Role of Macrofaunal Communities in the Vistula River Plume, the Baltic Sea—Bioturbation and Bioirrigation Potential
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Sampling
2.2. Macrofauna
2.3. Bioturbation Potential (BPC) and Irrigation Potential (IPC)
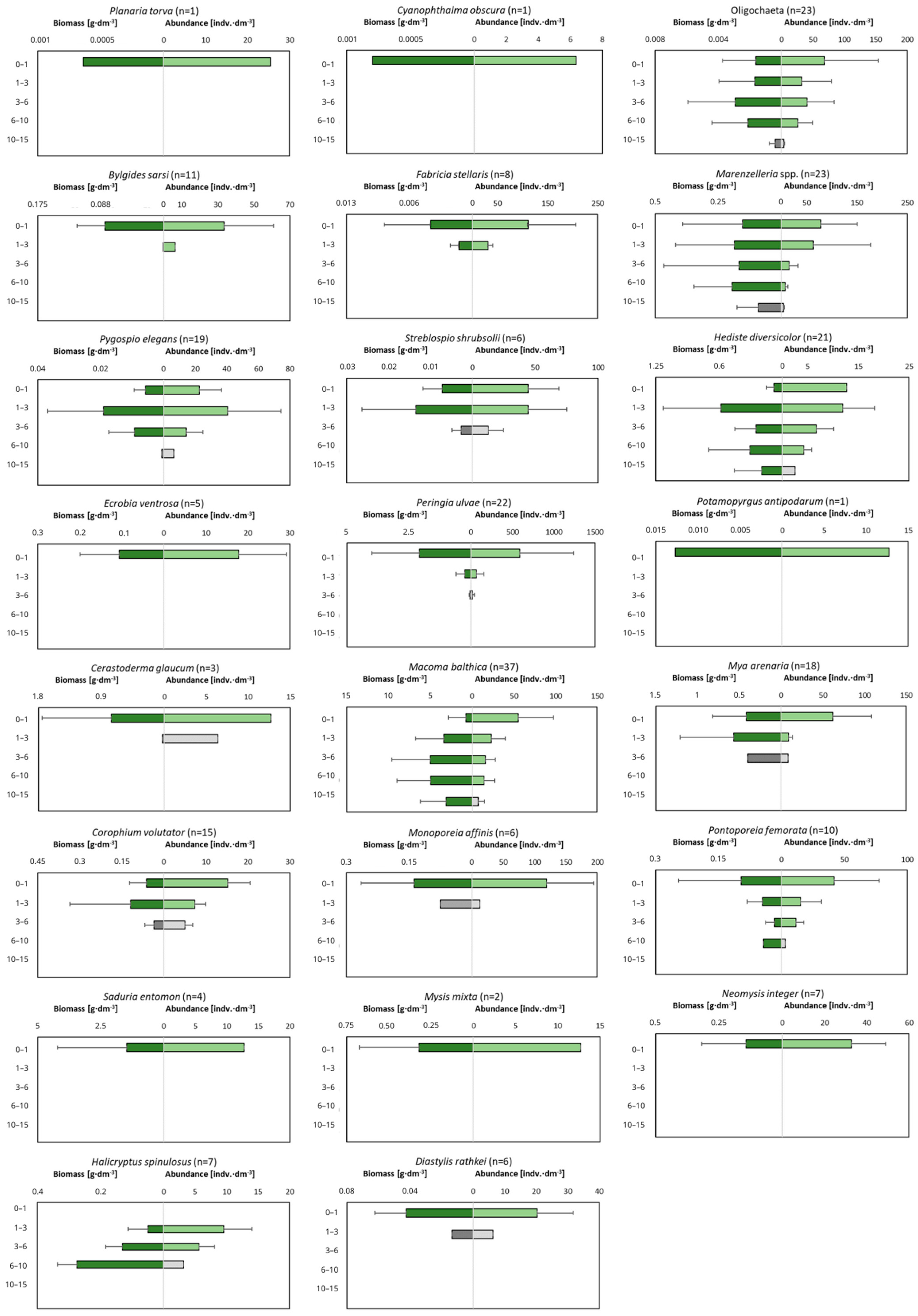
2.4. Vertical Distrbution of Macrofauna in Sediment
2.5. Data Analysis
3. Results
3.1. Environmental Conditions
3.2. Macrofauna
4. Discussion
4.1. Conditions of Bottom Water and Sediments and Their Impact on Macrozoobenthos
4.2. Macrozoobenthos
4.3. Bioturbation and Bioirrigation
4.4. Burrowing Depth
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
| Taxa | BPc | IPc | |||
|---|---|---|---|---|---|
| Mi | Ri | BTi | FTi | IDi | |
| Planaria torva | 1 | 1 | 1 | 2 | 1 |
| Cyanophthalma obscura | 3 | 1 | 3 | 2 | 2 |
| Oligochaeta | 3 | 2 | 3 | 3 | 4 |
| Bylgides sarsi | 3 | 1 | 1 | 2 | 2 |
| Fabricia stellaris | 2 | 1 | 3 | 1 | 2 |
| Marenzelleria spp. | 4 | 4 | 3 | 3 | 5 |
| Pygospio elegans | 2 | 2 | 3 | 3 | 3 |
| Streblospio shrubsolii | 2 | 2 | 3 | 3 | 2 |
| Hediste diversicolor | 4 | 3 | 2 | 3 | 5 |
| Ecrobia ventrosa | 1 | 1 | 1 | 3 | 1 |
| Peringia ulvae | 1 | 1 | 1 | 3 | 2 |
| Potamopyrgus antipodarum | 1 | 1 | 1 | 3 | 1 |
| Cerastoderma glaucum | 3 | 2 | 1 | 1 | 1 |
| Macoma balthica | 3 | 4 | 1 | 3 | 4 |
| Mya arenaria | 3 | 4 | 1 | 1 | 2 |
| Corophium volutator | 2 | 2 | 2 | 3 | 3 |
| Monoporeia affinis | 4 | 2 | 3 | 3 | 2 |
| Pontoporeia femorata | 4 | 4 | 3 | 3 | 4 |
| Diastylis rathkei | 3 | 2 | 3 | 3 | 2 |
| Saduria entomon | 4 | 2 | 3 | 3 | 1 |
| Mysis mixta | 4 | 1 | 3 | 3 | 1 |
| Neomysis integer | 4 | 1 | 3 | 3 | 1 |
| Halicryptus spinulosus | 3 | 4 | 3 | 3 | 4 |
| Site | No. of Taxa | Marenzelleria spp. | Hediste diversicolor | Peringia ulvae | Macoma balthica | Saduria entomon | Others * |
|---|---|---|---|---|---|---|---|
| VE03 | 8 ± 4 | 1.9 ± 2.0 | 21.7 ± 22.1 | 42.5 ± 26.1 | 370.7 ± 227.2 | 0.0 ± 0.0 | 14.6 ± 10.1 |
| VE04 | 5 ± 0 | 0.0 ± 0.0 | 16.2 ± 13.0 | 7.6 ± 8.4 | 90.1 ± 97.4 | 0.0 ± 0.0 | 3.4 ± 2 |
| VE05 | 8 ± 3 | 1.1 ± 2.1 | 35.4 ± 19.3 | 39.3 ± 8.6 | 375.5 ± 117.5 | 0.0 ± 0.0 | 10.8 ± 11.7 |
| VE06 | 5 ± 0 | 18.3 ± 6.8 | 0.0 ± 0.0 | 0.1 ± 0.2 | 81.8 ± 57.9 | 0.0 ± 0.0 | 3.2 ± 2.6 |
| VE07 | 4 ± 1 | 0.0 ± 0.0 | 0.0 ± 0.0 | 0.0 ± 0.0 | 177.2 ± 77.5 | 14.0 ± 27.9 | 3 ± 3.7 |
| VE09 | 5 ± 4 | 10.0 ± 8.7 | 10.6 ± 17 | 1.1 ± 1.9 | 109.1 ± 113.5 | 0.0 ± 0.0 | 6.1 ± 7.3 |
| VE18 | 9 ± 2 | 34.1 ± 30.3 | 4.6 ± 6.5 | 12.0 ± 7.4 | 158.7 ± 59.8 | 0.0 ± 0.0 | 18.2 ± 16.4 |
| VE23 | 6 ± 1 | 1.1 ± 0.9 | 0.0 ± 0.0 | 0.0 ± 0.0 | 118.0 ± 150.6 | 0.1 ± 0.1 | 10.3 ± 7.7 |
| VE38 | 0 ± 0 | 0.0 ± 0.0 | 0.0 ± 0.0 | 0.0 ± 0.0 | 0.0 ± 0.0 | 0.0 ± 0.0 | 1.8 ± 3.1 |
| VE39 | 0 ± 0 | 0.0 ± 0.0 | 0.0 ± 0.0 | 0.0 ± 0.0 | 0.0 ± 0.0 | 0.0 ± 0.0 | 0.2 ± 0.2 |
| VE43 | 0 ± 0 | 0.0 ± 0.0 | 0.0 ± 0.0 | 0.0 ± 0.0 | 0.0 ± 0.0 | 0.0 ± 0.0 | 0.4 ± 0.7 |
| VE46 | 5 ± 0 | 0.1 ± 0.1 | 0.0 ± 0.0 | 0.0 ± 0.0 | 458.1 ± 82.1 | 0.7 ± 1.4 | 5.8 ± 3.3 |
| VE49 | 7 ± 0 | 0.0 ± 0.0 | 15.9 ± 6.1 | 3.8 ± 0.8 | 374.7 ± 183.2 | 0.0 ± 0.0 | 10.2 ± 7.1 |
| TF0233 | 0 ± 0 | 0.0 ± 0.0 | 0.0 ± 0.0 | 0.0 ± 0.0 | 0.0 ± 0.0 | 0.0 ± 0.0 | 0.0 ± 0.0 |
References
- de Groot, R.S.; Wilson, M.A.; Boumans, R.M.J. A typology for the classification, description and valuation of ecosystem functions, goods and services. Ecol. Econ. 2002, 41, 393–408. [Google Scholar] [CrossRef]
- McGlathery, K.; Sundbäck, K.; Anderson, I. Eutrophication in shallow coastal bays and lagoons: The role of plants in the coastal filter. Mar. Ecol. Prog. Ser. 2007, 348, 1–18. [Google Scholar] [CrossRef]
- Carstensen, J.; Conley, D.J.; Almroth-Rosell, E.; Asmala, E.; Bonsdorff, E.; Fleming-Lehtinen, V.; Gustafsson, B.G.; Gustafsson, C.; Heiskanen, A.-S.; Janas, U.; et al. Factors regulating the coastal nutrient filter in the Baltic Sea. AMBIO 2019, 49, 1194–1210. [Google Scholar] [CrossRef] [PubMed]
