Increased Circulating H3 Histone in Response to Repeated Bouts of Exercise Does Not Associate with Parallel Alterations of Cell-Free DNA
Abstract
Simple Summary
Abstract
1. Introduction
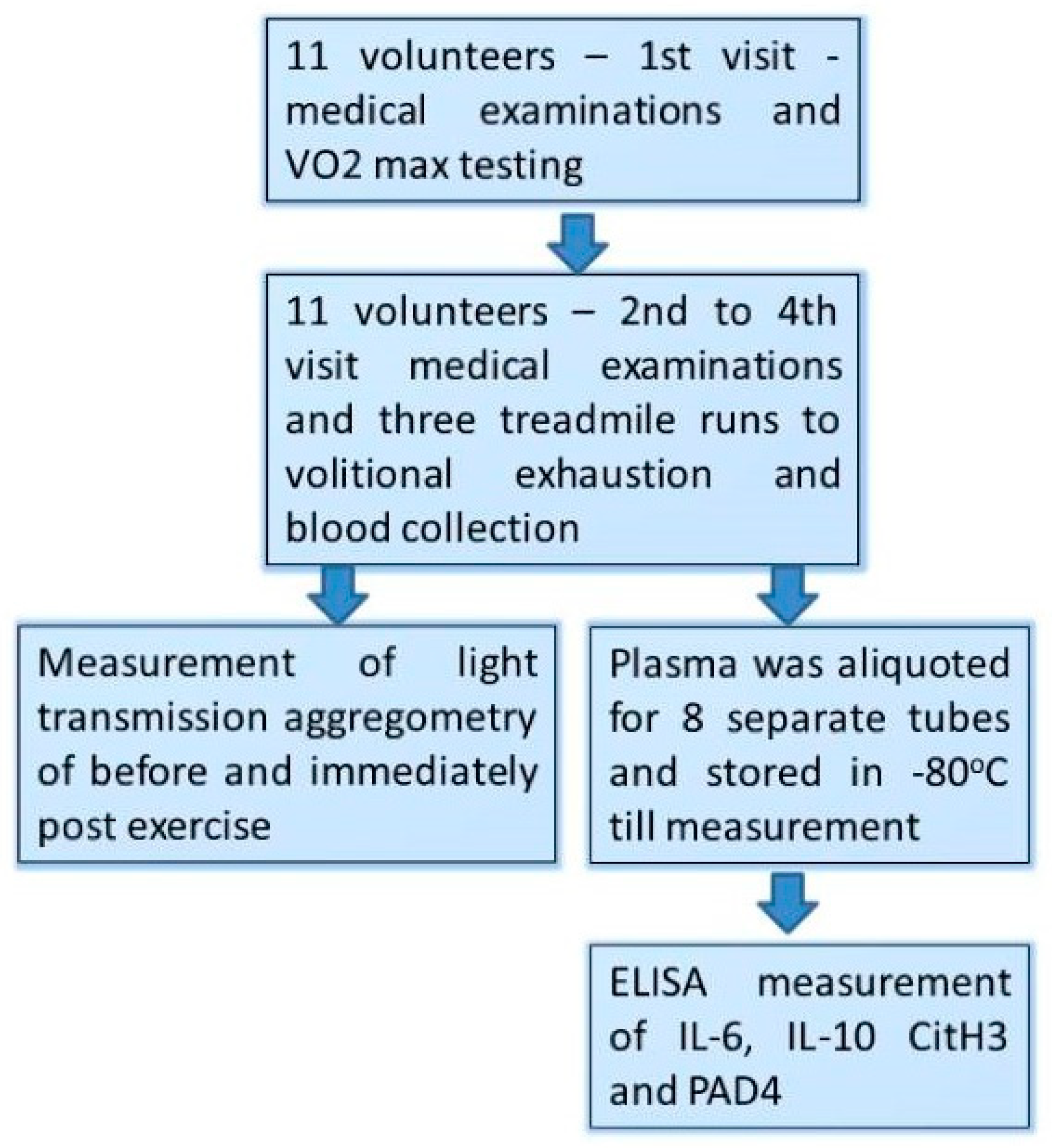
2. Materials and Methods
2.1. Studied Group
2.2. The Study Design
- (1)
- Those male volunteers who fulfilled the inclusion/exclusion criteria underwent a treadmill VO2max test at the first visit (day 1st).
