Growth Forms and Functional Guilds Distribution of Soil Fungi in Coastal Versus Inland Sites of Victoria Land, Antarctica
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Description of the Localities
2.2. Soil Physicochemical and Molecular Analyses
2.3. Statistical Analyses
3. Results
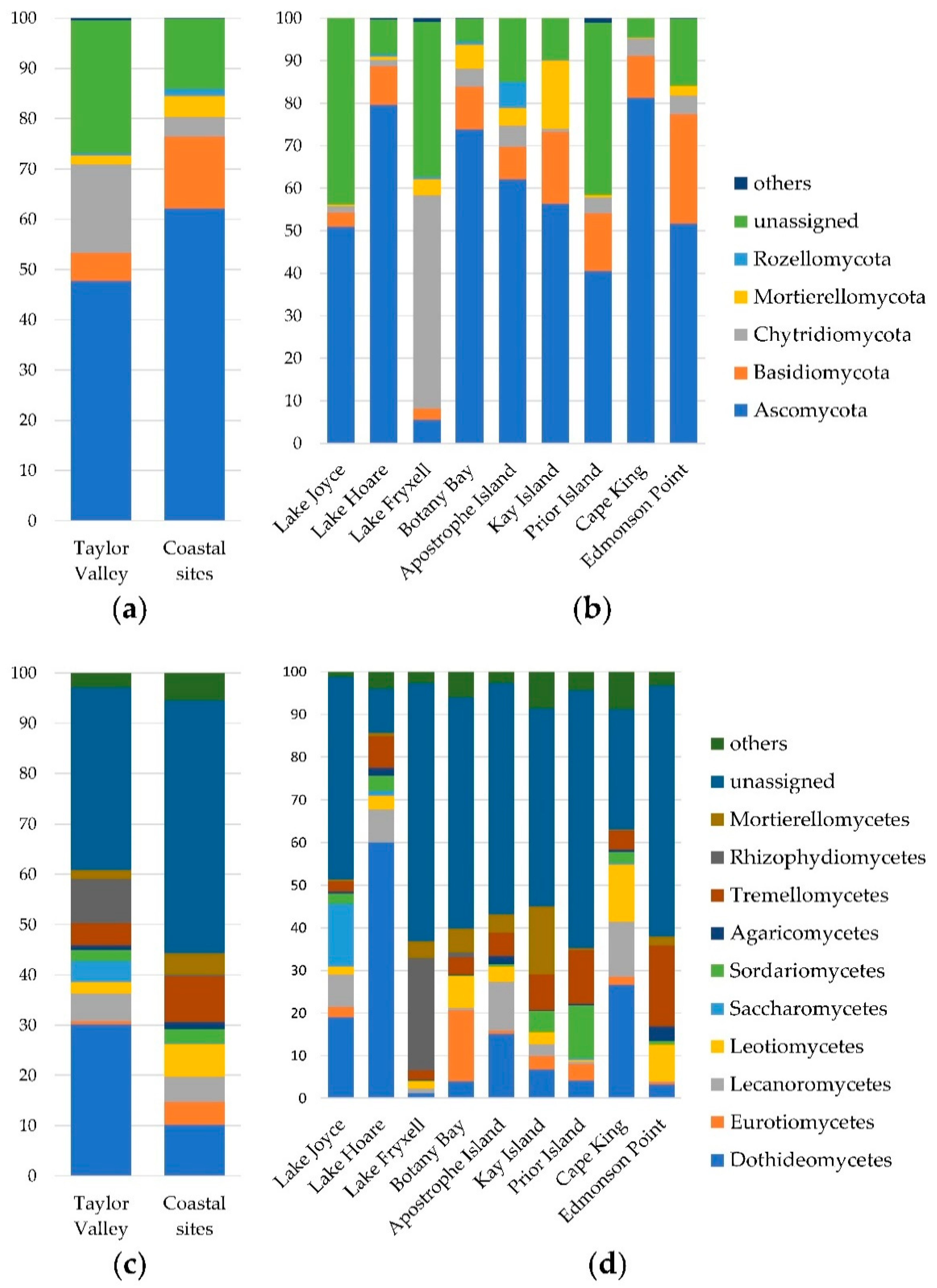
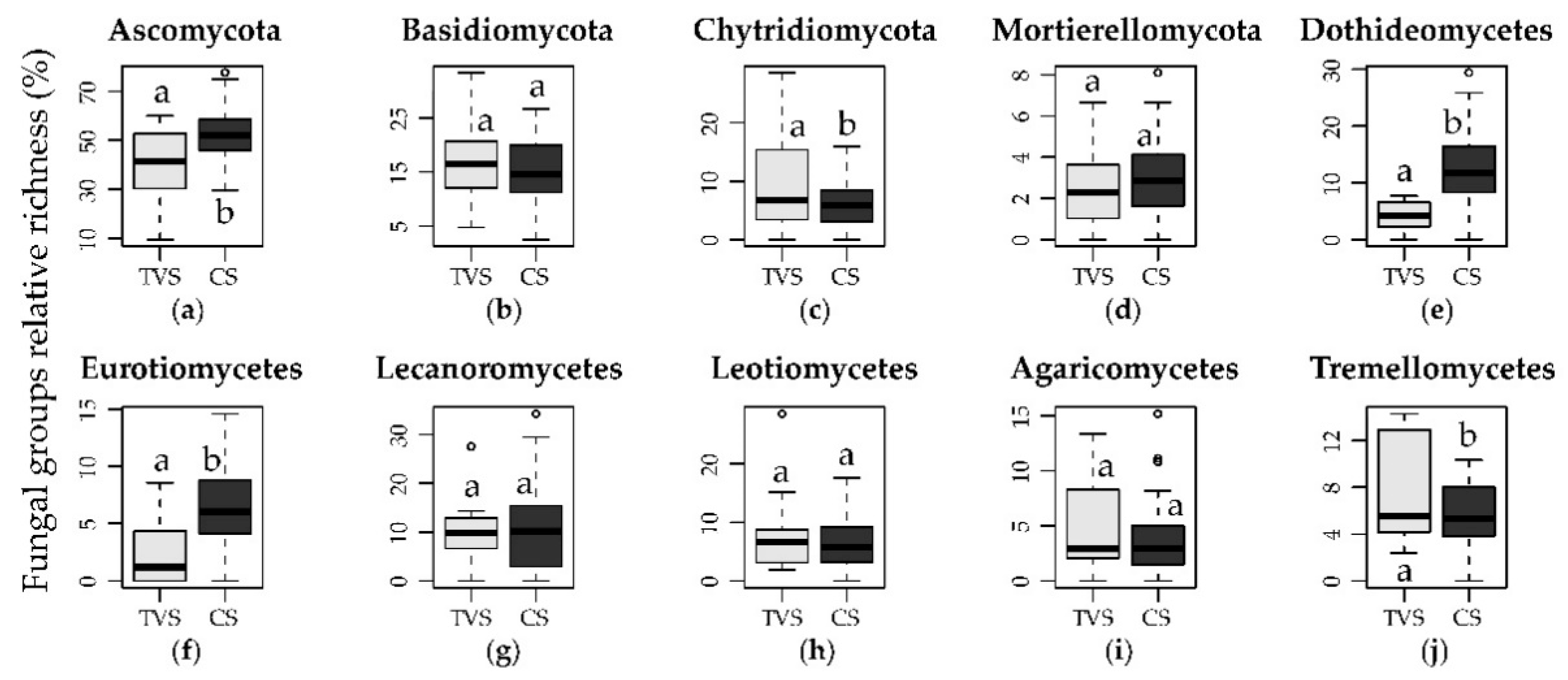
3.1. Taxonomic Characterization
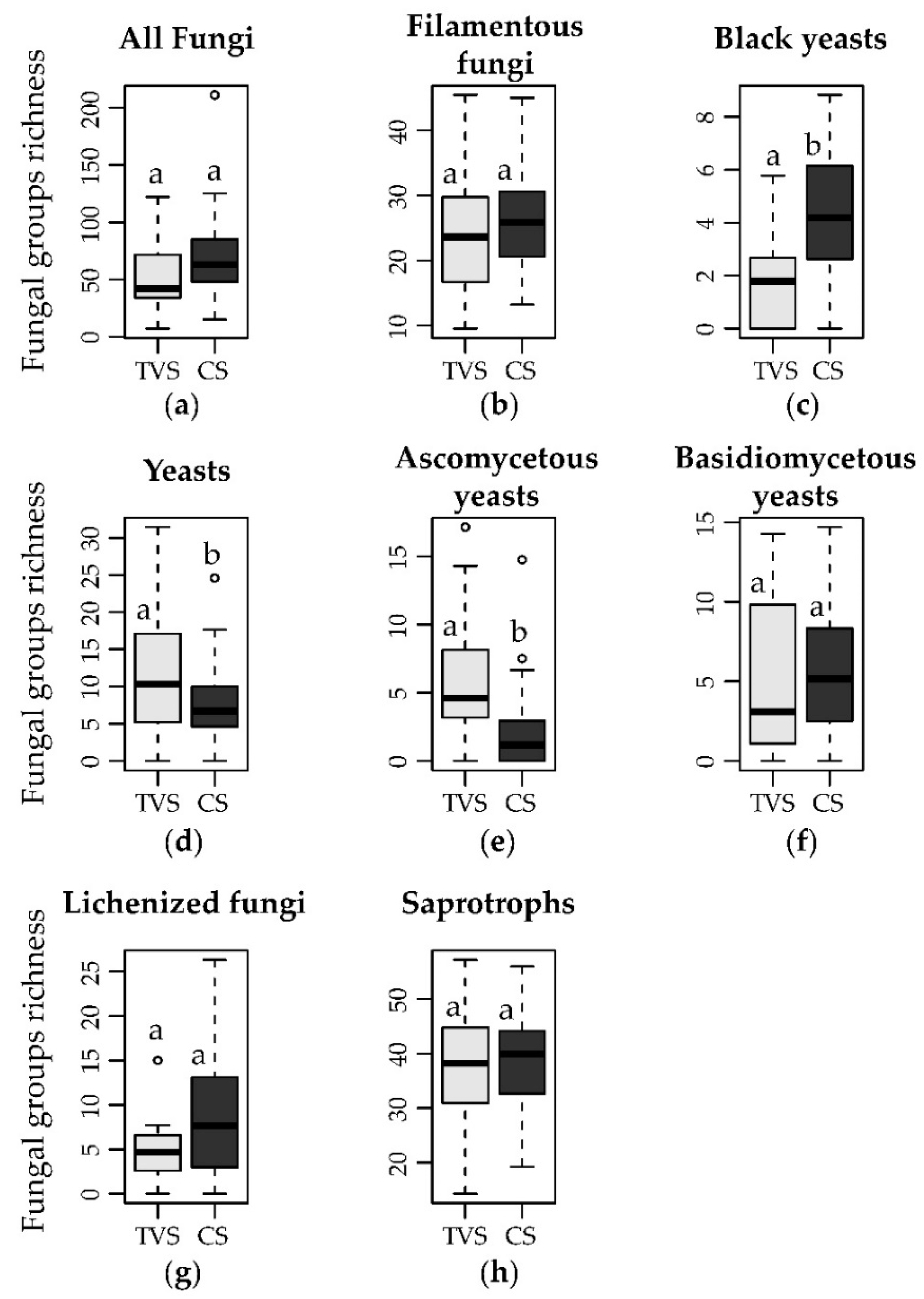
3.2. Distribution of the Different Growth Forms and Lifestyles
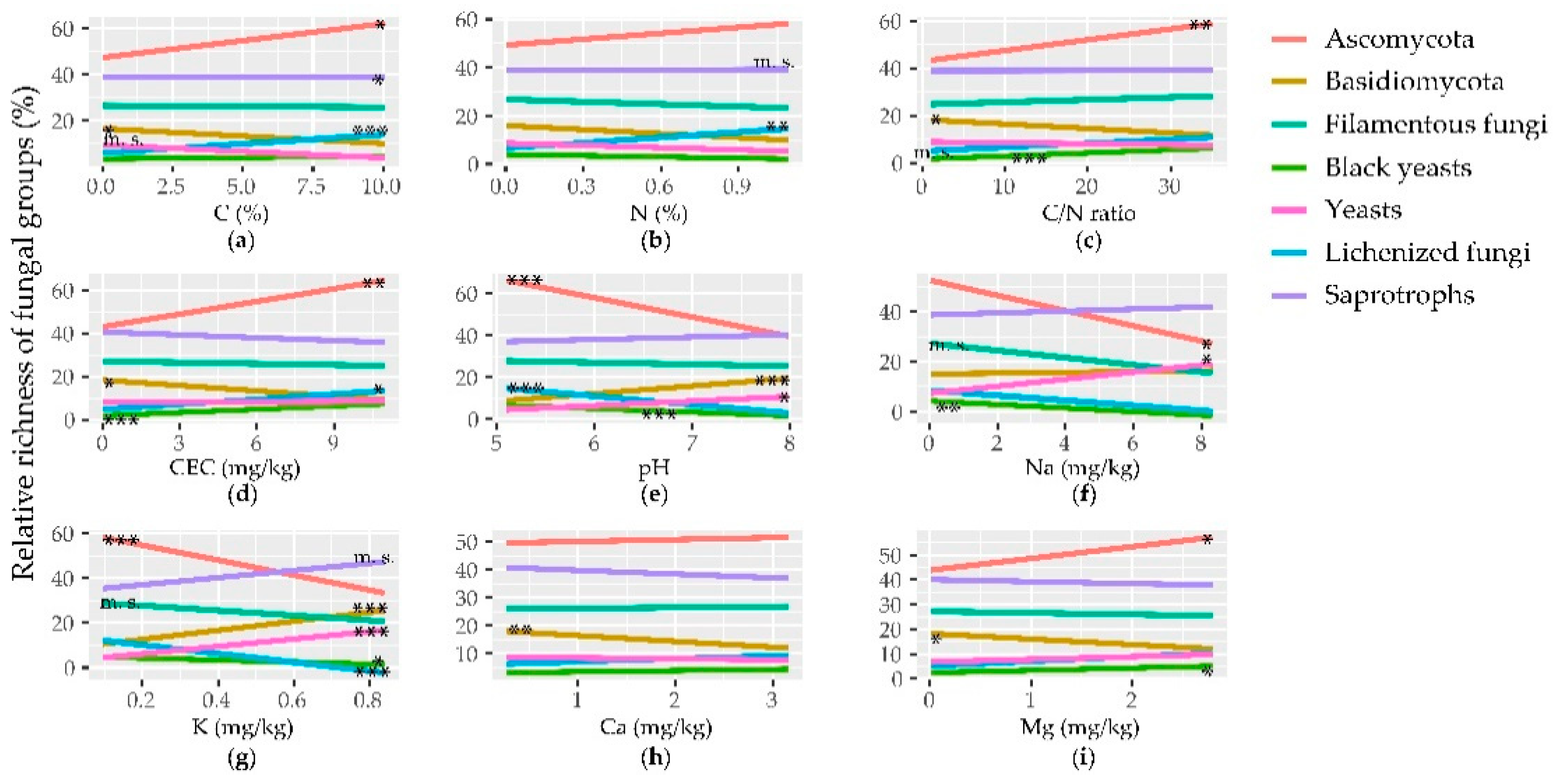
3.3. Correlation between Fungal Richness/Abundance and Edaphic Parameters
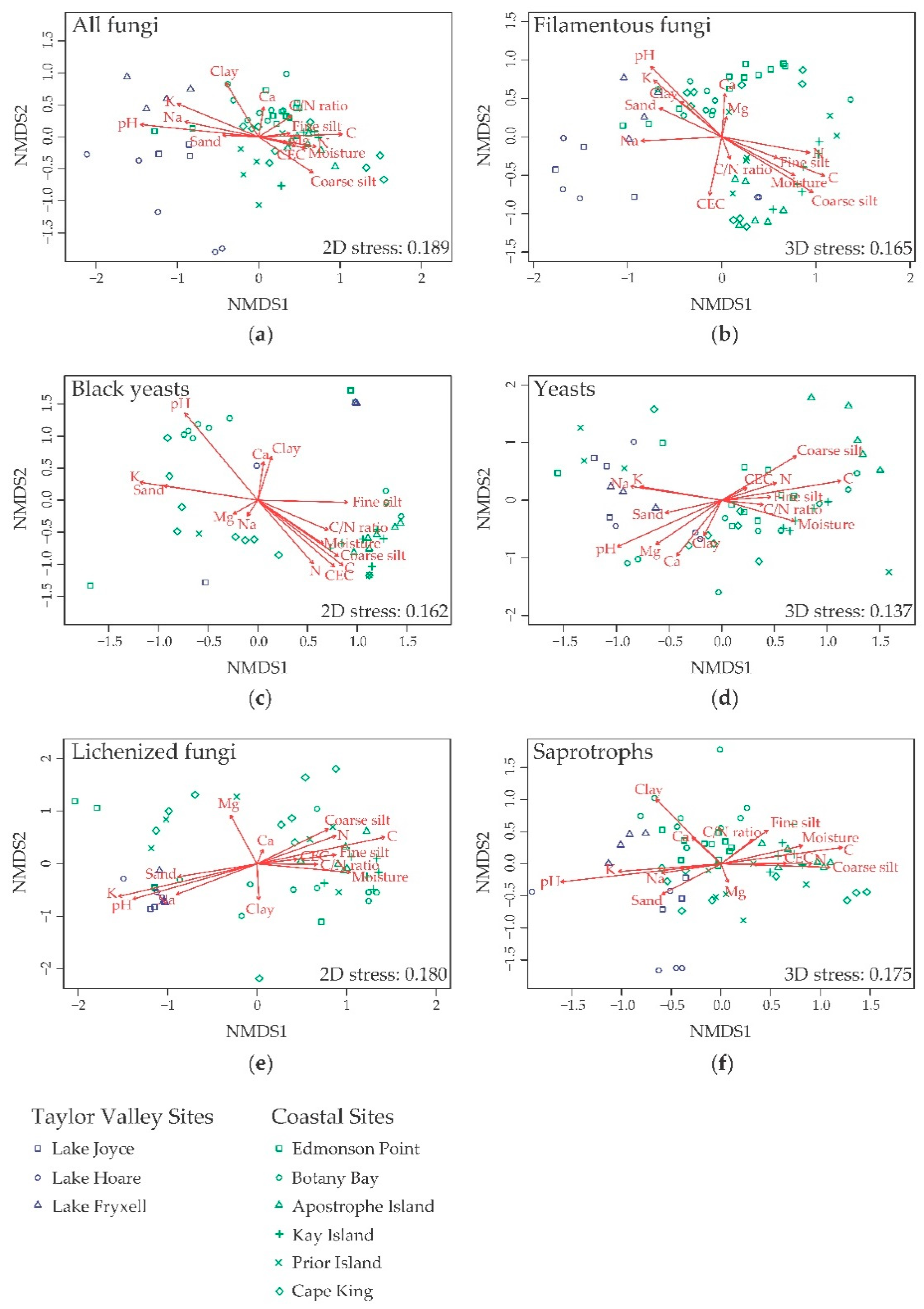
3.4. Effect of Edaphic Parameters on Community and Groups Composition
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Campbell, I.B.; Claridge, G.G.C. Salts in Antarctic Soils. In Antarctica: Soils, Weathering Processes and Environment; Campbell, I.B., Claridge, G.G.C., Eds.; Elsevier: Amsterdam, The Netherlands, 1987; pp. 239–273. [Google Scholar]
- Tsilinsky, P. Sur les mucédinées thermophiles. Ann. l’Istitute Pasteur 1899, 13, 500–505. [Google Scholar]
- Tsilinsky, P. La flore microbienne dans les regions du Pole Sud. In Expedition Antarctique Française 1903–1905; Masson et C.le Ed: Paris, France, 1908; Volume 3, 33p. [Google Scholar]
- Bommer, E.; Rousseau, M. Note preliminaire sur les champignons recueillis par l’Expedition Antarctique Belge 1897–1899. Bull. Acad. R Sci. Belgiq. Clas. Sci. 1900, 8, 640–646. [Google Scholar]
- Flint, E.A.; Stout, J.D. Microbiology of some soils from Antarctica. Nature 1960, 188, 767–768. [Google Scholar] [CrossRef] [PubMed]
- Tubaki, K. Notes on some fungi and yeasts from Antarctica. Antarct. Rec. Ser. E 1961, 11, 161–162. [Google Scholar]
- Tubaki, K. On some fungi isolated from Antarctic materials. Biological Results of the Japanese Antarctic Research Expedition 14. Spec. Publ. Seto Mar. Biol. Lab. 1961, 1, 1–9. [Google Scholar]
- Tubaki, K.; Asano, I. Additional species of fungi isolated from the Antarctic materials. Sci. Rep. Jpn. Antarct. Res. Exped. 1965, 27–28, 1–12. [Google Scholar]
- Ugolini, F.C.; Starkey, R.L. Soils and micro-organisms from Mount Erebus, Antarctica. Nature 1966, 211, 440–441. [Google Scholar] [CrossRef]
- Duarte, A.W.F.; Centurion, V.B.; Oliveira, V.M. Uncultivated Fungi from Antarctica. In Fungi of Antarctica; Rosa, L.H., Ed.; Springer: Cham, Switzerland, 2019; pp. 19–41. [Google Scholar]
- Godinho, V.M.; Gonçalves, V.N.; Santiago, I.F.; Figueredo, H.M.; Vitoreli, G.A.; Schaefer, C.E.; Barbosa, E.C.; Oliveira, J.G.; Alves, T.M.A.; Zani, C.L.; et al. Diversity and bioprospection of fungal community present in oligotrophic soil of continental Antarctica. Extremophiles 2015, 19, 585–596. [Google Scholar] [CrossRef]
