Tailoring Mechanical and Surface Properties of Epoxy Coatings with Synthesized Pigment Particles: Microhardness, Adhesion, and Wettability Studies
Highlights
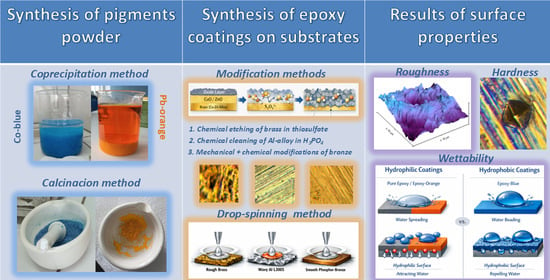
- Cobalt blue (CoAl2O4) and chrome orange (PbCrO4·Pb(OH)2) pigments were synthesized using the coprecipitation method and heat drying.
- Epoxy-based resin was deposited onto three modified substrates (brass B36, aluminum L3005, and phosphor-bronze 510).
- The 3.0 wt.% of pigment powder was mixed with epoxy resin and deposited onto substrates in the form of thin coatings using the drop-spinning technique.
- Chrome orange pigment exhibits a hollow micro-tube, while cobalt blue pigment displays an irregular morphology.
- All pigmented coatings show higher composite hardness and better adhesion compared to a pure epoxy matrix.
- All epoxy and epoxy/orange coatings showed a hydrophilic character, and epoxy/blue coatings showed a hydrophobic character.
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Synthesis of Pigment-Based Particles
2.2.1. Synthesis of Cobalt Blue (Co-Blue) Powder (Cobalt-(II)-Oxide-Aluminum-Oxide, CoO Al2O3)
2.2.2. Synthesis of Chrome Orange (Pb-Orange) Powder (Lead (II)-Chromate, PbCrO4 Pb(OH)2)
2.3. Substrate Preparation
2.4. Preparation of Composite Coatings
2.5. Characterizations of the Materials
3. Results and Discussion
3.1. Characterization of Pigments
3.1.1. Optical Analysis
3.1.2. FE-SEM and Image Analysis
3.1.3. XRD Analysis of Synthesized Pigment Powders
3.2. Characterization of Substrates
3.3. Characterization of the Epoxy-Based Coatings
3.3.1. Cross-Section Analysis and Measurement of Coating Thickness
3.3.2. FTIR Measurement of Epoxy-Based Coatings
3.3.3. AFM Measurement of Epoxy-Based Coatings
3.3.4. Microhardness Measurement of Epoxy-Based Coatings
Composite Hardness Measurement
Calculation of Intrinsic Hardness
The Adhesion Properties of Epoxy-Based Coatings
3.4. Wettability Properties of Epoxy-Based Coatings
4. Conclusions
- Microparticles of Co-blue pigment showed granular size in the range of 0.1–5 µm, with nano-scale crystallites decorating their surfaces. This hierarchical structure enhances color intensity, stability, and dispersibility in the epoxy coating. The Pb-orange pigment forms smooth rod- or needle-like forms, typically 100–500 nm in diameter and 1–5 µm long, often hollow at the ends. Both synthesized pigments are highly crystalline, which was confirmed by XRD analysis.
- The hardest substrate is brass (1.5505 GPa), and the softest is the AL-alloy (283.9 MPa), while the roughness parameter is smallest for the phosphor bronze substrate (69.1 nm).
- The maximum composite hardness of the epoxy coating was achieved on chemically etched brass foil (242.56 MPa), while the minimum hardness was shown by the pure epoxy coating on the aluminum alloy (32.165 MPa). All coatings with epoxy/Pb-orange pigment are harder than those with Co-blue pigment, while the pure epoxy matrix shows the lowest hardness on all substrates.
- The modified brass substrate shows the optimal substrate for the deposition of epoxy-based coatings. The addition of Pb-orange pigment increases the adhesion strength of epoxy on all substrates: 44.5% on brass, 92.8% on Al L3005, and 73.3% on bronze. The epoxy coatings with Co-blue pigment show the following improvements of adhesion parameter b over the pure epoxy matrix: 25.5% on brass, 14.1% on AL L3005, and 47.1% on bronze.
- The intrinsic hardness of epoxy-based coatings does not change significantly with the addition of pigment particles, indicating that the type of substrate has a dominant effect on the composite hardness of epoxy-based coatings. Both hardness values (Hc and Hi) are sensitive to variations in coating thickness.
- The pigments significantly influence the wettability of epoxy-based coatings. While pure epoxy and epoxy/Pb-orange coatings exhibited hydrophilic behavior, promoting water affinity, the epoxy/Co-blue coatings showed hydrophobic character, resisting water interaction.
- The Co-blue pigment increases the dispersion component of epoxy’s surface energy. In contrast, Pb-orange pigment decreases it and enhances the polar component, showing that pigment type is the dominant factor in surface energy modification.
