1. Introduction
Polymerous base materials have great potential in the building industry; they have high strength, excellent resistance, and harden quickly [
1]. For this research, selected binders were based on epoxide and polyurethane resins. Epoxide resins are known to have very good strength, surface hardness, bond to cement based composites, and resistance to aggressive substances [
2]. Polyurethanes offer very good flexibility, capability of bridging cracks, and small fissures [
3].
Solidification is an interesting way of using dangerous wastes as secondary raw materials. Solidification is based on mixing hazardous waste with a solidifying agent; it is advisable to use the maximum possible amount of hazardous waste for this process [
4,
5]. On using suitable solidification technology, hazardous waste (HW) can be reprocessed to a progressive material—solidification product [
6]. Portland cement is the most widely used for solidification as a solidifying agent. Many formulations have been developed for the process, according to the kind of hazardous wastes, containing heavy metals, polychlorinated biphenyl (PCB), etc. Fly ash, lime, slag, soluble silicates, clay, etc. can also be used as a solidifying agent instead or in combination with Portland cement for solidification of HW [
7].
Fly ash obtained from coal combustion is frequently used in concrete as a cost-effective substitute for Portland cement [
5]. Like most common industrial by-products, fly ash is a complex anthropogenic material, and processing it prevents many problems, like the toxicity caused by the presence of various organic and inorganic compounds derived from the type of coal used [
8]. This material is a mix of oxides rich in silicon (SiO
2), iron (Fe
2O
3), and aluminum (Al
2O
3) [
9]. The American US Energy Information Administration (USEIA, 2014) states that 40% of the energy produced worldwide is made from coal; India and China are the largest producers of coal. The growing consumption of energy has caused a considerable increase in the production of fly ash. Compared to 2005 when fly ash production was around 500 million tons, fly ash production increased to 750 million tons in 2015 [
10,
11]. This enormous amount of produced fly ash presents a major ecological threat connected to storage and disposal [
8]. The utilization of fly ash as a filler for epoxide resin is a topic dealt with by many authors all over the world. For example, Krishna et al. [
12] observed the influence of the action of various proportions of resin and filler (fly ash and wood dust) on the physical and mechanical properties of composites. They noted that increasing the proportion of filler not only reduces the price of the product, but also has a potential influence on its life span [
12]. Currently, quartz flour (dust) is the most widely used filler in organic coatings, mainly because of the quartz high Mohs hardness value. The quartz filler showed the mechanical and anticorrosive behavior of epoxy coatings of steel materials [
13]. In order to use the wastes in polymeric patching and grouting materials, and especially in coatings to improve the environmental aspects of its production, natural river and crushed fine aggregate, sand, and quartz flour can be replaced in different percentages by natural waste or sub-products, such as silica fume, milled recycle glass, fly ash, polystyrene granules, saw dust, polyethylene terephthalate (PET) particle, etc. [
14,
15,
16,
17,
18,
19,
20].
Lokuge and Aravinthan [
1] noted that the use of 0%–10% filler in the form of fly ash increases its physical and mechanical properties. However, they also concluded that further addition worsens these properties. In a study by Sosoi et al. [
14], fly ash was added as a filler, and aggregate fraction 0–4 mm was replaced in different dosages by saw dust, where epoxy resin was used as a binder. Waste substitution influenced the properties of the concrete. This waste decreased the density of the hardened polymer concrete to values less than 2000 kg/m
3, and saw dust decreased the workability of the polymer concrete. For dosages of 25% and 50% saw dust, the polymer concrete with waste substitution presented higher values of compressive strength than in the control mix (12.4% of epoxy resin, fly ash as filler in a dosage of 12.8%, and natural river aggregates: 37.4% of the size fraction 0–4 mm and 37.4% of the fraction 4–8 mm). The highest value of compressive strength—25%—was obtained for polymer concrete with saw dust substitution of aggregate, a value bigger than that of the control mix with 18.1% [
14].
Polyurethanes were discovered in 1937 when Bayer was synthesizing polymer fibers to compete with nylon—he developed the first fiber-forming polyurethane. His invention ranks among the major breakthroughs in polymer chemistry, but Bayer’s polymer was dismissed as impractical by his superiors at the company I.G. Farbenindustrie, which produced typical polyurethane, in addition to polyurethane linkages, aliphatic and aromatic hydrocarbons, esters, ethers, amides, urea, and isocyanurate groups [
21]. Polyurethane (PUR) elastomers are largely applied to industrial and consumer products, particularly in the fields of heavy pressure, load, impact, and wear because they possess excellent comprehensive properties, such as high wear, oil, and corrosion resistance, high elasticity and damping, good adhesion to other materials, etc. [
22].
