Influence of TiO2-Coating Layer on Nanoporous Alumina Membranes by ALD Technique
Abstract
:1. Introduction
2. Materials and Methods
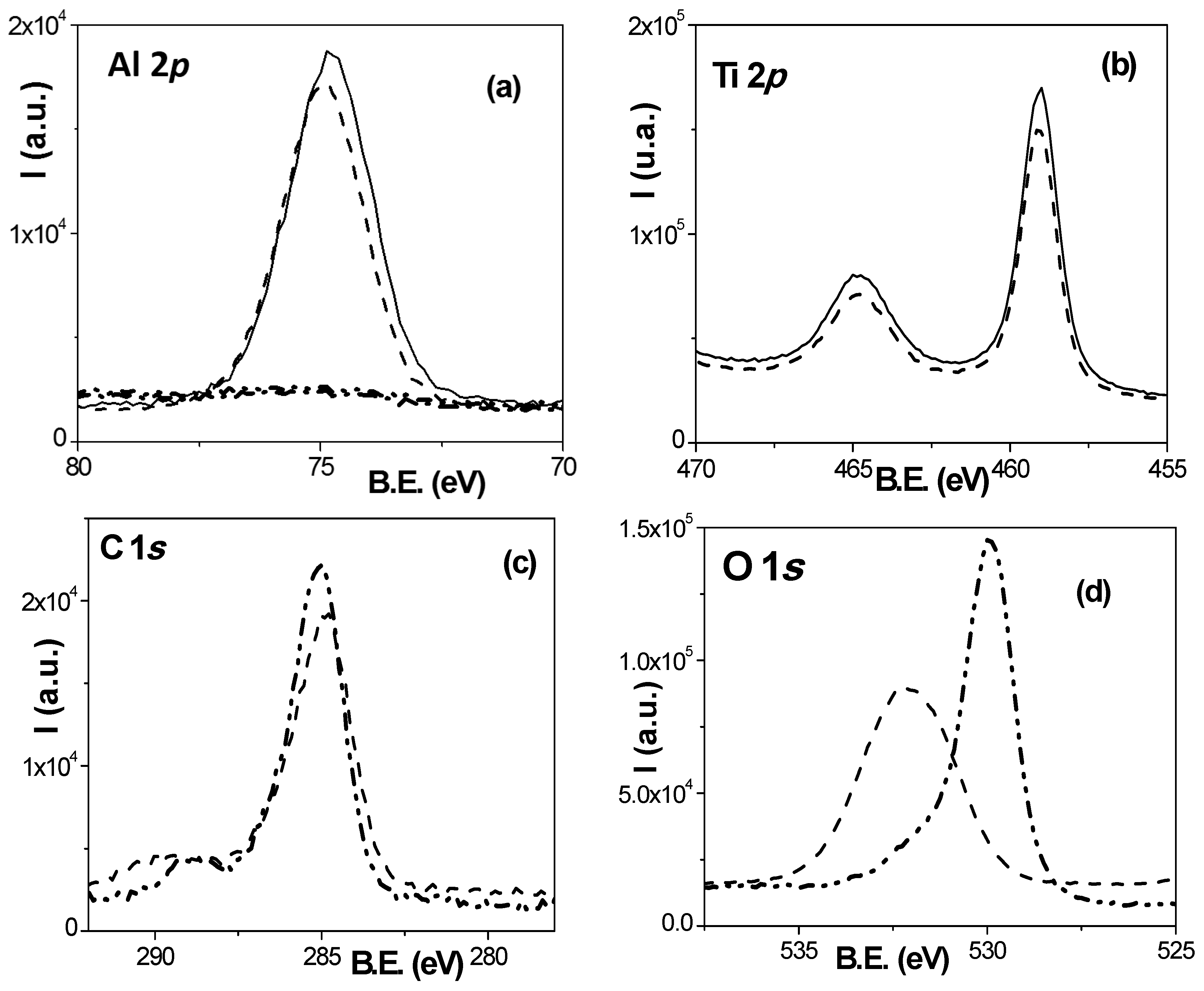
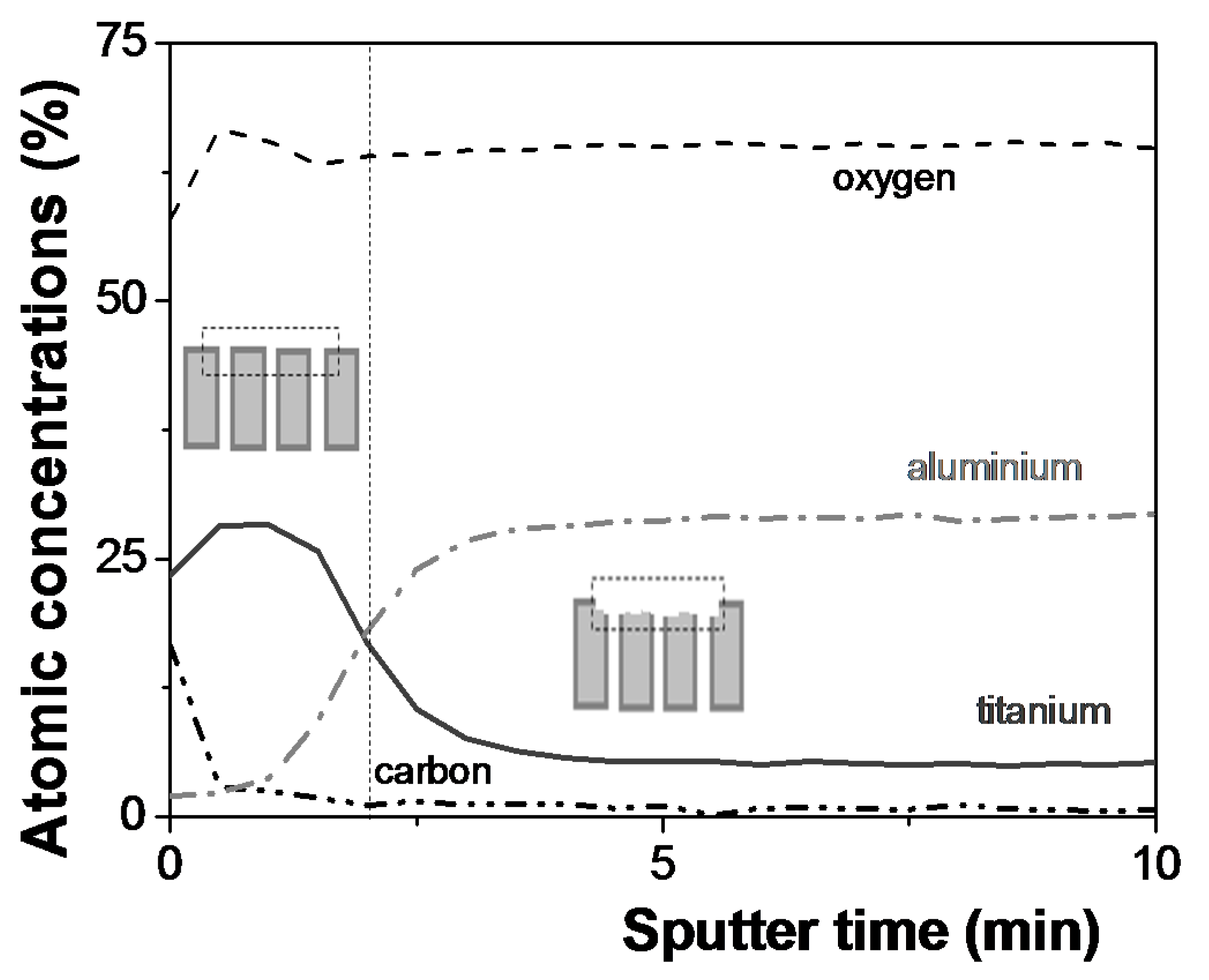
2.1. Chemical Surface Analysis
2.2. Scanning and Transmission Electron Microscopy Characterizations
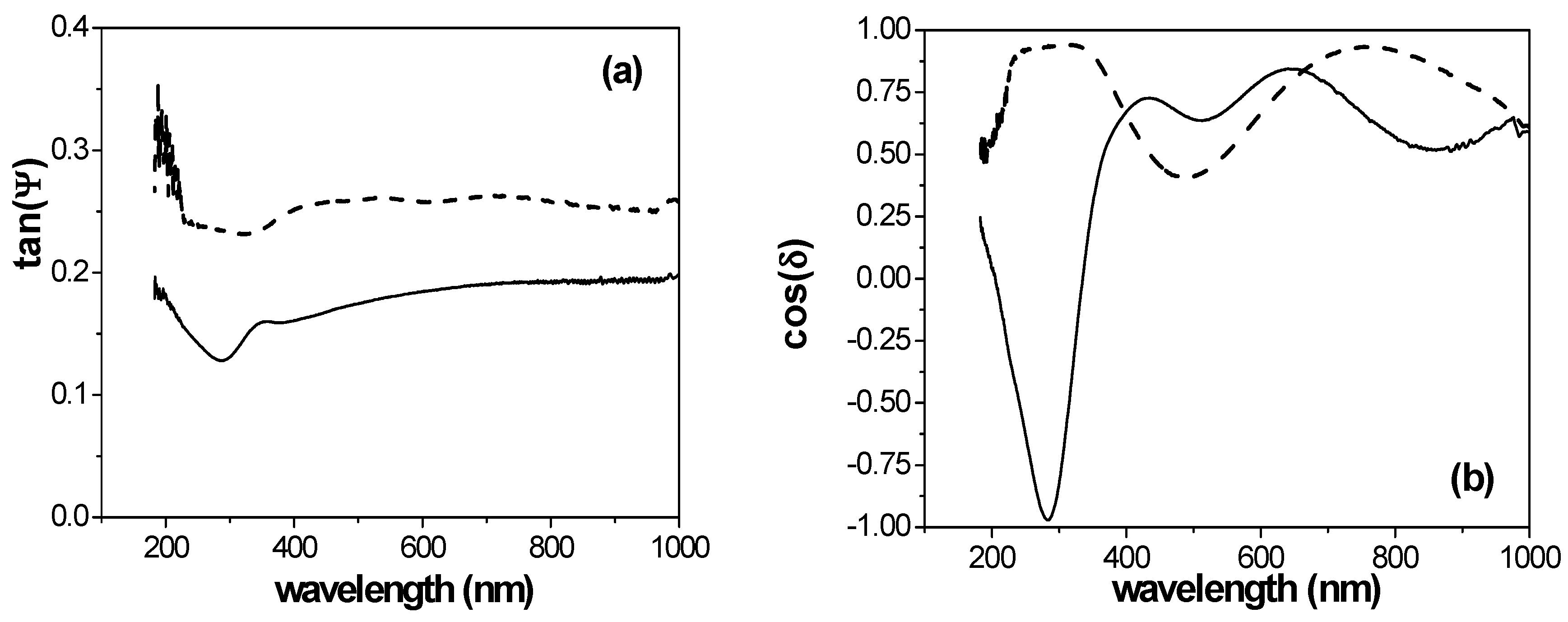
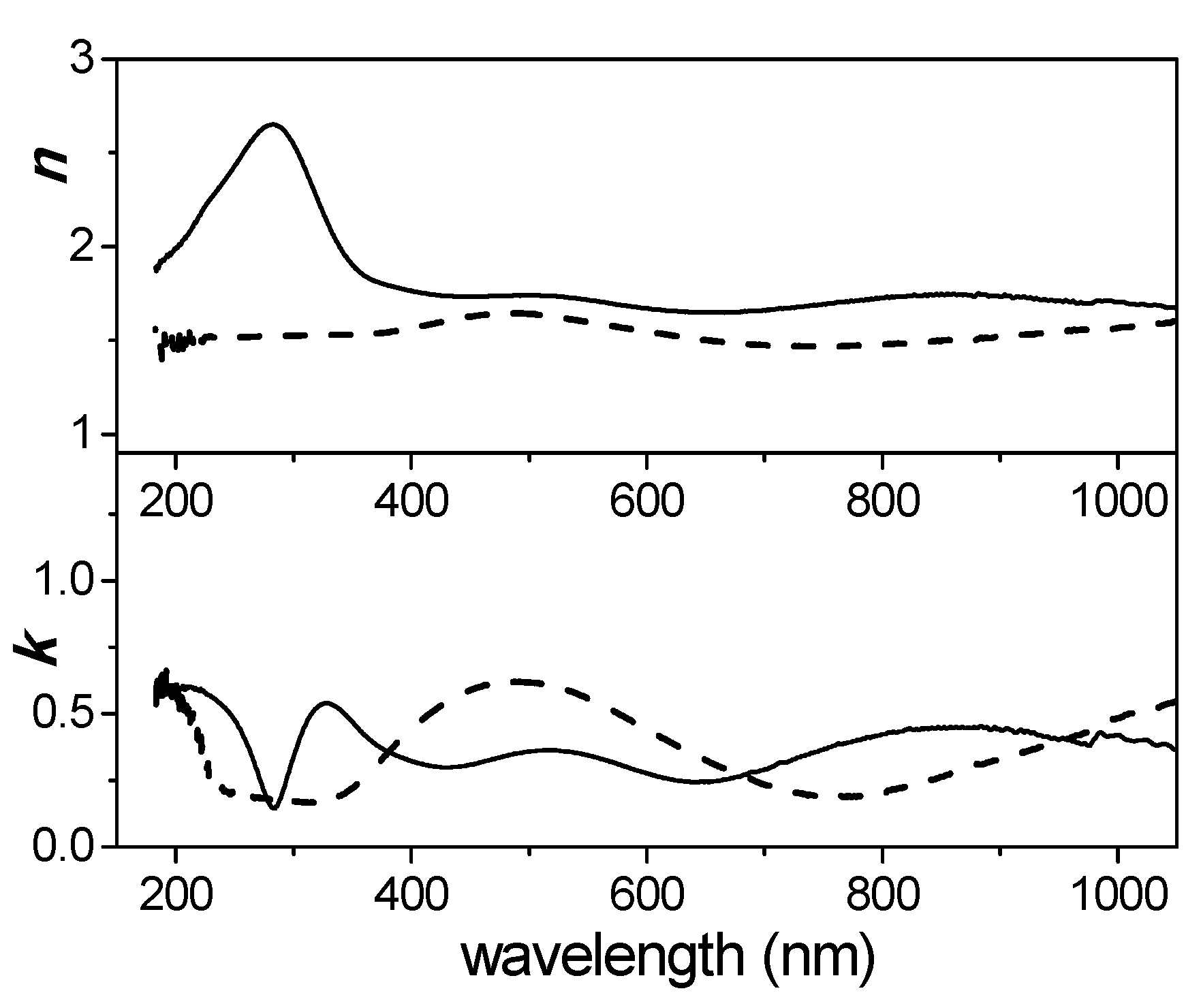
2.3. Spectroscopic Ellipsometry Measurements
2.4. Concentration Potential Measurements
3. Results
3.1. Chemical and Morphological Characterization of the Nanoporous Alumina-Based Membranes
3.2. Spectroscopic Ellipsometry Analysis
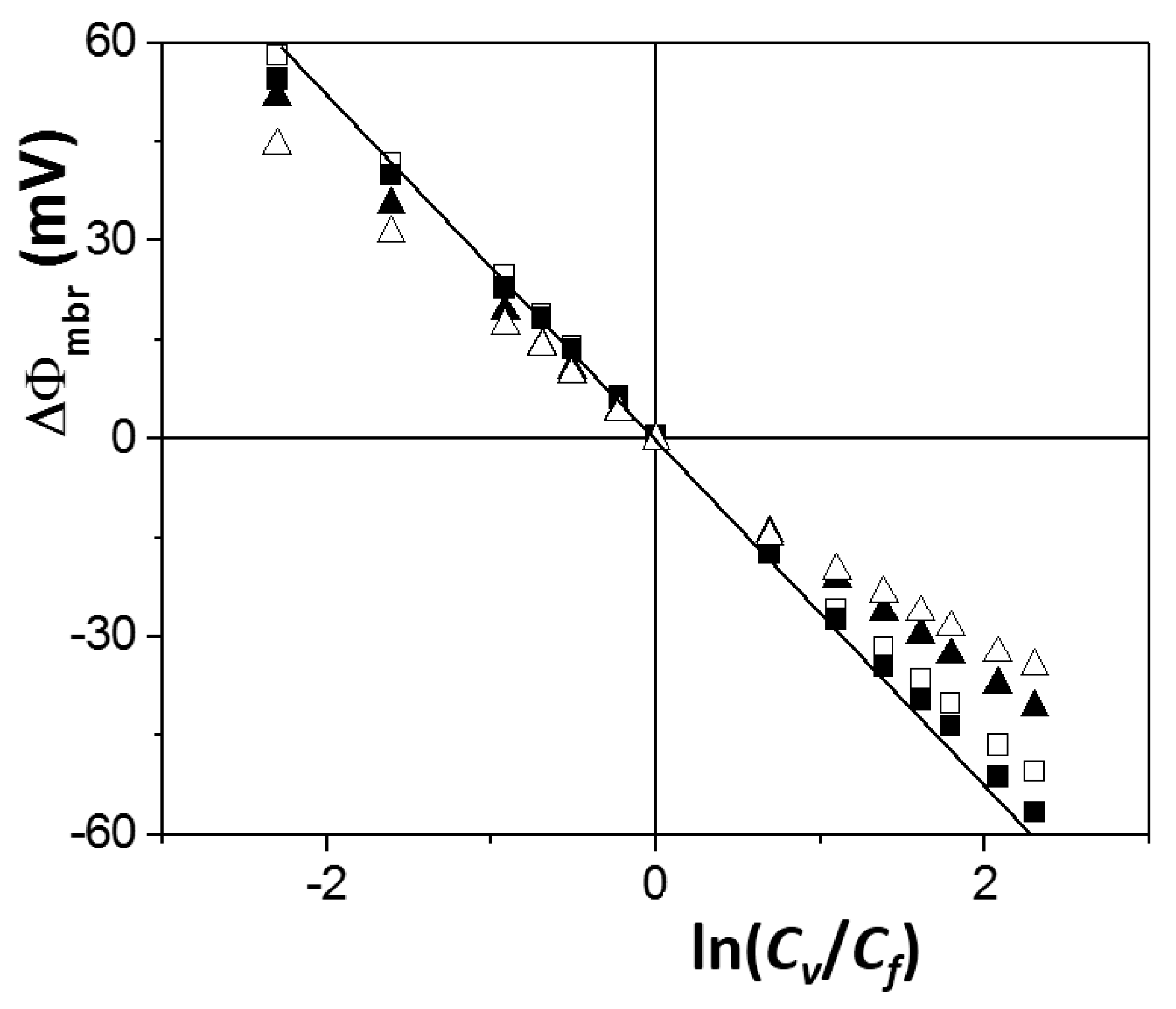
3.3. Concentration Potential Analysis
4. Discussion
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Masuda, H.; Fukuda, K. Ordered metal nanohole arrays made by a two-step replication of honeycomb structures of anodic alumina. Science 1995, 268, 1466–1468. [Google Scholar] [CrossRef] [PubMed]
- Hohlbein, J.; Steinhart, M.; Schiene-Fisher, C.; Benda, A.; Hof, M.; Hübner, C.G. Confined Diffusion in Ordered Nanoporous Alumina Membranes. Small 2007, 3, 380–385. [Google Scholar] [CrossRef] [PubMed]
- Jiang, X.; Mishra, N.; Turner, J.N.; Spencer, M.G. Diffusivity of sub-1000 Da molecules in 40 nm silicon-based alumina pores. Microfluid. Nanofluid. 2008, 5, 695–701. [Google Scholar] [CrossRef]