- (2)
- At the three consecutive visits separated by 72 h of resting period (day 7th, 10th, and 13th), participants performed a treadmill exercise to exhaustion at speed matching to 70% of their personal VO2max.
- (3)
- Venous blood (2.7 mL) was collected into vacutainer tubes with EDTA and sodium citrate (Becton Dickinson, Franklin Lakes, NJ, USA) before and just after each bout of exhaustive exercise.
- (4)
- Light Transmission Aggregometry (LTA) was executed immediately after the blood collection, while the obtained plasma was aliquoted, frozen, and stored at −80 °C until the measurements.
2.3. Variables Measured with ELISA Test
2.4. Platelets Function Measurement
2.5. Other Variables
2.6. Statistical Analysis
3. Results
4. Discussion
Study Limitations
- (1)
- Our study has several limitations, a relatively small number of subjects, and hence the inability to divide participants into subgroups (well-trained/untrained volunteers, female/male).
- (2)
- Second, the lack of kinetic analysis makes it impossible to confirm the trend of citrullinated histones or explain the mechanism that disrupts the relationship between circulating histone and cfDNA.
- (3)
- Finally, since CitH3 ELISA kit detects both DNA bound and free histones this may result in uncertainty in some observations.
5. Conclusions
- (1)
- This is the first study showing that the level of circulating histone and PAD4 protein increases in exhaustive exercises. Moreover, the presence of circulating histones in post-exercise serum might confirm the increase of NETosis process during exercise.
- (2)
- Blood aggregation status and interleukin expression increase in response to each bout of strenuous exercise.
- (3)
- Despite the parallel increase, no significant correlation between citrullinated histone or blood aggregation was found. However, positive correlations of cf nDNA with blood aggregation, and lactate with blood aggregation, and lactate with citrullinated histone have been observed.
- (4)
- Although each bout caused an increase in histones, all parameters normalized three days after the treadmill run. However, histones showed a downward trend in their increment.
- (5)
- The increment of H3Cit is relatively low comparing to the other diseases what might suggest limited exercise induced histone cytotoxicity.
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| PAD4 | peptidylarginine deiminase 4 |
| nDNA | Nuclear Deoxyribonucleic acid |
| NETs | Neutrophil extracellular traps |
| H3Cit | Citrullinated histone H3 |
| IL | Interleukin |
| LTA | Light Transmission Aggregometry |
| CVD | Cardiovascular disease |
| DAMPs | Damage-associated molecular patterns |
| HMGB1 | High Mobility Group Box 1 |
| MPO | Myeloperoxidase |
| ADP | Adenosine diphosphate |
| CK | Creatine kinase |
| AST | Aspartate aminotransferase |
| ALT | Alanine aminotransferase |
| CRP | C-reactive protein |
References
