Molybdenum Nitride Films: Crystal Structures, Synthesis, Mechanical, Electrical and Some Other Properties
Abstract
:1. Introduction
2. Structure of Molybdenum Nitride Phases
2.1. γ-Mo2N1 ± x and β-Mo2N1 ± x Phases
| Mo–N Phase | Structure | Space Group | Lattice Constants (nm) | References |
|---|---|---|---|---|
| γ-Mo2N b | Face centred cubic | Fm3m | a = 0.41613 | [24] |
| γ-Mo2N f | a = 0.416–0.419 | [25] | ||
| a = 0.416 | [26] | |||
| a = 0.4124 | [27] | |||
| a = 0.42 | [28] | |||
| a = 0.4165 | [12] | |||
| a = 0.4215–0.4303 | [29] | |||
| a = 0.421–0.425 | [30] | |||
| a = 0.419–0.429 | [31] | |||
| a = 0.418–0.429 | [32] | |||
| β-Mo2N f | Body centred tetragonal | I41/amd | a = 0.416 c = 0.800 | [28] |
| β-Mo2N0.85 f | a = 0.4199 c = 0.7996 | [33] | ||
| β-Mo2N f | a = 0.4182 c = 0.7993 | [34] | ||
| β-Mo16N7 b | Tetragonal | a = 0.841 c = 0.805 | [23] | |
| MoN0.5 th | Cubic | a = 0.4162 | [35] |
2.2. Hexagonal δ-MoN Phase
| Mo–N Phase | Structure Type | Space Group | Lattice Constants (nm) | References | |
|---|---|---|---|---|---|
| δ-MoN b | Hexagonal | P63mc | a = 0.57402 | [37] | |
| c = 0.56203 | |||||
| δ-MoN f | Hexagonal | a = 0.572 | [25] | ||
| c = 0.560 | |||||
| a = 0.572 | [26] | ||||
| b = 0.556 | |||||
| δ1-MoN f | Hexagonal WC-type | P6m2 | a = 0.2868 | [36] | |
| c = 0.2810 | |||||
| δ3-MoN f | Hexagonal NiAs-type | P63mc | a = 0.57356 | [36] | |
| c = 0.56281 | |||||
| Mo5N6 f | Hexagonal WC and NiAs-type | P63/m | a = 0.48924 | [36] | |
| c = 1.10643 | |||||
| MoN th | Hexagonal | a = 0.5787 | [35] | ||
| c = 0.5404 | |||||
| δ1-MoN th | a = 0.286 | [38] | |||
| c = 0.280 | |||||
| δ3-MoN th | a = 0.5710 | [38] | |||
| c = 0.5625 | |||||
| δ1-MoN th | P6m2 | a = 0.2861 | [39] | ||
| c = 0.2843 | |||||
| δ2-MoN th | P63/mmc | a = 0.5700 | [39] | ||
| c = 0.5661 | |||||
| δ3-MoN th | P63mc | a = 0.5721 | [39] | ||
| c = 0.5649 | |||||
| MoN f | Cubic NaCl-B1-type | Fm3m | a = 0.4212 | [40] | |
| a = 0.420–0.427 | [25] | ||||
| a = 0.4215–0.4253 | [41] | ||||
| a = 0.420–0.422 | [26] | ||||
| MoN th | a = 0.425 | [42] | |||
2.3. Metastable Cubic MoN Phase
2.4. Amorphous Structures
3. Formation of Molybdenum Nitride Compounds
3.1. Thermodynamic Analysis
3.2. Application to the Synthesis of Molybdenum Nitride Compounds
4. Deposition of Molybdenum Nitride Films and Coatings
4.1. Reactive Sputtering Method
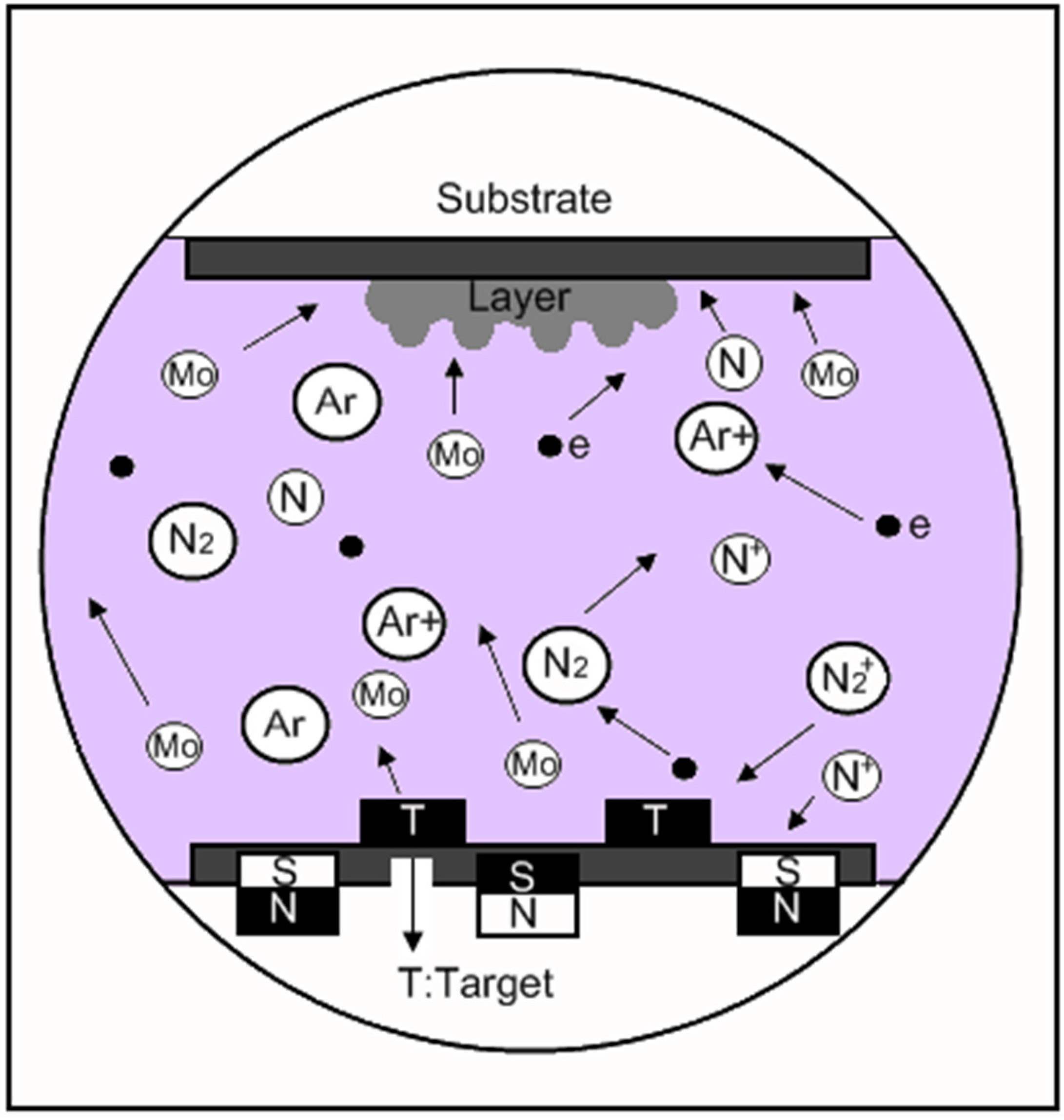
4.1.1. Influence of the Nitrogen Pressure on the Formation of Crystalline or Amorphous Mo2N Phases
