H.; Guan, Y.; Ma, C.; Eiler, J.M.; Gross, J.; Greenwood, J.P.; Stolper, E.M. The chlorine isotope fingerprint of the lunar magma ocean. Sci. Adv. 2015, 1, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Barr, A.C. On the origin of the Earth’s Moon. J. Geophys. Res. Planets 2016, 121, 1573–1601. [Google Scholar] [CrossRef]
- Sivakumar, V.; Neekakantan, R.; Santosh, M. Lunar surface mineralogy using hyperspectral data: Implications for primordial crust in the Earth-Moon system. Geosci. Front. 2017, 8, 457–465. [Google Scholar] [CrossRef]
- Sun, C.; Graff, M.; Liang, Y. Trace element partitioning between plagioclase and silicate melt: The importance of temperature and plagioclase composition, with implications for terrestrial and lunar magnetism. Geochim. Cosmochim. Acta 2017, 206, 273–295. [Google Scholar] [CrossRef]
- Google LUNAR XPRIZE, 2017. Available online: http://lunar.xprize.org/ (accessed on 4 June 2017).
- Moon Express. 2016. Available online: http://www.moonexpress.com/files/moon-express-press-kit.pdf (accessed on 4 June 2017).
- Caminiti, S. The Billionaire’s Race to Harness the Moon’s Resources. Available online: http://www.cnbc.com/2014/04/02/the-global-race-to-harness-the-Moon’s-resources.html (accessed on 5 August 2017).
- Goins, N.R.; Dainty, A.M.; Toksoz, M.N. Lunar seismology: The internal structure of the Moon. J. Geophys. Res. 1981, 86, 5061–5074. [Google Scholar] [CrossRef]
- Wieczorek, M.A.; Jolliff, B.L.; Khan, A.; Pritchard, M.E.; Weiss, B.P.; Williams, J.G.; Hood, L.L.; Righter, K.; Neal, C.R.; Shearer, C.K.; et al. The Constitution and Structure of the Lunar Interior. Rev. Mineral. Geochem. 2006, 60, 221–364. [Google Scholar] [CrossRef]
- Weber, R.C.; Lin, P.-Y.; Garnero, E.J.; Williams, Q.; Lognonne, P. Seismic Detection of the Lunar Core. Science 2011, 331, 309–312. [Google Scholar] [CrossRef] [PubMed]
- Jaumann, R.; Hiesinger, H.; Anand, M.; Crawford, I.A.; Wagner, R.; Sohl, F.; Jolliff, B.L.; Scholten, F.; Knapmeyer, M.; Hoffman, H.; et al. Geology, geochemistry and geophysics of the Moon: Status of current understanding. Planet. Space Sci. 2012, 74, 15–41. [Google Scholar] [CrossRef]
- Haskin, L.A.; Colson, R.O.; Vaniman, D.T.; Gillett, S.L. A geochemical assessment of possible lunar ore formation. In Resources of Near-Earth Space; Lewis, J.S., Matthews, M.S., Guerrieri, M.L, Eds.; University of Arizona Press: Tucson, AZ, USA, 1993; 977p. [Google Scholar]
- Solomon, S.C.; Toksoz, M.N. Internal Constitution and Evolution of the Moon. Phys. Earth Planet. Inter. 1973, 7, 15–38. [Google Scholar] [CrossRef]
- Elkins-Tanton, L.T.; Burgess, S.; Yin, Q.-Z. The lunar magma ocean: Reconciling the solidification process with lunar petrology and geochronology. Earth Planet. Sci. Lett. 2011, 304, 326–336. [Google Scholar] [CrossRef]
- McLeod, C.L. Lunar Magma Ocean, Size. In Encyclopedia of Lunar Science; Cudnik, B., Ed.; Springer: New York, NY, USA, 2016. [Google Scholar] [CrossRef]
- Wieczorek, M.A.; Neumann, G.A.; Nimmo, F.; Kiefer, W.S.; Taylor, G.J.; Melosh, H.J.; Phillips, R.J.; Solomon, S.C.; Andrews-Hanna, J.C.; Asmar, S.W.; et al. The Crust of the Moon as Seen by GRAIL. Science 2013, 339, 671–675. [Google Scholar] [CrossRef] [PubMed]
- Petro, N.E.; Jolliff, B.L. Thin Crust in the South Pole Aitken Basin and Samples from the Mantle? Implications for the South Pole-Aitken Basin Sampling in light of Recent GRAIL Results. In Proceedings of the 44th Lunar Planetary Science Conference, Woodlands, TX, USA, 18–22 March 2013; p. 2724. [Google Scholar]
- Green, D.H.; Ringwood, A.E. The stability fields of aluminous pyroxene peridotite and garnet peridotite. Earth Planet. Sci. Lett. 1967, 3, 151–160. [Google Scholar] [CrossRef]
- Longhi, J. A model of early lunar differentiation. In Proceedings of the 11st Lunar and Planetary Science Conference, Houston, TX, USA, 17–21 March 1980; pp. 289–315. [Google Scholar]