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| C-G | Chen–Gao |
| FTIR | Fourier-Transform Infrared Spectroscopy |
| FE-SEM | Field-emission scanning electron microscopy |
| XRD | X-ray diffraction |
| AFM | Atomic Force Microscopy |
References
- Jin, F.-L.; Li, X.; Park, S.-J. Synthesis and Application of Epoxy Resins: A Review. J. Ind. Eng. Chem. 2015, 29, 1–11. [Google Scholar] [CrossRef]
- Capricho, J.C.; Fox, B.; Hameed, N. Multifunctionality in Epoxy Resins. Polym. Rev. 2020, 60, 1–41. [Google Scholar] [CrossRef]
- Ogundana, T.O.; Oginni, O.T.; Onuh, F.A.; Morawo, J.; Olagonju, M.O. Characterization of Epoxy Resin Matrix Reinforced with Silica Nanoparticles for Enhanced Mechanical Performance. FUW Trends Sci. Technol. J. 2024, 9, 057–063. [Google Scholar]
- Collado, I.; Vázquez-López, A.; Jiménez-Suárez, A.; Prolongo, S.G. Multifunctional Sequential Dual Curing Electroactive Graphene Nanocomposites: Self-Heating, de-Icing and in-Situ Curing. Compos. Part B Eng. 2025, 307, 112911. [Google Scholar] [CrossRef]
- Jin, R.; Xu, B.; Guo, D.; Shi, B.; Chen, Y.; Jia, X.; Qu, L. Advanced Chemical Modification Technology of Inorganic Oxide Nanoparticles in Epoxy Resin and Mechanical Properties of Epoxy Resin Nanocomposites: A Review. Nano Mater. Sci. 2025, 7, 643–664. [Google Scholar] [CrossRef]
- Parvizi, P.; Jalilian, M.; Dearn, K.D. Epoxy Composites Reinforced with Nanomaterials and Fibres: Manufacturing, Properties, and Applications. Polym. Test. 2025, 146, 108761. [Google Scholar] [CrossRef]
- Ilangovan, S.; Kumaran, S.S.; Vasudevan, A.; Naresh, K. Effect of Silica Nanoparticles on Mechanical and Thermal Properties of Neat Epoxy and Filament Wounded E-Glass/Epoxy and Basalt/Epoxy Composite Tubes. Mater. Res. Express 2019, 6, 0850e2. [Google Scholar] [CrossRef]
- Białkowska, A.; Bakar, M.; Kucharczyk, W.; Zarzyka, I. Hybrid Epoxy Nanocomposites: Improvement in Mechanical Properties and Toughening Mechanisms—A Review. Polymers 2023, 15, 1398. [Google Scholar] [CrossRef]
- Gürses, A.; Açıkyıldız, M.; Güneş, K.; Gürses, M.S. Dyes and Pigments: Their Structure and Properties. In SpringerBriefs in Molecular Science; Springer: Berlin/Heidelberg, Germany, 2016; pp. 13–29. [Google Scholar]
- Völz, H.G.; Kischkewitz, J.; Woditsch, P.; Westerhaus, A.; Griebler, W.-D.; De Liedekerke, M.; Buxbaum, G.; Printzen, H.; Mansmann, M.; Räde, D.; et al. Pigments, Inorganic. In Ullmann’s Encyclopedia of Industrial Chemistry; Wiley-VCH Verlag GmbH & Co. KGaA: Weinheim, Germany, 2006. [Google Scholar]
- Lee, H.B.; Yoo, Y.M.; Han, Y.-H. Characteristic Optical Properties and Synthesis of Gold–Silica Core–Shell Colloids. Scr. Mater. 2006, 55, 1127–1129. [Google Scholar] [CrossRef][Green Version]
- Gholami, T.; Salavati-Niasari, M.; Varshoy, S. Investigation of the Electrochemical Hydrogen Storage and Photocatalytic Properties of CoAl2O4 Pigment: Green Synthesis and Characterization. Int. J. Hydrogen Energy 2016, 41, 9418–9426. [Google Scholar] [CrossRef]
- Enríquez, E.; Reinosa, J.J.; Fuertes, V.; Fernández, J.F. Advances and Challenges of Ceramic Pigments for Inkjet Printing. Ceram. Int. 2022, 48, 31080–31101. [Google Scholar] [CrossRef]
- Morán-Lázaro, J.P.; Blanco, O.; Rodríguez-Betancourtt, V.M.; Reyes-Gómez, J.; Michel, C.R. Enhanced CO2-Sensing Response of Nanostructured Cobalt Aluminate Synthesized Using a Microwave-Assisted Colloidal Method. Sens. Actuators B Chem. 2016, 226, 518–524. [Google Scholar] [CrossRef]
- Farrokhpay, S.; Morris, G.E.; Fornasiero, D.; Self, P. Influence of Polymer Functional Group Architecture on Titania Pigment Dispersion. Colloids Surf. A Physicochem. Eng. Asp. 2005, 253, 183–191. [Google Scholar] [CrossRef]
