Influence of Silicon-Modified Al Powders (SiO2@Al) on Anti-oxidation Performance of Al2O3-SiO2 Ceramic Coating for Carbon Steel at High Temperature
Abstract
:1. Introduction
2. Experiment Procedure
2.1. Preparation of Carbon Steel Sample
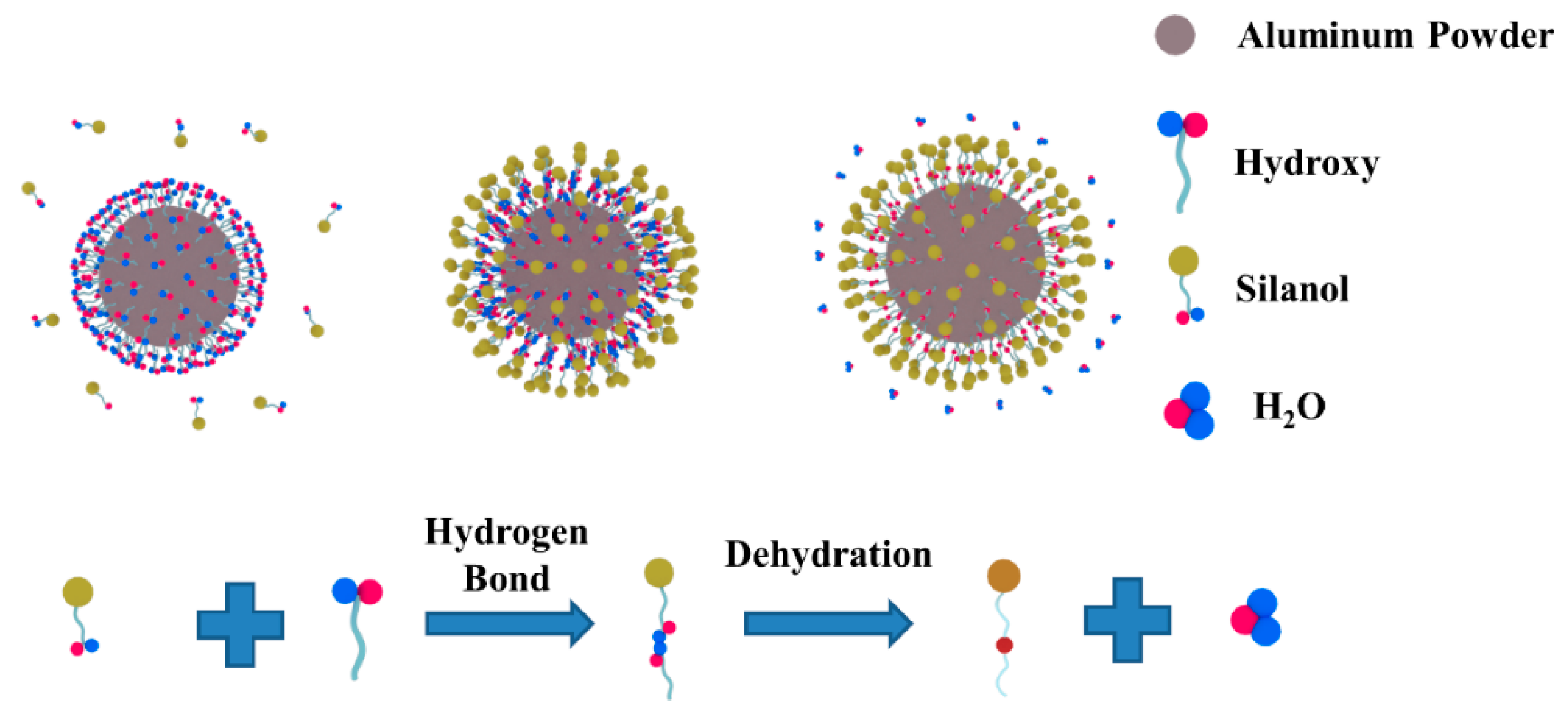
2.2. Preparation of Modified Al Powders (SiO2@Al)
2.3. Preparation of Coating Slurry
2.4. Evaluation of Anti-oxidation Performance
2.5. Characterization
3. Results and Discussion
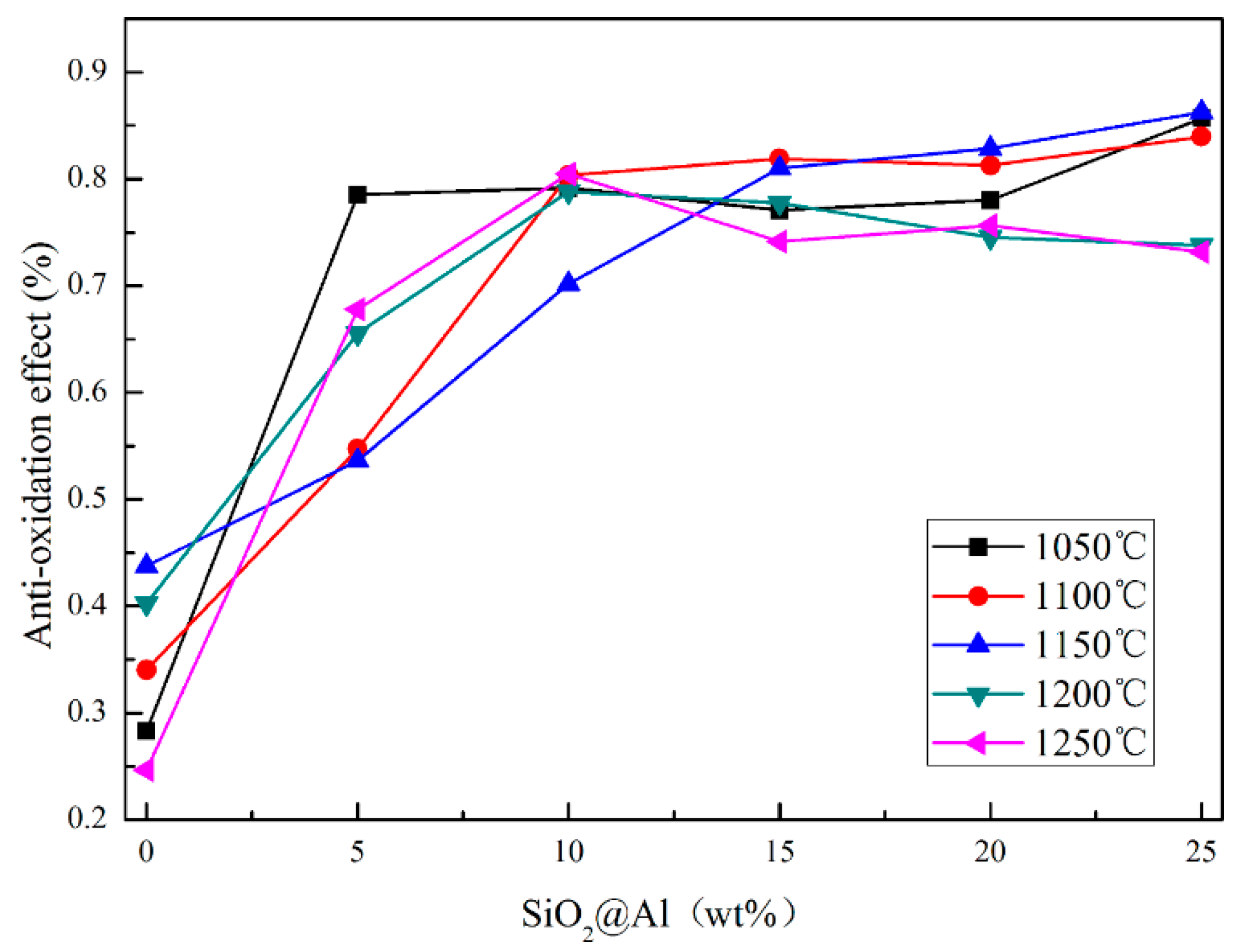