- Ashok, A.; Doriya, K.; Rao, J.V.; Qureshi, A.; Tiwari, A.K.; Kumar, D.S. Microbes Producing L-Asparaginase free of Glutaminase and Urease isolated from Extreme Locations of Antarctic Soil and Moss. Sci. Rep. 2019, 9, 1423. [Google Scholar] [CrossRef]
- Rosa, L.H.; Zani, C.L.; Cantrell, C.L.; Duke, S.O.; Van Dijck, P.; Desideri, A.; Rosa, C.A. Fungi in Antarctica: Diversity, ecology, effects of climate change, and bioprospection for bioactive compounds. In Fungi of Antarctica; Rosa, L.H., Ed.; Springer: Cham, Switzerland, 2019; pp. 1–17. [Google Scholar]
- Vaca, I.; Chávez, R. Bioactive compounds produced by Antarctic filamentous fungi. In Fungi of Antarctica; Rosa, L.H., Ed.; Springer: Cham, Switzerland, 2019; pp. 265–283. [Google Scholar]
- Zucconi, L.; Onofri, S.; Cecchini, C.; Isola, D.; Ripa, C.; Fenice, M.; Madonna, S.; Reboleiro-Rivas, P.; Selbmann, L. Mapping the lithic colonization at the boundaries of life in Northern Victoria Land, Antarctica. Polar Biol. 2016, 39, 91–102. [Google Scholar] [CrossRef]
- Hogg, I.D.; Cary, S.C.; Convey, P.; Newsham, K.K.; O’Donnell, A.G.; Adams, B.J.; Aislabie, J.; Frati, F.; Stevens, M.I.; Wall, D.H. Biotic interactions in Antarctic terrestrial ecosystems: Are they a factor? Soil Biol. Biochem. 2006, 38, 3035–3040. [Google Scholar] [CrossRef]
- Van Goethem, M.W.; Makhalanyane, T.P.; Valverde, A.; Cary, S.C.; Cowan, D.A. Characterization of bacterial communities in lithobionts and soil niches from Victoria Valley, Antarctica. FEMS Microbiol. Ecol. 2016, 92, fiw051. [Google Scholar] [CrossRef] [PubMed]
- Durán, P.; Barra, P.J.; Jorquera, M.A.; Viscardi, S.; Fernandez, C.; Mora, M.L.; Bol, R. Occurrence of soil fungi in Antarctic pristine environments. Front. Bioeng. Biotechnol. 2019, 7, 28. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.K.; Laughlin, D.C.; Bottos, E.M.; Caruso, T.; Joy, K.; Barrett, J.E.; Brabyn, L.; Nielsen, U.N.; Adams, B.J.; Wall, D.H.; et al. Biotic interactions are an unexpected yet critical control on the complexity of an abiotically driven polar ecosystem. Commun. Biol. 2019, 2, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Canini, F.; Geml, J.; D’Acqui, L.P.; Selbmann, L.; Onofri, S.; Ventura, S.; Zucconi, L. Exchangeable cations and pH drive diversity and functionality of fungal communities in biological soil crusts from coastal sites of Victoria Land, Antarctica. Fungal Ecol. 2020, 45, 100923. [Google Scholar] [CrossRef]
