Combined Effect of Cultivar and Peel Chromaticity on Figs’ Primary and Secondary Metabolites: Preliminary Study Using Biochemical and FTIR Fingerprinting Coupled to Chemometrics
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Plant Material
2.2. Growing Conditions
2.3. FTIR Fingerprinting
2.4. Fruit Peel Color
2.5. HPLC Sugars and Organic Acids Profile
2.6. Antioxidant Activity
2.7. Total Polyphenols Analysis
2.8. Quality Assurance/Quality Control (QA/QC)
2.9. Statistical Analysis
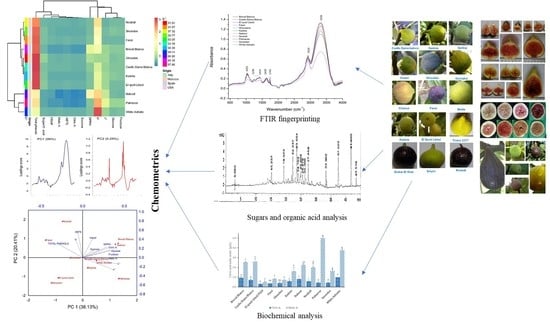
3. Results and Discussions
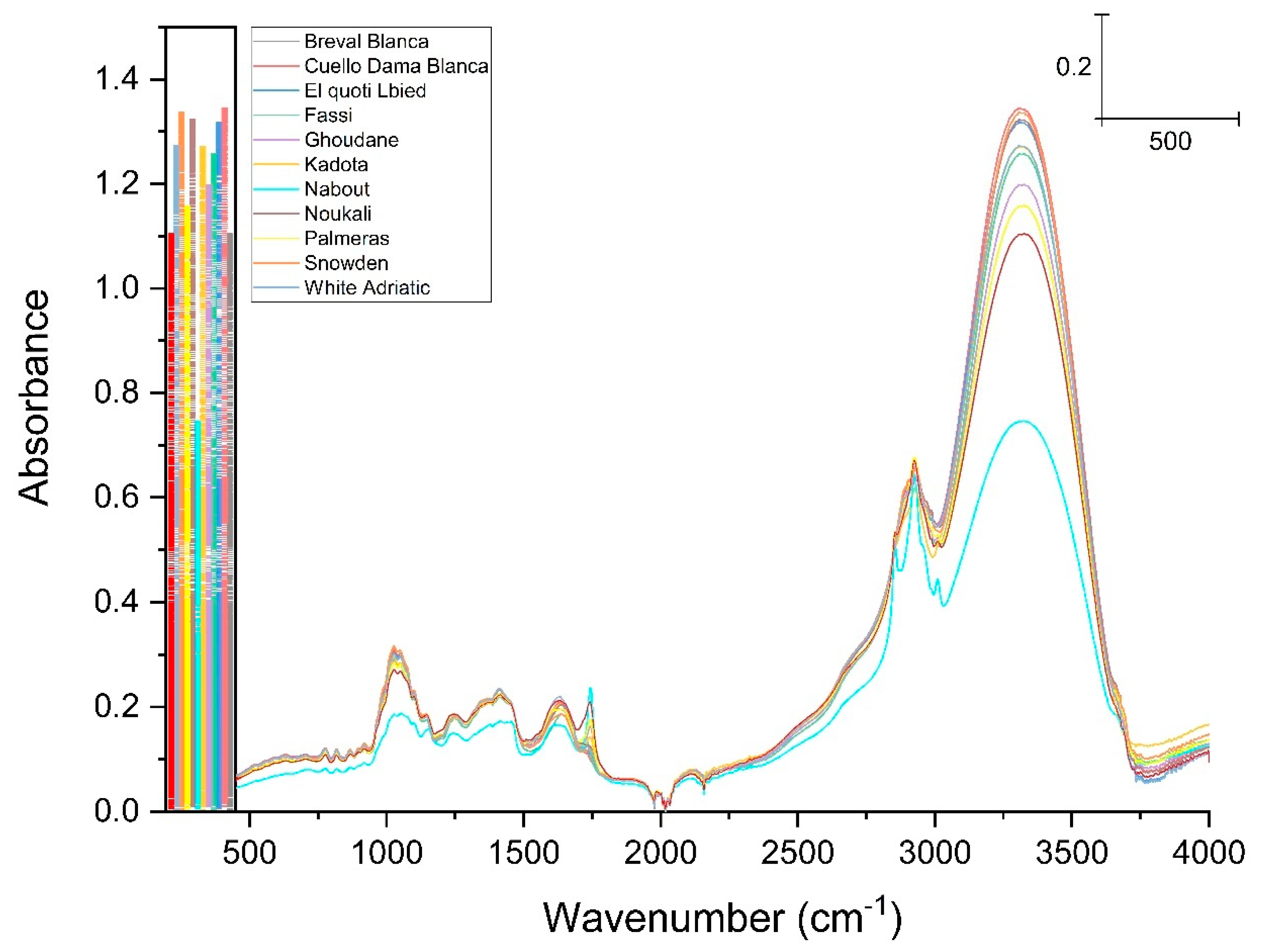
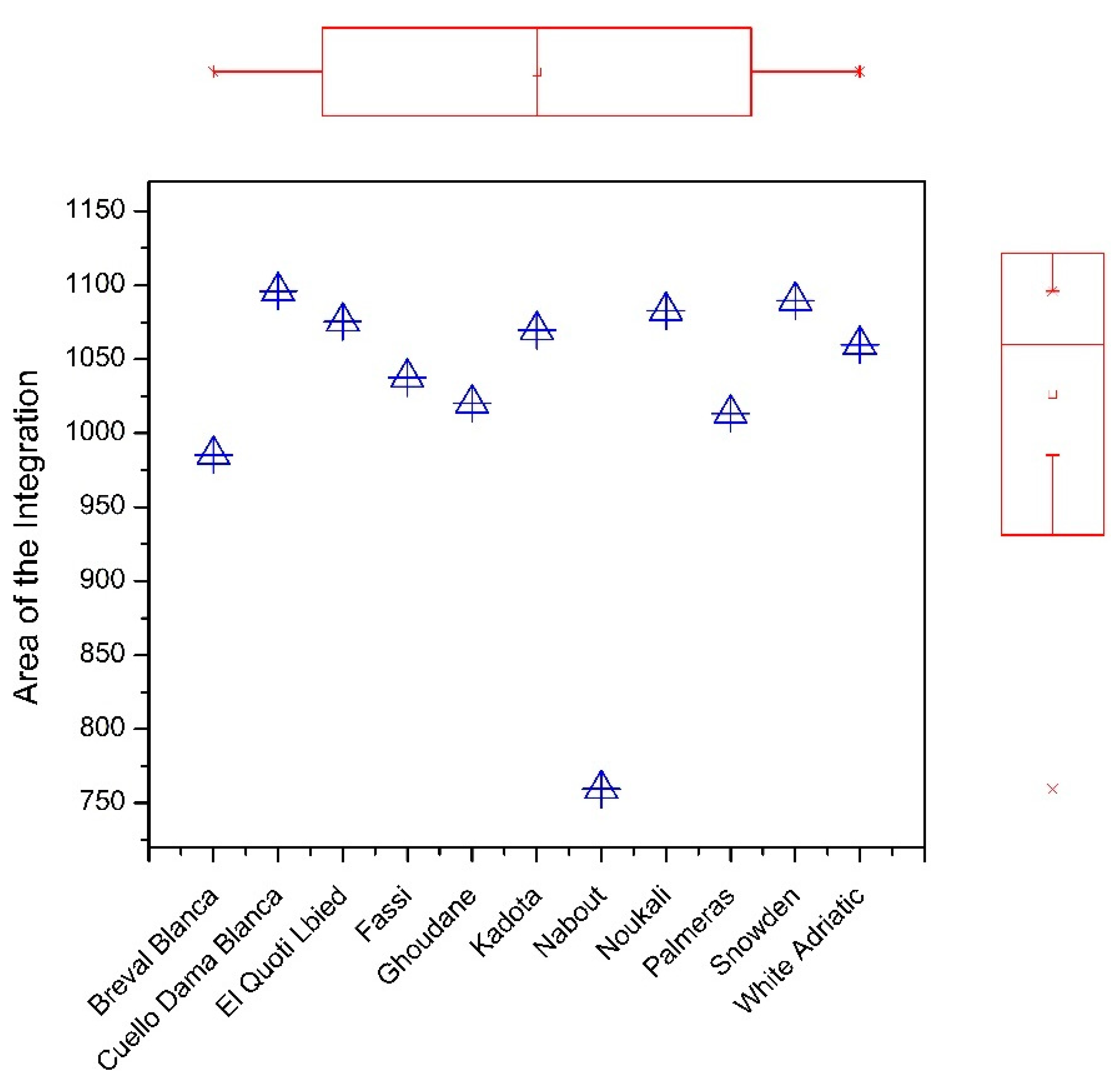
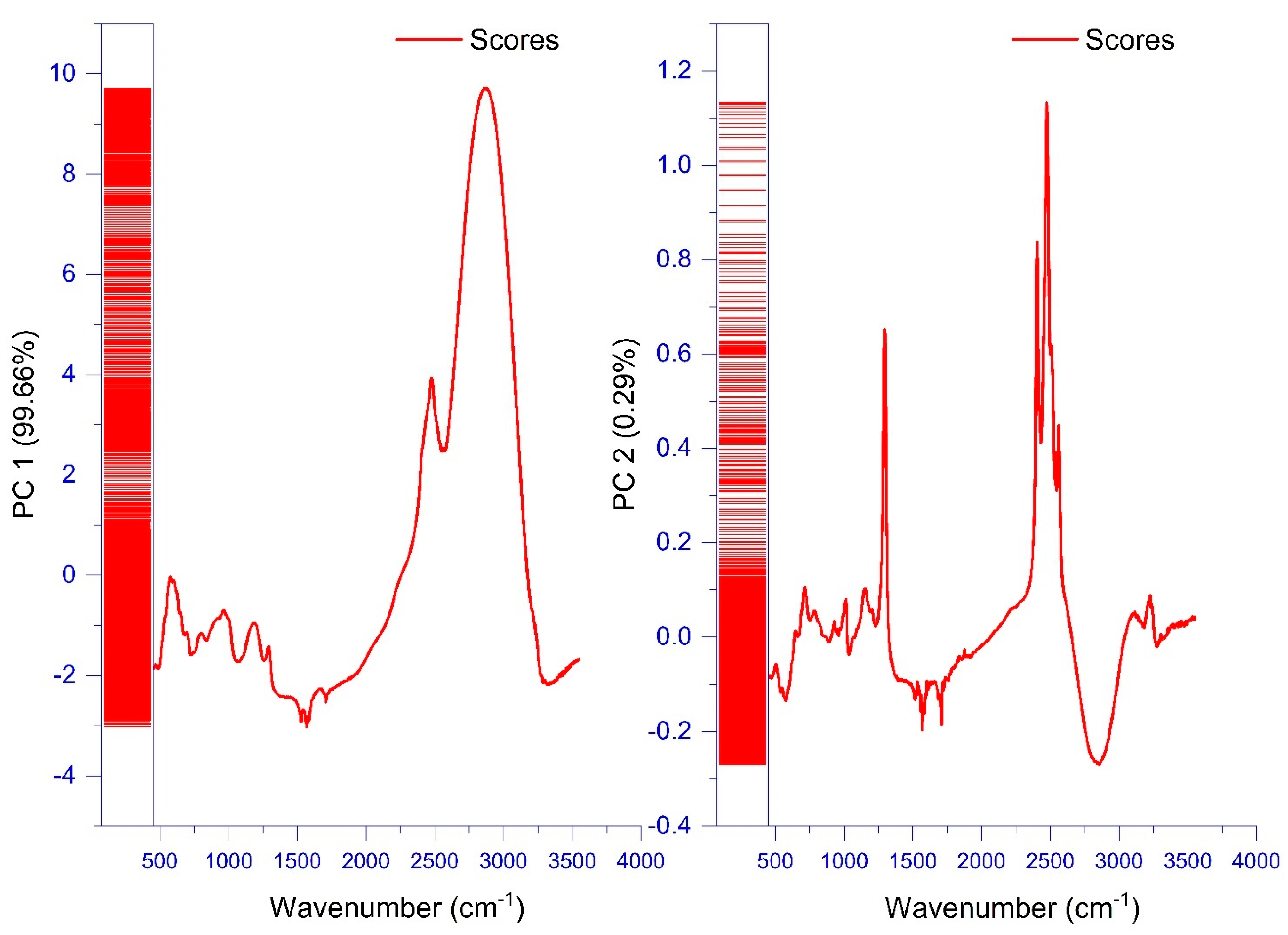
3.1. FTIR Fingerprinting
3.2. Peel Color
3.3. Reducing Sugars
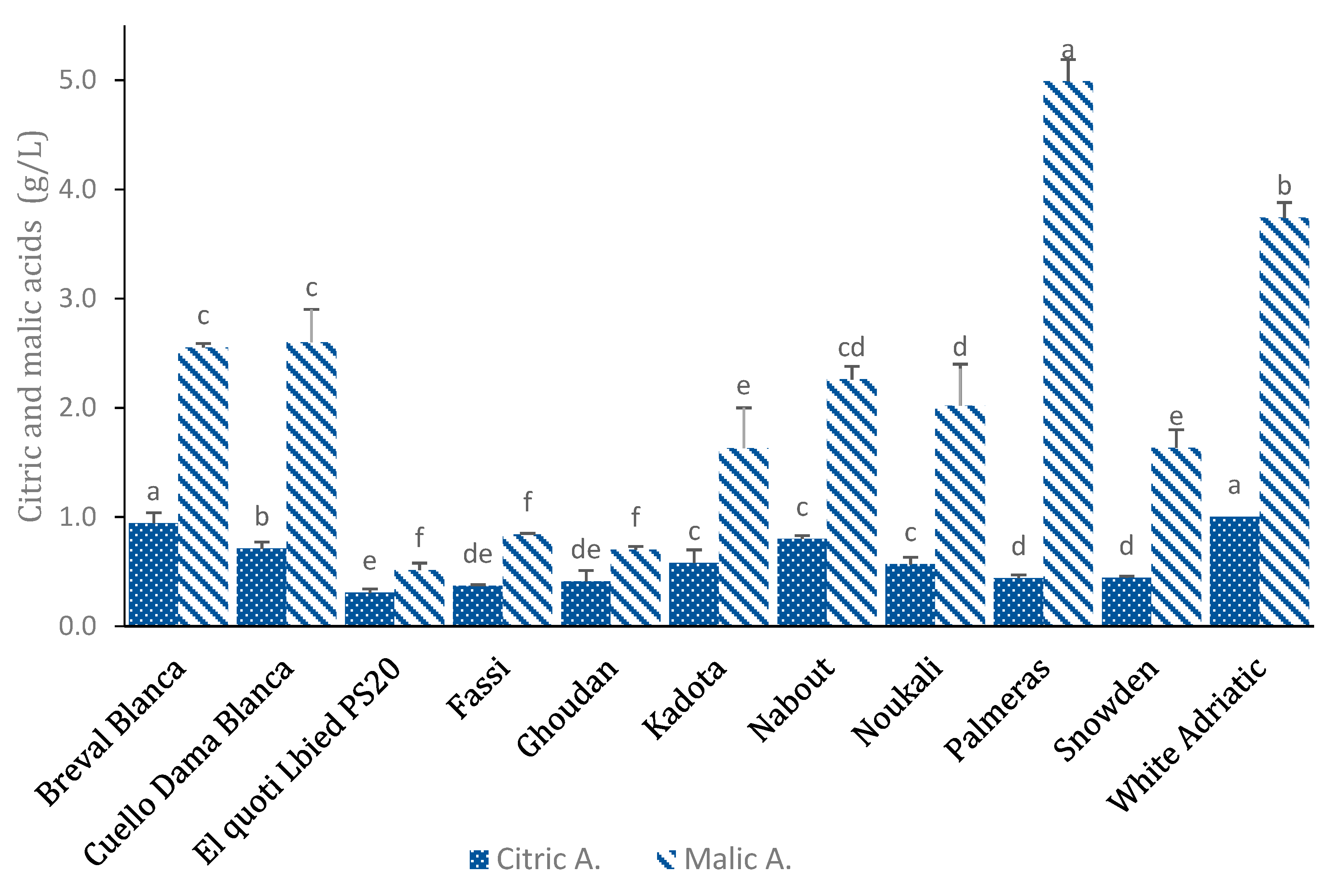
3.4. Organic Acids
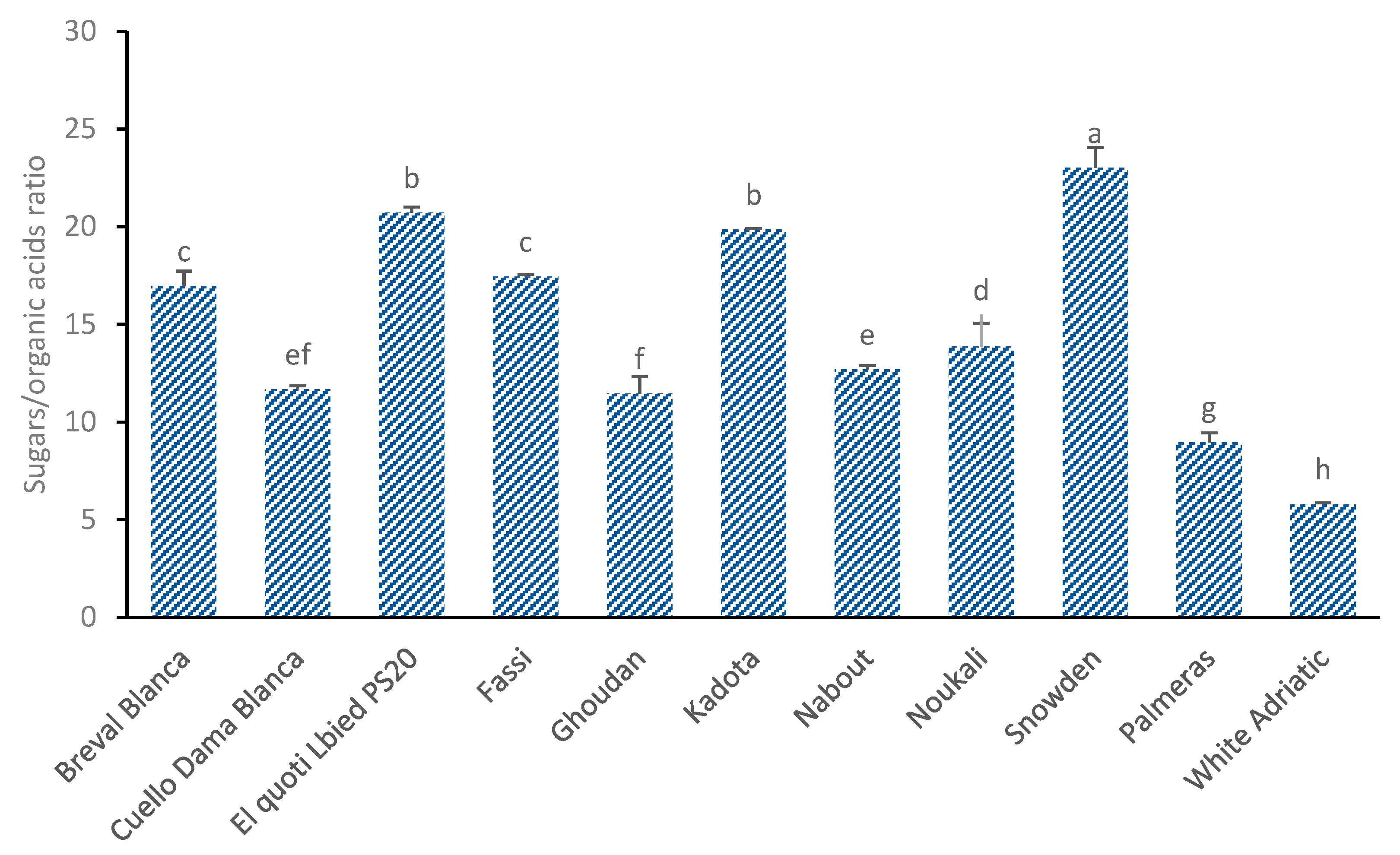
3.5. Sugars/Organic Acids Ratio
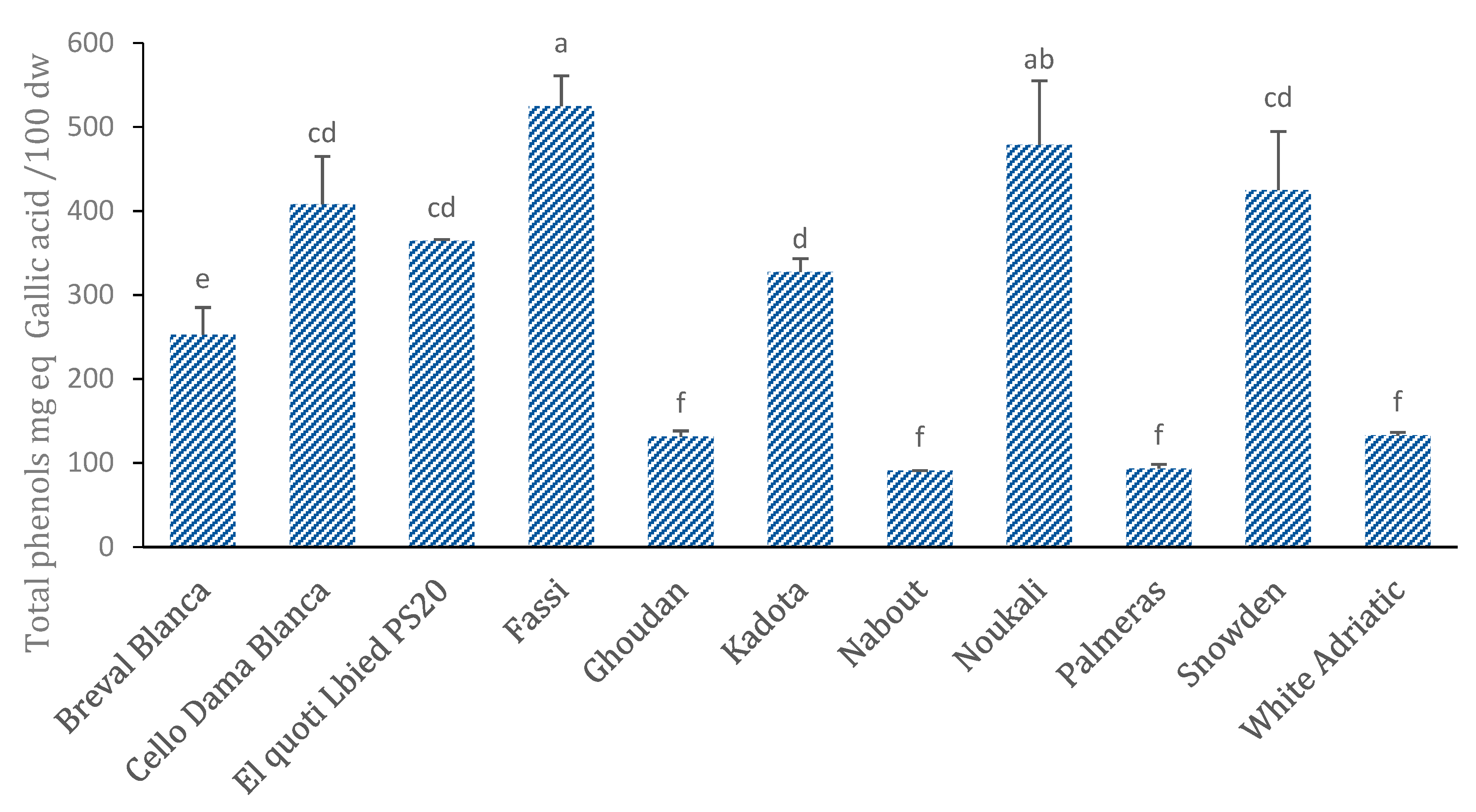
3.6. Total Phenol
3.7. Antioxidant Activity
3.8. Correlations Among Variables
3.9. Chemometric Analyses
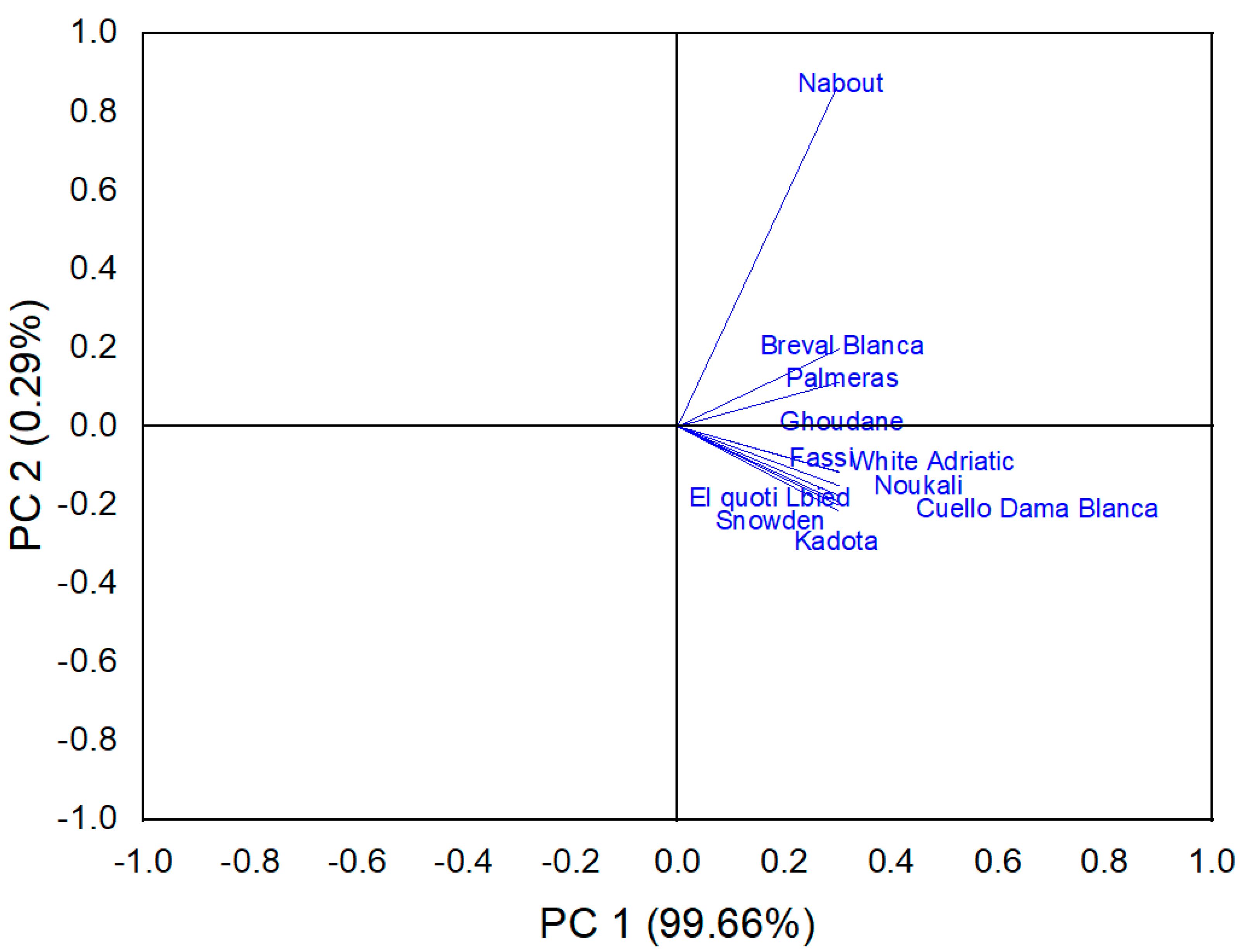
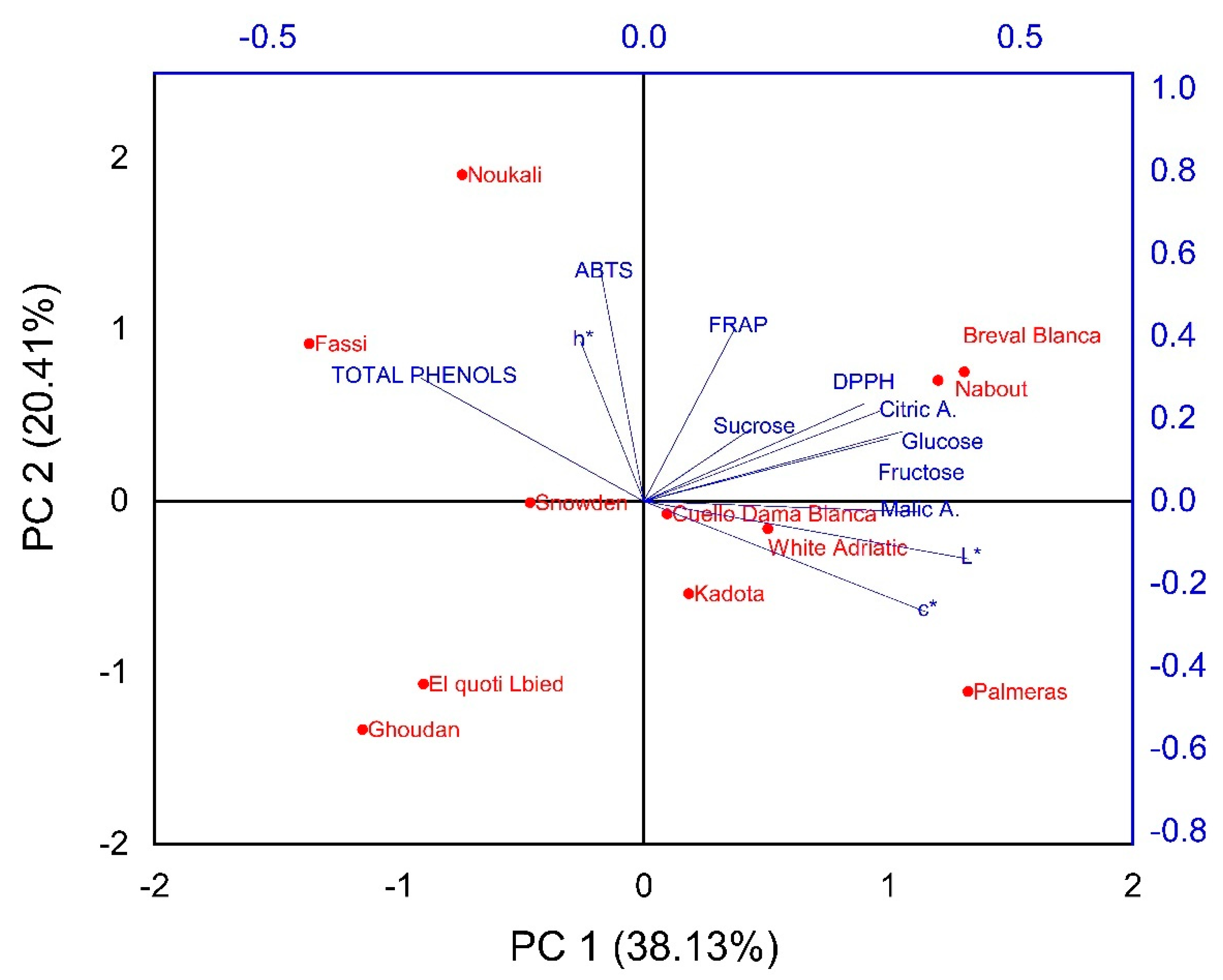
3.9.1. Principal Component Analysis
FTIR Fingerprinting
Biochemical Screening
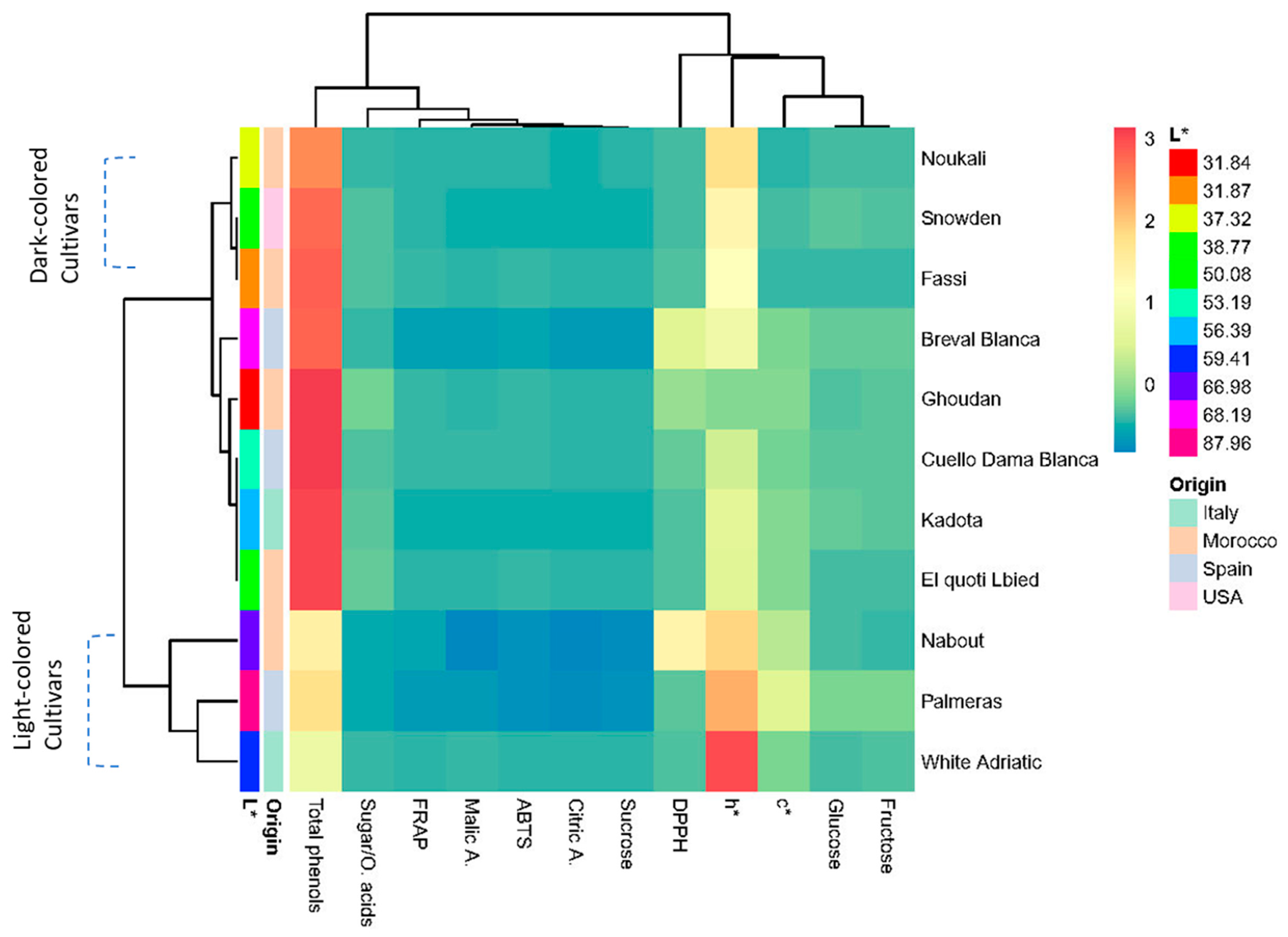
3.9.2. Two-Dimensional Clustered Heatmap
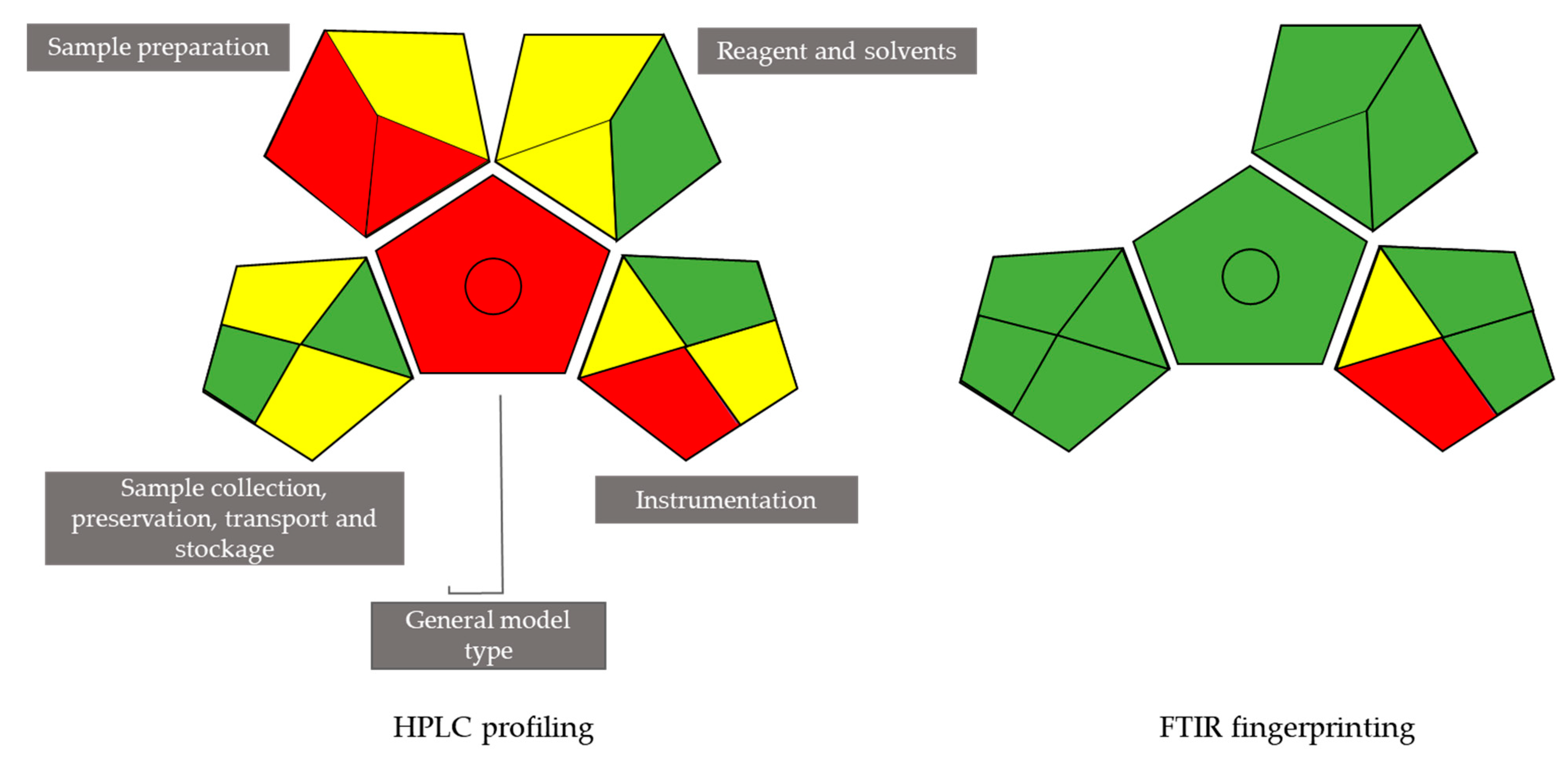
3.10. Evaluation of Greenness of the Analytical Techniques Used on the Scale of the Green Analytical Procedure Index (GAPI)
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Hssaini, L.; Hernandez, F.; Viuda-Martos, M.; Charafi, J.; Razouk, R.; Houmanat, K.; Ouaabou, R.; Ennahli, S.; Elothmani, D.; Hmid, I.; et al. Survey of Phenolic Acids, Flavonoids and In Vitro Antioxidant Potency Between Fig Peels and Pulps: Chemical and Chemometric Approach. Molecules 2021, 26, 2574. [Google Scholar] [CrossRef] [PubMed]
- Hssaini, L.; Ouaabou, R.; Charafi, J.; Idlimam, A.; Lamharrar, A.; Razouk, R.; Hanine, H. Hygroscopic proprieties of fig (Ficus carica L.): Mathematical modelling of moisture sorption isotherms and isosteric heat kinetics. S. Afr. J. Bot. 2020. [Google Scholar] [CrossRef]
- Hssaini, L.; Charafi, J.; Hanine, H.; Ennahli, S.; Mekaoui, A.; Mamouni, A.; Razouk, R. Comparative analysis and physio-biochemical screening of an ex-situ fig (Ficus carica L.) collection. Hortic. Environ. Biotechnol. 2019, 60, 671–683. [Google Scholar] [CrossRef]
- Hssaini, L.; Hanine, H.; Charafi, J.; Razouk, R.; Elantari, A.; Ennahli, S.; Hernández, F.; Ouaabou, R. First report on fatty acids composition, total phenolics and antioxidant activity in seeds oil of four fig cultivars (Ficus carica L.) grown in Morocco. OCL 2020, 27, 8. [Google Scholar] [CrossRef] [Green Version]