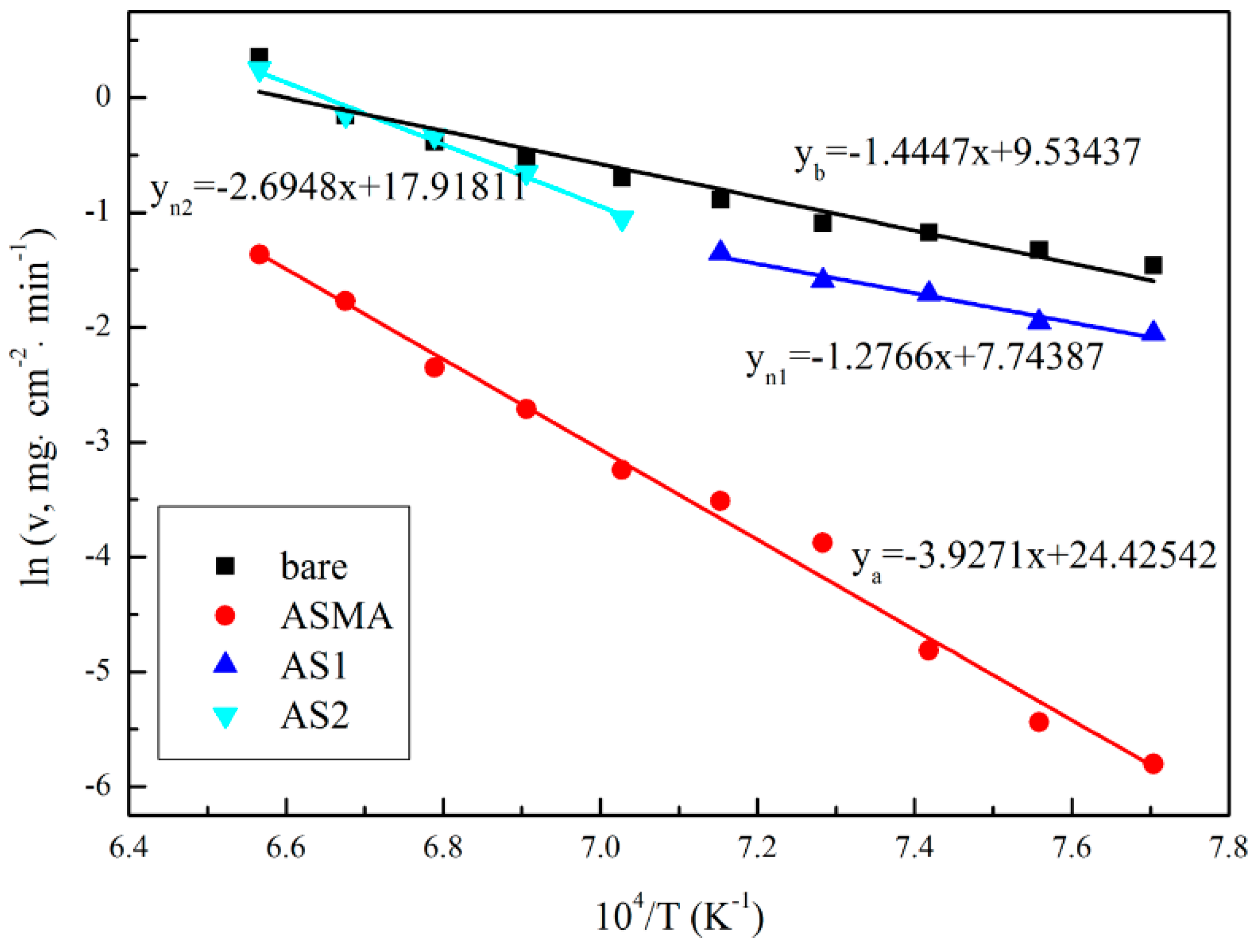
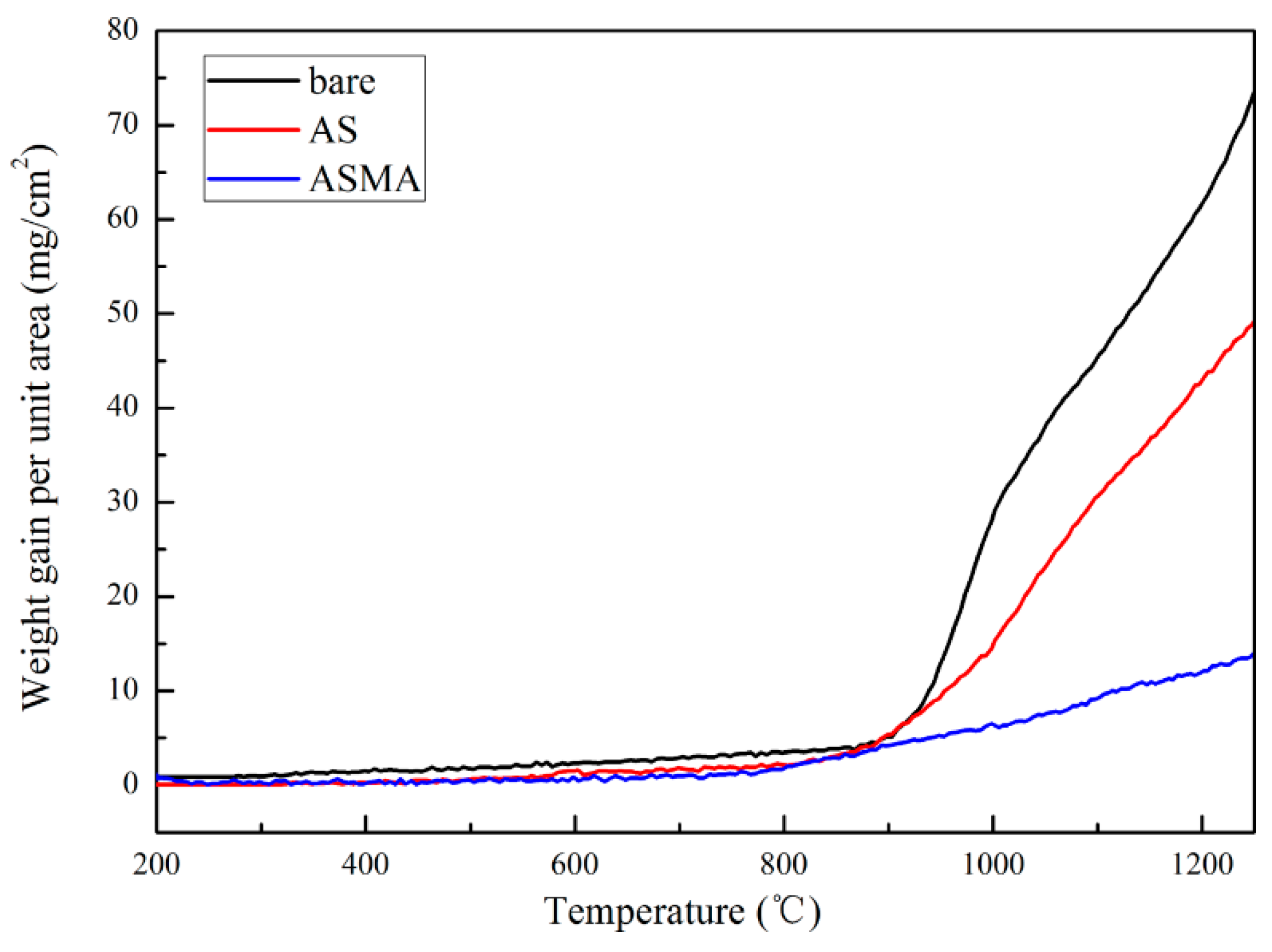
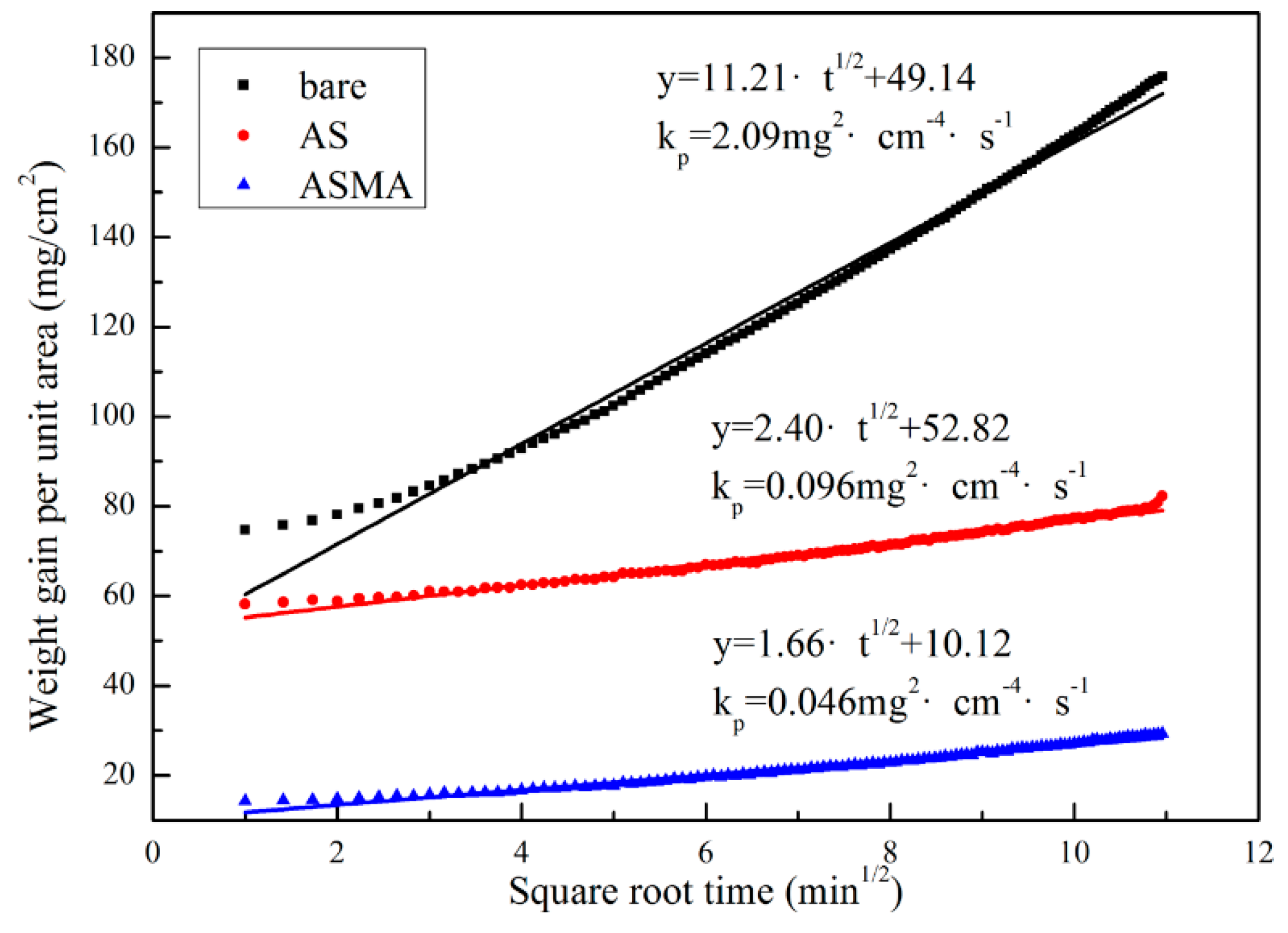
3.1. Performance of Coating
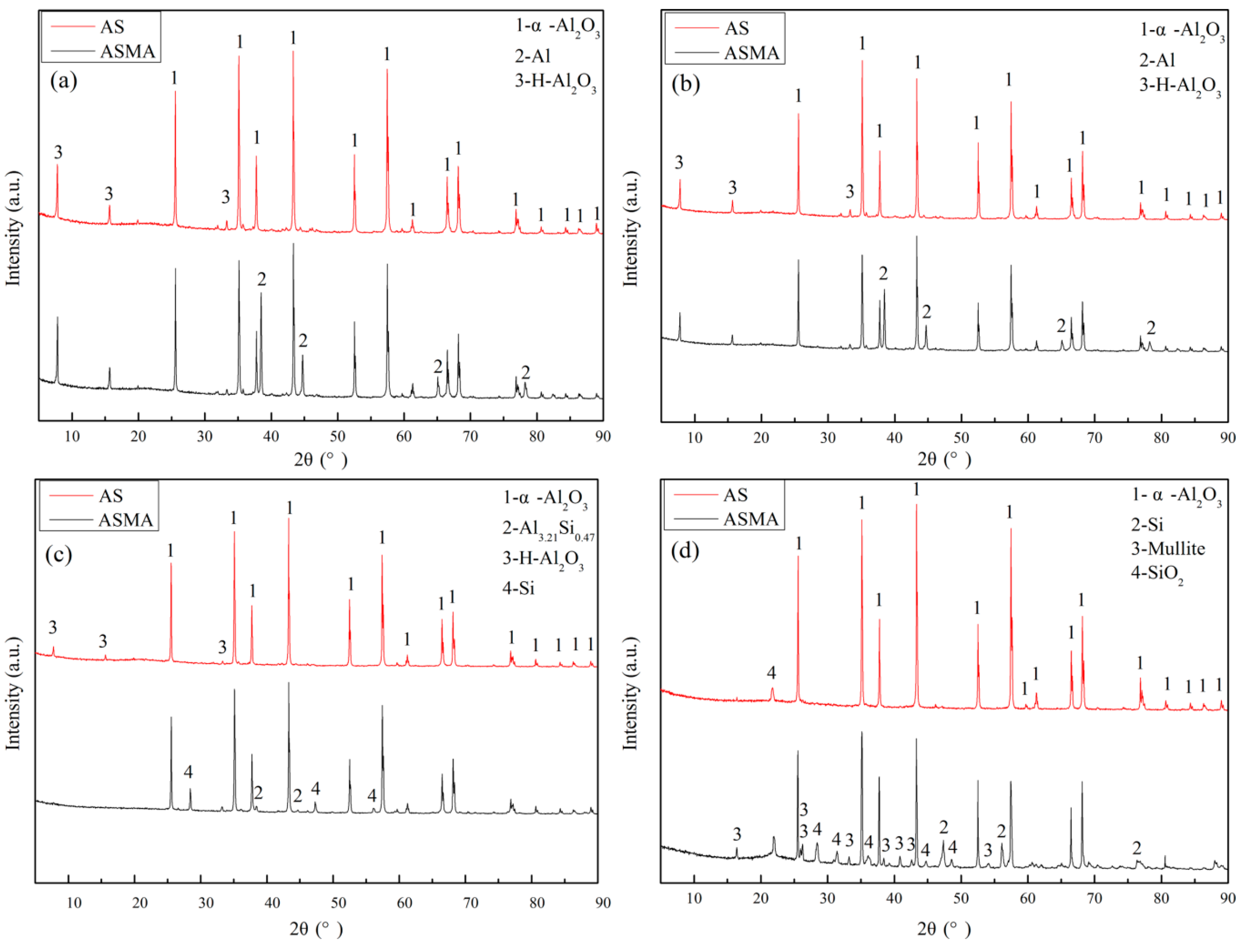
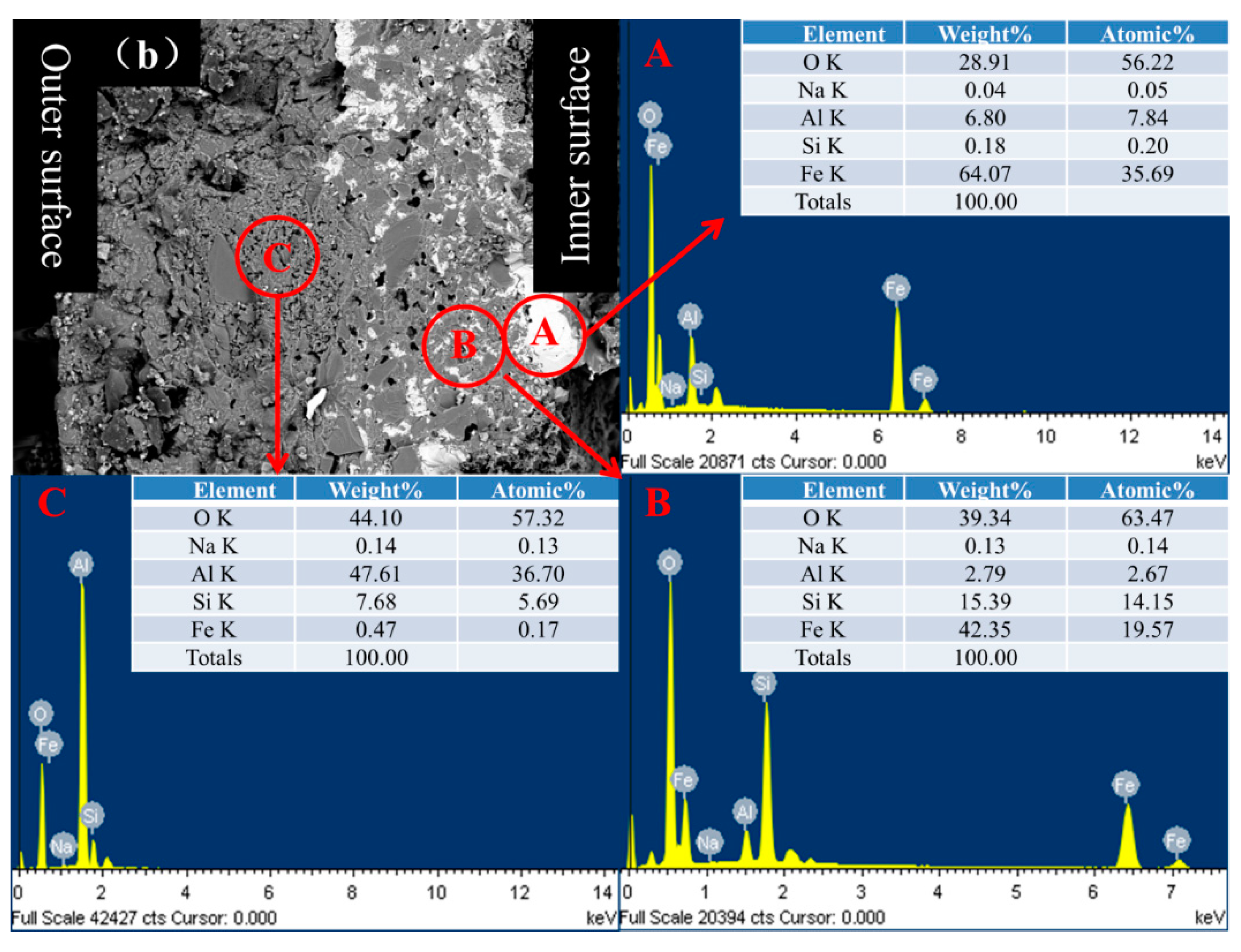
