Thermal Growth of Graphene: A Review
Abstract
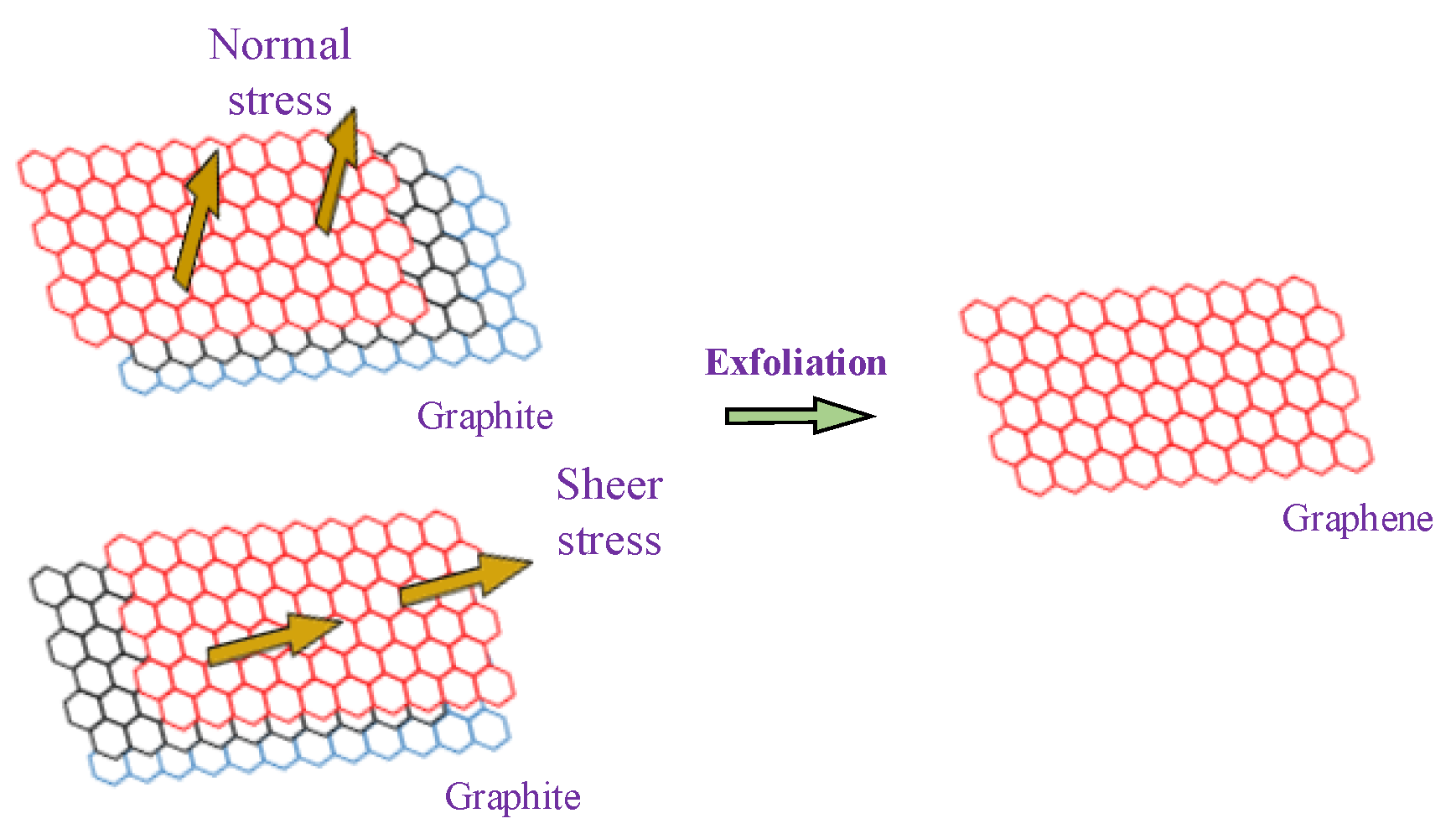
1. Introduction
2. Thermal Method for Growing of Graphene
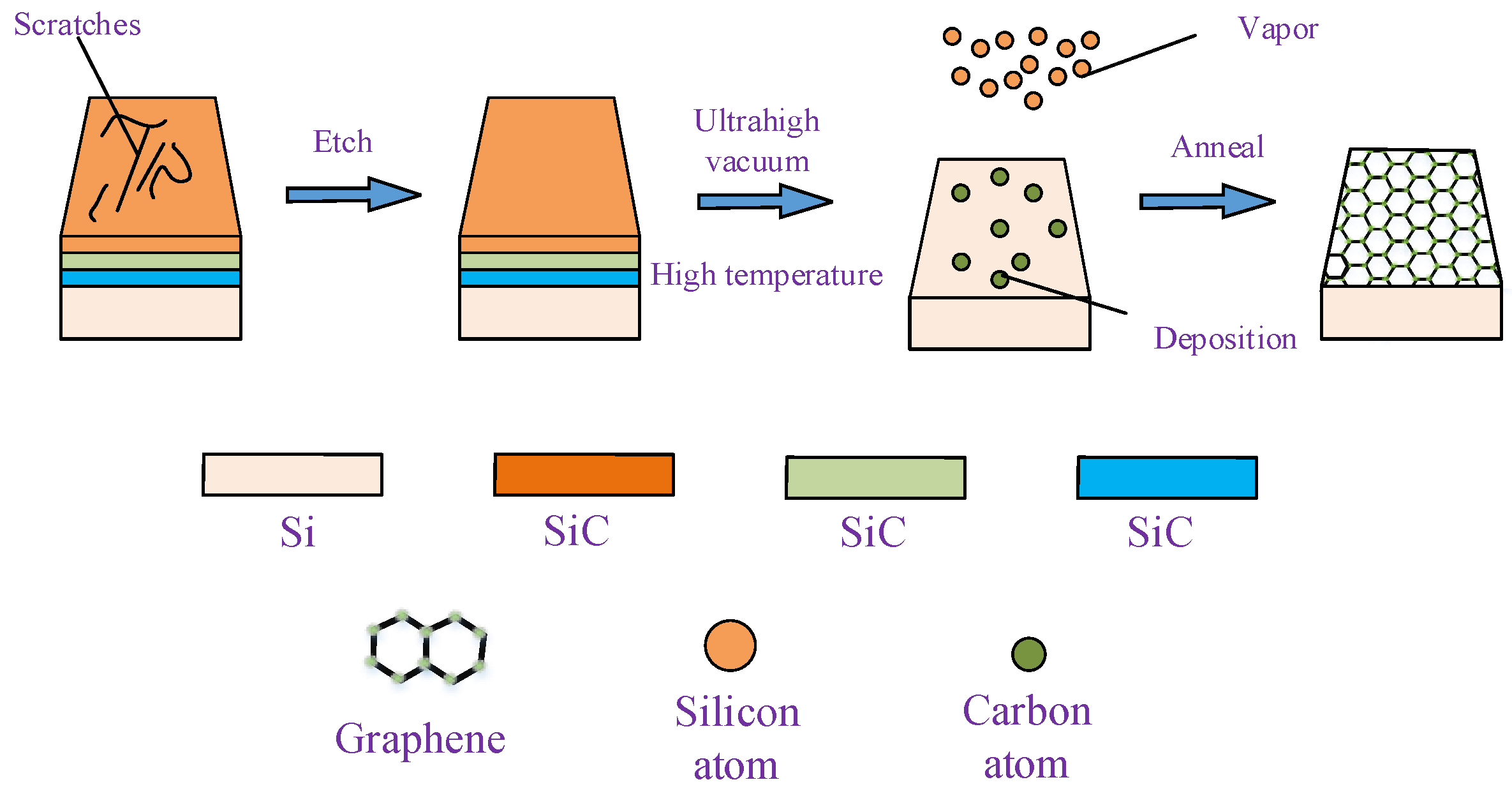
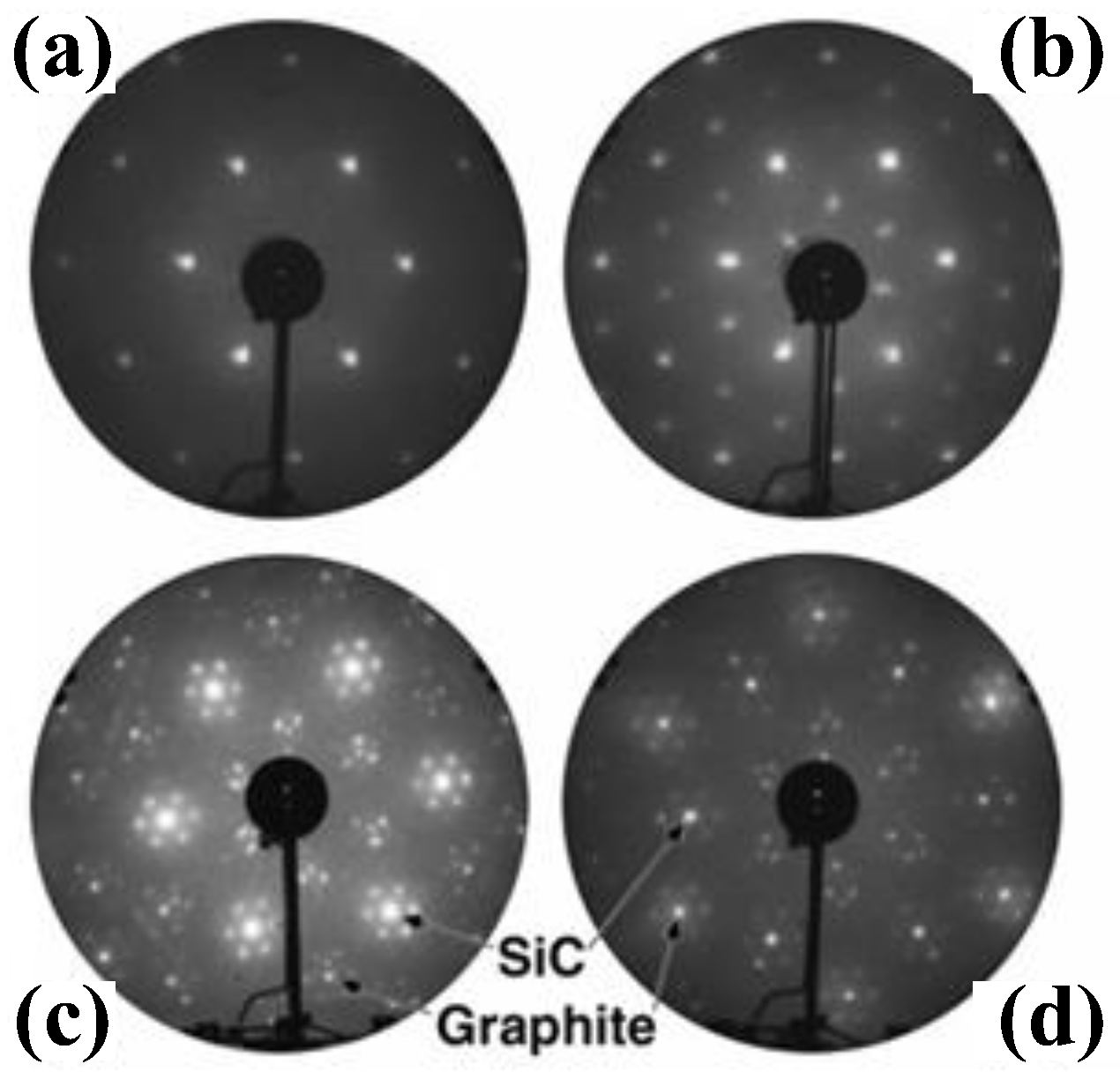
2.1. Epitaxial Growth of Graphene
2.2. Chemical Vapor Deposition Mechanism
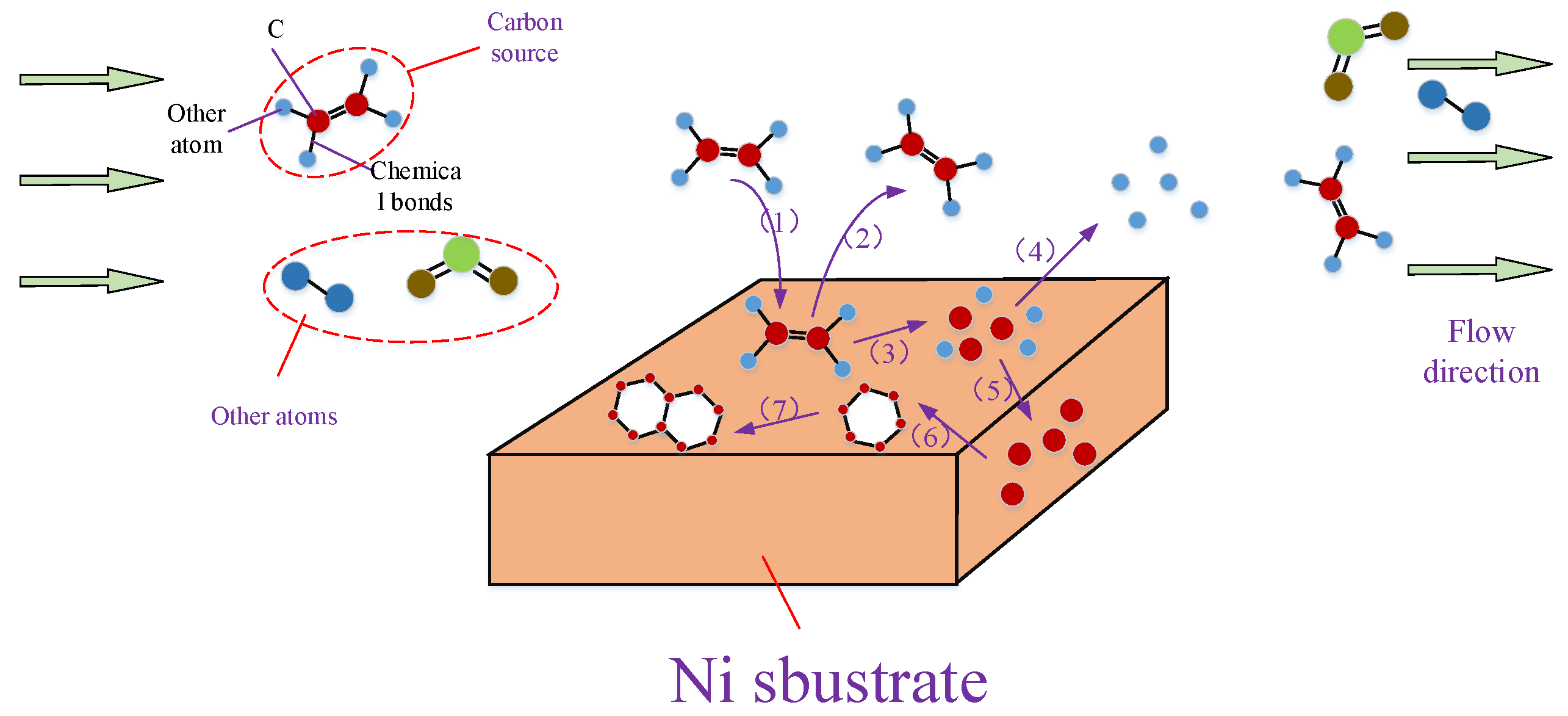
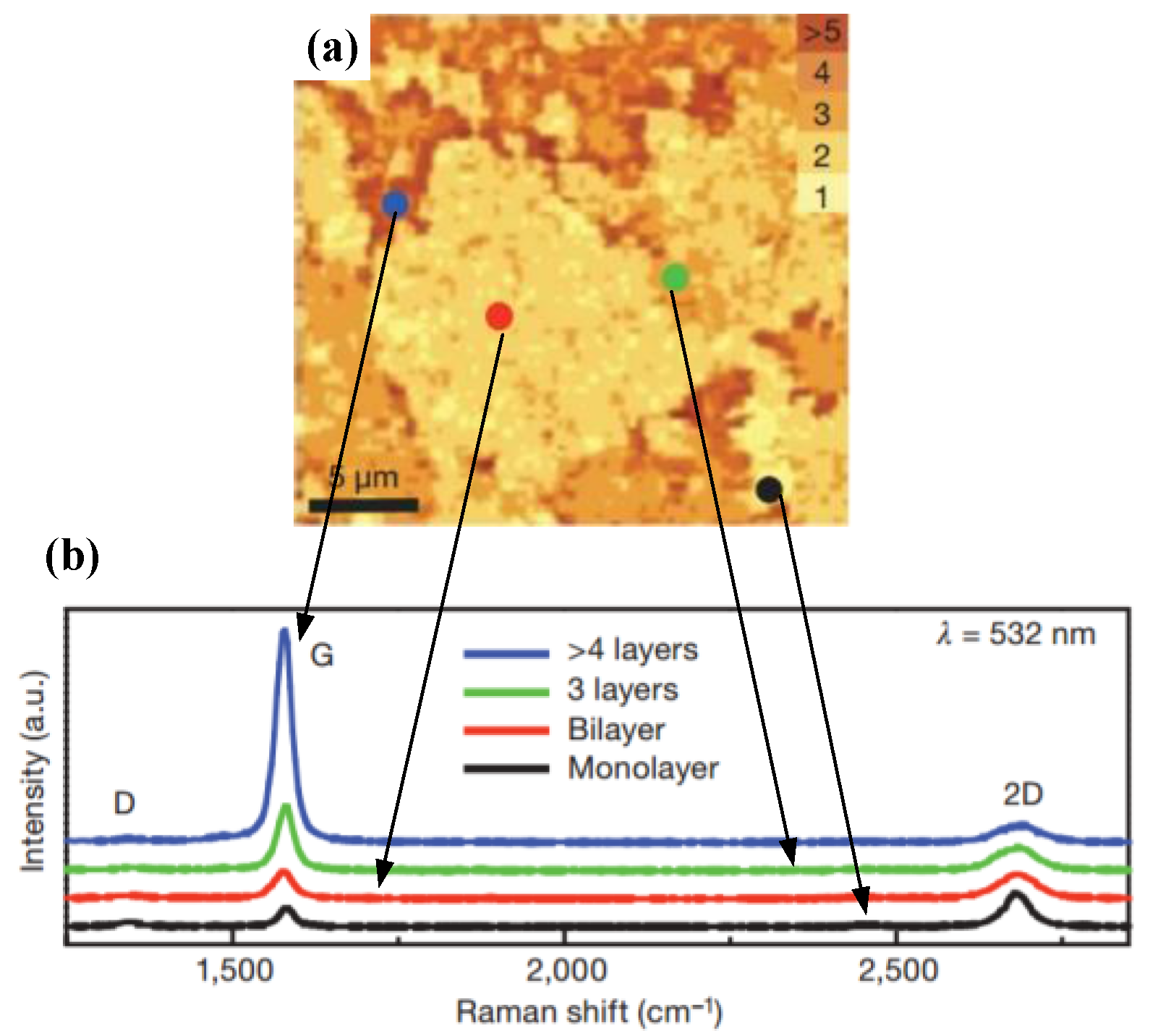
2.2.1. Conventional Chemical Vapor Deposition Growth on Ni
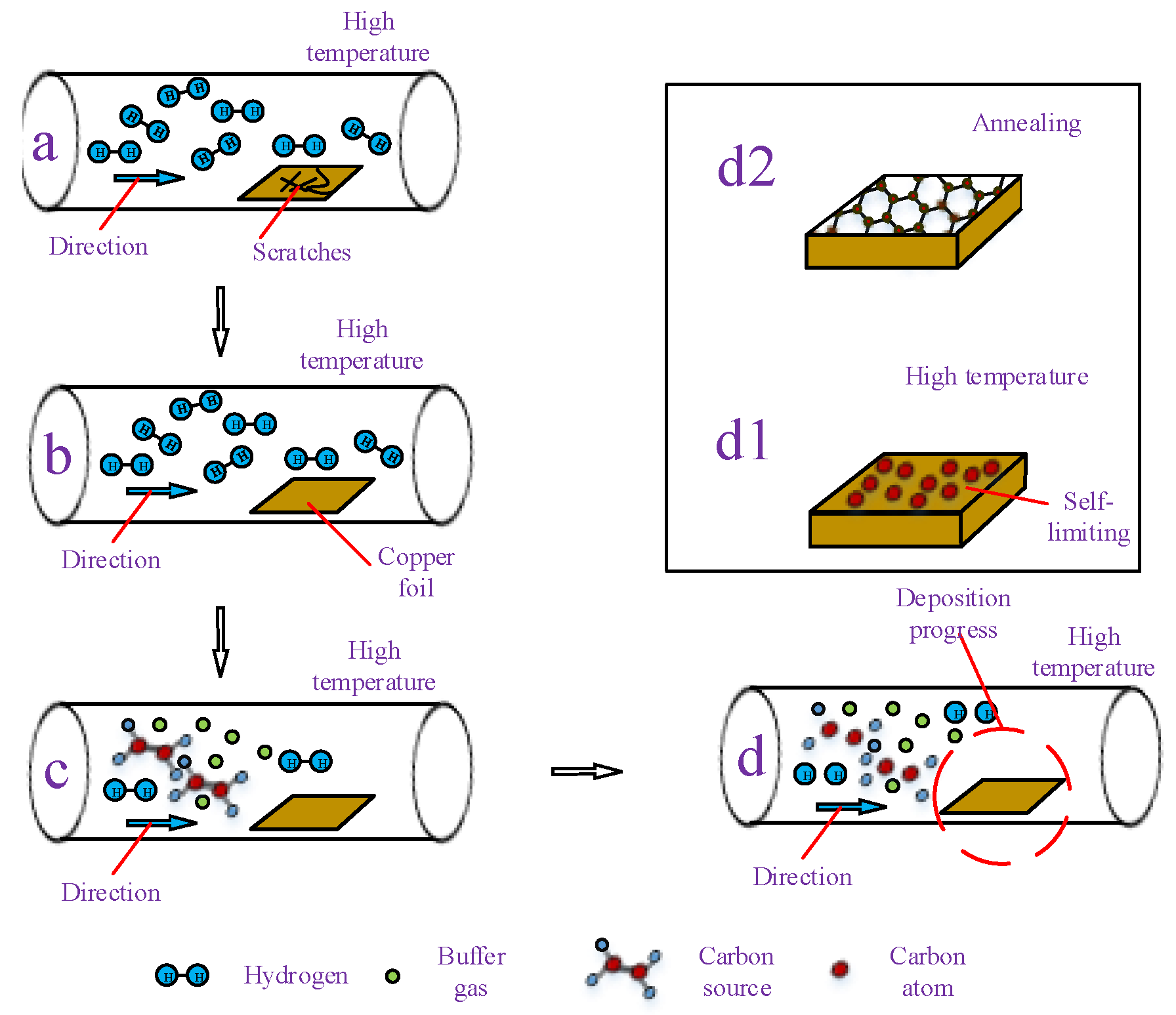
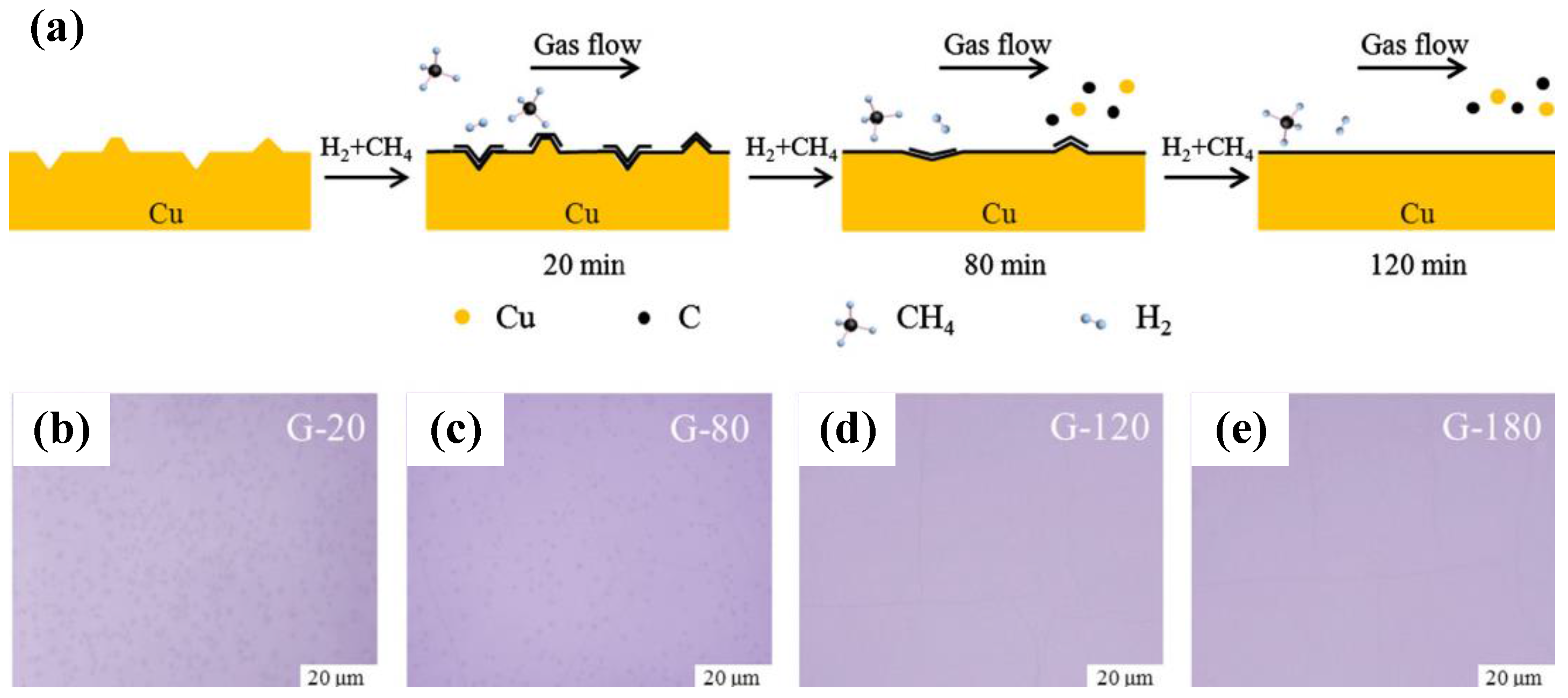
2.2.2. Conventional Chemical Vapor Deposition Growth on Cu
2.2.3. Plasma-Enhanced Chemical Vapor Deposition
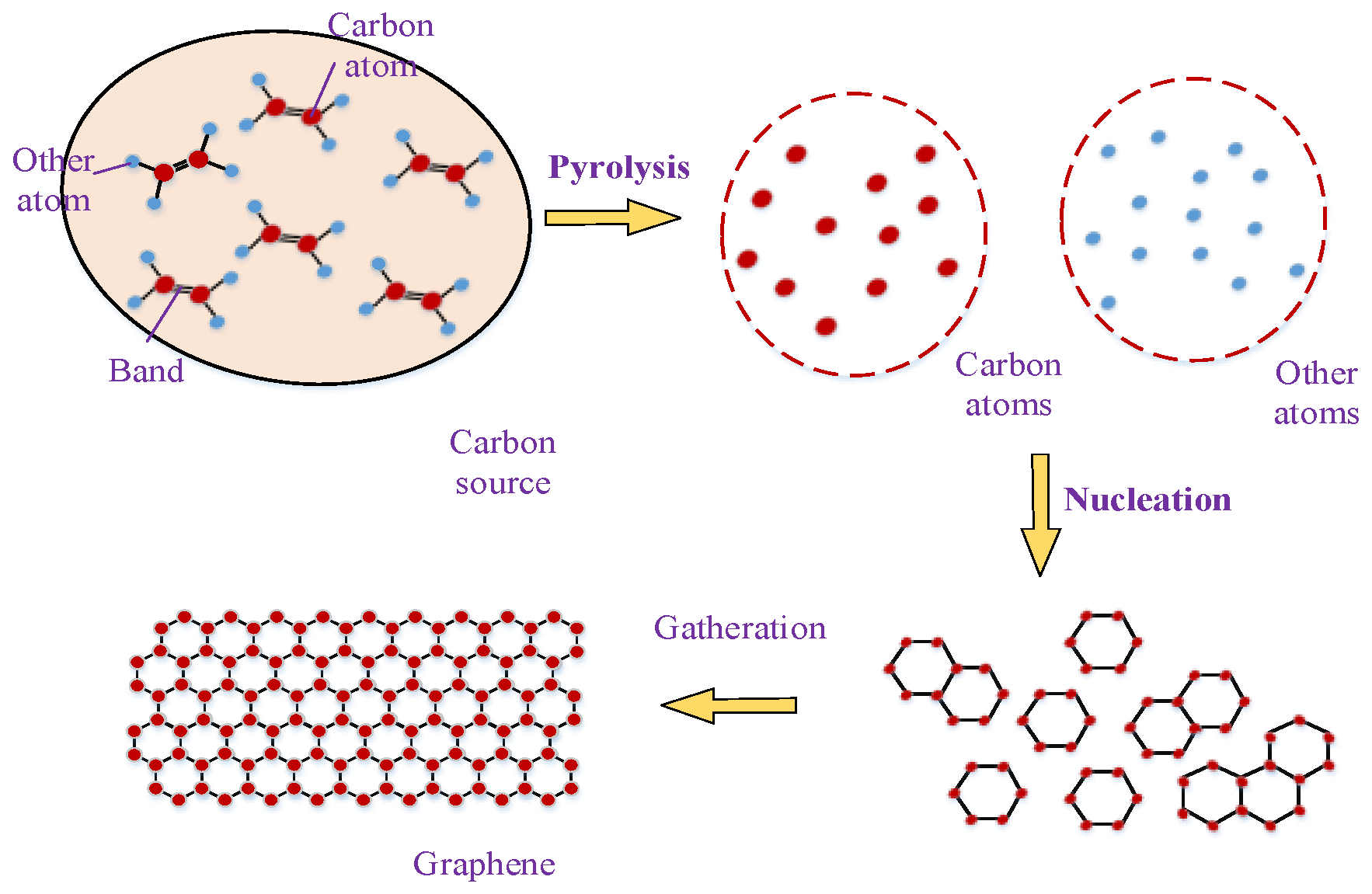
2.3. Combustion Method
3. Conclusions
Acknowledgments
Conflicts of Interest
References
- Peierls, R.E. Bemerkungen über umwandlungstemperaturen. Helv. Phys. Acta 1934, 7, 81–83. (In German) [Google Scholar]
- Peierls, R.E. Quelques proprietes typiques des corpses solides. Ann. I. H. Poincare 1935, 5, 177–222. (In German) [Google Scholar]
- Landau, L.D. Zur Theorie der phasenumwandlungen II. Phys. Z. Sowjetunion 1937, 11, 26–35. (In German) [Google Scholar]
- Novoselov, K.S.; Geim, A.K.; Morozov, S.V.; Jiang, D.; Zhang, Y.; Dubonos, S.V.; Grigorieva, I.V.; Firsov, A.A. Electric field effect in atomically thin carbon films. Science 2004, 306, 666–669. [Google Scholar] [CrossRef] [PubMed]
- Kroto, H.W.; Heath, J.R.; O’Brien, S.C.; Curl, R.F.; Smalley, R.E. C60: Buckminsterfullerene. Nature 1985, 318, 162–163. [Google Scholar] [CrossRef]
- Iijima, S. Helical microtubules of graphitic carbon. Nature 1991, 354, 56–58. [Google Scholar] [CrossRef]
- Zhang, Y.; Tan, Y.W.; Stormer, H.L.; Kim, P. Experimental observation of the quantum hall effect and berry’s phase in graphene. Nature 2005, 438, 201–204. [Google Scholar] [CrossRef] [PubMed]
- Sweetman, M.J.; May, S.; Mebberson, N.; Pendleton, P.; Vasilev, K.; Plush, S.E.; Hayball, J.D. Activated carbon, carbon nanotubes and graphene: Materials and composites for advanced water purification. C 2017, 3, 18. [Google Scholar] [CrossRef]
- Liu, B.; Xie, J.; Ma, H.; Zhang, X.; Pan, Y.; Lv, J.; Ge, H.; Ren, N.; Su, H.; Xie, X.; et al. From graphite to graphene oxide and graphene oxide quantum dots. Small 2017, 13. [Google Scholar] [CrossRef] [PubMed]
- Buzaglo, M.; Bar, I.P.; Varenik, M.; Shunak, L.; Pevzner, S.; Regev, O. Graphite-to-Graphene: Total conversion. Adv. Mater. 2017, 29. [Google Scholar] [CrossRef] [PubMed]
- Mayorov, A.S.; Gorbachev, R.V.; Morozov, S.V.; Britnell, L.; Jalil, R.; Ponomarenko, L.A.; Blake, P.; Novoselov, K.S.; Watanabe, K.; Taniguchi, T.; et al. Micrometer-scale ballistic transport in encapsulated graphene at room temperature. Nano Lett. 2011, 11, 2396–2399. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.; Wei, X.; Kysar, J.W.; Hone, J. Measurement of the elastic properties and intrinsic strength of monolayer graphene. Science 2008, 321, 385–388. [Google Scholar] [CrossRef] [PubMed]