- Zhang, K.; Zhang, Y.; Ye, M.; Han, A.; Chen, X. Preparation and Characterization of Multi-Component Composite Microspheres Used for Ceramic Toner. Ceram. Int. 2019, 45, 24601–24608. [Google Scholar] [CrossRef]
- Nwokolo, I.K.; Shi, H.; Liu, F. MOF-Based Protective Coatings for Metal Corrosion Protection: A Critical Review of Design/Synthesis, Performance, and Mechanism. Mater. Sci. Eng. B 2025, 313, 117932. [Google Scholar] [CrossRef]
- Amune, U.O.; Solomon, M.M.; Hu, D.; He, J.; Tesema, F.B.; Umoren, S.A.; Gerengi, H. A Comprehensive Review of Stimulus-Based Smart Self-Healing Coatings for Substrate Protection. Prog. Org. Coat. 2026, 210, 109669. [Google Scholar] [CrossRef]
- Gharieh, A.; Sharifian, A.; Dadkhah, S. Enhanced Long-Term Corrosion Resistance and Self-Healing of Epoxy Coating with HQ-Zn-PA Nanocomposite. Sci. Rep. 2025, 15, 8154. [Google Scholar] [CrossRef]
- Chopra, I.; Ola, S.K.; Priyanka; Dhayal, V.; Shekhawat, D.S. Recent Advances in Epoxy Coatings for Corrosion Protection of Steel: Experimental and Modelling Approach-A Review. Mater. Today Proc. 2022, 62, 1658–1663. [Google Scholar] [CrossRef]
- Fekiač, J.J.; Krbata, M.; Kohutiar, M.; Janík, R.; Kakošová, L.; Breznická, A.; Eckert, M.; Mikuš, P. Comprehensive Review: Optimization of Epoxy Composites, Mechanical Properties, & Technological Trends. Polymers 2025, 17, 271. [Google Scholar] [CrossRef]
- Antony Jose, S.; Lapierre, Z.; Williams, T.; Hope, C.; Jardin, T.; Rodriguez, R.; Menezes, P.L. Wear- and Corrosion-Resistant Coatings for Extreme Environments: Advances, Challenges, and Future Perspectives. Coatings 2025, 15, 878. [Google Scholar] [CrossRef]
- Hare, C.H. Corrosion Control of Steel by Organic Coatings. In Uhlig’s Corrosion Handbook; Wiley: Hoboken, NJ, USA, 2011; pp. 971–983. [Google Scholar]
- Mi, X.; Liang, N.; Xu, H.; Wu, J.; Jiang, Y.; Nie, B.; Zhang, D. Toughness and Its Mechanisms in Epoxy Resins. Prog. Mater. Sci. 2022, 130, 100977. [Google Scholar] [CrossRef]
- Kalenda, P. Improved Chemical Resistance of Epoxy Resin-Based Coating Compositions. Dye. Pigment. 1995, 27, 305–312. [Google Scholar] [CrossRef]
- Ramakrishnan, T.; Raja Karthikeyan, K.; Tamilselvan, V.; Sivakumar, S.; Gangodkar, D.; Radha, H.R.; Narain Singh, A.; Asrat Waji, Y. Study of Various Epoxy-Based Surface Coating Techniques for Anticorrosion Properties. Adv. Mater. Sci. Eng. 2022, 2022, 5285919. [Google Scholar] [CrossRef]
- Tomić, N.Z.; Saleh, M.N.; Vuksanović, M.M.; Egelja, A.; Obradović, V.; Marinković, A.; Jančić Heinemann, R. Tailored Adhesion Properties of Acrylate Adhesives on Al Alloys by the Addition of Mn-Al–LDH. Polymers 2021, 13, 1525. [Google Scholar] [CrossRef]
- Algellai, A.A.; Tomić, N.; Vuksanović, M.M.; Dojčinović, M.; Volkov-Husović, T.; Radojević, V.; Heinemann, R.J. Adhesion Testing of Composites Based on Bis-GMA/TEGDMA Monomers Reinforced with Alumina Based Fillers on Brass Substrate. Compos. Part B Eng. 2018, 140, 164–173. [Google Scholar] [CrossRef]
- Altwair, K.; Vuksanović, M.M.; Mladenović, I.O.; Heinemann, R.J. Development of PMMA–Silica–Alumina Nanocomposites for Enhanced Performance. Polym. Polym. Compos. 2026, 34, 09673911251414228. [Google Scholar] [CrossRef]
- Wang, K.; Li, J.; Yi, Y.; Lv, B.; Wu, Y.; Wang, C.; Li, H.; Li, Y.; Liu, Y.; Cai, X.; et al. Polyamine-Activated Carbonyl Stress Strategy for Oxidative Damage Therapy. Nano Today 2022, 42, 101355. [Google Scholar] [CrossRef]
- Wang, H.; Yan, Y.; Tian, L.; Li, X.; Yang, Y.; Niu, L.; Li, X.; Zhang, Z. High Content of Spherical Nanosilica Filled Epoxy Resin Master Batch with Low Viscosity and Superior Thermomechanical Performance. Compos. Commun. 2022, 36, 101355. [Google Scholar] [CrossRef]
- Available online: https://www.webexhibits.org/pigments/indiv/overview/crorange.html (accessed on 7 May 2026).