- Veberic, R.; Mikulic-Petkovsek, M. Phytochemical Composition of Common Fig (Ficus carica L.) Cultivars. In Nutritional Composition of Fruit Cultivars; Springer Science and Business Media B.V.: Berlin/Heidelberg, Germany, 2016; pp. 235–255. [Google Scholar] [CrossRef]
- Palassarou, M.; Melliou, E.; Liouni, M.; Michaelakis, A.; Balayiannis, G.; Magiatis, P. Volatile profile of Greek dried white figs (Ficus carica L.) and investigation of the role of β-damascenone in aroma formation in fig liquors. J. Sci. Food Agric. 2017, 97, 5254–5270. [Google Scholar] [CrossRef]
- Pourghayoumi, M.; Bakhshi, D.; Rahemi, M.; Noroozisharaf, A.; Jafari, M.; Salehi, M.; Chamane, R.; Hernandez, F. Phyto-chemical attributes of some dried fig (Ficus carica L.) fruit cultivars grown in Iran. Agric. Conspec. Sci. 2016, 81, 161–166. [Google Scholar]
- Hssaini, L.; Hanine, H.; Razouk, R.; Ennahli, S.; Mekaoui, A.; Charafi, J. Characterization of local fig clones (Ficus carica L.) collected in Northern Morocco. Fruits 2019, 74, 55–64. [Google Scholar] [CrossRef]
- Veberic, R.; Colaric, M.; Stampar, F. Phenolic acids and flavonoids of fig fruit (Ficus carica L.) in the northern Mediterranean region. Food Chem. 2008, 106, 153–157. [Google Scholar] [CrossRef]
- Hssaini, L.; Hanine, H.; Razouk, R.; Ennahli, S.; Mekaoui, A.; Ejjilani, A.; Charafi, J. Assessment of genetic diversity in Moroccan fig (Ficus carica L.) collection by combining morphological and physicochemical descriptors. Genet. Resour. Crop. Evol. 2020, 67, 457–474. [Google Scholar] [CrossRef]
- Piga, A.; Del Caro, A.; Milella, G.; Pinna, I.; Vacca, V.; Schirru, S. HPLC analysis of polyphenols in peel and pulp of fresh figs. In III International Symposium on Fig 798; Leitao, J., Neves, M.A., Eds.; ISHS: Algarve, Portugal, 2008; pp. 301–306. [Google Scholar] [CrossRef]
- Melgarejo, P.; Hernandez, F.; Martinez, J.; Sánchez, J.; Salazar, D. Organic acids and sugars from first and second crop fig juices. In II International Symposium on Fig 605; Corrales, M.L., García, M.J.B., Eds.; ISHS: Caceres, Spain, 2003; pp. 237–239. [Google Scholar] [CrossRef]
- Çalişkan, O.; Polat, A.A. Phytochemical and antioxidant properties of selected fig (Ficus carica L.) accessions from the eastern Mediterranean region of Turkey. Sci. Hortic. 2011, 128, 473–478. [Google Scholar] [CrossRef]
- Slatnar, A.; Klancar, U.; Stampar, F.; Veberic, R. Effect of Drying of Figs (Ficus carica L.) on the Contents of Sugars, Organic Acids, and Phenolic Compounds. J. Agric. Food Chem. 2011, 59, 11696–11702. [Google Scholar] [CrossRef] [PubMed]
- Vemmos, S.N.; Petri, E.; Stournaras, V. Seasonal changes in photosynthetic activity and carbohydrate content in leaves and fruit of three fig cultivars (Ficus carica L.). Sci. Hortic. 2013, 160, 198–207. [Google Scholar] [CrossRef]
