Antibacterial Properties of Zn Doped Hydrophobic SiO2 Coatings Produced by Sol-Gel Method
Abstract
1. Introduction
2. Materials and Methods
2.1. Preparation of Sols and Deposition of Coatings
2.2. Characterization of Coatings
2.3. Antibacterial Properties
2.3.1. Bacterial Colonization
2.3.2. Visualization of E. coli Cells at the Sample Surface
3. Results and Discussion
3.1. Characteristics of Coatings
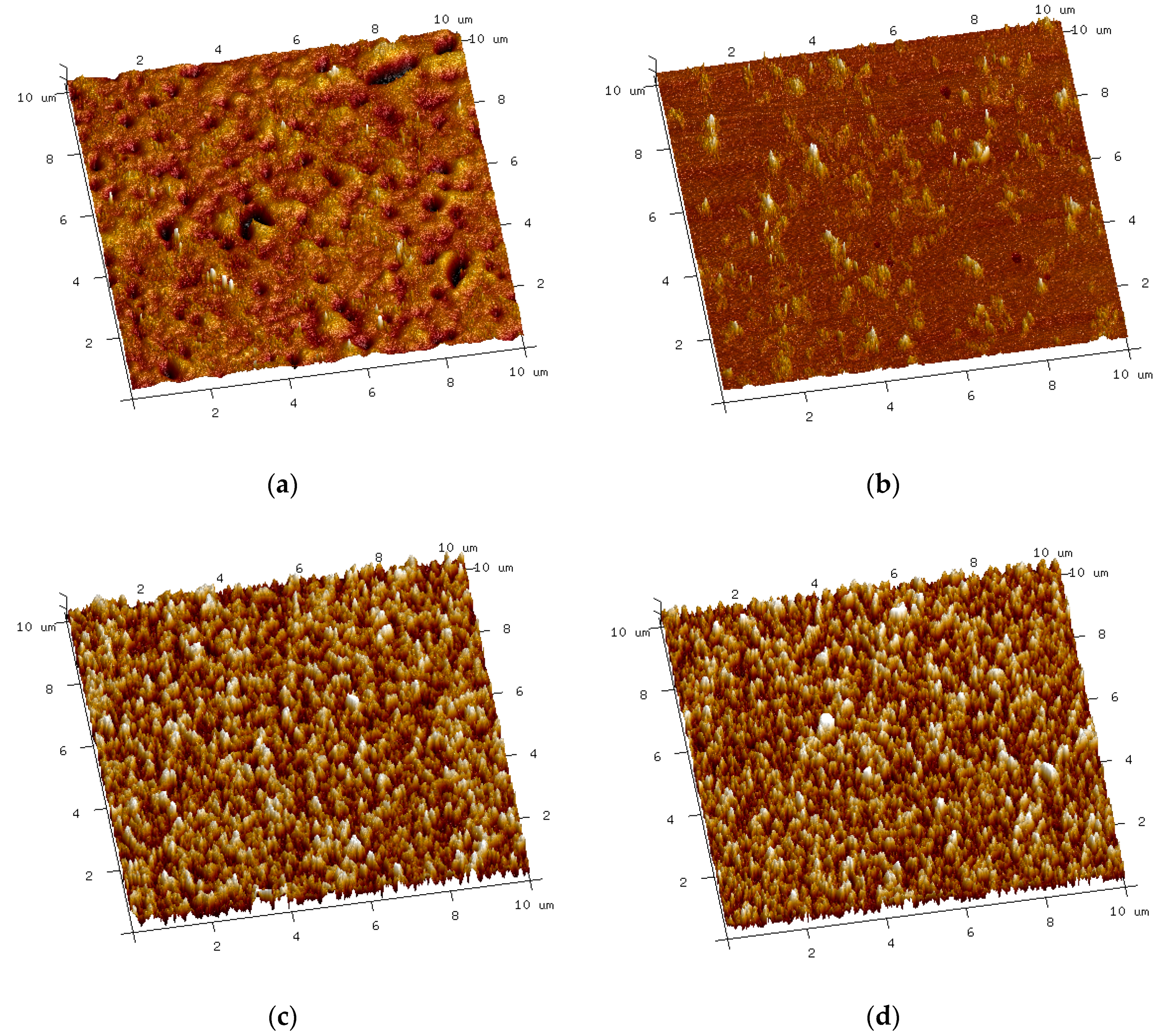
3.1.1. Microscopic Study
3.1.2. Thickness of Coatings
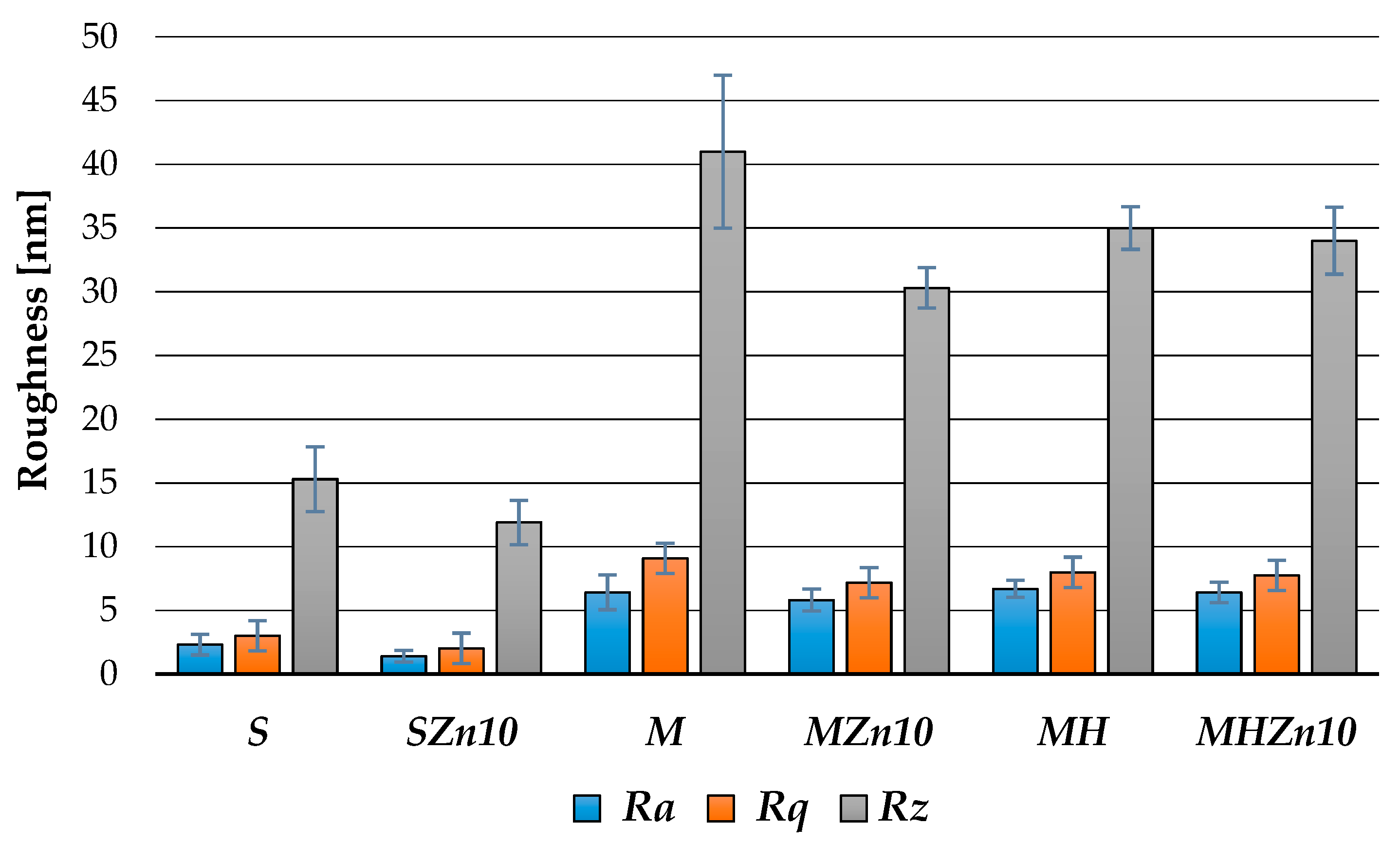
3.1.3. Topography and Roughness of Coatings
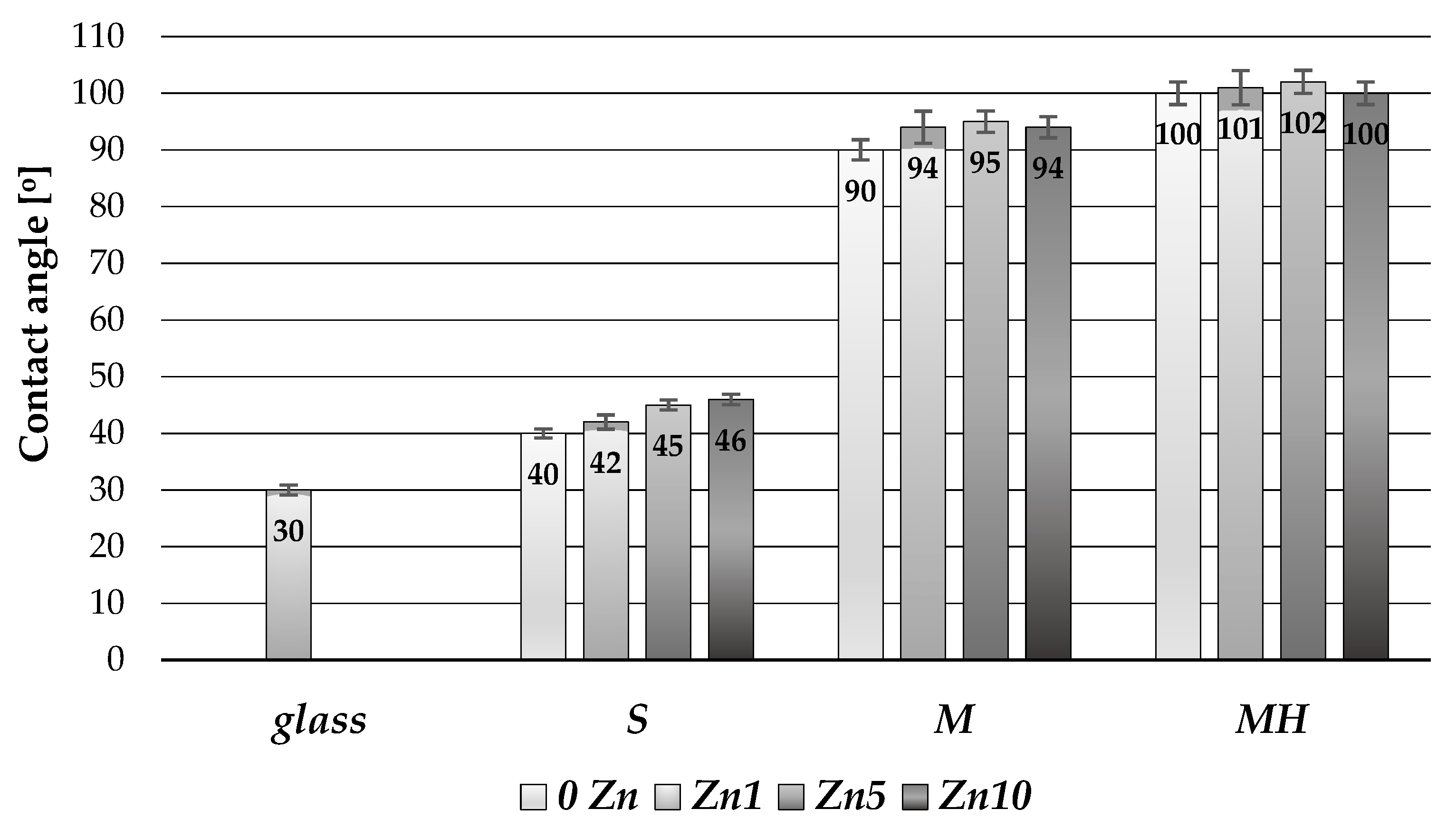
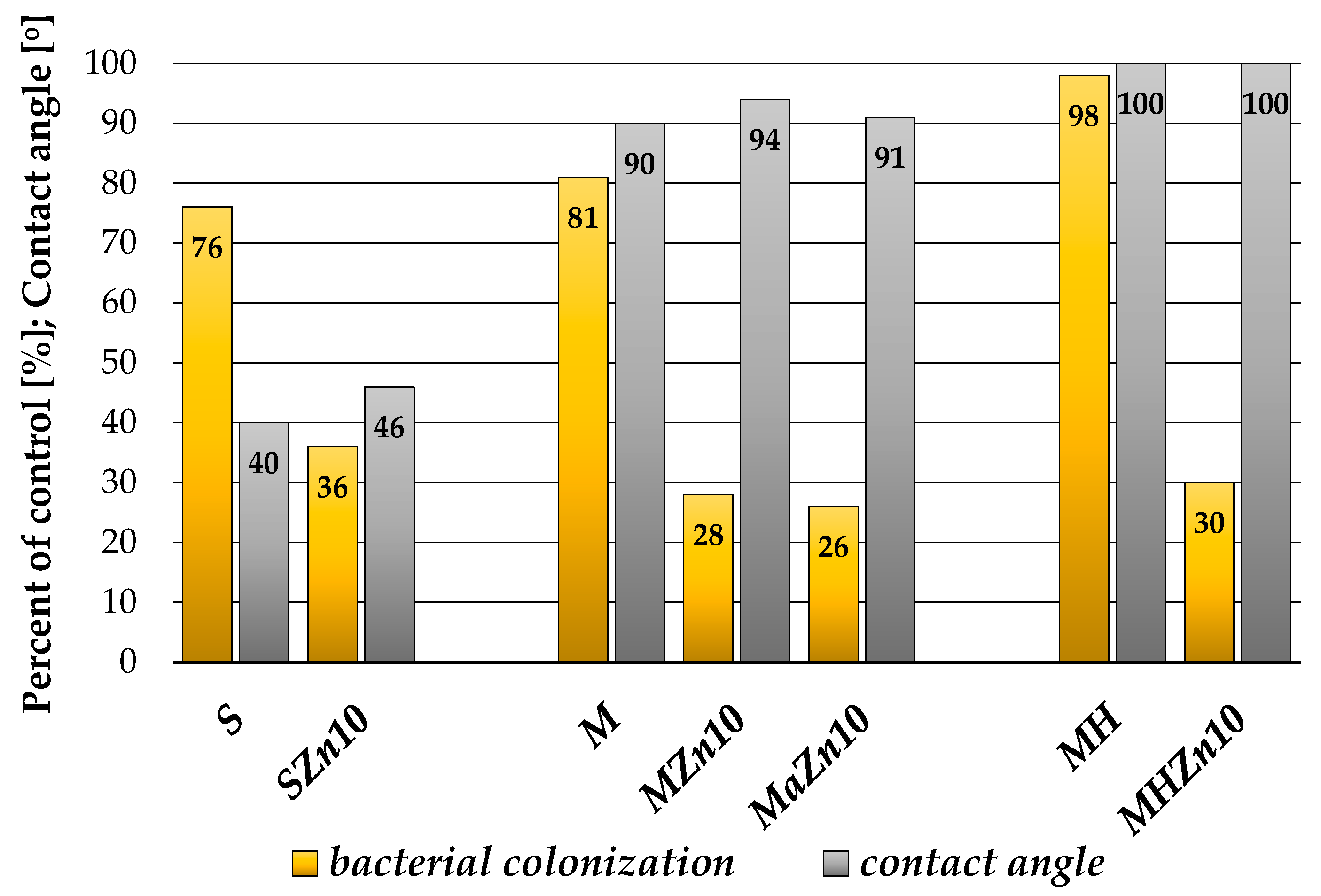
3.1.4. Wettability of Coatings
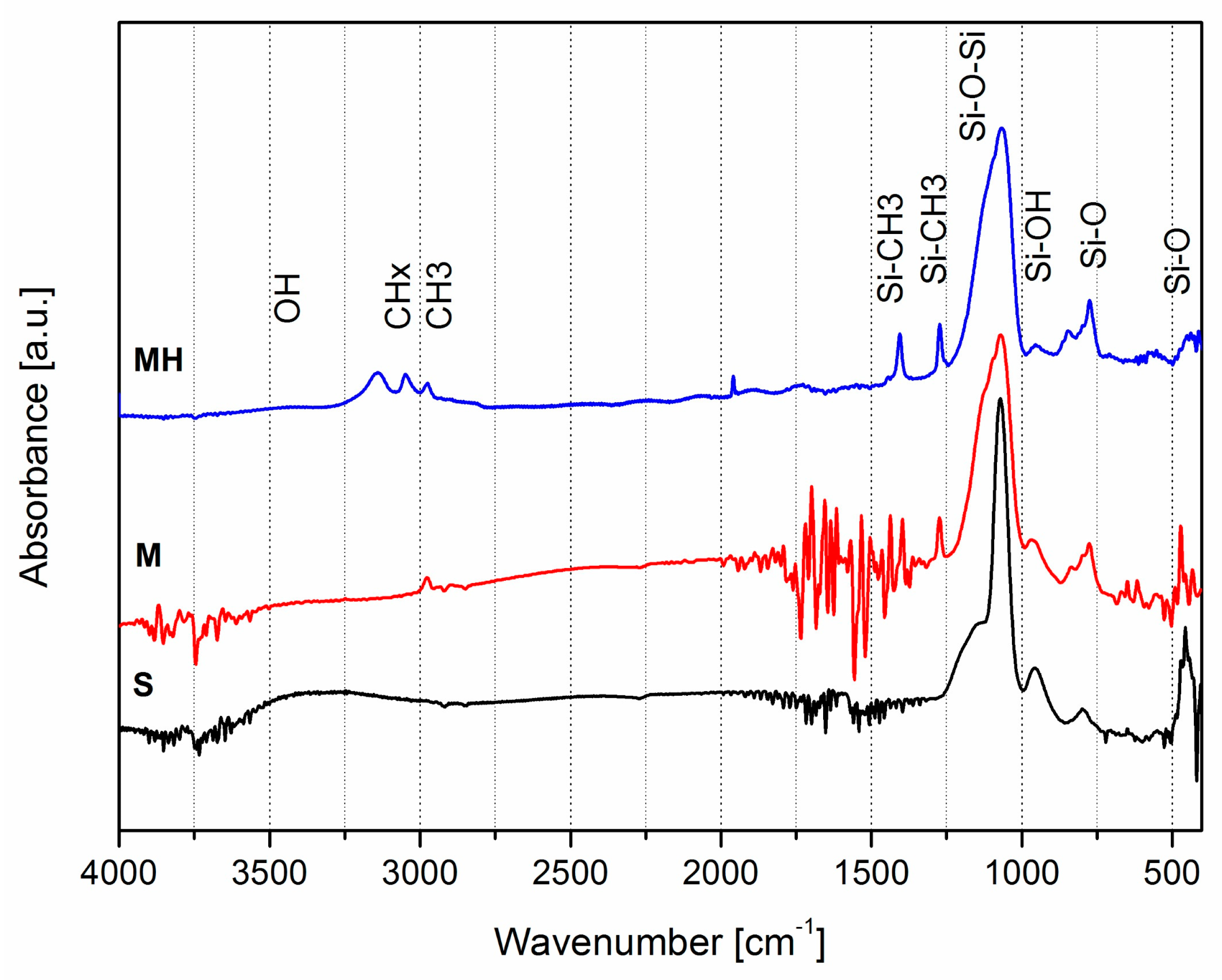
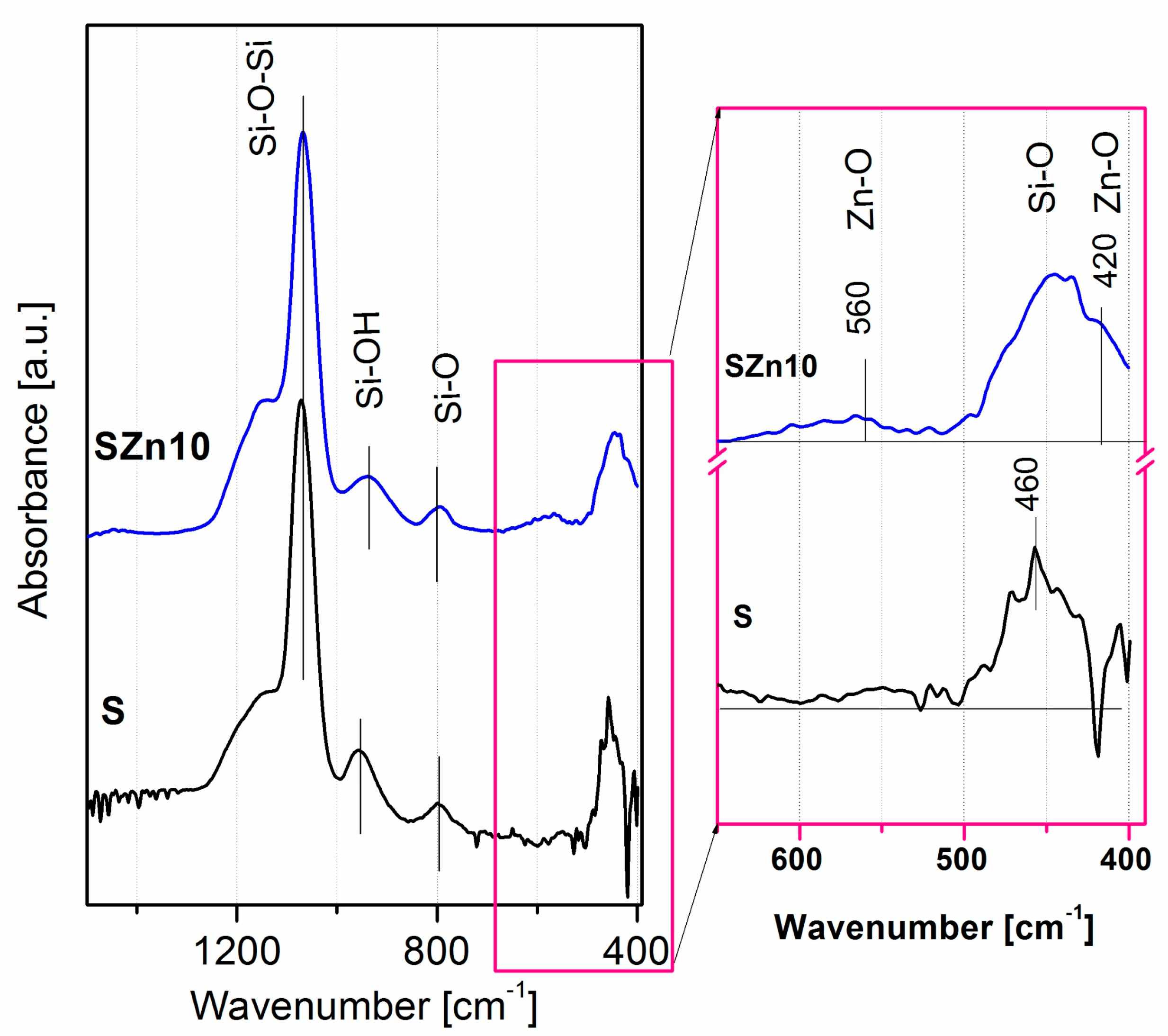
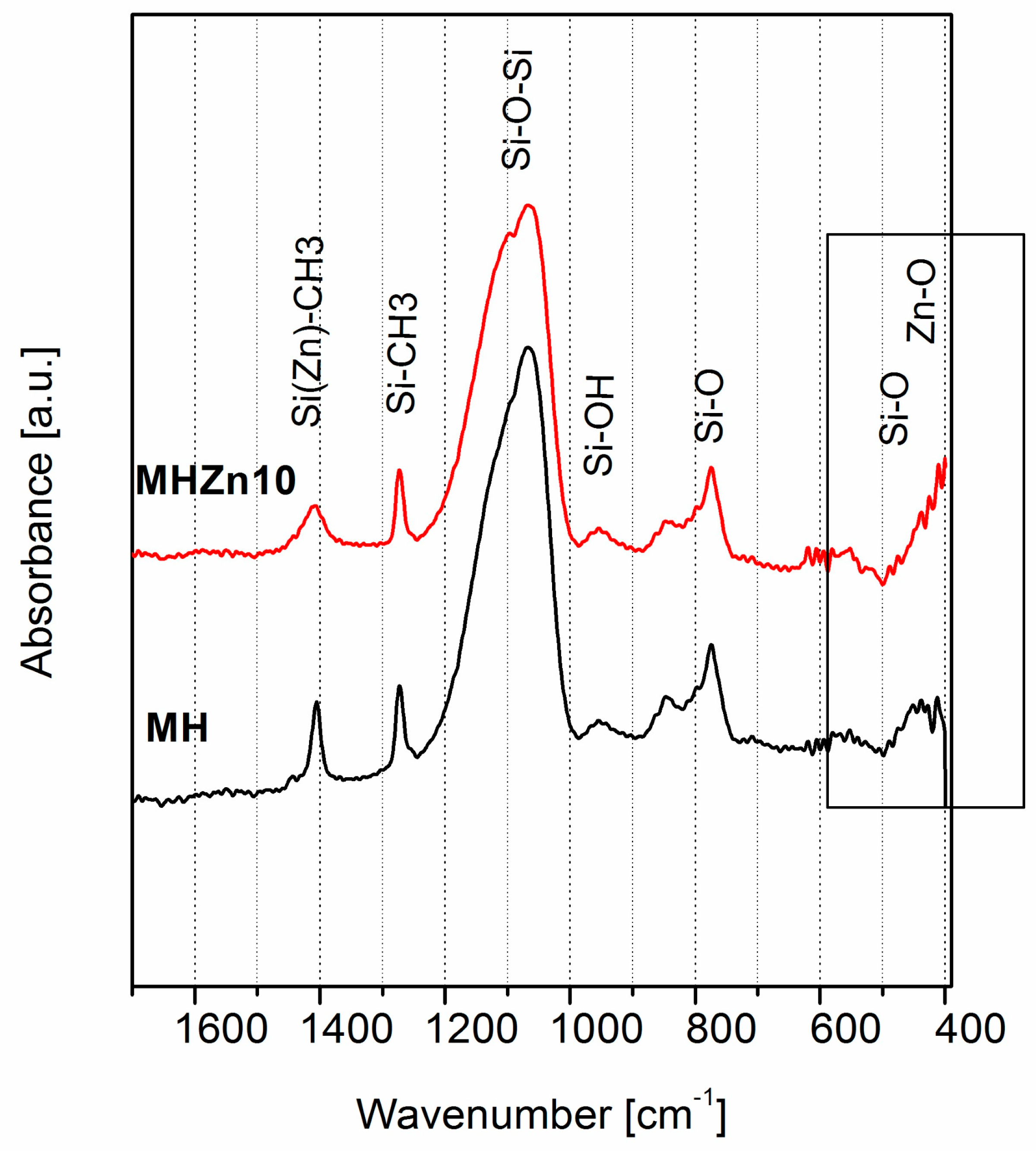
3.1.5. Chemical Structure of Coatings
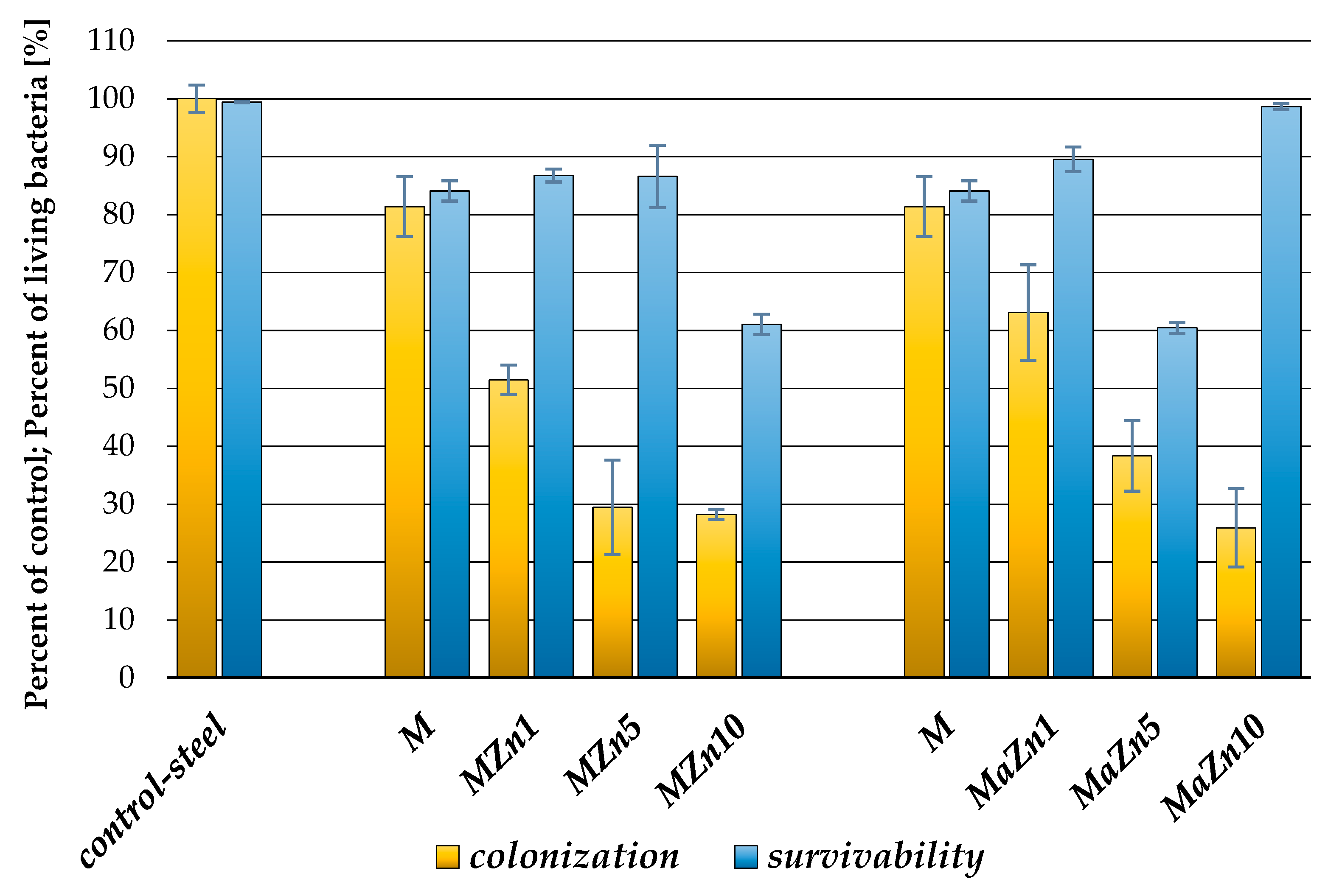
3.2. Antibacterial Properties of the Coatings
4. Summary and Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Villapún, V.M.; Dover, L.G.; Cross, A.; González, S. Antibacterial metallic touch surfaces. Materials 2016, 9, 736. [Google Scholar] [CrossRef]
- Page, K.; Wilson, M.; Parkin, I.P. Antimicrobial surfaces and their potential in reducing the role of the inanimate environment in the incidence of hospital-acquired infections. J. Mater. Chem. 2009, 19, 3819–3831. [Google Scholar] [CrossRef]
