Diversity and Co-Occurrence Patterns of Fungal and Bacterial Communities from Alkaline Sediments and Water of Julong High-Altitude Hot Springs at Tianchi Volcano, Northeast China
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Description of the Sample Sites and Measurement of pH Values
2.2. Cultivable Microbe Isolation, Microscopy, and Molecular Analyses
2.3. Culture-Independent Microbial Diversity and Statistical Analysis
2.4. Co-Occurrence Network Construction and Analysis
3. Results
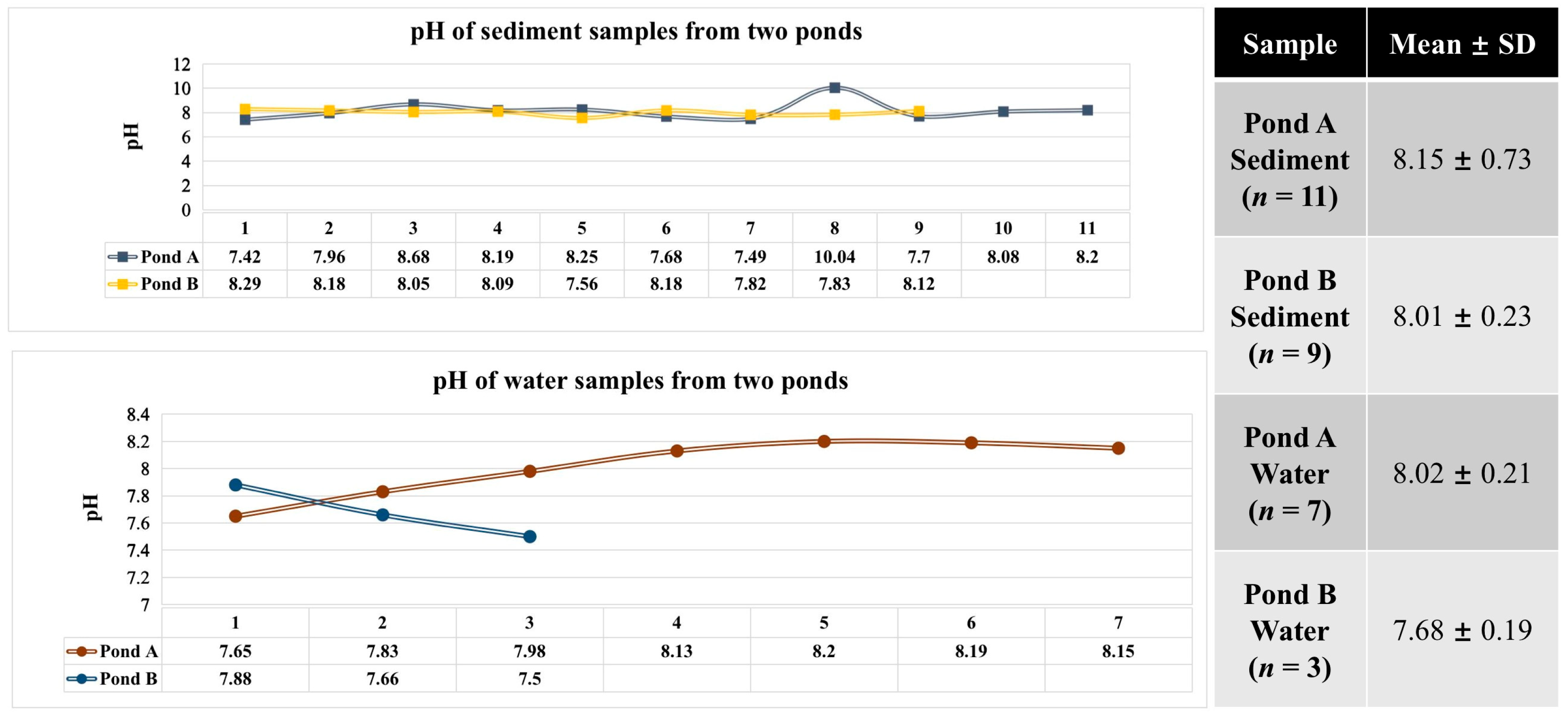
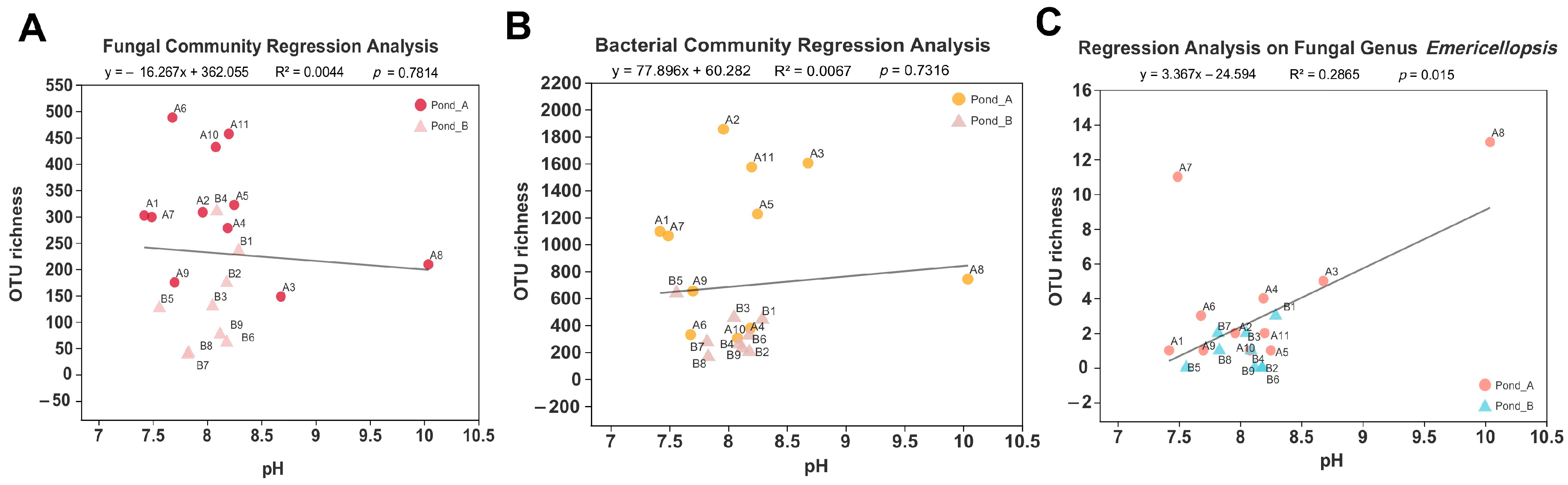
3.1. pH Measurement of Water and Sediment Samples
3.2. Diversity of Culturable Microbes
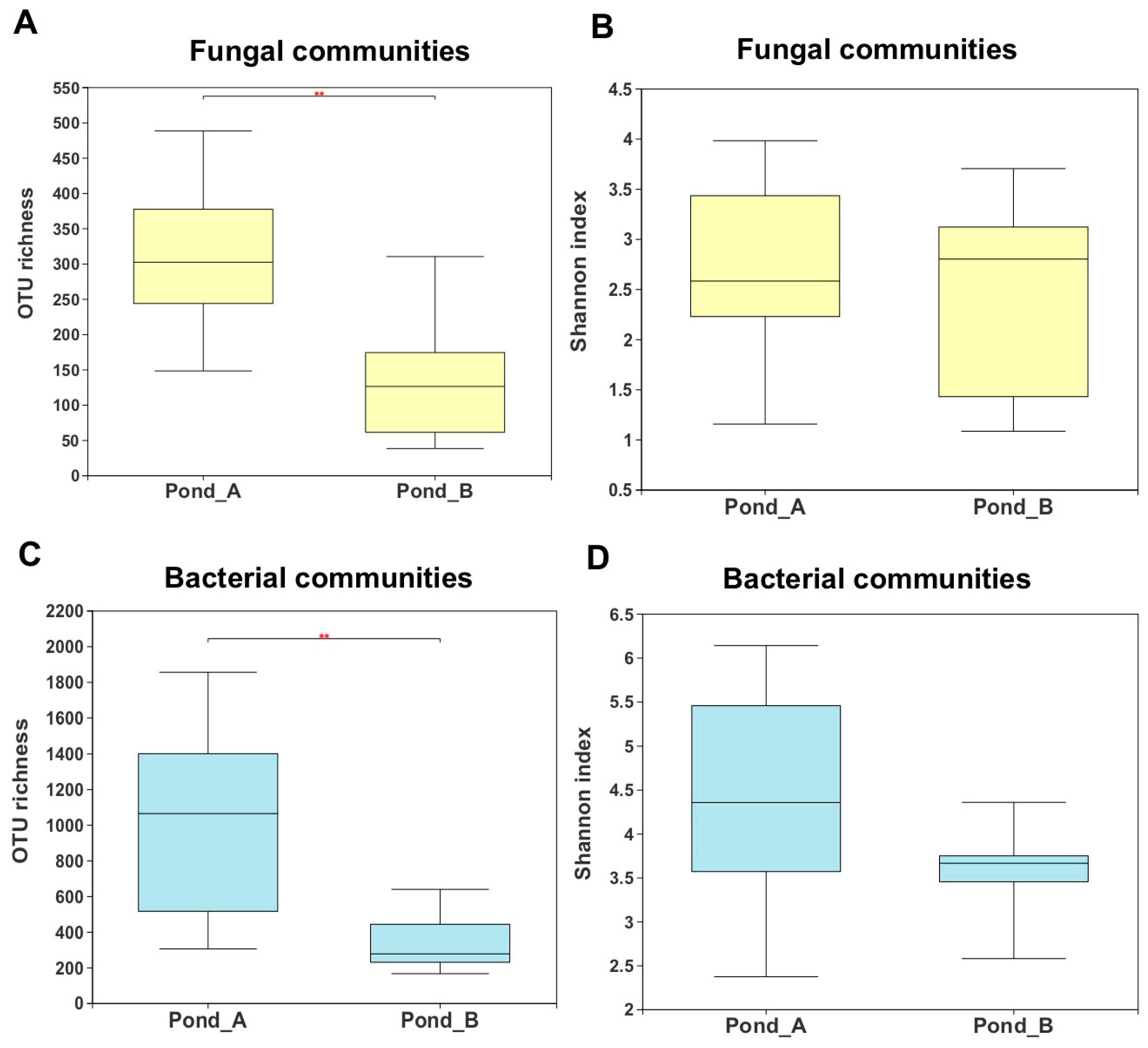
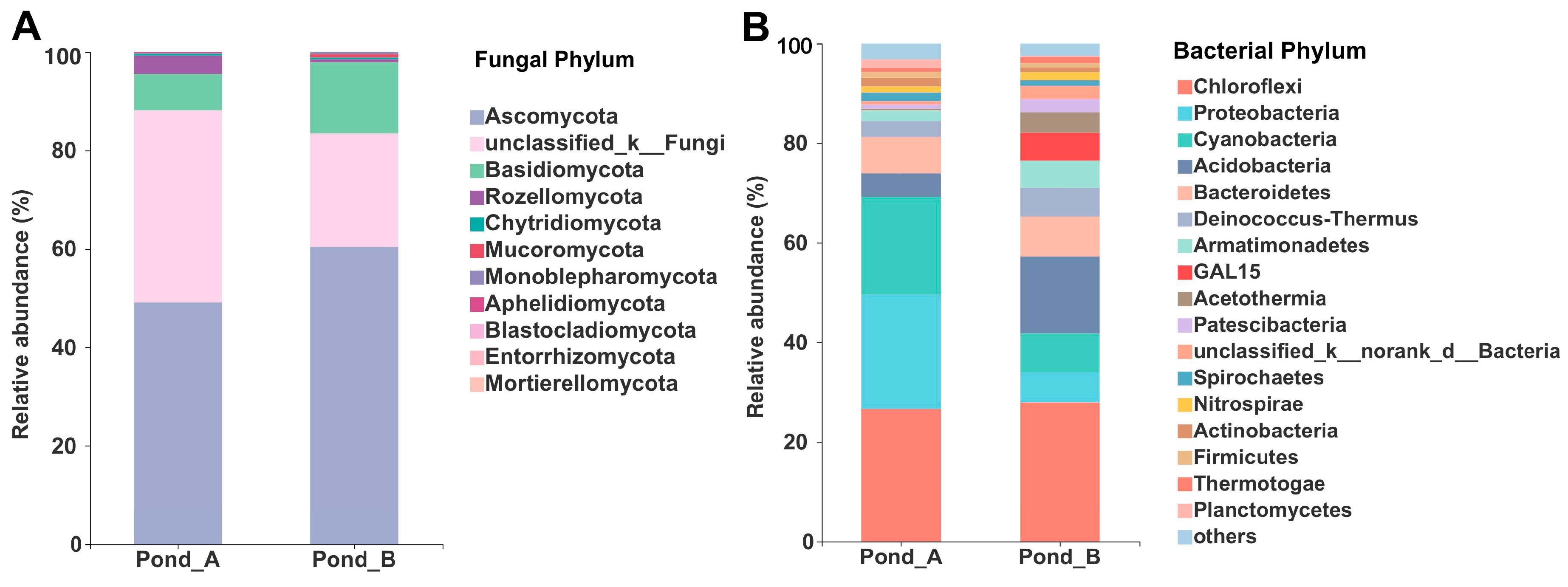
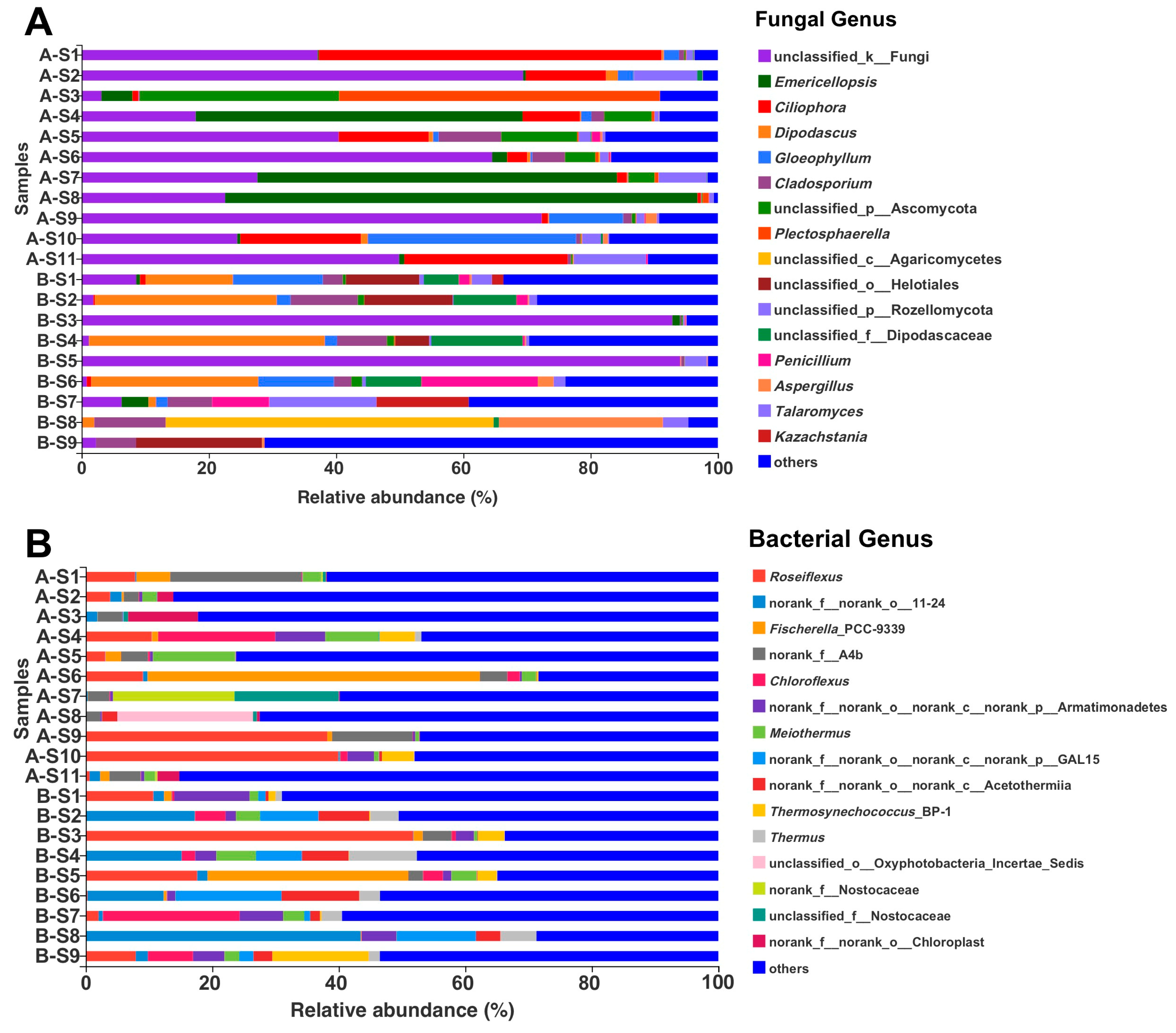
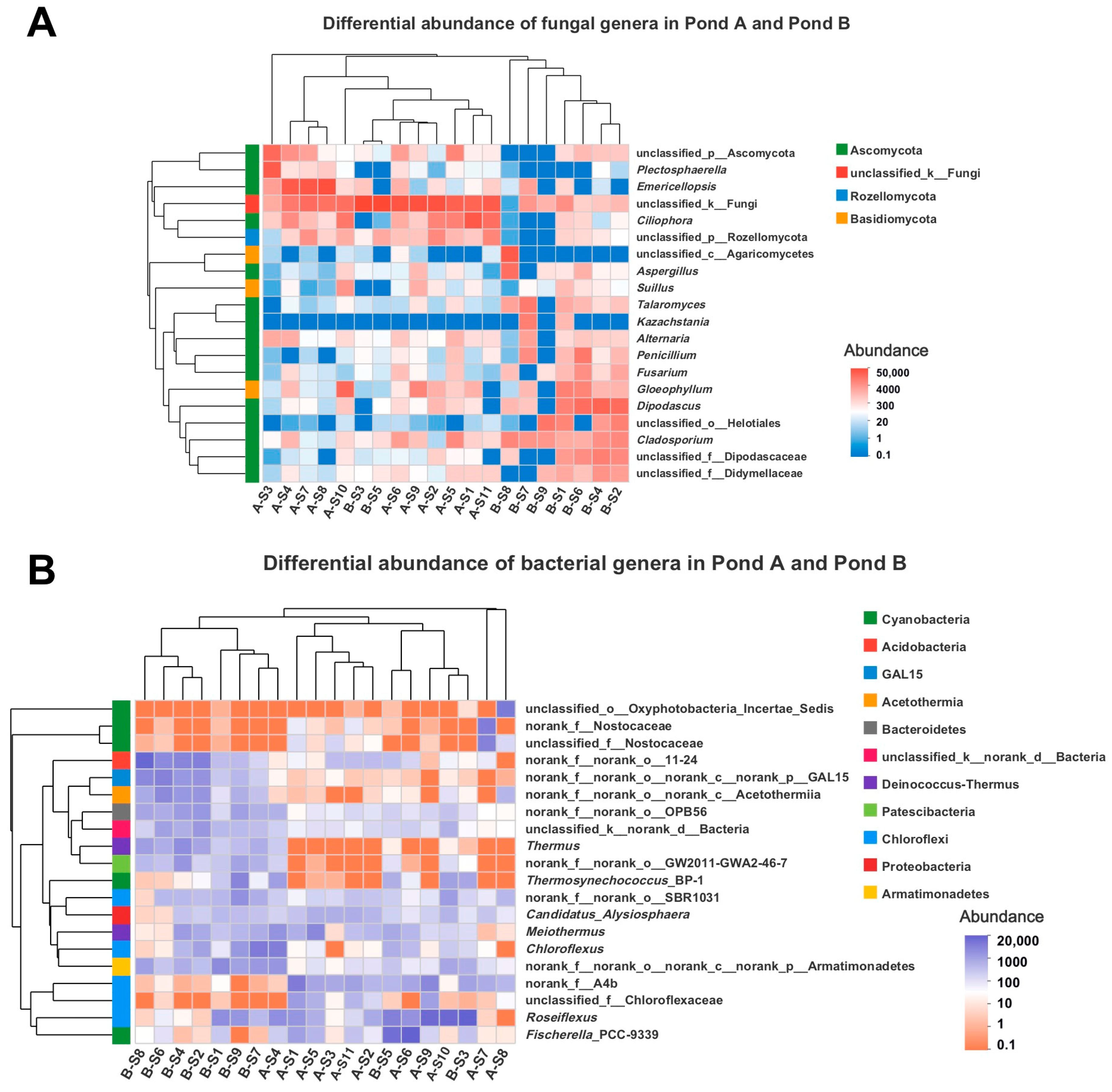
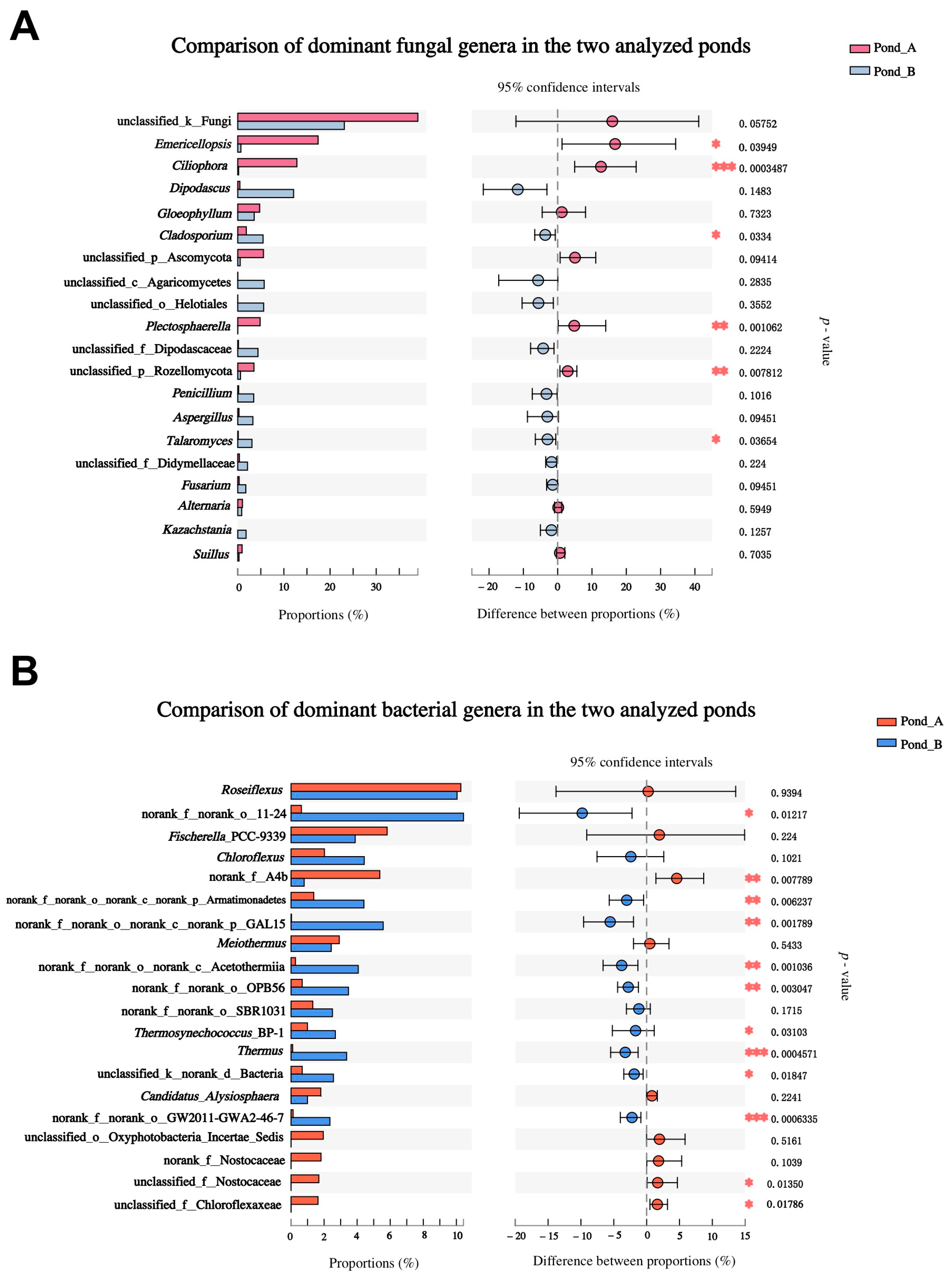
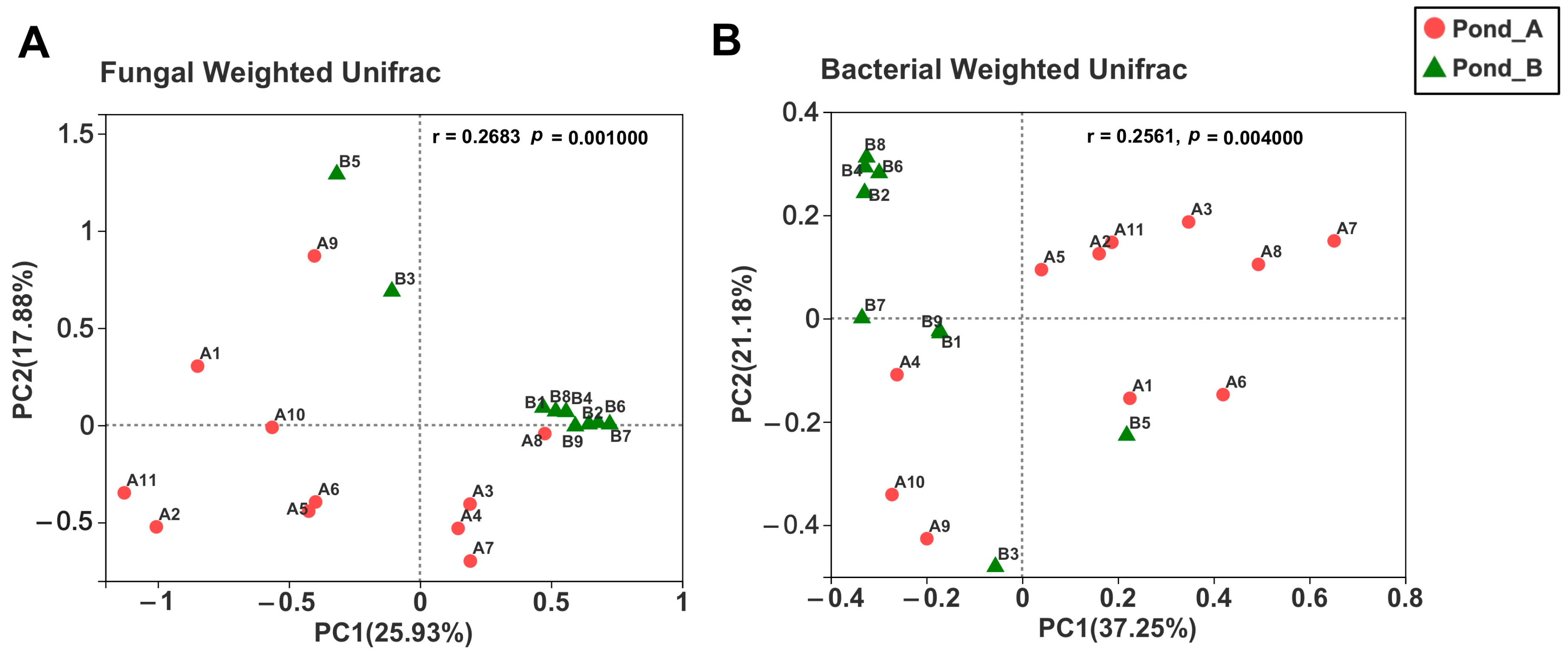
3.3. Microbial Diversity by Illumina Sequencing
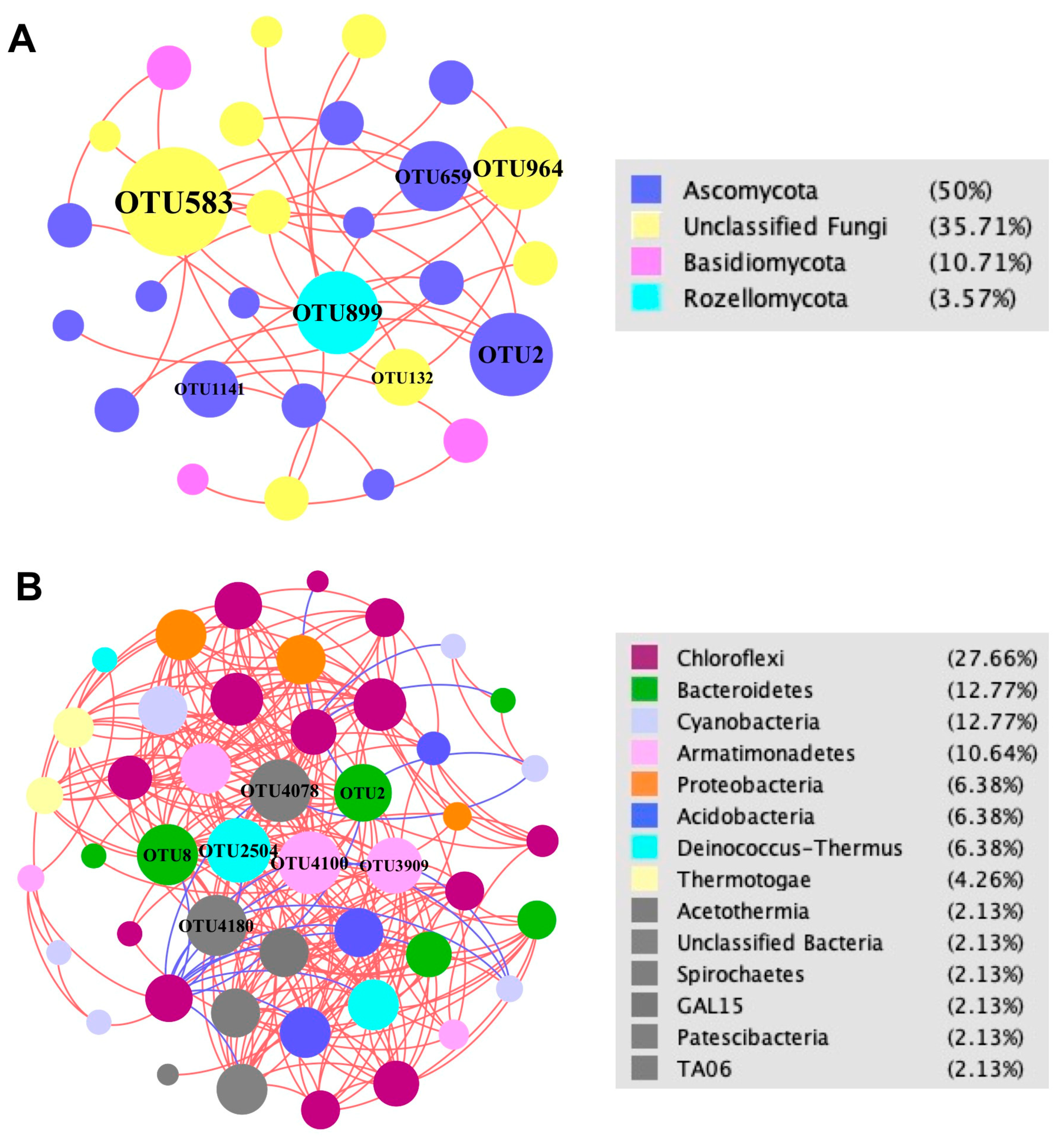
3.4. Fungal and Bacterial Co-Occurrence Networks
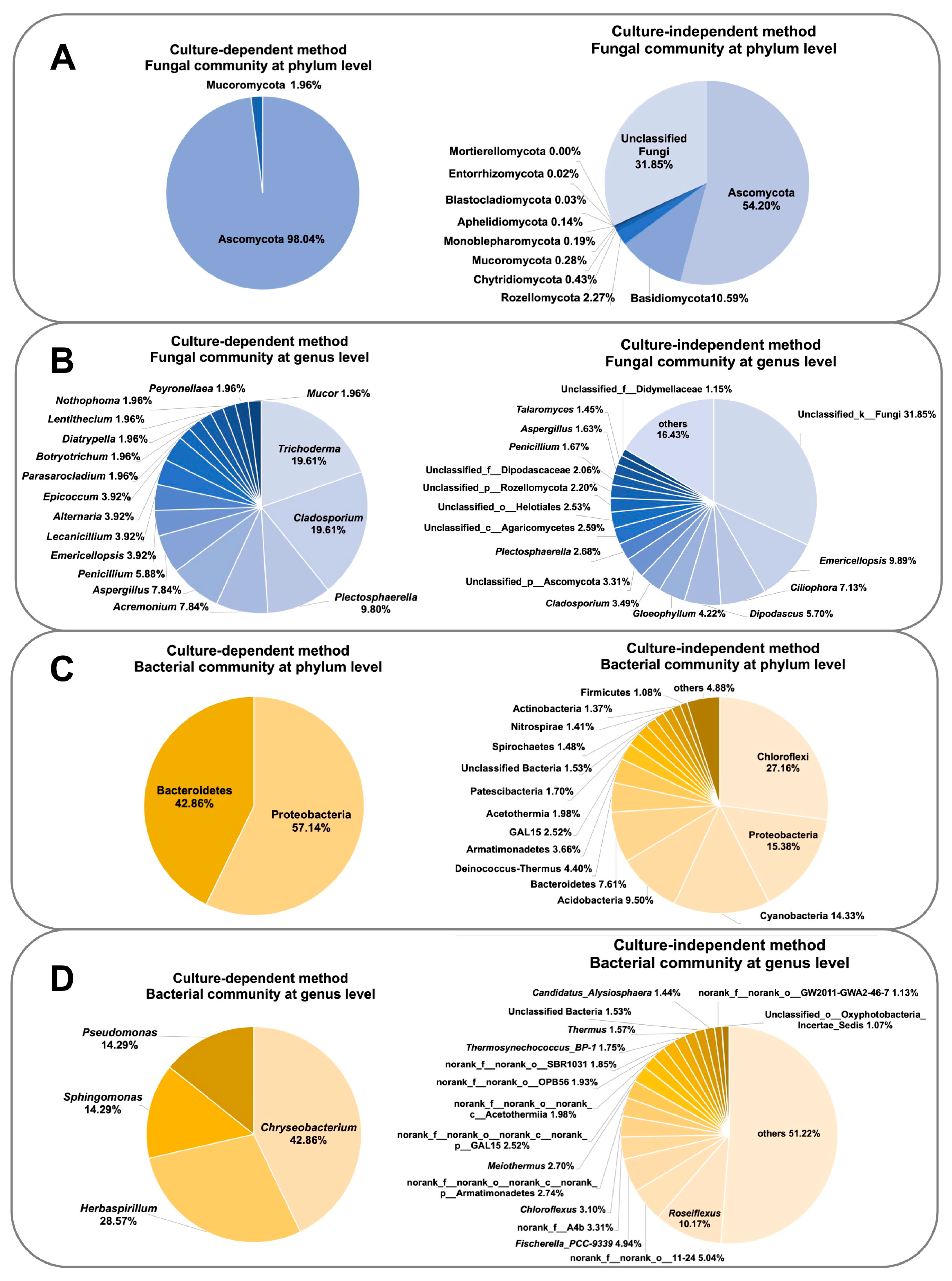
3.5. Comparison of Culture-Dependent and Culture-Independent Microbial Diversity Analyses
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Rawat, N.; Joshi, G.K. Bacterial community structure analysis of a hot spring soil by next generation sequencing of ribosomal RNA. Genomics 2018, 111, 1053–1058. [Google Scholar] [CrossRef] [PubMed]
- Weber, A.P.M.; Horst, R.J.; Barbier, G.G.; Oesterhelt, C. Metabolism and metabolomics of eukaryotes living under extreme conditions. Int. Rev. Cytol. 2007, 256, 1–34. [Google Scholar]
- Cowan, D.A.; Ramond, J.B.; Makhalanyane, T.P.; Maayer, P.D. Metagenomics of extreme environments. Curr. Opin. Microbiol. 2015, 25, 97–102. [Google Scholar] [CrossRef] [Green Version]
- Panda, A.K.; Bisht, S.S.; Rana, M.; Mandal, S.D.; Kumar, N.S. New and Future Developments in Microbial Biotechnology and Bioengineering; Elsevier: Amsterdam, The Netherlands, 2018; pp. 155–164. ISBN 978-0-444-63994-3. [Google Scholar]
- Ding, X.; Yang, W.J.; Min, H.; Peng, X.T.; Zhou, H.Y.; Lu, Z.M. Isolation and characterization of a new strain of Methanothermobacter marburgensis DX01 from hot springs in China. Anaerobe 2010, 16, 54–59. [Google Scholar] [CrossRef] [PubMed]
- Mirete, S.; Morgante, V.; González-Pastor, J.E. Functional metagenomics of extreme environments. Curr. Opin. Biotechnol. 2016, 38, 143–149. [Google Scholar] [CrossRef] [PubMed]
- Mueller, R.C.; Peach, J.T.; Skorupa, D.J.; Copié, V.; Bothner, B.; Peyton, B.M. An emerging view of the diversity, ecology and function of Archaea in alkaline hydrothermal environments. FEMS Microbiol. Ecol. 2021, 97, fiaa246. [Google Scholar] [CrossRef]