3.2. Morphology of Coating
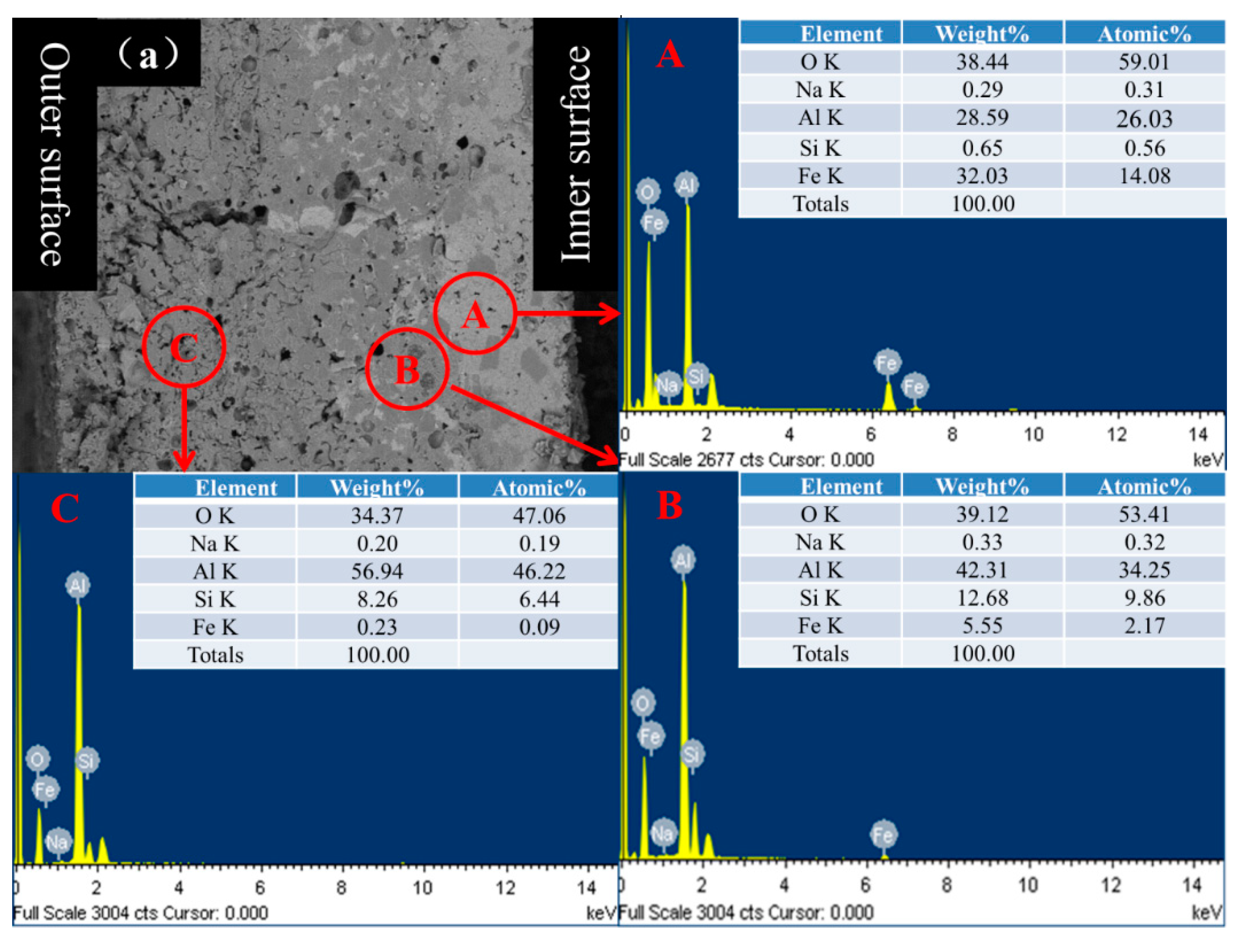
3.3. Protection Mechanism
4. Conclusions
- The Al powders were successfully encapsulated by SiO2 and the SiO2@Al stably existed in the as-prepared alkaline coating slurry. The