An Evolutionary Framework for Understanding the Origin of Eukaryotes
Abstract
:“When all are one, and one is all,” Stairway to Heaven, Led Zeppelin
1. Introduction
2. Conflict and Cooperation in the Darwinian Evolution of Endosymbiosis
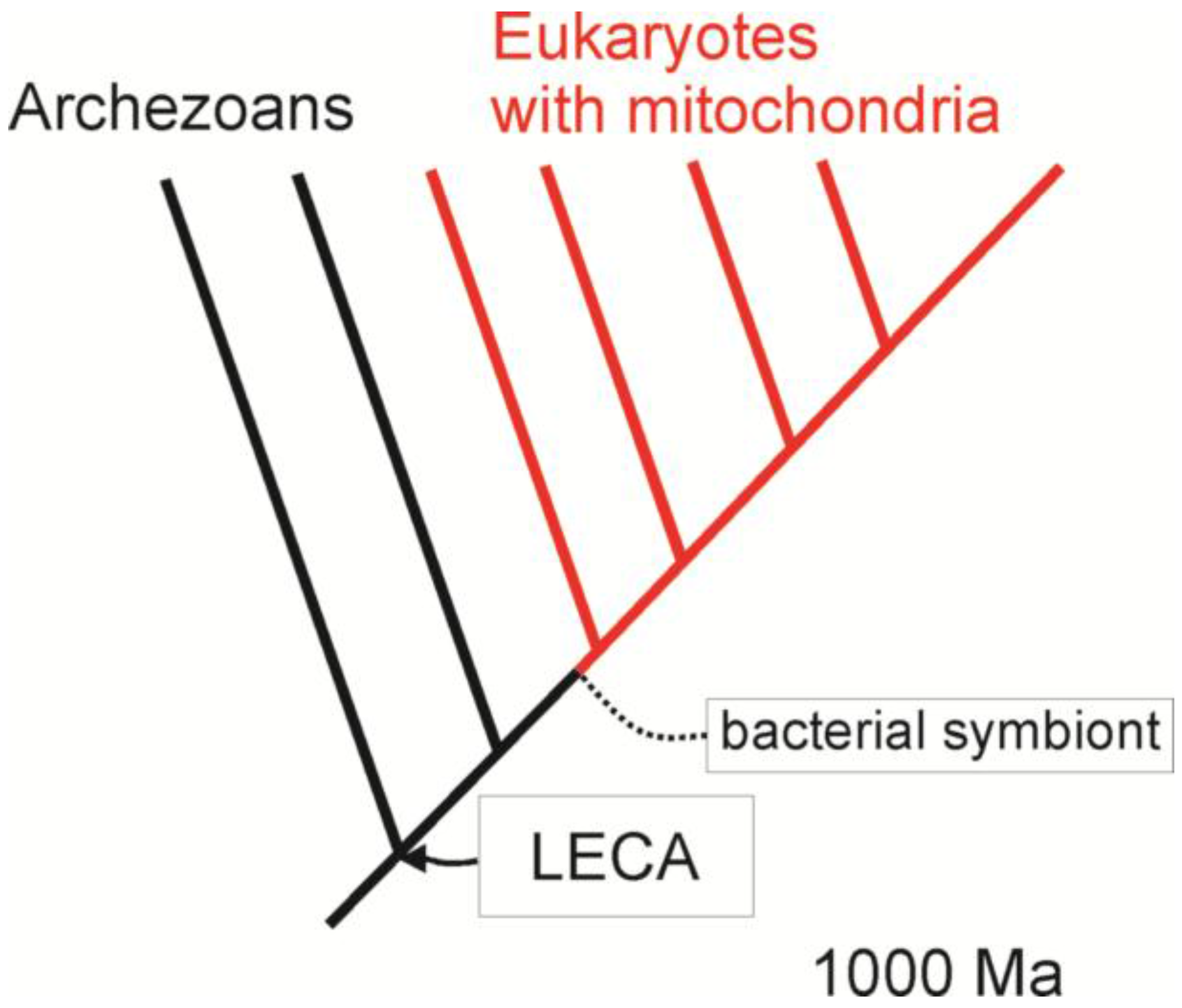
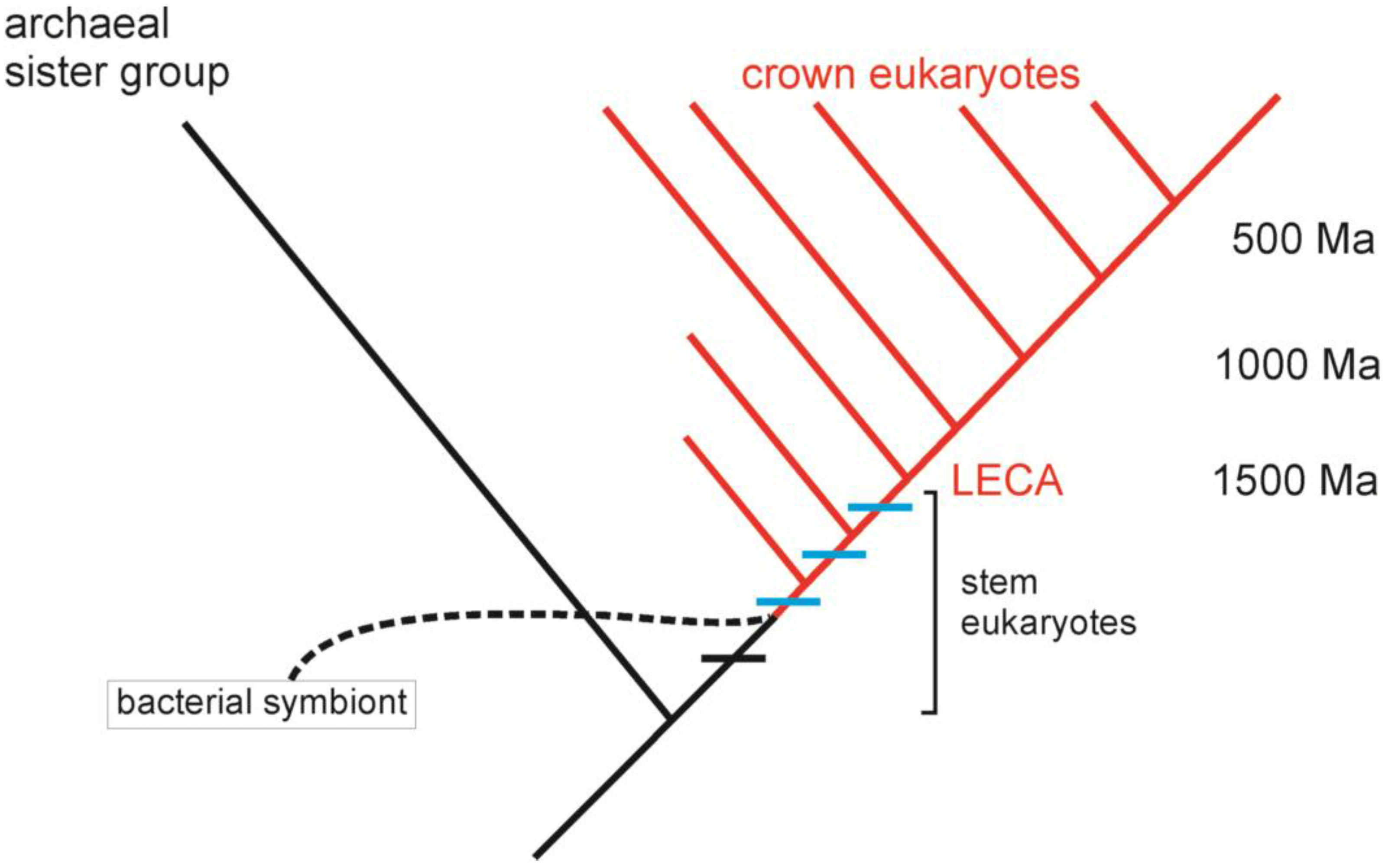
3. Comparative Methods and the Origin of Eukaryotes
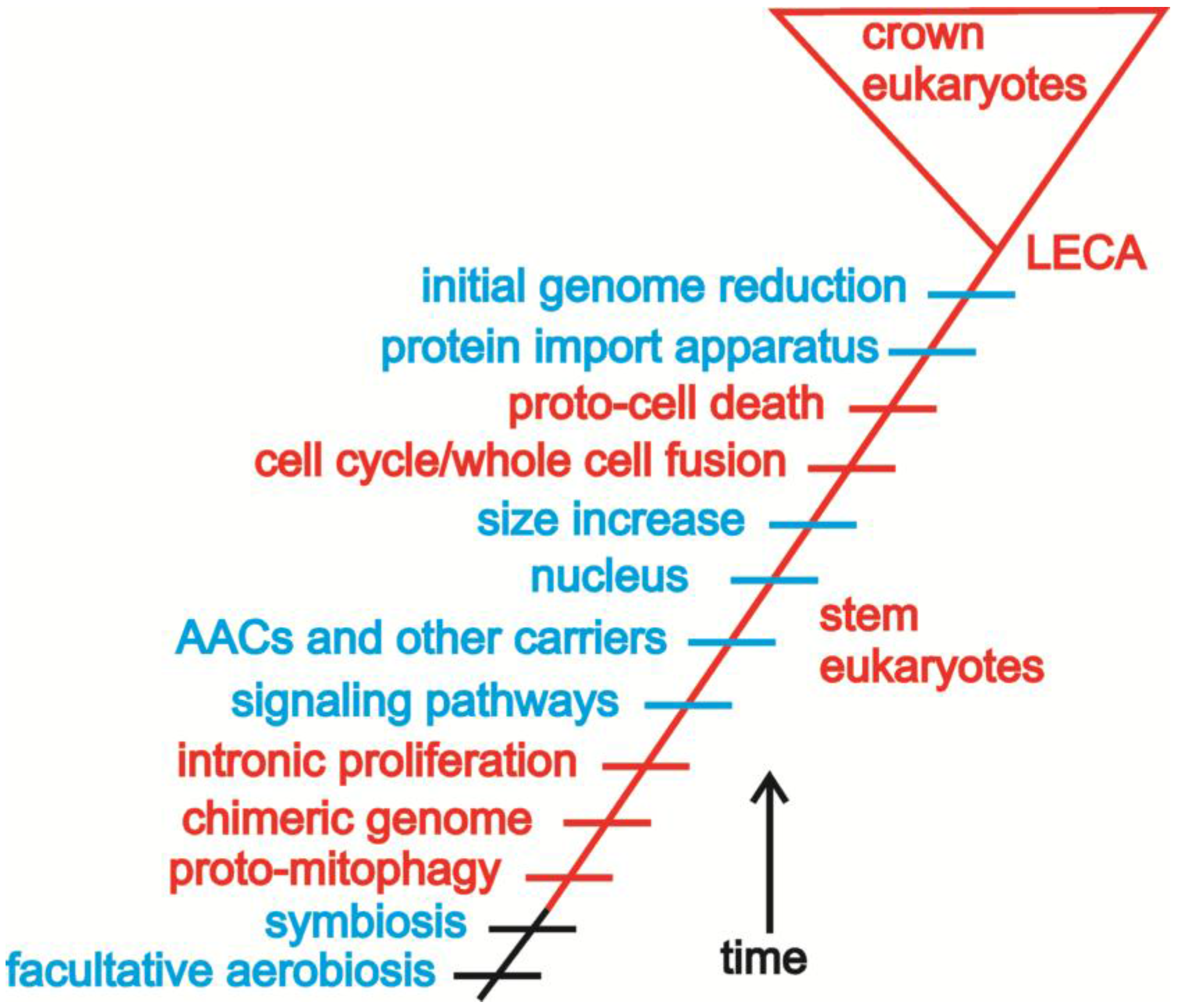
4. Building a Framework to Explore the Origin of Eukaryotes
4.1. Introns and Endosymbiosis: From Small Things, Big Things One Day Come
4.2. Conflict, Cooperation, and the Evolution of Eukaryotic Bioenergetics
4.3. Oxygen, Metabolism, Membranes, and Sex
5. Discussion
6. Conclusions
Acknowledgments
Conflicts of Interest
References
- Darwin, C. On the Origin of Species, 1st ed.; Harvard University Press: Cambridge, MA, USA, 1964; pp. 1–513. [Google Scholar]
- Darwin, C. Descent of Man; Penguin Books: London, UK, 2004; pp. 21–22, 157–158. [Google Scholar]
- Herron, J.C.; Freeman, S. Evolutionary Analysis, 5th ed.; Pearson: Boston, MA, USA, 2014; pp. 1–850. [Google Scholar]
- Harvey, P.H.; Pagel, M.D. The Comparative Method in Evolutionary Biology; Oxford University Press: Oxford, UK, 1991; pp. 1–239. [Google Scholar]
- Wallin, I.E. Symbionticism and the Origin of Species; Williams & Wilkins: Baltimore, MD, USA, 1927; pp. 1–171. [Google Scholar]