- Karnik, R.; Fan, R.; Yue, M.; Li, D.; Yang, P.; Majumdar, A. Electrostatic Control of Ions and Molecules in Nanofluidic Transistors. Nano Lett. 2005, 5, 943–948. [Google Scholar] [CrossRef] [PubMed]
- Lee, W.; Park, S.J. Porous Anodic Aluminum Oxide: Anodization and Templated Synthesis of Functional Nanostructures. Chem. Rev. 2014, 114, 7487–7556. [Google Scholar] [CrossRef] [PubMed]
- Romero, V.; Vega, V.; García, J.; Zierold, R.; Nielsch, K.; Prida, V.M.; Hernando, B.; Benavente, J. Changes in Morphology and Ionic Transport Induced by ALD SiO2 Coating of Nanoporous Alumina Membranes. ACS Appl. Mater. Interfaces 2013, 5, 3556–3564. [Google Scholar] [CrossRef] [PubMed]
- George, S.M. Atomic Layer Deposition: An Overview. Chem. Rev. 2010, 110, 111–131. [Google Scholar] [CrossRef] [PubMed]
- Narayan, R.J.; Monteiro-Riviere, N.A.; Brigmon, R.L.; Pellin, M.J.; Elam, J.W. Atomic Layer Deposition of TiO2 Thin Films on Nanoporous Alumina Templates: Medical Applications. JOM 2009, 61, 12–16. [Google Scholar] [CrossRef]
- Petrochenko, P.E.; Kumar, G.; Fu, W.; Zhang, Q.; Zheng, J.; Liang, C.; Goering, P.L.; Narayan, R.J. Nanoporous Aluminum Oxide Membranes Coated with Atomic Layer Deposition-Grown Titanium Dioxide for Biomedical Applications: An In Vitro Evaluation. J. Biomed. Nanotechnol. 2015, 11, 2275–2285. [Google Scholar] [CrossRef] [PubMed]