- Abrams, S.T.; Zhang, N.; Manson, J.; Liu, T.; Dart, C.; Baluwa, F.; Wang, S.S.; Brohi, K.; Kipar, A.; Yu, W.; et al. Circulating histones are mediators of trauma-associated lung injury. Am. J. Respir. Crit. Care Med. 2013, 187, 160–169. [Google Scholar] [CrossRef]
- Monach, P.A.; Hueber, W.; Kessler, B.; Tomooka, B.H.; BenBarak, M.; Simmons, B.P.; Wright, J.; Thornhill, T.S.; Monestier, M.; Ploegh, H.; et al. A broad screen for targets of immune complexes decorating arthritic joints highlights deposition of nucleosomes in rheumatoid arthritis. Proc. Natl. Acad. Sci. USA 2009, 106, 15867–15872. [Google Scholar] [CrossRef] [PubMed]
- Borissoff, J.I.; Joosen, I.A.; Versteylen, M.O.; Brill, A.; Fuchs, T.A.; Savchenko, A.S.; Gallant, M.; Martinod, K.; Ten Cate, H.; Hofstra, L.; et al. Elevated levels of circulating DNA and chromatin are independently associated with severe coronary atherosclerosis and a prothrombotic state. Arterioscler. Thromb. Vasc. Biol. 2013, 33, 2032–2040. [Google Scholar] [CrossRef]
- Xu, J.; Zhang, X.; Pelayo, R.; Monestier, M.; Ammollo, C.T.; Semeraro, F.; Taylor, F.B.; Esmon, N.L.; Lupu, F.; Esmon, C.T. Extracellular histones are major mediators of death in sepsis. Nat. Med. 2009, 15, 1318–1321. [Google Scholar] [CrossRef] [PubMed]
- Zuo, Y.; Yalavarthi, S.; Shi, H.; Gockman, K.; Zuo, M.; Madison, J.A.; Blair, C.; Weber, A.; Barnes, B.J.; Egeblad, M.; et al. Neutrophil extracellular traps (NETs) as markers of disease severity in COVID-19. medRxiv 2020. [Google Scholar] [CrossRef]
- Beiter, T.; Fragasso, A.; Hartl, D.; Niess, A.M. Neutrophil extracellular traps: A walk on the wild side of exercise immunology. Sport. Med. 2015, 45, 625–640. [Google Scholar] [CrossRef]
- Hojman, P.; Brolin, C.; Nørgaard-Christensen, N.; Dethlefsen, C.; Lauenborg, B.; Olsen, C.K.; Åbom, M.M.; Krag, T.; Gehl, J.; Pedersen, B.K. IL-6 release from muscles during exercise is stimulated by lactate-dependent protease activity. Am. J. Physiol. Endocrinol. Metab. 2019, 316, E940–E947. [Google Scholar] [CrossRef]
- Tsourouktsoglou, T.D.; Warnatsch, A.; Ioannou, M.; Hoving, D.; Wang, Q.; Papayannopoulos, V. Histones, DNA, and Citrullination Promote Neutrophil Extracellular Trap Inflammation by Regulating the Localization and Activation of TLR4. Cell Rep. 2020, 31, 107602. [Google Scholar] [CrossRef]
- Wang, Y.; Li, M.; Stadler, S.; Correll, S.; Li, P.; Wang, D.; Hayama, R.; Leonelli, L.; Han, H.; Grigoryev, S.A.; et al. Histone hypercitrullination mediates chromatin decondensation and neutrophil extracellular trap formation. J. Cell Biol. 2009, 184, 205–213. [Google Scholar] [CrossRef] [PubMed]
- Neeli, I.; Dwivedi, N.; Khan, S.; Radic, M. Regulation of extracellular chromatin release from neutrophils. J. Innate Immun. 2009, 1, 194–201. [Google Scholar] [CrossRef]