- Margulis, L.; Sagan, D. Microcosmos; Summit: New York, NY, USA, 1986; pp. 14–15. [Google Scholar]
- Frank, S.A. Repression of competition and the evolution of cooperation. Evolution 2003, 57, 693–705. [Google Scholar] [CrossRef] [PubMed]
- Okasha, S. Evolution and the Levels of Selection; Oxford University Press: Oxford, UK, 2006; pp. 1–263. [Google Scholar]
- Wynne-Edwards, V.C. Animal Dispersion in Relation to Social Behavior; Hafner: New York, NY, USA, 1962; pp. 1–653. [Google Scholar]
- Maynard Smith, J. Group selection and kin selection. Nature 1964, 201, 1145–1147. [Google Scholar] [CrossRef]
- Williams, G.C. Adaptation and Natural Selection; Princeton University Press: Princeton, NJ, USA, 1966; pp. 1–307. [Google Scholar]
- Hamilton, W.D. The genetical evolution of social behavior. I. J. Theor. Biol. 1964, 7, 1–16. [Google Scholar] [CrossRef]
- Wilson, D.S. A theory of group selection. Proc. Natl. Acad. Sci. USA 1975, 72, 143–146. [Google Scholar] [CrossRef] [PubMed]
- Wilson, D.S.; Wilson, E.O. Rethinking the theoretical foundation of sociobiology. Q. Rev. Biol. 2007, 82, 327–348. [Google Scholar] [CrossRef] [PubMed]
- Keller, L. Levels of Selection in Evolution; Princeton University Press: Princeton, NJ, USA, 1999; pp. 1–318. [Google Scholar]
- Buss, L. The Evolution of Individuality; Princeton University Press: Princeton, NJ, USA, 1987; pp. 1–201. [Google Scholar]