- Kemell, M.; Pore, V.; Tupala, J.; Ritala, M.; Leskelä, M. Atomic Layer Deposition of Nanostructured TiO2 Photocatalysts via Template Approach. Chem. Mater. 2007, 19, 1816–1820. [Google Scholar] [CrossRef]
- Ng, C.J.W.; Gao, H.; Tan, T.T.Y. Atomic Layer Deposition of TiO2 nanostructures for self-cleaning applications. Nanotechnology 2008, 19. [Google Scholar] [CrossRef] [PubMed]
- Liang, Y.-C.; Wang, C.-C.; Kei, C.-C.; Hsueh, Y.-C.; Cho, W.-H.; Perng, T.-P. Photocatalysis of Ag-Loaded TiO2 Nanotube Arrays Formed by Atomic Layer Deposition. J. Phys. Chem. C 2011, 115, 9498–9502. [Google Scholar] [CrossRef]
- Cameron, M.A.; Gartland, I.P.; Smith, J.A.; Diaz, S.F.; George, S.M. Atomic Layer Deposition of SiO2 and TiO2 in Alumina Tubular Membranes: Pore Reduction and Effect of Surface Species on Gas Transport. Langmuir 2000, 16, 7435–7444. [Google Scholar] [CrossRef]
- Romero, V.; Vega, V.; García, J.; Prida, V.M.; Hernando, B.; Benavente, J. Ionic transport across tailored nanoporous anodic alumina membranes. J. Colloids Interface Sci. 2012, 376, 40–46. [Google Scholar] [CrossRef] [PubMed]
- Prida, V.M.; Vega, V.; García, J.; Iglesias, L.; Hernando, B.; Minguez-Bacho, I. Electrochemical Methods for Template-Assisted Synthesis of Nanostructured Materials. In Magnetic Nano- and Microwires; Vázquez, M., Ed.; Woodhead Publishing: Cambridge, UK, 2015; pp. 1–39. ISBN 9780081001646. [Google Scholar]