- Shearer, C.K.; Papike, J.J. Magmatic evolution of the Moon. Am. Mineral. 1999, 84, 1469–1494. [Google Scholar] [CrossRef]
- Hiesinger, H.; Jaumann, R.; Neukum, G.; Head, J.W., III. Ages of mare basalts on the lunar nearside. J. Geophys. Res. 2000, 105, 29239–29275. [Google Scholar] [CrossRef]
- Braden, S.E.; Stopar, J.S.; Robinson, M.S.; Lawrence, S.J.; van der Bogert, C.H.; Hiesenger, H. Evidence for basaltic volcanism on the Moon within the past 100 million years. Nat. Geosci. 2014, 7, 787–791. [Google Scholar] [CrossRef]
- Green, D.H.; Ringwood, A.E.; Ware, N.G.; Hibberson, A.; Kiss, E. Experimental petrology and petrogenesis of Apollo 12 basalts. In Proceedings of the 2nd Lunar Science Conference, Houston, TX, USA, 11–14 January 1971; pp. 601–615. [Google Scholar]
- Green, D.H.; Ware, N.G.; Hibberson, A.; Major, A. Experimental petrology of Apollo 12 mare basalts, Part 1, sample 12009. Earth Planet. Sci. Lett. 1971, 13, 85–96. [Google Scholar] [CrossRef]
- Philpotts, J.A.; Schnetzler, C.C.; Nava, D.D.; Bottino, M.L.; Fullagar, P.D.; Thomas, H.H.; Schumann, S.; Kouns, C.W. Apollo 14: some geochemical aspects. Proceedings of Third Lunar Planetary Science Conference, Houston, TX, USA, 10–13 January 1972; The MIT Press; pp. 1293–1305. Available online: http://adsabs.harvard.edu/full/1972LPSC....3.1293P (accessed on 28 May 2017).
- Haskin, L.A.; Lindstrom, M.M.; Salpas, P.A.; Lindstrom, D. On compositional variations among lunar anorthosites. In Proceedings of the 12th Lunar Science Conference, Houston, TX, USA, 16–20 March 1981; pp. 41–66. [Google Scholar]
- Morse, S.A. Adcumulus growth of anorthosite at the base of the lunar crust. In Proceedings of the 13th Lunar and Planetary Science Conference, Houston, TX, USA, 15–19 March 1982; pp. A10–A18. [Google Scholar]
- Ryder, G. Lunar anorthosite 60025, the petrogenesis of lunar anorthosites, and the bulk composition of the Moon. Geochem. Cosmochim. Acta 1982, 46, 1591–1601. [Google Scholar] [CrossRef]
- Longhi, J. Origin of Picritic Green Glass Magmas by Polybaric Fractional Fusion. Proc. Lunar Planet. Sci. 1992, 22, 343–353. [Google Scholar]
- Warren, P.W.; Wasson, J.T. Compositional-petrographic investigation of pristine nonmare rocks. In Proceedings of the 9th Lunar and Planetary Science Conference, Houston, TX, USA, 13–17 March 1978; pp. 185–217. [Google Scholar]
- Yanhao, L.; Tronche, E.J.; Steenstra, E.S.; van Westrenen, W. Experimental constraints on the solidification of a nominally dry lunar magma ocean. Earth Planet. Sci. Lett. 2017, 471, 104–116. [Google Scholar]
- Alibert, C.; Norman, M.D.; McCulloch, M.T. An ancient Sm-Nd age for a ferroan noritic anorthosite clast from lunar breccia 67016. Geochim. Cosmochim. Acta 1994, 58, 2921–2926. [Google Scholar] [CrossRef]
- Borg, L.E.; Gaffney, A.M.; Shearer, C.K.; DePaolo, D.J.; Hutcheon, I.D.; Owens, T.L.; Ramon, E.; Brennecka, G. Mechanisms for incompatible-element enrichment on the Moon deduced from the lunar basaltic meteorite Northwest Africa 032. Geochim. Cosmochim. Acta 2009, 73, 3963–3980. [Google Scholar] [CrossRef]
- Meyer, J.; Elkins Tanton, L.T.; Wisdom, J. Coupled thermal-orbital evolution of the early Moon. Icarus 2010, 208, 1–10. [Google Scholar] [CrossRef]
- Taylor, D.J.; McKeegan, K.D.; Harrison, T.M. Lu-Hf zircon evidence for rapid lunar differentiation. Earth Planet. Sci. Lett. 2009, 279, 157–264. [Google Scholar] [CrossRef]
- Gaffney, A.M.; Borg, L.E. A Young Age for KREEP Formation Determined from Lu-Hf Isotope Systematics of KREEP Basalts and Mg-Suite Samples. In Proceedings of the 44th Lunar and Planetary Science Conference, Woodlands, TX, USA, 18–22 March 2013; p. 1719. [Google Scholar]
- Smith, J.V.; Steele, I.M. Lunar mineralogy: A heavenly detective story. Part II. Am. Mineral. 1976, 61, 1059–1116. [Google Scholar]
- Meyer, C. NASA Lunar Petrographic Educational Thin Section Set. 2003. Available online: https://curator.jsc.nasa.gov/lunar/letss/mineralogy.pdf (accessed on 18 March 2017).