- Maynard Smith, J.; Szathmáry, E. The Major Transitions in Evolution; Oxford University Press: Oxford, UK, 1995; pp. 1–346. [Google Scholar]
- Michod, R.E. Darwinian Dynamics: Evolutionary Transitions in Fitness and Individuality; Princeton University Press: Princeton, NJ, USA, 1999; pp. 1–262. [Google Scholar]
- Bonner, J.T. The origins of multicellularity. Integr. Biol. 1998, 1, 27–36. [Google Scholar] [CrossRef]
- Michod, R.E.; Nedelcu, A.M. On the reorganization of fitness during evolutionary transitions in individuality. Integr. Compar. Biol. 2003, 43, 64–73. [Google Scholar] [CrossRef] [PubMed]
- Cosmides, L.M.; Tooby, J. Cytoplasmic inheritance and intragenomic conflict. J. Theor. Biol. 1981, 89, 83–129. [Google Scholar] [CrossRef]
- Blackstone, N.W. A units-of-evolution perspective on the endosymbiont theory of the orgin of the mitochondrion. Evolution 1995, 49, 785–796. [Google Scholar] [CrossRef]
- Frade, J.M.; Michaelidis, T.M. Origin of eukaryotic programmed cell death—A consequence of aerobic metabolism. BioEssays 1997, 19, 827–832. [Google Scholar] [CrossRef] [PubMed]
- Kroemer, G. Mitochondrial implication in apoptosis: Towards an endosymbiont hypothesis of apoptosis evolution. Cell Death Differ. 1997, 4, 443–456. [Google Scholar] [CrossRef] [PubMed]
- Mignotte, B.; Vayssiere, J.-L. Mitochondria and apoptosis. Eur. J. Biochem. 1998, 252, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Blackstone, N.W.; Green, D.R. The evolution of a mechanism of cell suicide. BioEssays 1999, 21, 84–88. [Google Scholar] [CrossRef]