- Meng, X.; Norouzi Banis, M.; Geng, D.; Li, X.; Zhang, Y.; Li, R.; Abou-Rachid, H.; Sun, X. Controllable atomic layer deposition of one-dimensional nanotubular TiO2. Appl. Surf. Sci. 2013, 266, 132–140. [Google Scholar] [CrossRef]
- Moulder, J.F.; Stickle, W.F.; Sobol, P.E.; Bomben, K.D. Handbook of X-ray Photoelectron Spectroscopy; Chastain, J., Ed.; Perkin-Elmer Corporation: Minneapolis, MN, USA, 1992. [Google Scholar]
- Schneider, C.A.; Rasband, W.S.; Eliceiri, K.W. NIH Image to ImageJ: 25 years of image analysis. Nat. Methods 2012, 9, 671–675. [Google Scholar] [CrossRef] [PubMed]
- Romero, V.; Vázquez, M.I.; Benavente, J. Study of ionic and diffusive transport through a regenerated cellulose nanoporous membrane. J. Membr. Sci. 2013, 433, 152–159. [Google Scholar] [CrossRef]
- Nielsch, K.; Choi, J.; Schwirn, K.; Wehrspohn, R.B.; Gösele, U. Self-ordering regimes of porous alumina: The 10% porosity rule. Nano Lett. 2002, 2, 677–680. [Google Scholar] [CrossRef]
- Weirich, T.E.; Winterer, M.; Seifried, S.; Fuess, H.; Hahn, H. Rietveld analysis of electron powder diffraction data from nanocrystalline anatase TiO2. Ultramicroscopy 2000, 81, 263–270. [Google Scholar] [CrossRef]