- Balandin, A.A.; Ghosh, S.; Bao, W.; Calizo, I.; Teweldebrhan, D.; Miao, F.; Lau, C.N. Superior thermal conductivity of single-layer graphene. Nano Lett. 2008, 8, 902–907. [Google Scholar] [CrossRef] [PubMed]
- Nair, R.R.; Blake, P.; Grigorenko, A.N.; Novoselov, K.S.; Booth, T.J.; Stauber, T.; Peres, N.M.R.; Geim, A.K. Fine structure constant defines visual tranparency of graphene. Science 2008, 320, 1308. [Google Scholar] [CrossRef] [PubMed]
- Yi, M.; Shen, Z. A review on mechanical exfoliation for the scalable production of graphene. J. Mater. Chem. A 2015, 3, 11700–11715. [Google Scholar] [CrossRef]
- Ponomarenko, L.A.; Schedin, F.; Katsnelson, M.I.; Yang, R.; Hill, E.W.; Novoselov, K.S.; Geim, A.K. Chaotic Dirac billiard in graphene quantum dots. Science 2008, 320, 356–358. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.M.; Dimitrakopoulos, C.; Jenkins, K.A.; Farmer, D.B.; Chiu, H.Y.; Grill, A.; Avouris, P. 100-GHz transistors from wafer-scale epitaxial graphene. Science 2010, 327, 662. [Google Scholar] [CrossRef] [PubMed]
- Badami, D.V. Graphitization of α-silicon carbide. Nature 1962, 193, 569–570. [Google Scholar] [CrossRef]
- Hass, J.; Millán-Otoya, J.E.; First, P.N.; Conrad, E.H. Interface structure of epitaxial graphene grown on 4H-SiC (0001). Phys. Rev. B 2008, 78, 205424. [Google Scholar] [CrossRef]
- Jernigan, G.G.; VanMil, B.L.; Tedesco, J.L.; Tischler, J.G.; Glaser, E.R.; Davidson, A., III; Campbell, P.M.; Gaskill, D.K. Comparison of epitaxial graphene on Si-face and C-face 4H SiC formed by ultrahigh vacuum and RF furnace production. Nano Lett. 2009, 9, 2605–2609. [Google Scholar] [CrossRef] [PubMed]
- Berger, C.; Song, Z.; Li, T.; Li, X.; Ogbazghi, A.Y.; Feng, R.; Dai, Z.; Marchenkov, A.N.; Conrad, E.H.; First, P.N.; et al. Ultrathin epitaxial graphite: 2D electron gas properties and a route toward graphene-based nanoelectronics. J. Phys. Chem. B 2004, 108, 19912–19916. [Google Scholar] [CrossRef]
- Berger, C.; Song, Z.; Li, X.; Wu, X.; Brown, N.; Naud, C.; Mayou, D.; Li, T.; Hass, J.; Marchenkov, A.N.; et al. Electronic confinement and coherence in patterned epitaxial graphene. Science 2006, 312, 1191–1196. [Google Scholar] [CrossRef] [PubMed]
- Kang, C.Y.; Fan, L.L.; Chen, S.; Liu, Z.L.; Xu, P.S.; Zou, C.W. Few-layer graphene growth on 6H-SiC (0001) surface at low temperature via Ni-silicidation reactions. Appl. Phys. Lett. 2012, 100, 251604. [Google Scholar] [CrossRef]
- Tang, J.; Liu, Z.L.; Kang, C.Y.; Yan, W.S.; Xu, P.S.; Pan, H.B.; Wei, S.Q.; Gao, Y.Q.; Xu, X.G. Annealing time dependence of morphology and structure of epitaxial graphene on 6H-SiC(0001) surface. Acta Phys-Chim. Sin. 2010, 26, 253–258. (In Chinese) [Google Scholar]