appropriate proportion of SiO2@Al in the coating was 10%.
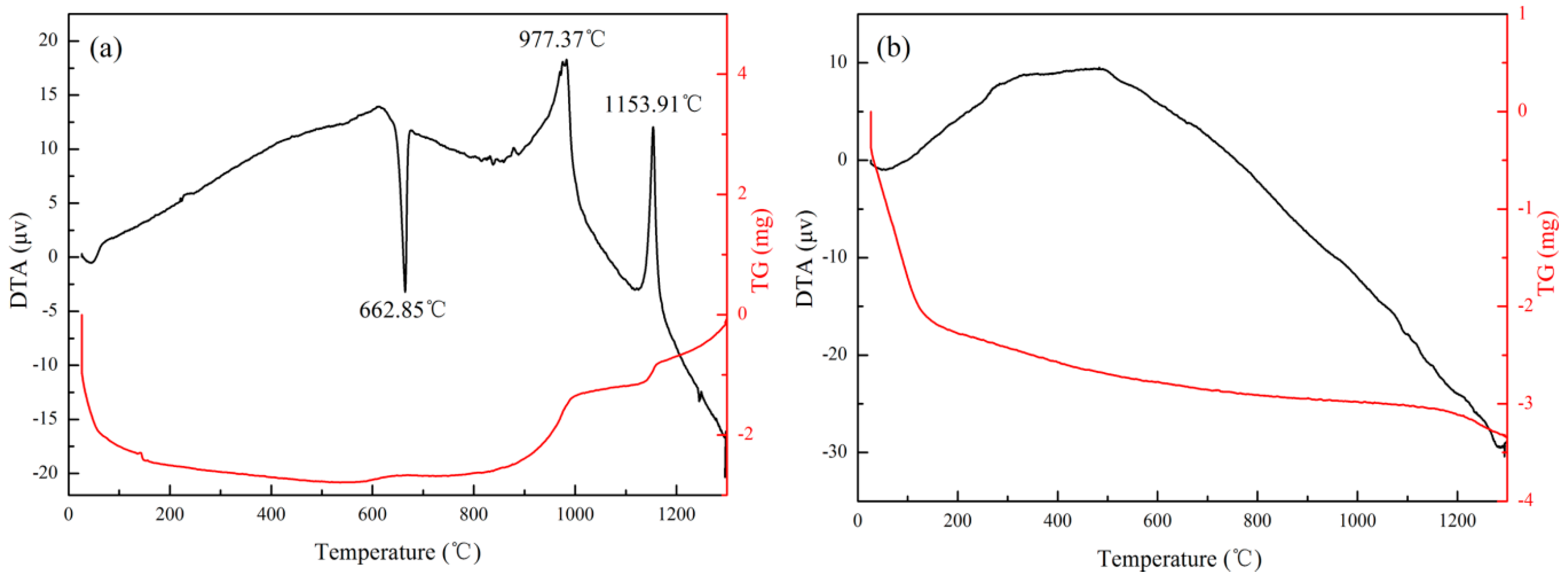
- The metallic Al in ASMA melted at 662.85 °C. What generated local high temperature and accelerated the sintering of ceramic raw materials was the exothermal reaction between the melted Al and SiO2 (4Al + 3SiO2 → 3Si + 2Al2O3). The well-sintered structure decreased the inner diffusion rate of oxygen and enhanced the anti-oxidation effect of ASMA during the temperature-rise period.
