The Epistemic Revolution Induced by Microbiome Studies: An Interdisciplinary View
Simple Summary
Abstract
1. Introduction
2. A Cross-Disciplinary Perspective
3. Commonalities across Novel Avenues of Research
4. Towards the Future: Some Outstanding Questions
- Will the original Koch postulate be extended even further than in medical research to demonstrate causal effects of dysbiotic microbial communities, not only on their hosts’ health, but also on the sustainability of their environment, in particular the microbiome impact on elemental biogeochemical cycles and on planetary boundaries?
- Will the extent of the regulation of host gene expression by microbiomes be sufficiently well deciphered to give rise to a ‘microbiomo-genetics’, a genetics of collectives inclusive of both host and microbiome genetic interactions?
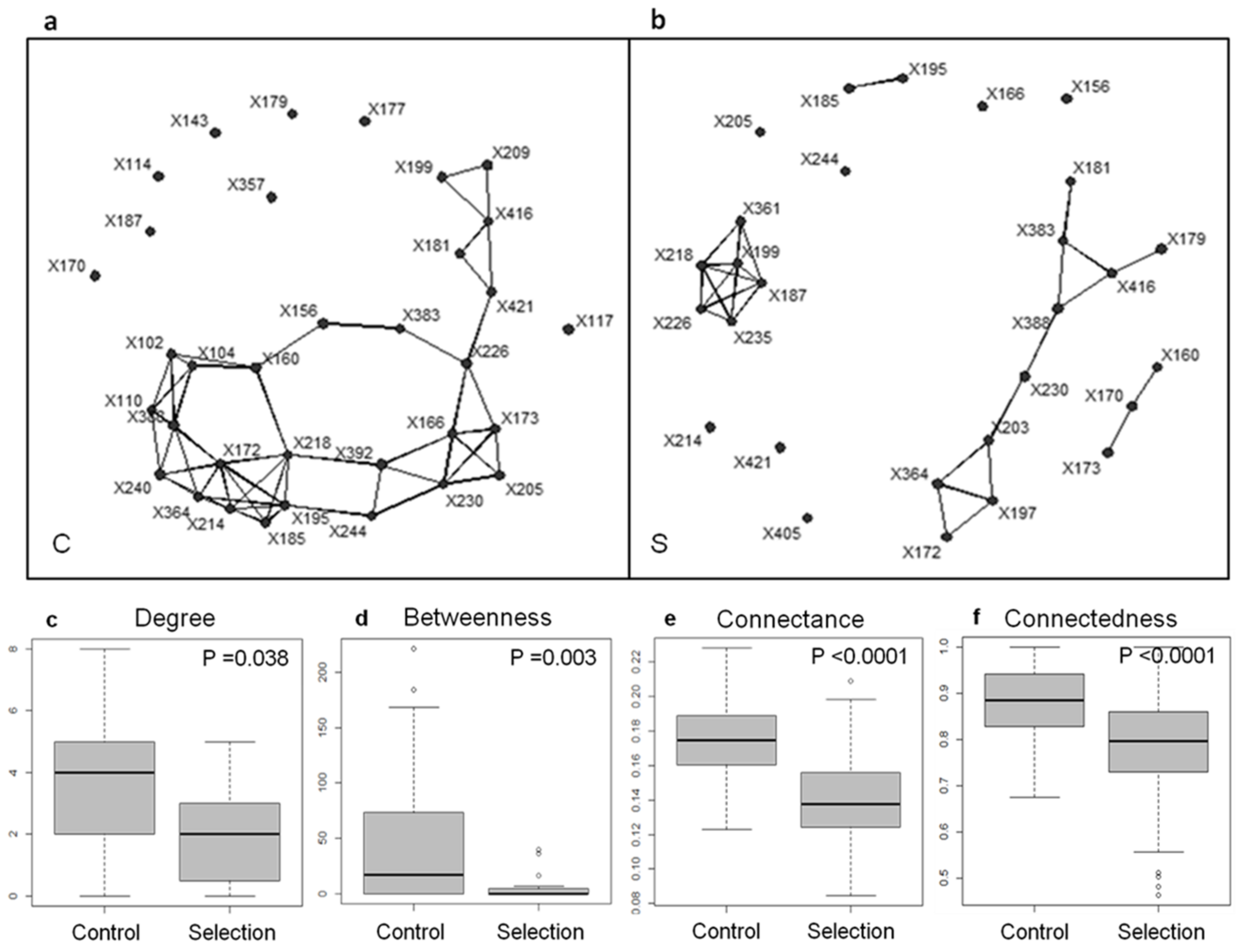
- Is clade selection prevalent in the microbial world? Whereas better dispersal abilities, or better cellular rejuvenation abilities are expected to favor microbial lineages over others by mere persistence, the ability to engage in productive interspecific interaction (e.g., to laterally exchange genes) constitutes another way through which microbial lineages may enhance their fitness. Therefore, might the persistence of microbiomes also rely on an expanded kind of clade selection, involving members of multiple phylogenetic groups, a form of ‘symbiosis selection’?