- Meyer, C. Lunar Sample Compendium; Lunar Planetary Institute: Houston, TX, USA, 2009. [Google Scholar]
- Frondel, J.W. Lunar Mineralogy; Wiley-Interscience: New York, NY, USA, 1975; pp. 84–87. [Google Scholar]
- Yazawa, Y.; Yamaguchi, A.; Takeda, H. Lunar Minerals and Their Resource Utilization with Particular Reference to Solar Power Satellite and Potential Roles for Humic substances for Lunar Agriculture. In Moon, Prospective Energy and Material Resources; Badescu, V., Ed.; Springer: New York, NY, USA, 2012; 771p. [Google Scholar]
- Dixon, J.R.; Papike, J.J. Petrology of anorthosites from the Descartes region of the moon: Apollo 16. In Proceedings of the 6th Lunar Science Conference, Houston, TX, USA, 17–21 March 1975; pp. 263–291. [Google Scholar]
- James, O.B.; Lindstrom, M.M.; McGee, J.J. Lunar ferroan anorthosite 60025: Petrology and chemistry of mafic lithologies. In Proceedings of the 21st Lunar Planetary Science Conference, Houston, TX, USA, 12–16 March 1990; Lunar Planetary Institute: Houston, TX, USA. [Google Scholar]
- Brown, G.M.; Emeleus, C.H.; Holland, G.J.; Peckett, A.; Phillips, R. Mineral-chemical variations in Apollo 14 and Apollo 15 basalts and granitic fractions. In Proceedings of the 3rd Lunar Science Conference, Houston, TX, USA, 10–13 January 1972; pp. 147–157. [Google Scholar]
- Papike, J.J.; Hodges, F.N.; Bence, A.E.; Cameron, M.; Rhodes, J.M. Mare basalts: Crystal chemistry, mineralogy and petrology. Rev. Geophys. Space Phys. 1976, 14, 475–540. [Google Scholar] [CrossRef]
- McGee, P.E.; Warner, J.L.; Simonds, C.H. Introduction to the Apollo Collections. Part I: Lunar Igneous Rocks; Curators Office, JSC, 1977; Available online: http://www.lpi.usra.edu/lunar/surface/apolloCollectionsPartI.pdf (accessed on 28 May 2017).
- Lunar Sample Atlas. Available online: http://www.lpi.usra.edu/lunar/samples/atlas/thin_sections/ (accessed on 12 March 2017).
- Warren, P.H.; Wasson, J.T. The Origin of KREEP. Rev. Geophys. Space Phys. 1979, 17, 73–88. [Google Scholar] [CrossRef]
- Shearer, C.K.; Hess, P.C.; Wieczorek, M.A.; Prichard, M.E.; Parmentier, E.M.; Borg, L.E.; Longhi, J.; Elkins-Tanton, L.T.; Neal, C.R.; Antonenko, I.; et al. Thermal and Magmatic Evolution of the Moon. Rev. Mineral. Geochem. 2006, 60, 365–518. [Google Scholar] [CrossRef]
- Lawrence, D.J.; Feldman, W.C.; Barraclough, B.L.; Elphic, R.C.; Maurice, S.; Binder, A.B.; Miller, M.C.; Prettyman, T.H. High resolution measurements of absolute thorium abundances on the lunar surface from the Lunar Prospector gamma-ray spectrometer. Geophys. Res. Lett. 1999, 26, 2681–2684. [Google Scholar] [CrossRef]
- Haskin, L.A.; Gillis, J.J.; Korotev, R.L.; Jolliff, B.L. The Materials of the lunar Procellarum KREEP Terrane: A synthesis of data from geomorphological mapping, remote-sensing, and sample analysis. J. Geophys. Res. 2000, 105, 20403–20415. [Google Scholar] [CrossRef]
- Jolliff, B.L.; Gillis, J.J.; Haskin, L.A.; Korotev, R.L.; Wieczorek, M.A. Major lunar crustal terranes: Surface expressions and crust-mantle origins. J. Geophys. Res. Planets 2000, 105, 4197–4216. [Google Scholar] [CrossRef]
- Global Map of the Element Thorium. Available online: https://solarsystem.nasa.gov/galleries/global-map-of-the-element-thorium (accessed 21 August 2017).
- Wieczorek, M.A.; Phillips, R.J. Lunar multi-ring basins and the cratering process. Icarus 1999, 139, 246–259. [Google Scholar] [CrossRef]
- Lawrence, D.J.; Feldman, W.C.; Barraclough, B.L.; Binder, A.B.; Elphic, R.C.; Maurice, S.; Miller, M.C.; Prettyman, T.H. Thorium abundances on the lunar surface. J. Geophys. Res. 2000, 105, 20307–20331. [Google Scholar] [CrossRef]
- Zou, Y.; Xu, L.; Ouyang, Z. KREEP Rocks. Chin. J. Geochem. 2004, 23, 65–70. [Google Scholar]
- Neal, C.L.; Taylor, L.A. Definition of a Pristine, Unadulterated urKREEP Composition Using the “K-FRAC/REEP-FRAC” Hypothesis. Abstracts of the Lunar and Planetary Science Conference. 1989, Volume 20, pp. 772–773. Available online: http://adsabs.harvard.edu/full/1989LPI....20..772N (accessed on 27 May 2017).