4.1.2. Influence of the Nitrogen Pressure on the Formation of MoN Phases
4.2. Reactive Cathodic Arc Evaporation Method
4.3. Ion Implantation Method
4.3.1. Influence of the Temperature on the Formation of Mo2N Phases
4.3.2. Influence of Nitrogen Doses on the Formation of Mo2N and MoN Phases
4.4. Low-Energy Ion-Assisted Method
4.5. Pulsed Laser Method
4.6. Atomic Layer Deposition (ALD)
4.7. Plasma Enhanced Chemical Vapor Deposition Techniques
4.7.1. Hexagonal MoN Films
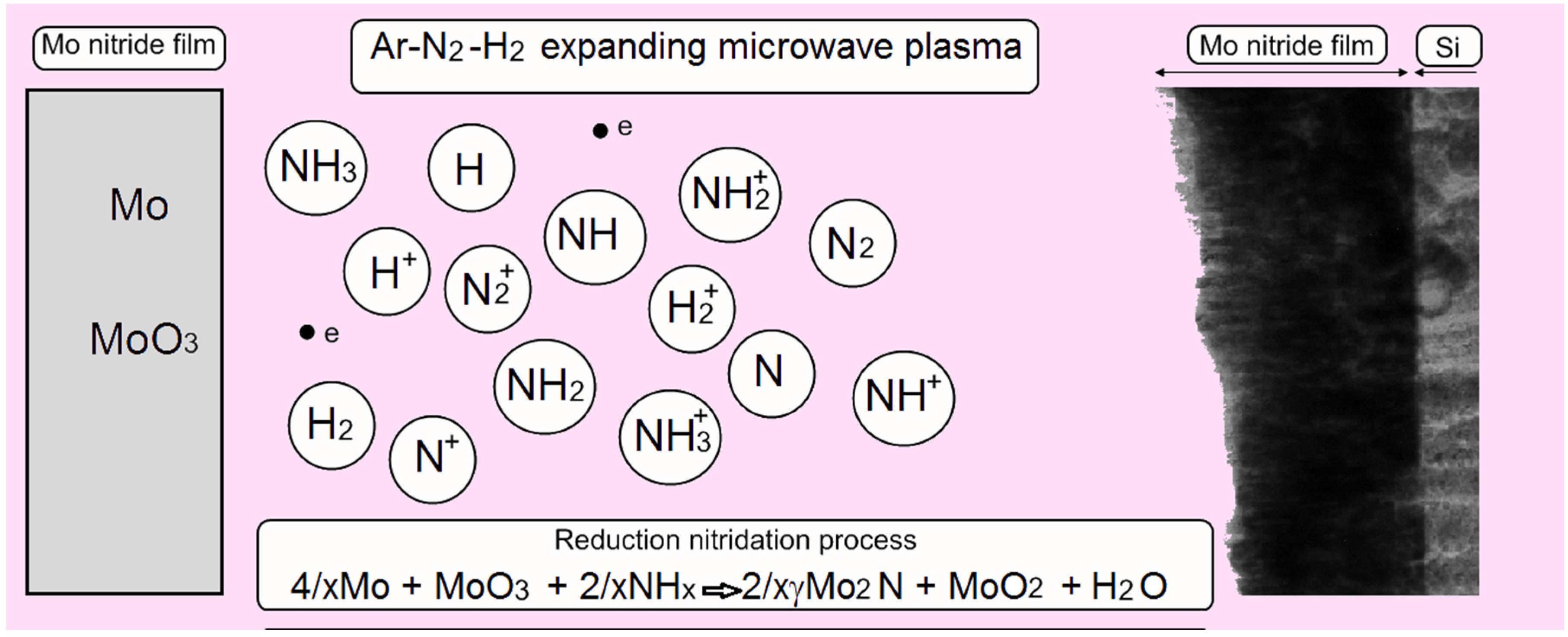
4.7.2. β-Mo2N and γ-Mo2N Films in Expanding Plasma

4.8. Metal Organic Chemical Vapor Deposition
4.9. Deposition of MoN Coatings by Chemical Process
4.9.1. Polymer Assisted Deposition (PAD)
4.9.2. Thermal Processes
5. Mechanical Properties of Molybdenum Nitride Coatings
5.1. γ-Mo2N Phase
| Mo–N Phase Thickness (μm) | Bulk Modulus (GPa) | Young’s Modulus (GPa) | Hardness (GPa) | References |
|---|---|---|---|---|
| Mo | 264.7 | 320 | 1.53 | [2,38,39] |
| γ-Mo2N b | 16 | [2] | ||
| 304 | [24] | |||
| γ-Mo1.8N f (1) | 365 | 28–29 | [30] | |
| γ-Mo2N f (0.1) | 210 | 12 | [8] | |
| β-Mo2N f (0.1) | 130 | 6–7 | [8] | |
| cubic MoN0.5 th | 348 | [35] | ||
| δ3-MoN b | 345 | [58] | ||
| δ-MoN1.2 f (1) | 420 | 26–27 | [30] | |
| hex MoN th | 392 | [35] | ||
| δ3-MoN th | 338–351 | [59] | ||
| δ1-MoN th | 377 | 640 | [38] | |
| δ3-MoN th | 379 | 611 | [38] | |
| 327 | 534 | 29 | [39] | |
| δ2-MoN th | 356 | 600 | 34 | [39] |
| δ1-MoN th | 353 | 463 | [39] | |
| cubic MoN th | 389 | [35] | ||
| cubic MoN b | 390 | [60] | ||
| cubic MoN th | 352 | 462 | [38] | |
| cubic BN b | 370 | 60 | [24] | |
| C (diamond) b | 443 | 900 | 110 | [2] |
5.2. Stoichiometric δ-MoN and Cubic MoN Phases
6. Resistivity
| Mo–N Phase Thickness (μm) | Resistivity (μΩcm) | Tc (K) | References |
|---|---|---|---|
| Mo b | 5.47 | [2] | |
| Mo f | 7.5–12 | [31] | |
| Mo f (0.3) | 13 | [9] | |
| Mo2N b | 19.8 | [2] | |
| γ-Mo2N b | 5.2 | [24] | |
| Cubic Mo2N f | 6–7 | [25] | |
| γ-Mo2N f (0.15) | 25–80 | 4–6 | [26] |
| Mo2N f (0.2) | 180 | [9] | |
| Mo2N f | 500–900 | [29] | |
| 300–550 | [31] | ||
| γ-Mo2N f (0.01) | 100 | [11] | |
| γ-Mo2N f (1) | 2483 | [10] | |
| γ-Mo2N f | 245 | [12] | |
| 4.5 | [56] | ||
| Mo2N f | 150–460 | [32] | |
| β-Mo2N f (0.036) | 5.2 | [33] | |
| (a) Mo–N f | 1250 | [31] | |
| (a) Mo2N3 f | 104 | [43] | |
| δ-MoN f (0.15) | 110 | 4 | [26] |
| δ1-MoN f | 4 | [36] | |
| δ3-MoN f | 12 | [36] | |
| Mo5N6 b | 12 | [36] | |
| δ-MoN f | 1000 | [12] | |
| 12 | [56] | ||
