Recent Developments in Edible Films and Coatings for Fruits and Vegetables
Abstract
1. Introduction
- primary production (in agricultural production and harvesting),
- secondary production (in postharvest handling and storage, processing, distribution, and retailing),
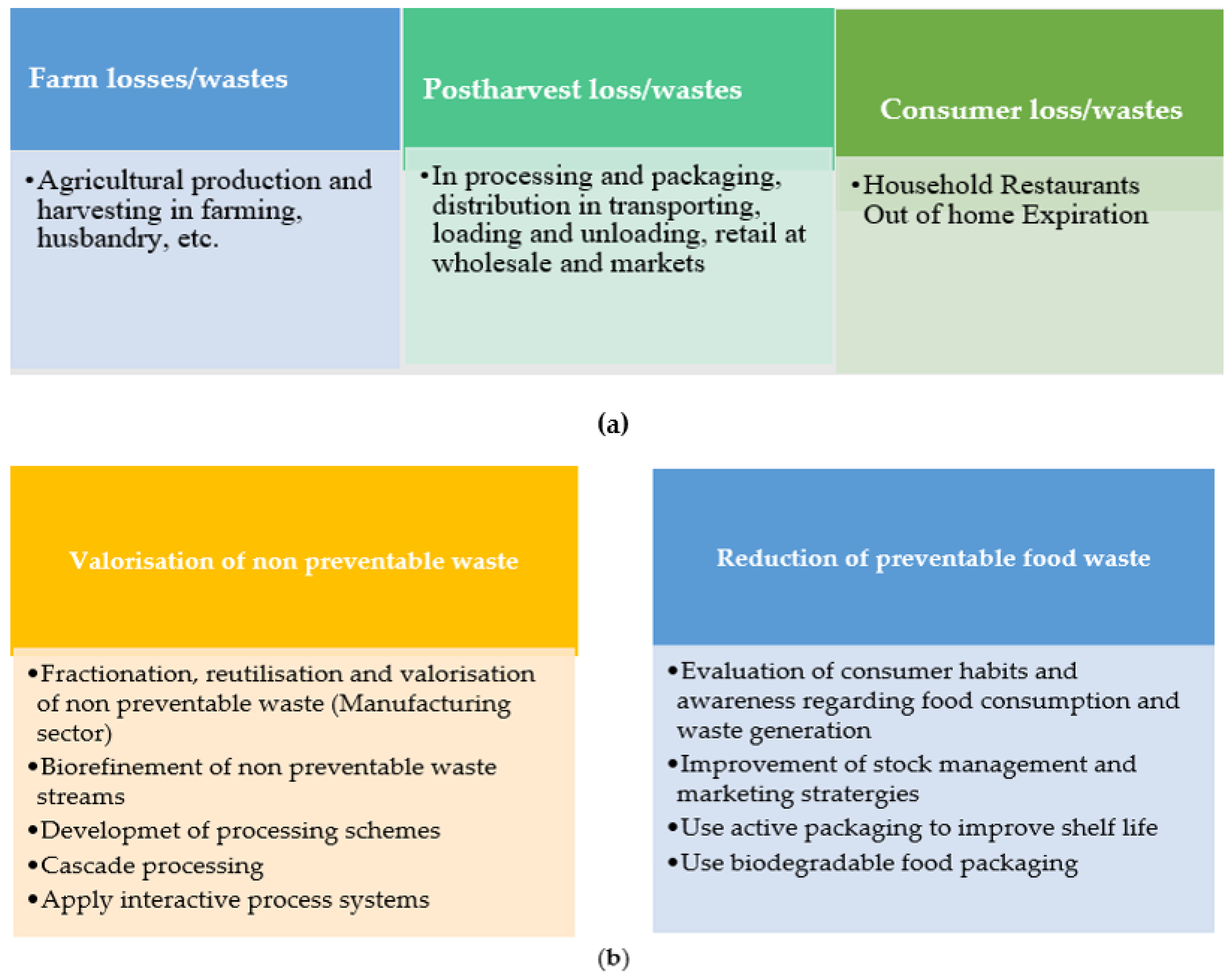
- consumption (in-household and out-of-home). Figure 1a depicts the major wastes and losses in food consumption.