- Mahajan, G.B.; Balachandran, L. Sources of antibiotics: Hot springs. Biochem. Pharmacol. 2016, 134, 35–41. [Google Scholar] [CrossRef] [PubMed]
- Brazelton, W.J.; Schrenk, M.O.; Kelley, D.S.; Baross, J.A. Methane- and Sulfur-Metabolizing Microbial Communities Dominate the Lost City Hydrothermal Field Ecosystem. Appl. Environ. Microbiol. 2006, 72, 6257–6270. [Google Scholar] [CrossRef] [Green Version]
- Najar, I.N.; Sherpa, M.T.; Das, S.; Das, S.; Thakur, N. Microbial ecology of two hot springs of Sikkim: Predominate population and geochemistry. Sci. Total Environ. 2018, 637–638, 730–745. [Google Scholar] [CrossRef]
- Blank, C.E.; Cady, S.L.; Pace, N.R. Microbial Composition of Near-Boiling Silica-Depositing Thermal Springs throughout Yellowstone National Park. Appl. Environ. Microbiol. 2002, 68, 5123–5135. [Google Scholar] [CrossRef] [Green Version]
- Wang, S.; Hou, W.G.; Dong, H.L.; Jiang, H.C.; Huang, L.Q.; Wu, G.; Zhang, C.L.; Song, Z.Q.; Zhang, Y.; Ren, H.L.; et al. Control of Temperature on Microbial Community Structure in Hot Springs of the Tibetan Plateau. PLoS ONE 2013, 8, e62901. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hobel, C.F.V.; Marteinsson, V.T.; Hreggvidsson, G.O.; Kristjansson, J.K. Investigation of the microbial ecology of intertidal hot springs by using diversity analysis of 16S rRNA and chitinase genes. Appl. Environ. Microbiol. 2005, 71, 2771–2776. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mardanov, A.V.; Gumerov, V.M.; Beletsky, A.V.; Ravin, N.V. Microbial diversity in acidic thermal pools in the Uzon Caldera, Kamchatka. Antonie Van Leeuwenhoek 2018, 111, 35–43. [Google Scholar] [CrossRef] [PubMed]
- Kambura, A.K.; Mwirichia, R.K.; Kasili, R.W.; Karanja, E.N.; Makonde, H.M.; Boga, H.I. Diversity of fungi in sediments and water sampled from the hot springs of Lake Magadi and Little Magadi in Kenya. Afr. J. Microbiol. Res. 2016, 10, 330–338. [Google Scholar]
- Baker, G.C.; Gaffar, S.; Cowan, D.A.; Suharto, A.R. Bacterial community analysis of Indonesian hot springs. FEMS Microbiol. Lett. 2001, 200, 103–109. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Salam, N.; Jiao, J.Y.; Jiang, H.C.; Zhou, E.M.; Yin, Y.R.; Ming, H.; Li, W.J. Diversity of culturable thermophilic actinobacteria in hot springs in Tengchong, China and studies of their biosynthetic gene profiles. Microb. Ecol. 2016, 72, 150–162. [Google Scholar] [CrossRef]
- Narsing Rao, M.P.; Dong, Z.Y.; Luo, Z.H.; Li, M.M.; Liu, B.B.; Guo, S.X.; Hozzein, W.N.; Xiao, M.; Li, W.J. Physicochemical and Microbial Diversity Analyses of Indian Hot Springs. Front. Microbiol. 2021, 12, 627200. [Google Scholar] [CrossRef] [PubMed]