| 13.2 | [25] | ||
| cubic MoN f (0.15–0.4) | 850–1700 | 3–6 | [40] |
| cubic MoN f | 40–70 | 6.8 | [26] |
| cubic MoN f (0.4) | 120–200 | 5–6.15 | [41] |
| cubic MoN th | 29.4 | [42] | |
| C (diamond) b | 1018 | [2] |
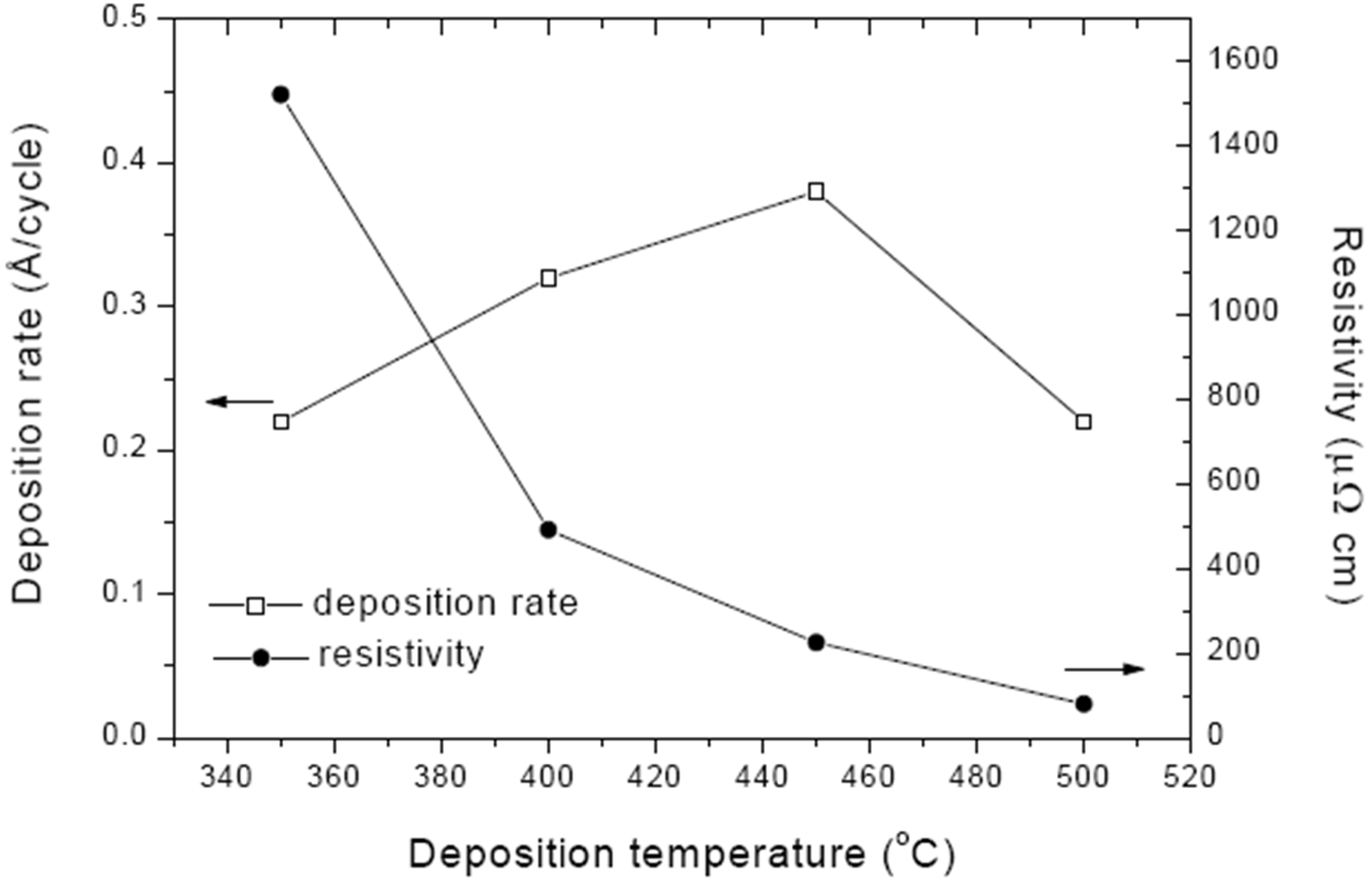
6.1. Molybdenum Nitride Films Prepared by Reactive Sputtering
6.2. Molybdenum Nitride Films Prepared by Low-Energy Ion-Assisted Process
6.3. Molybdenum Nitride Films Prepared by Ion Implantation
7. Superconducting Properties
7.1. γ-Mo2N and β-Mo2N Phase
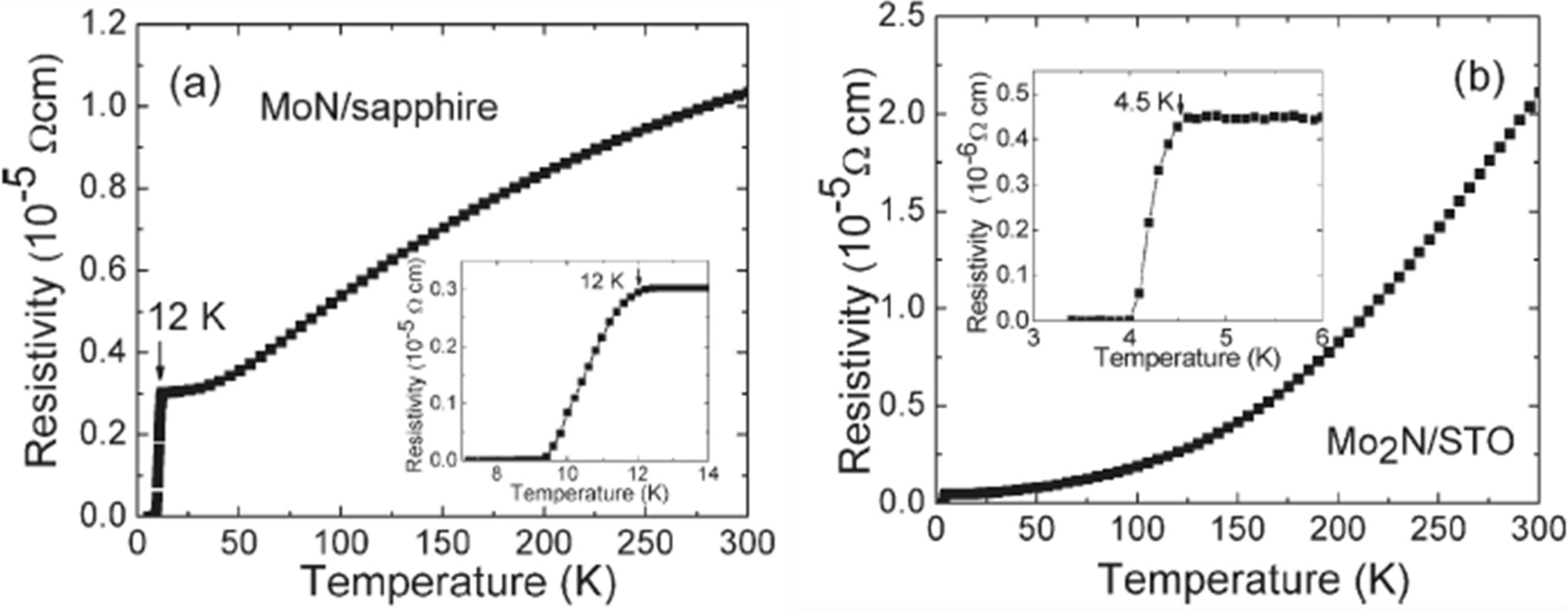
7.2. δ-MoN Phase
7.3. Stoichiometric MoN Phase of Cubic Structure
8. Some Other Properties
9. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Toth, L. Transition Metal Carbides and Nitrides; Academic Press: New York, NY, USA; London, UK, 1971; pp. 29, 247–261. [Google Scholar]
- Oyama, S.T. Introduction to the chemistry of transition metal carbides and nitrides. In The Chemistry of Transition Carbides and Nitrides; Oyama, S.T., Ed.; Blackie Academic and Professional: London, Glasgow, UK, 1996; pp. 2–10. [Google Scholar]
- Gubanov, V.A.; Ivanovsky, A.L.; Zhukov, V.P. Electronic structure and interatomic interactions in transition-metal nitrides. In Electronic Structure of Refractory Carbides and Nitrides; Gubanov, V.A., Ivanovsky, A.L., Zhukov, V.P., Eds.; Cambridge University Press: Cambridge, UK, 1994; pp. 70–75. [Google Scholar]
- Chen, W.; Jiang, J.Z. Elastic properties and electronic structures of 4d- and 5d-transition metal mononitrides. J. Alloys Compd. 2010, 499, 243–254. [Google Scholar] [CrossRef]
- Aouadi, S.M.; Paudel, Y.; Luster, B.; Stadler, S.; Kohli, P.; Muratore, C.; Hager, C.; Voevodin, A.A. AdaptiveMo2N/MoS2/Ag Tribological nanocomposite coatings for aerospace applications. Tribol. Lett. 2008, 29, 95–103. [Google Scholar] [CrossRef]
- Aouadi, S.M.; Paudel, Y.; Simonson, W.J.; Ge, Q.; Kohli, P.; Muratore, C.; Voevodin, A.A. Tribological investigations of adaptiveMo2N/MoS2/Ag coatings with high sulfur content. Surf. Coat. Technol. 2009, 203, 1304–1309. [Google Scholar] [CrossRef]
- Gulbinski, W.; Suszko, T. Thin films of Mo2N/Ag nanocomposite-the structure, mechanical and tribological properties. Surf. Coat. Technol. 2006, 201, 1469–1476. [Google Scholar] [CrossRef]
- Khojier, K.; Karami Mehr, M.R.; Savaloni, H. Annealing temperature effect on the mechanical and tribological properties of molybdenum nitride thin films. J. Nanostruct. Chem. 2013, 3, 1–7. [Google Scholar] [CrossRef]