- Neal, C.L.; Taylor, L.A. Metasomatic products of the lunar magma ocean: The role of KREEP dissemination. Geochim. Cosmochim. Acta 1989, 53, 529–541. [Google Scholar] [CrossRef]
- Thompson, R.N. British Tertiary volcanic province. Scott. J. Geol. 1982, 18, 49–107. [Google Scholar] [CrossRef]
- Lindstrom, M.M.; Knapp, S.A.; Shervais, J.A.; Taylor, L.A. Magnesian anorthosites and associated troctolites and dunite in Apollo 14 breccias. In Proceedings of the 15th Lunar and Planetary Science Conference, Houston, TX, USA, 12–16 March 1984; pp. C41–C49. [Google Scholar]
- Rudnick, R.L.; Gao, S. Composition of the Continental Crust. Treatise Geochem. 2003, 3, 1–64. [Google Scholar]
- Warren, P.H.; Jerde, E.A.; Kalleymeyn, G.W. Pristine moon rocks: A “large” felsite and metal-rich ferroan anorthosite. In Proceedings of the 17th Lunar and Planetary Science Conference, Houston, TX, USA.
- Jolliff, B.L.; Wadhwa, M. The distribution of Rare Earth Elements between Lunar apatite and whitlockite. In Proceedings of the 23rd Lunar and Planetary Science Conference, Houston, TX, USA, 16–20 March 1992; pp. 625–626. [Google Scholar]
- Kartashov, P.M.; Bogatikov, O.A.; Mokhov, A.V.; Gorshkov, A.I.; Ashikhmina, N.A.; Magazina, L.O.; Koporulina, E.V. Lunar Monazites. In Doklady Earth Sciences; MAIK Nauka/Interperiodica, 2006; Volume 407A, pp. 498–502. Available online: https://link.springer.com/article/10.1134/S1028334X06030342 (accessed on 21 August 2017).
- Jolliff, B.L.; Hughes, J.M.; Freeman, J.J.; Ziegler, R.A. Crystal chemistry of lunar merrillite and comparison to other meteoritic and planetary suites of whitlockite and merrillite. Am. Mineral. 2006, 91, 1583–1595. [Google Scholar] [CrossRef]
- Jolliff, B.L. Fragments of Quartz Monzodiorite and Felsite in Apollo 14 Soil Particles. In Proceedings of the Lunar and Planetary Science Conference, Houston, TX, USA, 12–16 March 1990; Volume 21, pp. 101–118. [Google Scholar]
- Jolliff, B.L. Large-Scale Separation of K-frac and REEP-frac in the source Regions of Apollo Impact-Melt Breccias, and a Revised Estimate of the KREEP Composition. Int. Geol. Rev. 1998, 40, 916–935. [Google Scholar] [CrossRef]
- Meyer, C.; Yang, S.V. Tungsten-bearing yttrobetafite in lunar granophyre. Am. Mineral. 1988, 73, 1420–1425. [Google Scholar]
- Ramdohr, P.; El Gorsey, A. Opaque minerals of the lunar rocks and dust from Mare Tranquillitatis. Science 1970, 167, 615–618. [Google Scholar] [CrossRef] [PubMed]
- Cameron, E.N. Opaque minerals in certain lunar rocks from Apollo 11. In Proceedings of the Apollo 11 Lunar Science Conference, Houston, TX, USA, 5–8 January 1970; pp. 221–245. [Google Scholar]
- Lovering, J.F.; Wark, D.A.; Reid, A.F.; Ware, N.G.; Keil, K.; Prinz, M.; Bunch, T.E.; El Gorsey, A.; Ramdohr, P.; Brown, G.M.; et al. Tranquillityite: A new silicate mineral from Apollo 11 and Apollo 12 basaltic rocks. In Proceedings of the Lunar Science Conference, Houston, TX, USA, 11–14 January 1971; Volume 2, pp. 39–45. [Google Scholar]
- Rasmussen, B.; Fletcher, I.R.; Gregory, C.J.; Muhling, J.R.; Suvorova, A.A. Tranquillityite: The last lunar mineral comes down to Earth. Geology 2012, 40, 83–86. [Google Scholar] [CrossRef]
- Haskin, L.A.; Warren, P. Lunar Chemistry. In The Lunar Sourcebook: A User’s Guide to the Moon; Cambridge University Press: Cambridge, UK, 1991; pp. 357–474. [Google Scholar]
- McKay, D.S.; Heiken, G.; Basu, A.; Blanford, G.; Simon, S.; Reedy, R.; French, B.M.; Papike, J. The lunar regolith. In Lunar Sourcebook; Heiken, G.H., Vaniman, D.T., French, B.M., Eds.; Cambridge University Press: Cambridge, UK, 1991; pp. 285–356. [Google Scholar]
- Blair, B. Quantitative Approaches to Lunar Economic Analysis. In Proceedings of the Annual Meeting of the Lunar Exploration Analysis Group (LEAG), Houston, TX, USA; 2009. Available online: http://www.lpi.usra.edu/meetings/leag2009/presentations/Day-1%20PM/02-15_Blair.pdf (accessed on 6 August 2017).