- Singh, N.; Puri, M.; Tuli, D.K.; Gupta, R.P.; Barrow, C.J.; Mathur, A.S. Bioethanol production potential of a novel thermophilic isolate Thermoanaerobacter sp. DBT-IOC-X2 isolated from Chumathang hot spring. Biomass Bioenergy 2018, 116, 122–130. [Google Scholar] [CrossRef]
- Adams, M.W.W.; Kelly, R.M. Finding and using hyperthermophilic enzymes. Trends Biotechnol. 1998, 16, 329–332. [Google Scholar] [CrossRef]
- Tang, J.; Jiang, D.; Luo, Y.F.; Liang, Y.M.; Li, L.H.; Shah, M.M.R.; Daroch, M. Potential new genera of cyanobacterial strains isolated from thermal springs of western Sichuan, China. Algal Res. 2018, 31, 14–20. [Google Scholar] [CrossRef]
- Nakagawa, T.; Fukui, M. Phylogenetic characterization of microbial mats and streamers from a Japanese alkaline hot spring with a thermal gradient. J. Gen. Appl. Microbiol. 2002, 48, 211–222. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Purcell, D.; Sompong, U.; Yim, L.C.; Barraclough, T.G.; Peerapornpisal, Y.; Pointing, S.B. The effects of temperature, pH and sulphide on the community structure of hyperthermophilic streamers in hot springs of northern Thailand. FEMS Microbiol. Ecol. 2007, 60, 456–466. [Google Scholar] [CrossRef] [Green Version]
- Pearson, A.; Pi, Y.; Zhao, W.; Li, W.; Li, Y.; Inskeep, W.; Perevalova, A.; Romanek, C.; Li, S.; Zhang, C.L. Factors controlling the distribution of archaeal tetraethers in terrestrial hot springs. Appl. Environ. Microbiol. 2008, 74, 3523–3532. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, K.H.; Ding, X.W.; Salam, N.; Zhang, B.; Tang, X.F.; Deng, B.W.; Li, W.J. Unexpected fungal communities in the Rehai thermal springs of Tengchong influenced by abiotic factors. Extremophiles 2018, 22, 525–535. [Google Scholar] [CrossRef]
- Li, J.W.; Peng, X.T.; Zhang, L.X.; Jiang, L.; Chen, S. Linking Microbial Community Structure to S, N and Fe Biogeochemical Cycling in the Hot Springs at the Tengchong Geothermal Fields, Southwest China. Geomicrobiol. J. 2015, 33, 135–150. [Google Scholar] [CrossRef]
- Hou, W.G.; Wang, S.; Dong, H.L.; Jiang, H.C.; Briggs, B.R.; Peacock, J.P.; Huang, Q.Y.; Huang, L.Q.; Wu, G.; Zhi, X.Y. A Comprehensive Census of Microbial Diversity in Hot Springs of Tengchong, Yunnan Province China Using 16S rRNA Gene Pyrosequencing. PLoS ONE 2013, 8, e53350. [Google Scholar]
- Pan, W.Z.; Huang, X.W.; Wei, K.B.; Zhang, C.M.; Yang, D.M.; Ding, J.M.; Zhang, K.Q. Diversity of thermophilic fungi in Tengchong Rehai National Park revealed by ITS nucleotide sequence analyses. J. Microbiol. 2010, 48, 146–152. [Google Scholar] [CrossRef] [PubMed]
- Cai, J.G.; Wang, Y.P.; Liu, D.B.; Zeng, Y.; Xue, Y.F.; Ma, Y.H.; Feng, Y. Fervidobacterium changbaicum sp. nov., a novel thermophilic anaerobic bacterium isolated from a hot spring of the Changbai Mountains, China. Int. J. Syst. Evol. Microbiol. 2007, 57, 2333–2336. [Google Scholar] [CrossRef] [PubMed]