- Biles, C.L.; Paterson, D.M.; Ford, R.B.; Solan, M.; Raffaelli, D.G. Bioturbation, ecosystem functioning and community structure. Hydrol. Earth Syst. Sci. 2002, 6, 999–1005. [Google Scholar] [CrossRef]
- Braeckman, U.; Provoost, P.; Gribsholt, B.; Van Gansbeke, D.; Middelburg, J.; Soetaert, K.; Vincx, M.; Vanaverbeke, J. Role of macrofauna functional traits and density in biogeochemical fluxes and bioturbation. Mar. Ecol. Prog. Ser. 2010, 399, 173–186. [Google Scholar] [CrossRef]
- Kauppi, L.; Bernard, G.; Bastrop, R.; Norkko, A.; Norkko, J. Increasing densities of an invasive polychaete enhance bioturbation with variable effects on solute fluxes. Sci. Rep. 2018, 8, 1–12. [Google Scholar] [CrossRef]
- Benelli, S.; Bartoli, M.; Ribaudo, C.; Fano, E.A. Contrasting Effects of an Alien Worm on Benthic N Cycling in Muddy and Sandy Sediments. Water 2019, 11, 465. [Google Scholar] [CrossRef]
- Bartoli, M.; Benelli, S.; Magri, M.; Ribaudo, C.; Moraes, P.; Castaldelli, G. Contrasting Effects of Bioturbation Studied in Intact and Reconstructed Estuarine Sediments. Water 2020, 12, 3125. [Google Scholar] [CrossRef]
- Borawska, Z.; Szymczycha, B.; Silberberger, M.J.; Koziorowska-Makuch, K.; Szczepanek, M.; Kędra, M. Benthic fluxes of dissolved silica are an important component of the marine Si cycle in the coastal zone. Estuar. Coast. Shelf Sci. 2022, 273, 107880. [Google Scholar] [CrossRef]
- Politi, T.; Zilius, M.; Castaldelli, G.; Bartoli, M.; Daunys, D. Estuarine Macrofauna Affects Benthic Biogeochemistry in a Hypertrophic Lagoon. Water 2019, 11, 1186. [Google Scholar] [CrossRef]
- Politi, T.; Barisevičiūte, R.; Bartoli, M.; Bonaglia, S.; Cardini, U.; Castaldelli, G.; Kančauskaitė, A.; Marzocchi, U.; Petkuviene, J.; Samuiloviene, A.; et al. A bioturbator, a holobiont, and a vector: The multifaceted role of Chironomus plumosus in shaping N-cycling. Freshw. Biol. 2021, 66, 1036–1048. [Google Scholar] [CrossRef]
- Samuiloviene, A.; Bartoli, M.; Bonaglia, S.; Cardini, U.; Vybernaite-Lubiene, I.; Marzocchi, U.; Petkuviene, J.; Politi, T.; Zaiko, A.; Zilius, M. The Effect of Chironomid Larvae on Nitrogen Cycling and Microbial Communities in Soft Sediments. Water 2019, 11, 1931. [Google Scholar] [CrossRef]
- Kristensen, E.; Mikkelsen, O.L. Impact of the burrow-dwelling polychaete Nereis diversicolor on the degradation of fresh and aged macroalgal detritus in a coastal marine sediment. Mar. Ecol. Prog. Ser. 2003, 265, 141–153. [Google Scholar] [CrossRef]
- Gilbert, F.; Rivet, L.; Bertrand, J.-C. The in vitro influence of the burrowing polychaete Nereis diversicolor on the fate of petroleum hydrocarbons in marine sediments. Chemosphere 1994, 29, 1–12. [Google Scholar] [CrossRef]
- Granberg, M.E.; Gunnarsson, J.S.; Hedman, J.E.; Rosenberg, R.; Jonsson, P. Bioturbation-Driven Release of Organic Contaminants from Baltic Sea Sediments Mediated by the Invading Polychaete Marenzelleria neglecta. Environ. Sci. Technol. 2008, 42, 1058–1065. [Google Scholar] [CrossRef] [PubMed]
- Lohrer, A.M.; Thrush, S.F.; Gibbs, M.M. Bioturbators enhance ecosystem function through complex biogeochemical interactions. Nature 2004, 431, 1092–1095. [Google Scholar] [CrossRef] [PubMed]
- Snelgrove, P.V.; Soetaert, K.; Solan, M.; Thrush, S.; Wei, C.-L.; Danovaro, R.; Fulweiler, R.W.; Kitazato, H.; Ingole, B.; Norkko, A.; et al. Global Carbon Cycling on a Heterogeneous Seafloor. Trends Ecol. Evol. 2018, 33, 96–105. [Google Scholar] [CrossRef] [PubMed]
- Elliott, M.; Day, J.; Ramachandran, R.; Wolanski, E.; Fang, Q.; Sheehan, M.; Seen, A.; Ellison, J. A Synthesis: What Is the Future for Coasts, Estuaries, Deltas and Other Transitional Habitats in 2050 and Beyond? In Coasts and Estuaries: The Future; Wolanski, E., Day, J.W., Elliott, M., Ramachandran, R., Eds.; Elsevier: Oxford, UK, 2019; pp. 1–28. ISBN 9780128140031. [Google Scholar]
