Surface Free Energy Utilization to Evaluate Wettability of Hydrocolloid Suspension on Different Vegetable Epicarps
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Preparation of Coating Formulations
2.3. Wettability of Coating Formulations on Banana and Eggplant Epicarps
2.4. Statistical Design and Analysis
3. Results and Discussion
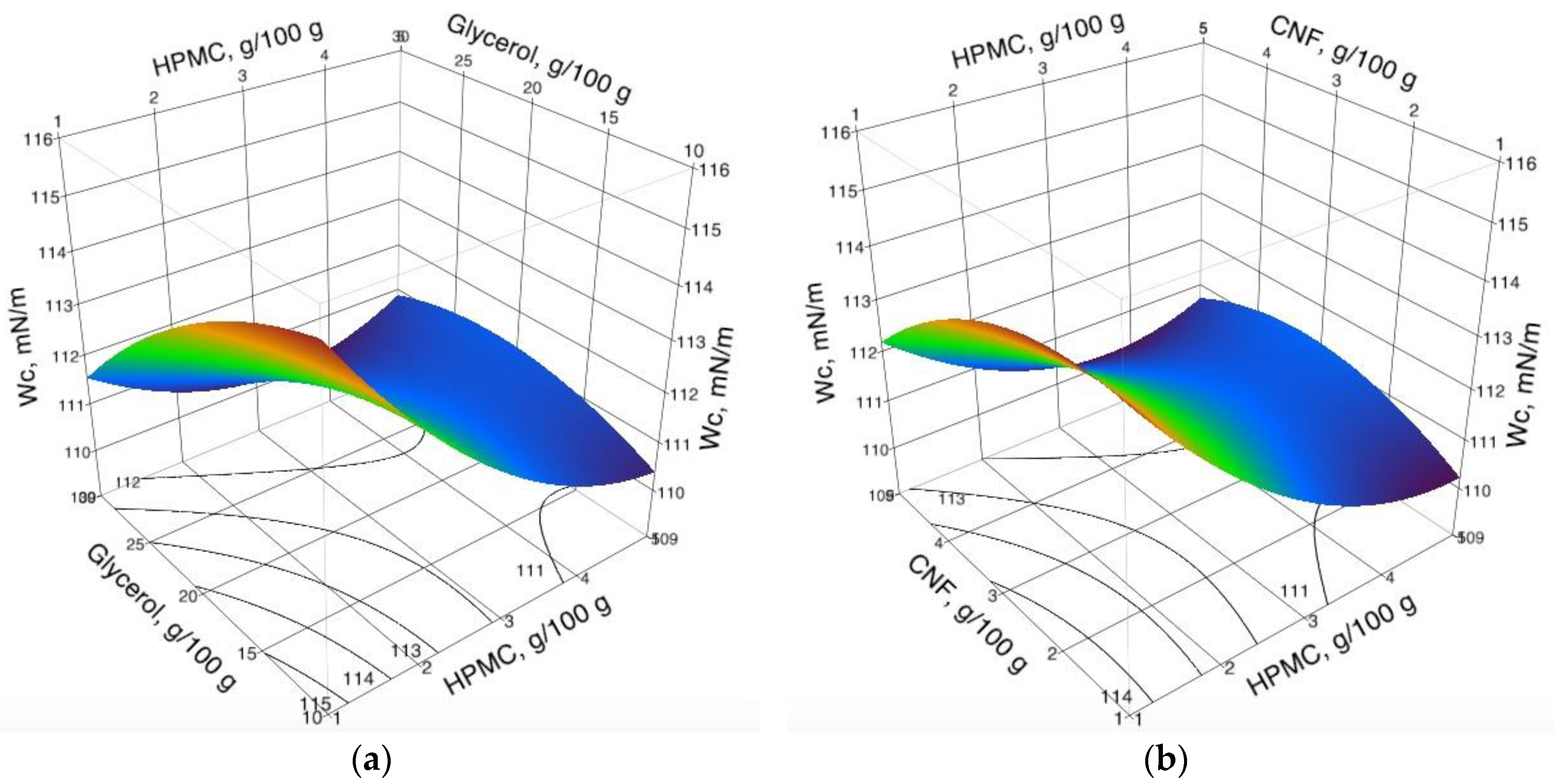
3.1. Cohesion Work (WC) of Coating Formulations
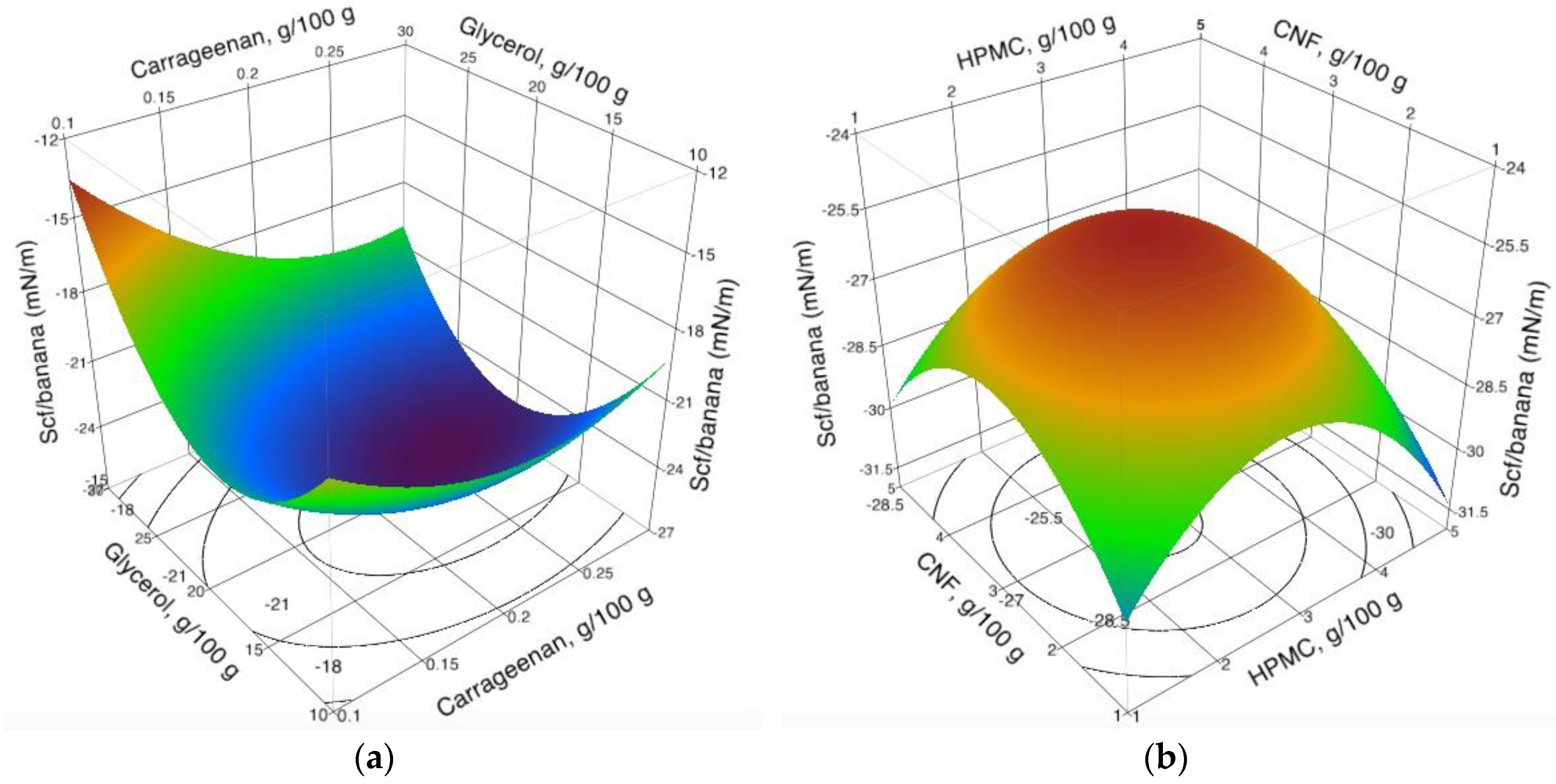
3.2. Spreading Coefficient of Coating Formulations on Banana Epicarp
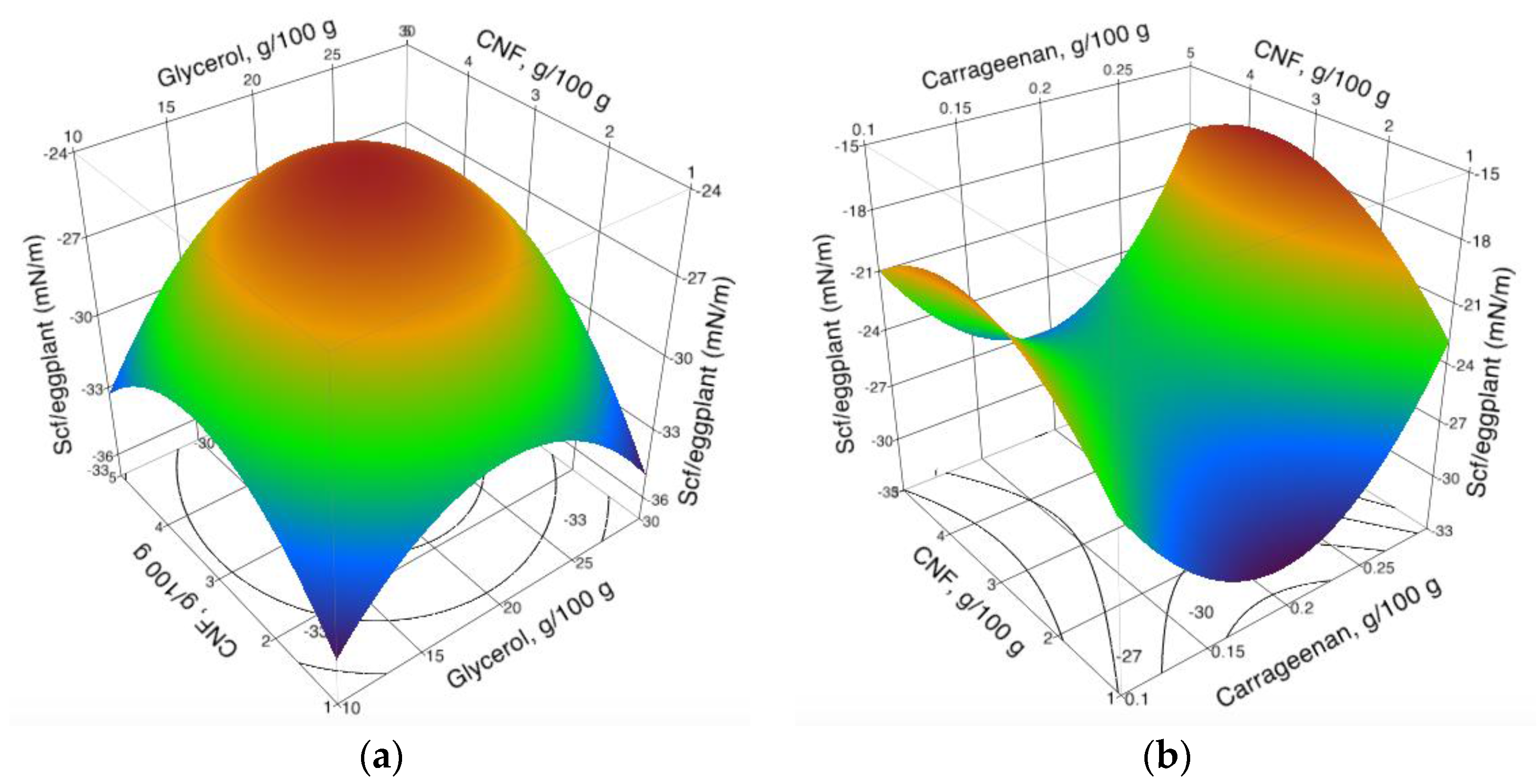
3.3. Spreading Coefficient of Coating Formulations on Eggplant Epicarp
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Andrade, R.D.; Skurtys, O.; Osorio, F.A. Atomizing spray systems for application of edible coatings. Compr. Rev. Food Sci. Food Saf. 2012, 11, 323–337. [Google Scholar] [CrossRef]
- Lin, D.; Zhao, Y. Innovations in the development and application of edible coatings for fresh and minimally processed fruits and vegetables. Compr. Rev. Food Sci. Food Saf. 2007, 6, 60–75. [Google Scholar] [CrossRef]
- Liu, Y.; Wang, S.; Lan, W.; Qin, W. Fabrication and testing of PVA/Chitosan bilayer films for strawberry packaging. Coatings 2017, 7, 109. [Google Scholar] [CrossRef]
- Gennadios, A.; McHugh, T.; Weller, C.; Krochta, J.M. Edible coatings and films based on proteins. In Edible Coatings and Films to Improve Food Quality; Krochta, J., Baldwin, E., Nisperos-Carriedo, M., Eds.; Technomic Publishing Company: Lancaster, PA, USA, 1994; pp. 201–277. [Google Scholar]
