Assessment on the Effects of ZnO and Coated ZnO Particles on iPP and PLA Properties for Application in Food Packaging
Abstract
:1. Introduction
2. Experimental
2.1. Materials and Sample Preparation
2.2. Characterization Methodologies
2.2.1. Scanning Electron Microscopy (SEM)
2.2.2. Antibacterial Activity
2.2.3. UV-Visible Spectrophotometry
2.2.4. Thermogravimetric Analysis (TGA)
2.2.5. Mechanical Properties (Tensile)
2.2.6. Percentage Variation of Oxygen Permeability
3. Results and Discussion
3.1. Overall Morphology
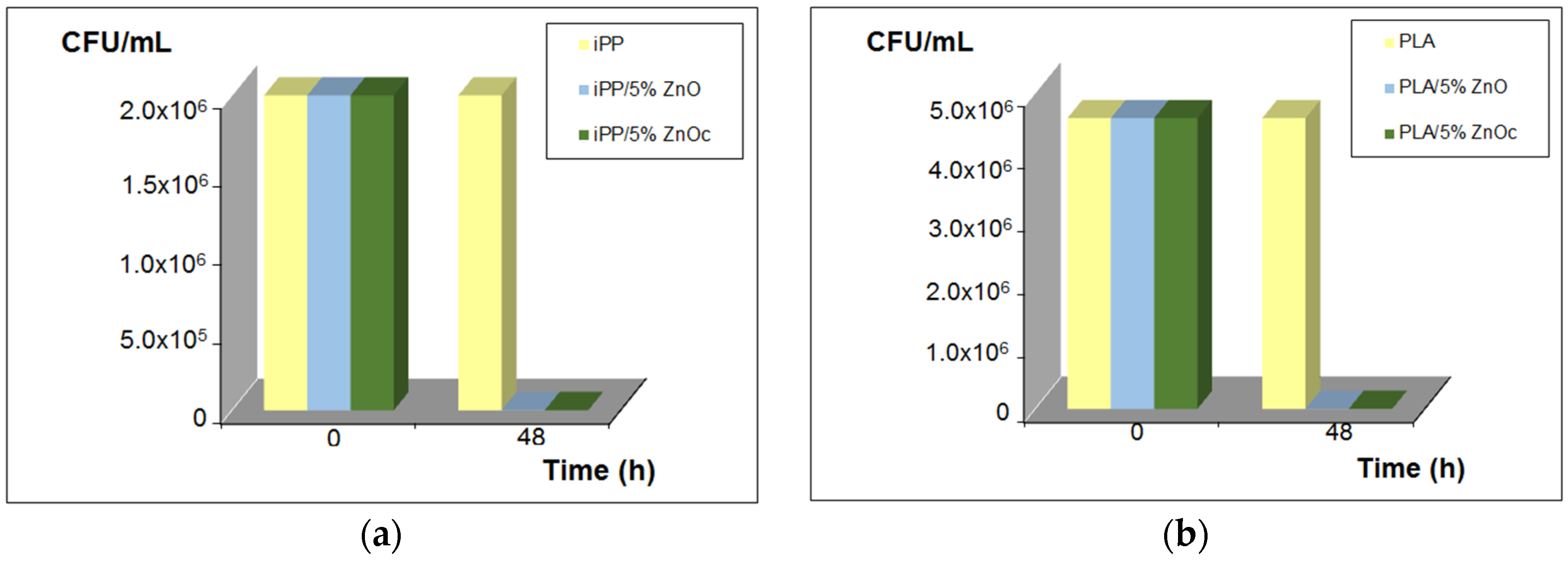
3.2. Antibacterial Activity
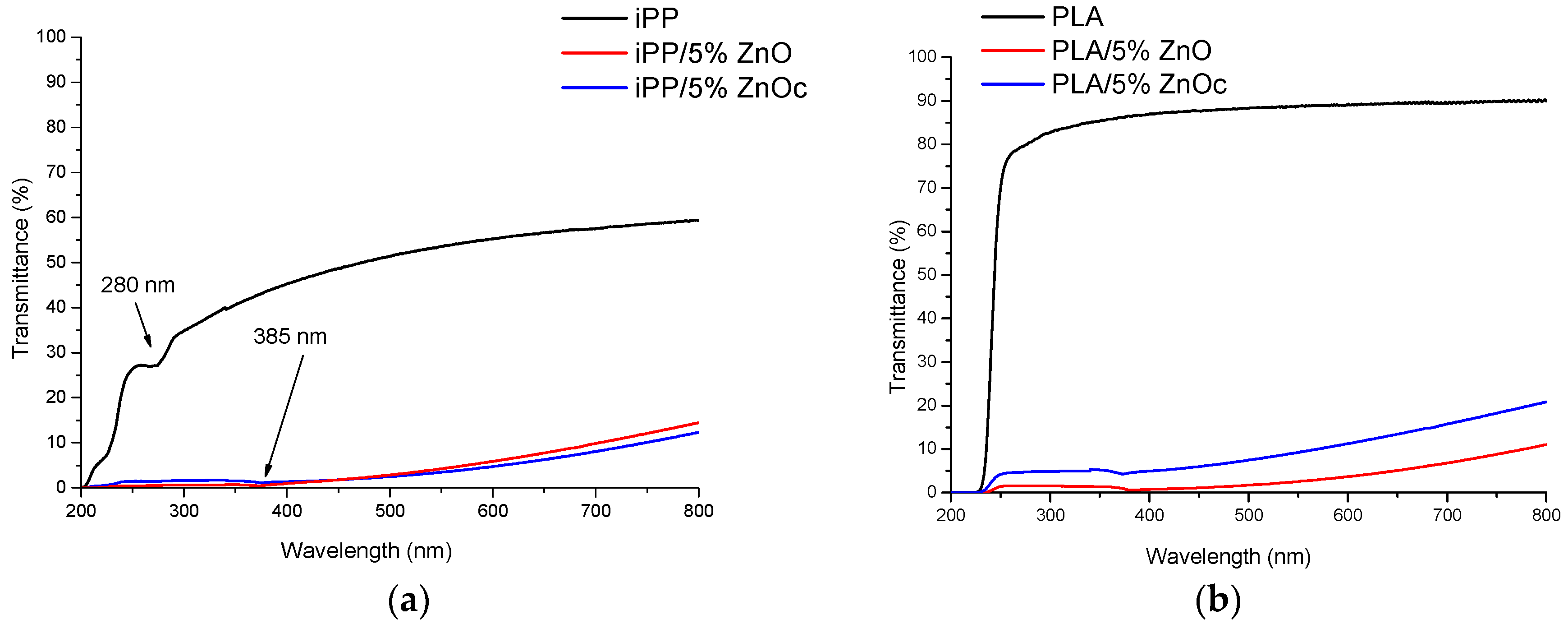
3.3. Photostability and Thermostability
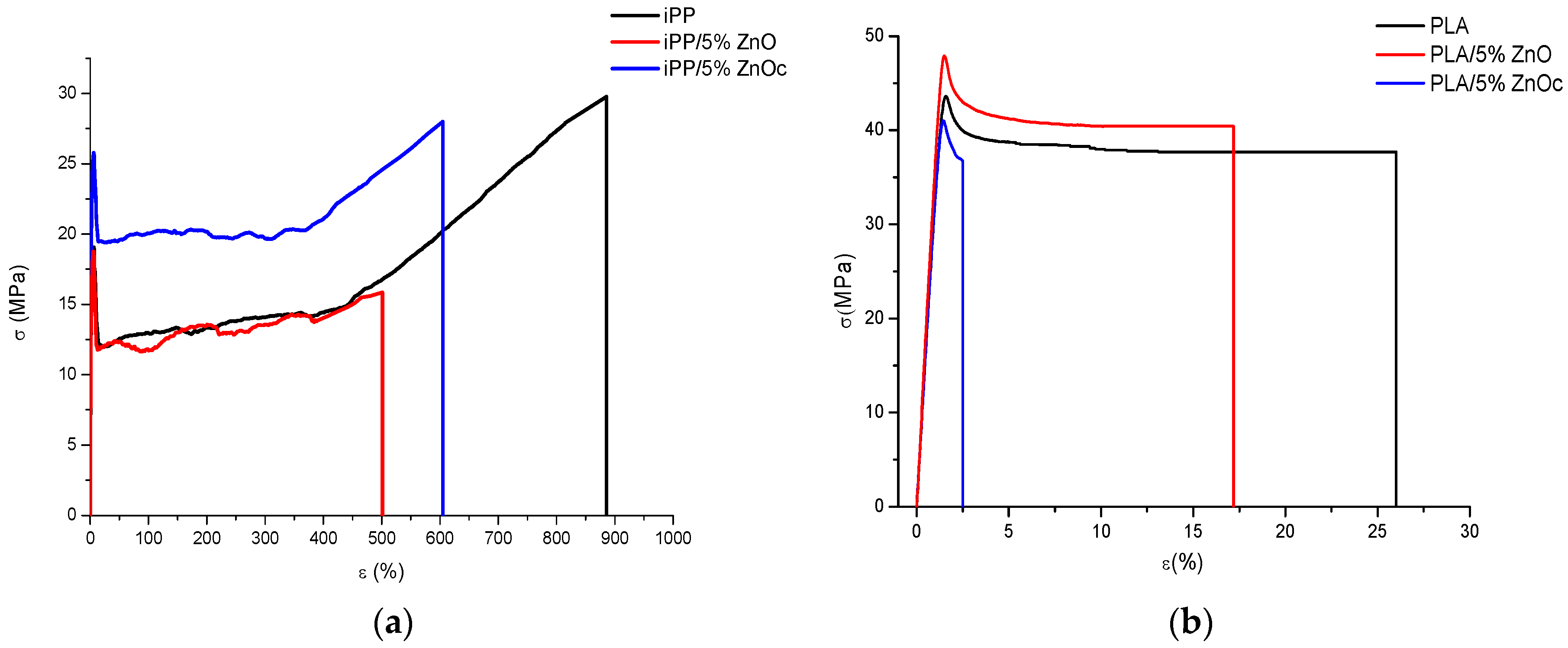