- Is it possible to ‘Darwinize microbiomes’, in the same way Gaia was Darwinized, and to identify sets of interacting microbes selected for their collective persistence? Consistently, is the stability of microbial community structure a necessary condition to convey heritable variations?
- Will we be able to delineate and count microbiomes (within an environment, within a host, etc.)? Notably, many biological explanations depend on the ability to identify and count individual members of a given population. For this reason, the terms “individual”, “member of a population”, and “organism” are often used interchangeably. This approach assumes (often wrongly) that there is some sort of homogeneity within and between individuals. It is assumed that biological individuals are constituted of parts that share a common genetic and developmental history and it is often taken for granted that populations are necessarily constituted of these related homogeneous individuals. For most metazoans this assumption may not be a major concern. However, microbiome research highlights that natural entities (individuals or collections of individuals) are often constituted of unrelated heterogeneous individuals functioning as wholes. It is an ever-more accepted fact that unrelated micro-organisms develop complex functioning ecologies with emergent properties or adaptations. This raises fundamental questions about how to identify these collectives, recognize their emergent wholeness, and deal with the fact that they may not always display the fixed boundaries of larger single genome biological individuals. The absence of determinate and fixed boundaries must not deter us from explaining their functional reality and emergent wholeness. The transiency of the boundaries of these systems does not alter the fact that they exist as genuine emergent individual and that we can count them and describe their properties. This being considered, one may wonder how many different microbiomes exist out there, transiently or more permanently?
- Will we be able to determine which human traits (if any) escape microbiome influence?
- In order to explain any destabilized biological phenomenon (e.g., to explain host health issues, tipping ecosystems or altered biogeochemical cycles), is it a priori sensible or a microbiome-centric bias to consider changes in microbiome communities, rather than macroscopic biological influences, as critical?
- Will microbiome-based signatures on crime scenes prove to be as effective proxies for non-human species as for human species?
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Cordovez, V.; Dini-Andreote, F.; Carrión, V.J.; Raaijmakers, J.M. Ecology and Evolution of Plant Microbiomes. Annu. Rev. Microbiol. 2019, 73, 69–88. [Google Scholar] [CrossRef]
- Mendes, R.; Raaijmakers, J.M. Cross-Kingdom Similarities in Microbiome Functions. ISME J. 2015, 9, 1905–1907. [Google Scholar] [CrossRef]
- Rosenberg, E.; Zilber-Rosenberg, I. The Hologenome Concept of Evolution after 10 Years. Microbiome 2018, 6, 78. [Google Scholar] [CrossRef]
- Berg, G.; Rybakova, D.; Fischer, D.; Cernava, T.; Vergès, M.-C.C.; Charles, T.; Chen, X.; Cocolin, L.; Eversole, K.; Corral, G.H.; et al. Microbiome Definition Re-Visited: Old Concepts and New Challenges. Microbiome 2020, 8, 103. [Google Scholar] [CrossRef]
- Blaser, M.J.; Cardon, Z.G.; Cho, M.K.; Dangl, J.L.; Donohue, T.J.; Green, J.L.; Knight, R.; Maxon, M.E.; Northen, T.R.; Pollard, K.S.; et al. Toward a Predictive Understanding of Earth’s Microbiomes to Address 21st Century Challenges. mBio 2016, 7. [Google Scholar] [CrossRef]