- Yan, B.Z.; Qiu, S.W.; Xiao, C.L.; Liang, X.J. Characteristics of Mineral Fluids and Geothermal Reservoir in Changbai Mountain, Northeast of China. Geochem. Int. 2019, 57, 83–97. [Google Scholar]
- Yan, B.Z.; Liang, X.J.; Xiao, C.L. Hydrogeochemical Characteristics and Genesis Model of Jinjiang and Julong Hot Springs in Changbai Mountain, Northeast China. Geofluids 2018, 2018, 1–16. [Google Scholar] [CrossRef]
- Gao, Q.W. Volcanic Hytrothermal Activities and Gas-releasing characteristics of the Tianchi Lake Region, Changbai Mountains. Acta Geosci. Sin. 2004, 25, 345–350. [Google Scholar]
- Liu, J.Q.; Chen, S.S.; Guo, Z.F.; Guo, W.F.; He, H.Y.; You, H.T.; Kim, H.M.; Sung, G.H.; Kim, H. Geological background and geodynamic mechanism of Mt. Changbai volcanoes on the China-Korea border. Lithos 2015, 236–237, 46–73. [Google Scholar] [CrossRef]
- Yan, B.Z.; Qiu, S.W.; Liu, Z.; Changlai, X. Characteristics of the geothermal water in Changbai Mountain volcanic region, northeast of China. Arab. J. Geosci. 2017, 10, 261. [Google Scholar] [CrossRef]
- Boudreau, A.E.; Lynne, B.Y. The growth of siliceous sinter deposits around high-temperature eruptive hot springs. J. Volcanol. Geotherm. Res. 2012, 247–248, 1–8. [Google Scholar] [CrossRef]
- Bing, W.; Wang, H.; Zheng, B.; Zhang, F.; Zhu, G.; Feng, Y.; Zhang, Z. Caldicellulosiruptor changbaiensis sp. nov., a cellulolytic and hydrogen-producing bacterium from a hot spring. Int. J. Syst. Evol. Microbiol. 2015, 65, 293–297. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.L.; Guo, Z.F.; Liu, J.Q.; Liu, G.M.; Zhang, L.H.; Lei, M.; Zhao, W.B.; Ma, L.; Sepe, V.; Ventura, G. The intraplate Changbaishan volcanic field (China/North Korea): A review on eruptive history, magma genesis, geodynamic significance, recent dynamics and potential hazards. Earth-Sci. Rev. 2018, 187, 19–52. [Google Scholar] [CrossRef]
- Wang, X.; Pecoraro, L. Analysis of Soil Fungal and Bacterial Communities in Tianchi Volcano Crater, Northeast China. Life 2021, 11, 280. [Google Scholar] [CrossRef] [PubMed]
- Gilman, J.C. A Manual of Soil Fungi; The Iowa State University Press: Ames, IA, USA, 1957; pp. 1–450. ISBN 978-70-0001-234-5. [Google Scholar]
- Alexopoulos, C.J.; Mims, C.W. Introductory Mycology, 3rd ed.; John Wiley and Sons: New York, NY, USA, 1979; pp. 1–632. ISBN 978-71-2456-557-2. [Google Scholar]
- Hall, T. Bioedit: A user-friendly biological sequence alignment editor and analysis program for windows 95/98/NT. Nucleic Acids Symp. Ser. 1999, 41, 95–98. [Google Scholar]