- Ariza, M.J.; Benavente, J.; Rodríguez-Castellón, E. The Capability of X-ray Photoelectron Spectroscopy in the Characterization of Membranes: Correlation between Surface Chemical and Transport Properties in Polymeric Membranes. In Handbook of Membrane Research: Properties, Performance and Applications; Nova Science Publishers, Inc.: New York, NY, USA, 2009; Chapter 7; pp. 257–290. ISBN 978-1-60741-638-8. [Google Scholar]
- Vega, V.; Gelde, L.; González, A.S.; Prida, V.M.; Hernando, B.; Benavente, J. Diffusive transport through surface functionalized nanoporous alumina membranes by atomic layer deposition of metal oxides. J. Ind. Eng. Chem. 2017, 52, 66–72. [Google Scholar] [CrossRef]
- Thompson, D.W.; Snyder, P.G.; Castro, L.; Yan, L. Optical characterization of porous alumina from vacuum ultraviolet to midinfrared. J. App. Phys. 2005, 97, 113511–113519. [Google Scholar] [CrossRef]
- Gâlca, A.C.; Kooij, E.S.; Wormeester, H.; Salm, C.; Lecca, V.; Rector, J.H.; Poelsema, B. Structural and optical characterization of porous anodic aluminium oxide. J. Appl. Phys. 2003, 94, 4296–4305. [Google Scholar] [CrossRef]
- Ogielo, W.; Wormeester, H.; Wessling, M.; Benes, N.E. Spectroscopic ellipsometry analysis of a thin film composite membrane consisting of polysulphone on a porous α-alumina support. ACS Appl. Mater. Interfaces 2012, 4, 935–943. [Google Scholar] [CrossRef] [PubMed]