- Available online: http://www.seilnacht.tuttlingen.com (accessed on 7 May 2026).
- Available online: https://www.webexhibits.org/pigments/indiv/recipe/crorange.html (accessed on 7 May 2026).
- Habashi, F. Pigments through the Ages. Interceram—Int. Ceram. Rev. 2016, 65, 156–165. [Google Scholar] [CrossRef]
- Robert, L. Feller Artists’ Pigments: A Handbook of Their History and Characteristics; Cambridge University Press: London, UK, 1986; ISBN 9780521303743. [Google Scholar]
- Available online: https://www.metallography.com (accessed on 7 May 2026).
- Luo, J.; Liu, J.; Xia, H.; Ao, X.; Yin, H.; Guo, L. Surface Treatments for Enhancing the Bonding Strength of Aluminum Alloy Joints. Materials 2023, 16, 5674. [Google Scholar] [CrossRef]
- Lee, S.; Cho, Y.J. Phosphoric Acid Anodizing Effect on Morphology and Corrosion Resistance of Nanostructured Anodic Oxide Layers on 6061 Aluminum Alloy. Met. Mater. Int. 2025, 31, 2598–2608. [Google Scholar] [CrossRef]
- Chen, Z.; Yang, Y.; Lou, H.; Wang, H.; Xiang, C. Effects of the Grain Size, Boundary Type and Texture on the Bending Properties of Phosphorus Bronze Strips. Mater. Charact. 2025, 225, 115090. [Google Scholar] [CrossRef]
- Broitman, E. Indentation Hardness Measurements at Macro-, Micro-, and Nanoscale: A Critical Overview. Tribol. Lett. 2017, 65, 23. [Google Scholar] [CrossRef]
- Petrík, J.; Blaško, P.; Markulík, Š.; Šolc, M.; Palfy, P. The Indentation Size Effect (ISE) of Metals. Crystals 2022, 12, 795. [Google Scholar] [CrossRef]
- Magagnin, L.; Maboudian, R.; Carraro, C. Adhesion Evaluation of Immersion Plating Copper Films on Silicon by Microindentation Measurements. Thin Solid Film. 2003, 434, 100–105. [Google Scholar] [CrossRef]
- Magagnin, L.; Cojocaru, P.; Raygani, A.; Brivio, D.; Secundo, F.; Turolla, A.; Ottolina, G. Galvanic Displacement of Nanostructured Gold for Flavoenzyme Adsorption in Biotechnology. ECS Trans. 2011, 33, 59–66. [Google Scholar] [CrossRef]
- Ou, J.F.; Amirfazli, A.; Chini, S.F. The Sessile Drop Work of Adhesion Revisited. Int. J. Adhes. Adhes. 2025, 136, 103872. [Google Scholar] [CrossRef]
- Zukiene, K.; Monastyreckis, G.; Kilikevicius, S.; Procházka, M.; Micusik, M.; Omastová, M.; Aniskevich, A.; Zeleniakiene, D. Wettability of MXene and Its Interfacial Adhesion with Epoxy Resin. Mater. Chem. Phys. 2021, 257, 123820. [Google Scholar] [CrossRef]
- Royaux, A.; El Haitami, A.; Fichet, O.; Cantin, S. Surface Free-Energy Determination of Copper Wire Using a Large Range of Model Liquids. SN Appl. Sci. 2020, 2, 48. [Google Scholar] [CrossRef]
- Sagar, R.; Gaur, M.S.; Kushwah, V.; Rathore, A.; Piliptsou, D.G.; Rogachev, A.A. Preparation, Characterization and Microhardness Measurements of Hybrid Nanocomposites Based on PMMA + P(VDF–TrFE) and Graphene Oxide. Polym. Bull. 2021, 78, 7279–7300. [Google Scholar] [CrossRef]
- Embirsh, H.S.A.; Mladenović, I.O.; Radojević, V.; Marinković, A.; Vuksanović, M.M. Evaluation of Surface Properties in Biosilica-Reinforced Biobased Polyester Nanocomposites. Appl. Sci. 2025, 15, 9244. [Google Scholar] [CrossRef]
- Huhtamäki, T.; Tian, X.; Korhonen, J.T.; Ras, R.H.A. Surface-Wetting Characterization Using Contact-Angle Measurements. Nat. Protoc. 2018, 13, 1521–1538. [Google Scholar] [CrossRef]
- Mohamed, S.S.M.; Vuksanović, M.M.; Vasiljević-Radović, D.G.; Janković Mandić, L.; Jančić Heinneman, R.M.; Marinković, A.D.; Mladenović, I.O. Sol–Gel Derived Alumina Particles for the Reinforcement of Copper Films on Brass Substrates. Gels 2024, 10, 648. [Google Scholar] [CrossRef]