- Strokal, M.; Bai, Z.; Franssen, W.; Hofstra, N.; Koelmans, A.A.; Ludwig, F.; Ma, L.; van Puijenbroek, P.; Spanier, J.E.; Vermeulen, L.C.; et al. Urbanization: An increasing source of multiple pollutants to rivers in the 21st century. NPJ Urban Sustain. 2021, 1, 1–13. [Google Scholar] [CrossRef]
- Kruk-Dowgiałło, L.; Szaniawska, A. Gulf of Gdańsk and Puck Bay. In Ecology of Baltic Coastal Waters. Ecological Studies; Schiewer, U., Ed.; Springer: Berlin/Heidelberg, Germany, 2008; Volume 197. [Google Scholar]
- Bartl, I.; Hellemann, D.; Rabouille, C.; Schulz, K.; Tallberg, P.; Hietanen, S.; Voss, M. Particulate organic matter controls benthic microbial N retention and N removal in contrasting estuaries of the Baltic Sea. Biogeosciences 2019, 16, 3543–3564. [Google Scholar] [CrossRef]
- Łukawska-Matuszewska, K.; Burska, D.; Niemirycz, E. Toxicity Assessment by Microtox® in Sediments, Pore Waters and Sediment Saline Elutriates in the Gulf of Gdańsk (Baltic Sea). CLEAN–Soil Air Water 2009, 37, 592–598. [Google Scholar] [CrossRef]
- HELCOM. Input of Nutrients by the Seven Biggest Rivers in the Baltic Sea Region; Baltic Sea Environment Proceedings; Helsinki Commission: Helsinki, Finland, 2018; No.161. [Google Scholar]
- Witek, Z.; Humborg, C.; Savchuk, O.; Grelowski, A.; Łysiak-Pastuszak, E. Nitrogen and phosphorus budgets of the Gulf of Gdańsk (Baltic Sea). Estuar. Coast. Shelf Sci. 2003, 57, 239–248. [Google Scholar] [CrossRef]
- Staniszewska, M.; Koniecko, I.; Falkowska, L.; Krzymyk, E. Occurrence and distribution of bisphenol A and alkylphenols in the water of the gulf of Gdansk (Southern Baltic). Mar. Pollut. Bull. 2015, 91, 372–379. [Google Scholar] [CrossRef]
- Zaborska, A.; Siedlewicz, G.; Szymczycha, B.; Dzierzbicka-Głowacka, L.; Pazdro, K. Legacy and emerging pollutants in the Gulf of Gdańsk (southern Baltic Sea)–loads and distribution revisited. Mar. Pollut. Bull. 2019, 139, 238–255. [Google Scholar] [CrossRef] [PubMed]
- Saniewska, D.; Bełdowska, M.; Szymczak, E.; Kuliński, K.; Bełdowski, J.; Voss, M.; Pryputniewicz-Flis, D.; Burska, D. Processes affecting the transformation of mercury in the coastal zone in the vicinity of two river mouths in the southern Baltic Sea. Mar. Chem. 2022, 238, 104065. [Google Scholar] [CrossRef]
- Van Hoey, G.; Permuy, D.C.; Vandendriessche, S.; Vincx, M.; Hostens, K. An ecological quality status assessment procedure for soft-sediment benthic habitats: Weighing alternative approaches. Ecol. Indic. 2013, 25, 266–278. [Google Scholar] [CrossRef]
- Rees, W.E. Ecological Footprints and Appropriated Carrying Capacity: What Urban Economics Leaves Out. Environ. Urban. 1992, 4, 121–130. [Google Scholar] [CrossRef]
- Solan, M.; Cardinale, B.J.; Downing, A.L.; Engelhardt, K.A.M.; Ruesink, J.L.; Srivastava, D.S. Extinction and Ecosystem Function in the Marine Benthos. Science 2004, 306, 1177–1180. [Google Scholar] [CrossRef]
- Queirós, A.M.; Birchenough, S.N.R.; Bremner, J.; Godbold, J.A.; Parker, R.E.; Romero-Ramirez, A.; Reiss, H.; Solan, M.; Somerfield, P.J.; Van Colen, C.; et al. A bioturbation classification of European marine infaunal invertebrates. Ecol. Evol. 2013, 3, 3958–3985. [Google Scholar] [CrossRef]
- Renz, J.; Powilleit, M.; Gogina, M.; Zettler, M.; Morys, C.; Forster, S. Community bioirrigation potential (BIPc), an index to quantify the potential for solute exchange at the sediment-water interface. Mar. Environ. Res. 2018, 141, 214–224. [Google Scholar] [CrossRef]
- Wrede, A.; Holstein, J.; Brey, T. Choosing the lesser evil—A case study on quantification of sediment reworking rates in multi corer type sediment cores. J. Exp. Mar. Biol. Ecol. 2018, 514–515, 1–9. [Google Scholar] [CrossRef]