3.4. Mechanical Properties
- A slight increase in the modulus and in the stress at the yield point;
- No consistent variation in ɛy and σb;
- A decrease in the elongation at the break;
- A strong decrease of the elongation at the break mainly for the PLA/ZnOc system.
3.5. Percentage Variation of Oxygen Permeability
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Silvestre, C.; Cimmino, S. Ecosustainable Polymer Nanomaterials for Food Packaging: Innovative Solutions, Characterization Needs, Safety and Environmental Issues; Silvestre, C., Cimmino, S., Eds.; CRC Press: Boca Raton, FL, USA, 2013. [Google Scholar]
- Sahoo, S. Socio-ethical issues and nanotechnology development: perspectives from India. In Proceedings of 10th IEEE Conference on Nanotechnology (IEEE-NANO), Seoul, Korea, 17–20 August 2010.
- Sirelkhatim, A.; Mahmud, S.; Seeni, A.; Mohamad, K.N.H.; Ann, L.C.; Bakhori, S.K.M.; Hasan, H.; Mohamad, D. Review on Zinc Oxide Nanoparticles: Antibacterial Activity and Toxicity Mechanism. Nano-Micro Lett. 2015, 7, 219–242. [Google Scholar] [CrossRef]
- Garcia, M.; Forbe, T.; Gonzalez, E. Potential applications of nanotechnology in the agro-food sector. Cienc. Technol. Aliment. 2010, 30, 573–581. [Google Scholar] [CrossRef]
- Ayhan, Z. Potential Applications of Nanotechnology in Food Packaging. In Proceedings of 1st International Congress on Food Technology, Antalya, Turkey, 3–6 November 2010.