- McHugh, T.; Senesi, E. Apple wraps: A novel method to improve the quality and extend the shelf life of fresh-cut apples. J. Food Sci. 2000, 65, 480–485. [Google Scholar] [CrossRef]
- Fahs, A.; Brogly, M.; Bistac, S.; Schmitt, M. Hydroxypropyl methylcellulose (HPMC) formulated films: Relevance to adhesion and friction surface properties. Carbohydr. Polym. 2010, 80, 105–114. [Google Scholar] [CrossRef]
- Arora, A.; Padua, G.W. Review: Nanocomposites in food packaging. J. Food Sci. 2010, 75, R43–R49. [Google Scholar] [CrossRef] [PubMed]
- Azeredo, H.; Mattoso, L.; Avena-Bustillos, R.; Filho, G.; Munford, M.; Wood, D.; McHugh, T. Nanocellulose reinforced chitosan composite films as affected by nanofiller loading and plasticizer content. J. Food Sci. 2010, 75, N1–N7. [Google Scholar] [CrossRef] [PubMed]
- Savadekar, N.R.; Karande, V.S.; Vigneshwaran, N.; Bharimalla, A.K.; Mhaske, S.T. Preparation of nano cellulose fibers and its application in kappa-carrageenan based film. Int. J. Biol. Macromol. 2012, 51, 1008–1013. [Google Scholar] [CrossRef] [PubMed]
- Hershko, V.; Klein, E.; Nussinovith, A. Relationships between edible coatings and garlic skin. J. Food Sci. 1996, 61, 769–777. [Google Scholar] [CrossRef]
- Andrade, R.D.; Skurtys, O.; Osorio, F.A.; Zuluaga, R.; Gañán, P.; Castro, C. Wettability of gelatin coating formulations containing cellulose nanofibers on banana and eggplant epicarps. LWT Food Sci. Technol. 2014, 58, 158–165. [Google Scholar] [CrossRef]