- Stawski, R.; Walczak, K.; Kosielski, P.; Meissner, P.; Budlewski, T.; Padula, G.; Nowak, D. Repeated bouts of exhaustive exercise increase circulating cell free nuclear and mitochondrial DNA without development of tolerance in healthy men. PLoS ONE 2017, 12, e0178216. [Google Scholar] [CrossRef]
- Beiter, T.; Fragasso, A.; Hudemann, J.; Nieß, A.M.; Simon, P. Short-Term Treadmill Running as a Model for Studying Cell-Free DNA Kinetics In Vivo. Clin. Chem. 2011, 57, 633–636. [Google Scholar] [CrossRef]
- Beiter, T.; Fragasso, A.; Hudemann, J.; Schild, M.; Steinacker, J.; Mooren, F.C.; Niess, A.M. Neutrophils release extracellular DNA traps in response to exercise. J. Appl. Physiol. 2014, 117, 325–333. [Google Scholar] [CrossRef]
- Haller, N.; Helmig, S.; Taenny, P.; Petry, J.; Schmidt, S.; Simon, P. Circulating, cell-free DNA as a marker for exercise load in intermittent sports. PLoS ONE 2018, 13, e0191915. [Google Scholar] [CrossRef]
- Vittori, L.N.; Tarozzi, A.; Latessa, P.M. Circulating Cell-Free DNA in Physical Activities. Methods Mol. Biol. 2019, 1909, 183–197. [Google Scholar] [CrossRef] [PubMed]
- Stawski, R.; Walczak, K.; Perdas, E.; Wlodarczyk, A.; Sarniak, A.; Kosielski, P.; Meissner, P.; Budlewski, T.; Padula, G.; Nowak, D. Decreased integrity of exercise-induced plasma cell free nuclear DNA—negative association with the increased oxidants production by circulating phagocytes. Sci. Rep. 2019, 9, 15970. [Google Scholar] [CrossRef] [PubMed]
- Gmiat, A.; Mieszkowski, J.; Prusik, K.; Prusik, K.; Kortas, J.; Kochanowicz, A.; Radulska, A.; Lipiński, M.; Tomczyk, M.; Jaworska, J.; et al. Changes in pro-inflammatory markers and leucine concentrations in response to Nordic Walking training combined with vitamin D supplementation in elderly women. Biogerontology 2017, 18, 535–548. [Google Scholar] [CrossRef] [PubMed]
- Collier, D.M.; Villalba, N.; Sackheim, A.; Bonev, A.D.; Miller, Z.D.; Moore, J.S.; Shui, B.; Lee, J.C.; Lee, F.K.; Reining, S.; et al. Extracellular histones induce calcium signals in the endothelium of resistance-sized mesenteric arteries and cause loss of endothelium-dependent dilation. Am. J. Physiol. Heart Circ. Physiol. 2019, 316, H1309–H1322. [Google Scholar] [CrossRef] [PubMed]
- Michiels, C. Endothelial cell functions. J. Cell. Physiol. 2003, 196, 430–443. [Google Scholar] [CrossRef] [PubMed]
- Elaskalani, O.; Abdol Razak, N.B.; Metharom, P. Neutrophil extracellular traps induce aggregation of washed human platelets independently of extracellular DNA and histones. Cell Commun. Signal. 2018, 16, 24. [Google Scholar] [CrossRef] [PubMed]
- Nomura, K.; Miyashita, T.; Yamamoto, Y.; Munesue, S.; Harashima, A.; Takayama, H.; Fushida, S.; Ohta, T. Citrullinated Histone H3: Early Biomarker of Neutrophil Extracellular Traps in Septic Liver Damage. J. Surg. Res. 2019, 234, 132–138. [Google Scholar] [CrossRef] [PubMed]
- Silk, E.; Zhao, H.; Weng, H.; Ma, D. The role of extracellular histone in organ injury. Cell Death Dis. 2017, 8, e2812. [Google Scholar] [CrossRef] [PubMed]