- Jasper, S.; Subash, R.; Muthuneelakandan, K.; Vijayakumar, D.; Jhansi Ida, S. The Mechanical Properties of Brass Alloys: A Review. Eng. Proc. 2025, 93, 11. [Google Scholar]
- Chang, S.C.; Jahn, M.T.; Wan, C.M.; Lee, J.Y.M.; Hsu, T.K. The Determination of Tensile Properties from Hardness Measurements for Al-Zn-Mg Alloys. J. Mater. Sci. 1976, 11, 623–630. [Google Scholar] [CrossRef]
- Yang, K.; Fu, Z.H.; Chen, H.; Wang, X.; Liu, B. Micro-Indentation Analysis of Aluminium Alloys: Simultaneous Evaluation of Tensile Properties and Hardness Characterisation via Knoop Indentation. Nondestruct. Test. Eval. 2026, 1–21. [Google Scholar] [CrossRef]
- Mladenović, I.O.; Lamovec, J.S.; Vasiljević Radović, D.G.; Vasilić, R.; Radojević, V.J.; Nikolić, N.D. Morphology, Structure and Mechanical Properties of Copper Coatings Electrodeposited by Pulsating Current (PC) Regime on Si(111). Metals 2020, 10, 488. [Google Scholar] [CrossRef]
- Chen, M.; Gao, J. The Adhesion of Copper Films Coated on Silicon and Glass Substrates. Mod. Phys. Lett. B 2000, 14, 103–108. [Google Scholar] [CrossRef]
- Hou, Q.R.; Gao, J.; Li, S.J. Adhesion and Its Influence on Micro-Hardness of DLC and SiC Films. Eur. Phys. J. B 1999, 8, 493–496. [Google Scholar] [CrossRef]
- He, J.L. Hardness Measurement of Thin Films: Separation from Composite Hardness. Appl. Phys. Lett. 1996, 25, 2002. [Google Scholar] [CrossRef]
- Rajabi, M.; Kharaziyan, P.; Montazeri-Pour, M. Microwave-Assisted Processing of Cobalt Aluminate Blue Nano-Ceramic Pigment Using Sol–Gel Method. J. Aust. Ceram. Soc. 2019, 55, 219–227. [Google Scholar] [CrossRef]
- Horsth, D.F.L.; de O. Primo, J.; Anaissi, F.J.; Umek, P.; Bittencourt, C. Sustainable Near-Infrared Reflective Blue Pigments: Recycled Aluminum from Can Seals for Cobalt Aluminates in Cool Coatings. Colorants 2024, 3, 253–262. [Google Scholar] [CrossRef]
- Barbar, S.K. Structural, Optical and Luminescence Characteristics of a Novel Blue Pigment Ba2CaCo2Si6O17. J. Coat. Technol. Res. 2025, 23, 993–1000. [Google Scholar] [CrossRef]
- Zhang, W.; Li, Z.; Wu, G.; Wu, W.; Zeng, H.; Jiang, H.; Zhang, W.; Wu, R.; Xue, Q. Effects of Coloration of Spinel CoAl2O4 Cobalt Blue Pigments: Composition, Structure, and Cation Distribution. Inorganics 2023, 11, 368. [Google Scholar] [CrossRef]
- Ghazitabar, A.; Ataeefard, M.; Moradi, Z.; Naderi, M. Influence of the Chemistry and Morphology of Recycled Iron Oxide Pigments on the Colorimetric Properties of Flexographic Ink. J. Mater. Cycles Waste Manag. 2025, 27, 4641–4654. [Google Scholar] [CrossRef]
- Liu, M.; Jia, Z.; Li, P.; Liu, Y.; Zhao, M.; Yang, Y.; Huang, Q.; Yu, C. High Catalytic Activity of Fe3−xCuxO4/Graphene Oxide (0 ≤ x ≤ 0.1) Nanocomposites as Heterogeneous Fenton Catalysts for p-Nitrophenol Degradation. Water Air Soil Pollut. 2019, 230, 64. [Google Scholar] [CrossRef]
- Yap, F.Y.; Berkes, J.L.; Knuttinen, M.G.; Walzer, N.M.; Cotler, S.J.; Owens, C.A.; Bui, J.T.; Gaba, R.C. Quantitative Morphometric Analysis of Hepatocellular Carcinoma: Programmed Algorithm Development and Preliminary Application. Diagn. Interv. Radiol. 2012, 19, 97–105. [Google Scholar] [CrossRef]
- Kudekar, V.P.; Rane, B.R.; Jain, A.S.; Kothawade, S.N. Characterization Techniques for Polymer Nanocomposites. In Polymer Nanocomposites; Springer Nature: Cham, Switzerland, 2026; pp. 139–159. [Google Scholar]
- Allen, T. Particle Size Measurement: Volume 1–Powder Sampling and Particle Size Measurement; Springer: Dordrecht, The Netherlands, 1997. [Google Scholar] [CrossRef]
- ISO 9276-1:1998; Representation of Results of Particle Size Analysis—Part 1: Graphical Representation. International Organization for Standardization: Geneva, Switzerland, 1998.
