Resistance of Injection Molded Wood-Polypropylene Composites against Basidiomycetes According to EN 15534-1: New Insights on the Test Procedure, Structural Alterations, and Impact of Wood Source
Abstract
1. Introduction
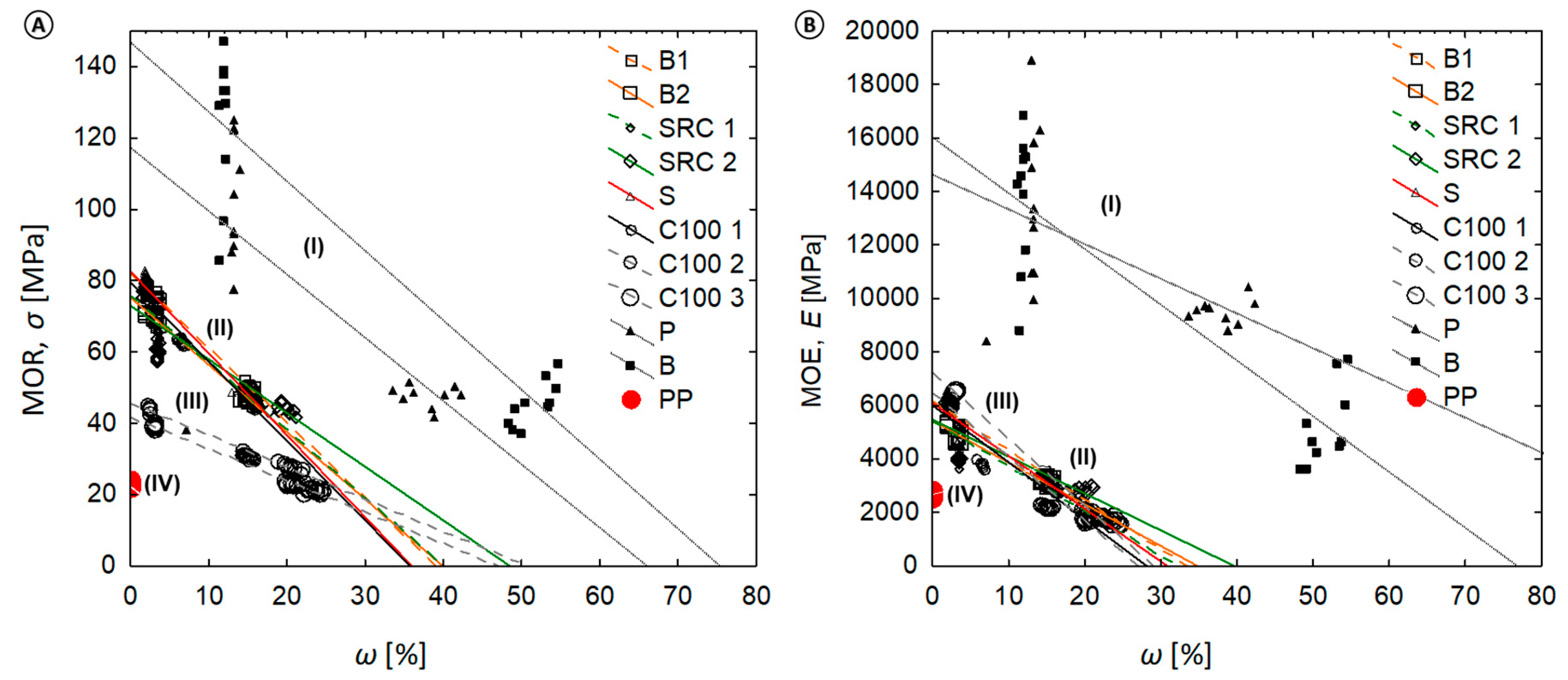
- (I)
- Evaluation of the relative loss in mechanical properties caused by the test procedure such as moisture and fungi;
- (II)
- Examine the relationship between moisture content (ω)/mass loss and modulus of rupture (MOR)/modulus of elasticity (MOE);
- (III)
- Study the impact of wood source on decay resistance; and
- (IV)
- Study the impact of fungal decay on the internal composite structure.