- Kattelus, H.; Koskenala, J.; Nurmela, A.; Niskanen, A. Stress control of sputter-deposited Mo–N films for micromechanical applications. Microelectron. Eng. 2002, 60, 97–105. [Google Scholar] [CrossRef]
- Rische, D. MOCVD of Tungsten and Molybdenum Nitrides. Ph.D. Thesis, University of Bochum, Bochum, Germany, 2007. [Google Scholar]
- Alen, P. Atomic Layer Deposition of TaN, NbN and MoN films for Cu Metallizations. Ph.D. Thesis, University of Helsinski, Helsinski, Finland, 2005. [Google Scholar]
- Guntur, V. Molybdenum Nitride Films in the Back Contact Structure of Flexible Substrates CdTe Solar Cells. Ph.D. Thesis, University of South Florida, Tampa, USA, 2011. [Google Scholar]
- Shuangxi, S.; Yuzhang, L.; Dali, M.; Huiqin, L.; Ming, L. Diffusion barrier performances of thin Mo, Mo–N and Mo/Mo–N films between Cu and Si. Thin Solid Films 2005, 476, 142–147. [Google Scholar]
- Reid, J.S. MEMS with Flexible Portions Made of Novel Materials. U.S. Patent 7071520B2, 4 July 2006. [Google Scholar]
- Nagai, M. Transition-metal nitrides for hydrotreating catalyst-synthesis, surface properties, and reactivities. Appl. Catal. A Gen. 2007, 322, 178–190. [Google Scholar] [CrossRef]
- McKay, D. Catalysis over Molybdenum Containing Nitride Materials. Ph.D. Thesis, University of Glasgow, Glasgow, UK, 2008. [Google Scholar]
- Zhu, J.F.; Guo, J.C.; Zhai, R.S.; Bao, X.; Zhang, X.Y.; Zhuang, S. Preparation and adsorption properties of Mo2N model catalyst. Appl. Surf. Sci. 2000, 161, 86–93. [Google Scholar] [CrossRef]
- Shi, C.; Zhu, A.M.; Yang, X.F.; Au, C.T. On the catalytic nature of VN, Mo2N and W2N nitrides for NO reduction with hydrogen. Appl. Catal. A Gen. 2004, 276, 223–230. [Google Scholar] [CrossRef]
- Tagliazucca, V.; Schlichte, K.; Schüth, F.; Weidenthaler, C. Molybdenum-based catalysts for the decomposition of ammonia: In situ X-ray diffraction studies, microstructure, and catalytic properties. J. Catal. 2013, 305, 277–289. [Google Scholar] [CrossRef]
- Tagliazucca, V.; Leoni, M.; Weidenthaler, C. Crystal structure and microstructural changes of molybdenum nitrides traces during catalytic reaction by in situ X-ray diffraction studies. Phys. Chem. Chem. Phys. 2014, 16, 6182–6188. [Google Scholar] [CrossRef] [PubMed]
- Gong, S.W.; Chen, H.K.; Li, W.; Li, B.Q. Catalytic behaviors of β-Mo2N0.78 as a hydrodesulfurization catalyst. Energy Fuels 2006, 20, 1372–1376. [Google Scholar] [CrossRef]
- Jehn, H.; Ettmayer, P. The molybdenum-nitrogen phase diagram. J. Less-Common Met. 1978, 58, 85–98. [Google Scholar] [CrossRef]