2. Characteristics and Functionalities of Edible Films and Coatings
- (a)
- interactions with the food texture and surface;
- (b)
- aging and prolonged performance of the shelf life of the food in contact;
- (c)
- changes in flavor, color, and texture of food due to interactions with edible films and coatings;
- (d)
- response and sensitivity under storage/environmental conditions;
- (e)
- processing conditions, including temperature, color and thickness.
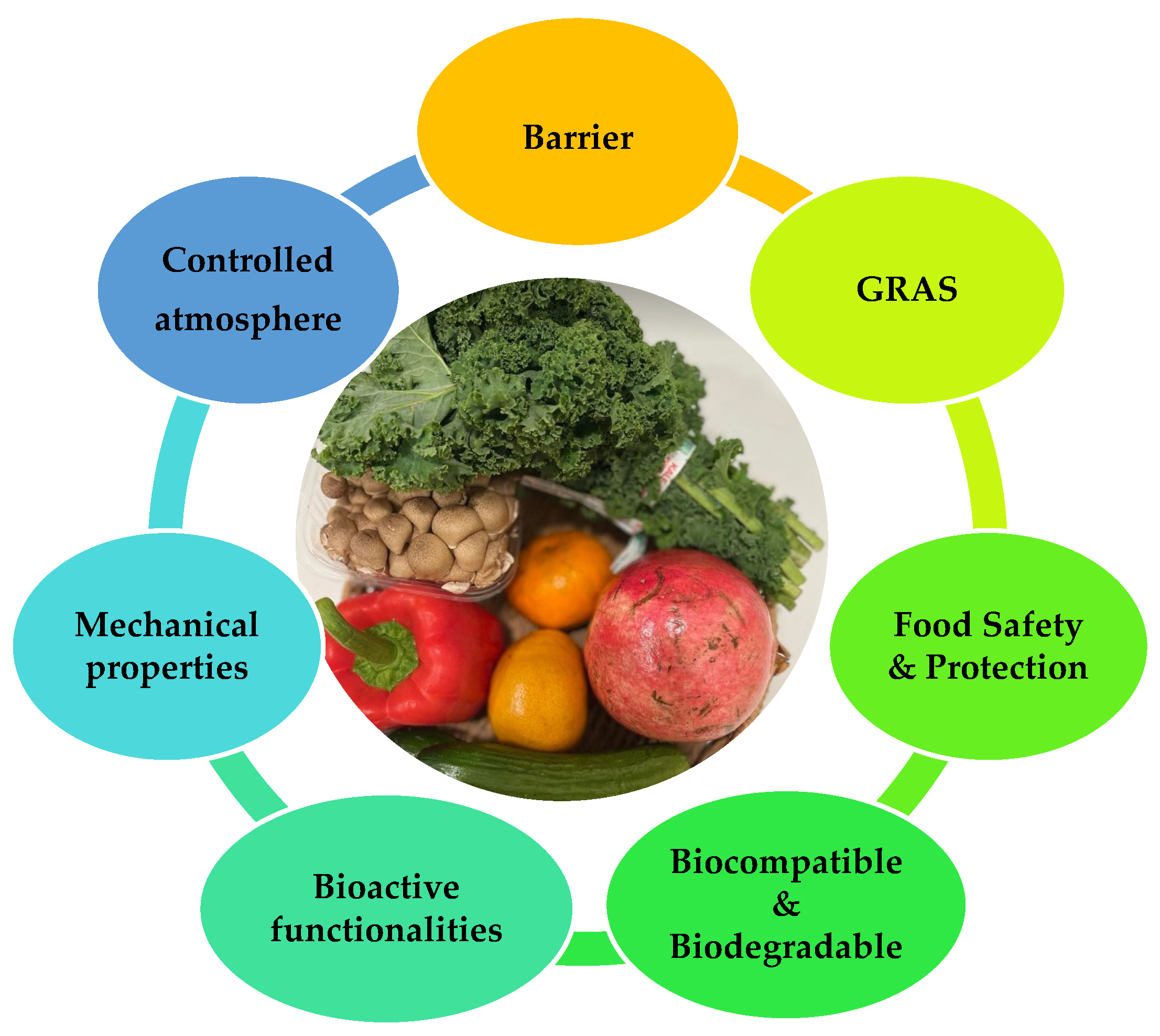
2.1. Key Functionalities of Edible Films and Coatings
- Protection from transport, handling, mechanical damages, and UV radiation;
- Barrier properties:
- (a)
- Moisture barrier: minimizes water vapor transmission to prevent dehydration;
- (b)
- Gas barrier: controls oxygen and carbon dioxide levels as they pass through the protective layer;
- (c)
- Volatile organic compounds (VOC) barrier: protects against organic vapors such as aromas and solvents and other additives and pigments.