- Canini, F.; Geml, J.; D’Acqui, L.P.; Buzzini, P.; Turchetti, B.; Onofri, S.; Ventura, S.; Zucconi, L. Fungal diversity and functionality are driven by soil texture in Taylor Valley, Antarctica. Fungal Ecol. 2021, 50, 101041. [Google Scholar] [CrossRef]
- Lawley, B.; Ripley, S.; Bridge, P.; Convey, P. Molecular analysis of geographic patterns of eukaryotic diversity in Antarctic soils. Appl. Environ. Microbiol. 2004, 70, 5963–5972. [Google Scholar] [CrossRef]
- Howard-Williams, C.; Hawes, I.; Gordon, S. The environmental basis of ecosystem variability in Antarctica: Research in the Latitudinal Gradient Project. Antarct. Sci. 2010, 22, 591–602. [Google Scholar] [CrossRef]
- Czechowski, P.; Clarke, L.J.; Breen, J.; Cooper, A.; Stevens, M.I. Antarctic eukaryotic soil diversity of the Prince Charles Mountains revealed by high-throughput sequencing. Soil Biol. Biochem. 2016, 95, 112–121. [Google Scholar] [CrossRef]
- Cowan, D.A.; Makhalanyane, T.P.; Dennis, P.G.; Hopkins, D.W. Microbial ecology and biogeochemistry of continental Antarctic soils. Front. Microbiol. 2014, 5, 154. [Google Scholar] [CrossRef]
- Onofri, S.; Zucconi, L.; Tosi, S. Continental Antarctic Fungi, 1st ed.; IHW: München, Germany, 2007. [Google Scholar]
- Rippin, M.; Lange, S.; Sausen, N.; Becker, B. Biodiversity of biological soil crusts from the Polar Regions revealed by metabarcoding. FEMS Microbiol. Ecol. 2018, 94, fiy036. [Google Scholar] [CrossRef]
- Belnap, J.; Büdel, B.; Lange, O.L. Biological soil crusts: Characteristics and distribution. In Biological Soil Crusts: Structure, Function, and Management, 1st ed.; Belnap, J., Lange, O.L., Eds.; Springer: Berlin/Heidelberg, Germany, 2001; pp. 3–30. [Google Scholar]
- Pushkareva, E.; Pessi, I.S.; Namsaraev, Z.; Mano, M.J.; Elster, J.; Wilmotte, A. Cyanobacteria inhabiting biological soil crusts of a polar desert: Sør Rondane Mountains, Antarctica. Syst. Appl. Microbiol. 2018, 41, 363–373. [Google Scholar] [CrossRef] [PubMed]
- Peterson, D.; Howard-Williams, C. The Latitudinal Gradient Project; Antarctica New Zealand: Christchurch, New Zealand, 2001. [Google Scholar]
- Fountain, A.G.; Nylen, T.H.; Monaghan, A.; Basagic, H.J.; Bromwich, D. Snow in the McMurdo Dry Valleys, Antarctica. Int. J. Climatol. 2010, 30, 633–642. [Google Scholar] [CrossRef]
- Buzzini, P.; Turk, M.; Perini, L.; Turchetti, B.; Gunde-Cimerman, N. Yeasts in Polar and Subpolar Habitats. In Yeasts in Natural Ecosystems: Diversity, 1st ed.; Buzzini, P., Lachance, M.A., Yurkov, A., Eds.; Springer: Cham, Switzerland, 2017; pp. 331–366. [Google Scholar]