- Cavicchioli, R.; Ripple, W.J.; Timmis, K.N.; Azam, F.; Bakken, L.R.; Baylis, M.; Behrenfeld, M.J.; Boetius, A.; Boyd, P.W.; Classen, A.T.; et al. Scientists’ Warning to Humanity: Microorganisms and Climate Change. Nat. Rev. Microbiol. 2019, 17, 569–586. [Google Scholar] [CrossRef] [PubMed]
- Roux, S.; Brum, J.R.; Dutilh, B.E.; Sunagawa, S.; Duhaime, M.B.; Loy, A.; Poulos, B.T.; Solonenko, N.; Lara, E.; Poulain, J.; et al. Ecogenomics and Potential Biogeochemical Impacts of Globally Abundant Ocean Viruses. Nature 2016, 537, 689–693. [Google Scholar] [CrossRef] [PubMed]
- Dutreuil, S. James Lovelock’s Gaia hypothesis: “A New Look at Life on Earth” … for the Life and the Earth sciences. In Dreamers, Visionaries, and Revolutionaries in the Life Sciences; Chicago University Press: Chicago, IL, USA, 2018; pp. 272–287. [Google Scholar]
- Lenton, T.M.; Dutreuil, S.; Latour, B. Life on Earth Is Hard to Spot. Anthr. Rev. 2020, 7, 248–272. [Google Scholar] [CrossRef]
- Sunagawa, S.; Coelho, L.P.; Chaffron, S.; Kultima, J.R.; Labadie, K.; Salazar, G.; Djahanschiri, B.; Zeller, G.; Mende, D.R.; Alberti, A.; et al. Structure and Function of the Global Ocean Microbiome. Science 2015, 348. [Google Scholar] [CrossRef]
- Dittami, S.M.; Arboleda, E.; Auguet, J.-C.; Bigalke, A.; Briand, E.; Cárdenas, P.; Cardini, U.; Decelle, J.; Engelen, A.H.; Eveillard, D.; et al. A Community Perspective on the Concept of Marine Holobionts: Current Status, Challenges, and Future Directions. PeerJ 2021, 9, e10911. [Google Scholar] [CrossRef]
- Troussellier, M.; Escalas, A.; Bouvier, T.; Mouillot, D. Sustaining Rare Marine Microorganisms: Macroorganisms As Repositories and Dispersal Agents of Microbial Diversity. Front. Microbiol. 2017, 8. [Google Scholar] [CrossRef]
- Guidi, L.; Chaffron, S.; Bittner, L.; Eveillard, D.; Larhlimi, A.; Roux, S.; Darzi, Y.; Audic, S.; Berline, L.; Brum, J.; et al. Plankton Networks Driving Carbon Export in the Oligotrophic Ocean. Nature 2016, 532, 465–470. [Google Scholar] [CrossRef]
- Bergk Pinto, B.; Maccario, L.; Dommergue, A.; Vogel, T.M.; Larose, C. Do Organic Substrates Drive Microbial Community Interactions in Arctic Snow? Front. Microbiol. 2019, 10. [Google Scholar] [CrossRef]
- Zhu, C.; Miller, M.; Lusskin, N.; Bergk Pinto, B.; Maccario, L.; Häggblom, M.; Vogel, T.; Larose, C.; Bromberg, Y. Snow Microbiome Functional Analyses Reveal Novel Aspects of Microbial Metabolism of Complex Organic Compounds. Microbiologyopen 2020, 9, e1100. [Google Scholar] [CrossRef]
- Margesin, R.; Collins, T. Microbial Ecology of the Cryosphere (Glacial and Permafrost Habitats): Current Knowledge. Appl. Microbiol. Biotechnol. 2019, 103, 2537–2549. [Google Scholar] [CrossRef] [PubMed]
- Bapteste, E. Tous Entrelacés: Des Gènes Aux Super-Organismes: Les Réseaux de L’évolution; Belin: Paris, France, 2017. [Google Scholar]
- Barr, B.; Blackall, L.; Crocetti, G.; Reed, A.; Stock, P.; Wild, A. Nema and the Xenos: A Story of Soil Cycles; Csiro Publishing: Clayton, Australia, 2019. [Google Scholar]
- Barr, B.; Crocetti, G.; Reed, A.; Wild, A. The Squid, the Vibrio and the Moon; Csiro Publishing: Clayton, Australia, 2019. [Google Scholar]
- Barr, B.; Crocetti, G.; Reed, A.; Wild, A. Zobi and the Zoox, a Story of Coral Bleaching; Csiro Publishing: Clayton, Australia, 2018. [Google Scholar]
- Eden Project. Invisible You: The Human Microbiome. Exhibition Catalogue. 2015. Available online: https://www.edenproject.com/sites/default/files/invisible-you-catalogue.pdf (accessed on 30 April 2021).