- Yamamura, M.; Yamada, Y.; Momita, S.; Kamihira, S.; Tomonaga, M. Circulating interleukin-6 levels are elevated in adult T-cell leukaemia/lymphoma patients and correlate with adverse clinical features and survival. Br. J. Haematol. 1998, 100, 129–134. [Google Scholar] [CrossRef] [PubMed]
- Born, G.V. Aggregation of blood platelets by adenosine diphosphate and its reversal. Nature 1962, 194, 927–929. [Google Scholar] [CrossRef]
- Perdas, E.; Stawski, R.; Kaczka, K.; Nowak, D.; Zubrzycka, M. Altered levels of circulating nuclear and mitochondrial DNA in patients with Papillary Thyroid Cancer. Sci. Rep. 2019, 9, 14438. [Google Scholar] [CrossRef] [PubMed]
- Gauthier, V.J.; Tyler, L.N.; Mannik, M. Blood clearance kinetics and liver uptake of mononucleosomes in mice. J. Immunol. 1996, 156, 1151–1156. [Google Scholar] [PubMed]
- Qaddoori, Y.; Abrams, S.T.; Mould, P.; Alhamdi, Y.; Christmas, S.E. Extracellular Histones Inhibit Complement Activation through Interacting with Complement Component 4. J. Immunol. 2018, 200, 4125–4133. [Google Scholar] [CrossRef]
- Masuda, S.; Nakazawa, D.; Shida, H.; Miyoshi, A.; Kusunoki, Y.; Tomaru, U.; Ishizu, A. NETosis markers: Quest for specific, objective, and quantitative markers. Clin. Chim. Acta 2016, 459, 89–93. [Google Scholar] [CrossRef]
- Shi, Y.; Shi, H.; Nieman, D.C.; Hu, Q.; Yang, L.; Liu, T.; Zhu, X.; Wei, H.; Wu, D.; Li, F.; et al. Lactic Acid Accumulation During Exhaustive Exercise Impairs Release of Neutrophil Extracellular Traps in Mice. Front. Physiol. 2019, 10. [Google Scholar] [CrossRef]
- Paues Göranson, S.; Thålin, C.; Lundström, A.; Hållström, L.; Lasselin, J.; Wallén, H.; Soop, A.; Mobarrez, F. Circulating H3Cit is elevated in a human model of endotoxemia and can be detected bound to microvesicles. Sci. Rep. 2018, 8, 12641. [Google Scholar] [CrossRef] [PubMed]
- Nakahara, M.; Ito, T.; Kawahara, K.; Yamamoto, M.; Nagasato, T.; Shrestha, B.; Yamada, S.; Miyauchi, T.; Higuchi, K.; Takenaka, T.; et al. Recombinant thrombomodulin protects mice against histone-induced lethal thromboembolism. PLoS ONE 2013, 8, e75961. [Google Scholar] [CrossRef]
- Marijon, E.; Uy-Evanado, A.; Reinier, K.; Teodorescu, C.; Narayanan, K.; Jouven, X.; Gunson, K.; Jui, J.; Chugh, S.S. Sudden cardiac arrest during sports activity in middle age. Circulation 2015, 131, 1384–1391. [Google Scholar] [CrossRef]
- Tozzi-Ciancarelli, M.G.; Penco, M.; Di Massimo, C. Influence of acute exercise on human platelet responsiveness: Possible involvement of exercise-induced oxidative stress. Eur. J. Appl. Physiol. 2002, 86, 266–272. [Google Scholar] [CrossRef]
- Aldemir, H.; Kiliç, N. The effect of time of day and exercise on platelet functions and platelet–neutrophil aggregates in healthy male subjects. Mol. Cell. Biochem. 2005, 280, 119–124. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.S.; Jen, C.J.; Kung, H.C.; Lin, L.J.; Hsiue, T.R.; Chen, H.I. Different effects of strenuous exercise and moderate exercise on platelet function in men. Circulation 1994, 90, 2877–2885. [Google Scholar] [CrossRef] [PubMed]