- Carniello, V.; Peterson, B.W.; van der Mei, H.C.; Busscher, H.J. Physico-chemistry from initial bacterial adhesion to surface-programmed biofilm growth. Adv. Colloid Interface Sci. 2018, 261, 1–14. [Google Scholar] [CrossRef]
- Song, F.; Koo, H.; Ren, D. Effects of material properties on bacterial adhesion and biofilm formation. J. Dent. Res. 2015, 94, 1027–1034. [Google Scholar] [CrossRef] [PubMed]
- Ramasamy, M.; Lee, J. Recent nanotechnology approaches for prevention and treatment of biofilm-associated infections on medical devices. BioMed Res. Int. 2016, 2016, 1851242. [Google Scholar] [CrossRef] [PubMed]
- Jamal, M.; Ahmad, W.; Andleeb, S.; Jalil, F.; Imran, M.; Nawaz, M.A.; Hussain, T.; Ali, M.; Rafiq, M.; Kamil, M.A. Bacterial biofilm and associated infections. J. Chin. Med. Assoc. 2018, 81, 7–11. [Google Scholar] [CrossRef]
- Kip, N.; van Veen, J.A. The dual role of microbes in corrosion. ISME J. 2014, 9, 542–551. [Google Scholar] [CrossRef] [PubMed]
- Campoccia, D.; Montanaro, L.; Arciola, C.R. A review of the biomaterials technologies for infection-resistant surfaces. Biomaterials 2013, 34, 8533–8554. [Google Scholar] [CrossRef] [PubMed]
- Sun, D.; Babar Shahzad, M.; Li, M.; Wang, G.; Xu, D. Antimicrobial materials with medical applications. Mater. Technol. 2015, 30, B90–B95. [Google Scholar] [CrossRef]
- Elbourne, A.; Crawford, R.J.; Ivanova, E.P. Nano-structured antimicrobial surfaces: From nature to synthetic analogues. J. Colloid Interface Sci. 2017, 508, 603–616. [Google Scholar] [CrossRef] [PubMed]