- Andrade, R.D.; Skurtys, O.; Osorio, F.A. Drop impact behavior on food using spray coating: Fundamentals and applications. Food Res. Int. 2013, 54, 397–405. [Google Scholar] [CrossRef]
- Ribeiro, C.; Vicente, A.A.; Teixeira, J.A.; Miranda, C. Optimization of edible coating composition to retard strawberry fruit senescence. Postharvest Biol. Technol. 2007, 44, 63–70. [Google Scholar] [CrossRef]
- Skurtys, O.; Velásquez, P.; Henriquez, O.; Matiacevich, S.; Enrione, J.; Osorio, F. Wetting behavior of chitosan solutions on blueberry epicarp with or without epicuticular waxes. LWT Food Sci. Technol. 2011, 44, 1449–1457. [Google Scholar] [CrossRef]
- Ramírez, C.; Gallegos, I.; Ihl, M.; Bifani, V. Study of contact angle, wettability and water vapor permeability in carboxymethylcellulose (CMC) based film with murta leaves (Ugni molinae Turcz) extract. J. Food Eng. 2012, 109, 424–429. [Google Scholar] [CrossRef]
- Castro, C.; Zuluaga, R.; Putaux, J.; Caro, G.; Mondragon, I.; Gañan, P. Structural characterization of bacterial Cellulose produced by Gluconacetobacter swingii sp. from Colombian agroindustrial wastes. Carbohydr. Polym. 2011, 84, 96–102. [Google Scholar] [CrossRef]
- Kwok, D.Y.; Neumann, A.W. Contact angle measurement and contact angle interpretation. Adv. Colloid Interface Sci. 1999, 81, 167–249. [Google Scholar] [CrossRef]
- Neter, J.; Wasserman, W.; Kutner, M. Applied Linear Statistical Models: Regresion, Analysis of Variance, and Experimental Design, 2nd ed.; Richard D. Irwin, Inc.: Homewood, IL, USA, 1985. [Google Scholar]