- Golyshenkov, S.P.; Mel′nikova, N.A.; Lapshina, M.V. The Effect of Exercise on Platelet Aggregability and Lipid Peroxidation. Hum. Physiol. 2004, 30, 708–713. [Google Scholar] [CrossRef]
- Gould, T.J.; Vu, T.T.; Swystun, L.L.; Dwivedi, D.J.; Mai, S.H.; Weitz, J.I.; Liaw, P.C. Neutrophil extracellular traps promote thrombin generation through platelet-dependent and platelet-independent mechanisms. Arterioscler. Thromb. Vasc. Biol. 2014, 34, 1977–1984. [Google Scholar] [CrossRef]
- Fuchs, T.A.; Brill, A.; Duerschmied, D.; Schatzberg, D.; Monestier, M.; Myers, D.D., Jr.; Wrobleski, S.K.; Wakefield, T.W.; Hartwig, J.H.; Wagner, D.D. Extracellular DNA traps promote thrombosis. Proc. Natl. Acad. Sci. USA 2010, 107, 15880–15885. [Google Scholar] [CrossRef]
- Gleeson, M.; Bishop, N.C.; Stensel, D.J.; Lindley, M.R.; Mastana, S.S.; Nimmo, M.A. The anti-inflammatory effects of exercise: Mechanisms and implications for the prevention and treatment of disease. Nat. Rev. Immunol. 2011, 11, 607–615. [Google Scholar] [CrossRef] [PubMed]
- Ostapiuk-Karolczuk, J.; Zembron-Lacny, A.; Naczk, M.; Gajewski, M.; Kasperska, A.; Dziewiecka, H.; Szyszka, K. Cytokines and cellular inflammatory sequence in non-athletes after prolonged exercise. J. Sport. Med. Phys. Fit. 2012, 52, 563–568. [Google Scholar]
- Danesh, J.; Kaptoge, S.; Mann, A.G.; Sarwar, N.; Wood, A.; Angleman, S.B.; Wensley, F.; Higgins, J.P.; Lennon, L.; Eiriksdottir, G.; et al. Long-term interleukin-6 levels and subsequent risk of coronary heart disease: Two new prospective studies and a systematic review. PLoS Med. 2008, 5, e78. [Google Scholar] [CrossRef] [PubMed]
- Thålin, C.; Lundström, S.; Seignez, C.; Daleskog, M.; Lundström, A.; Henriksson, P.; Helleday, T.; Phillipson, M.; Wallén, H.; Demers, M. Citrullinated histone H3 as a novel prognostic blood marker in patients with advanced cancer. PLoS ONE 2018, 13, e0191231. [Google Scholar] [CrossRef]
| Parameter | Bouts of Exhaustive Treadmill Exercise | |||||
|---|---|---|---|---|---|---|
| 1st Bout | 2nd Bout | 3rd Bout | ||||
| Before | After | Before | After | Before | After | |
| H3Cit ng/mL | 3.08 ± 2.9 (1.96 ± 2.9) | 5.96 ± 3.0 * (4.63 ± 4.4) | 3.65 ± 3.5 (2.23 ± 3.4) | 6.37 ± 5.2 (4.29 ± 5.4) | 3.86 ± 3.1 (2.57 ± 4.4) † | 4.75 ± 3.04 * (5.05 ± 5.7) |
| PAD4 ng/mL | 2.03 ± 3.2 (0.68 ± 0.6) | 3.08 ± 3.7 * (1.40 ± 1.3) | 1.91 ± 3.4 (0.41 ± 0.6) | 3.53 ± 4.1 * (1.42 ± 1.8) | 2.38 ± 3.6 (0.67 ± 1.6) | 3.25 ± 3.8 * (1.67 ± 2.4) |
| Parameter | Bouts of Exhaustive Treadmill Exercise | |||||
|---|---|---|---|---|---|---|
| 1st Bout | 2nd Bout | 3rd Bout | ||||
| Before | After | Before | After | Before | After | |