- Henriques, P.C.; Borges, I.; Pinto, A.M.; Magalhães, F.D.; Gonçalves, I.C. Fabrication and antimicrobial performance of surfaces integrating graphene-based materials. Carbon 2018, 132, 709–732. [Google Scholar] [CrossRef]
- Champagne, V.; Sundberg, K.; Helfritch, D. Kinetically deposited copper antimicrobial surfaces. Coatings 2019, 9, 257. [Google Scholar] [CrossRef]
- Swartjes, J.J.T.M.; Sharma, P.K.; van Kooten, T.G.; van der Mei, H.C.; Mahmoudi, M.; Busscher, H.J.; Rochford, E.T.J. Current developments in antimicrobial surface coatings for biomedical applications. Curr. Med. Chem. 2015, 22, 2116–2129. [Google Scholar] [CrossRef] [PubMed]
- Cloutier, M.; Mantovani, D.; Rosei, F. Antibacterial coatings: Challenges, perspectives, and opportunities. Trends Biotechnol. 2015, 33, 637–652. [Google Scholar] [CrossRef] [PubMed]
- Adlhart, C.; Verran, J.; Azevedo, N.F.; Olmez, H.; Keinänen-Toivola, M.M.; Gouveia, I.; Melo, L.F.; Crijns, F. Surface modifications for antimicrobial effects in the healthcare setting: a critical overview. J. Hosp. Infection 2018, 99, 239–249. [Google Scholar] [CrossRef]
- Li, R.; Jin, Z.T.; Liu, Z.; Liu, L. Antimicrobial double-layer coating prepared from pure or doped-titanium dioxide and binders. Coatings 2018, 8, 41. [Google Scholar] [CrossRef]