- Sannino, C.; Tasselli, G.; Filippucci, S.; Turchetti, B.; Buzzini, P. Yeasts in Nonpolar Cold Habitats. In Yeasts in Natural Ecosystems: Diversity, 1st ed.; Buzzini, P., Lachance, M.A., Yurkov, A., Eds.; Springer: Cham, Switzerland, 2017; pp. 367–396. [Google Scholar]
- De Hoog, G.S.; McGinnis, M.R. Ascomycetous black yeasts. Stud. Mycol. 1987, 30, 187–199. [Google Scholar]
- Sterflinger, K. Black yeasts and meristematic fungi: Ecology, diversity and identification. In Biodiversity and Ecophysiology of Yeasts, 1st ed.; Péter, G., Rosa, C., Eds.; Springer: Berlin/Heidelberg, Germany, 2006; pp. 501–514. [Google Scholar]
- Ruisi, S.; Barreca, D.; Selbmann, L.; Zucconi, L.; Onofri, S. Fungi in Antarctica. Rev. Environ. Sci. Biotechnol. 2007, 6, 127–141. [Google Scholar] [CrossRef]
- Palmer, J.M.; Jusino, M.A.; Banik, M.T.; Lindner, D.L. Non-biological synthetic spike-in controls and the AMPtk software pipeline improve mycobiome data. PeerJ 2018, 6, e4925. [Google Scholar] [CrossRef] [PubMed]
- Edgar, R.C. Search and clustering orders of magnitude faster than BLAST. Bioinformatics 2010, 26, 2460–2461. [Google Scholar] [CrossRef]
- Rognes, T.; Flouri, T.; Nichols, B.; Quince, C.; Mahé, F. VSEARCH: A versatile open source tool for metagenomics. PeerJ 2016, 4, e2584. [Google Scholar] [CrossRef]
- Nguyen, N.H.; Song, Z.; Bates, S.T.; Branco, S.; Tedersoo, L.; Menke, J.; Schilling, J.S.; Kennedy, P.G. FUNGuild: An open annotation tool for parsing fungal community datasets by ecological guild. Fungal Ecol. 2016, 20, 241–248. [Google Scholar] [CrossRef]
- Oksanen, J.; Blanchet, F.G.; Friendly, M.; Kindt, R.; Legendre, P.; McGlinn, D.; Minchin, P.R.; O’Hara, R.B.; Simpson, G.L.; Solymos, P.; et al. Vegan: Community Ecology Package. R Package 2018, Version 2.5-2. Available online: https://CRAN.R-project:package=vegan (accessed on 25 March 2021).
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2018. [Google Scholar]
- Anderson, M.J. A new method for non-parametric multivariate analysis of variance. Austral Ecol. 2001, 26, 32–46. [Google Scholar] [CrossRef]
- Selbmann, L.; Onofri, S.; Coleine, C.; Buzzini, P.; Canini, F.; Zucconi, L. Effect of environmental parameters on biodiversity of the fungal component in lithic Antarctic communities. Extremophiles 2017, 21, 1069–1080. [Google Scholar] [CrossRef] [PubMed]
- Coleine, C.; Stajich, J.E.; Zucconi, L.; Onofri, S.; Pombubpa, N.; Egidi, E.; Franks, A.; Buzzini, P.; Selbmann, L. Antarctic Cryptoendolithic Fungal Communities Are Highly Adapted and Dominated by Lecanoromycetes and Dothideomycetes. Front. Microbiol. 2018, 9, 1392. [Google Scholar] [CrossRef] [PubMed]