- Duraccio, D.; Silvestre, C.; Cimmino, S.; Marra, A.; Pezzuto, M. Polymer Morphology: Principles, Characterization, and Processing; Qi, P.G., Ed.; Wiley: New York, NY, USA, 2016; pp. 374–396. [Google Scholar]
- Malucelli, G. Hybrid Organic/Inorganic Coatings through Dual-Cure Processes: State of the Art and Perspectives. Coatings 2016, 6, 10. [Google Scholar] [CrossRef]
- Silvestre, C.; Cimmino, S.; Pezzuto, M.; Marra, A.; Ambrogi, V.; Dexpert-Ghys, J.; Verelst, M.; Augier, S.; Romano, I.; Duraccio, D. Preparation and characterization of isotactic polypropylene/zinc oxide microcomposites with antibacterial activity. Polym. J. 2013, 45, 938–945. [Google Scholar] [CrossRef]
- Marra, A.; Silvestre, C.; Duraccio, D.; Cimmino, S. Polylactic Acid/Zinc Oxide biocomposite films for food packaging application. Int. J. Biol. Macromol. 2016, 88, 254–262. [Google Scholar] [CrossRef] [PubMed]
- Silvestre, C.; Duraccio, D.; Marra, A.; Strongone, V.; Cimmino, S. Development of Antibacterial Composite Films Based on Isotactic Polypropylene and Coated ZnO Particles for Active Food Packaging. Coatings 2016, 6, 4. [Google Scholar] [CrossRef]
- Duncan, T.V. Applications of nanotechnology in food packaging and food safety: Barrier materials, antimicrobials and sensors. J. Colloid Interface Sci. 2011, 363, 1–24. [Google Scholar] [CrossRef] [PubMed]
- Mihindukulasuriya, S.D.F.; Lim, L.T. Nanotechnology development in food packaging: A review. Trends Food Sci. Technol. 2014, 40, 149–167. [Google Scholar] [CrossRef]
- Reuge, N.; Caussat, B.; Joffin, N.; Dexpert-Ghys, J.; Verelst, M.; Dxpert, H. Modelling of Spray Pyrolysis-Why the Synthesized Y2O3 Microparticles hollow. AlChE J. 2008, 54, 394–405. [Google Scholar] [CrossRef]
- Rossignol, C.; Verelst, M.; Dexpert-Ghys, J.; Rul, S. Synthesis of undoped ZnO nanoparticles by spray pyrolysis. Adv. Sci. Thecnol. 2006, 45, 237–241. [Google Scholar] [CrossRef]
- Caiut, J.M.A.; Dexpert-Ghys, J.; Kihn, Y.; Veelst, M.; Dexpert, H.; Ribeiro, S.J.L.; Messaddeq, Y. Elaboration of boehmite nano-powders by spray-pyrolisis. Powder Thecnol. 2009, 190, 95–98. [Google Scholar] [CrossRef]
- Bacsa, R.; Kihn, Y.; Verelst, M.; Dexpert-Ghys, J.; Bacsa, W.; Serp, P. Large scale synthesis of zinc oxide nanorods by homogeneous chemical vapour deposition and their characterization. Surf. Coat. Technol. 2007, 201, 9200–9204. [Google Scholar] [CrossRef]
- Zhou, N.; Liu, Y.; Li, L.; Meng, N.; Huang, Y.; Zhang, J.; Wei, S.; Shen, J. A new nanocomposite biomedical material of polymer. Curr. Appl. Phys. 2007, 7, 58–62. [Google Scholar] [CrossRef]