- The hercynite layer formed within ASMA played a significant role for slowing down the external diffusion of iron ions due to the lower fraction of Fe2+ occupied the octahedral site, which facilitated the improvement of anti-oxidation ability at 1250 °C.
Author Contributions
Funding
Conflicts of Interest
References
- Frolenkov, K.Y.; Frolenkova, L.Y.; Shadrin, I.F. High-temperature oxidation of low-alloyed steel under glass coatings. Prot. Met. Phys. Chem. Surf. 2010, 46, 103–109. [Google Scholar] [CrossRef]
- Fu, G.; Wei, L.; Zhang, X.; Cui, Y.; Wang, Y.; Yu, B.; Lv, C.; Ye, S. A MgO-SiO2-Al2O3-ZnO ceramic-glass coating to improve the anti-oxidation of carbon steel at high temperature. ISIJ Int. 2018, 58, 929–935. [Google Scholar] [CrossRef]
- Hu, X.J.; Zhang, B.M.; Chen, S.H.; Fang, F.; Jiang, J.Q. Oxide scale growth on high carbon steel at high temperatures. J. Iron Steel Res. Int. 2013, 20, 47–52. [Google Scholar] [CrossRef]
- Liu, Y.; Zhang, W.; Tong, Q.; Sun, Q.; Chao, Y.; Yang, W.; Li, T.; Li, L. Effects of chemical composition on decarburization layer depth of high carbon steels in 2% oxygen atmosphere. Heat Treat. Met. 2017, 42, 143–148. [Google Scholar]
- Heo, N.H.; Lee, J.K. Grain boundary segregation of phosphorus and intergranular surface cracking accompanied by decarburization in plain carbon steels. ISIJ Int. 2011, 51, 673–678. [Google Scholar] [CrossRef]
- Torkar, M.; Glogovac, B. Diminution of scaling by the application of a protective coating. J. Mater. Process. Technol. 1996, 58, 217–222. [Google Scholar] [CrossRef]
- Munther, P.; Lenard, J. The effect of scaling on interfacial friction in hot rolling of steels. J. Mater. Process. Technol. 1999, 88, 105–113. [Google Scholar] [CrossRef]
- Mao, Z.L.; Yang, X.J.; Zhu, S.L.; Cui, Z.D.; Lu, Y. Pack cementation processing parameters for SiC coatings on C/C for optimum tribological properties. Surf. Coat. Technol. 2014, 254, 54–60. [Google Scholar] [CrossRef]
- Fu, G.; Wei, L.; Shan, X.; Zhang, X.; Ding, J.; Lv, C.; Liu, Y.; Ye, S. Influence of a Cr2O3 glass coating on enhancing the oxidation resistance of 20MnSiNb structural steel. Surf. Coat. Technol. 2016, 294, 8–14. [Google Scholar] [CrossRef]
- Fu, G.Y.; Wei, L.Q.; Zhang, X.M.; Cui, Y.B.; Lv, C.C.; Ding, J.; Yu, B.; Ye, S.F. A high-silicon anti-oxidation coating for carbon steel at high temperature. Surf. Coat. Technol. 2017, 310, 166–172. [Google Scholar] [CrossRef]
- Wei, L.; Peng, L.; Ye, S.; Xie, Y.; Chen, Y. Preparation and properties of anti-oxidation inorganic nano-coating for low carbon steel at an elevated temperature. J. Wuhan Univ. Technol. 2006, 21, 48–52. [Google Scholar]
- Wang, D. Formation and property of ceramic layer on a low-carbon steel. Int. J. Process. Sci. Charact. Appl. Adv. Mater. 2007, 134–138. [Google Scholar]
- Zhou, X.; Ye, S.F.; Xu, H.W.; Liu, P.; Wang, X.J.; Wei, L.Q. Influence of ceramic coating of MgO on oxidation behavior and descaling ability of low alloy steel. Surf. Coat. Technol. 2012, 206, 3619–3625. [Google Scholar] [CrossRef]
- Zhou, X.; Wei, L.Q.; Liu, P.; Wang, X.J.; Ye, S.F.; Chen, Y.F. Preparation and characterization of high temperature protective ceramic coating for plain carbon steel. Chin. J. Process Eng. 2010, 10, 167–172. [Google Scholar]
- Li, G.; Jie, X.; He, L. High temperature oxidation behavior of (Ti,Al) C ceramic coatings on carbon steel prepared by electrical discharge coating in kerosene. Adv. Mat. Res. 2011, 189–193, 186–192. [Google Scholar] [CrossRef]
- Nguyen, M.D.; Bang, J.W.; Kim, Y.H.; Bin, A.S.; Hwang, K.H.; Pham, V.H.; Kwon, W.T. Slurry spray coating of carbon steel for use in oxidizing and humid environments. Ceram. Int. 2018, 44, 8306–8313. [Google Scholar] [CrossRef]
- Wang, C.; Chen, S. The high-temperature oxidation behavior of hot-dipping Al–Si coating on low carbon steel. Surf. Coat. Technol. 2006, 200, 6601–6605. [Google Scholar] [CrossRef]