- Enders, G. Gut: The inside Story of Our Body’s Most Underrated Organ; Scribe: London, UK, 2014. [Google Scholar]
- Margulis, L.; Sagan, D. Microcosmos: Four Billion Years of Evolution from Our Microbial Ancestors; University of California Press: Berkeley, CA, USA, 1997. [Google Scholar]
- Yong, E. I Contain Multitudes: The Microbes within Us and a Grander View of Life; Vintage: London, UK, 2016. [Google Scholar]
- Selosse, M.-A. Jamais Seul. Ces Microbes Qui Construisent Les Plantes, Les Animaux et Les Civilisations; Actes Sud: Arles, France, 2017. [Google Scholar]
- Selosse, M.-A. Les Goûts et Les Couleurs Du Monde. Une Histoire Naturelle Des Tannins, de L’écologie à La Santé; Actes Sud: Arles, France, 2019. [Google Scholar]
- Burniat, M. Sous Terre; Dargaud: Paris, France, 2021. [Google Scholar]
- Vonaesch, P.; Anderson, M.; Sansonetti, P.J. Pathogens, Microbiome and the Host: Emergence of the Ecological Koch’s Postulates. FEMS Microbiol. Rev. 2018, 42, 273–292. [Google Scholar] [CrossRef] [PubMed]
- Llopis, M.; Cassard-Doulcier, A.-M.; Wrzosek, L.; Boschat, L.; Bruneau, A.; Ferrere, G.; Puchois, V.; Martin, J.-C.; Lepage, P.; Le Roy, T.; et al. Intestinal Microbiota Contributes to Individual Susceptibility to Alcoholic Liver Disease. Gut 2015, 65. [Google Scholar] [CrossRef] [PubMed]
- Le Roy, T.; Lécuyer, E.; Chassaing, B.; Rhimi, M.; Lhomme, M.; Boudebbouze, S.; Ichou, F.; Haro Barceló, J.; Huby, T.; Guerin, M.; et al. The Intestinal Microbiota Regulates Host Cholesterol Homeostasis. BMC Biol. 2019, 17, 94. [Google Scholar] [CrossRef]
- Javan, G.T.; Finley, S.J.; Can, I.; Wilkinson, J.E.; Hanson, J.D.; Tarone, A.M. Human Thanatomicrobiome Succession and Time Since Death. Sci. Rep. 2016, 6, 29598. [Google Scholar] [CrossRef]
- Javan, G.T.; Finley, S.J.; Abidin, Z.; Mulle, J.G. The Thanatomicrobiome: A Missing Piece of the Microbial Puzzle of Death. Front. Microbiol. 2016, 7, 225. [Google Scholar] [CrossRef]
- Can, I.; Javan, G.T.; Pozhitkov, A.E.; Noble, P.A. Distinctive Thanatomicrobiome Signatures Found in the Blood and Internal Organs of Humans. J. Microbiol. Methods 2014, 106, 1–7. [Google Scholar] [CrossRef]
- Franzosa, E.A.; Huang, K.; Meadow, J.F.; Gevers, D.; Lemon, K.P.; Bohannan, B.J.M.; Huttenhower, C. Identifying Personal Microbiomes Using Metagenomic Codes. Proc. Natl. Acad. Sci. USA 2015, 112, E2930–E2938. [Google Scholar] [CrossRef] [PubMed]
- Hanssen, E.N.; Avershina, E.; Rudi, K.; Gill, P.; Snipen, L. Body Fluid Prediction from Microbial Patterns for Forensic Application. Forensic Sci. Int. Genet. 2017, 30, 10–17. [Google Scholar] [CrossRef] [PubMed]
- Walker, A.R.; Grimes, T.L.; Datta, S.; Datta, S. Unraveling Bacterial Fingerprints of City Subways from Microbiome 16S Gene Profiles. Biol. Direct. 2018, 13, 10. [Google Scholar] [CrossRef]
- Schmedes, S.E.; Woerner, A.E.; Novroski, N.M.M.; Wendt, F.R.; King, J.L.; Stephens, K.M.; Budowle, B. Targeted Sequencing of Clade-Specific Markers from Skin Microbiomes for Forensic Human Identification. Forensic Sci. Int. Genet. 2018, 32, 50–61. [Google Scholar] [CrossRef] [PubMed]
- Adserias-Garriga, J.; Quijada, N.M.; Hernandez, M.; Rodríguez Lázaro, D.; Steadman, D.; Garcia-Gil, L.J. Dynamics of the Oral Microbiota as a Tool to Estimate Time since Death. Mol. Oral Microbiol. 2017, 32, 511–516. [Google Scholar] [CrossRef] [PubMed]