2. Materials and Methods
2.1. Processing and Wood-Polypropylene Composite Manufacturing
2.2. Sample Preparation and Fungal Test
2.3. Structural Characterization
3. Results
3.1. Mass Loss of Decayed Specimens
3.2. Structural Characteristics and Alterations of Decayed Specimens
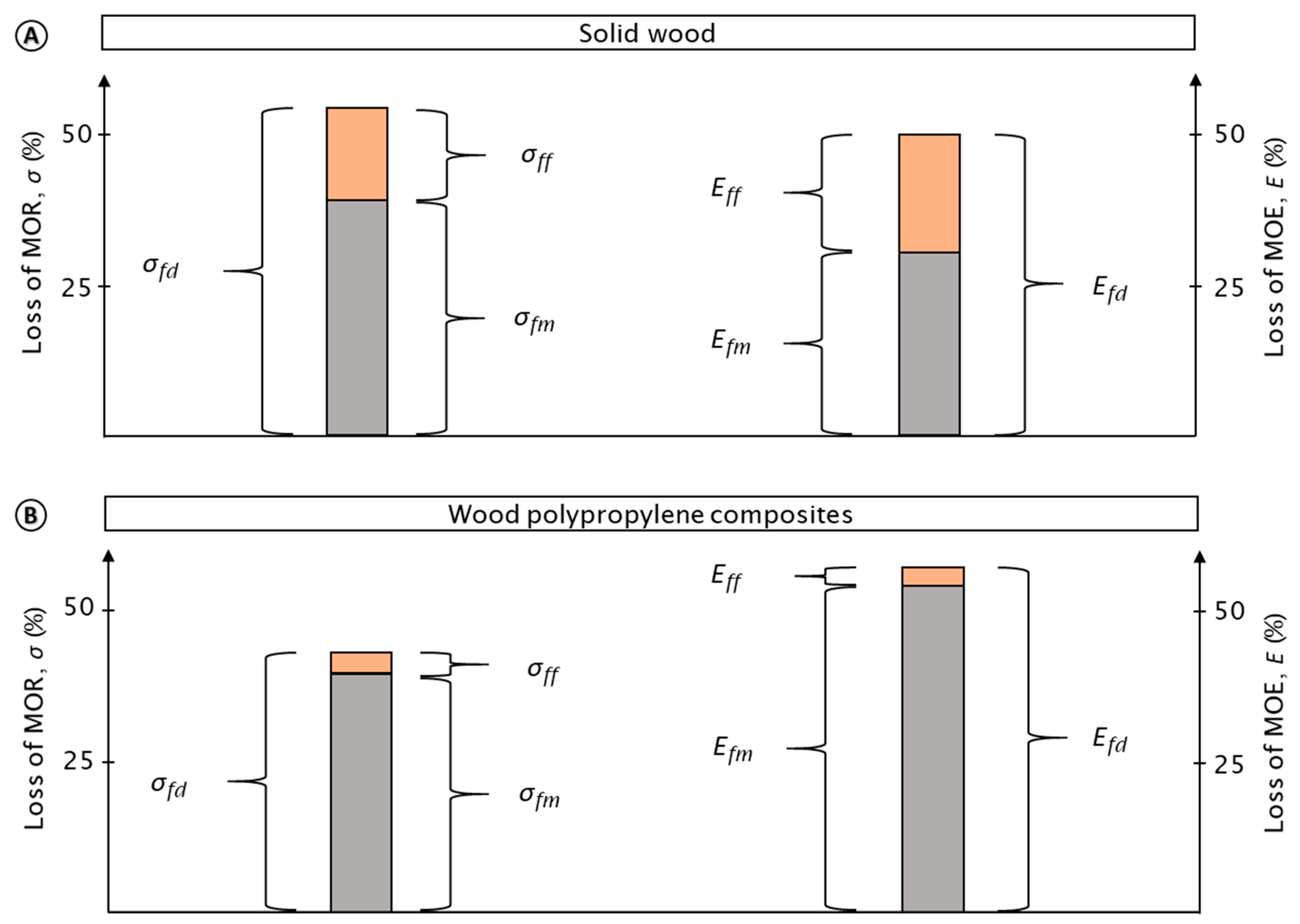
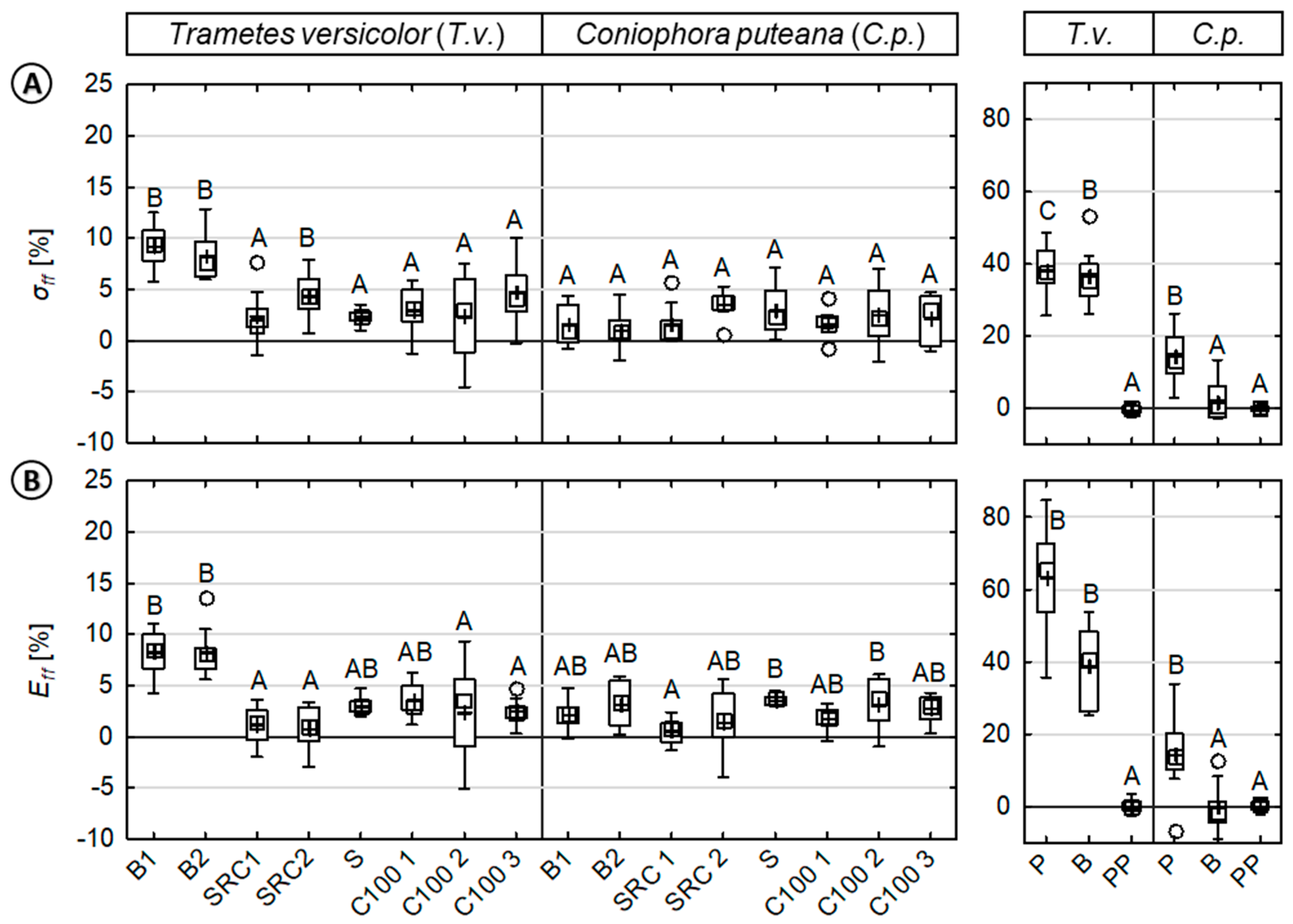
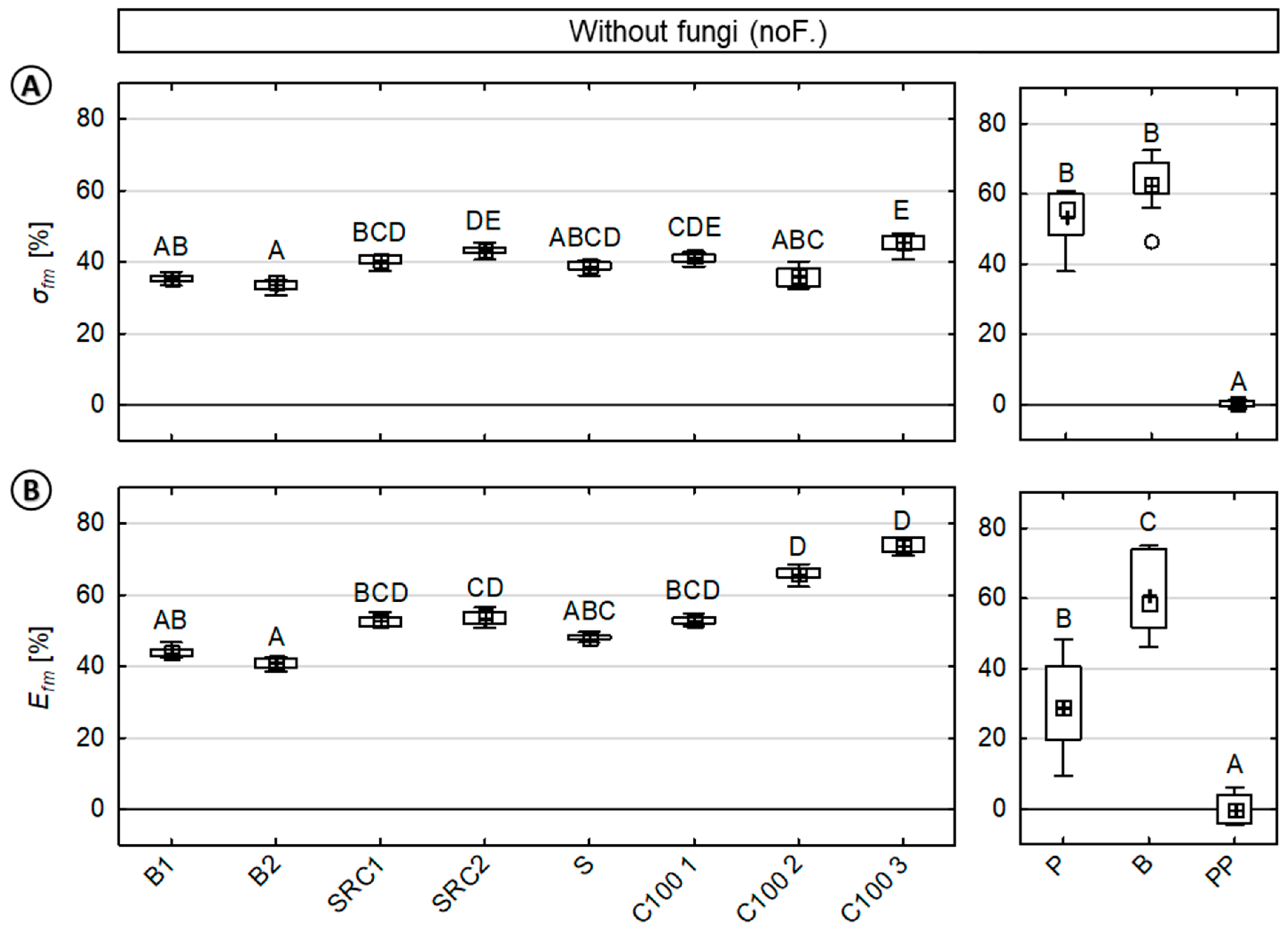
3.3. Fungal Test Procedure—Effect of Moisture and Fungi on Mechanical Properties
3.4. Impact of Different Wood Sources on Decay Resistance
3.5. Relationship between Moisture Content (ω %) and Modulus of Rupture (MOR) or Modulus of Elasticity (MOE)
4. Discussion
4.1. Fungal Test Procedure—Effect of Moisture and Fungi
4.2. Correlation between Moisture Content (MC) and MOR, MOE
4.3. The Effect of Wood Particle Source and Wood Species
4.4. Structural Characterization
5. Conclusions
- ▪
- the test procedure is suitable to test injection molded WPC;
- ▪
- moisture is key and wood–moisture interactions play a vital role when accessing different WPC variants and fungal infestation;
- ▪
- after preconditioning, the wood moisture content of many injection molded WPC is still below fiber saturation;
- ▪
- mass losses up to 3.7% (wood mass losses up to 5.3%) were determined for WPC;
- ▪
- wood mass loss is a valuable parameter for the actual degradation of wooden material caused by fungi within polyolefin based composites;
- ▪