- Yang, C.H.; Lin, S.N.; Chen, C.H.; Tsai, W.T. Effects of temperature and straining on the oxidation behavior of electrical steels. Oxid. Met. 2009, 72, 145–157. [Google Scholar] [CrossRef]
- Ren, Q.; He, X.; Wu, X. Effects of nano-additives on the structure and properties of 99% alumina ceramics. Rare Met. Mater. Eng. 2008, 37, 452–454. [Google Scholar]
- Yin, J.; Wang, X.; Zhang, Y.; Zhao, C.; Wang, X.; Gao, H.; Yang, J. Effect of sintering aids on high purity alumina ceramic densification process. Chin. Rare Earths 2014, 35, 16–20. [Google Scholar]
- Chen, R.Y.; Yuen, W.Y.D. The effects of steel composition on the oxidation kinetics, scale structure, and scale-steel interface adherence of low and ultra-low carbon steels. Mater. Sci. Forum 2006, 522–523, 451–460. [Google Scholar] [CrossRef]
- Le, M.T.; Kim, C.; Lee, J. Effect of silica addition on ceramic layer in centrifugal-thermit reaction. Mater. Trans. 2008, 49, 1410–1414. [Google Scholar] [CrossRef]
- Jiang, Z. Research on TG-DSC of carbon steel oxidation kinetics. Hot Work. Technol. 2015, 44, 89–91. [Google Scholar]
- Nagelberg, A.S. Observations on the role of Mg and Si in the directed oxidation of Al-Mg-Si alloys. J. Mater. Res. 1992, 7, 265–268. [Google Scholar] [CrossRef]
- Aksay, I.A.; Pask, J. A stable and metastable equilibria in system SiO2-Al2O3. J. Am. Ceram. Soc. 1975, 58, 507–512. [Google Scholar] [CrossRef]
- Zhu, Z.; Wei, Z.; Sun, W.; Hou, J.; He, B.; Dong, Y. Cost-effective utilization of mineral-based raw materials for preparation of porous mullite ceramic membranes via in-situ reaction method. Appl. Clay Sci. 2016, 120, 135–141. [Google Scholar] [CrossRef]
- Odashima, H.; Kitayama, M. Oxidation-inhibition mechanism and performance of a new protective coating for slab reheating of 3% Si-steel. ISIJ Int. 1990, 30, 255–264. [Google Scholar] [CrossRef]
- Dong, Y.; Lu, H.; Cui, J.; Yan, D.; Yin, F.; Li, D. Mechanical characteristics of FeAl2O4 and AlFe2O4 spinel phases in coatings—A study combining experimental evaluation and first-principles calculations. Ceram. Int. 2017, 43, 16094–16100. [Google Scholar] [CrossRef]
- Hazen, R.M.; Jeanloz, R. Wüstite (Fe1−xO): A review of its defect structure and physical-properties. Rev. Geophys. 1984, 22, 37–46. [Google Scholar] [CrossRef]
- Freer, R.; Oreilly, W. The diffusion of Fe2+ ions in spinels with relevance to the process of maghemitization. Miner. Mag. 1980, 43, 889–899. [Google Scholar] [CrossRef]



| No. | Al2O3 | Sodium Silicate Solution (40 wt %) | SiO2@Al | Surface Active Agent Sodium Polyacrylate | H2O |
|---|---|---|---|---|---|
| 1 | 70.5 | 23.5 | 0 | 4 | 2 |
| 2 | 66 | 21 | 5 | 4 | 4 |
| 3 | 62 | 18.5 | 10 | 4 | 5.5 |
| 4 | 58 | 16 | 15 | 4 | 7 |
| 5 | 54 | 13.5 | 20 | 4 | 8.5 |
| 6 | 50 | 11 | 25 | 4 | 10 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yu, B.; Fu, G.; Cui, Y.; Zhang, X.; Tu, Y.; Du, Y.; Zuo, G.; Ye, S.; Wei, L. Influence of Silicon-Modified Al Powders (SiO2@Al) on Anti-oxidation Performance of Al2O3-SiO2 Ceramic Coating for Carbon Steel at High Temperature. Coatings 2019, 9, 167. https://doi.org/10.3390/coatings9030167
Yu B, Fu G, Cui Y, Zhang X, Tu Y, Du Y, Zuo G, Ye S, Wei L. Influence of Silicon-Modified Al Powders (SiO2@Al) on Anti-oxidation Performance of Al2O3-SiO2 Ceramic Coating for Carbon Steel at High Temperature. Coatings. 2019; 9(3):167. https://doi.org/10.3390/coatings9030167
Chicago/Turabian StyleYu, Bo, Guoyan Fu, Yanbin Cui, Xiaomeng Zhang, Yubo Tu, Yingchao Du, Gaohong Zuo, Shufeng Ye, and Lianqi Wei. 2019. "Influence of Silicon-Modified Al Powders (SiO2@Al) on Anti-oxidation Performance of Al2O3-SiO2 Ceramic Coating for Carbon Steel at High Temperature" Coatings 9, no. 3: 167. https://doi.org/10.3390/coatings9030167
APA StyleYu, B., Fu, G., Cui, Y., Zhang, X., Tu, Y., Du, Y., Zuo, G., Ye, S., & Wei, L. (2019). Influence of Silicon-Modified Al Powders (SiO2@Al) on Anti-oxidation Performance of Al2O3-SiO2 Ceramic Coating for Carbon Steel at High Temperature. Coatings, 9(3), 167. https://doi.org/10.3390/coatings9030167