| LTA U/min | 40.8 ± 14.7 (37.2 ± 15.5) | 55.0 ± 14.3 * (56.7 ± 11.7) | 33.3 ± 16.8 (31.1 ± 24.6) | 57.0 ± 14.7 * (63.1 ± 17.2) | 39.3 ± 12.4 (35.3 ± 13.3) | 56.43 ± 14.77 * (57.05 ± 7.68) |
| Parameter | Bouts of Exhaustive Treadmill Exercise | |||||
|---|---|---|---|---|---|---|
| 1st Bout | 2nd Bout | 3rd Bout | ||||
| Before | After | Before | After | Before | After | |
| Il-6 (pg/mL) | 1 ± 0 (1 ± 0) † | 8.44 ± 12.84 * (2.43 ± 6.27) | 1 ± 0 (1 ± 0) † | 8.12 ± 9.59 (2.49 ± 6.31) | 1 ± 0 (1 ± 0) † | 7.28 ± 7.54 * (3.01 ± 6.3) |
| IL-10 (pg/mL) | 2.63 ± 1.25 (2.66 ± 1.2) | 6.81 ± 6.27 * (4.18 ± 5.56) | 2.34 ± 1.13 (2.23 ± 0.26) | 6.99 ± 11.09 * (2.77 ± 1.62) | 2.02 ± 1.04 (2.08 ± 1.51) | 6.04 ± 6.59 * (2.37 ± 6.1) |
| Spearman Rang Correlation Variables | Correlation Coefficient (r) Spearman R | p-Value |
|---|---|---|
| H3Cit vs. PAD4 | 0.786 | (below 0.000005) |
| IL-10 vs. IL-6 | 0.374 | (p = 0.0021) |
| H3Cit vs. Aggregation | 0.157 | (ns) |
| H3Cit vs. IL-6 | 0.312 | (p = 0.0078) |
| PAD4 vs. IL6 | 0.485 | (p = 0.000036) |
| H3Cit vs. IL-10 | 0.207 | (ns) |
| Aggregation vs. IL-6 | 0.409 | (p = 0.00049) |
| Aggregation vs. IL-10 | 0.051 | (ns) |
| H3Cit vs. cf nDNA * | 0.224 | (ns) |
| PAD4 vs. cf nDNA | 0.348 | (p = 0.0041) |
| PAD4 vs. Aggregation | 0.487 | (p = 0.000092) |
| Aggregation vs. cf nDNA * | ||
| Aggregation vs. Lactate * | 0.412 | (p = 0.00058) |
| H3Cit vs. Lactate * | 0.266 | (p = 0.030) |
| PAD4 vs. Lactate * | 0.369 | (p = 0.0023) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Stawski, R.; Walczak, K.; Perdas, E.; Prymont-Przymińska, A.; Zwolińska, A.; Kosielski, P.; Budlewski, T.; Padula, G.; Jerczynska, H.; Nowak, D. Increased Circulating H3 Histone in Response to Repeated Bouts of Exercise Does Not Associate with Parallel Alterations of Cell-Free DNA. Biology 2021, 10, 181. https://doi.org/10.3390/biology10030181
Stawski R, Walczak K, Perdas E, Prymont-Przymińska A, Zwolińska A, Kosielski P, Budlewski T, Padula G, Jerczynska H, Nowak D. Increased Circulating H3 Histone in Response to Repeated Bouts of Exercise Does Not Associate with Parallel Alterations of Cell-Free DNA. Biology. 2021; 10(3):181. https://doi.org/10.3390/biology10030181
Chicago/Turabian StyleStawski, Robert, Konrad Walczak, Ewelina Perdas, Anna Prymont-Przymińska, Anna Zwolińska, Piotr Kosielski, Tomasz Budlewski, Gianluca Padula, Hanna Jerczynska, and Dariusz Nowak. 2021. "Increased Circulating H3 Histone in Response to Repeated Bouts of Exercise Does Not Associate with Parallel Alterations of Cell-Free DNA" Biology 10, no. 3: 181. https://doi.org/10.3390/biology10030181
APA StyleStawski, R., Walczak, K., Perdas, E., Prymont-Przymińska, A., Zwolińska, A., Kosielski, P., Budlewski, T., Padula, G., Jerczynska, H., & Nowak, D. (2021). Increased Circulating H3 Histone in Response to Repeated Bouts of Exercise Does Not Associate with Parallel Alterations of Cell-Free DNA. Biology, 10(3), 181. https://doi.org/10.3390/biology10030181