- Merkus, H.G. Particle Size Measurements; Particle Technology Series; Springer: Dordrecht, The Netherlands, 2009; Volume 17, ISBN 978-1-4020-9015-8. [Google Scholar]
- Mrkwitschka, P.; Abram, S.-L.; Rühle, B.; Hodoroaba, V.-D. Understanding Correlative Electron Microscopy Imaging with SEM, STEM-in-SEM and TEM for the Accurate Characterization of Size and Shape of Iron Oxide Nanoparticles. Microsc. Microanal. 2024, 30, ozae044.339. [Google Scholar] [CrossRef]
- Pfaff, G. Iron Oxide Pigments. Phys. Sci. Rev. 2021, 6, 535–548. [Google Scholar] [CrossRef]
- Zhang, W.; Li, J.; Zhong, F.; Wu, G.; Jiang, H.; Zhang, W.; Liu, Q. Preparation and Characterization of Supercritical Fluid—Fried (CoAl2O4) Cobalt Blue Nano-Pigment. J. Asian Ceram. Soc. 2022, 10, 33–39. [Google Scholar] [CrossRef]
- Zavalij, P.Y.; Pecharsky, V.K. Fundamentals of Powder Diffraction and Structural Characterization of Materials; Springer Nature: Cham, Switzerland, 2025; ISBN 978-3-031-91503-1. [Google Scholar]
- Zhiani, R.; Es-haghi, A.; Sadeghzadeh, S.M.; Shamsa, F. Green Synthesis of PbCrO 4 Nanostructures Using Gum of Ferula Assa-Foetida for Enhancement of Visible-Light Photocatalytic Activity. RSC Adv. 2018, 8, 40934–40940. [Google Scholar] [CrossRef]
- Kaiser, M.; Wosylus, A.; Gerke, B.; Pöttgen, R.; Prokeš, K.; Ruck, M.; Doert, T. Crystal Growth and Structure Determination of Pigment Orange 82. Z. Anorg. Allg. Chem. 2019, 645, 564–569. [Google Scholar] [CrossRef]
- Mahmoudian, M.; Goharpey, F.; Behzadnasab, M.; Daneshfar, Z. Shear Thickening and Hysteresis in Dense Suspensions: The Effect of Particle Shape. J. Rheol. 2024, 68, 479–490. [Google Scholar] [CrossRef]
- Nikolic, G.; Zlatkovic, S.; Cakic, M.; Cakic, S.; Lacnjevac, C.; Rajic, Z. Fast Fourier Transform IR Characterization of Epoxy GY Systems Crosslinked with Aliphatic and Cycloaliphatic EH Polyamine Adducts. Sensors 2010, 10, 684–696. [Google Scholar] [CrossRef]
- Zhang, W.; Fina, A.; Ferraro, G.; Yang, R. FTIR and GCMS Analysis of Epoxy Resin Decomposition Products Feeding the Flame during UL 94 Standard Flammability Test. Application to the Understanding of the Blowing-out Effect in Epoxy/Polyhedral Silsesquioxane Formulations. J. Anal. Appl. Pyrolysis 2018, 135, 271–280. [Google Scholar] [CrossRef]
- Zhang, A.; Mu, B.; Wang, X.; Wang, A. CoAl2O4/Kaoline Hybrid Pigment Prepared via Solid-Phase Method for Anticorrosion Application. Front. Chem. 2018, 6, 586. [Google Scholar] [CrossRef]
- Ngasoh, O.F.; Anye, V.C.; Ojo, E.B.; Stanislas, T.T.; Bello, A.; Agyei-Tuffour, B.; Orisekeh, K.; Oyewole, O.K.; Rahbar, N.; Soboyejo, W.O. Mechanical Properties of Epoxy/Clay Composite Coatings on an X65 Steel Substrate. Cogent Eng. 2021, 8, 1944015. [Google Scholar] [CrossRef]
- Collado, I.; Vázquez-López, A.; Jiménez-Suárez, A.; Prolongo, S.G. Dual-Curing MWCNT Nanocomposites for Energy-Efficient Electroactive Shape Memory and In-Situ Processing. Chem. Eng. J. 2025, 515, 163471. [Google Scholar] [CrossRef]
- Bratasyuk, N.A.; Latyshev, A.V.; Zuev, V.V. Water in Epoxy Coatings: Basic Principles of Interaction with Polymer Matrix and the Influence on Coating Life Cycle. Coatings 2023, 14, 54. [Google Scholar] [CrossRef]