- Pande, G.; Akoh, C.C. Organic acids, antioxidant capacity, phenolic content and lipid characterisation of Georgia-grown underutilized fruit crops. Food Chem. 2010, 120, 1067–1075. [Google Scholar] [CrossRef]
- Mikulic-Petkovsek, M.; Slatnar, A.; Schmitzer, V.; Stampar, F.; Veberic, R.; Koron, D. Chemical profile of black currant fruit modified by different degree of infection with black currant leaf spot. Sci. Hortic. 2013, 150, 399–409. [Google Scholar] [CrossRef]
- Das, T.; Harshey, A.; Nigam, K.; Yadav, V.K.; Srivastava, A. Analytical approaches for bloodstain aging by vibrational spectroscopy: Current trends and future perspectives. Microchem. J. 2020, 158, 105278. [Google Scholar] [CrossRef]
- Haarman, B.C.B.; Riemersma-Van der Lek, R.F.; Nolen, W.A.; Mendes, R.; Drexhage, H.A.; Burger, H. Feature-expression heat maps–A new visual method to explore complex associations between two variable sets. J. Biomed. Inform. 2015, 53, 156–161. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hernández, F.; Noguera-Artiaga, L.; Burló, F.; Wojdyło, A.; Carbonell-Barrachina, Á.A.; Legua, P. Physico-chemical, nutritional, and volatile composition and sensory profile of Spanish jujube (Ziziphus jujuba Mill.) fruits. J. Sci. Food Agric. 2016, 96, 2682–2691. [Google Scholar] [CrossRef]
- Brand-Williams, W.; Cuvelier, M.E.; Berset, C. Use of a free radical method to evaluate antioxidant activity. LWT-Food sci. Technol. 1995, 28, 25–30. [Google Scholar] [CrossRef]
- Re, R.; Pellegrini, N.; Proteggente, A.; Pannala, A.; Yang, M.; Rice-Evans, C. Antioxidant activity applying an improved ABTS radical cation decolorization assay. Free Radic. Biol. Med. 1999, 26, 1231–1237. [Google Scholar] [CrossRef]
- Benzie, I.F.F.; Strain, J.J. The ferric reducing ability of plasma (FRAP) as a measure of “antioxidant power”: The FRAP assay. Anal. Biochem. 1996, 239, 70–76. [Google Scholar] [CrossRef] [Green Version]
- Singleton, V.L.; Rossi, J.A. Colorimetry of total phenolics with phosphomolybdic-phosphotungstic acid reagents. Am. J. Enol. Vitic. 1965, 16, 144–153. [Google Scholar]
- Shen, X.; Xu, L.; Ye, S.; Hu, R.; Jin, L.; Xu, H.; Liu, W. Automatic baseline correction method for the open-path Fourier transform infrared spectra by using simple iterative averaging. Opt. Express 2018, 26, A609–A614. [Google Scholar] [CrossRef]
- Hssaini, L.; Razouk, R.; Charafi, J.; Houmanat, K.; Hanine, H. Fig seeds: Combined approach of lipochemical assessment using gas chromatography and FTIR-ATR spectroscopy using chemometrics. Vib. Spectrosc. 2021, 114, 103251. [Google Scholar] [CrossRef]
- Baranowska-Wójcik, E.; Szwajgier, D. Characteristics and pro-health properties of mini kiwi (Actinidia arguta). Hortic. Environ. Biotechnol. 2019, 60, 217–225. [Google Scholar] [CrossRef]