- Zhang, X.; Wang, L.; Levänen, E. Superhydrophobic surfaces for the reduction of bacterial adhesion. RSC Adv. 2013, 3, 12003–12020. [Google Scholar] [CrossRef]
- Krasowska, A.; Sigler, K. How microorganisms use hydrophobicity and what does this mean for human needs? Front. Cell. Infect. Microbiol. 2014, 4, 112. [Google Scholar] [CrossRef] [PubMed]
- Salwiczek, M.; Qu, Y.; Gardiner, J.; Strugnell, R.A.; Lithgow, T.; McLean, K.M.; Thissen, H. Emerging rules for effective antimicrobial coatings. Trends Biotechnol. 2014, 32, 82–90. [Google Scholar] [CrossRef] [PubMed]
- Levy, D.; Zayat, M. The Sol-Gel Handbook: Synthesis, Characterization and Applications, 1st ed.; Wiley-VCH Verlag GmbH & Co. KGaA: Weindheim, Germany, 2015. [Google Scholar]
- Jaiswal, S.; McHale, P.; Duffy, B. Preparation and rapid analysis of antibacterial silver, copper and zinc doped sol-gel surfaces. Colloids Surf. B Biointerfaces 2012, 94, 170–176. [Google Scholar] [CrossRef]
- Owens, G.J.; Singh, R.K.; Foroutan, F.; Alqaysi, M.; Han, C.-M.; Mahapatra, C.; Kim, H.-W.; Knowles, J.C. Sol-gel based materials for biomedical applications. Prog. Mater. Sci. 2016, 77, 1–79. [Google Scholar] [CrossRef]
- Khokhlova, M.; Dykas, M.; Krishnan-Kutty, V.; Patra, A.; Venkatesan, T.; Prellier, W. Oxide thin films as bioactive coatings. J. Phys. Condens. Matt. 2018, 31, 033001. [Google Scholar] [CrossRef]
- Chernousova, S.; Epple, M. Silver as antibacterial agent: Ion, nanoparticle, and metal. Angew. Chem. Int. Ed. 2013, 52, 1636–1653. [Google Scholar] [CrossRef] [PubMed]