- Santiago-Maldonado, E.; Gleaton, J.; Devor, R.; Captain, J. Creating Methane from Plastic: Recycling at a Lunar Outpost. In Proceedings of the 48th AIAA Aerospace Sciences Meeting including the New Horizons Forum and Aerospace Exposition, Orlando, FL, USA, 4–7 January 2010; American Institute of Aeronautics and Astronautics: Reston, VA, USA, 2010; Volume 21, pp. 18271–18278. [Google Scholar]
- Hintze, P.E.; Quintana, S. Building a Lunar or Martian Launch Pad with In Situ Materials: Recent Laboratory and Field Studies. J. Aerosp. Eng. 2013, 26, 134–142. [Google Scholar] [CrossRef]
- Schrunk, D.; Sharp, B.; Cooper, B.; Thangavelu, M. The Moon: Resources, Future Development, and Settlement; Springer: Chichester, UK, 2008. [Google Scholar]
- Anand, M.; Crawford, I.A.; Balat-Pichelin, M.; Abanades, S.; van Westrenen, W.; Péraudeau, G.; Jaumann, R.; Seboldt, W. A brief review of chemical and mineralogical resources on the Moon and likely in situ resource utilization (ISRU) applications. Planet. Space Sci. 2012, 74, 42–48. [Google Scholar] [CrossRef]
- Fegley, B.; Swindle, T.D. Lunar volatiles: Implications for lunar resource utilization. In Resources of Near Earth Space; Lewis, J., Matthews, M.S., Guerrieri, M.L., Eds.; Tucson University Press: Tucson, AZ, USA, 1993; pp. 367–426. [Google Scholar]
- Fa, W.; Jin, Y.-Q. Quantitative estimation of helium-3 spatial distribution in the lunar regolith layer. Icarus 2007, 190, 15–23. [Google Scholar] [CrossRef]
- Feldman, W.C.; Maurice, S.; Binder, A.B.; Barraclough, B.L.; Elphic, R.C.; Lawrence, D.J. Fluxes of fast and epithermal neutrons from Lunar Prospector: Evidence for water ice at the lunar poles. Science 1998, 281, 1496–1500. [Google Scholar] [CrossRef] [PubMed]
- Feldman, W.C.; Maurice, S.; Lawrence, D.J.; Little, R.C.; Lawson, S.L.; Gasnault, O.; Wiens, R.C.; Barraclough, B.L.; Elphic, R.C.; Prettyman, T.H.; et al. Evidence for water ice near the lunar poles. J. Geophys. Res. 2011, 106, 23231–23252. [Google Scholar] [CrossRef]
- Colaprete, A.; Schultz, P.; Heldmann, J.; Wooden, D.; Shirley, M.; Ennico, K.; Hermalyn, B.; Marshall, W.; Ricco, A.; Elphic, R.C.; et al. Detection of Water in the LCROSS Ejecta Plume. Science 2010, 330, 463–468. [Google Scholar] [CrossRef] [PubMed]
- USGS. 2016. Available online: https://water.usgs.gov/edu/qa-home-percapita.html (accessed on 19 March 2017).
- Taylor, L.A.; Carrier, W.D. Oxygen production on the Moon: An overview and evaluation. In Resources of Near Earth Space; Lewis, J., Matthews, M.S., Guerrieri, M.L., Eds.; Tucson University: Tucson, AZ, USA, 1993. [Google Scholar]
- Schwandt, C.; Dimitrov, A.T.; Fray, D.J. High-yield synthesis of multi-walled carbon nanotubes from graphite by molten salt electrolysis. Carbon 2012, 50, 1311–1315. [Google Scholar] [CrossRef]
- Benaroya, H.; Mottaghi, S.; Porter, Z. Magnesium as an ISRU-derived resource for lunar structures. J. Aerosp. Eng. 2013, 26, 152–159. [Google Scholar] [CrossRef]
- Mottaghi, S.; Benaroya, H. Design of a Lunar Surface Structure. I: Design Configuration and Thermal Analysis. J. Aerosp. Eng. 2015, 28, 0401405. [Google Scholar] [CrossRef]
- NASA. Metals from Regolith. 2012. Available online: https://isru.nasa.gov/MetalsfromRegolith.html (accessed on 7 August 2017).