- Blackstone, N.W. Why did eukaryotes evolve only once? Genetic and energetic aspects of conflict and conflict mediation. Philos. Trans. R. Soc. Lond. B 2013, 368, 20120266. [Google Scholar] [CrossRef] [PubMed]
- Blackstone, N.W. Evolution and cell physiology. 2. The evolution of cell signaling from mitochondria to Metazoa. Am. J. Physiol. Cell Physiol. 2013, 305, C909–C915. [Google Scholar] [CrossRef] [PubMed]
- Blackstone, N.W. sAC as a model for understanding the impact of endosymbiosis on cell signaling. Biochim. Biophys. Acta 2014, 1842, 2548–2554. [Google Scholar] [CrossRef] [PubMed]
- Blackstone, N.W. The impact of mitochondrial endosymbiosis on the evolution of calcium signaling. Cell Calcium 2015, 57, 133–139. [Google Scholar] [CrossRef] [PubMed]
- Radzvilavicius, A.L.; Blackstone, N.W. Conflict and cooperation in eukaryogenesis: Implications for the timing of endosymbiosis and the evolution of sex. J. R. Soc. Lond. Interface 2015, 12. [Google Scholar] [CrossRef] [PubMed]
- Felsenstein, J. Phylogenies and the comparative method. Am. Nat. 1985, 125, 1–15. [Google Scholar] [CrossRef]
- Harvey, P.H.; Leigh Brown, A.J.; Maynard Smith, J.; Nee, S. New Uses for New Phylogenies; Oxford University Press: Oxford, UK, 1996; pp. 1–349. [Google Scholar]