- Fujiwara, H. Spectroscopic Ellipsometry Principles and Applications; John Wiley & Sons: Chischester, UK, 2007; ISBN 9780470016084. [Google Scholar]
- Jeon, G.; Yang, S.Y.; Kim, J.K. Functional nanoporous membranes for drug delivery. J. Mater. Chem. 2012, 22, 14814–14834. [Google Scholar] [CrossRef]
- Kuo, T.-C.; Sloan, L.A.; Sweedler, J.V.; Bohn, P.W. Manipulating Molecular Transport through Nanoporous Membranes by Control of Electrokinetic Flow: Effect of Surface Charge Density and Debye Length. Langmuir 2001, 17, 6298–6303. [Google Scholar] [CrossRef]
- Robinson, R.A.; Stokes, R.H. Electrolyte Solutions, 2nd ed.; Dover Publications: New York, NY, USA, 2002; ISBN 0486422259. [Google Scholar]
- Sollner, K. The Electrochemistry of Porous Membranes, with Particular Reference to Ion Exchange Membranes and Their Use in Model Studies of Biophysical Interest. J. Macromol. Sci. Pure Appl. Chem. 1969, 3, 1–86. [Google Scholar] [CrossRef]
- Meyer, K.H.; Sievers, J.F. La perméabilité des membranes I. Théorie de la perméabilité ionique. Helv. Chim. Acta 1936, 19, 649–664. [Google Scholar] [CrossRef]
- Teorell, T. Transport phenomena in membranes eighth Spiers Memorial Lecture. Discuss. Faraday Soc. 1956, 21, 9–26. [Google Scholar] [CrossRef]
- Lakshminarayanaiah, N. Transport Phenomena in Membranes; Academic Press Inc.: New York, NY, USA, 1970; ISBN1 10: 0124342507. ISBN2 13: 9780124342507. [Google Scholar]