- Lucey, P.G.; Taylor, G.J.; Malaret, E. Abundance and distribution of iron on the Moon. Science 1995, 268, 1150–1153. [Google Scholar] [CrossRef] [PubMed]
- Meyers, C.; Toutanji, H. Analysis of Lunar-Habitat Structure Using Waterless Concrete and Tension glass Fibers. J. Aerosp. Eng. 2007, 20, 220–226. [Google Scholar] [CrossRef]
- Masafumi, I.; Gertsch, L. Excavation of Lunar Regolith with Large Grains by Rippers for Improved Excavation Efficiency. J. Aerosp. Eng. 2013, 26, 97–104. [Google Scholar]
- Wilhelm, S.; Curbach, M. Review of possible mineral materials and production techniques for a building material on the moon. Struct. Concr. 2014, 15, 419–428. [Google Scholar] [CrossRef]
- Turkevich, A. Average chemical composition of the lunar surface. In Proceedings of the 4th Lunar Science Conference, Houston, TX, USA, 5–8 March 1973; Volume 2, pp. 1119–1168. [Google Scholar]
- Papike, J.J.; Simon, S.B.; Laul, J.C. Lunar Regolith: Chemistry, Mineralogy, and Petrology. Rev. Geophys. 1982, 20, 761–826. [Google Scholar] [CrossRef]
- Lucey, P.G.; Korotev, R.L.; Gillis, J.J.; Taylor, L.A.; Lawrence, D.; Campbell, B.A.; Elphic, R.; Feldman, B.; Hood, L.L.; Hunten, D.; et al. Understanding the lunar surface and space-Moon interaction. Rev. Mineral. Geochem. 2006, 60, 82–219. [Google Scholar] [CrossRef]
- Ceccanti, F.; Dini, E.; De Kestelier, X.; Colla, V.; Pambaguian, L. 3D printing technology for a moon outpost exploiting lunar soil. In Proceedings of the 61st International Astronautical Congress, Prague, Czech Republic, 27 September–1 October 2010; Available online: http://esmat.esa.int/Publications/Published_papers/IAC-10-D3.3.5.pdf (accessed on 23 August 2017).
- Cesaretti, G.; Dini, E.; De Kestelier, X.; Colla, V.; Pambaguian, L. Building components for an outpost on the Lunar soil by means of a novel 3D printing technology. Acta Astron. 2014, 93, 430–450. [Google Scholar] [CrossRef]
- Norman, M.D. The composition and thickness of the crust of Mars estimated from rare earth elements and neodymium-isotopic compositions of Martian meteorites. Meteorit. Planet. Sci. 1999, 34, 439–449. [Google Scholar] [CrossRef]
- An up-to-date List of Martian Meteorites. Available online: http://www.imca.cc/mars/martian-meteorites-list.htm (accessed on 21 August 2017).
- List of Lunar Meteorites. Available online: http://meteorites.wustl.edu/lunar/moon_meteorites_list_alpha.htm (accessed on 21 August 2017).
- Apollo Missions. Available online: http://www.lpi.usra.edu/lunar/missions/apollo/ (accessed on 21 August 2017).
- Taylor, G.J. A Primordial and Complicated Ocean of Magma on Mars. Planetary Science Research Discoveries, 2006. Available online: http://www.psrd.hawaii.edu/Mar06/mars_magmaOcean.html (accessed on 7 August 2017).
- Harper, C.L.; Nyquist, L.E.; Bansal, B.; Wiesmann, H.; Shih, C.-Y. Rapid accretion and early differentiation of Mars as indicated by 142Nd/144Nd in SNC meteorites. Science 1995, 267, 213–216. [Google Scholar] [CrossRef] [PubMed]
- Borg, L.E.; Nyquist, L.E.; Taylor, L.A.; Wiesmann, H.; Shih, C.-Y. Constraints on Martian differentiation processes from Rb-Sr and Sm-Nd isotopic analyses of the basaltic shergottite QUE 94201. Geochim. Cosmochim. Acta 1997, 61, 4915–4931. [Google Scholar] [CrossRef]
- Lee, D-C.; Halliday, A.N. Core formation on Mars and differentiated asteroids. Nature 1997, 388, 854–857. [Google Scholar] [CrossRef]
- Halliday, A.N.; Wänke, H.; Birck, J.-L.; Clayton, R.N. The Accretion, Composition and Early Differentiation of Mars. Space Sci. Rev. 2001, 96, 197–230. [Google Scholar] [CrossRef]
- Elkins-Tanton, L.T.; Parmentier, E.M.; Hess, P.C. Magma ocean fractional crystallization and cumulate overturn in terrestrial planets: Implications for Mars. Meteorit. Planet. Sci. 2003, 38, 1753–1771. [Google Scholar] [CrossRef]
- Elkins-Tanton, L.T.; Hess, P.C.; Parmentier, E.M. Possible formation of ancient crust on Mars through magma ocean processes. J. Geophys. Res. 2005, 110, E12S01. [Google Scholar] [CrossRef]
- Foley, C.N.; Wadhwa, M.; Borg, L.E.; Janney, P.E.; Hines, R.; Grove, T.L. The early differentiation history of Mars from 182W-142Nd isotope systematics in the SNC meteorites. Geochim. Cosmochim. Acta 2005, 69, 4557–4571. [Google Scholar] [CrossRef]