- Cavalier-Smith, T. Archaebacteria and archezoa. Nature 1989, 339, 100–101. [Google Scholar] [CrossRef] [PubMed]
- Lane, N. The Vital Question; Norton: New York, NY, USA, 2014; pp. 1–360. [Google Scholar]
- Koonin, E.V. The origin and early evolution of eukaryotes in the light of phylogenomics. Genome Biol. 2010, 11. [Google Scholar] [CrossRef] [PubMed]
- Guy, L.; Saw, J.H.; Ettema, T.J.G. The archaeal legacy of eukaryotes: A phylogenomic perspective. Cold Spring Harbor Perspect. Biol. 2014, 6. [Google Scholar] [CrossRef] [PubMed]
- Pittis, A.A.; Gabaldón, T. Late acquisition of mitochondria by a host with chimaeric prokaryotic ancestry. Nature 2016, 531, 101–104. [Google Scholar] [CrossRef] [PubMed]
- Poole, A.M.; Gribaldo, S. Eukaryotic origins: How and when was the mitochondrion acquired? Cold Spring Harbor Perspect. Biol. 2014, 6. [Google Scholar] [CrossRef] [PubMed]
- Martin, W.F.; Koonin, E.V. Introns and the origin of the nucleus-cytosol compartmentation. Nature 2006, 440, 41–45. [Google Scholar] [CrossRef] [PubMed]
- Henze, K.; Martin, W.F. How do mitochondrial genes get into the nucleus? Trends Genet. 2001, 17, 383–387. [Google Scholar] [CrossRef]
- Martin, W.F.; Garg, S.; Zimorski, V. Endosymbiotic theories for eukaryotic origin. Philos. Trans. R. Soc. Lond. B 2015, 370. [Google Scholar] [CrossRef] [PubMed]
- Garg, S.; Martin, W.F.; Heinrich-Heine University, Düsseldorf, Germany. Unpublished work. 2016.