- Prolongation of shelf life;
- Bioactivity: shows antimicrobial and antifungal properties and acts as probiotics;
- Biodegradability;
- Structural integrity: melts above 40 °C without decomposition, water resistant, easily emulsifiable, non-sticky, or non-tacky, and delivery efficient drying;
- Maintenance of food quality: minimal influence on texture, flavor, or color;
- Formulated from economical, relatively abundant, consumer-safety GRAS materials.
2.2. Common Preparation Methods of Edible Films and Coatings
2.2.1. Melt Extrusion Method
2.2.2. Solvent Casting Method
3. Types of Edible Films and Coatings and Their Structural and Chemical Significance
3.1. Hydrocolloids
3.2. Lipid Colloids
3.3. Composites
4. Polysaccharide-Based Edible Films and Coatings
4.1. Cellulose and Cellulosic Derivatives
4.2. Carrageenan
4.3. Pectin
4.4. Chitin and Chitosan
4.5. Gums
4.6. Starch
5. Protein-Based Edible Films and Coatings
5.1. Animal Proteins—Casein
5.2. Animal Proteins—Whey Protein
5.3. Animal Proteins—Collagen
5.4. Animal Proteins—Gelatin
5.5. Plant Proteins—Soy and Wheat Proteins
6. Lipid-Based Edible Films and Coatings
6.1. Essential Oils
6.2. Waxes and Resins
- (a)
- Animal waxes: bees wax, spermaceti wax, shellac wax, lanolin wax, Chinese insect wax;
- (b)
- Vegetable waxes: carnauba wax, candelilla wax, bayberry wax, rosin wood wax, sugarcane wax, palm wax, esparto wax, cotton seed wax, oricury wax, rice bran wax, Japan wax, and waxol;
- (c)
- Mineral and synthetic waxes: montan wax, paraffin wax, ozocerite, synthetic wax, and microcrystalline wax.
7. Health Effects
8. Conclusions and Current Trends
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- FAO. Food Wastage Footprint: Impacts on Natural Resources; FAO: Rome, Italy, 2013. [Google Scholar]
- Liu, C.; Hotta, Y.; Santo, A.; Hengesbaugh, M.; Watabe, A.; Totoki, Y.; Allen, D.; Bengtsson, M. Food waste in Japan: Trends, current practices and key challenges. J. Clean. Prod. 2016, 133, 557–564. [Google Scholar] [CrossRef]
- Gustavsson, J.; Cederberg, C.; Sonesson, U.; Van Otterdijk, R.; Meybeck, A. Global Food Losses and Food Waste; FAO: Rome, Italy, 2011. [Google Scholar]
- Amicarelli, V.; Lagioia, G.; Bux, C. Global warming potential of food waste through the life cycle assessment: An analytical review. Environ. Impact Assess. Rev. 2021, 91, 106677. [Google Scholar] [CrossRef]
- Poore, J.; Nemecek, T. Reducing food’s environmental impacts through producers and consumers. Science 2018, 360, 987–992. [Google Scholar] [CrossRef] [PubMed]
- Ritchie, H.; Roser, M. Environmental impacts of food production. 2020. Available online: https://ourworldindata.org/environmental-impacts-of-food (accessed on 28 May 2023).