- moisture content is strongly correlated with the mechanical properties of WPC;
- ▪
- structural analysis via XµCT provides valuable insights into the degradation pattern of wood decay fungi;
- ▪
- calcium oxalates were present in decayed WPC;
- ▪
- more data and further research on the fungal test procedure for WPC are required;
- ▪
- potential fields of interest are the impact of preconditioning, specimen size and shape, and fungi species; and
- ▪
- open issues are the informative value of mass losses and the classification to durability classes validated by a round robin test.
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Strohmeyer, A.; Walther, H. Overview of the German Wood-Plastic Composite Industry. In Proceedings of the 7th Biocomposites Conference Cologne, Cologne, Germany, 6 December 2017. [Google Scholar]
- Schirp, A.; Ibach, R.E.; Pendleton, D.E.; Wolcott, M.P. Biological degradation of Wood-Plastic Composites (WPC) and strategies for improving the resistance of WPC against biological decay. In ACS Symposium Series (982); American Chemical Society: Washington, DC, USA, 2008; pp. 480–507. ISBN 978-0-8412-3951-7. [Google Scholar]
- Sun, G.; Ibach, R.E.; Faillace, M.; Gnatowski, M.; Glaeser, J.A.; Haight, J. Laboratory and exterior decay of wood-plastic composite boards: Voids analysis and computed tomography. Wood Mater. Sci. Eng. 2017, 12, 263–278. [Google Scholar] [CrossRef]