- Karam, R.; Ward, R. The Preparation of β-Molybdenum Nitride. Inorg. Chem. 1970, 9, 1385–1387. [Google Scholar] [CrossRef]
- Bull, C.L.; Kawashima, T.; McMillan, P.F.; Machon, D.; Shebanova, O.; Daisenberger, D.; Soignard, E.; Takayama-Muromachi, E.; Chapon, L.C. Crystal structure and high-pressure properties of γ-Mo2N determined by neutron powder diffraction and X-ray diffraction. J. Solid State Chem. 2006, 179, 1762–1767. [Google Scholar] [CrossRef]
- Ihara, H.; Kimura, Y.; Senzaki, K.; Zezuka, H.; Hirabayashi, M. Electronic structures of B1 MoN, fcc Mo2N and hexagonal MoN. Phys. Rev. B 1985, 31, 3177–3178. [Google Scholar] [CrossRef]
- Saito, K.; Asada, Y. Superconductivity and structural changes of nitrogen-ion implanted Mo thin films. J. Phys. F Met. Phys. 1987, 17, 2273–2283. [Google Scholar] [CrossRef]
- Wu, J.D.; Wu, C.Z.; Zhong, X.X.; Song, Z.M.; Li, F.M. Surface nitridation of transition metals by pulsed laser irradiation in gaseous nitrogen. Surf. Coat. Technol. 1997, 96, 330–336. [Google Scholar] [CrossRef]
- Mändl, S.; Manova, D.; Gerlach, J.W.; Assmann, W.; Neumann, H.; Rauschenbach, B. High temperature nitrogen plasma immersion ion implantation into molybdenum. Surf. Coat. Technol. 2004, 180–181, 362–366. [Google Scholar] [CrossRef]
- Anitha, V.P.; Major, S.; Chandrashekharam, D.; Bhatnagar, M. Deposition of molybdenum nitride films by r.f. reactive magnetron sputtering. Surf. Coat. Technol. 1996, 79, 50–54. [Google Scholar] [CrossRef]
- Hones, P.; Martin, N.; Regula, M.; Lévy, F. Structural and mechanical properties of chromium nitride, molybdenum nitride, and tungsten nitride thin films. J. Phys. D Appl. Phys. 2003, 36, 1023–1029. [Google Scholar] [CrossRef]
- Wang, Y.; Lin, R.Y. Amorphous molybdenum nitride thin films prepared by reactive sputter deposition. Mater. Sci. Eng. B 2004, 112, 42–49. [Google Scholar] [CrossRef]
- Stöber, L.; Konrath, J.P.; Krivec, S.; Patocka, F.; Schwartz, S.; Bittner, A.; Schneider, M.; Schmid, U. Impact of sputter deposition parameters on molybdenum nitride thin films properties. J. Micromech. Microeng. 2015, 25. [Google Scholar] [CrossRef]
- Inumaru, K.; Baba, K.; Yamanaka, S. Synthesis and Characterization of superconducting β-MoN crystalline phase on a Si substrate: An application of pulsed laser deposition to nitride chemistry. Chem. Mater. 2005, 17, 5935–5940. [Google Scholar] [CrossRef]