- Seyfriedsberger, G.; Rametsteine, K.; Kern, W. Polyethylene compounds with antimicrobial surface properties. Eur. Polym. J. 2006, 42, 3383–3389. [Google Scholar] [CrossRef]
- Kumar, R.; Münstedt, H. Silver ion release from antimicrobial polyamide/silver composites. Biomaterials 2005, 26, 2081–2088. [Google Scholar] [CrossRef] [PubMed]
- Soriano, C.B.; Martínez, V.P.; López, A.M.C.; Ortega, B.I.; Grande, B.M.J.; Pérez, P.R.; Gálvez, A.; Lucas, L.R. Effect of Activated Plastic Films on Inactivation of Foodborne Pathogens. Coatings 2016, 6, 28. [Google Scholar] [CrossRef]
- Silvestre, C.; Duraccio, D.; Cimmino, S. Food packaging based on polymer nanomaterials. Prog. Polym. Sci. 2011, 36, 1766–1782. [Google Scholar] [CrossRef]
- Liau, S.Y.; Read, D.C.; Pugh, W.; Furr, J.R.; Russell, A.D. Interaction of silver nitrate with readily identifiable groups: Relationship to the antibacterial action of silver ions. Lett. Appl. Microbiol. 1997, 25, 279–283. [Google Scholar] [CrossRef] [PubMed]
- Fujishima, A.; Rao, T.N.; Tryk, D.A. Titanium dioxide photocatalysis. Photochem. Photobiol. C 2000, 1, 1–21. [Google Scholar] [CrossRef]
- Santhosh, S.M.; Natarajan, K. Antibiofilm Activity of Epoxy/Ag-TiO2 Polymer Nanocomposite Coatings against Staphylococcus Aureus and Escherichia Coli. Coatings 2015, 5, 95–114. [Google Scholar]
- Santos, G.N.C.; Tibayan, E.B.; Castillon, G.B.; Estacio, E.; Furuya, T.; Iwamae, A.; Yamamoto, K.; Tani, M. Tin Oxide-Silver Composite Nanomaterial Coating for UV Protection and Its Bactericidal Effect on Escherichia coli (E. coli). Coatings 2014, 4, 320–328. [Google Scholar] [CrossRef]
- Jones, N.; Ray, B.; Ranjit, K.T.; Manna, A.C. Antibacterial activity of ZnO nanoparticle suspensions on a broad spectrum of microorganisms. FEMS Microbiol. Lett. 2008, 279, 71–76. [Google Scholar] [CrossRef] [PubMed]
- Sawai, J.; Yoshikawa, T.J. Quantitative evaluation of antifungal activity of metallic oxide powders (MgO, CaO and ZnO) by an indirect conductimetric assay. Appl. Microbiol. 2004, 96, 803–809. [Google Scholar] [CrossRef]
- Yamamoto, O.; Nakakoshi, K.; Sasamoto, T.; Nakagawa, H.; Miura, K. Adsorption and growth inhibition of bacteria on carbon materials containing zinc oxide. Carbon 2001, 39, 1643–1651. [Google Scholar] [CrossRef]
- Brayner, R.; Ferrari-lliou, R.; Brivois, N.; Djediat, S.; Benedetti, M.F.; Fiévet, F. Toxicological Impact Studies Based on Escherichia coli Bacteria in Ultrafine ZnO Nanoparticles Colloidal Medium. Nano Lett. 2006, 6, 866–870. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, O.; Komatsu, M.; Sawai, J.; Nakagawa, Z. Effect of lattice constant of zinc oxide on antibacterial characteristics. J. Mater. Sci. Mater. 2004, 15, 847–851. [Google Scholar] [CrossRef] [PubMed]