- Emtsev, K.V.; Bostwick, A.; Horn, K.; Jobst, J.; Kellogg, G.L.; Ley, L.; McChesney, J.L.; Ohta, T.; Reshanov, S.A.; Röhrl, J.; et al. Towards wafer-size graphene layers by atmospheric pressure graphitization of silicon carbide. Nat. Mater. 2009, 8, 203–207. [Google Scholar] [CrossRef] [PubMed]
- Heer, W.A.D.; Berger, C.; Wu, X.; First, P.N.; Conrad, E.H.; Li, X.; Li, T.; Sprinkle, M.; Hass, J.; Sadowski, M.L.; et al. Epitaxial graphene. Solid State Commun. 2007, 143, 92–100. [Google Scholar] [CrossRef]
- Hass, J.; De Heer, W.A.; Conrad, E.H. The growth and morphology of epitaxial multilayer graphene. J. Phys. Condens. Matter 2008, 20, 323202. [Google Scholar] [CrossRef]
- Hicks, J.; Shepperd, K.; Wang, F.; Conrad, E.H. The structure of graphene grown on the SiC surface. J. Phys. D Appl. Phys. 2012, 45, 154002. [Google Scholar] [CrossRef]
- Terai, M.; Hasegawa, N.; Okusawa, M.; Otani, S.; Oshima, C. Electronic states of monolayer micrographite on TiC (111)-faceted and TiC (410) surfaces. Appl. Surf. Sci. 1998, 130, 876–882. [Google Scholar] [CrossRef]
- Itchkawitz, B.S.; Lyman, P.F.; Ownby, G.W.; Zehner, D.M. Monolayer graphite on TaC (111): Electronic band structure. Surf. Sci. 1994, 318, 395–402. [Google Scholar] [CrossRef]
- Yang, W.; Chen, G.; Shi, Z.; Liu, C.C.; Zhang, L.; Xie, G.; Cheng, M.; Wang, D.; Yang, R.; Shi, D.; et al. Epitaxial growth of single-domain graphene on hexagonal boron nitride. Nat. Mater. 2013, 12, 792–797. [Google Scholar] [CrossRef] [PubMed]
- Reina, A.; Jia, X.; Ho, J.; Nezich, D.; Son, H.; Bulovic, V.; Dresselhaus, M.S.; Kong, J. Large area, few-layer graphene films on arbitrary substrates by chemical vapor deposition. Nano Lett. 2009, 9, 30–35. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.S.; Zhao, Y.; Jang, H.; Lee, S.Y.; Kim, J.M.; Kim, K.S.; Ahn, J.H.; Kim, P.; Choi, J.Y.; Hong, B.H. Large-scale pattern growth of graphene films for stretchable transparent electrodes. Nature 2009, 457, 706–710. [Google Scholar] [CrossRef] [PubMed]
- Seo, D.H.; Pineda, S.; Fang, J.; Gozukara, Y.; Yick, S.; Bendavid, A.; Lam, S.K.H.; Murdock, A.T.; Murphy, A.B.; Han, Z.J.; et al. Single-step ambient-air synthesis of graphene from renewable precursors as electrochemical genosensor. Nat. Commun. 2017, 8, 14217. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Cai, W.; An, J.; Kim, S.; Nah, J.; Yang, D.; Piner, R.; Velamakanni, A.; Jung, I.; Tutuc, E.; et al. Large-area synthesis of high-quality and uniform graphene films on copper foils. Science 2009, 324, 1312–1314. [Google Scholar] [CrossRef] [PubMed]
- Bae, S.; Kim, H.; Lee, Y.; Xu, X.; Park, J.S.; Zheng, Y.; Balakrishnan, J.; Lei, T.; Kim, H.R.; Song, Y., II; et al. Roll-to-roll production of 30-inch graphene films for transparent electrodes. Nat. Nanotechnol. 2010, 5, 574–578. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Fu, C.; Sun, H.; Meng, L.; Xia, Y.; Zhang, C.; Yi, X.; Yang, W.; Guo, P.; Wang, C.; et al. Large-area, high-quality monolayer graphene from polystyrene at atmospheric pressure. Nanotechnology 2017, 28, 155605. [Google Scholar] [CrossRef] [PubMed]