- Palmeira, L.; Pereira, C.; Dias, M.I.; Abreu, R.M.; Corrêa, R.C.G.; Pires, T.C.; Alves, M.J.; Barros, L.; Ferreira, I.C. Nutritional, chemical and bioactive profiles of different parts of a Portuguese common fig (Ficus carica L.) variety. Food Res. Int. 2019, 126, 108572. [Google Scholar] [CrossRef] [Green Version]
- Bouafif, H.; Koubaa, A.; Perré, P.; Cloutier, A.; Riedl, B. Analysis of among-species variability in wood fiber sur-face using DRIFTS and XPS: Effects on esterification efficiency. J. Wood Chem. Technol. 2008, 28, 296–315. [Google Scholar] [CrossRef]
- Oh, S.Y.; Yoo, D.I.; Shin, Y.; Seo, G. FTIR analysis of cellulose treated with sodium hydroxide and carbon dioxide. Carbohydr. Res. 2005, 340, 417–428. [Google Scholar] [CrossRef]
- Gok, S.; Severcan, M.; Goormaghtigh, E.; Kandemir, I.; Severcan, F. Differentiation of Anatolian honey samples from different botanical origins by ATR-FTIR spectroscopy using multivariate analysis. Food Chem. 2015, 170, 234–240. [Google Scholar] [CrossRef] [PubMed]
- Vongsvivut, J.; Heraud, P.; Gupta, A.; Puri, M.; McNaughton, D.; Barrow, C.J. FTIR microspectroscopy for rap-id screening and monitoring of polyunsaturated fatty acid production in commercially valuable marine yeasts and protists. Analyst 2013, 138, 6016–6031. [Google Scholar] [CrossRef] [Green Version]
- Terpugov, E.L.; Degtyareva, O.V.; Savransky, V.V. Possibility of Light-Induced Mid-IR Emission in Situ Analysis of Plants. J. Russ. Laser Res. 2016, 37, 507–510. [Google Scholar] [CrossRef]
- Cassani, L.; Santos, M.; Gerbino, E.; del Rosario Moreira, M.; Gómez-Zavaglia, A. A combined approach of infrared spectroscopy and multivariate analysis for the simultaneous determination of sugars and fructans in strawberry juices during storage. J. Food Sci. 2018, 83, 631–638. [Google Scholar] [CrossRef]
- Agrebi, F.; Ghorbel, N.; Bresson, S.; Abbas, O.; Kallel, A. Study of nanocomposites based on cellulose nanoparticles and natural rubber latex by ATR/FTIR spectroscopy: The impact of reinforcement. Polym. Compos. 2019, 40, 2076–2087. [Google Scholar] [CrossRef]
- Viuda-Martos, M.; Barber, X.; Pérez-Álvarez, J.A.; Fernández-López, J. Assessment of chemical, physico-chemical, techno-functional and antioxidant properties of fig (Ficus carica L.) powder co-products. Ind. Crop. Prod. 2015, 69, 472–479. [Google Scholar] [CrossRef]
- Sedaghat, S.; Rahemi, M. Effects of physio-chemical changes during fruit development on nutritional quality of fig (Ficus carica L. var. ‘Sabz’) under rain-fed condition. Sci. Hortic. 2018, 237, 44–50. [Google Scholar] [CrossRef]
- Souza, M.; Jemni, M.; Otón, M.; Leonel, S.; Melgarejo, P.; Artés, F. Caracterización morfológica, química y sensorial de cuatro variedades de brevas. Rev. Iberoam. Tecnol. Postcosecha 2013, 14, 48–52. [Google Scholar]