- Chen, S.; Zhou, Y.; Chen, Y.; Gu, J. fastp: An ultra-fast all-in-one FASTQ preprocessor. Bioinformatics 2018, 34, i884–i890. [Google Scholar] [CrossRef]
- Magoč, T.; Salzberg, S.L. FLASH: Fast length adjustment of short reads to improve genome assemblies. Bioinformatics 2011, 27, 2957–2963. [Google Scholar] [CrossRef]
- Edgar, R.C.; Haas, B.J.; Clemente, J.C.; Christopher, Q.; Rob, K. UCHIME improves sensitivity and speed of chimera detection. Bioinformatics 2011, 27, 2194–2200. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Edgar, R.C. UPARSE: Highly accurate OTU sequences from microbial amplicon reads. Nat. Methods 2013, 10, 96–998. [Google Scholar] [CrossRef]
- Cole, J.R.; Wang, Q.; Fish, J.A.; Chai, B.; McGarrell, D.M.; Sun, Y.; Brown, C.T.; Porras-Alfaro, A.; Kuske, C.R.; Tiedje, J.M. Ribosomal database project: Data and tools for high throughput rRNA analysis. Nucleic Acids Res. 2014, 42, D633–D642. [Google Scholar] [CrossRef] [Green Version]
- Lozupone, C.; Knight, R. UniFrac: A new phylogenetic method for comparing microbial communities. Appl. Environ. Microbiol. 2005, 71, 8228–8235. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bastian, M.; Heymann, S.; Jacomy, M. Gephi: An Open Source Software for Exploring and Manipulating Networks. In Proceedings of the Third International Conference on Weblogs and Social Media (ICWSM 2009), San Jose, CA, USA, 17–20 May 2009; pp. 361–362. [Google Scholar]
- Poddar, A.; Das, S.K. Microbiological studies of hot springs in India: A review. Arch. Microbiol. 2018, 200, 1–18. [Google Scholar] [CrossRef]
- Sahoo, R.K.; Subudhi, E.; Kumar, M. Investigation of bacterial diversity of hot springs of Odisha, India. Genom. Data 2015, 6, 188–190. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zimik, H.V.; Farooq, S.H.; Prusty, P. Geochemical evaluation of thermal springs in Odisha, India. Environ. Earth Sci. 2017, 76, 593. [Google Scholar] [CrossRef]
- Santoyo, G. Unveiling Taxonomic Diversity and Functional Composition Differences of Microbial Mat Communities Through Comparative Metagenomics. Geomicrobiol. J. 2021, 39, 639–648. [Google Scholar] [CrossRef]
- Bennett, A.C.; Murugapiran, S.K.; Hamilton, T.L. Temperature impacts community structure and function of phototrophic Chloroflexi and Cyanobacteria in two alkaline hot springs in Yellowstone National Park. Environ. Microbiol. Rep. 2020, 12, 503–513. [Google Scholar] [CrossRef] [PubMed]
- Bian, X.M.; Guo, J.J.; Zhao, Y.; Li, C.L.; Li, W. Four new Chinese fungal records from marine sediments. Mycosystema 2016, 35, 1273–1279. [Google Scholar]
- Marchese, P.; Garzoli, L.; Young, R.; Allcock, L.; Barry, F.; Tuohy, M.; Murphy, M. Fungi populate deep-sea coral gardens as well as marine sediments in the Irish Atlantic Ocean. Environ. Microbiol. 2021, 23, 4168–4184. [Google Scholar] [CrossRef]