- Avranas, A.; Tasopoulos, V. Aqueous solutions of Sodium deoxycholate and Hydroxypropylmethylcellulose: Dynamic surface tension measurements. J. Colloid Interface Sci. 2000, 221, 223–229. [Google Scholar] [CrossRef] [PubMed]
- Abdelmouleh, M.; Boufi, S.; Belgacem, M.N.; Duarte, A.P.; Ben Salah, A.; Gandini, A. Modification of cellulosic fibers with functionalized Silanes: Development of surface properties. Int. J. Adhes. Adhes. 2004, 24, 43–54. [Google Scholar] [CrossRef]
- Lara, I.; Belge, B.; Goulao, L.F. A focus on the biosynthesis and composition of cuticle in fruits. J. Agric. Food Chem. 2015, 63, 4005–4019. [Google Scholar] [CrossRef] [PubMed]
- Brieskorn, C.H.; Briner, M.; Leiner, U. Untersuchungen fiber die Bedeutung der Ursolsäure für die pftanzliche Kutikula. Arch. Pharm. 1954, 287, 305–309. (In German) [Google Scholar] [CrossRef]
- Wasan, D.; Nikolov, A.; Kondiparty, K. The wetting and spreading of nanofluids on solids: Role of the structural disjoining pressure. Curr. Opin. Colloid Interface Sci. 2011, 16, 344–349. [Google Scholar] [CrossRef]
- Brochard-Wyart, F.; de Gennes, P.; Hervetc, H. Wetting of stratified solids. Adv. Colloid Interface Sci. 1991, 34, 561–582. [Google Scholar] [CrossRef]
- Andrade, R.; Skurtys, O.; Osorio, F. Experimental study of drop impacts and spreading on epicarps: Effect of fluid properties. J. Food Eng. 2012, 109, 430–437. [Google Scholar] [CrossRef]
- Liu, D.; Pourrahimi, A.M.; Pallon, L.K.H.; Andersson, R.L.; Hedenqvist, M.S.; Gedde, U.W.; Olsson, R.T. Morphology and properties of silica-based coatings with different functionalities for Fe3O4, ZnO and Al2O3 nanoparticles. RSC Adv. 2015, 5, 48094–48103. [Google Scholar] [CrossRef]