- Nasi, F.; Ferrante, L.; Alvisi, F.; Bonsdorff, E.; Auriemma, R.; Cibic, T. Macrofaunal bioturbation attributes in relation to riverine influence: What can we learn from the Po River lagoonal system (Adriatic Sea)? Estuar. Coast. Shelf Sci. 2020, 232, 106405. [Google Scholar] [CrossRef]
- Braeckman, U.; Foshtomi, M.Y.; Van Gansbeke, D.; Meysman, F.; Soetaert, K.; Vincx, M.; Vanaverbeke, J. Variable Importance of Macrofaunal Functional Biodiversity for Biogeochemical Cycling in Temperate Coastal Sediments. Ecosystems 2014, 17, 720–737. [Google Scholar] [CrossRef]
- Villnäs, A.; Norkko, J.; Lukkari, K.; Hewitt, J.; Norkko, A. Consequences of Increasing Hypoxic Disturbance on Benthic Communities and Ecosystem Functioning. PLoS ONE 2012, 7, e44920. [Google Scholar] [CrossRef]
- Birchenough, S.; Parker, R.; McManus, E.; Barry, J. Combining bioturbation and redox metrics: Potential tools for assessing seabed function. Ecol. Indic. 2012, 12, 8–16. [Google Scholar] [CrossRef]
- Queirós, A.M.; Stephens, N.; Cook, R.; Ravaglioli, C.; Nunes, J.; Dashfield, S.; Harris, C.; Tilstone, G.H.; Fishwick, J.; Braeckman, U.; et al. Can benthic community structure be used to predict the process of bioturbation in real ecosystems? Prog. Oceanogr. 2015, 137, 559–569. [Google Scholar] [CrossRef]
- Thoms, F.; Burmeister, C.; Dippner, J.W.; Gogina, M.; Janas, U.; Kendzierska, H.; Liskow, I.; Voss, M. Impact of Macrofaunal Communities on the Coastal Filter Function in the Bay of Gdansk, Baltic Sea. Front. Mar. Sci. 2018, 5, 1–19. [Google Scholar] [CrossRef]
- Janas, U.; Burska, D.; Kendzierska, H.; Pryputniewicz-Flis, D.; Łukawska-Matuszewska, K. Importance of benthic macrofauna and coastal biotopes for ecosystem functioning–Oxygen and nutrient fluxes in the coastal zone. Estuar. Coast. Shelf Sci. 2019, 225, 106238. [Google Scholar] [CrossRef]
- Kendzierska, H.; Łukawska-Matuszewska, K.; Burska, D.; Janas, U. Benthic fluxes of oxygen and nutrients under the influence of macrobenthic fauna on the periphery of the intermittently hypoxic zone in the Baltic Sea. J. Exp. Mar. Biol. Ecol. 2020, 530–531, 151439. [Google Scholar] [CrossRef]
- Silberberger, M.J.; Koziorowska-Makuch, K.; Borawska, Z.; Szczepanek, M.; Kędra, M. Disentangling the Drivers of Benthic Oxygen and Dissolved Carbon Fluxes in the Coastal Zone of the Southern Baltic Sea. Estuaries Coasts 2022, 45, 2450–2471. [Google Scholar] [CrossRef]
- Szczepanek, M.; Silberberger, M.J.; Koziorowska-Makuch, K.; Nobili, E.; Kędra, M. The response of coastal macrobenthic food-web structure to seasonal and regional variability in organic matter properties. Ecol. Indic. 2021, 132, 108326. [Google Scholar] [CrossRef]
- Szczepanek, M.; Silberberger, M.J.; Koziorowska-Makuch, K.; Kędra, M. Utilization of riverine organic matter by macrobenthic communities in a temperate prodelta. Front. Mar. Sci. 2022, 9, 1–15. [Google Scholar] [CrossRef]
- Dye, A.H. Seasonal Fluctuations in the Vertical Distribution of Meiofauna in Estuarine Sediments. Zool. Afr. 1978, 13, 207–212. [Google Scholar] [CrossRef]
- Reading, C.; McGrorty, S. Seasonal variations in the burying depth of Macoma balthica (L.) and its accessibility to wading birds. Estuar. Coast. Mar. Sci. 1978, 6, 135–144. [Google Scholar] [CrossRef]
- Hill, C.; Elmgren, R. Vertical Distribution in the Sediment in the Co-Occurring Benthic Amphipods Pontoporeia affinis and P. femorata. Oikos 1987, 49, 221. [Google Scholar] [CrossRef]
- Renz, J.; Forster, S. Effects of bioirrigation by the three sibling species of Marenzelleria spp. on solute fluxes and porewater nutrient profiles. Mar. Ecol. Prog. Ser. 2014, 505, 145–159. [Google Scholar] [CrossRef]
- Haque, A.; Szymelfenig, M.; Wȩsławski, J.M. Small-scale vertical distribution of zoobenthos in the sandy littoral of the Gulf of Gdańsk. Oceanologia 1997, 39, 433–446. [Google Scholar]