- Jung, W.K.; Koo, H.C.; Kim, K.W.; Shin, S.; Kim, S.H.; Park, Y.H. Antibacterial activity and mechanism of action of the silver ion in staphylococcus aureus and Escherichia coli. Appl. Environ. Microbiol. 2008, 74, 2171. [Google Scholar] [CrossRef] [PubMed]
- Jeon, H.-J.; Yi, S.-C.; Oh, S.-G. Preparation and antibacterial effects of Ag-SiO2 thin films by sol-gel method. Biomaterials 2003, 24, 4921–4928. [Google Scholar] [CrossRef]
- Jäger, E.; Schmidt, J.; Pfuch, A.; Spange, S.; Beier, O.; Jäger, N.; Jantschner, O.; Daniel, R.; Mitterer, C. Antibacterial silicon oxide thin films doped with zinc and copper grown by atmospheric pressure plasma chemical vapor deposition. Nanomaterials 2019, 9, 255. [Google Scholar] [CrossRef]
- Trapalis, C.C.; Kokkoris, M.; Perdikakis, G.; Kordas, G. Study of antibacterial composite Cu/SiO2 thin coatings. J. Sol-Gel Sci. Technol. 2003, 26, 1213–1218. [Google Scholar] [CrossRef]
- Turner, R.J. Metal-based antimicrobial strategies. Microb. Biotechnol. 2017, 10, 1062–1065. [Google Scholar] [CrossRef] [PubMed]
- Kumar, V.V.; Anthony, S.P. Antimicrobial studies of metal and metal oxide nanoparticles. In Surface Chemistry of Nanobiomaterials; Grumezescu, A.M., Ed.; William Andrew Publishing: Norwich, NY, USA, 2016; pp. 265–300. [Google Scholar]