- De Menezes, G.C.; Amorim, S.S.; Gonçalves, V.N.; Godinho, V.M.; Simões, J.C.; Rosa, C.A.; Rosa, L.H. Diversity, Distribution, and Ecology of Fungi in the Seasonal Snow of Antarctica. Microorganisms 2019, 7, 445. [Google Scholar] [CrossRef] [PubMed]
- Connell, L.; Staudigel, H. Fungal diversity in a dark oligotrophic volcanic ecosystem (DOVE) on Mount Erebus, Antarctica. Biology 2013, 2, 798–809. [Google Scholar] [CrossRef]
- Goordial, J.; Davila, A.; Lacelle, D.; Pollard, W.; Marinova, M.M.; Greer, C.W.; DiRuggiero, J.; McKay, C.P.; Whyte, L.G. Nearing the cold-arid limits of microbial life in permafrost of an upper dry valley, Antarctica. ISME J. 2016, 10, 1613–1624. [Google Scholar] [CrossRef]
- Isola, D.; Zucconi, L.; Onofri, S.; Caneva, G.; de Hoog, S.; Selbmann, L. Extremotolerant rock inhabiting black fungi from Italian monumental sites. Fungal Divers. 2016, 76, 75–96. [Google Scholar] [CrossRef]
- Antonelli, F.; Esposito, A.; Calvo, L.; Licursi, V.; Tisseyre, P.; Ricci, S.; Romagnoli, M.; Piazza, S.; Guerrieri, F. Characterization of black patina from the Tiber River embankments using Next-Generation Sequencing. PLoS ONE 2020, 15, e0227639. [Google Scholar] [CrossRef]
- Coleine, C.; Pombubpa, N.; Zucconi, L.; Onofri, S.; Stajich, J.E.; Selbmann, L. Endolithic Fungal Species Markers for Harshest Conditions in the McMurdo Dry Valleys, Antarctica. Life 2020, 10, 13. [Google Scholar] [CrossRef]
- Egidi, E.; de Hoog, G.S.; Isola, D.; Onofri, S.; Quaedvlieg, W.; de Vries, M.; Verkley, G.J.M.; Stielow, J.B.; Zucconi, L.; Selbmann, L. Phylogeny and taxonomy of meristematic rock-inhabiting black fungi in the dothidemycetes based on multi-locus phylogenies. Fungal Divers. 2014, 65, 127–165. [Google Scholar] [CrossRef]
- Pacelli, C.; Selbmann, L.; Zucconi, L.; Coleine, C.; de Vera, J.P.; Rabbow, E.; Böttger, U.; Dadachova, E.; Onofri, S. Responses of the Black Fungus Cryomyces antarcticus to Simulated Mars and Space Conditions on Rock Analogs. Astrobiology 2019, 19, 209–220. [Google Scholar] [CrossRef]
- Bottos, E.M.; Woo, A.C.; Zawar-Reza, P.; Pointing, S.B.; Cary, S.C. Airborne Bacterial populations above desert soils of the McMurdo Dry Valleys, Antarctica. Microb. Ecol. 2014, 67, 120–128. [Google Scholar] [CrossRef] [PubMed]
- Archer, S.D.; Lee, K.C.; Caruso, T.; Maki, T.; Lee, C.K.; Cary, S.C.; Cowan, D.A.; Maestre, F.T.; Pointing, S.B. Airborne microbial transport limitation to isolated Antarctic soil habitats. Nat. Microbiol. 2019, 4, 925–932. [Google Scholar] [CrossRef] [PubMed]
- Jones, T.C.; Hogg, I.D.; Wilkins, R.J.; Green, T.G.A. Microsatellite analyses of the Antarctic endemic lichen Buellia frigida Darb. (Physciaceae) suggest limited dispersal and the presence of glacial refugia in the Ross Sea region. Polar Biol. 2015, 38, 941–949. [Google Scholar] [CrossRef]