- Malesevic, A.; Vitchev, R.; Schouteden, K.; Volodin, A.; Zhang, L.; Van Tendeloo, G.; Vanhulsel, A.; Van Haesendonck, C. Synthesis of few-layer graphene via microwave plasma-enhanced chemical vapor deposition. Nanotechnology 2008, 19, 305604. [Google Scholar] [CrossRef] [PubMed]
- Boyd, D.A.; Lin, W.H.; Hsu, C.C.; Teague, M.L.; Chen, C.C.; Lo, Y.Y.; Chan, W.Y.; Su, W.B.; Cheng, T.C.; Chang, C.S.; et al. Single-step deposition of high-mobility graphene at reduced temperatures. Nat. Commun. 2015, 6, 6620. [Google Scholar] [CrossRef] [PubMed]
- Dato, A.; Radmilovic, V.; Lee, Z.; Phillips, J.; Frenklach, M. Substrate-free gas-phase synthesis of graphene sheets. Nano Lett. 2008, 8, 2012–2016. [Google Scholar] [CrossRef] [PubMed]
- Subrahmanyam, K.S.; Panchakarla, L.S.; Govindaraj, A.; Rao, C.N.R. Simple method of preparing graphene flakes by an arc-discharge method. J. Phys. Chem. C 2009, 113, 4257–4259. [Google Scholar] [CrossRef]
- Son, M.; Lim, H.; Hong, M.; Choi, H.C. Direct growth of graphene pad on exfoliated hexagonal boron nitride surface. Nanoscale 2011, 3, 3089–3093. [Google Scholar] [CrossRef] [PubMed]
- Tang, S.; Ding, G.; Xie, X.; Chen, J.; Wang, C.; Ding, X.; Huang, F.; Lu, W.; Jiang, M. Nucleation and growth of single crystal graphene on hexagonal boron nitride. Carbon 2012, 50, 329–331. [Google Scholar] [CrossRef]
- Liu, Z.; Song, L.; Zhao, S.; Huang, J.; Ma, L.; Zhang, J.; Lou, J.; Ajayan, P.M. Direct growth of graphene/hexagonal boron nitride stacked layers. Nano Lett. 2011, 11, 2032–2037. [Google Scholar] [CrossRef] [PubMed]
- Kwieciñski, W.; Sotthewes, K.; Poelsema, B.; Zandvliet, H.J.; Bampoulis, P. Chemical vapor deposition growth of bilayer graphene in between molybdenum disulfide sheets. J. Colloid Interface Sci. 2017, 505, 776–782. [Google Scholar] [CrossRef] [PubMed]
- Fu, W.; Du, F.H.; Su, J.; Li, X.H.; Wei, X.; Ye, T.N.; Wang, K.X.; Chen, J.S. In situ catalytic growth of large-area multilayered graphene/MoS2 heterostructures. Sci. Rep. 2014, 4, 4673. [Google Scholar] [CrossRef] [PubMed]
- Oshima, C.; Nagashima, A. Ultra-thin epitaxial films of graphite and hexagonal boron nitride on solid surfaces. J. Phys. Condens. Matter 1997, 9, 1. [Google Scholar] [CrossRef]
- Kong, J.; Cassell, A.M.; Dai, H. Chemical vapor deposition of methane for single-walled carbon nanotubes. Chem. Phys. Lett. 1998, 292, 567–574. [Google Scholar] [CrossRef]
- Reina, A.; Thiele, S.; Jia, X.; Bhaviripudi, S.; Dresselhaus, M.S.; Schaefer, J.A.; Kong, J. Growth of large-area single-and bi-layer graphene by controlled carbon precipitation on polycrystalline Ni surfaces. Nano Res. 2009, 2, 509–516. [Google Scholar] [CrossRef]