- Romero, V.; Vega, V.; García, J.; Prida, V.M.; Hernando, B.; Benavente, J. Effect of porosity and concentration polarization on electrolyte diffusive transport parameters through ceramic membranes with similar nanopore size. Nanomaterials 2014, 4, 700–711. [Google Scholar] [CrossRef] [PubMed]
- Romero, V.; Vázquez, M.I.; Cañete, S.; Vega, V.; García, J.; Prida, V.M.; Hernando, B.; Benavente, J. Frictional and Electrical Effects Involved in the Diffusive Transport through a Nanoporous Alumina Membrane. J. Phys. Chem. C 2013, 117, 25513–25518. [Google Scholar] [CrossRef]


| Membrane | rp (nm) | Dint (nm) | Θ (%) |
|---|---|---|---|
| Ox-NPAM | 16 ± 2 | 105 ± 5 | 8 |
| Ox-NPAM + TiO2 | 13 ± 2 | 105 ± 5 | 6 |
| Sf-NPAM | 13 ± 2 | 65 ± 5 | 13 |
| Sf-NPAM + TiO2 | 10 ± 2 | 65 ± 5 | 9 |
| Sample | C (%) | Al (%) | Ti (%) | O (%) | N (%) | Si (%) |
|---|---|---|---|---|---|---|
| Sf-NPAM a | 16.9 | 25.7 | – | 53.0 | – | 0.3 |
| Sf-NPAM + TiO2 | 19.2 | 0.6 | 23.5 | 53.5 | 0.7 | 0.5 |
| Ox-NPAM b | 23.8 | 25.3 | – | 48.3 | – | 0.3 |
| Ox-NPAM + TiO2 | 24.4 | 1.5 | 21.4 | 52.2 | 0.2 | 0.4 |
| Sample | n | k |
|---|---|---|
| Sf-NPAM | 1.6091 | 0.4909 |
| Sf-NPAM + TiO2 | 1.4606 | 0.3359 |
| Ox-NPAM | 1.5151 | 0.3657 |
| Ox-NPAM + TiO2 | 1.6527 | 0.2458 |