- Santiago, I.F.; Soares, M.A.; Rosa, C.A.; Rosa, L.H. Lichensphere: A protected natural microhabitat of the non-lichenised fungal communities living in extreme environments of Antarctica. Extremophiles 2015, 19, 1087–1097. [Google Scholar] [CrossRef]
- Connell, L.B.; Redman, R.; Rodriguez, R.; Barrett, A.; Iszard, M.; Fonseca, A. Dioszegia antarctica sp. nov. and Dioszegia cryoxerica sp. nov., psychrophilic basidiomycetous yeasts from polar desert soils in Antarctica. Int. J. Syst. Evol. Microbiol. 2010, 60, 1466–1472. [Google Scholar] [CrossRef] [PubMed]
- Fell, J.W.; Statzell, A.C.; Hunter, I.L.; Phaff, H.J. Leucosporidium gen. n., the heterobasidiomycetous stage of several yeasts of the genus Candida. Antonie Leeuwenhoek 1969, 35, 433–462. [Google Scholar] [CrossRef]
- Vishniac, H.S. Cryptococcus friedmannii, a new species of yeast from the Antarctic. Mycologia 1985, 77, 149–153. [Google Scholar] [CrossRef]
- Branda, E.; Turchetti, B.; Diolaiuti, G.; Pecci, M.; Smiraglia, C.; Buzzini, P. Yeast and yeast-like diversity in the southernmost glacier of Europe (Calderone Glacier, Apennines, Italy). FEMS Microbiol. Ecol. 2010, 72, 354–369. [Google Scholar] [CrossRef]
- Butinar, L.; Strmole, T.; Gunde-Cimerman, N. Relative incidence of ascomycetous yeasts in arctic coastal environments. Microb. Ecol. 2011, 61, 832–843. [Google Scholar] [CrossRef]
- Buzzini, P.; Turchetti, B.; Yurkov, A. Extremophilic yeasts: The toughest yeasts around? Yeast 2018, 35, 487–497. [Google Scholar] [CrossRef]
- Fowler, M.S.; Lindström, J. Extinctions in simple and complex communities. Oikos 2002, 99, 511–517. [Google Scholar] [CrossRef]
- Wedin, M.; Döring, H.; Gilenstam, G. Saprotrophy and lichenization as options for the same fungal species on different substrata: Environmental plasticity and fungal lifestyles in the Stictis–Conotrema complex. New Phytol. 2004, 164, 459–465. [Google Scholar] [CrossRef]
- Selbmann, L.; de Hoog, G.S.; Zucconi, L.; Isola, D.; Onofri, S. Black Yeasts in Cold Habitats. In Yeasts from Cold Habitats, 1st ed.; Buzzini, P., Margesin, R., Eds.; Springer: Berlin/Heidelberg, Germany, 2014; pp. 173–189. [Google Scholar]
- Coleine, C.; Zucconi, L.; Onofri, S.; Pombubpa, N.; Stajich, J.E.; Selbmann, L. Sun Exposure Shapes Functional Grouping of Fungi in Cryptoendolithic Antarctic Communities. Life 2018, 8, 19. [Google Scholar] [CrossRef] [PubMed]
- Newsham, K.K.; Davey, M.L.; Hopkins, D.W.; Dennis, P.G. Regional diversity of maritime Antarctic soil fungi and predicted responses of guilds and growth forms to climate change. Front. Microbiol. 2021, 11, 3575. [Google Scholar] [CrossRef] [PubMed]