- Zhang, Y.; Gomez, L.; Ishikawa, F.N.; Madaria, A.; Ryu, K.; Wang, C.; Badmaev, A.; Zhou, C. Comparison of graphene growth on single-crystalline and polycrystalline Ni by chemical vapor deposition. J. Phys. Chem. Lett. 2010, 1, 3101–3107. [Google Scholar] [CrossRef]
- Chae, S.J.; Güneş, F.; Kim, K.K.; Kim, E.S.; Han, G.H.; Kim, S.M.; Shin, H.J.; Yoon, S.M.; Choi, J.Y.; Park, M.H.; et al. Synthesis of large-area graphene layers on poly-nickel substrate by chemical vapor deposition: Wrinkle formation. Adv. Mater. 2009, 21, 2328–2333. [Google Scholar] [CrossRef]
- Liu, J.; Li, P.; Chen, Y.; Wang, Z.; He, J.; Tian, H.; Qi, F.; Zheng, B.; Zhou, J.; Lin, W.; et al. Large-area synthesis of high-quality and uniform monolayer graphene without unexpected bilayer regions. J. Alloys Compd. 2014, 615, 415–418. [Google Scholar] [CrossRef]
- Vlassiouk, I.; Smirnov, S.; Regmi, M.; Surwade, S.P.; Srivastava, N.; Feenstra, R.; Eres, G.; Parish, C.; Lavrik, N.; Datskos, P.; et al. Graphene nucleation density on copper: Fundamental role of background pressure. J. Phys. Chem. C 2013, 117, 18919–18926. [Google Scholar] [CrossRef]
- Liu, L.; Zhou, H.; Cheng, R.; Chen, Y.; Lin, Y.C.; Qu, Y.; Bai, J.; Ivanov, I.A.; Liu, G.; Huang, Y.; et al. A systematic study of atmospheric pressure chemical vapor deposition growth of large-area monolayer graphene. J. Mater. Chem. 2012, 22, 1498–1503. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Cai, W.; Colombo, L.; Ruoff, R.S. Evolution of graphene growth on Ni and Cu by carbon isotope labeling. Nano Lett. 2009, 9, 4268–4272. [Google Scholar] [CrossRef] [PubMed]
- Whiteway, E.; Yang, W.; Yu, V.; Hilke, M. Time evolution of the growth of single graphene crystals and high resolution isotope labeling. Carbon 2017, 111, 173–181. [Google Scholar] [CrossRef]
- Chen, C.; Avila, J.; Asensio, M.C. Chemical and electronic structure imaging of graphene on Cu: A NanoARPES study. J. Phys. Condens. Matter 2017, 29, 183001. [Google Scholar] [CrossRef] [PubMed]
- Yang, F.; Liu, Y.; Wu, W.; Chen, W.; Gao, L.; Sun, J. A facile method to observe graphene growth on copper foil. Nanotechnology 2012, 23, 475705. [Google Scholar] [CrossRef] [PubMed]
- Vinogradov, N.A.; Schulte, K.; Ng, M.L.; Mikkelsen, A.; Lundgren, E.; Martensson, N.; Preobrajenski, A.B. Impact of atomic oxygen on the structure of graphene formed on Ir (111) and Pt (111). J. Phys. Chem. C 2011, 115, 9568–9577. [Google Scholar] [CrossRef]
- Coraux, J.; Engler, M.; Busse, C.; Wall, D.; Buckanie, N.; Zu Heringdorf, F.J.M.; Van Gastel, R.; Poelsema, B.; Michely, T. Growth of graphene on Ir (111). New J. Phys. 2009, 11, 023006. [Google Scholar] [CrossRef]
- Lukosius, M.; Dabrowski, J.; Kitzmann, J.; Fursenko, O.; Akhtar, F.; Lisker, M.; Lippert, G.; Schulze, S.; Yamamoto, Y.; Schubert, M.A.; et al. Metal-free CVD graphene synthesis on 200 mm Ge/Si (001) substrates. ACS Appl. Mater. Interfaces 2016, 8, 33786–33793. [Google Scholar] [CrossRef] [PubMed]