- Cecchi, G.; Cutroneo, L.; Piazza, S.D.; Capello, M.; Zotti, M. Culturable fungi from dredged and marine sediments from six ports studied in the framework of the SEDITERRA Project. J. Soils Sediments 2021, 21, 1563–1573. [Google Scholar] [CrossRef]
- Khusnullina, A.I.; Bilanenko, E.N.; Kurakov, A.V. Microscopic Fungi of White Sea Sediments. Contemp. Probl. Ecol. 2018, 11, 503–513. [Google Scholar] [CrossRef]
- Oppong-Danquah, E.; Parrot, D.; Blümel, M.; Labes, A.; Tasdemir, D. Molecular Networking-Based Metabolome and Bioactivity Analyses of Marine-Adapted Fungi Co-cultivated with Phytopathogens. Front. Microbiol. 2018, 9, 2072. [Google Scholar] [CrossRef]
- Oppong-Danquah, E.; Passaretti, C.; Chianese, O.; Blümel, M.; Tasdemir, D. Mining the Metabolome and the Agricultural and Pharmaceutical Potential of Sea Foam-Derived Fungi. Mar. Drugs 2020, 18, 128. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Agrawala, S.; Barrowa, C.J.; Deshmukh, S.K. Structural deformation in pathogenic bacteria cells caused by marine fungal metabolites: An in vitro investigation. Microb. Pathog. 2020, 146, 104248. [Google Scholar] [CrossRef] [PubMed]
- Inostroza, A.; Lara, L.; Paz, C.; Perez, A.; Galleguillos, F.; Hernandez, V.; Becerra, J.; González-Rocha, G.; Silva, M. Antibiotic activity of Emerimicin IV isolated from Talcahuano Bay, Chile. Nat. Prod. Res. 2018, 32, 1361–1364. [Google Scholar] [CrossRef]
- Pinheiroa, Â.; Dethoupab, T.; Bessaa, J.; Silvac, A.M.S.; Kijjoa, A. A new bicyclic sesquiterpene from the marine sponge associated fungus Emericellopsis minima. Phytochem. Lett. 2012, 5, 68–70. [Google Scholar] [CrossRef]
- Elíades, L.A.; Cabello, M.N.; Voget, C.E. Contribution to the study of alkalophilic and alkali-tolerant Ascomycota from Argentina. Darwiniana 2006, 44, 64–73. [Google Scholar]
- Klatt, C.G.; Bryant, D.A.; Ward, D.M. Comparative genomics provides evidence for the 3-hydroxypropionate autotrophic pathway in filamentous anoxygenic phototrophic bacteria and in hot spring microbial mats. Environ. Microbiol. 2007, 9, 2067–2078. [Google Scholar] [CrossRef] [PubMed]
- Alcorta, J.; Vergara-Barros, P.; Antonaru, L.A.; Alcamán-Arias, M.E.; Nürnberg, D.J.; Díez, B. Fischerella thermalis: A model organism to study thermophilic diazotrophy, photosynthesis and multicellularity in cyanobacteria. Extremophiles 2019, 23, 635–647. [Google Scholar] [CrossRef]
- Nishida, A.; Thiel, V.; Nakagawa, M.; Ayukawa, S.; Yamamura, M. Effect of light wavelength on hot spring microbial mat biodiversity. PLoS ONE 2018, 13, e0191650. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sasaki, K.; Uejima, Y.; Sakamoto, A.; Yu, Q.Q.; Ishibashi, J.; Okibe, N.; Hirajima, T. Geochemical and Microbiological Analysis of Sambe Hot Springs, Shimane Prefecture, Japan. Resour. Geol. 2013, 63, 155–165. [Google Scholar] [CrossRef]