- Connell, L.; Redman, R.; Craig, S.; Rodriguez, R. Distribution and abundance of fungi in the soils of Taylor Valley, Antarctica. Soil Biol. Biochem. 2006, 38, 3083–3094. [Google Scholar] [CrossRef]
- Newsham, K.K.; Garnett, M.H.; Robinson, C.H.; Cox, F. Discrete taxa of saprotrophic fungi respire different ages of carbon from Antarctic soils. Sci. Rep. 2018, 8, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Hemkemeyer, M.; Dohrmann, A.B.; Christensen, B.T.; Tebbe, C.C. Bacterial preferences for specific soil particle size fractions revealed by community analyses. Front. Microbiol. 2018, 9, 149. [Google Scholar] [CrossRef]
- Almela, P.; Justel, A.; Quesada, A. Heterogeneity of microbial communities in soils from the Antarctic Peninsula region. Front. Microbiol. 2021, 12, 280. [Google Scholar] [CrossRef]
- Clem, K.R.; Fogt, R.L.; Turner, J.; Lintner, B.R.; Marshall, G.J.; Miller, J.R.; Renwick, J.A. Record warming at the South Pole during the past three decades. Nat. Clim. Chang. 2020, 10, 762–770. [Google Scholar] [CrossRef]
- Buelow, H.N.; Winter, A.S.; Van Horn, D.J.; Barrett, J.E.; Gooseff, M.N.; Schwartz, E.; Takacs-Vesbach, C.D. Microbial community responses to increased water and organic matter in the arid soils of the McMurdo Dry Valleys, Antarctica. Front. Microbiol. 2016, 7, 1040. [Google Scholar] [CrossRef]

| All FUNGI | Black YEASTS | Filamentous FUNGI | All YEASTS | Ascomycetous YEASTS | Basidiomycetous YEASTS | Lichenized FUNGI | Saprotrophs | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Par | V | p | Par | V | p | Par | V | p | Par | V | p | Par | V | p | Par | V | p | Par | V | p | Par | V | p |
| pH | 7.93 | 0.0001 | pH | 18.9 | 0.0001 | pH | 9.676 | 0.0001 | pH | 8.23 | 0.0001 | Clay | 23.186 | 0.0001 | pH | 7.91 | 0.0001 | K | 13.239 | 0.0001 | pH | 8.774 | 0.0001 |
| C | 3.555 | 0.0002 | C | 4.678 | 0.0002 | C | 4.621 | 0.0001 | Moist | 6.038 | 0.0002 | Fs | 4.75 | 0.0301 | K | 3.904 | 0.017 | pH | 5.534 | 0.0004 | C | 3.459 | 0.0002 |
| Cs | 4.284 | 0.0001 | K | 7.263 | 0.0001 | Cs | 4.264 | 0.0001 | K | 3.774 | 0.0052 | Sand | 4.126 | 0.0562 | Moist | 5.672 | 0.0007 | C | 5.293 | 0.0002 | K | 3.946 | 0.0001 |
| K | 3.687 | 0.0001 | Cs | 1.493 | 0.1663 | K | 3.851 | 0.0001 | Sand | 5.532 | 0.0005 | Ca | 1.578 | 0.413 | C/N | 2.838 | 0.0802 | Cs | 2.551 | 0.0319 | Cs | 3.653 | 0.0002 |
| Na | 3.259 | 0.0003 | N | 1.784 | 0.0858 | Moist | 3.073 | 0.0002 | Fs | 3.676 | 0.0093 | Moist | 1.803 | 0.3874 | Na | 2.563 | 0.1102 | Moist | 3.182 | 0.0072 | CEC | 4.256 | 0.0001 |