- Megerlin, F.; Fouassier, E.; Lopert, R.; Bourlioux, P. Microbiota Transplantation: A Sui Generis Biological Drug, Not a Tissue. Ann. Pharm. Fr. 2014, 72, 217–220. [Google Scholar] [CrossRef]
- Bocquet, F.; Peigné, J. La Transplantation Du Microbiote Fécal: Les Enjeux Juridiques et Sanitaires d’une Révolution Thérapeutique. Rev. Gén. Droit Méd. 2016, 374, 281–294. [Google Scholar]
- Aïdan, G.; Bourcier, D. Humain Non Humain, Repenser L’intériorité du Sujet de Droit; Droit et Société. Maison des Sciences de l’Homme; LGDJ: Gif-sur-Yvette, France, 2021; ISBN 978-2-275-09074-0. [Google Scholar]
- Suárez, J.; Triviño, V. What Is a Hologenomic Adaptation? Emergent Individuality and Inter-Identity in Multispecies Systems. Front. Psychol. 2020, 11, 187. [Google Scholar] [CrossRef]
- O’Malley, M.; Parke, E. Philosophy of Microbiology. In The Stanford Encyclopedia of Philosophy; Stanford University: Stanford, CA, USA, 2020; Available online: https://plato.stanford.edu/cgi-bin/encyclopedia/archinfo.cgi?entry=microbiology (accessed on 30 April 2021).
- Parke, E.C.; Calcott, B.; O’Malley, M.A. A Cautionary Note for Claims about the Microbiome’s Impact on the “Self”. PLoS Biol. 2018, 16, e2006654. [Google Scholar] [CrossRef]
- Rees, T.; Bosch, T.; Douglas, A.E. How the Microbiome Challenges Our Concept of Self. PLoS Biol. 2018, 16, e2005358. [Google Scholar] [CrossRef]
- Gayon, J. Darwinism’s Struggle for Survival. Heredity and the Hypothesis of Natural Selection; Cambridge University Press: Cambridge, UK, 1998. [Google Scholar]
- Arias-Sánchez, F.I.; Vessman, B.; Mitri, S. Artificially Selecting Microbial Communities: If We Can Breed Dogs, Why Not Microbiomes? PLoS Biol. 2019, 17, e3000356. [Google Scholar] [CrossRef] [PubMed]
- Goodnight, C.J. Experimental studies of community evolution I: The response to selection at the community level. Evolution 1990, 44, 1614–1624. [Google Scholar] [CrossRef] [PubMed]
- Goodnight, C.J. Experimental studies of community evolution II: The ecological basis of the response to community selection. Evolution 1990, 44, 1625–1636. [Google Scholar] [CrossRef]
- Swenson, W.; Arendt, J.; Wilson, D.S. Artificial Selection of Microbial Ecosystems for 3-Chloroaniline Biodegradation. Environ. Microbiol. 2000, 2, 564–571. [Google Scholar] [CrossRef]
- Swenson, W.; Wilson, D.S.; Elias, R. Artificial Ecosystem Selection. Proc. Natl. Acad. Sci. USA 2000, 97, 9110–9114. [Google Scholar] [CrossRef] [PubMed]
- Penn, A. Modelling artificial ecosystem selection: A preliminary investigation. In Advances in Artificial Life, Lecture Notes in Artificial Intelligence; Springer: Berlin/Heidelberg, Germany, 2003; pp. 659–666. [Google Scholar]
- Penn, A.; Harvey, I. The Role of Non-Genetic Change in the Heritability, Variation and Response to Selection of Artificially Selected Ecosystems. In Proceedings of the Ninth International Conference on Artificial Life, Boston, MA, USA, 12–15 September 2004; pp. 352–357. [Google Scholar]
- Doolittle, W.F. Making the Most of Clade Selection. Philos. Sci. 2017, 84, 275–295. [Google Scholar] [CrossRef]