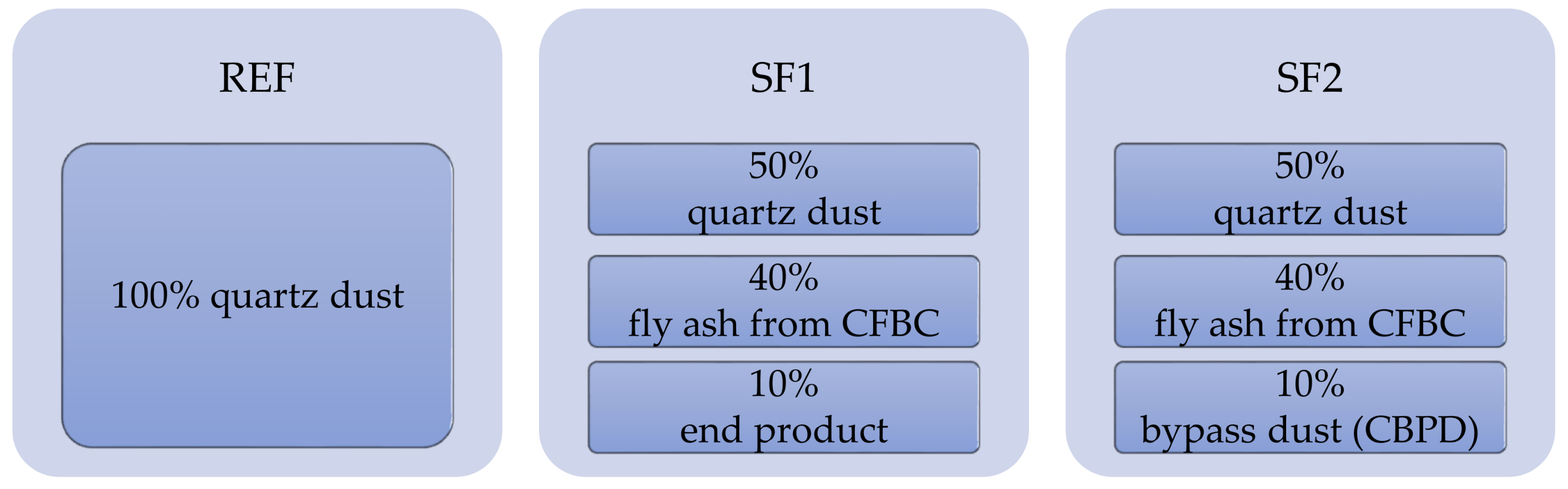
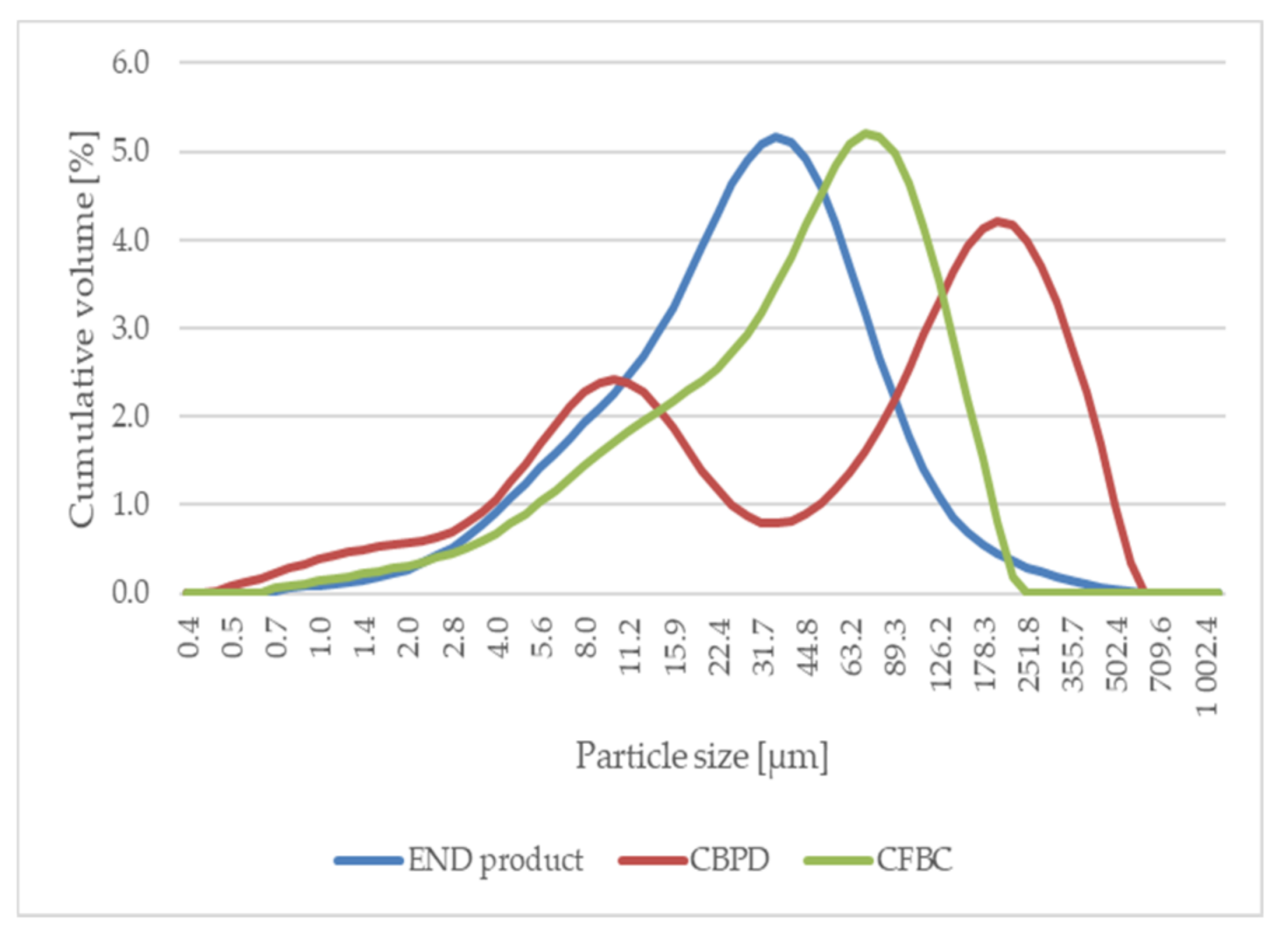
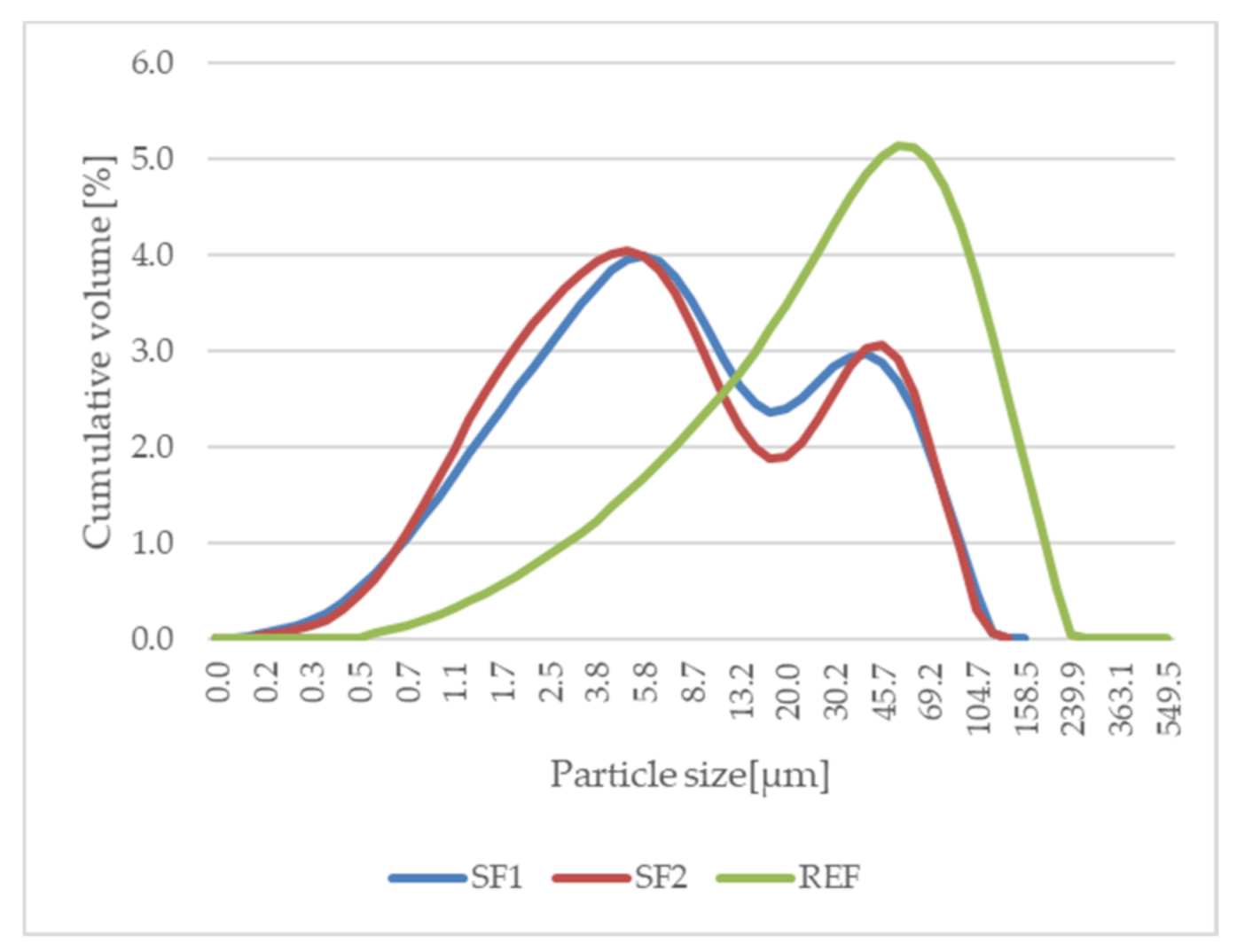
The main aim of this research was, in particular, to determine ways of using specific type of dry by-products (end product and cement bypass dust (CBPD)). These are classified as hazardous waste based on their dangerous properties. For storage and handling of such waste products, it is necessary to prevent the possible release of pollutants (lead, mercury, arsenic, etc.) into the ambient environment. Successful utilization of these wastes will, therefore, have