- Racz, A.; Vasiljev Marchesi, V.; Crnković, I. Economical, environmental and ethical impact of food wastage in hospitality and other global industries. Jahr Eur. Časopis Bioetiku 2018, 9, 25–42. [Google Scholar]
- FAO. Food Wastage Footprint Full-Cost Accounting; FAO: Rome, Italy, 2014. [Google Scholar]
- Gross Domestic Product, Fourth Quarter and Year 2021 (Second Estimate). 2022. Available online: https://www.bea.gov/news/2023/gross-domestic-product-fourth-quarter-and-year-2022-second-estimate (accessed on 28 May 2023).
- United Nations Environment Programme. Think, Eat, Save. Reduce Your Footprint; UNEP: Nairobi, Kenya, 2021. [Google Scholar]
- United Nations General Assembly. Transforming Our World: The 2030 Agenda for Sustainable Development; A/RES/70/1; United Nations: New York, NY, USA, 2015.
- Xue, L.; Liu, G. 1—Introduction to global food losses and food waste. In Saving Food; Galanakis, C.M., Ed.; Academic Press: Cambridge, MA, USA, 2019; pp. 1–31. [Google Scholar]
- Xue, L.; Liu, G.; Parfitt, J.; Liu, X.; Van Herpen, E.; Stenmarck, Å.; O’Connor, C.; Östergren, K.; Cheng, S. Missing Food, Missing Data? A Critical Review of Global Food Losses and Food Waste Data. Environ. Sci. Technol. 2017, 51, 6618–6633. [Google Scholar] [CrossRef]
- Chen, C.; Chaudhary, A.; Mathys, A. Nutritional and environmental losses embedded in global food waste. Resour. Conserv. Recycl. 2020, 160, 104912. [Google Scholar] [CrossRef]
- Blakeney, M. Food Loss and Food Waste: Causes and Solutions; Edward Elgar Publishing: Cheltenham, UK, 2019. [Google Scholar]
- Gustafsson, J.; Cederberg, C.; Sonesson, U.; Emanuelsson, A. The Methodology of the FAO Study: Global Food Losses and Food Waste-Extent, Causes and Prevention-FAO, 2011; FAO: Rome, Italy, 2013. [Google Scholar]
- Sachdeva, S.; Sachdev, T.R.; Sachdeva, R. Increasing fruit and vegetable consumption: Challenges and opportunities. Indian J. Community Med. Off. Publ. Indian Assoc. Prev. Soc. Med. 2013, 38, 192. [Google Scholar] [CrossRef]
- Southon, S.; Faulks, R. Health benefits of increased fruit and vegetable consumption. Fruit Veg. Process. Improv. Qual. 2002, 1, 2–20. [Google Scholar]
- FAO. International Year of Fruits and Vegetables 2021; Global Action Plan: London, UK, 2021. [Google Scholar]
- Morone, P.; Koutinas, A.; Gathergood, N.; Arshadi, M.; Matharu, A. Food waste: Challenges and opportunities for enhancing the emerging bio-economy. J. Clean. Prod. 2019, 221, 10–16. [Google Scholar] [CrossRef]
- Lintas, C. Nutritional aspects of fruits and vegetables consumption. Options Mediterraennes 1992, 19, 79–87. [Google Scholar]
- Embuscado, M.E.; Huber, K.C. Edible Films and Coatings for Food Applications; Springer: Berlin/Heidelberg, Germany, 2009; Volume 9. [Google Scholar]
- Baldwin, E.A.; Hagenmaier, R.; Bai, J.; Krochta, J.M. Edible Coatings and Films to Improve Food Quality; Taylor & Francis: Abingdon, UK, 1994. [Google Scholar]
- Rossman, J.M. Commercial Manufacture of Edible Films. In Edible Films and Coatings for Food Applications; Huber, K.C., Embuscado, M.E., Eds.; Springer New York: New York, NY, USA, 2009; pp. 367–390. [Google Scholar]
- United States Food and Drug Administration. CFR—Code of Federal Regulations Title 21; FDA: Montgomery, MD, USA, 2022.