- Pourrahimi, A.M.; Olsson, R.T.; Hedenqvist, M.S. The role of interfaces in Polyethylene/Metal-Oxide nanocomposites for ultrahigh-voltage insulating material. Adv. Mater. 2017, 1703624. [Google Scholar] [CrossRef] [PubMed]
- Xie, X. A review of recent advances in surface defect detection using texture analysis techniques. Electron. Lett. Comput. Vis. Image Anal. 2008, 7, 1–22. [Google Scholar]
| Run | Level of Variables | WC ± SD mN·m−1 | Banana | Eggplant | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| HPMC | κ-C † | G * | CNF | CA ± SD | WA ± SD mN·m−1 | Scf/Banana ± SD mN·m−1 | CA ± SD | WA ± SD mN·m−1 | Scf/Eggplant ± SD mN·m−1 | ||
| S1 | 1 | 0.1 | 10 | 1 | 114.02 ± 2.27 | 49.0 ± 1.7 | 94.42 ± 2.78 | −19.6 ± 0.51 | 61.5 ± 1.1 | 84.22 ± 1.93 | −29.8 ± 0.34 |
| S2 | 1 | 0.1 | 10 | 5 | 114.26 ± 2.64 | 65.0 ± 0.9 | 81.26 ± 1.51 | −33.0 ± 1.13 | 67.2 ± 1.7 | 79.26 ± 2.81 | −35.0 ± 0.17 |
| S3 | 1 | 0.1 | 30 | 1 | 115.64 ± 3.47 | 45.4 ± 1.6 | 98.44 ± 2.76 | −17.2 ± 0.71 | 59.2 ± 1.0 | 87.44 ± 1.72 | −28.2 ± 1.75 |
| S4 | 1 | 0.1 | 30 | 5 | 108.9 ± 2.38 | 39.8 ± 1.8 | 96.3 ± 3.03 | −12.6 ± 0.65 | 49.8 ± 1.3 | 89.6 ± 2.27 | −19.3 ± 0.11 |
| S5 | 1 | 0.3 | 10 | 1 | 124.7 ± 1.85 | 51.9 ± 1.6 | 100.8 ± 2.32 | −23.9 ± 0.47 | 48.3 ± 0.7 | 103.8 ± 1.17 | −20.9 ± 0.68 |
| S6 | 1 | 0.3 | 10 | 5 | 115.5 ± 2.06 | 53.5 ± 0.9 | 92.1 ± 1.58 | −23.4 ± 0.48 | 53.4 ± 1.8 | 92.2 ± 2.98 | −23.3 ± 0.92 |