- Ibach, R.; Gnatowski, M.; Sun, G.; Glaeser, J.A.; Leung, M.; Haight, J. Laboratory and environmental decay of wood-plastic composite boards: Flexural properties. Wood Mater. Sci. Eng. 2018, 13, 81–96. [Google Scholar] [CrossRef]
- Krause, A.; Gellerich, A. Evaluating durability of thermoplastic wood composites against basidiomycetes and development of a suitable test design. Wood Mater. Sci. Eng. 2014, 9, 179–185. [Google Scholar] [CrossRef]
- European Committee for Standardization (CEN). Composites Made from Cellulose-Based Materials and Thermoplastics (Usually Called Wood-Polymer Composites (WPC) or Natural Fibre Composites (NFC))—Part 1: Test Methods for Characterisation of Compounds and Products, EN 15534-1, German version: DIN EN 15534-1; European Committee for Standardization: Brussels, Belgium, 2014. [Google Scholar]
- European Committee for Standardization (CEN). Wood Preservatives—Accelerated Ageing of Treated Wood Prior to Biological Testing—Leaching Procedure; European Committee for Standardization: Brussels, Belgium, 1997. [Google Scholar]
- Mankowski, M.; Morrell, J.J. Patterns of fungal attack in wood-plastic composites following exposure in a soil block test. Wood Fiber Sci. 2000, 32, 340–345. [Google Scholar]
- Kim, J.W.; Harper, D.P.; Taylor, A.M. Effect of wood species on water sorption and durability of wood-plastic composites. Wood Fiber Sci. 2008, 40, 519–531. [Google Scholar]
- Dritte Bundeswaldinventur (BWi3)—Third National Forest Inventory. Available online: https://bwi.info (accessed on 6 June 2018).