- Veberic, R.; Jakopic, J.; Stampar, F. Internal fruit quality of figs (Ficus carica L.) in the Northern Mediterranean Region. Italian J. Food Sci. 2008, 20, 255–262. [Google Scholar]
- Veberic, R.; Schmitzer, V.; Petkovsek, M.M.; Stampar, F. Impact of shelf life on content of primary and second-ary metabolites in apple (Malus domestica Borkh.). J.Food Sci. 2010, 75, S461–S468. [Google Scholar] [CrossRef] [PubMed]
- Colaric, M.; Veberic, R.; Stampar, F.; Hudina, M. Evaluation of peach and nectarine fruit quality and correlations between sensory and chemical attributes. J. Sci. Food Agric. 2005, 85, 2611–2616. [Google Scholar] [CrossRef]
- Pereira, C.; López Corrales, M.; Martín, A.; Villalobos, M.D.C.; Córdoba, M.D.G.; Serradilla, M.J. Physico-chemical and nutritional characterization of brebas for fresh consumption from nine fig varieties (Ficus carica L.) grown in Extremadura (Spain). J. Food Qual. 2017, 2017, 1–12. [Google Scholar] [CrossRef] [Green Version]
- Serradilla, M.J.; Lozano, M.; Bernalte, M.J.; Ayuso, M.C.; López-Corrales, M.; González-Gómez, D. Physicochemical and bioactive properties evolution during ripening of ‘Ambrunés’ sweet cherry cultivar. LWT-Food Sci. Technol. 2011, 44, 199–205. [Google Scholar] [CrossRef]
- Wojdyło, A.; Nowicka, P.; Carbonell-Barrachina, Á.A.; Hernández, F. Phenolic compounds, antioxidant and antidiabetic activity of different cultivars of Ficus carica L. fruits. J. Funct. Foods 2016, 25, 421–432. [Google Scholar] [CrossRef]
- Harzallah, A.; Bhouri, A.M.; Amri, Z.; Soltana, H.; Hammami, M. Phytochemical content and antioxidant activity of different fruit parts juices of three figs (Ficus carica L.) varieties grown in Tunisia. Ind. Crop. Prod. 2016, 83, 255–267. [Google Scholar] [CrossRef]
- Del Caro, A.; Piga, A. Polyphenol composition of peel and pulp of two Italian fresh fig fruits cultivars (Ficus carica L.). Eur. Food Res. Technol. 2008, 226, 715–719. [Google Scholar] [CrossRef]
- Turkmen, N.; Sari, F.; Velioglu, Y.S. Effects of extraction solvents on concentration and antioxidant activity of black and black mate tea polyphenols determined by ferrous tartrate and Folin–Ciocalteu methods. Food Chem. 2006, 99, 835–841. [Google Scholar] [CrossRef]
- Clark, N.R.; Ma’Ayan, A. Introduction to Statistical Methods to Analyze Large Data Sets: Principal Components Analysis. Sci. Signal. 2011, 4, tr3. [Google Scholar] [CrossRef] [Green Version]
- Harvey, T.J.; Gazi, E.; Henderson, A.; Snook, R.D.; Clarke, N.; Brown, M.; Gardner, P. Factors influencing the discrimination and classification of prostate cancer cell lines by FTIR microspectroscopy. Analyst 2009, 134, 1083–1091. [Google Scholar] [CrossRef]
- Mohamed, D.; Hegazy, M.A.; El-Sayed, G.M.; Youssef, S.H. Greenness evaluation of different chromato-graphic approaches for the determination of dextromethorphan, phenylephrine & brompheniramine in their pharmaceutical formulation. Microchem. J. 2020, 157, 104893. [Google Scholar]