| Moist | 2.815 | 0.0009 | Clay | 7.541 | 0.0001 | CEC | 4.744 | 0.0001 | C | 0.872 | 0.7834 | K | 3.412 | 0.1036 | Ca | 4.781 | 0.0045 | Na | 2.003 | 0.0651 | Clay | 5.097 | 0.0001 |
| Sand | 3.957 | 0.0002 | CEC | 3.173 | 0.0051 | Ca | 4.323 | 0.0001 | Cs | 1.713 | 0.2634 | Na | 7.937 | 0.0013 | Fs | 3.004 | 0.0653 | Mg | 6.968 | 0.0001 | Moist | 2.881 | 0.0004 |
| Clay | 3.103 | 0.0004 | Ca | 6.085 | 0.0002 | N | 1.91 | 0.0306 | Na | 4.142 | 0.0015 | pH | 4.516 | 0.0404 | R | 69.329 | N | 1.798 | 0.1495 | Ca | 2.534 | 0.002 | |
| Fs | 5.76 | 0.0001 | Sand | 4.523 | 0.0002 | Mg | 5.374 | 0.0001 | Clay | 3.752 | 0.0071 | R | 48.692 | Sand | 4.858 | 0.0005 | Na | 3.337 | 0.0001 | ||||
| N | 1.482 | 0.1115 | Fs | 4.343 | 0.0005 | Sand | 2.333 | 0.0061 | Ca | 4.092 | 0.0048 | Fs | 3.287 | 0.0066 | N | 1.625 | 0.064 | ||||||
| CEC | 4.21 | 0.0001 | Na | 2.151 | 0.0329 | Clay | 4.677 | 0.0001 | R | 58.179 | Ca | 0.714 | 0.8647 | Mg | 4.757 | 0.0001 | |||||||
| Ca | 4.314 | 0.0001 | Moist | 2.042 | 0.0587 | Fs | 2.06 | 0.0106 | Clay | 2.669 | 0.0271 | Sand | 5.489 | 0.0001 | |||||||||
| Mg | 2.797 | 0.0005 | Mg | 0.739 | 0.6523 | C/N | 1.566 | 0.1026 | R | 47.905 | Fs | 2.58 | 0.0009 | ||||||||||
| C/N | 1.572 | 0.0805 | C/N | 0.83 | 0.5776 | R | 47.53 | C/N | 1.35 | 0.1885 | |||||||||||||
| R | 47.312 | R | 34.454 | R | 46.261 | ||||||||||||||||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Canini, F.; Geml, J.; Buzzini, P.; Turchetti, B.; Onofri, S.; D’Acqui, L.P.; Ripa, C.; Zucconi, L. Growth Forms and Functional Guilds Distribution of Soil Fungi in Coastal Versus Inland Sites of Victoria Land, Antarctica. Biology 2021, 10, 320. https://doi.org/10.3390/biology10040320
Canini F, Geml J, Buzzini P, Turchetti B, Onofri S, D’Acqui LP, Ripa C, Zucconi L. Growth Forms and Functional Guilds Distribution of Soil Fungi in Coastal Versus Inland Sites of Victoria Land, Antarctica. Biology. 2021; 10(4):320. https://doi.org/10.3390/biology10040320
Chicago/Turabian StyleCanini, Fabiana, József Geml, Pietro Buzzini, Benedetta Turchetti, Silvano Onofri, Luigi Paolo D’Acqui, Caterina Ripa, and Laura Zucconi. 2021. "Growth Forms and Functional Guilds Distribution of Soil Fungi in Coastal Versus Inland Sites of Victoria Land, Antarctica" Biology 10, no. 4: 320. https://doi.org/10.3390/biology10040320
APA StyleCanini, F., Geml, J., Buzzini, P., Turchetti, B., Onofri, S., D’Acqui, L. P., Ripa, C., & Zucconi, L. (2021). Growth Forms and Functional Guilds Distribution of Soil Fungi in Coastal Versus Inland Sites of Victoria Land, Antarctica. Biology, 10(4), 320. https://doi.org/10.3390/biology10040320