- United States Food and Drug Administration. Generally Recognized as Safe (GRAS); FDA: Montgomery, MD, USA, 2019.
- Díaz-Montes, E.; Castro-Muñoz, R. Edible Films and Coatings as Food-Quality Preservers: An Overview. Foods 2021, 10, 249. [Google Scholar] [CrossRef]
- Davis, G.; Song, J. Biodegradable packaging based on raw materials from crops and their impact on waste management. Ind. Crops Prod. 2006, 23, 147–161. [Google Scholar] [CrossRef]
- Bourtoom, T. Edible films and coatings: Characteristics and properties. Int. Food Res. J. 2008, 15, 237–248. [Google Scholar]
- Guimaraes, A.; Abrunhosa, L.; Pastrana, L.M.; Cerqueira, M.A. Edible films and coatings as carriers of living microorganisms: A new strategy towards biopreservation and healthier foods. Compr. Rev. Food Sci. Food Saf. 2018, 17, 594–614. [Google Scholar] [CrossRef] [PubMed]
- Olivas, G.I.I.; Barbosa-Cánovas, G. Edible Films and Coatings for Fruits and Vegetables. In Edible Films and Coatings for Food Applications; Huber, K.C., Embuscado, M.E., Eds.; Springer: New York, NY, USA, 2009; pp. 211–244. [Google Scholar]
- Rossi-Márquez, G.; Dávalos-Saucedo, C.A.; Di Pierro, P. Edible Films and Coatings Applied in the Food Industry. Coatings 2023, 13, 670. [Google Scholar] [CrossRef]
- Han, J.H. Edible films and coatings: A review. In Innovations in Food Packaging; Academic Press: Cambridge, MA, USA, 2014; pp. 213–255. [Google Scholar]
- Hernandez-Izquierdo, V.; Krochta, J. Thermoplastic processing of proteins for film formation—A review. J. Food Sci. 2008, 73, R30–R39. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Castellanos, W.; Martínez-Bustos, F.; Rodrigue, D.; Trujillo-Barragán, M. Extrusion blow molding of a starch–gelatin polymer matrix reinforced with cellulose. Eur. Polym. J. 2015, 73, 335–343. [Google Scholar] [CrossRef]
- Dahiya, M.; Saha, S.; Shahiwala, A.F. A review on mouth dissolving films. Curr. Drug Deliv. 2009, 6, 469–476. [Google Scholar] [CrossRef]
- Pavlath, A.E.; Orts, W. Edible films and coatings: Why, what, and how? In Edible Films and Coatings for Food Applications; Springer: Berlin/Heidelberg, Germany, 2009; pp. 1–23. [Google Scholar]
- Mellinas, C.; Valdés, A.; Ramos, M.; Burgos, N.; Garrigos, M.d.C.; Jiménez, A. Active edible films: Current state and future trends. J. Appl. Polym. Sci. 2016, 133, 42631. [Google Scholar] [CrossRef]
- Galus, S.; Kadzińska, J. Food applications of emulsion-based edible films and coatings. Trends Food Sci. Technol. 2015, 45, 273–283. [Google Scholar] [CrossRef]
- Bhattacharya, T. Techniques of Preparing Edible Protein Films. Asian J. Sci. Technol. 2013, 4, 39–41. [Google Scholar]
- Safaya, M.; Rotliwala, Y. Nanoemulsions: A review on low energy formulation methods, characterization, applications and optimization technique. Mater. Today Proc. 2020, 27, 454–459. [Google Scholar] [CrossRef]
- Gutoff, E.B.; Cohen, E.D. Water-and solvent-based coating technology. In Multilayer Flexible Packaging; Elsevier: Amsterdam, The Netherlands, 2016; pp. 205–234. [Google Scholar]