- Okibe, N.; Maki, M.; Sasaki, K.; Hirajima, T. Mn(II)-Oxidizing Activity of Pseudomonas sp. Strain MM1 is Involved in the Formation of Massive Mn Sediments around Sambe Hot Springs in Japan. Mater. Trans. 2013, 54, 2027–2031. [Google Scholar] [CrossRef] [Green Version]
- Francis, C.; Co, E.M.; Tebo, B.R. Enzymatic manganese(II) oxidation by a marine α-proteobacterium. Appl. Environ. Microbiol. 2001, 67, 4024–4029. [Google Scholar] [CrossRef] [Green Version]
- Prondzinsky, P.; Berkemer, S.J.; Ward, L.M.; McGlynn, S.E. The Thermosynechococcus Genus: Wide Environmental Distribution, but a Highly Conserved Genomic Core. Microbes Environ. 2021, 36, ME20138. [Google Scholar] [CrossRef] [PubMed]
- Power, J.F.; Carere, C.R.; Lee, C.K.; Wakerley, G.L.J.; Evans, D.W.; Mathew, B.; Duncan, W.; Climo, M.D.; Hinze, A.M.; Morgan, X.C. Microbial biogeography of 925 geothermal springs in New Zealand. Nat. Commun. 2018, 9, 2876. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Uribe-Lorío, L.; Brenes-Guillén, L.; Hernández-Ascencio, W.; Mora-Amador, R.; González, G.; Ramírez-Umaña, C.J.; Díez, B.; Pedrós-Alió, C. The influence of temperature and pH on bacterial community composition of microbial mats in hot springs from Costa Rica. MicrobiologyOpen 2019, 8, e893. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- López-López, O.; Knapik, K.; Cerdán, M.E.; González-Siso, M.I. Metagenomics of an Alkaline Hot Spring in Galicia (Spain): Microbial Diversity Analysis and Screening for Novel Lipolytic Enzymes. Front. Microbiol. 2015, 6, 1291. [Google Scholar] [CrossRef] [Green Version]
- Brasen, C.; Esser, D.; Rauch, B.; Siebers, B. Carbohydrate Metabolism in Archaea: Current Insights into Unusual Enzymes and Pathways and Their Regulation. Microbiol. Mol. Biol. Rev. 2014, 78, 89–175. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Brock, T.D. Thermophilic Microorganisms and Life at High Temperatures. BioScience 1979, 29, 691. [Google Scholar]
- Straub, C.T.; Counts, J.A.; Nguyen, D.M.N.; Wu, C.H.; Zeldes, B.M.; Crosby, J.R.; Conway, J.M.; Otten, J.K.; Lipscomb, G.L.; Schut, G.J. Biotechnology of extremely thermophilic archaea. FEMS Microbiol. Rev. 2018, 42, 543–578. [Google Scholar] [CrossRef] [PubMed] [Green Version]

| Community | Phylum | Class | Order | Genus | Number of Species | Number of Strains |
|---|---|---|---|---|---|---|
| Fungi | Ascomycota | Sordariomycetes | Hypocreales | Acremonium | 2 | 4 |
| Emericellopsis | 1 | 2 | ||||
| Lecanicillium | 1 | 2 | ||||
| Parasarocladium | 1 | 1 | ||||