- Scaparro, A.M.; Miseikis, V.; Coletti, C.; Notargiacomo, A.; Pea, M.; De Seta, M.; Di Gaspare, L. Investigating the CVD Synthesis of Graphene on Ge (100): Toward layer-by-layer growth. ACS Appl. Mater. Interfaces 2016, 8, 33083–33090. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; You, H.; Liu, F.; Li, M.; Wan, L.; Li, S.; Li, Q.; Xu, Y.; Tian, R.; Yu, Z.; et al. Large-scale synthesis of few-layered graphene using CVD. Chem. Vapor Depos. 2009, 15, 53–56. [Google Scholar] [CrossRef]
- Ryu, J.; Kim, Y.; Won, D.; Kim, N.; Park, J.S.; Lee, E.K.; Cho, D.; Cho, S.P.; Kim, S.J.; Ryu, G.H.; et al. Fast synthesis of high-performance graphene films by hydrogen-free rapid thermal chemical vapor deposition. ACS Nano 2014, 8, 950–956. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.Z.; Medina, H.; Tsai, H.W.; Wang, Y.C.; Yen, Y.T.; Manikandan, A.; Chueh, Y.L. Low temperature growth of graphene on glass by carbon-enclosed chemical vapor deposition process and its application as transparent electrode. Chem. Mater. 2015, 27, 1646–1655. [Google Scholar] [CrossRef]
- Chan, S.H.; Chen, S.H.; Lin, W.T.; Li, M.C.; Lin, Y.C.; Kuo, C.C. Low-temperature synthesis of graphene on Cu using plasma-assisted thermal chemical vapor deposition. Nanoscale Res. Lett. 2013, 8, 285. [Google Scholar] [CrossRef] [PubMed]
- Mueller, N.S.; Morfa, A.J.; Abou-Ras, D.; Oddone, V.; Ciuk, T.; Giersig, M. Growing graphene on polycrystalline copper foils by ultra-high vacuum chemical vapor deposition. Carbon 2014, 78, 347–355. [Google Scholar] [CrossRef]
- Jang, J.; Son, M.; Chung, S.; Kim, K.; Cho, C.; Lee, B.H.; Ham, M.H. Low-temperature-grown continuous graphene films from benzene by chemical vapor deposition at ambient pressure. Sci. Rep. 2015, 5, 17955. [Google Scholar] [CrossRef] [PubMed]
- John, P.I. Plasma Sciences and the Creation of Wealth; Tata McGraw-Hill Education: New York, NY, USA, 2005. [Google Scholar]
- Qi, J.; Zhang, L.; Cao, J.; Zheng, W.; Wang, X.; Feng, J. Synthesis of graphene on a Ni film by radio-frequency plasma-enhanced chemical vapor deposition. Chin. Sci. Bull. 2012, 57, 3040–3044. [Google Scholar] [CrossRef]
- Karmakar, S.; Kulkarni, N.V.; Nawale, A.B.; Lalla, N.P.; Mishra, R.; Sathe, V.G.; Bhoraskar, S.V.; Das, A.K. A novel approach towards selective bulk synthesis of few-layer graphenes in an electric arc. J. Phys. D Appl. Phys. 2009, 42, 115201. [Google Scholar] [CrossRef]
- Tatarova, E.; Dias, A.; Henriques, J.; do Rego, A.B.; Ferraria, A.M.; Abrashev, M.V.; Luhrs, C.C.; Phillips, J.; Dias, F.M.; Ferreira, C.M. Microwave plasmas applied for the synthesis of free standing graphene sheets. J. Phys. D Appl. Phys. 2014, 47, 38550. [Google Scholar] [CrossRef]
- Park, H.J.; Ahn, B.W.; Kim, T.Y.; Lee, J.W.; Jung, Y.H.; Choi, Y.S.; Song, Y., II; Suh, S.J. Direct synthesis of multi-layer graphene film on various substrates by microwave plasma at low temperature. Thin Solid Films 2015, 587, 8–13. [Google Scholar] [CrossRef]