- Jauberteau, I.; Jauberteau, J.L.; Goudeau, P.; Soulestin, B.; Marteau, M.; Cahoreau, M.; Aubreton, J. Investigations on a nitriding process of molybdenum thin films exposed to (Ar–N2–H2) expanding microwave plasma. Surf. Coat. Technol. 2009, 203, 1127–1132. [Google Scholar] [CrossRef]
- Lowther, J.E. Lattice model for the properties of non-stoichiometric cubic and hexagonal molybdenum nitride. J. Alloys Compd. 2004, 364, 13–16. [Google Scholar] [CrossRef]
- Ganin, A.Y.; Kienle, L.; Vajenine, G.V. Synthesis and characterization of hexagonal molybdenum nitrides. J. Solid State Chem. 2006, 179, 2339–2348. [Google Scholar] [CrossRef]
- Bull, C.L.; McMillan, P.F.; Soignard, E.; Leinenweber, K. Determination of the crystal structure of δ-MoN by neutron diffraction. J. Solid State Chem. 2004, 177, 1488–1492. [Google Scholar] [CrossRef]
- Kanoun, M.B.; Goumri-Said, S.; Jaouen, M. Structure and mechanical stability of molybdenum nitrides: A first-principles study. Phys. Rev. B 2007, 76. [Google Scholar] [CrossRef]
- Zhao, E.; Wang, J.; Wu, Z. Displacive phase transition, structural stability, and mechanical properties of the ultra-incompressible and hard MoN by first principles. Phys. Status Solidi B 2010, 247, 1207–1213. [Google Scholar] [CrossRef]
- Linker, G.; Smithey, R.; Meyer, O. Superconductivity in MoN films with NaCl structure. J. Phys. F Met. Phys. 1984, 14, L115–L119. [Google Scholar] [CrossRef]
- Savvides, N. High Tc superconducting B1 phase MoN films prepared by low-energy ion-assisted deposition. J. Appl. Phys. 1987, 62, 600–610. [Google Scholar] [CrossRef]
- Papaconstantopoulos, D.A.; Pickett, W.E.; Klein, B.M.; Boyer, L.L. Electronic properties of transition-metal nitrides: The group-V and group-VI nitrides VN, NbN, TaN, CrN, MoN, and WN. Phys. Rev. B 1985, 31, 752–761. [Google Scholar] [CrossRef]
- Fix, R.; Gordon, R.G.; Hoffman, D.M. Low-temperature atmospheric-pressure metal-organic chemical vapor deposition of molybdenum nitride thin films. Thin Solid Films 1996, 288, 116–119. [Google Scholar] [CrossRef]
- Maoujoud, M.; Jardinier-Offergeld, M.; Bouillon, F. Synthesis and characterization of thin-film molybdenum nitrides. Appl. Surf. Sci. 1993, 64, 81–89. [Google Scholar] [CrossRef]
- Lyutaya, M.D. Formation of nitrides of the group VI transition metals. Powder Metall. Met. Ceram. 1979, 18, 190–196. [Google Scholar] [CrossRef]
- McKay, D.; Hargreaves, J.S.J.; Rico, J.L.; Rivera, J.L.; Sun, X.-L. The influence of phase and morphology of molybdenum nitrides on ammonia synthesis activity and reduction characteristics. J. Solid State Chem. 2008, 181, 325–333. [Google Scholar] [CrossRef]