- Nasr, J.J.M.; Al-Shaalan, N.H.; Shalan, S.M. Sustainable environment-friendly quantitative determination of three anti-hyperlipidemic statin drugs and ezetimibe in binary mixtures by first derivative Fourier transform infrared (FTIR) spectroscopy. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2020, 237, 118332. [Google Scholar] [CrossRef]
- Gałuszka, A.; Migaszewski, Z.M.; Konieczka, P.; Namieśnik, J. Analytical Eco-Scale for assessing the greenness of analytical procedures. TrAC Trends Anal. Chem. 2012, 37, 61–72. [Google Scholar] [CrossRef]
- Kurowska-Susdorf, A.; Zwierżdżyński, M.; Bevanda, A.M.; Talić, S.; Ivanković, A.; Płotka-Wasylka, J. Green analytical chemistry: Social dimension and teaching. TrAC Trends Anal. Chem. 2019, 111, 185–196. [Google Scholar] [CrossRef]

| Cultivar | Geographical Origin | August | September | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| (1–5) | (6–10) | (11–15) | (16–20) | (21–25) | (26–30) | (31–4) | (5–9) | |||
| Local | El Quoti Lbied | Morocco | ||||||||
| Nabout | ||||||||||
| Fassi | ||||||||||
| Noukali | ||||||||||
| Ghoudan | ||||||||||
| Introduced | Snowden | USA | ||||||||
| White Adriatic | Italy | |||||||||
| Kadota | Italy | |||||||||
| Cuello Dama Blanca | Spain | |||||||||
| Breval Blanca | Spain | |||||||||
| Palmeras | Spain | |||||||||
| Total rainfall (mm) | 0 | 0 | 0 | 0 | 0 | 26.4 | 0 | 0 | ||
| Average temperature (°C) | 25.84 | 28.5 | 27.56 | 29.24 | 29.44 | 23.64 | 25.6 | 25.42 | ||
| Average solar radiation (W/m2) | 169.29 | 208.74 | 243.83 | 238.28 | 185.35 | 123.5 | 270.21 | 271.38 | ||
| Soil type | Sandy clay loam with an average organic matter of 1% (0–30 cm soil layer) | |||||||||
| Soil pH | 7.2 | |||||||||
| Wavenumbers (cm−1) | Assignment | References |
|---|---|---|
| 3326 | Intra-molecular hydrogen bonding C(3)OH···O(5) C(6)O···(O)H | [27,28] |
| 2929 | C-H symmetric and asymmetric stretching of CH3 and CH2 groups | [29,30] |
| 1745 | Stretching of C=O | [28,29,30] |
| 1630 | N–H bending and C–N stretching of amide I | [30,31] |
| 1414 and 1238 | N-H in-bending and C–N stretching of amid II CH, CH2 and CH3 deformations | [32,33,34] |
| 1025 | Stretching of C–OH, C–C and C–O | [34,35] |
| Cultivar | L* | c* | h* |
|---|---|---|---|
| Breval Blanca | 63.92 ± 6.92 b | 36.34 ± 3.48 cd | 103.55 ± 3.23 cd |
| Cuello Dama Blanca | 60.26 ± 16.53 bc | 33.44 ± 2.39 d | 100.14 ± 4.82 cd |
| El Quoti Lbied | 48.8 ± 1.79 d | 37.29 ± 1.13 bc | 95.72 ± 9.06 cd |
| Fassi | 31.78 ± 3.75 f | 10.28 ± 0.46 f | 215.93 ± 16.81 c |
| Ghoudan | 34.91 ± 6.32 ef | 15.83 ± 2.99 e | 19.24 ± 14.93 d |
| Kadota | 56.12 ± 7 c | 37.44 ± 4.62 bc | 106.47 ± 5.66 cd |