| Trichoderma | 7 | 10 | ||||
| Glomerellales | Plectosphaerella | 1 | 5 | |||
| Sordariales | Botryotrichum | 1 | 1 | |||
| Xylariales | Diatrypella | 1 | 1 | |||
| Dothideomycetes | Pleosporales | Alternaria | 2 | 2 | ||
| Epicoccum | 1 | 2 | ||||
| Lentithecium | 1 | 1 | ||||
| Nothophoma | 1 | 1 | ||||
| Peyronellaea | 1 | 1 | ||||
| Capnodiales | Cladosporium | 4 | 10 | |||
| Eurotiomycetes | Eurotiales | Aspergillus | 3 | 4 | ||
| Penicillium | 2 | 3 | ||||
| Mucoromycota | Mucoromycetes | Mucorales | Mucor | 1 | 1 | |
| Bacteria | Bacteroidetes | Flavobacteriia | Flavobacteriales | Chryseobacterium | 2 | 3 |
| Proteobacteria | Alphaproteobacteria | Sphingomonadales | Sphingomonas | 1 | 1 | |
| Betaproteobacteria | Burkholderiales | Herbaspirillum | 2 | 2 | ||
| Gammaproteobacteria | Pseudomonadales | Pseudomonas | 1 | 1 |
| Community | Phylum | Class | Order | Genus | Number of Species | Number of Strains |
|---|---|---|---|---|---|---|
| Fungi | Ascomycota | Dothideomycetes | Pleosporales | Alternaria | 3 | 3 |
| Dothideales | Aureobasidium | 1 | 1 | |||
| Botryosphaeriales | Neofusicoccum | 1 | 1 | |||
| Eurotiomycetes | Eurotiales | Aspergillus | 2 | 6 | ||
| Basidiomycota | Tremellomycetes | Tremellales | Cryptococcus | 1 | 2 | |
| Microbotryomycetes | Sporidiobolales | Sporobolomyces | 1 | 1 | ||
| Bacteria | Firmicutes | Bacilli | Bacillales | Bacillus | 1 | 1 |
| Proteobacteria | Gammaproteobacteria | Enterobacterales | Enterobacter | 1 | 1 | |
| Pseudomonadales | Acinetobacter | 1 | 1 | |||
| Pseudomonas | 8 | 11 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, X.; Pecoraro, L. Diversity and Co-Occurrence Patterns of Fungal and Bacterial Communities from Alkaline Sediments and Water of Julong High-Altitude Hot Springs at Tianchi Volcano, Northeast China. Biology 2021, 10, 894. https://doi.org/10.3390/biology10090894
Wang X, Pecoraro L. Diversity and Co-Occurrence Patterns of Fungal and Bacterial Communities from Alkaline Sediments and Water of Julong High-Altitude Hot Springs at Tianchi Volcano, Northeast China. Biology. 2021; 10(9):894. https://doi.org/10.3390/biology10090894
Chicago/Turabian StyleWang, Xiao, and Lorenzo Pecoraro. 2021. "Diversity and Co-Occurrence Patterns of Fungal and Bacterial Communities from Alkaline Sediments and Water of Julong High-Altitude Hot Springs at Tianchi Volcano, Northeast China" Biology 10, no. 9: 894. https://doi.org/10.3390/biology10090894
APA StyleWang, X., & Pecoraro, L. (2021). Diversity and Co-Occurrence Patterns of Fungal and Bacterial Communities from Alkaline Sediments and Water of Julong High-Altitude Hot Springs at Tianchi Volcano, Northeast China. Biology, 10(9), 894. https://doi.org/10.3390/biology10090894