- Vincent, M.; Hartemann, P.; Engels-Deutsch, M. Antimicrobial applications of copper. Int. J. Hyg. Environ. Health 2016, 219, 585–591. [Google Scholar] [CrossRef] [PubMed]
- Pasquet, J.; Chevalier, Y.; Pelletier, J.; Couval, E.; Bouvier, D.; Bolzinger, M.-A. The contribution of zinc ions to the antimicrobial activity of zinc oxide. Colloids Surf. A Physicochem. Eng. Aspects 2014, 457, 263–274. [Google Scholar] [CrossRef]
- Lemire, J.A.; Harrison, J.J.; Turner, R.J. Antimicrobial activity of metals: mechanisms, molecular targets and applications. Nat. Rev. Microbiol. 2013, 11, 371. [Google Scholar] [CrossRef] [PubMed]
- Mittapally, S.; Taranum, R.; Parveen, S. Metal ions as antibacterial agents. JDDT 2018, 8, 411–419. [Google Scholar] [CrossRef]
- Jakubowski, W.; Bartosz, G.; Niedzielski, P.; Szymanski, W.; Walkowiak, B. Nanocrystalline diamond surface is resistant to bacterial colonization. Diam. Relat. Mater. 2004, 13, 1761–1763. [Google Scholar] [CrossRef]
- Cai, S.; Zhang, Y.; Zhang, H.; Yan, H.; Lv, H.; Jiang, B. Sol-gel preparation of hydrophobic silica antireflective coatings with low refractive index by base/acid two-step catalysis. ACS Appl. Mater. Interfaces 2014, 6, 11470–11475. [Google Scholar] [CrossRef]
- Philipavičius, J.; Kazadojev, I.; Beganskienė, A.; Melninkaitis, A.; Sirutkaitis, V.; Kareiva, A. Hydrophobic antireflective silica coatings via sol-gel process. Mater. Sci. 2008, 14, 283–287. [Google Scholar]