| S7 | 1 | 0.3 | 30 | 1 | 111.04 ± 2.56 | 60.8 ± 1.5 | 82.64 ± 2.51 | −28.4 ± 0.50 | 59.9 ± 1.2 | 83.34 ± 1.96 | −27.7 ± 0.60 |
| S8 | 1 | 0.3 | 30 | 5 | 115.8 ± 2.95 | 49.1 ± 1.2 | 95.8 ± 1.85 | −20.0 ± 1.10 | 53.9 ± 1.3 | 92.0 ± 2.27 | −23.8 ± 0.68 |
| S9 | 5 | 0.1 | 10 | 1 | 112.78 ± 2.49 | 47.0 ± 1.8 | 94.88 ± 2.76 | −17.9 ± 0.27 | 61.6 ± 1.9 | 83.18 ± 2.92 | −29.6 ± 0.43 |
| S10 | 5 | 0.1 | 10 | 5 | 111.64 ± 2.52 | 50.5 ± 1.9 | 91.34 ± 3.13 | −20.3 ± 0.69 | 57.7 ± 1.8 | 85.64 ± 2.91 | −26.0 ± 0.39 |
| S11 | 5 | 0.1 | 30 | 1 | 109.78 ± 2.46 | 50.1 ± 1.1 | 90.08 ± 1.77 | −19.7 ± 0.61 | 59.1 ± 1.7 | 83.08 ± 2.77 | −26.7 ± 0.31 |
| S12 | 5 | 0.1 | 30 | 5 | 110.26 ± 3.11 | 57.5 ± 1.6 | 84.76 ± 2.61 | −25.5 ± 0.50 | 58.0 ± 1.4 | 84.36 ± 2.46 | −25.9 ± 0.65 |
| S13 | 5 | 0.3 | 10 | 1 | 109.96 ± 3.34 | 56.7 ± 1.7 | 85.16 ± 2.88 | −24.8 ± 0.46 | 64.8 ± 1.2 | 78.36 ± 1.90 | −31.6 ± 1.44 |
| S14 | 5 | 0.3 | 10 | 5 | 111.96 ± 3.33 | 57.3 ± 1.8 | 86.26 ± 2.88 | −25.7 ± 0.45 | 56.6 ± 1.7 | 86.76 ± 2.81 | −25.2 ± 0.52 |
| S15 | 5 | 0.3 | 30 | 1 | 114.00 ± 2.68 | 61.4 ± 1.7 | 84.3 ± 2.98 | −29.7 ± 0.30 | 61.6 ± 1.4 | 84.10 ± 2.40 | −29.9 ± 0.28 |
| S16 | 5 | 0.3 | 30 | 5 | 113.00 ± 2.69 | 53.9 ± 1.7 | 89.8 ± 2.75 | −23.2 ± 0.60 | 55.3 ± 1.4 | 88.70 ± 2.20 | −24.3 ± 0.49 |
| S17 | 1 | 0.2 | 20 | 3 | 112.24 ± 2.31 | 58.0 ± 1.6 | 85.84 ± 2.84 | −26.4 ± 0.53 | 59.0 ± 1.1 | 85.04 ± 1.90 | −27.2 ± 0.41 |
| S18 | 5 | 0.2 | 20 | 3 | 112.74 ± 2.96 | 66.8 ± 1.8 | 78.54 ± 2.83 | −34.2 ± 0.13 | 57.3 ± 1.5 | 86.84 ± 2.57 | −25.9 ± 0.39 |