- Doolittle, W.F. Making Evolutionary Sense of Gaia. Trends Ecol. Evol. 2019, 34, 889–894. [Google Scholar] [CrossRef]
- Doolittle, W.F. Darwinizing Gaia. J. Theor. Biol. 2017, 434, 11–19. [Google Scholar] [CrossRef]
- Bouchard, F. Ecosystem Evolution Is About Variation and Persistence, Not Populations and Reproduction. Biol. Theory 2014, 9, 382–391. [Google Scholar] [CrossRef]
- Bouchard, F. What Is a Symbiotic Superindividual and How Do You Measure Its Fitness? In From Groups to Individuals: Evolution and Emerging Individuality; Vienna Series in Theoretical Biology; MIT Press: Cambridge, MA, USA, 2013; p. 288. [Google Scholar]
- Hutter, T.; Gimbert, C.; Bouchard, F.; Lapointe, F.-J. Being Human Is a Gut Feeling. Microbiome 2015, 3, 9. [Google Scholar] [CrossRef]
- Bouchard, F. Symbiosis, Transient Biological Individuality, and Evolutionary Processes. In Everything Flows: Towards a Processual Philosophy of Biology; Oxford University Press: Oxford, UK, 2018; pp. 186–198. [Google Scholar]
- Bouchard, F. Darwinism without Populations: A More Inclusive Understanding of the “Survival of the Fittest”. Stud. Hist. Philos. Sci. Part C Stud. Hist. Philos. Biol. Biomed. Sci. 2011, 42, 106–114. [Google Scholar] [CrossRef]
- Bouchard, F. Causal Processes, Fitness, and the Differential Persistence of Lineages. Philos. Sci. 2008, 75, 560–570. [Google Scholar] [CrossRef]
- Bapteste, E.; Huneman, P. Towards a Dynamic Interaction Network of Life to Unify and Expand the Evolutionary Theory. BMC Biol. 2018, 16, 56. [Google Scholar] [CrossRef]
- Bakker, M.G.; Manter, D.K.; Sheflin, A.M.; Weir, T.L.; Vivanco, J.M. Harnessing the Rhizosphere Microbiome through Plant Breeding and Agricultural Management. Plant Soil 2012, 360, 1–13. [Google Scholar] [CrossRef]
- Clements, F.E. Plant Succession: Analysis of the Development of Vegetation. Carnegie Inst. Wash. Publ. Sci. 1916, 242, 1–512. [Google Scholar]
- Allee, W.C.; Park, O.; Emerson, A.E.; Park, T.; Schmidt, K.P. Principles of Animal Ecology; W. B. Saunders Company: Philadelphia, PA, USA, 1950. [Google Scholar]
- Huneman, P. Biological Individuals as ‘Weak Individuals’ and their Identity: Exploring a Radical Hypothesis in the Metaphysics of Science. In Biological Identity; Routledge: Abingdon-on-Thames, Oxfordshire, UK, 2020; pp. 40–62. [Google Scholar]
- Scadden, D.T. Nice Neighborhood: Emerging Concepts of the Stem Cell Niche. Cell 2014, 157, 41–50. [Google Scholar] [CrossRef] [PubMed]
- Costello, E.K.; Stagaman, K.; Dethlefsen, L.; Bohannan, B.J.M.; Relman, D.A. The Application of Ecological Theory toward an Understanding of the Human Microbiome. Science 2012, 336, 1255–1262. [Google Scholar] [CrossRef] [PubMed]
- Morar, N.; Bohannan, B.J.M. The Conceptual Ecology of the Human Microbiome. Q. Rev. Biol. 2019, 94, 149–175. [Google Scholar] [CrossRef]
- Hodgetts, T.; Grenyer, R.; Greenhough, B.; McLeod, C.; Dwyer, A.; Lorimer, J. The Microbiome and Its Publics. EMBO Rep. 2018, 19, e45786. [Google Scholar] [CrossRef] [PubMed]
- Bouchard, F.; Huneman, P. From Groups to Individuals: Evolution and Emerging Individuality; Vienna Series in Theoretical Biology; MIT Press: Cambridge, MA, USA, 2013. [Google Scholar]