- Long, D.A. Infrared and Raman Characteristic Group Frequencies. Tables and charts George Socrates John Wiley and Sons, Ltd, Chichester, Third Edition, 2001, J. Raman Spectrosc. 2004, 35, 905. [Google Scholar]
- Preedy, E.; Perni, S.; Nipiĉ, D.; Bohinc, K.; Prokopovich, P. Surface roughness mediated adhesion forces between borosilicate glass and Gram-Positive Bacteria. Langmuir 2014, 30, 9466–9476. [Google Scholar] [CrossRef]
- Tang, L.; Pillai, S.; Revsbech, N.P.; Schramm, A.; Bischoff, C.; Meyer, R.L. Biofilm retention on surfaces with variable roughness and hydrophobicity. Biofouling 2011, 27, 111–121. [Google Scholar] [CrossRef]
- Bazaka, K.; Crawford, R.J.; Ivanova, E.P. Do bacteria differentiate between degrees of nanoscale surface roughness? Biotechnol. J. 2011, 6, 1103–1114. [Google Scholar] [CrossRef]
- Garrett, T.R.; Bhakoo, M.; Zhang, Z. Bacterial adhesion and biofilms on surfaces. Prog. Nat. Sci. 2008, 18, 1049–1056. [Google Scholar] [CrossRef]
- Bjarnsholt, T. The role of bacterial biofilms in chronic infections. APMIS 2013, 121, 1–58. [Google Scholar] [CrossRef] [PubMed]
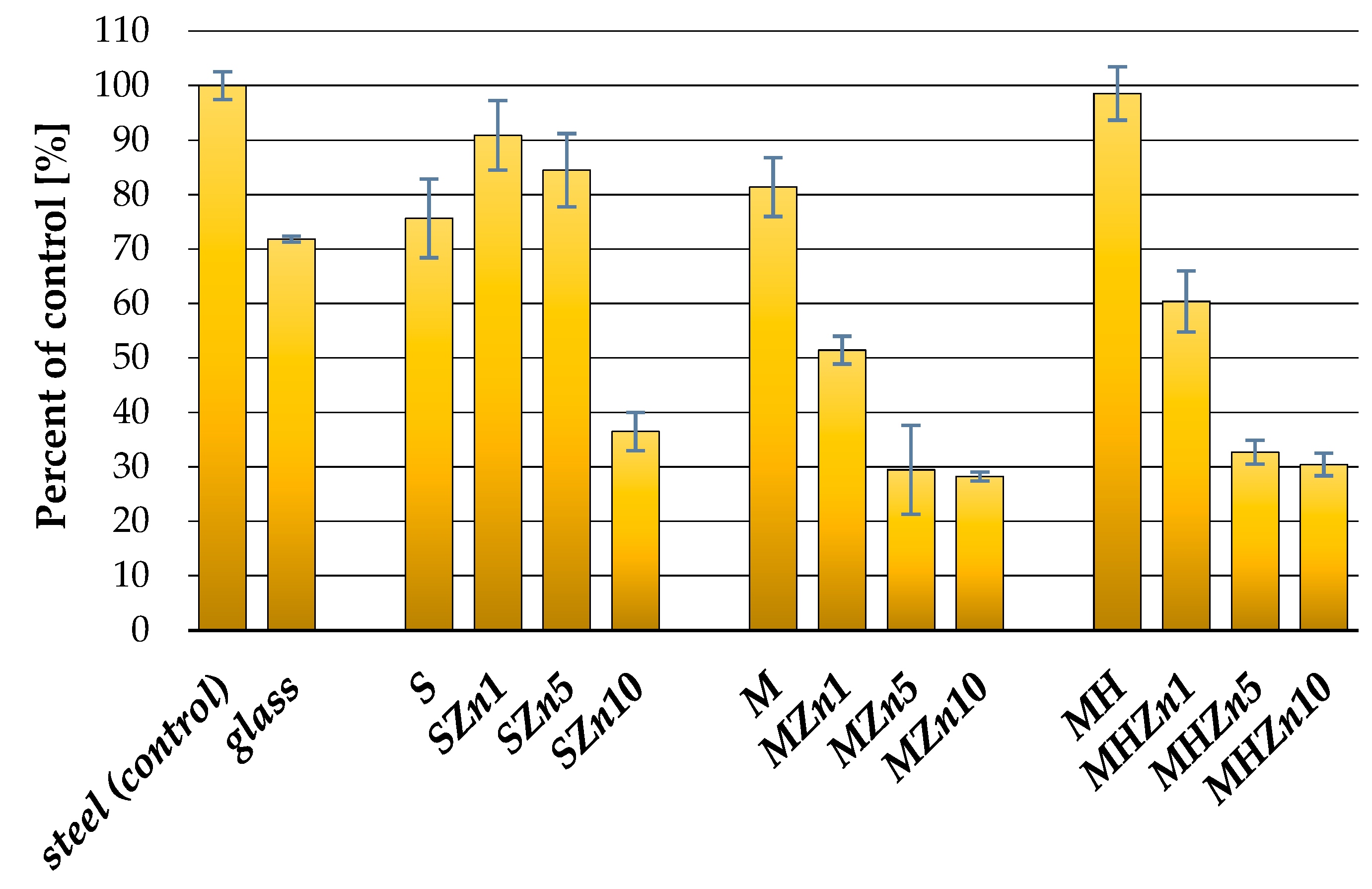
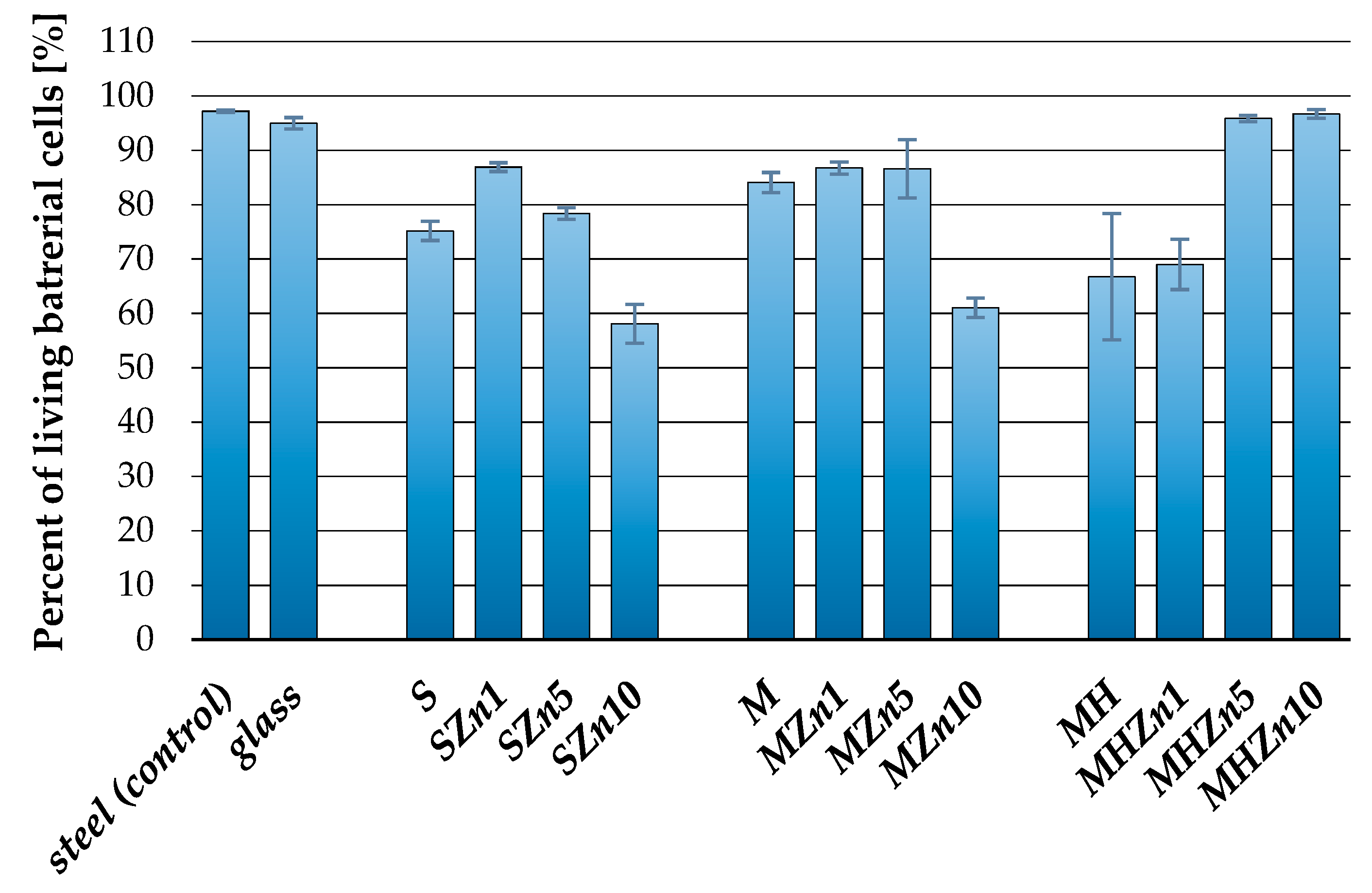
| Sol Label | Precursor | Hydrophobizer | Zinc Compound | Molar Ratio Si/Zn |
|---|---|---|---|---|
| S | TEOS | – | – | – |
| M | TEOS | MTMS | – | – |
| MH | TEOS | MTMS + HMDS | – | – |
| SZn1 | TEOS | – | Zinc nitrate | 0.01 |
| SZn5 | TEOS | – | Zinc nitrate | 0.05 |