- Mantau, U.; Prins, K.; Lindner, M.; Verkerk, H.; Eggers, J.; Leel, N.; Oldenburger, J.; Asikainer, A.; Anttila, P. Real Potential for Changes in Growth and Use of EU Forests. Methodology Report. Available online: https://www.ab.gov.tr/files/ardb/evt/1_avrupa_birligi/1_9_politikalar/1_9_6_enerji_politikasi/euwood_methodology_report.pdf (accessed on 6 June 2018).
- Krause, K.C.; Müller, M.; Krause, A. Converting wood from short rotation coppice and low-value beech wood into thermoplastic composites. In Bioenergy from Dendromass for the Sustainable Development of Rural Areas; Wiley-Vch: Weinheim, Germany, 2015; pp. 483–496. ISBN 978-3-527-68298-0. [Google Scholar]
- Krause, K.C.; Müller, M.; Militz, H.; Krause, A. Enhanced water resistance of extruded wood–polypropylene composites based on alternative wood sources. Eur. J. Wood Prod. 2017, 75, 125–134. [Google Scholar] [CrossRef]
- European Committee for Standardization (CEN). Durability of Wood and Wood-Based Products. Wood-Based Panels: Method of Test for Determining the Resistance Against Wood-Destroying Basidiomycetes; European Committee for Standardization: Brussels, Belgium, 2002. [Google Scholar]
- Koddenberg, T.; Krause, K.C.; Krause, A. Tomographic analysis of siliceous particulates in Australian turpentine wood (Syncarpia glomulifera) through X-ray micro-computed tomography. Micron 2019, 117, 22–28. [Google Scholar] [CrossRef] [PubMed]
- Schirp, A.; Wolcott, M.P. Influence of fungal decay and moisture absorption on mechanical properties of extruded wood-plastic composites. Wood Fiber Sci. 2005, 37, 643–652. [Google Scholar]
- Defoirdt, N.; Gardin, S.; Van den Bulcke, J.; Van Acker, J. Moisture dynamics of WPC and the impact on fungal testing. Int. Biodeterior. Biodegrad. 2010, 64, 65–72. [Google Scholar] [CrossRef][Green Version]
- Morris, P.I.; Cooper, P. Recycled plastic/wood composite lumber attacked by fungi. For. Prod. J. 1998, 48, 86–88. [Google Scholar]
- Fabiyi, J.S.; McDonald, A.G. Degradation of polypropylene in naturally and artificially weathered plastic matrix composites. Maderas Cienc. Tecnol. 2014, 16, 275–290. [Google Scholar] [CrossRef]
- Bay, R.S.; Tucker, C.L. Fiber orientation in simple injection moldings. Part I: Theory and numerical methods. Polym. Compos. 1992, 13, 317–331. [Google Scholar] [CrossRef]
- Bay, R.S.; Tucker, C.L. Fiber orientation in simple injection moldings. Part II: Experimental results. Polym. Compos. 1992, 13, 332–341. [Google Scholar] [CrossRef]
- Bledzki, A.K.; Faruk, O. Injection moulded microcellular wood fibre-polypropylene composites. Compos. Part A Appl. Sci. Manuf. 2006, 37, 1358–1367. [Google Scholar] [CrossRef]
- Correa, R.A.; Nunes, R.C.R.; Filho, W.Z.F. Effect of injections on the orientation of short fibre composites. An optical microscopic analysis. Polym. Test. 1996, 15, 467–475. [Google Scholar] [CrossRef]
- Mertens, O. Performance and Processing Evaluation of Thermoplastic Wood Fiber Composites. Master’s Thesis, Universität Hamburg, Hamburg, Germany, 2018. [Google Scholar]
- Bourmaud, A.; Ausias, G.; Lebrun, G.; Baley, C.; Tachon, M.L. Observation of the structure of a composite polypropylene/flax and damage mechanisms under stress. Ind. Crops Prod. 2013, 43, 225–236. [Google Scholar] [CrossRef]
- Peters, S.T. Handbook of Composites, 2nd ed.; Springer: Boston, MA, USA, 1998; ISBN 978-0-412-54020-2. [Google Scholar]
- Khavkine, M.; Kazayawoko, M.; Law, S.; Balatinecz, J.J. Durability of Wood Flour-Thermoplastic Composites under Extreme Environmental Conditions and Fungal Exposure. Int. J. Polym. Mater. 2000, 46, 255–269. [Google Scholar] [CrossRef]
- Segerholm, B.K.; Ibach, R.E.; Westin, M. Moisture sorption, biological durability, and mechanical performance of WPC containing modified wood and polylactates. BioRes 2012, 7, 4575–4585. [Google Scholar] [CrossRef]
- Candelier, K.; Atli, A.; Alteyrac, J. Termite and decay resistance of bioplast-spruce green wood-plastic composites. Eur. J. Wood Prod. 2019, 77, 157–169. [Google Scholar] [CrossRef]
- Clemons, C.M.; Ibach, R.E. Application of Laboratory Fungal Resistance Tests to Solid Wood and Wood-Plastic Composites. Plast. Build. Constr. 2003, 27, 7–14. [Google Scholar]
- Sobczak, L.; Lang, R.W.; Haider, A. Polypropylene composites with natural fibers and wood—General mechanical property profiles. Compos. Sci. Technol. 2012, 72, 550–557. [Google Scholar] [CrossRef]
- Müller, M. Influence of Wood Modification on the Properties of Polyvinyl Chloride Based Wood Polymer Composites (WPC). Bachelor’s Thesis, Georg-August University of Goettingen, Göttingen, Germany, 2011. [Google Scholar]
- Radovanovic, I. Verarbeitung und Optimierung der Rezeptur von Wood Plastic Composites (WPC). Master’s Thesis, University of Osnabrück, Osnabrück, Germany, 2007. [Google Scholar]
- Grünwald, L.K. Influence of Specimen Size and Shape on the Durability of Wood in Laboratory Tests. Master’s Thesis, Georg-August University of Goettingen, Göttingen, Germany, 2019. [Google Scholar]
- Ansell, M.P. Wood Composites; Elsevier/WP, Woodhead: Amsterdam, The Netherlands, 2015; p. 437. ISBN 978-1-78242-477-2. [Google Scholar]
- Ross, R.J. Wood Handbook: Wood as an Engineering Material; U.S. Dept. of Agriculture, Forest Service, Forest Products Laboratory: Madison, WI, USA, 2010; p. 508. ISBN 978-1-48485-970-4.