- Gong, J.; Wei, H.; Hao, P.; Li, S.; Zhao, X.; Tang, Y.; Zuo, Y. Study on the Influence of Metal Substrates on Protective Performance of the Coating by EIS. Materials 2024, 17, 378. [Google Scholar] [CrossRef]
- van den Brand, J.; Van Gils, S.; Beentjes, P.C.J.; Terryn, H.; Sivel, V.; de Wit, J.H.W. Improving the Adhesion between Epoxy Coatings and Aluminium Substrates. Prog. Org. Coat. 2004, 51, 339–350. [Google Scholar] [CrossRef]
- Rodič, P.; Kapun, B.; Milošev, I. Superhydrophobic Aluminium Surface to Enhance Corrosion Resistance and Obtain Self-Cleaning and Anti-Icing Ability. Molecules 2022, 27, 1099. [Google Scholar] [CrossRef]
- Wiame, F.; Salgin, B.; Swiatowska-Mrowiecka, J.; Maurice, V.; Marcus, P. Brass Surface Nanochemistry: The Role of Alloying Cu with Zn. J. Phys. Chem. C 2008, 112, 7540–7543. [Google Scholar] [CrossRef]
- Giuliani, C.; Pascucci, M.; Riccucci, C.; Messina, E.; Salzano de Luna, M.; Lavorgna, M.; Ingo, G.M.; Di Carlo, G. Chitosan-Based Coatings for Corrosion Protection of Copper-Based Alloys: A Promising More Sustainable Approach for Cultural Heritage Applications. Prog. Org. Coat. 2018, 122, 138–146. [Google Scholar] [CrossRef]
- Huang, F.; Wei, Q.; Wang, X.; Xu, W. Dynamic Contact Angles and Morphology of PP Fibres Treated with Plasma. Polym. Test. 2006, 25, 22–27. [Google Scholar] [CrossRef]
- Kim, J.-H.; Shin, P.-S.; Kwon, D.-J. Evaluation of Static and Dynamic Contact Angle Methods for Their Correlation with Interfacial Properties in Composites. Adv. Ind. Eng. Chem. 2025, 1, 16. [Google Scholar] [CrossRef]
- Hanh, V.T.H.; Truong, M.X.; Ha, C.V.; Thuy, N.T.M.; Do, T.C.; Ha, D.T.; Thuy, T.G.T.; Hien, N.T.T.; Nguyen, T.-B. Evaluating the Morphology Contribution and Modeling of Solidification Time for Icephobic Surface Design. Europhys. Lett. 2026, 153, 16001. [Google Scholar] [CrossRef]
- Takamura, K.; Fischer, H.; Morrow, N.R. Physical Properties of Aqueous Glycerol Solutions. J. Pet. Sci. Eng. 2012, 98–99, 50–60. [Google Scholar] [CrossRef]
- Golewski, P.; Budka, M. Manufacturing Technology and Mechanical Properties of Novel Pre-Impregnated Coatings as Applied to FRP “Sandwich” Composites. Materials 2025, 18, 4725. [Google Scholar] [CrossRef]
- Ulaeto, S.B.; Ravi, R.P.; Udoh, I.I.; Mathew, G.M.; Rajan, T.P.D. Polymer-Based Coating for Steel Protection, Highlighting Metal–Organic Framework as Functional Actives: A Review. Corros. Mater. Degrad. 2023, 4, 284–316. [Google Scholar] [CrossRef]



















| H1 | |||
| Substrate → | Brass B36 | Bronze 510 | Al L3005 |
| Coatings | |||
| Epoxy | 161.8 ± 11.5 | 73.07 ± 5.1 | 32.16 ± 0.5 |
| Epoxy/Co-blue | 187.2 ± 16.4 | 120.7 ± 12.0 | 38.59 ± 0.8 |
| Epoxy/Pb-orange | 242.6 ± 20.2 | 139.3 ± 17.0 | 80.93 ± 1.0 |
| HV20.05 | |||
| Substrate → | Brass B36 | Bronze 510 | Al L3005 |
| Coatings | |||
| Epoxy | 16.5 ± 1.2 | 7.45 ± 0.5 | 3.27 ± 0.1 |
| Epoxy/Co-blue | 19.09 ± 1.7 | 12.31 ± 1.2 | 3.94 ± 0.1 |
| Epoxy/Pb-orange | 24.74 ± 2.1 | 14.20 ± 1.7 | 8.25 ± 0.1 |
| Substrate | Epoxy Coatings | A | B | C | MS | p-Value | Hi/GPa |
|---|---|---|---|---|---|---|---|
| Brass B36 | Epoxy | 0.338 ± 0.05 | −2.171 ± 0.642 | 119.9 ± 54.6 | 0.0073 | 0.017 | 0.423 |
| Brass B36 | Epoxy/Co-blue | 0.362 ± 0.05 | −1.744 ± 0.677 | 65.83 ± 36.1 | 0.0071 | 0.052 | 0.446 |