- Solomon, S.C.; Aharonson, O.; Aurnou, J.M.; Banerdt, B.W.; Carr, M.H.; Dombard, A.J.; Frey, H.V.; Golombek, M.P.; Hauck, S.A., II; Head, J.W., III; et al. New Perspectives on American Mars. Science 2005, 307, 1214–1220. [Google Scholar] [CrossRef] [PubMed]
- Borg, L.E.; Draper, D.S. A petrogenetic model for the origin and compositional variation of the Martian basaltic meteorites. Meteorit. Planet. Sci. 2003, 38, 1713–1731. [Google Scholar] [CrossRef]
- Boynton, W.V.; Taylor, G.J.; Evans, L.G.; Reedy, R.C.; Starr, R.; Janes, D.M.; Kerry, K.E.; Drake, D.M.; Kim, K.J.; Williams, R.M.S.; et al. Concentration of H, Si, Cl, K, Fe, and Th in the low- and mid-latitude regions of Mars. J. Geophys. Res. Planets 2007, 112, E12. [Google Scholar] [CrossRef]
- Gasnault, O.; Taylor, G.J.; Karunatillake, S.; Dohm, J.; Newsom, H.; Forni, O.; Pinet, P.; Boynton, W.V. Quantitative geochemical mapping of Martian elemental provinces. Icarus 2010, 207, 226–247. [Google Scholar] [CrossRef]
- Meslin, P.-Y.; Gasnault, O.; Forni, O.; Schröder, S.; Cousin, A.; Berger, G.; Clegg, S.M.; Lasue, J.; Maurice, S.; Sautter, V.; et al. Soil Diversity and Hydration as Observed by ChemCam at Gale Crater, Mars. Science 2013, 341, 1238670. [Google Scholar] [CrossRef] [PubMed]
- McSween, H.Y., Jr.; Taylor, G.J.; Wyatt, M.B. Elemental composition of the Martian Crust. Science 2009, 324, 736–739. [Google Scholar] [CrossRef] [PubMed]
- Agee, C.B.; Wilson, N.V.; McCubbin, F.M.; Ziegler, K.; Polyak, V.J.; Sharp, Z.D.; Asmerom, Y.; Nunn, M.H.; Shaheen, R.; Thiemens, M.H.; et al. Unique Meteorite from Early Amazonian Mars: Water-Rich Basaltic Breccia Northwest Africa 7034. Science 2013, 339, 780–785. [Google Scholar] [CrossRef] [PubMed]
- Humayun, M.; Nemchin, A.; Zanda, B.; Hewins, R.H.; Grange, M.; Kennedy, A.; Lorand, J.P.; Goepel, C.; Fieni, C.; Pont, S.; et al. Origin and age of the earliest Martian crust from meteorite NWA 7533. Nature 2013, 503, 513–517. [Google Scholar] [CrossRef] [PubMed]
- Wittmann, A.; Korotev, R.L.; Jolliff, B.L.; Irving, A.J.; Moser, D.E.; Barker, I.; Rumble, D. Petrography and composition of Martian regolith breccia meteorite Northwest Africa 7475. Meteorit. Planet. Sci. 2015, 50, 326–352. [Google Scholar] [CrossRef]
- Liu, Y.; Ma, C.; Beckett, J.R.; Chen, Y.; Guan, Y. Rare-earth element minerals in Martian breccia meteorites NWA 7034 and 7533: Implications for fluid-rock interaction in the Martian crust. Earth Planet. Sci. Lett. 2016, 451, 251–262. [Google Scholar] [CrossRef]
- The Martian Meteorite Compendium. Available online: https://curator.jsc.nasa.gov/antmet/mmc/ (accessed on 21 August).
- Shearer, C.K.; Burger, P.V.; Papike, J.J.; McCubbin, F.M.; Bell, A.S. Crystal chemistry of merrillite from Martian meteorites: Mineralogical recorders of magmatic processes and planetary differentiation. Meteorit. Planet. Sci. 2015, 50, 649–673. [Google Scholar] [CrossRef]
- Symes, S.J.; Borg, L.E.; Shearer, C.K.; Irving, A.J. The age of the Martian meteorite Northwest Africa 1195 and the differentiation history of the shergottites. Geochim. Cosmochim. Acta 2008, 72, 1696–1710. [Google Scholar] [CrossRef]
- Treiman, A.H. The parent magma of Nakhla (SNC) meteorite, inferred from magmatic inclusions. Geochim. Cosmochim. Acta 1993, 57, 4753–4767. [Google Scholar] [CrossRef]
- Treiman, A.H. The nakhlite meteorites: Augite-rich igneous rocks from Mars. Chem. Erde-Geochem. 2005, 65, 203–270. [Google Scholar] [CrossRef]
- Jones, J.H. Isotopic relationships among the shergottites, the nakhlites and Chassigny. In Proceedings of the 4th Lunar and Planetary Science Conference, Houston, TX, USA, 14–18 March 1988; pp. 465–474. [Google Scholar]
- Nyquist, L.E.; Shih, C.-Y.; McCubbin, F.M.; Santos, A.R.; Shearer, C.K.; Peng, Z.X.; Burger, P.V.; Agee, C.B. Rb-Sr and Sm-Nd isotopic and REE studies of igneous components in the bulk matrix domain of Martian breccia Northwest Africa 7034. Meteorit. Planet. Sci. 2016, 51, 483–498. [Google Scholar] [CrossRef]
- Shirai, N.; Ebihara, M. The Magmatism of Mars Inferred From Chemical Composition of Shergottites. In Proceedings of the 37th Lunar and Planetary Science Conference, Woodlands, TX, USA, 13–17 March 2006; p. 1917. [Google Scholar]