- Niemz, P. Physik des Holzes und der Holzwerkstoffe/Physics of Wood and Wood-Based Materials; ETH—Eidgenössische Technische Hochschule: Zürich, Zwitzerland, 2005; p. 159. ISBN 3-87191-324-9. [Google Scholar]
- Xie, Y.; Hill, C.A.S.; Xiao, Z.; Militz, H.; Mai, C. Silane coupling agents used for natural fiber/polymer composites: A review. Compos. Part A Appl. Sci. Manuf. 2010, 41, 806–819. [Google Scholar] [CrossRef]
- Schwarze, F.W.M.R. Wood decay under the microscope. Fungal Biol. Rev. 2007, 21, 133–170. [Google Scholar] [CrossRef]
- Da Silveira, M.F.; Gouveia, F.N.; Moreira, A.C.O.; Oliveira, J.R.V.; Silva, A.S.V.S.; Almeida, G.F.; Costa, A.F. Natural resistance of eight Brazilian wood species from the region Caatinga determined by an accelerated laboratory decay test against four fungi. Holzforschung 2019, 73, 151–154. [Google Scholar] [CrossRef]
- Laks, P.; Vehring, K.; Verhey, S.; Richter, D. Mold growth on wood-plastic composites. In Canadian Wood Preservation Association (CWPA) Proceedings; CWPA: Vancouver, BC, Canada, 2007; pp. 60–69. [Google Scholar]
- Laks, P.E.; McKaig, P.A. Flavonoid Biocides: Wood Preservatives Based on Condensed Tannins. Holzforschung 1988, 42, 299–306. [Google Scholar] [CrossRef]
- Hale, M.D.C.; Eaton, R.A. Soft Rot Cavity Widening—A Consideration of the Kinetics; IRG/WP/1227; International Research Group on Wood Preservation (IRG): Stockholm, Sweden, 1984; pp. 1–12. [Google Scholar]
- Schmidt, O.; Czeschlik, D. Wood and Tree Fungi: Biology, Damage, Protection, and Use; Springer: Berlin/Heidelberg, Germany, 2006. [Google Scholar]
- Müller, M.; Gellerich, A.; Militz, H.; Krause, A. Resistance of modified polyvinyl chloride/wood flour composites to basidiomycetes. Eur. J. Wood Prod. 2013, 71, 199–204. [Google Scholar] [CrossRef][Green Version]
- Morrell, J.J.; Stark, N.M.; Pendleton, D.E.; McDonald, A.G. Durability of wood-plastic composites. In Proceedings of the 10th International Conference on Wood & Biofiber Plastic Composites Forest Products Society, Madison, WI, USA, 11–13 May 2009; pp. 71–75. [Google Scholar]
- Van den Bulcke, J.; Biziks, V.; Andersons, B.; Mahnert, C.M.; Militz, H.; Van loo, D.; Dierick, M.; Masschaele, B.; Boone, M.N.; Brabant, L.; et al. Potential of X-ray computed tomography for 3D anatomical analysis and microdensitometrical assessment in wood research with focus on wood modification. Int. Wood Prod. J. 2013, 4, 183–190. [Google Scholar] [CrossRef]