- Łęczyński, L.; Szymczak, E. Własności fizyczne osadów dennych. In Fizyczne, Biologiczne i Chemiczne Badania Morskich Osadów Dennych; Wydawnictwo UG: Gdańsk, Poland, 2010. [Google Scholar]
- Wentworth, C.K. A Scale of Grade and Class Terms for Clastic Sediments. J. Geol. 1922, 30, 377–392. [Google Scholar] [CrossRef]
- Salonen, K.; Sarvala, J.; Hakala, I.; Viljanen, M.-L. The relation of energy and organic carbon in aquatic invertebrates1. Limnol. Oceanogr. 1976, 21, 724–730. [Google Scholar] [CrossRef]
- Rumohr, H.; Brey, T.; Ankar, S. A Compilation of Biometric Conversion Factors for Benthic Invertebrates of the Baltic Sea. Balt. Mar. Biol. 1987, 9, 62. [Google Scholar]
- Zettler, M.L. Population dynamics, growth and production of the neozoon Marenzelleria cf. viridis (Verrill, 1873) (Polychaeta: Spionidae) in a coastal water of the southern Baltic Sea. Aquat. Ecol. 1997, 13, 177–186. [Google Scholar] [CrossRef]
- Ricciardi, A.; Bourget, E. Weight-to-weight conversion factors for marine benthic macroinvertebrates. Mar. Ecol. Prog. Ser. 1998, 163, 245–251. [Google Scholar] [CrossRef]
- Wrede, A.; Beermann, J.; Dannheim, J.; Gutow, L.; Brey, T. Organism functional traits and ecosystem supporting services—A novel approach to predict bioirrigation. Ecol. Indic. 2017, 91, 737–743. [Google Scholar] [CrossRef]
- Clarke, K.R. Non-parametric multivariate analyses of changes in community structure. Aust. J. Ecol. 1993, 18, 117–143. [Google Scholar] [CrossRef]
- Anderson, M.; Gorley, R.; Clarke, K.P. For PRIMER: Guide to Software and Statistical Methods; PRIMER-E: Plymouth, UK, 2008. [Google Scholar]
- Elliott, M.; Whitfield, A. Challenging paradigms in estuarine ecology and management. Estuar. Coast. Shelf Sci. 2011, 94, 306–314. [Google Scholar] [CrossRef]
- Ducrotoy, J.P.; Michael, E.; Cutts, N.; Franco, A.; Little, S.; Mazik, K.; Wilkinson, M. Temperate Estuaries: Their Ecology Under Future Environmental Changes. In Coasts and Estuaries: The Future; Wolanski, E., Day, J.W., Elliott, M., Ramachandran, R., Eds.; Elsevier: Oxford, UK, 2019; pp. 577–594. ISBN 9780128140031. [Google Scholar]
- Janas, U.; Szaniawska, A. The influence of hydrogen sulphide on macrofaunal biodiversity in the Gulf of Gdańsk. Oceanologia 1996, 38, 127–142. [Google Scholar]
- Pempkowiak, J.; Kupryszewski, G. The input of organic matter to the Baltic from the Vistula River. Oceanology 1980, 12, 79–98. [Google Scholar]
- Voss, M.; Liskow, I.; Pastuszak, M.; Rüβ, D.; Schulte, U.; Dippner, J. Riverine discharge into a coastal bay: A stable isotope study in the Gulf of Gdańsk, Baltic Sea. J. Mar. Syst. 2005, 57, 127–145. [Google Scholar] [CrossRef]
- Voss, M.; Asmala, E.; Bartl, I.; Carstensen, J.; Conley, D.J.; Dippner, J.W.; Humborg, C.; Lukkari, K.; Petkuviene, J.; Reader, H.; et al. Origin and fate of dissolved organic matter in four shallow Baltic Sea estuaries. Biogeochemistry 2020, 154, 385–403. [Google Scholar] [CrossRef]
- Reader, H.E.; Thoms, F.; Voss, M.; Stedmon, C.A. The Influence of Sediment-Derived Dissolved Organic Matter in the Vistula River Estuary/Gulf of Gdansk. J. Geophys. Res. Biogeosci. 2019, 124, 115–126. [Google Scholar] [CrossRef]
- Asmala, E.; Carstensen, J.; Conley, D.J.; Slomp, C.P.; Stadmark, J.; Voss, M. Efficiency of the coastal filter: Nitrogen and phosphorus removal in the Baltic Sea. Limnol. Oceanogr. 2017, 62, S222–S238. [Google Scholar] [CrossRef]
- Nybom, I.; Horlitz, G.; Gilbert, D.; Berrojalbiz, N.; Martens, J.; Arp, H.P.H.; Sobek, A. Effects of Organic Carbon Origin on Hydrophobic Organic Contaminant Fate in the Baltic Sea. Environ. Sci. Technol. 2021, 55, 13061–13071. [Google Scholar] [CrossRef]
- Kuliński, K.; Rehder, G.; Asmala, E.; Bartosova, A.; Carstensen, J.; Gustafsson, B.; Hall, P.O.J.; Humborg, C.; Jilbert, T.; Jürgens, K.; et al. Biogeochemical functioning of the Baltic Sea. Earth Syst. Dyn. 2022, 13, 633–685. [Google Scholar] [CrossRef]