- Yamada, T.; Ishihara, M.; Kim, J.; Hasegawa, M.; Iijima, S. A roll-to-roll microwave plasma chemical vapor deposition process for the production of 294 mm width graphene films at low temperature. Carbon 2012, 50, 2615–2619. [Google Scholar] [CrossRef]
- Phillips, J. Plasma Generation of Supported Metal Catalysts. U.S. Patent 5,989,648, 23 November 1999. [Google Scholar]
- Dato, A.M. Substrate-Free Gas-Phase Synthesis of Graphene; University of California: Berkeley, CA, USA, 2009. [Google Scholar]
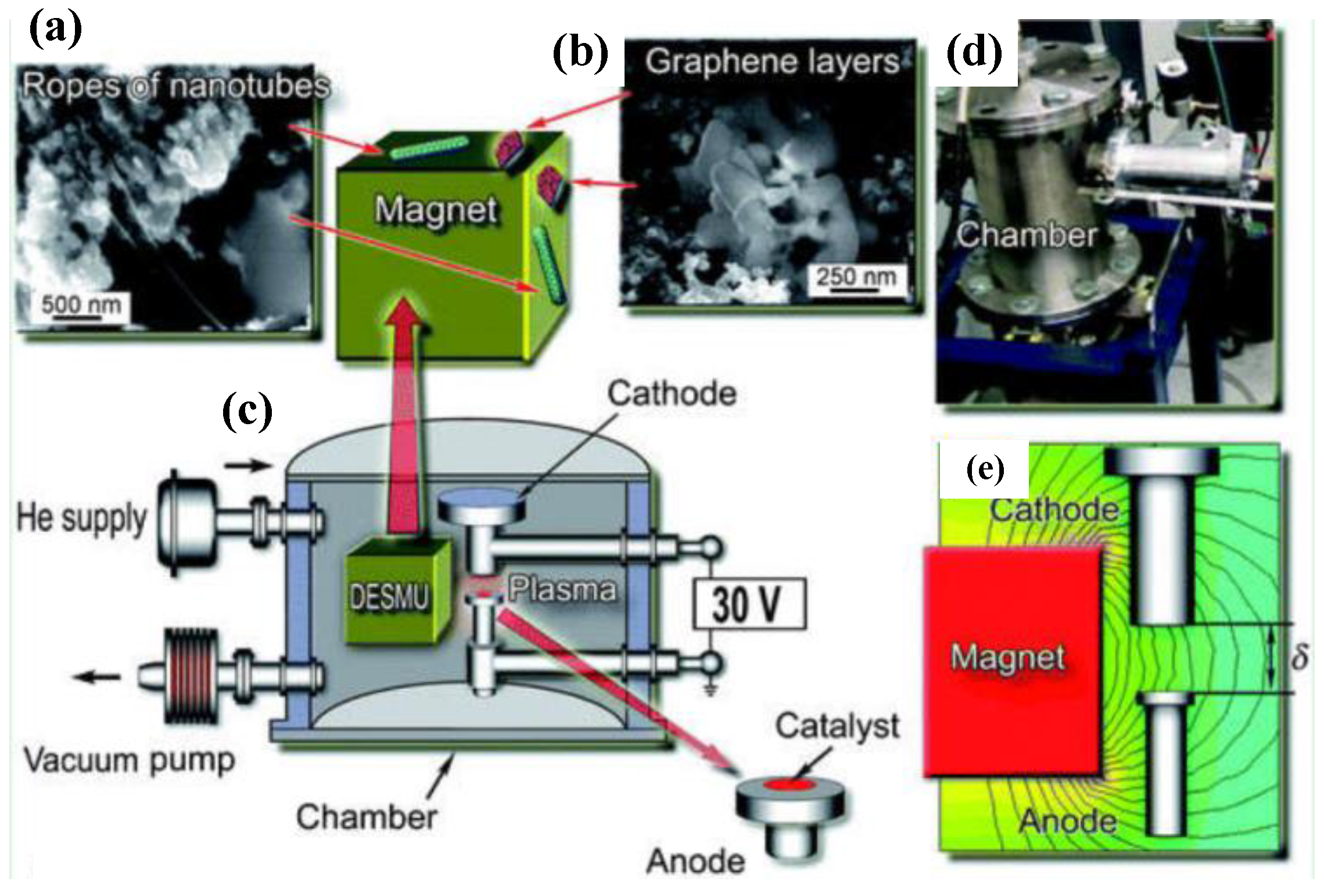
- Volotskova, O.; Levchenko, I.; Shashurin, A.; Raitses, Y.; Ostrikov, K.; Keidar, M. Single-step synthesis and magnetic separation of graphene and carbon nanotubes in arc discharge plasmas. Nanoscale 2010, 2, 2281–2285. [Google Scholar] [CrossRef] [PubMed]
- Levchenko, I.; Volotskova, O.; Shashurin, A.; Raitses, Y.; Ostrikov, K.; Keidar, M. The large-scale production of graphene flakes using magnetically-enhanced arc discharge between carbon electrodes. Carbon 2010, 48, 4570–4574. [Google Scholar] [CrossRef]
- Wang, Z.; Li, N.; Shi, Z.; Gu, Z. Low-cost and large-scale synthesis of graphene nanosheets by arc discharge in air. Nanotechnology 2010, 21, 175602. [Google Scholar] [CrossRef] [PubMed]
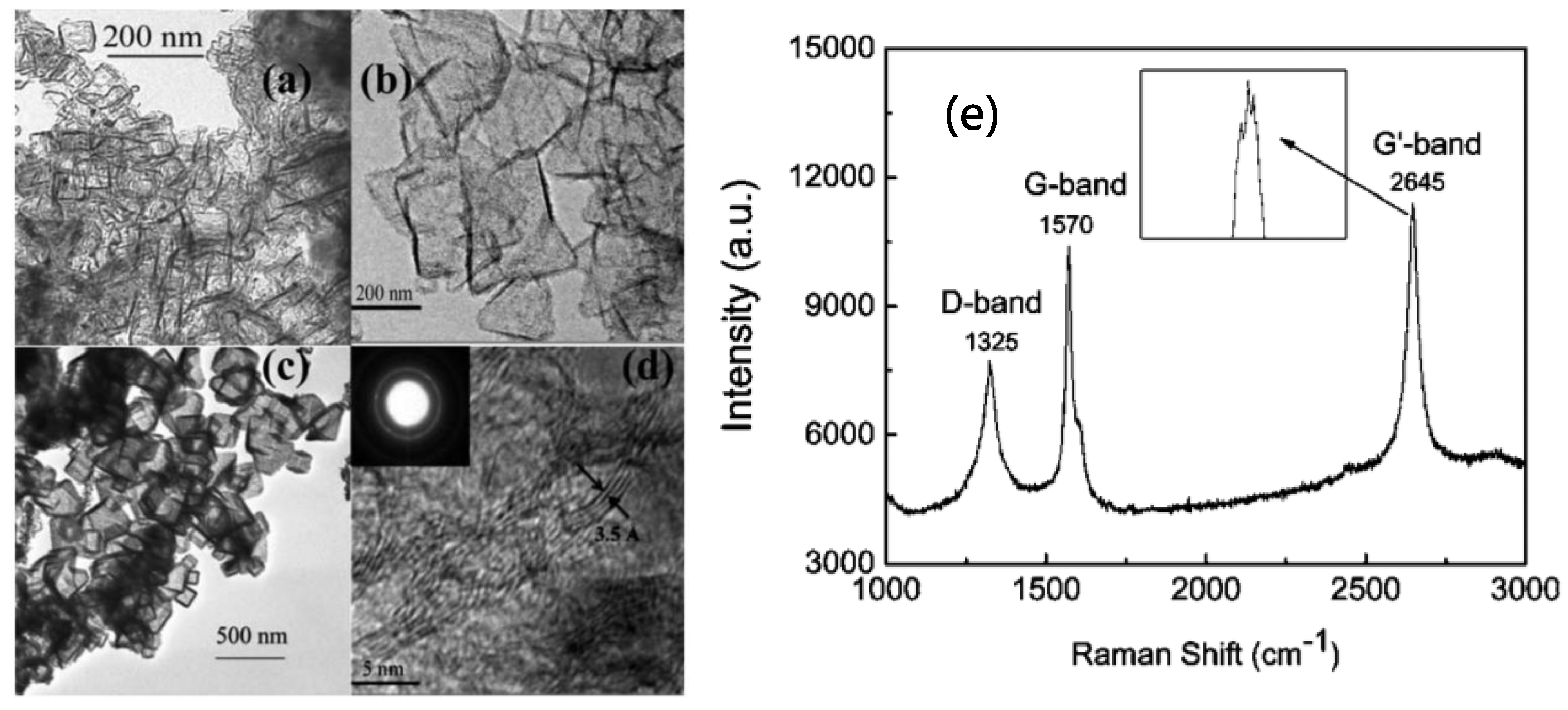
- Shen, B.; Ding, J.; Yan, X.; Feng, W.; Li, J.; Xue, Q. Influence of different buffer gases on synthesis of few-layered graphene by arc discharge method. Appl. Surf. Sci. 2012, 258, 4523–4531. [Google Scholar] [CrossRef]
- Sorensen, C.; Nepal, A.; Singh, G.P. Process for High-Yield Production of Graphene via Detonation of Carbon-Containing Material. U.S. Patent 9,440,857, 13 September 2016. [Google Scholar]
- Nepal, A.; Singh, G.P.; Flanders, B.N.; Sorensen, C.M. One-step synthesis of graphene via catalyst-free gas-phase hydrocarbon detonation. Nanotechnology 2013, 24, 245602. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Zhu, H.; Xie, D.; Wang, K.; Cao, A.; Wei, J.; Li, X.; Fan, L.; Wu, D. Flame synthesis of few-layered graphene/graphite films. Chem. Commun. 2011, 47, 3520–3522. [Google Scholar] [CrossRef] [PubMed]
- Memon, N.K.; Stephen, D.T.; Al-Sharab, J.F.; Yamaguchi, H.; Goncalves, A.M.B.; Kear, B.H.; Jaluria, Y.; Andrei, E.Y.; Chhowalla, M. Flame synthesis of graphene films in open environments. Carbon 2011, 49, 5064–5070. [Google Scholar] [CrossRef]
- Chakrabarti, A.; Lu, J.; Skrabutenas, J.C.; Xu, T.; Xiao, Z.; Maguire, J.A.; Hosmane, N.S. Conversion of carbon dioxide to few-layer graphene. J. Mater. Chem. 2011, 21, 9491–9493. [Google Scholar] [CrossRef]
- Zhang, J.; Tian, T.; Chen, Y.; Niu, Y.; Tang, J.; Qin, L.C. Synthesis of graphene from dry ice in flames and its application in supercapacitors. Chem. Phys. Lett. 2014, 591, 78–81. [Google Scholar] [CrossRef]