- Yamauchi, D.; Tamaoki, D.; Hayami, M.; Takeuchi, M.; Karahara, I.; Sato, M.; Toyooka, K.; Nishioka, H.; Terada, Y.; Uesugi, K.; et al. Micro-CT observations of the 3D distribution of calcium oxalate crystals in cotyledons during maturation and germination in Lotus miyakojimae seeds. Microsc. Oxf. Engl. 2013, 62, 353–361. [Google Scholar] [CrossRef]
- Daniel, G. Use of electron microscopy for aiding our understanding of wood biodegradation. FEMS Microbiol. Rev. 1996, 13, 199–233. [Google Scholar] [CrossRef]
- Eriksson, K.-E.L.; Blanchette, R.; Ander, P. Microbial and Enzymatic Degradation of Wood and Wood Components; Springer: Berlin/Heidelberg, Germany, 2012. [Google Scholar] [CrossRef]
- Guggiari, M.; Bloque, R.; Aragno, M.; Verrecchia, E.; Job, D.; Junier, P. Experimental calcium-oxalate crystal production and dissolution by selected wood-rot fungi. Int. Biodeterior. Biodegrad. 2011, 65, 803–809. [Google Scholar] [CrossRef]
- Schilling, P.J.; Karedla, B.R.; Tatiparthi, A.K.; Verges, M.; Herrington, P.D. X-ray computed microtomography of internal damage in fiber reinforced polymer matrix composites. Compos. Sci. Technol. 2005, 65, 2071–2078. [Google Scholar] [CrossRef]
- Koddenberg, T.; Zauner, M.; Militz, H. REM und µCT-Aufnahmen von Holz nach Pilzbefall im Labortest—SEM and µCT imaging of wood after fungal decay in laboratory test. In Proceedings of the Deutsche Holzschutztagung, Dresden, Germany, 4–5 April 2019; pp. 115–125. [Google Scholar]
- Dutton, M.V.; Evans, C.S. Oxalate production by fungi: Its role in pathogenicity and ecology in the soil environment. Can. J. Microbiol. 1996, 42, 881–895. [Google Scholar] [CrossRef]
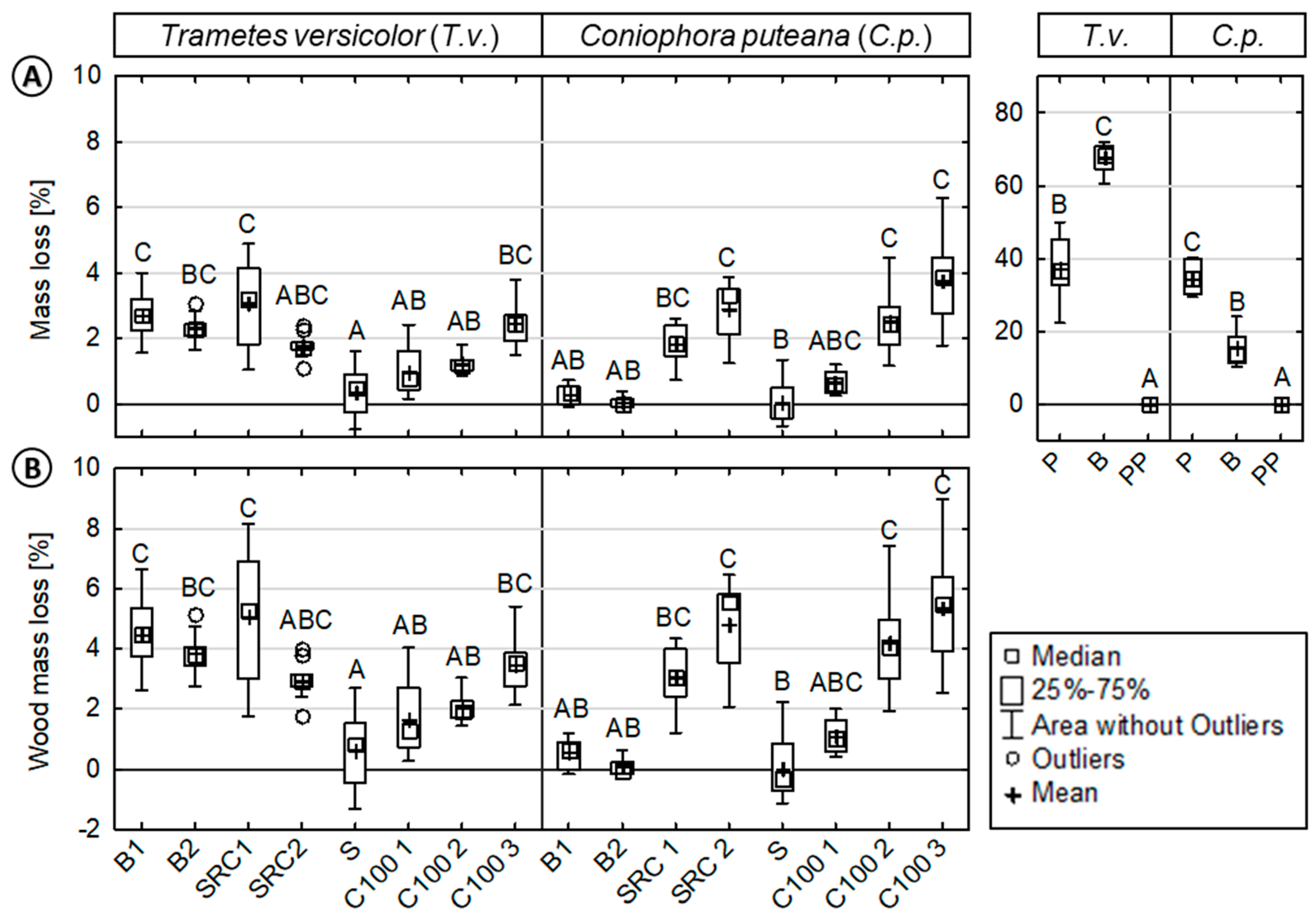

| SID | Species | Wood | PP | MA-PP | MB | Characteristics |
|---|---|---|---|---|---|---|
| (wt %) | ||||||
| B1 | Fagus sylvatica | 60 | 35 | 3 | 2 | Juvenile beech stems, debarked |
| B2 | Fagus sylvatica | 60 | 35 | 3 | 2 | Old and colored beech stems, debarked |
| SRC 1 | Populus spp. | 60 | 35 | 3 | 2 | Short rotation coppice including bark |
| SRC 2 | Salix spp. | 60 | 35 | 3 | 2 | Short rotation coppice including bark |
| S | Picea abies | 60 | 35 | 3 | 2 | Norway spruce stems, debarked |
| C100 1 | assorted Picea abies | 60 | 35 | 3 | 2 | Commercially available softwood source |
| C100 2 | assorted Picea abies | 60 | 38 | 0 | 2 | Commercially available softwood source |
| C100 3 | assorted Picea abies | 70 | 28 | 0 | 2 | Commercially available softwood source |
| P | Picea abies | 100 | - | - | - | Norway spruce, solid wood |
| B | Fagus sylvatica | 100 | - | - | - | European beech, solid wood |
| PP | - | 0 | 95 | 3 | 2 | Sabic 575P, Isotactic polypropylene |