- Janas, U.; Wocial, J.; Szaniawska, A. Seasonal and annual changes in the macrozoobenthic populations of the Gulf of Gdańsk with respect to hypoxia and hydrogen sulphide. Oceanologia 2004, 46, 85–102. Available online: http://www.iopan.gda.pl/oceanologia/86 (accessed on 15 November 2022).
- Lukawska-Matuszewska, K.; Kielczewska, J. Effects of near-bottom water oxygen concentration on biogeochemical cycling of C, N and S in sediments of the Gulf of Gdansk (southern Baltic). Cont. Shelf Res. 2016, 117, 30–42. [Google Scholar] [CrossRef]
- Łukawska-Matuszewska, K.; Graca, B. Pore water alkalinity below the permanent halocline in the Gdańsk Deep (Baltic Sea)-Concentration variability and benthic fluxes. Mar. Chem. 2018, 204, 49–61. [Google Scholar] [CrossRef]
- Kotwicki, L. Macrozoobenthos of the sandy littoral zone of the Gulf of Gdańsk. Oceanologia 1997, 39, 447–460. [Google Scholar]
- Włodarska-Kowalczuk, M.; Mazurkiewicz, M.; Jankowska, E.; Kotwicki, L.; Damrat, M.; Zajączkowski, M. Effects of fluvial discharges on meiobenthic and macrobenthic variability in the Vistula River prodelta (Baltic Sea). J. Mar. Syst. 2016, 157, 135–146. [Google Scholar] [CrossRef]
- Zajaczkowski, M.; Włodarska-Kowalczuk, M. Dynamic sedimentary environments of an Arctic glacier-fed river estuary (Adventfjorden, Svalbard). I. Flux, deposition, and sediment dynamics. Estuar. Coast. Shelf Sci. 2007, 74, 285–296. [Google Scholar] [CrossRef]
- Wodarska-Kowalczuk, M.; Kedra, M. Surrogacy in natural patterns of benthic distribution and diversity: Selected taxa versus lower taxonomic resolution. Mar. Ecol. Prog. Ser. 2007, 351, 53–63. [Google Scholar] [CrossRef]
- Rhoads, D.C.; Boesch, D.F.; Zhican, T.; Fengshan, X.; Liqiang, H.; Nilsen, K.J. Macrobenthos and sedimentary facies on the Changjiang delta platform and adjacent continental shelf, East China Sea. Cont. Shelf Res. 1985, 4, 189–213. [Google Scholar] [CrossRef]
- Diaz, R.J.; Rosenberg, R. Marine benthic hypoxia: A review of its ecological effects and the behavioural responses of benthic macrofauna. Oceanogr. Mar. Biol. Annu. Rev. 1995, 33, 245–303. [Google Scholar]
- Levin, L.A.; Ekau, W.; Gooday, A.J.; Jorissen, F.; Middelburg, J.J.; Naqvi, S.W.A.; Neira, C.; Rabalais, N.N.; Zhang, J. Effects of natural and human-induced hypoxia on coastal benthos. Biogeosciences 2009, 6, 2063–2098. [Google Scholar] [CrossRef]
- Villnäs, A.; Norkko, J.; Hietanen, S.; Josefson, A.B.; Lukkari, K.; Norkko, A. The role of recurrent disturbances for ecosystem multifunctionality. Ecology 2013, 94, 2275–2287. [Google Scholar] [CrossRef] [PubMed]
- Michaud, E.; Desrosiers, G.; Mermillod-Blondin, F.; Sundby, B.; Stora, G. The functional group approach to bioturbation: The effects of biodiffusers and gallery-diffusers of the Macoma balthica community on sediment oxygen uptake. J. Exp. Mar. Biol. Ecol. 2005, 326, 77–88. [Google Scholar] [CrossRef]
- Michaud, E.; Desrosiers, G.; Mermillod-Blondin, F.; Sundby, B.; Stora, G. The functional group approach to bioturbation: II. The effects of the Macoma balthica community on fluxes of nutrients and dissolved organic carbon across the sediment–water interface. J. Exp. Mar. Biol. Ecol. 2006, 337, 178–189. [Google Scholar] [CrossRef]
- Quintana, C.O.; Tang, M.; Kristensen, E. Simultaneous study of particle reworking, irrigation transport and reaction rates in sediment bioturbated by the polychaetes Heteromastus and Marenzelleria. J. Exp. Mar. Biol. Ecol. 2007, 352, 392–406. [Google Scholar] [CrossRef]
- Kristensen, E.; Hansen, T.; Delefosse, M.; Banta, G.T.; Quintana, C.O. Contrasting effects of the polychaetes Marenzelleria viridis and Nereis diversicolor on benthic metabolism and solute transport in sandy coastal sediment. Mar. Ecol. Prog. Ser. 2011, 425, 125–139. [Google Scholar] [CrossRef]