- Lane, N.; Martin, W.F. The energetic of genome complexity. Nature 2010, 467, 929–934. [Google Scholar] [CrossRef] [PubMed]
- Kunji, E.R.S.; Crichton, P.G. Mitochondrial carriers function as monomers. Biochim. Biophys. Acta 2010, 1797, 817–831. [Google Scholar] [CrossRef] [PubMed]
- Butterfield, N.J. Oxygen, animals and oceanic ventilation: An alternative view. Geobiology 2009, 7, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Parfrey, L.W.; Lahr, D.J.G.; Knoll, A.H.; Katz, L.A. Estimating the timing of early eukaryotic diversification with multigene molecular clocks. Proc. Natl. Acad. Sci. USA 2011, 108, 13624–13629. [Google Scholar] [CrossRef] [PubMed]
- Ramussen, B.; Fletcher, I.R.; Brocks, J.J.; Kilburn, M.R. Reassessing the first appearance of eukaryotes and cyanobacteria. Nature 2008, 455, 1101–1104. [Google Scholar] [CrossRef] [PubMed]
- Müller, M.; Mentel, M.; van Hellemond, J.J.; Henze, K.; Woehle, C.; Gould, S.B.; Yu, R.-Y.; van der Giezen, M.; Tielens, A.G.M.; Martin, W.F. Biochemistry and evolution of anaerobic energy metabolism in eukaryotes. Microbiol. Mol. Biol. Rev. 2012, 76, 444–495. [Google Scholar] [CrossRef] [PubMed]
- Waldbauer, J.R.; Newman, D.K.; Summons, R.E. Microaerobic steroid biosynthesis and the molecular fossil record of Archean life. Proc. Natl. Acad. Sci. USA 2011, 108, 13409–13414. [Google Scholar] [CrossRef] [PubMed]
- Preisig, O.; Zufferey, R.; Thony-Meyer, L.; Appleby, C.A.; Hennecke, H. A high-affinity cbb3-type cytochrome oxidase terminates the symbiosis-specific respiratory chain of Bradyrhizobium japonicum. J. Bacteriol. 1996, 178, 1532–1538. [Google Scholar] [PubMed]
- Cohan, F.M.; Aracena, S. Prokaryotic sex: Eukaryote-like qualities of recombination in an archaean lineage. Curr. Biol. 2012, 22, R601–R602. [Google Scholar] [CrossRef] [PubMed]
- Naor, A.; Lapierre, P.; Mevarech, M.; Papke, R.T.; Gophna, U. Low species barriers in halophilic Archaea and the formation of recombinant hybrids. Curr. Biol. 2012, 22, 1444–1448. [Google Scholar] [CrossRef] [PubMed]
- Errington, J. L-form bacteria, cell walls and the origins of life. Open Biol. 2013, 3. [Google Scholar] [CrossRef] [PubMed]
- Řezanka, T.; Siristova, L.; Melzoch, K.; Sigler, K. Hopanoids in bacteria and cyanobacteria—Their role in cellular biochemistry and physiology, analysis and occurrence. Mini-Rev. Org. Chem. 2010, 7, 300–313. [Google Scholar] [CrossRef]
- Adl, S.M.; Simpson, A.G.B.; Lane, C.E.; Lukes, J.; Bass, D.; Bowser, S.S.; Brown, M.W.; Burki, F.; Dunthorn, M.; Hampl, V.; et al. The revised classification of the eukaryotes. J. Eukaryot. Microbiol. 2012, 59, 429–493. [Google Scholar] [CrossRef] [PubMed]