| Nabout | 64.03 ± 3.19 b | 40.04 ± 3.96 ab | 105.2 ± 3.48 cd |
| Noukali | 32.43 ± 11.87 f | 6.2 ± 0.37 g | 356.14 ± 12.85 b |
| Palmeras | 83.64 ± 5.37 a | 42.12 ± 7.67 a | 102.63 ± 7.43 cd |
| Snowden | 39.8 ± 1.48 ef | 17.71 ± 0.59 e | 185.47 ± 19.81 c |
| White Adriatic | 60.49 ± 3.43 bc | 36.08 ± 1.16 cd | 115.7 ± 15.42 a |
| Mean | 51.23 ± 6.07 | 27.64 ± 2.54 | 181.47 ± 11.03 |
| ANOVA mean square | 2686.33 *** | 1725.34 *** | 215052.33 *** |
| Cultivar | SUCR (g kg−1 dw) | GLUC (g kg−1 dw) | FRUC (g kg−1 dw) | Total Reducing Sugars (g kg−1 dw) |
|---|---|---|---|---|
| Breval Blanca | 1.19 ± 0.07 f | 29.94 ± 0.81 a | 28.15 ± 0.78 a | 59.28 a |
| CuelloDama Blanca | 1.68 ± 0.05 e | 18.52 ± 2.4 de | 18.47 ± 2.34 d | 38.67 d |
| El Quoti Lbied | 1.61 ± 0.25 e | 7.82 ± 1.05 fg | 7.57 ± 1.01 fg | 17 h |
| Fassi | 2.03 ± 0.06 cd | 9.48 ± 0.18 f | 9.6 ± 0.26 f | 21.11 g |
| Ghoudan | 0.86 ± 0.01 g | 5.55 ± 0.27 g | 6.23 ± 0.28 g | 12.63 i |
| Kadota | 2.36 ± 0.34 c | 21.41 ± 4.8 cd | 20.11 ± 4.5 cd | 43.88 c |
| Nabout | 2.93 ± 0.14 b | 18.46 ± 0.62 de | 17.4 ± 0.51 d | 38.79 d |
| Noukali | 2.25 ± 0.07 c | 16 ± 2.16 e | 17.23 ± 0.86 d | 35.47 e |
| Palmeras | 1.80 ± 0.31 de | 23.29 ± 0.35 bc | 23.55 ± 0.41 b | 48.63 b |
| Snowden | 1.13 ± 0.04 fg | 24.68 ± 1.01 b | 21.85 ± 0.9 bc | 47.65 b |
| White Adriatic | 3.66 ± 0.28 a | 10.98 ± 0.1 f | 12.85 ± 0.61 e | 27.49 f |
| Mean | 1.95 ± 0.15 | 16.92 ± 1.25 | 16.64 ± 1.13 | 17 h |
| ANOVA mean square | 2.04 *** | 179.61 *** | 143.47 *** |
| Cultivar | DPPH (mmol TE eq/g dw) | ABTS (mmol TE eq/g dw) | FRAP (mmol TE eq/g dw) |
|---|---|---|---|
| Breval Blanca | 83.16 ± 6.93 b | 6.41 ± 0.66 b | 4.29 ± 0.53 ed |
| Cuello Dama Blanca | 20.84 ± 2.3 c | 3.73 ± 0.61 c | 4.14 ± 0.58 def |
| El Quoti Lbied | 14.28 ± 1.42 d | 3.59 ± 0.43 c | 1.09 ± 0.04 g |
| Fassi | 17.52 ± 0.98 cd | 5.96 ± 0.52 b | 6.06 ± 0.9 bc |
| Ghoudan | 17.78 ± 0.73 cd | 1.76 ± 0.48 d | 2.89 ± 0.33 ef |
| Kadota | 17.3 ± 1.27 cd | 2.43 ± 1.02 d | 2.55 ± 1.27 efg |
| Nabout | 88.1 ± 3.37 a | 4.44 ± 0.55 c | 10.65 ± 0.18 a |
| Noukali | 17.95 ± 1.47 cd | 8.04 ± 0.3 a | 7.44 ± 2.2 bc |
| Palmeras | 18.02 ± 1.96 cd | 1.43 ± 0.25 d | 4.95 ± 0.12 cd |
| Snowden | 14.76 ± 1.61 d | 1.75 ± 0.51 d | 3.53 ± 0.81 ef |
| White Adriatic | 15.21 ± 1.37 d | 2.11 ± 0.46 d | 1.97 ± 0.88 fg |
| Mean | 38.16 ± 2.12 | 2.78 ± 0.66 | 4.50 ± 0.71 |
| ANOVA mean square | 1967.25 *** | 14.53 *** | 18.51 *** |
| Citric Acid | Malic Acid | SUCR | GLUC | FRUC | ABTS | DPPH | FRAP | TP | L* | c* | h* | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Citric acid | 1 | 0.488 ** | 0.518 ** | 0.388 * | 0.441 * | 0.141 | 0.548 ** | 0.125 | −0.323 | 0.391 * | 0.372 * | 0.155 |
| Malic acid | 1 | 0.379 * | 0.487 ** | 0.583 ** | −0.226 | 0.115 | 0.146 | −0.492 ** | 0.734 ** | 0.499 ** | 0.103 | |