- Gogina, M.; Renz, J.R.; Forster, S.; Zettler, M.L. Benthic Macrofauna Community Bioirrigation Potential (BIPc): Regional Map and Utility Validation for the South-Western Baltic Sea. Biology 2022, 11, 1085. [Google Scholar] [CrossRef]
- Toussaint, E.; De Borger, E.; Braeckman, U.; De Backer, A.; Soetaert, K.; Vanaverbeke, J. Faunal and environmental drivers of carbon and nitrogen cycling along a permeability gradient in shallow North Sea sediments. Sci. Total. Environ. 2021, 767, 144994. [Google Scholar] [CrossRef]
- De Borger, E.; Tiano, J.; Braeckman, U.; Ysebaert, T.; Soetaert, K. Biological and biogeochemical methods for estimating bioirrigation: A case study in the Oosterschelde estuary. Biogeosciences 2020, 17, 1701–1715. [Google Scholar] [CrossRef]
- Ouellette, D.; Desrosiers, G.; Gagné, J.; Gilbert, F.; Poggiale, J.; Blier, P.; Stora, G. Effects of temperature on in vitro sediment reworking processes by a gallery biodiffusor, the polychaete Neanthes virens. Mar. Ecol. Prog. Ser. 2004, 266, 185–193. [Google Scholar] [CrossRef]
- Wohlgemuth, S.; Taylor, A.; Grieshaber, M. Ventilatory and metabolic responses to hypoxia and sulphide in the lugworm Arenicola marina (L.). J. Exp. Biol. 2000, 203, 3177–3188. [Google Scholar] [CrossRef] [PubMed]
- Hagerman, L. Physiological flexibility; a necessity for life in anoxic and sulphidic habitats. Hydrobiologia 1998, 375, 241–254. [Google Scholar] [CrossRef]
- Haselmair, A.; Stachowitsch, M.; Zuschin, M.; Riedel, B. Behaviour and mortality of benthic crustaceans in response to experimentally induced hypoxia and anoxia in situ. Mar. Ecol. Prog. Ser. 2010, 414, 195–208. [Google Scholar] [CrossRef]
- Lindström, M.; Sandberg-Kilpi, E. Breaking the boundary—The key to bottom recovery? The role of mysid crustaceans in oxygenizing bottom sediments. J. Exp. Mar. Biol. Ecol. 2008, 354, 161–168. [Google Scholar] [CrossRef]
- Tallqvist, M. Burrowing behaviour of the Baltic clam Macoma balthica: Effects of sediment type, hypoxia and predator presence. Mar. Ecol. Prog. Ser. 2001, 212, 183–191. [Google Scholar] [CrossRef]
- Zettler, M.; Bick, A.; Arch, R.B. Fish. Mar. Res, and Undefined 1995, Distribution and population dynamics of Marenzelleria viridis (Polychaeta, Spionidae) in a coastal water of the southern Baltic. Arch. Fish. Mar. Res. 2005, 42, 209–224. Available online: https://www.academia.edu/download/45531524/Distribution_and_population_dynamics_of_20160511-21785-periqn.pdf (accessed on 7 November 2022).
- Bonaglia, S.; Bartoli, M.; Gunnarsson, J.; Rahm, L.; Raymond, C.; Svensson, O.; Yekta, S.S.; Brüchert, V. Effect of reoxygenation and Marenzelleria spp. bioturbation on Baltic Sea sediment metabolism. Mar. Ecol. Prog. Ser. 2014, 482, 43–55. [Google Scholar] [CrossRef]
- Maximov, A.; Bonsdorff, E.; Eremina, T.; Kauppi, L.; Norkko, A.; Norkko, J. Context-dependent consequences of Marenzelleria spp. (Spionidae: Polychaeta) invasion for nutrient cycling in the Northern Baltic Sea. Oceanologia 2015, 57, 342–348. [Google Scholar] [CrossRef]
- Johansson, B. Behavioural response to gradually declining oxygen concentration by Baltic Sea macrobenthic crustaceans. Mar. Biol. 1997, 129, 71–78. [Google Scholar] [CrossRef]
- Villnäs, A.; Norkko, A.; Lehtonen, K.K. Multi-level responses of Macoma balthica to recurring hypoxic disturbance. J. Exp. Mar. Biol. Ecol. 2019, 510, 64–72. [Google Scholar] [CrossRef]
- Wrede, A.; Dannheim, J.; Gutow, L.; Brey, T. Who really matters: Influence of German Bight key bioturbators on biogeochemical cycling and sediment turnover. J. Exp. Mar. Biol. Ecol. 2017, 488, 92–101. [Google Scholar] [CrossRef]
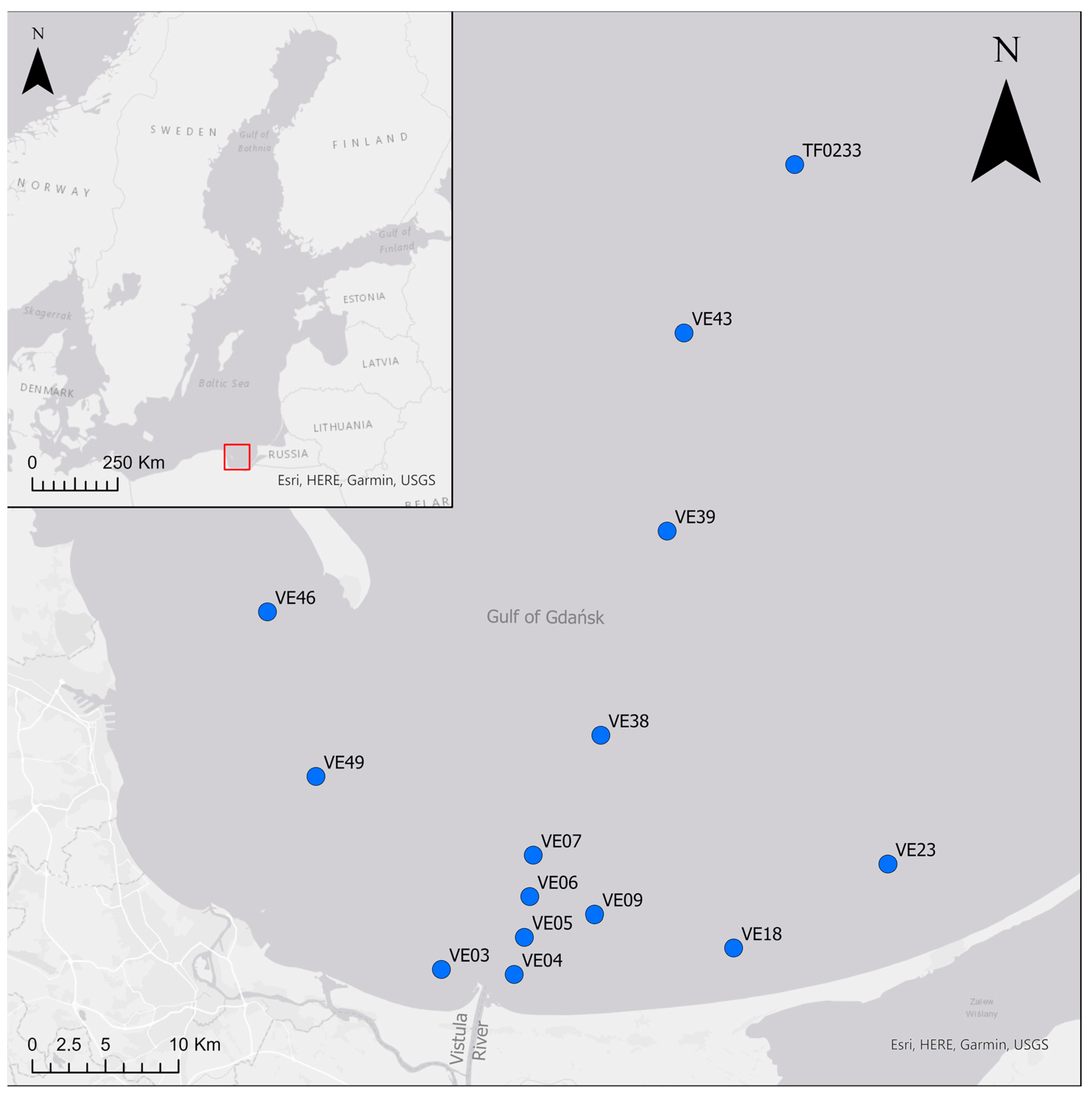



| Station | Temperature [°C] | Salinity | Oxygen [mL·dm−3] | Sediment Type | LOI [%] | Depth [m] |
|---|---|---|---|---|---|---|
| VE04 | 4.2 | 7.6 | 8.10 | Fine-grained sand | 1.25 | 15 |