| S19 | 3 | 0.1 | 20 | 3 | 113.46 ± 2.42 | 53.8 ± 1.7 | 90.26 ± 2.70 | −23.2 ± 0.28 | 50.0 ± 1.6 | 93.16 ± 2.71 | −20.3 ± 0.29 |
| S20 | 3 | 0.3 | 20 | 3 | 113.42 ± 2.42 | 58.4 ± 1.7 | 86.42 ± 2.58 | −27.0 ± 0.16 | 44.8 ± 1.1 | 96.92 ± 1.84 | −16.5 ± 0.58 |
| S21 | 3 | 0.2 | 10 | 3 | 110.42 ± 1.91 | 53.5 ± 1.8 | 88.02 ± 3.07 | −22.4 ± 1.16 | 64.7 ± 0.9 | 78.82 ± 1.47 | −31.6 ± 0.44 |
| S22 | 3 | 0.2 | 30 | 3 | 111.18 ± 2.13 | 51.8 ± 1.9 | 89.98 ± 2.93 | −21.2 ± 0.80 | 60.5 ± 1.2 | 82.98 ± 2.07 | −28.2 ± 0.60 |
| S23 | 3 | 0.2 | 20 | 1 | 109.52 ± 2.95 | 62.5 ± 1.7 | 80.02 ± 3.00 | −29.5 ± 0.50 | 69.6 ± 1.7 | 73.82 ± 2.80 | −35.7 ± 0.15 |
| S24 | 3 | 0.2 | 20 | 5 | 111.74 ± 2.76 | 62.8 ± 1.6 | 81.44 ± 2.49 | −30.3 ± 0.27 | 53.5 ± 1.4 | 89.14 ± 2.44 | −22.6 ± 0.32 |
| S25 | 3 | 0.2 | 20 | 3 | 110.62 ± 2.40 | 53.9 ± 1.6 | 87.92 ± 2.71 | −22.7 ± 0.31 | 55.8 ± 1.0 | 86.42 ± 1.66 | −25.2 ± 0.74 |
| S26 | 3 | 0.2 | 20 | 3 | 111.64 ± 2.33 | 54.7 ± 1.7 | 88.04 ± 2.91 | −23.6 ± 0.58 | 56.2 ± 1.4 | 86.84 ± 2.48 | −24.8 ± 0.15 |
| S27 | 3 | 0.2 | 20 | 3 | 112.28 ± 2.82 | 54.5 ± 1.6 | 88.78 ± 2.84 | −23.5 ± 0.20 | 56.6 ± 1.2 | 87.08 ± 2.21 | −25.2 ± 0.61 |
| S28 | 3 | 0.2 | 20 | 3 | 111.94 ± 1.91 | 54.9 ± 1.6 | 88.14 ± 2.80 | −23.8 ± 0.89 | 55.5 ± 1.2 | 87.64 ± 2.19 | −24.3 ± 0.28 |
| S29 | 3 | 0.2 | 20 | 3 | 113.36 ± 2.76 | 55.4 ± 1.7 | 88.86 ± 2.92 | −24.5 ± 0.16 | 56.5 ± 1.6 | 87.96 ± 2.90 | −25.4 ± 0.34 |
| S30 | 3 | 0.2 | 20 | 3 | 112.34 ± 2.44 | 54.8 ± 1.8 | 88.54 ± 2.89 | −23.8 ± 0.45 | 56.9 ± 1.0 | 86.84 ± 1.83 | −25.5 ± 0.61 |