- Blouin, M.; Karimi, B.; Mathieu, J.; Lerch, T.Z. Levels and Limits in Artificial Selection of Communities. Ecol. Lett. 2015, 18, 1040–1048. [Google Scholar] [CrossRef] [PubMed]
- Doolittle, W.F.; Inkpen, S.A. Processes and Patterns of Interaction as Units of Selection: An Introduction to ITSNTS Thinking. Proc. Natl. Acad. Sci. USA 2018, 115, 4006–4014. [Google Scholar] [CrossRef] [PubMed]
- Bapteste, E.; Papale, F. Modeling the Evolution of Interconnected Processes: It Is the Song and the Singers. BioEssays 2021, 43, 2000077. [Google Scholar] [CrossRef] [PubMed]
- Huneman, P. Individuality as a Theoretical Scheme. I. Formal and Material Concepts of Individuality. Biol. Theory 2014, 9, 361–373. [Google Scholar] [CrossRef]
- Huneman, P. Individuality as a Theoretical Scheme. II. About the Weak Individuality of Organisms and Ecosystems. Biol. Theory 2014, 9, 374–381. [Google Scholar] [CrossRef]
| Human Pathologies Affected by Microbiomes | Planetary Effects of Microbial Communities |
|---|---|
| Crohn’s disease, Ulcerative colitis, Obesity, Diabetes, Colon cancer, Non Alcoholic Liver Disease, Alcoholic Hepatitis, Atherosclerosis, Hypercholesterolemia, Depression | First evolution of photosynthesis, associated with the Great Oxidation Event, Contribution to the Sulfur cycle, associated with the Permo-Trias crisis, DMS production and global impact on climate |
| Entity | Law |
|---|---|
| “Nature” as a whole, granting rights to Pacha Mama | Ecuadorian constitution, chapter 7 (2008), Bolivian law (n° 071, 21 December 2010, explicitly including interacting microorganisms) |
| The Amazonian Forest | Bolivia Supreme Court 5 April 2018) |
| The Ganges and the Yamuna rivers, and the Whanganui river | Indian law (2017), and New-Zealand law (20 March 2017) |
| The Gantori and Yamunmonotri glaciers | The Gantori and Yamunmonotri glaciers: (High court of Uttarakhand, Nainital, 30 March 2017) |
| The ape Cecilia | Mendoza court, 3 November 2016 (n° XPTE.NRO.P-72.254/15) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bapteste, E.; Gérard, P.; Larose, C.; Blouin, M.; Not, F.; Campos, L.; Aïdan, G.; Selosse, M.A.; Adénis, M.S.; Bouchard, F.; et al. The Epistemic Revolution Induced by Microbiome Studies: An Interdisciplinary View. Biology 2021, 10, 651. https://doi.org/10.3390/biology10070651
Bapteste E, Gérard P, Larose C, Blouin M, Not F, Campos L, Aïdan G, Selosse MA, Adénis MS, Bouchard F, et al. The Epistemic Revolution Induced by Microbiome Studies: An Interdisciplinary View. Biology. 2021; 10(7):651. https://doi.org/10.3390/biology10070651
Chicago/Turabian StyleBapteste, Eric, Philippe Gérard, Catherine Larose, Manuel Blouin, Fabrice Not, Liliane Campos, Géraldine Aïdan, M. André Selosse, M. Sarah Adénis, Frédéric Bouchard, and et al. 2021. "The Epistemic Revolution Induced by Microbiome Studies: An Interdisciplinary View" Biology 10, no. 7: 651. https://doi.org/10.3390/biology10070651
APA StyleBapteste, E., Gérard, P., Larose, C., Blouin, M., Not, F., Campos, L., Aïdan, G., Selosse, M. A., Adénis, M. S., Bouchard, F., Dutreuil, S., Corel, E., Vigliotti, C., Huneman, P., Lapointe, F. J., & Lopez, P. (2021). The Epistemic Revolution Induced by Microbiome Studies: An Interdisciplinary View. Biology, 10(7), 651. https://doi.org/10.3390/biology10070651