- Birtill, J.; Dickens, P.G. Thermochemistry of Hydrogen Molybdenum Bronze Phases HxMoO3. J. Solid State Chem. 1979, 29, 367–372. [Google Scholar] [CrossRef]
- Cardenas-Lizana, F.; Gomez-Quero, S.; Perret, N.; Kiwi-Minsker, L.; Keane, M.A. β-Molybdenum nitride: Synthesis mechanism and catalytic response in the gas phase hydrogenation of p-chloronitrobenzene. Catal. Sci. Technol. 2011, 1, 794–801. [Google Scholar] [CrossRef]
- Thornton, J.A. High rate thick film growth. Annu. Rev. Mater. Sci. 1977, 7, 239–260. [Google Scholar] [CrossRef]
- Perry, A.J.; Baouchi, A.W.; Petersen, J.H.; Pozder, S.D. Crystal structure of molybdenum nitride films made by reactive cathodic arc evaporation. Surf. Coat. Technol. 1992, 54/55, 261–265. [Google Scholar] [CrossRef]
- Mändl, S.; Gerlach, J.W.; Assmann, W.; Rauschenbach, B. Phase formation and diffusion after nitrogen PIII in molybdenum. Surf. Coat. Technol. 2003, 174–175, 1238–1242. [Google Scholar] [CrossRef]
- Mändl, S.; Gerlach, W.; Rauschenbach, B. Nitride formation in transition metals during high fluence-high temperature implantation. Surf. Coat. Technol. 2005, 200, 584–588. [Google Scholar] [CrossRef]
- Jauberteau, I.; Merle-Méjean, T.; Touimi, S.; Weber, S.; Bessaudou, A.; Passelergue, A.; Jauberteau, J.L.; Aubreton, J. Expanding microwave plasma process for thin molybdenum films nitriding: Nitrogen diffusion and structure investigations. Surf. Coat. Technol. 2011, 205, S271–S274. [Google Scholar] [CrossRef]
- Jauberteau, I.; Jauberteau, J.L.; Touimi, S.; Merle-Méjean, T.; Weber, S.; Bessaudou, A. A thermochemical process using expanding plasma for nitriding thin molybdenum films at low temperature. Engineering 2012, 4, 857–868. [Google Scholar] [CrossRef]
- Jauberteau, I.; Mayet, R.; Cornette, J.; Bessaudou, A.; Carles, P.; Jauberteau, J.L.; Merle-Méjean, T. A reduction-nitridation process of molybdenum films in expanding microwave plasma: Crystal structure of molybdenum nitrides. Surf. Coat. Technol. 2015, 270, 77–85. [Google Scholar] [CrossRef]
- Luo, H.; Zou, G.; Wang, H.; Lee, J.H.; Lin, Y.; Peng, H.; Lin, Q.; Deng, S.; Bauer, E.; McKleskey, T.M.; et al. Controlling crystal structure and oxidation state in molybdenum nitrides through epitaxial stabilization. J. Phys. Chem. C 2011, 115, 17880–17883. [Google Scholar] [CrossRef]
- Nagae, M.; Yoshio, T.; Takemoto, Y.; Takada, J. Microstructure of a molybdenum nitride layer formed by nitriding molybdenum metal. J. Am. Ceram. Soc. 2001, 84, 1175–1177. [Google Scholar] [CrossRef]