- Liu, Y.; Ma, C. Monazite in Martian breccia meteorite NWA 7034. In Proceedings of the 8th International Mars Conference, Pasadena, CA, USA, 14–18 July 2014; p. 1250. [Google Scholar]
- Liu, Y.; Ma, C. Monazite, chevkinite-perrierite and xenotime in Martian breccia meteorite NWA 7034. In Proceedings of the 46th Lunar Planetary Science Conference, Woodlands, TX, USA, 16–20 March 2015; p. 1287. [Google Scholar]
- Dauphaus, N.; Pourmand, A. Thulium anomalies and rare earth element patterns in meteorites and Earth: Nebular fractionation and the nugget effect. Geochim. Cosmochim. Acta 2015, 163, 234–261. [Google Scholar] [CrossRef]
- Floss, C.; Crozaz, G. Heterogeneous REE patterns in oldhamite from aubrites: Their nature and origin. Geochim. Cosmochim. Acta 1993, 57, 4039–4057. [Google Scholar] [CrossRef]
- Terada, K.; Sano, Y. Ion microprobe U-Pb dating and REE analyses of phosphates in H4-chondrite, Yamato-74371. Geophys. Res. Lett. 2002, 29, 1460. [Google Scholar] [CrossRef]
- Reed, S.J.B.; Smith, D.G.W.; Long, J.V.P. Rare earth elements in chondritic phosphates—Implications for 244Pu chronology. Nature 1983, 306, 172–173. [Google Scholar] [CrossRef]
- Martínez-Jiménez, M.; Moyano-Cambero, C.E.; Trigo-Rodríguezm, J.M.; Alonso-Azcárate, J.; Jordi, L. Asteroid Mining: Mineral Resources in Undifferentiated Bodies from the Chemical Composition of Carbonaceous Chondrites; Assessment and Mitigation of Asteroid Impact hazards, Astrophysics and Space Science Proceedings; Springer: New York, NY, USA, 2017; pp. 73–101. [Google Scholar]
- USGS. Platinum-Group Elements-So Many Excellent Properties. 2014. Available online: https://pubs.usgs.gov/fs/2014/3064/pdf/fs2014-3064.pdf (accessed on 2 August 2017).
- NASA Approves 2018 Launch of Mars InSight Mission. Available online: https://insight.jpl.nasa.gov/home.cfm (accessed on 21 August 2017).
- Mars 2020 Mission Overview. Available online: https://mars.nasa.gov/mars2020/mission/overview (accessed on 21 August 2017).
- Cofield, C. NASA’s Mars Plan May Include Yearlong Mission to the Moon. 2017. Available online: https://www.space.com/36781-nasa-yearlong-crew-moon-mission-ahead-of-mars.html (accessed on 6 August 2017).
- Progress in Defining the Deep Space Gateway and Transport Plan. Available online: https://www.nasa.gov/sites/default/files/atoms/files/nss_chart_v23.pdf (accessed on 21 August 2017).
- Zubrin, R. The Case for Colonizing Mars. Ad Astra, 1996. Available online: http://www.ifa.hawaii.edu/~meech/a281/handouts/mars_case.pdf (accessed on 6 August 2017).
- Wall, M. Private Mars Missions: A Red Planet Exploration Roundup. 2016. Available online: https://www.space.com/32721-private-mars-missions-spacex-red-dragon.html (accessed on 6 August 2017).
- National Aeronautics and Space Administration. Available online: https://solarsystem.nasa.gov/missions/near/indepth (accessed on 21 August 2017).
- Asteroid Explorer “HAYABUSA” (MUSES-C). Available online: http://global.jaxa.jp/projects/sat/muses_c/ (accessed on 21 August 2017).
- Asteroid Explorer “Hayabusa2”. Available online: http://global.jaxa.jp/projects/sat/hayabusa2/ (accessed on 21 August 2017).
- OSIRIS-Rex. Available online: https://www.nasa.gov/osiris-rex (accessed on 21 August 2017).
- Crawford, I.A. The long-term scientific benefits of a space economy. Space Policy 2016, 37, 58–61. [Google Scholar] [CrossRef]




© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
McLeod, C.L.; Krekeler, M.P.S. Sources of Extraterrestrial Rare Earth Elements: To the Moon and Beyond. Resources 2017, 6, 40. https://doi.org/10.3390/resources6030040
McLeod CL, Krekeler MPS. Sources of Extraterrestrial Rare Earth Elements: To the Moon and Beyond. Resources. 2017; 6(3):40. https://doi.org/10.3390/resources6030040
Chicago/Turabian StyleMcLeod, Claire L., and Mark. P. S. Krekeler. 2017. "Sources of Extraterrestrial Rare Earth Elements: To the Moon and Beyond" Resources 6, no. 3: 40. https://doi.org/10.3390/resources6030040
APA StyleMcLeod, C. L., & Krekeler, M. P. S. (2017). Sources of Extraterrestrial Rare Earth Elements: To the Moon and Beyond. Resources, 6(3), 40. https://doi.org/10.3390/resources6030040