| SUCR | 1 | −0.122 | −0.052 | 0.031 | −0.015 | 0.185 | −0.178 | 0.240 | 0.255 | 0.295 | ||
| GLUC | 1 | 0.988 ** | 0.052 | 0.473 ** | 0.207 | −0.015 | 0.496 ** | 0.350 * | −0.048 | |||
| FRUC | 1 | 0.078 | 0.455 ** | 0.221 | −0.059 | 0.548 ** | 0.358 * | −0.021 | ||||
| ABTS | 1 | 0.371 * | 0.517 ** | 0.524 ** | −0.185 | −0.349 | 0.195 | |||||
| DPPH | 1 | 0.473 ** | −0.316 | 0.361 * | 0.345 | −0.168 | ||||||
| FRAP | 1 | 0.075 | 0.019 | −0.193 | 0.050 | |||||||
| TP | 1 | −0.558 ** | −0.563 ** | 0.228 | ||||||||
| L* | 1 | 0.855 ** | −0.168 | |||||||||
| c* | 1 | −0.285 | ||||||||||
| h* | 1 |
| Variables | Components | ||
|---|---|---|---|
| 1 | 2 | 3 | |
| Citric acid | 0.672 | 0.236 | 0.403 |
| Malic acid | 0.797 | −0.126 | 0.260 |
| Sucrose | 0.284 | 0.856 | |
| Glucose | 0.717 | 0.359 | −0.408 |
| Fructose | 0.764 | 0.355 | −0.326 |
| ABTS | −0.124 | 0.853 | |
| DPPH | 0.587 | 0.460 | −0.189 |
| FRAP | 0.182 | 0.694 | 0.101 |
| Total phenols | −0.568 | 0.548 | −0.119 |
| L* | 0.871 | −0.238 | |
| c* | 0.760 | −0.430 | |
| h* | −0.108 | 0.297 | 0.626 |
| Percentage of variance | 36.091 | 19.825 | 14.152 |
| Cumulative percentage | 36.091 | 55.916 | 70.068 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hssaini, L.; Elfazazi, K.; Razouk, R.; Ouaabou, R.; Hernandez, F.; Hanine, H.; Charafi, J.; Houmanat, K.; Aboutayeb, R. Combined Effect of Cultivar and Peel Chromaticity on Figs’ Primary and Secondary Metabolites: Preliminary Study Using Biochemical and FTIR Fingerprinting Coupled to Chemometrics. Biology 2021, 10, 573. https://doi.org/10.3390/biology10070573
Hssaini L, Elfazazi K, Razouk R, Ouaabou R, Hernandez F, Hanine H, Charafi J, Houmanat K, Aboutayeb R. Combined Effect of Cultivar and Peel Chromaticity on Figs’ Primary and Secondary Metabolites: Preliminary Study Using Biochemical and FTIR Fingerprinting Coupled to Chemometrics. Biology. 2021; 10(7):573. https://doi.org/10.3390/biology10070573
Chicago/Turabian StyleHssaini, Lahcen, Kaoutar Elfazazi, Rachid Razouk, Rachida Ouaabou, Francisca Hernandez, Hafida Hanine, Jamal Charafi, Karim Houmanat, and Rachid Aboutayeb. 2021. "Combined Effect of Cultivar and Peel Chromaticity on Figs’ Primary and Secondary Metabolites: Preliminary Study Using Biochemical and FTIR Fingerprinting Coupled to Chemometrics" Biology 10, no. 7: 573. https://doi.org/10.3390/biology10070573
APA StyleHssaini, L., Elfazazi, K., Razouk, R., Ouaabou, R., Hernandez, F., Hanine, H., Charafi, J., Houmanat, K., & Aboutayeb, R. (2021). Combined Effect of Cultivar and Peel Chromaticity on Figs’ Primary and Secondary Metabolites: Preliminary Study Using Biochemical and FTIR Fingerprinting Coupled to Chemometrics. Biology, 10(7), 573. https://doi.org/10.3390/biology10070573