significant environmental benefits. It is assumed that the two-stage stabilization and solidification (S/S) of these wastes could lead to perfect incorporation of the pollutants. The first stage of S/S is the formation of a solidification product in the form of a fine filler. The second step is to incorporate this filler into a polymer matrix, possibly improving the physical and mechanical properties and chemical resistance of the developed polymer coatings.
4. Conclusions
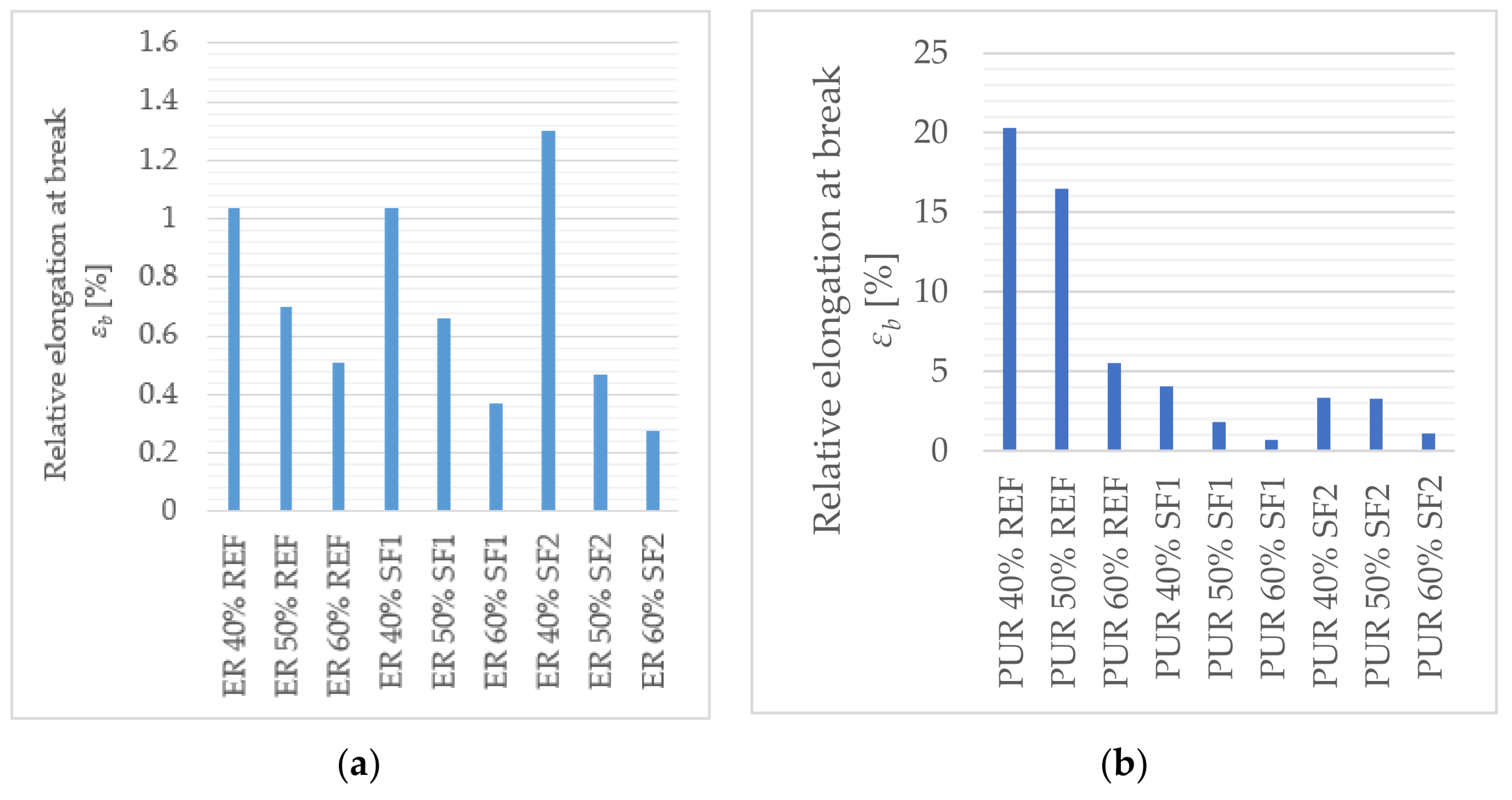
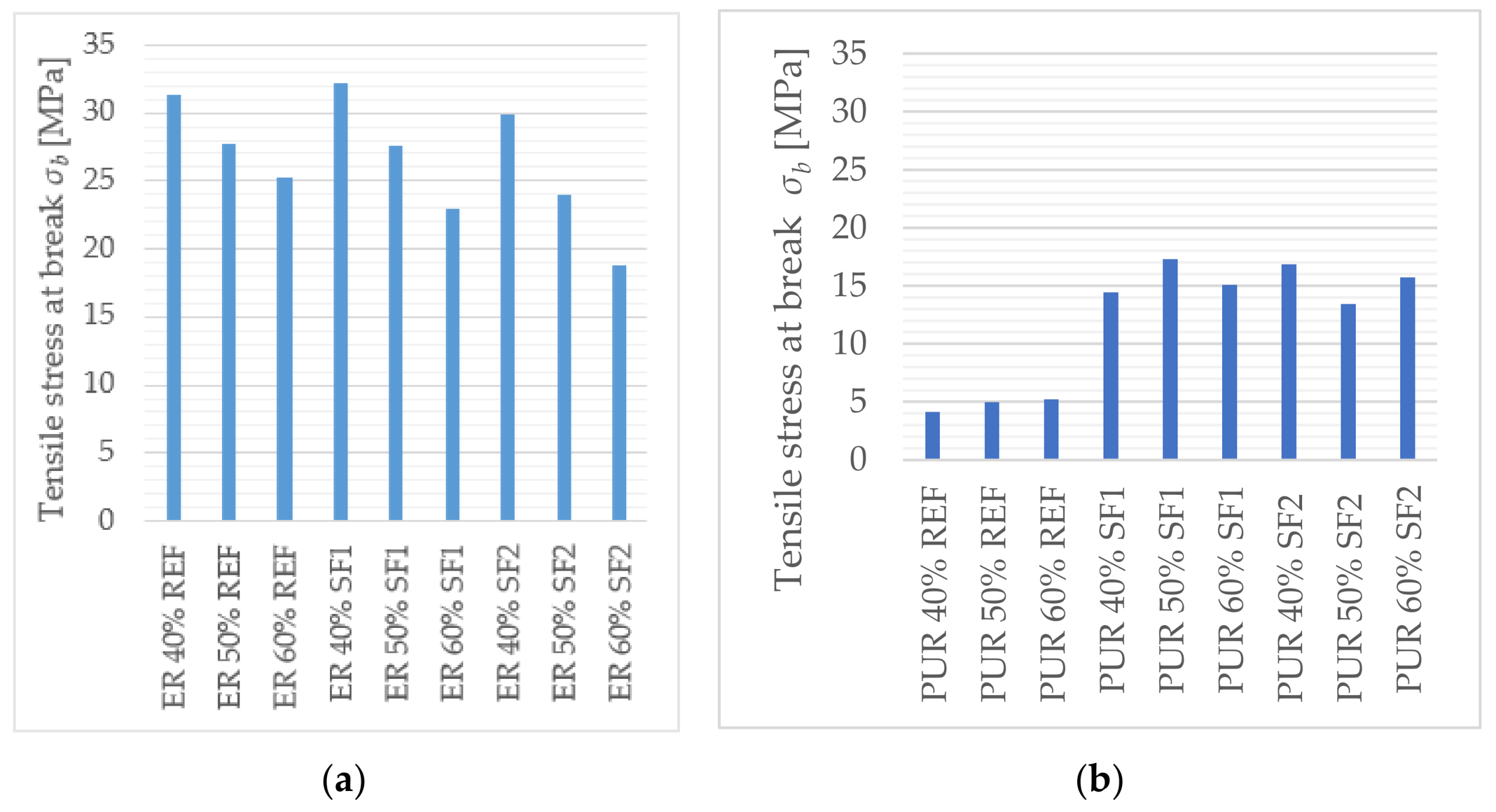