| Brass B36 | Epoxy/Pb-orange | 0.364 ± 0.07 | −2.627 ± 0.742 | 63.25 ± 23.76 | 0.0294 | 0.001 | 0.527 |
| Al L3005 | Epoxy | 0.076 ± 0.02 | −2.552 ± 0.359 | 1220 ± 218 | 0.0012 | 0.001 | 0.106 |
| Al L3005 | Epoxy/Co-blue | 0.089 ± 0.02 | −0.880 ± 0.425 | 222.4 ± 119 | 0.0004 | 0.009 | 0.115 |
| Al L3005 | Epoxy/Pb-orange | 0.098 ± 0.01 | −0.071 ± 0.092 | 29.04 ± 9.45 | 0.0005 | 0.002 | 0.117 |
| Bronze 510 | Epoxy | 0.107 ± 0.03 | 2.552 ± 0.600 | 480.5 ± 249 | 0.0041 | 0.001 | 0.157 |
| Bronze 510 | Epoxy/Co-blue | 0.157 ± 0.04 | 0.577 ± 0.715 | −471.0 ± 205 | 0.0038 | 0.012 | 0.162 |
| Bronze 510 | Epoxy/Pb-orange | 0.162 ± 0.04 | 0.972 ± 0.651 | −451.9 ± 153 | 0.0043 | 0.011 | 0.174 |
| Substrate | Epoxy Coatings | k | t/μm | b |
|---|---|---|---|---|
| Brass B36 | Epoxy | 0.1908 | 12 ± 0.8 | 64.35 |
| Brass B36 | Epoxy/Co-blue | 0.1392 | 15 ± 0.9 | 86.40 |
| Brass B36 | Epoxy/Pb-orange | 0.0961 | 20 ± 1.2 | 115.9 |
| Al L3005 | Epoxy | 1.0599 | 18 ± 1.1 | 1.83 |
| Al L3005 | Epoxy/Co-blue | 0.8630 | 20 ± 1.3 | 2.13 |
| Al L3005 | Epoxy/Pb-orange | 1.0599 | 25 ± 1.9 | 25.54 |
| Bronze 510 | Epoxy | 1.4551 | 3 ± 0.3 | 5.76 |
| Bronze 510 | Epoxy/Co-blue | 0.7435 | 9 ± 0.6 | 10.89 |
| Bronze 510 | Epoxy/Pb-orange | 0.0801 | 15 ± 0.8 | 21.57 |
| Substrate | Coating Type | WAwater (mN/m2) | WAglycerin (mN/m2) |
|---|---|---|---|
| Al-L3005 | Epoxy/Co-blue | 58.02 | 83.38 |
| Al-L3005 | Epoxy | 88.77 | 88.32 |
| Al-L3005 | Epoxy/Pb-orange | 117.8 | 91.11 |
| Brass B36 | Epoxy/Co-blue | 70.95 | 76.63 |
| Brass B36 | Epoxy | 97.43 | 87.50 |
| Brass B36 | Epoxy/Pb-orange | 97.99 | 92.73 |
| Bronze 510 | Epoxy/Co-blue | 74.96 | 84.06 |
| Bronze 510 | Epoxy | 93.30 | 87.14 |
| Bronze 510 | Epoxy/Pb-orange | 101.52 | 89.66 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Nedeljković, N.; Mladenović, I.O.; Vuksanović, M.M.; Lamovec, J.; Mušicki Bogdanović, M.Ž.; Vasiljević-Radović, D.G.; Heinemann, R.J. Tailoring Mechanical and Surface Properties of Epoxy Coatings with Synthesized Pigment Particles: Microhardness, Adhesion, and Wettability Studies. Coatings 2026, 16, 584. https://doi.org/10.3390/coatings16050584
Nedeljković N, Mladenović IO, Vuksanović MM, Lamovec J, Mušicki Bogdanović MŽ, Vasiljević-Radović DG, Heinemann RJ. Tailoring Mechanical and Surface Properties of Epoxy Coatings with Synthesized Pigment Particles: Microhardness, Adhesion, and Wettability Studies. Coatings. 2026; 16(5):584. https://doi.org/10.3390/coatings16050584
Chicago/Turabian StyleNedeljković, Nikola, Ivana O. Mladenović, Marija M. Vuksanović, Jelena Lamovec, Milica Ž. Mušicki Bogdanović, Dana G. Vasiljević-Radović, and Radmila Jančić Heinemann. 2026. "Tailoring Mechanical and Surface Properties of Epoxy Coatings with Synthesized Pigment Particles: Microhardness, Adhesion, and Wettability Studies" Coatings 16, no. 5: 584. https://doi.org/10.3390/coatings16050584
APA StyleNedeljković, N., Mladenović, I. O., Vuksanović, M. M., Lamovec, J., Mušicki Bogdanović, M. Ž., Vasiljević-Radović, D. G., & Heinemann, R. J. (2026). Tailoring Mechanical and Surface Properties of Epoxy Coatings with Synthesized Pigment Particles: Microhardness, Adhesion, and Wettability Studies. Coatings, 16(5), 584. https://doi.org/10.3390/coatings16050584