| Sample | tK+ | DCl−/DK+ |
|---|---|---|
| Sf-NPAM | 0.292 | 2.43 |
| Sf-NPAM + TiO2 | 0.162 | 5.17 |
| Ox-NPAM | 0.310 | 2.23 |
| Ox-NPAM + TiO2 | 0.187 | 4.35 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gelde, L.; Cuevas, A.L.; Martínez de Yuso, M.D.V.; Benavente, J.; Vega, V.; González, A.S.; Prida, V.M.; Hernando, B. Influence of TiO2-Coating Layer on Nanoporous Alumina Membranes by ALD Technique. Coatings 2018, 8, 60. https://doi.org/10.3390/coatings8020060
Gelde L, Cuevas AL, Martínez de Yuso MDV, Benavente J, Vega V, González AS, Prida VM, Hernando B. Influence of TiO2-Coating Layer on Nanoporous Alumina Membranes by ALD Technique. Coatings. 2018; 8(2):60. https://doi.org/10.3390/coatings8020060
Chicago/Turabian StyleGelde, Lourdes, Ana L. Cuevas, María Del Valle Martínez de Yuso, Juana Benavente, Víctor Vega, Ana Silvia González, Víctor M. Prida, and Blanca Hernando. 2018. "Influence of TiO2-Coating Layer on Nanoporous Alumina Membranes by ALD Technique" Coatings 8, no. 2: 60. https://doi.org/10.3390/coatings8020060
APA StyleGelde, L., Cuevas, A. L., Martínez de Yuso, M. D. V., Benavente, J., Vega, V., González, A. S., Prida, V. M., & Hernando, B. (2018). Influence of TiO2-Coating Layer on Nanoporous Alumina Membranes by ALD Technique. Coatings, 8(2), 60. https://doi.org/10.3390/coatings8020060