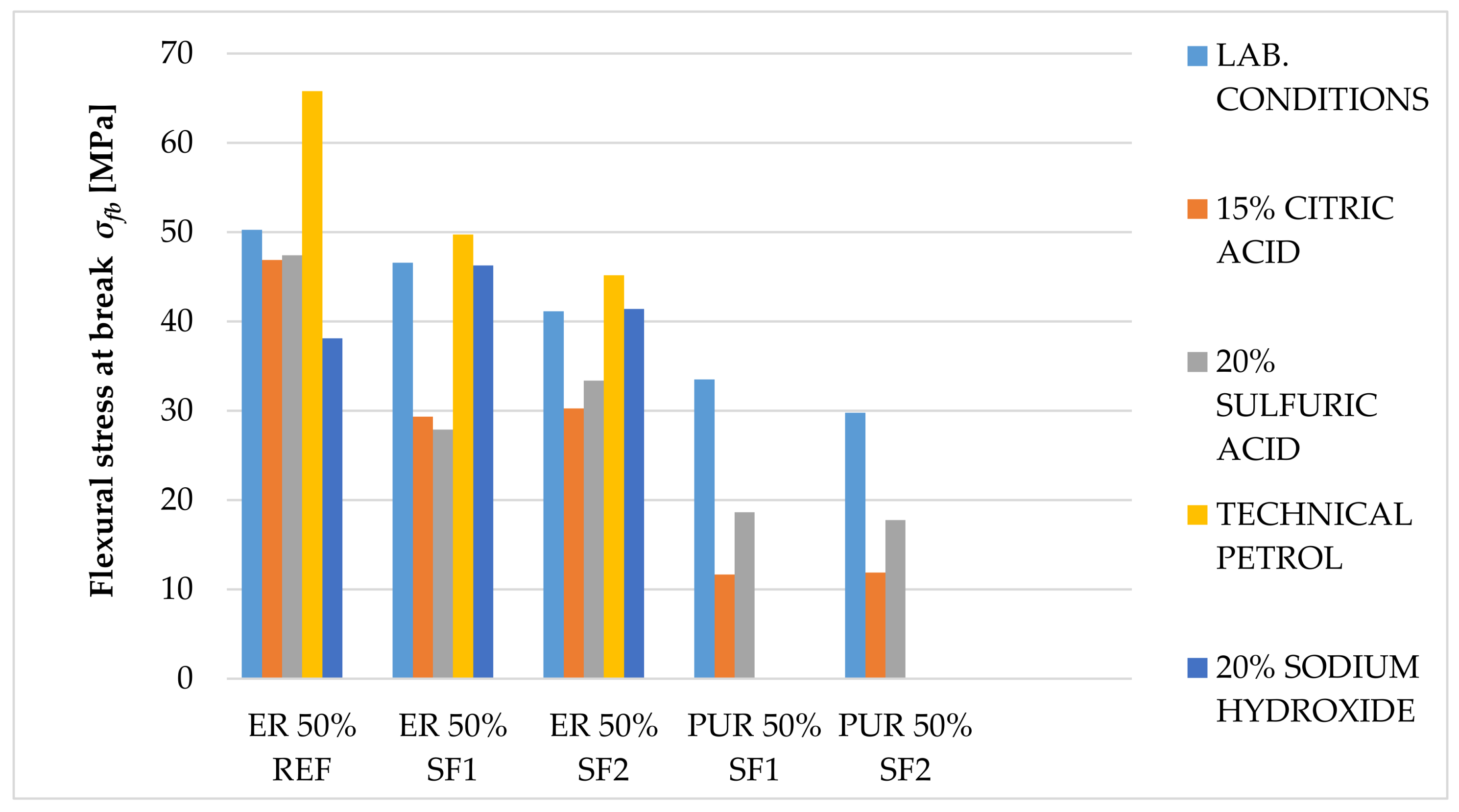
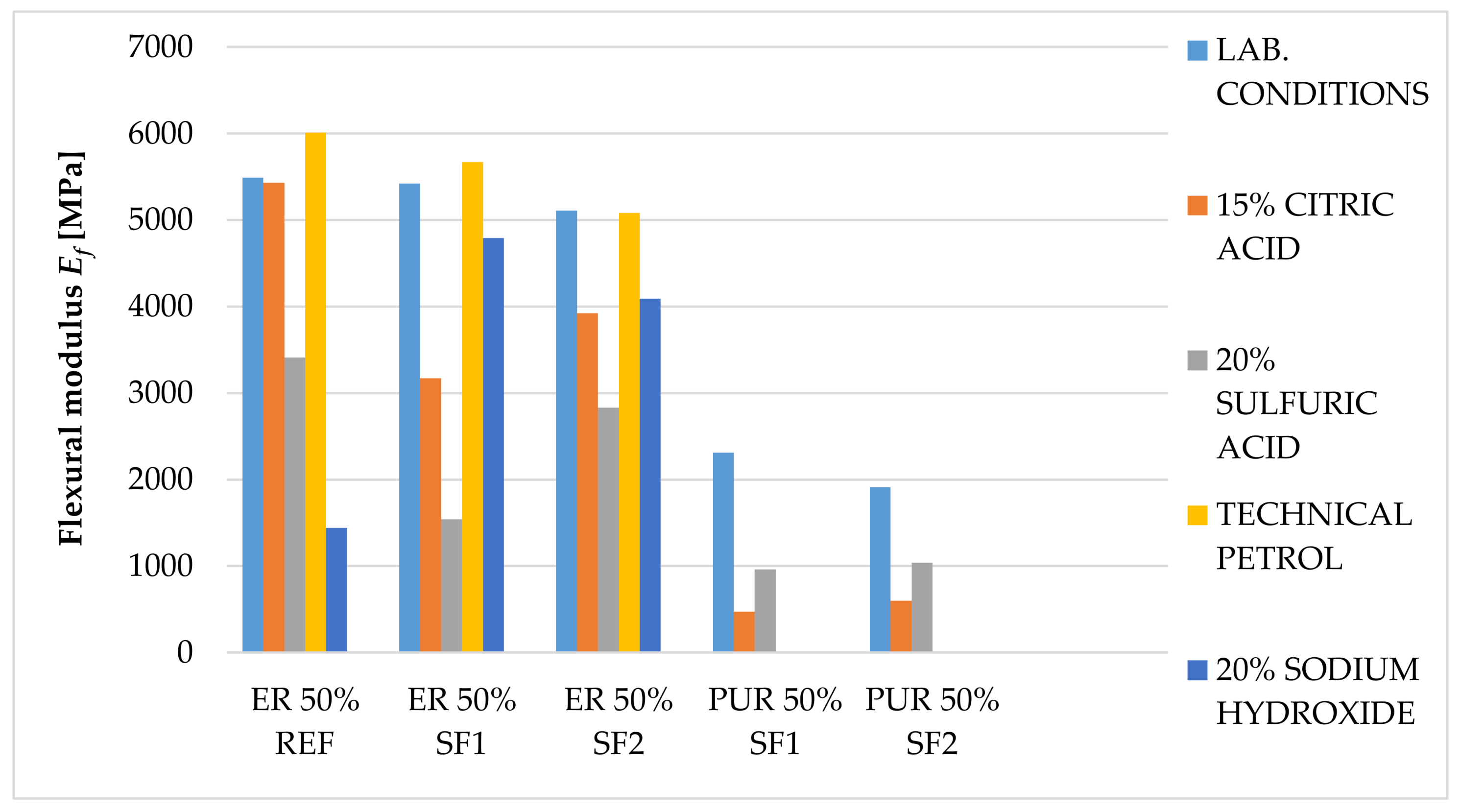
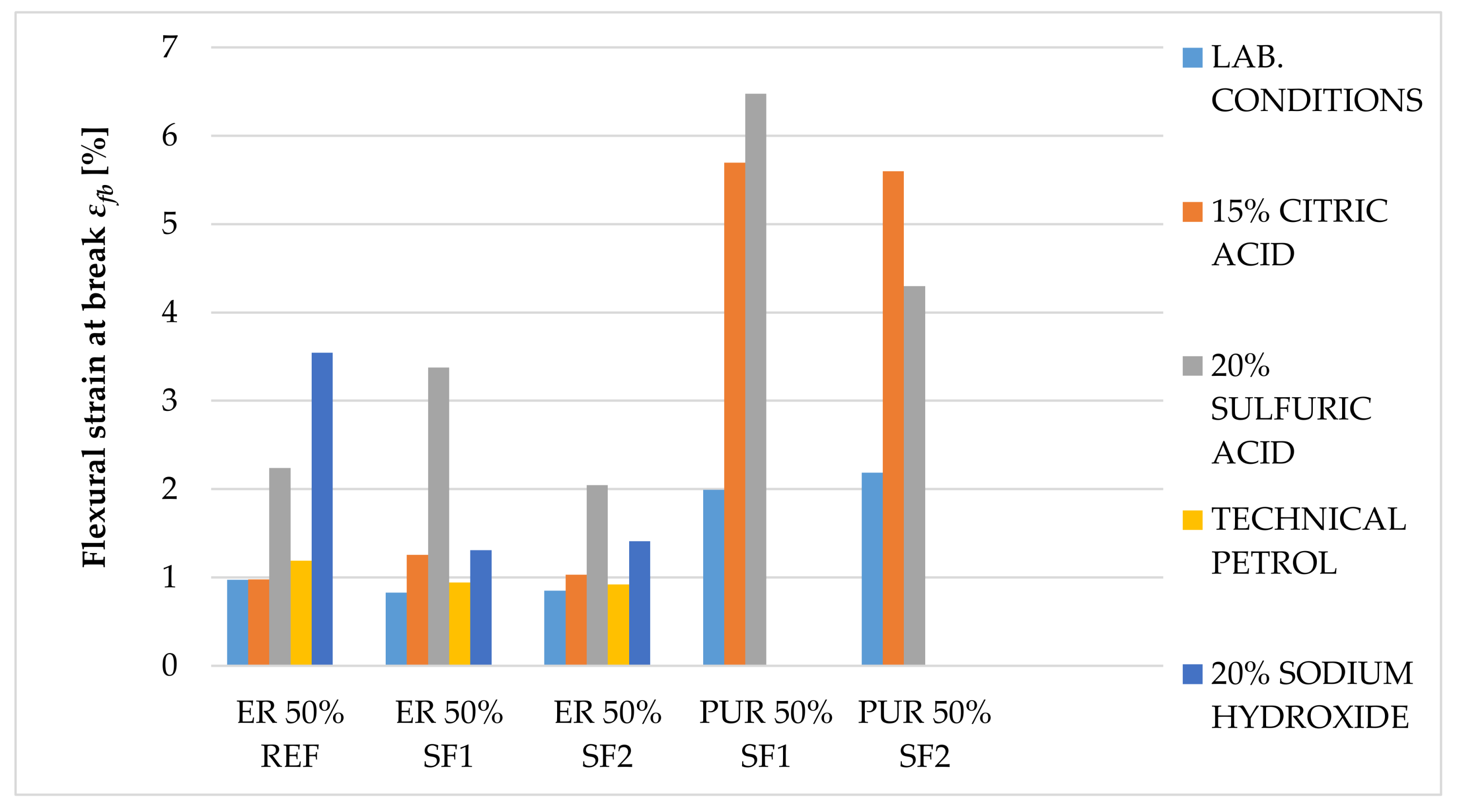
The main goal of this paper was to verify the possibility of using solidified hazardous waste (solidification products), a progressive filler produced during the dry process of solidification, as a filler in polymeric protective coatings for the secondary protection of reinforced concrete and metal structures. With respect to the planned use of these materials, in particular for concrete and metal surfaces, the following properties were determined: surface