| VE03 | 14.2 | 7.4 | 5.65 | Fine-grained sand | 4.49 | 16 |
| VE05 | 12.6 | 7.4 | 4.68 | Fine-grained sand | 4.03 | 24 |
| VE18 | 11.2 | 7.3 | 5.83 | Fine-grained sand | 0.91 | 24 |
| VE49 | 6.2 | 7.6 | 5.93 | Medium-grained sand | 0.89 | 25 |
| VE09 | 5.3 | 8.0 | 6.32 | Medium-grained sand | 0.81 | 32 |
| VE06 | 3.8 | 8.0 | 8.19 | Fine-grained sand | 0.88 | 38 |
| VE23 | 5.0 | 8.0 | 6.47 | Sandy silt | 4.24 | 48 |
| VE46 | 4.6 | 8.2 | 5.90 | Silt | 13.26 | 48 |
| VE07 | 3.7 | 8.0 | 8.33 | Fine-grained sand | 3.07 | 59 |
| VE38 | 4.5 | 9.1 | 3.41 | Silt | 4.40 | 67 |
| VE39 | 5.3 | 11.2 | 2.56 | Silty clay | 18.54 | 84 |
| VE43 | 5.8 | 12.3 | 1.52 | Silty clay | 3.18 | 94 |
| TF0233 | 5.6 | 12.7 | 1.59 | Silty clay | 15.49 | 105 |
| Variable | PC1 | PC2 | PC3 |
|---|---|---|---|
| Variation [%] | 59.6 | 26.9 | 11.7 |
| Temperature (T) | 0.128 | 0.939 | 0.104 |
| Salinity (S) | −0.612 | −0.100 | −0.366 |
| Oxygen (DO) | 0.576 | −0.329 | 0.366 |
| LOI | −0.527 | −0.015 | 0.849 |
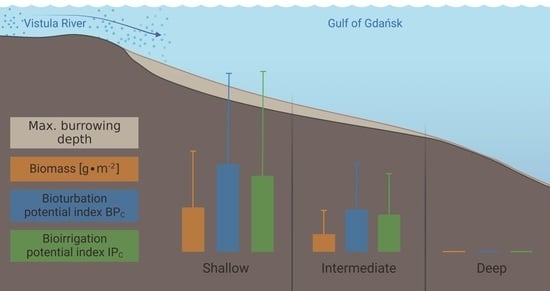
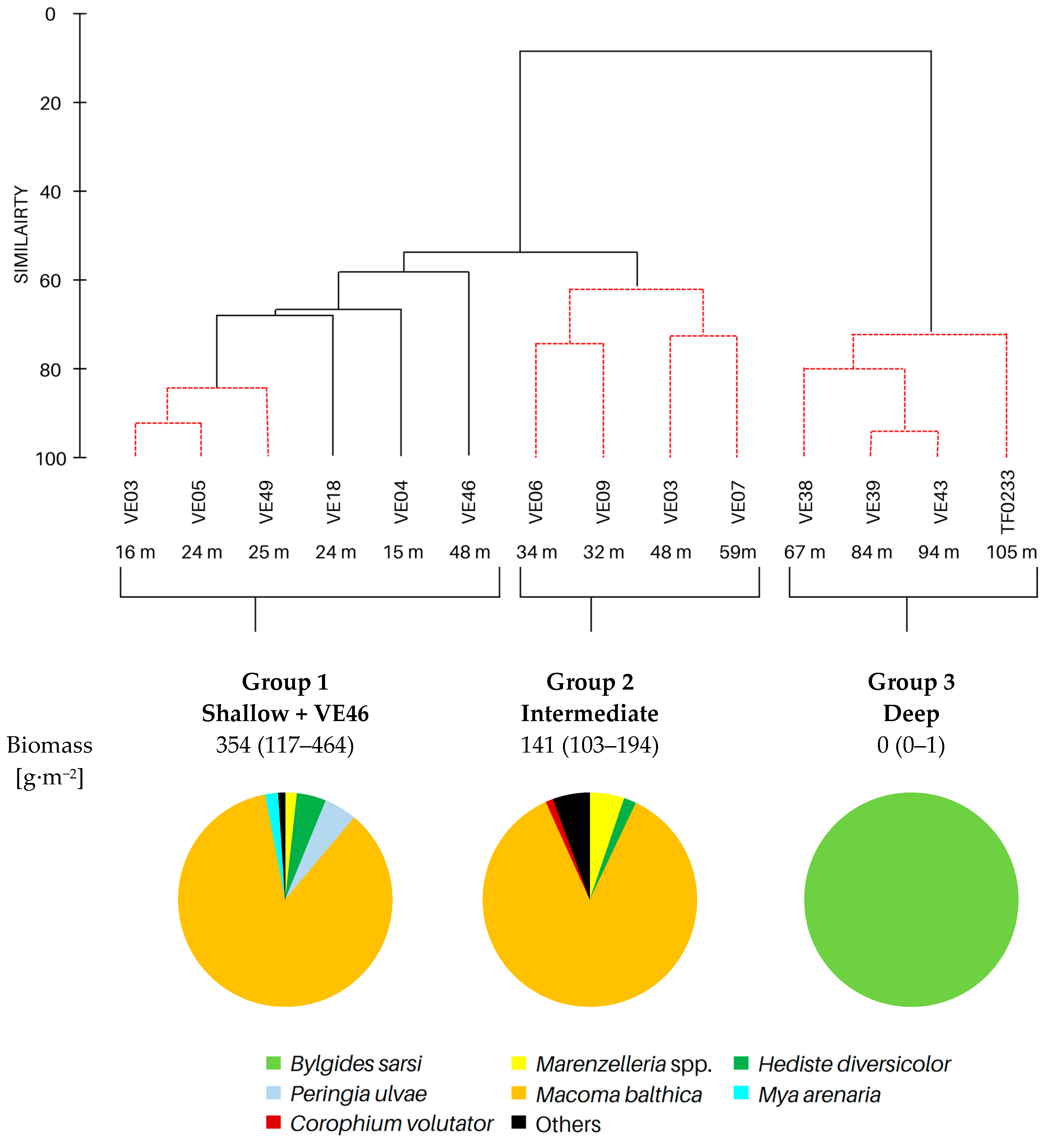
| Group 1 | Group 2 | Group 3 | |
|---|---|---|---|
| No. of taxa | 11 (6–16) | 8 (7–10) | 0 (0–1) |
| Max. burial depth [cm] | 14 (10–15) | 11 (6–15) | 0 (0–1) |
| Abundance [ind.∙m−2] | 11,030 (3628–30,557) | 3188 (2578–3851) | 63 (0–127) |
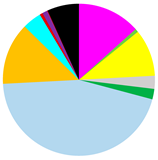 |  |  | |
| BPC | 3412 (1576–5000) | 1683 (1412–1890) | 5 (0–13) |
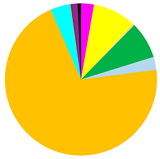 | 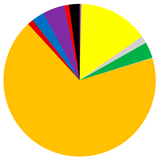 |  | |
| IPC | 1451 (581–1985) | 714 (632–785) | 1 (0–4) |
 |  |  | |
 | |||
| Biomass | Burial Depth | BPC | IPC | |||||
|---|---|---|---|---|---|---|---|---|
| Marginal Test | Sequential Test | Marginal Test | Sequential Test | Marginal Test | Sequential Test | Marginal Test | Sequential Test | |
| Salinity | 0.442 ** | 0.442 * | 0.644 ** | 0.644 ** | 0.529 ** | 0.529 ** | 0.564 ** | 0.564 ** |
| Temperature | 0.302 * | 0.153 | 0.03 | 0.425 * | 0.23 ** | 0.456 ** | 0.247 * | |
| LOI | 0.01 | 0.1021 | 0.250 | 0.027 | 0.076 * | 0.062 | ||
| Total | 0.697 | 0.644 | 0.835 | 0.811 | ||||
IDi lit. | Max. Burrowing Depth | No. of Taxa | Abundance | Biomass WW | Biomass AFDW | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| 0.999 | 0.991 | 0.978 | 0.982 | 0.982 | 0.672 | 0.756 | 0.934 | 0.968 | 0.970 | ||
| 0.990 | 0.990 | 0.986 | 0.985 | 0.678 | 0.766 | 0.938 | 0.965 | 0.968 | |||
| 0.984 | 0.977 | 0.976 | 0.663 | 0.732 | 0.915 | 0.989 | 0.990 | ||||
| 0.999 | 0.997 | 0.708 | 0.785 | 0.940 | 0.980 | 0.963 | |||||
| 0.997 | 0.720 | 0.792 | 0.942 | 0.953 | 0.956 | ||||||
IDi lit. | 0.718 | 0.785 | 0.935 | 0.949 | 0.953 | ||||||
| Max. burrowing depth | 0.730 | 0.717 | 0.637 | 0.637 | |||||||
| No. of taxa | 0.836 | 0.706 | 0.706 | ||||||||
| Abundance | 0.880 | 0.883 | |||||||||
| Biomass WW | 0.999 | ||||||||||
| Biomass AFDW |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Miernik, N.A.; Janas, U.; Kendzierska, H. Role of Macrofaunal Communities in the Vistula River Plume, the Baltic Sea—Bioturbation and Bioirrigation Potential. Biology 2023, 12, 147. https://doi.org/10.3390/biology12020147
Miernik NA, Janas U, Kendzierska H. Role of Macrofaunal Communities in the Vistula River Plume, the Baltic Sea—Bioturbation and Bioirrigation Potential. Biology. 2023; 12(2):147. https://doi.org/10.3390/biology12020147
Chicago/Turabian StyleMiernik, Natalia Anna, Urszula Janas, and Halina Kendzierska. 2023. "Role of Macrofaunal Communities in the Vistula River Plume, the Baltic Sea—Bioturbation and Bioirrigation Potential" Biology 12, no. 2: 147. https://doi.org/10.3390/biology12020147
APA StyleMiernik, N. A., Janas, U., & Kendzierska, H. (2023). Role of Macrofaunal Communities in the Vistula River Plume, the Baltic Sea—Bioturbation and Bioirrigation Potential. Biology, 12(2), 147. https://doi.org/10.3390/biology12020147