| Method | Advantage | Disadvantage | |
|---|---|---|---|
| Epitaxial growth | High quality; highly compatible with electronics | High costs of SiC wafers; Low yield; Hard to transfer | |
| Chemical vapor deposition | Conventional chemical vapor deposition | Large graphene films; Possible to transfer onto multitudes of materials; High quality and large-scale production | Required substrates are often expensive; Complicated synthetic and transfer process; Introducing new defects in the transfer process |
| Plasma-enhanced chemical vapor deposition | Relative low temperature; Short reaction time | ||
| Combustion method | Simple facility; Quick synthetic process; | Hard to control the combustible process; Non-uniform distribution; Low quality | |
| Substrate | Precursor Gas | Pressure (Torr) | Temperature (°C) | Charge Carrier Mobility (cm2·V−1·s−1) | Square Resistance (kΩ/sq) | Ref. |
|---|---|---|---|---|---|---|
| 6H–SiC | – | 1 × 10−10 | 1450 | 1100 (4 K) | 1.5 (4 K) | [21] |
| 4H–SiC | – | ultra-high vacuum | – | 2.5 × 104 (2490 K) | 1.41 (30 K) | [22] |
| Ni/6H–SiC | – | 4.5 × 10−10 | 950 | – | – | [23] |
| 6H–SiC | – | 4.5 × 10−10 | 1300 | – | – | [24] |
| 6H–SiC | Argon | 750 | 1550 | 2000 (27 K) | – | [25] |
| Method | Substrate | Precursor Gas | Temperature (°C) | Number of Layer | Size (cm2) | Ref. | |
|---|---|---|---|---|---|---|---|
| Conventional chemical vapor deposition | Ni | CH4; H2 | 900; 1000 | 1–12 | 2 | [32] | |
| CH4; H2; Ar | 1000 | 1–10 | 4 | [33] | |||
| Soybean | 800 | – | 4 | [34] | |||
| Cu | CH4; H2 | 1000 | 1–3 | 1 | [35] | ||
| CH4; H2 | 1000 | 1 | 30 (inch) | [36] | |||
| Polystyrene; H2; Ar | 1000 | 1 | 1 | [37] | |||
| Plasma-enhanced chemical vapor deposition | Micro-wave-assisted | Various | CH4; H2 | 700 | 4–6 | – | [38] |
| Cu | CH4; H2 | <420 | 1 | 1.04 | [39] | ||
| Non | C2H5OH; Ar | – | – | – | [40] | ||
| Arc-discharge | – | H2; graphite; Ar | – | 2–4 | – | [41] | |
| Method | Temperature | Energy Cost | Throughput | Electrical Property | |
|---|---|---|---|---|---|
| Epitaxial growth | Medium | High | Low | High | |
| CVD | CCVD | Medium | High | Low | High |
| PECVD | Low | Medium | Low | High | |
| Arc discharge | High | Low | High | Low | |
| Combustion method | High | Low | – | Low | |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tan, H.; Wang, D.; Guo, Y. Thermal Growth of Graphene: A Review. Coatings 2018, 8, 40. https://doi.org/10.3390/coatings8010040
Tan H, Wang D, Guo Y. Thermal Growth of Graphene: A Review. Coatings. 2018; 8(1):40. https://doi.org/10.3390/coatings8010040
Chicago/Turabian StyleTan, Hai, Deguo Wang, and Yanbao Guo. 2018. "Thermal Growth of Graphene: A Review" Coatings 8, no. 1: 40. https://doi.org/10.3390/coatings8010040
APA StyleTan, H., Wang, D., & Guo, Y. (2018). Thermal Growth of Graphene: A Review. Coatings, 8(1), 40. https://doi.org/10.3390/coatings8010040