| S31 | 3 | 0.2 | 20 | 3 | 110.32 ± 2.25 | 55.2 ± 1.4 | 86.62 ± 2.42 | −23.7 ± 0.17 | 55.5 ± 1.6 | 86.42 ± 2.88 | −25.9 ± 0.63 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Osorio, F.; Valdés, G.; Skurtys, O.; Andrade, R.; Villalobos-Carvajal, R.; Silva-Weiss, A.; Silva-Vera, W.; Giménez, B.; Zamorano, M.; Lopez, J. Surface Free Energy Utilization to Evaluate Wettability of Hydrocolloid Suspension on Different Vegetable Epicarps. Coatings 2018, 8, 16. https://doi.org/10.3390/coatings8010016
Osorio F, Valdés G, Skurtys O, Andrade R, Villalobos-Carvajal R, Silva-Weiss A, Silva-Vera W, Giménez B, Zamorano M, Lopez J. Surface Free Energy Utilization to Evaluate Wettability of Hydrocolloid Suspension on Different Vegetable Epicarps. Coatings. 2018; 8(1):16. https://doi.org/10.3390/coatings8010016
Chicago/Turabian StyleOsorio, Fernando, Gonzalo Valdés, Olivier Skurtys, Ricardo Andrade, Ricardo Villalobos-Carvajal, Andrea Silva-Weiss, Wladimir Silva-Vera, Begoña Giménez, Marcela Zamorano, and Johana Lopez. 2018. "Surface Free Energy Utilization to Evaluate Wettability of Hydrocolloid Suspension on Different Vegetable Epicarps" Coatings 8, no. 1: 16. https://doi.org/10.3390/coatings8010016
APA StyleOsorio, F., Valdés, G., Skurtys, O., Andrade, R., Villalobos-Carvajal, R., Silva-Weiss, A., Silva-Vera, W., Giménez, B., Zamorano, M., & Lopez, J. (2018). Surface Free Energy Utilization to Evaluate Wettability of Hydrocolloid Suspension on Different Vegetable Epicarps. Coatings, 8(1), 16. https://doi.org/10.3390/coatings8010016