- Soignard, E.; McMillan, P.F.; Chaplin, T.D.; Farag, S.M.; Bull, C.L.; Somayazulu, M.S.; Leinenweber, K. High-Pressure synthesis and study of low-compressibility molybdenum nitride (MoN and MoN1 − x phases). Phys. Rev. B 2003, 68. [Google Scholar] [CrossRef]
- Sahu, B.R.; Kleinman, L. Theoretical study of structural an electronic properties of δ-MoN. Phys. Rev. B 2004, 70. [Google Scholar] [CrossRef]
- Hart, G.L.W.; Klein, B.M. Phonon and elastic instabilities in MoC and MoN. Phys. Rev. B 2000, 61, 3151–3154. [Google Scholar] [CrossRef]
- Choi, D.; Kumta, P.N. Synthesis and characterization of nanostructured niobium and molybdenum nitrides by a two steps transition metal halide approach. J. Am. Ceram. Soc. 2011, 94, 2371–2378. [Google Scholar] [CrossRef]
- Ma, L.; Ting, L.R.L.; Molinari, V.; Giordano, C.; Yeo, B.S. Efficient hydrogen evolution catalyzed by molybdenum carbide and molybdenum nitride nanocatalysts synthesized via the urea glass route. J. Mater. Chem. A 2015, 3, 8361–8368. [Google Scholar] [CrossRef]
- Chen, W.F.; Muckerman, J.T.; Fujita, E. Recent developments in transition metal carbides and nitrides as hydrogen evolution eletrocatalysts. Chem. Commun. 2013, 49, 8896–8909. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jauberteau, I.; Bessaudou, A.; Mayet, R.; Cornette, J.; Jauberteau, J.L.; Carles, P.; Merle-Méjean, T. Molybdenum Nitride Films: Crystal Structures, Synthesis, Mechanical, Electrical and Some Other Properties. Coatings 2015, 5, 656-687. https://doi.org/10.3390/coatings5040656
Jauberteau I, Bessaudou A, Mayet R, Cornette J, Jauberteau JL, Carles P, Merle-Méjean T. Molybdenum Nitride Films: Crystal Structures, Synthesis, Mechanical, Electrical and Some Other Properties. Coatings. 2015; 5(4):656-687. https://doi.org/10.3390/coatings5040656
Chicago/Turabian StyleJauberteau, Isabelle, Annie Bessaudou, Richard Mayet, Julie Cornette, Jean Louis Jauberteau, Pierre Carles, and Thérèse Merle-Méjean. 2015. "Molybdenum Nitride Films: Crystal Structures, Synthesis, Mechanical, Electrical and Some Other Properties" Coatings 5, no. 4: 656-687. https://doi.org/10.3390/coatings5040656
APA StyleJauberteau, I., Bessaudou, A., Mayet, R., Cornette, J., Jauberteau, J. L., Carles, P., & Merle-Méjean, T. (2015). Molybdenum Nitride Films: Crystal Structures, Synthesis, Mechanical, Electrical and Some Other Properties. Coatings, 5(4), 656-687. https://doi.org/10.3390/coatings5040656