- Stoimenov, P.K.; Klinger, R.L.; Marchin, G.L.; Klabunde, K.J. Metal Oxide Nanoparticles as Bactericidal Agents. Langmuir 2002, 18, 6679–6686. [Google Scholar] [CrossRef]
- Tam, K.H.; Djurišic, A.B.; Chan, C.M.N.; Xi, Y.Y.; Tse, C.W.; Leung, Y.H.; Chan, W.K.; Leung, F.C.C.; Au, D.W.T. Antibacterial activity of ZnO nanorods prepared by a hydrothermal method. Thin Solid Films 2008, 516, 6167–6174. [Google Scholar] [CrossRef]
- Espitia, P.J.P.; Soares, N.F.F.; Coimbra, J.S.R.; de Andrade, N.J.; Cruz, R.S.; Medeiros, E.A. A Zinc Oxide Nanoparticles: Synthesis, Antimicrobial Activity and Food Packaging Applications. Food Bioprocess Technol. 2012, 5, 1447–1464. [Google Scholar] [CrossRef]
- Martínez-Bueno, M.J.; Cimmino, S.; Silvestre, C.; Tadeo, J.L.; Garcia-Valcárcel, A.I.; Fernández-Alba, A.R.; Hernando, M.D. Characterization of non-intentionally added substances (NIAS) and zinc oxide nanoparticle release from evaluation of new antimicrobial food contact materials by both LC-QTOF-MS, GC-QTOF-MS and ICP-MS. Anal. Methods 2016, 8, 7209–7216. [Google Scholar] [CrossRef]
- Marra, A.; Boumail, A.; Cimmino, S.; Criado, P.; Silvestre, C.; Lacroix, M. Effect of PLA/ZnO Packaging and Gamma Radiation on the Content of Listeria innocua, Escherichia coli and Salmonella enterica on Ham during Storage at 4 °C. J. Food Sci. Eng. 2016, 6, 245–259. [Google Scholar]
- Salvatore, M.; Marra, A.; Duraccio, D.; Shayanfar, S.; Pillai, S.D.; Cimmino, S.; Silvestre, C. Effect of electron beam irradiation on the properties of polylactic acid/montmorillonite nanocomposites for food packaging applications. J. Appl. Polym. Sci. 2016, 133, 42219. [Google Scholar] [CrossRef]
- Weber, C.J.; Haugaard, V.; Festersen, R.; Bertelsen, G. Production and applications of biobased packaging materials for the food industry. J. Food Addit. Contam. 2002, 19, 172–177. [Google Scholar] [CrossRef] [PubMed]
- Gurav, A.; Kodas, T.; Pluym, T.; Xiong, Y. Aerosol processing of materials. Aerosol Sci. Technol. 1993, 19, 411–452. [Google Scholar] [CrossRef]
- Pratsinis, S.E.; Vemury, S. Particle formation in gases. Powder Technol. 1996, 88, 267–273. [Google Scholar] [CrossRef]
- Messing, G.L.; Zhang, S.C.; Jayanthi, G.V. Ceramic powder synthesis by spray pyrolysis. J. Am. Ceram. Soc. 1993, 76, 2707–2726. [Google Scholar] [CrossRef]
- Alavi, S.; Caussat, B.; Couderc, J.P.; Dexpert-Ghys, J.; Joffin, N.; Neumeyer, D.; Verelst, M. Spray pyrolysis synthesis of submicronic particles. Possibilities and limits. Adv. Sci. Technol. A 2003, 30, 417–424. [Google Scholar]