| SID | ω0 | ω1 | ω2 | Trametes versicolor | Coniophora puteana | ||
|---|---|---|---|---|---|---|---|
| [%] | ML [%] | ωtv [%] | ML [%] | ωcp [%] | |||
| B1 | 2.0 (3.3) | 3.5 (5.8) | 15.2 (25.3) | 2.1 (3.5) | 17.3 (28.8) | 0.7 (1.1) | 16.3 (27.1) |
| B2 | 1.9 (3.2) | 3.1 (5.1) | 14.8 (24.7) | 2.3 (3.9) | 17.6 (29.3) | 0.0 (0.1) | 16.0 (26.6) |
| SRC 1 | 1.9 (3.2) | 6.2 (10.3) | 16.2 (27.0) | 3.1 (5.1) | 18.0 (29.9) | 1.8 (3.1) | 18.1 (30.2) |
| SRC 2 | 1.9 (3.2) | 6.0 (10.0) | 19.7 (32.8) | 1.8 (3.0) | 23.4 (39.0) | 2.9 (4.8) | 25.1 (41.9) |
| S | 1.9 (3.2) | 5.0 (8.3) | 14.5 (24.2) | 0.4 (0.6) | 16.4 (27.3) | 0.0 (0.1) | 16.3 (27.2) |
| C100 1 | 2.0 (3.3) | 6.4 (10.7) | 15.9 (26.4) | 1.0 (1.6) | 16.9 (28.2) | 0.7 (1.1) | 16.6 (27.60) |
| C100 2 | 2.3 (3.8) | 14.9 (24.8) | 20.3 (33.8) | 1.2 (2.0) | 23.4 (39.0) | 2.5 (4.2) | 25.1 (41.9) |
| C100 3 | 3.1 (4.4) | 20.5 (29.3) | 23.4 (33.4) | 2.4 (3.5) | 28.8 (41.1) | 3.7 (5.3) | 30.0 (42.9) |
| P | 13.2 | - | 34.9 | 37.3 | 106.4 | 34.7 | 178.2 |
| B | 11.8 | - | 51.6 | 67.6 | 140.8 | 15.4 | 103.4 |
| PP | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Krause, K.C.; Brischke, C.; Koddenberg, T.; Buschalsky, A.; Militz, H.; Krause, A. Resistance of Injection Molded Wood-Polypropylene Composites against Basidiomycetes According to EN 15534-1: New Insights on the Test Procedure, Structural Alterations, and Impact of Wood Source. Fibers 2019, 7, 92. https://doi.org/10.3390/fib7100092
Krause KC, Brischke C, Koddenberg T, Buschalsky A, Militz H, Krause A. Resistance of Injection Molded Wood-Polypropylene Composites against Basidiomycetes According to EN 15534-1: New Insights on the Test Procedure, Structural Alterations, and Impact of Wood Source. Fibers. 2019; 7(10):92. https://doi.org/10.3390/fib7100092
Chicago/Turabian StyleKrause, Kim Christian, Christian Brischke, Tim Koddenberg, Andreas Buschalsky, Holger Militz, and Andreas Krause. 2019. "Resistance of Injection Molded Wood-Polypropylene Composites against Basidiomycetes According to EN 15534-1: New Insights on the Test Procedure, Structural Alterations, and Impact of Wood Source" Fibers 7, no. 10: 92. https://doi.org/10.3390/fib7100092
APA StyleKrause, K. C., Brischke, C., Koddenberg, T., Buschalsky, A., Militz, H., & Krause, A. (2019). Resistance of Injection Molded Wood-Polypropylene Composites against Basidiomycetes According to EN 15534-1: New Insights on the Test Procedure, Structural Alterations, and Impact of Wood Source. Fibers, 7(10), 92. https://doi.org/10.3390/fib7100092