hardness, impact resistance, tensile properties and flexural properties, adhesion, and chemical resistance, as well as a comparison of these parameters with the reference coating material containing only the primary input materials. In the preliminary verification (the application test and consistency determination), it was found that 50% filler seems to be optimal. At this proportion, the material showed optimal consistency for the proposed application, and the cured surface was smooth and free from irregularities. The test results imply that the coating materials created using pretreated hazardous wastes offer adhesion, resistance to abrasion, surface hardness, impact resistance, and relative elongation at break, compared to the reference material. The epoxide binder (ER) created a stronger but less flexible structure, compared to the polyurethane binder (PUR), which was proven mainly in the tests of impact resistance and tensile stress. For tensile strengths, coatings with epoxy resin had significantly higher values than polyurethane coatings. Polyurethane-based coatings have better resistance to mechanical damage because of their lower modulus of elasticity, which implies that they can better distribute mechanical loads in their inner structure. Epoxide-based coatings showed better resistance to chemical exposure. Chemical stress caused the flexural properties to change significantly. Acetic acid caused the highest level of degradation for all tested samples (their flexural properties could not be determined); all tested specimens showed a decrease in their flexural strength and modulus of elasticity after exposure to citric and sulfuric acid, and technical petrol caused a slight increase in flexural strength with the epoxy samples. Samples with the most severe degradation also showed a maximal decrease in their flexural modulus (acetic acid and sodium hydroxide with PUR coatings). In conclusion, the use and implementation of specifically prepared fillers, solidification products containing hazardous waste (SF1, SF2), enabled the incorporation of pollutants into the polymerous matrix and the developed coatings portrayed the required physical and mechanical properties, comparable to the reference materials. These coatings can thus be used for secondary protection in construction.