- Butterfield, N.J. Early evolution of the Eukaryota. Palaeontology 2015, 58, 5–17. [Google Scholar] [CrossRef]
- Ratcliff, W.C.; Denison, R.F.; Borrello, M.; Travisano, M. Experimental evolution of multicellularity. Proc. Natl. Acad. Sci. USA 2012, 109, 1595–1600. [Google Scholar] [CrossRef] [PubMed]
- Spang, A.; Saw, J.H.; Jørgensen, S.L.; Zaremba-Niedzwiedzka, K.; Martijn, J.; Lind, A.E.; van Eijk, R.; Schleper, C.; Guy, L.; Ettema, T.J.G. Complex archaea that bridge the gap between prokaryotes and eukaryotes. Nature 2015, 521, 173–179. [Google Scholar] [CrossRef] [PubMed]
- Lynch, M.; Marinov, G.K. The bioenergetic cost of a gene. Proc. Natl. Acad. Sci. USA 2015, 112, 15690–15695. [Google Scholar] [CrossRef] [PubMed]
- Lane, N. Power, Sex, Suicide: Mitochondria and the Meaning of Life; Oxford University Press: Oxford, UK, 2005; pp. 1–354. [Google Scholar]
- Blackstone, N.W.; Kirkwood, T.B.L. Mitochondria and programmed cell death: “Slave revolt” or community homeostasis? In Genetic and Cultural Evolution of Cooperation; Hammerstein, P., Ed.; MIT Press: Cambridge, MA, USA, 2003; pp. 309–325. [Google Scholar]
- Lachmann, M.; Blackstone, N.W.; Haig, D.; Kowald, A.; Michod, R.E.; Szathmáry, E.; Werren, J.H.; Wolpert, L. Group 3: Cooperation and conflict in the evolution of genomes, cells, and multicellular organisms. In Genetic and Cultural Evolution of Cooperation; Hammerstein, P., Ed.; MIT Press: Cambridge, MA, USA, 2003; pp. 327–356. [Google Scholar]
- Allen, J.F. Control of gene expression by redox potential and the requirement for chloroplast and mitochondrial genomes. J. Theor. Biol. 1993, 165, 609–631. [Google Scholar] [CrossRef] [PubMed]
- Allen, J.F. The function of genomes in bioenergetic organelles. Philos. Trans. R. Soc. Lond. B 2003, 358, 19–38. [Google Scholar] [CrossRef] [PubMed]

© 2016 by the author; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Blackstone, N.W. An Evolutionary Framework for Understanding the Origin of Eukaryotes. Biology 2016, 5, 18. https://doi.org/10.3390/biology5020018
Blackstone NW. An Evolutionary Framework for Understanding the Origin of Eukaryotes. Biology. 2016; 5(2):18. https://doi.org/10.3390/biology5020018
Chicago/Turabian StyleBlackstone, Neil W. 2016. "An Evolutionary Framework for Understanding the Origin of Eukaryotes" Biology 5, no. 2: 18. https://doi.org/10.3390/biology5020018
APA StyleBlackstone, N. W. (2016). An Evolutionary Framework for Understanding the Origin of Eukaryotes. Biology, 5(2), 18. https://doi.org/10.3390/biology5020018