- ASTM E2149-10 Standard Test Method for Determining the Antimicrobial Activity of Immobilized Antimicrobial Agents Under Dynamic Contact Conditions; ASTM International: West Conshohocken, PA, USA, 2010.
- Bocchini, S.; Fukushima, K.; Di Blasio, A.; Fina, A.; Frache, A.; Geobaldo, F. Polylactic Acid and Polylactic Acid-Based Nanocomposite Photooxidation. Biomacromolecules 2010, 11, 2919–2926. [Google Scholar] [CrossRef] [PubMed]
- Utracki, L.A. Compatibilization of polymer blends. Can. J. Chem. Eng. 2002, 80, 1008–1016. [Google Scholar] [CrossRef]


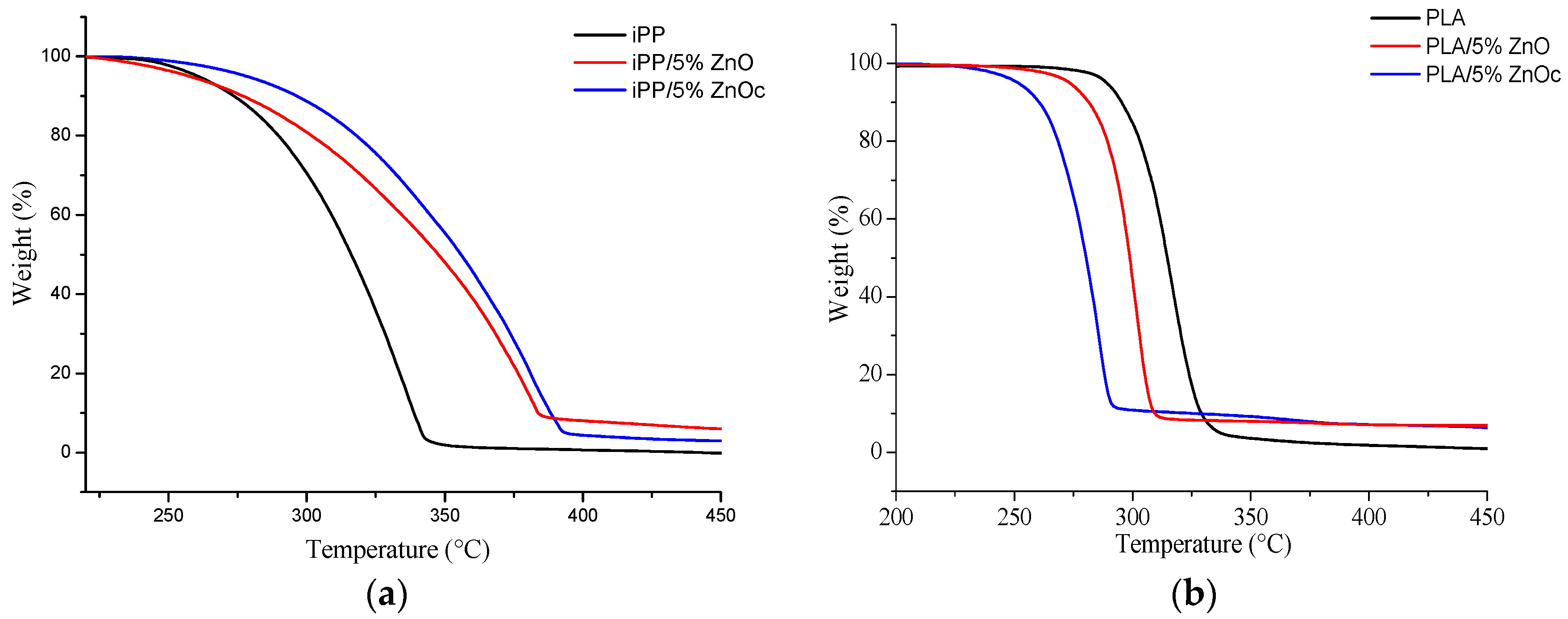
| Sample | T50% (°C) |
|---|---|
| iPP | 315 ± 1 |
| iPP/5%ZnO | 347 ± 1 |
| iPP/5%ZnOc | 355 ± 1 |
| PLA | 315 ± 1 |
| PLA/5%ZnO | 299 ± 1 |
| PLA/5%ZnOc | 280 ± 1 |
| Sample | E (MPa) | σy (MPa) | ɛy (%) | σb (MPa) | ɛb (%) |
|---|---|---|---|---|---|
| iPP | 1350 ± 100 | 19 ± 3 | 7 ± 2 | 30 ± 3 | 890 ± 65 |
| iPP/5%ZnO | 1430 ± 166 | 19 ± 2 | 6 ± 1 | 16 ± 4 | 500 ± 120 |
| iPP/5%ZnOc | 1515 ± 79 | 26 ± 1 | 7 ± 1 | 28 ± 3 | 605 ± 76 |
| Samples | E (MPa) | σy (MPa) | ɛy (%) | σb (MPa) | ɛb (%) |
|---|---|---|---|---|---|
| PLA | 3470 ± 189 | 44 ± 2 | 2 ± 1 | 38 ± 2 | 26 ± 5 |
| PLA/5%ZnO | 3853 ± 107 | 48 ± 3 | 2 ± 1 | 40 ± 3 | 17 ± 6 |
| PLA/5%ZnOc | 3492 ± 184 | 37 ± 3 | 3 ± 1 | 41 ± 3 | 2 ± 1 |
| Sample | ΔO2 Permeability (%) |
|---|---|
| iPP/5%ZnO | +2% |
| iPP/5%ZnOc | −3% |
| PLA/5%ZnO | −18% |
| PLA/5%ZnOc | +1% |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Marra, A.; Rollo, G.; Cimmino, S.; Silvestre, C. Assessment on the Effects of ZnO and Coated ZnO Particles on iPP and PLA Properties for Application in Food Packaging. Coatings 2017, 7, 29. https://doi.org/10.3390/coatings7020029
Marra A, Rollo G, Cimmino S, Silvestre C. Assessment on the Effects of ZnO and Coated ZnO Particles on iPP and PLA Properties for Application in Food Packaging. Coatings. 2017; 7(2):29. https://doi.org/10.3390/coatings7020029
Chicago/Turabian StyleMarra, Antonella, Gennaro Rollo, Sossio Cimmino, and Clara Silvestre. 2017. "Assessment on the Effects of ZnO and Coated ZnO Particles on iPP and PLA Properties for Application in Food Packaging" Coatings 7, no. 2: 29. https://doi.org/10.3390/coatings7020029
APA StyleMarra, A., Rollo, G., Cimmino, S., & Silvestre, C. (2017). Assessment on the Effects of ZnO and Coated ZnO Particles on iPP and PLA Properties for Application in Food Packaging. Coatings, 7(2), 29. https://doi.org/10.3390/coatings7020029