| SZn10 | TEOS | – | Zinc nitrate | 0.1 |
| MZn1 | TEOS | MTMS | Zinc nitrate | 0.01 |
| MZn5 | TEOS | MTMS | Zinc nitrate | 0.05 |
| MZn10 | TEOS | MTMS | Zinc nitrate | 0.1 |
| MHZn1 | TEOS | MTMS+HMDS | Zinc nitrate | 0.01 |
| MHZn5 | TEOS | MTMS+HMDS | Zinc nitrate | 0.05 |
| MHZn10 | TEOS | MTMS+HMDS | Zinc nitrate | 0.1 |
| MaZn1 | TEOS | MTMS | Zinc acetate | 0.01 |
| MaZn5 | TEOS | MTMS | Zinc acetate | 0.5 |
| MaZn10 | TEOS | MTMS | Zinc acetate | 0.1 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pietrzyk, B.; Porębska, K.; Jakubowski, W.; Miszczak, S. Antibacterial Properties of Zn Doped Hydrophobic SiO2 Coatings Produced by Sol-Gel Method. Coatings 2019, 9, 362. https://doi.org/10.3390/coatings9060362
Pietrzyk B, Porębska K, Jakubowski W, Miszczak S. Antibacterial Properties of Zn Doped Hydrophobic SiO2 Coatings Produced by Sol-Gel Method. Coatings. 2019; 9(6):362. https://doi.org/10.3390/coatings9060362
Chicago/Turabian StylePietrzyk, Bożena, Katarzyna Porębska, Witold Jakubowski, and Sebastian Miszczak. 2019. "Antibacterial Properties of Zn Doped Hydrophobic SiO2 Coatings Produced by Sol-Gel Method" Coatings 9, no. 6: 362. https://doi.org/10.3390/coatings9060362
APA StylePietrzyk, B., Porębska, K., Jakubowski, W., & Miszczak, S. (2019). Antibacterial Properties of Zn Doped Hydrophobic SiO2 Coatings Produced by Sol-Gel Method. Coatings, 9(6), 362. https://doi.org/10.3390/coatings9060362