- Wang, Q.; Chen, W.; Zhu, W.; McClements, D.J.; Liu, X.; Liu, F. A review of multilayer and composite films and coatings for active biodegradable packaging. NPJ Sci. Food 2022, 6, 18. [Google Scholar] [CrossRef]
- Gupta, V.; Biswas, D.; Roy, S. A Comprehensive Review of Biodegradable Polymer-Based Films and Coatings and Their Food Packaging Applications. Materials 2022, 15, 5899. [Google Scholar] [CrossRef] [PubMed]
- Dickinson, E. Hydrocolloids at interfaces and the influence on the properties of dispersed systems. Food Hydrocoll. 2003, 17, 25–39. [Google Scholar] [CrossRef]
- Li, J.-M.; Nie, S.-P. The functional and nutritional aspects of hydrocolloids in foods. Food Hydrocoll. 2016, 53, 46–61. [Google Scholar] [CrossRef]
- Phillips, G.O.; Williams, P.A. Handbook of Hydrocolloids; Elsevier: Amsterdam, The Netherlands, 2009. [Google Scholar]
- Nishinari, K.; Zhang, H.; Ikeda, S. Hydrocolloid gels of polysaccharides and proteins. Curr. Opin. Colloid Interface Sci. 2000, 5, 195–201. [Google Scholar] [CrossRef]
- Goff, H.D.; Guo, Q. Chapter 1 The Role of Hydrocolloids in the Development of Food Structure. In Handbook of Food Structure Development; The Royal Society of Chemistry: London, UK, 2020; pp. 1–28. [Google Scholar]
- Godoi, F.C.; Ningtyas, D.W.; Geoffroy, Z.; Prakash, S. Protein-based hydrocolloids: Effect on the particle size distribution, tribo-rheological behaviour and mouthfeel characteristics of low-fat chocolate flavoured milk. Food Hydrocoll. 2021, 115, 106628. [Google Scholar] [CrossRef]
- Yemenicioğlu, A.; Farris, S.; Turkyilmaz, M.; Gulec, S. A review of current and future food applications of natural hydrocolloids. Int. J. Food Sci. Technol. 2020, 55, 1389–1406. [Google Scholar] [CrossRef]
- Kristl, J.; Šmid-Korbar, J.; Štruc, E.; Schara, M.; Rupprecht, H. Hydrocolloids and gels of chitosan as drug carriers. Int. J. Pharm. 1993, 99, 13–19. [Google Scholar] [CrossRef]
- Wüstenberg, T. Cellulose and Cellulose Derivatives in the Food Industry: Fundamentals and Applications; John Wiley & Sons: Hoboken, NJ, USA, 2014. [Google Scholar]
- Picot-Allain, M.C.N.; Ramasawmy, B.; Emmambux, M.N. Extraction, characterisation, and application of pectin from tropical and sub-tropical fruits: A review. Food Rev. Int. 2022, 38, 282–312. [Google Scholar] [CrossRef]
- Chen, J.; Liu, W.; Liu, C.-M.; Li, T.; Liang, R.-H.; Luo, S.-J. Pectin modifications: A review. Crit. Rev. Food Sci. Nutr. 2015, 55, 1684–1698. [Google Scholar] [CrossRef] [PubMed]
- Nieto, M.B. Structure and function of polysaccharide gum-based edible films and coatings. In Edible Films and Coatings for Food Applications; Springer: Berlin/Heidelberg, Germany, 2009; pp. 57–112. [Google Scholar]
- Alsabagh, A.; Abdou, M.; Khalil, A.; Ahmed, H.; Aboulrous, A. Investigation of some locally water-soluble natural polymers as circulation loss control agents during oil fields drilling. Egypt. J. Pet. 2014, 23, 27–34. [Google Scholar] [CrossRef]
- Marcotte, M.; Hoshahili, A.R.T.; Ramaswamy, H. Rheological properties of selected hydrocolloids as a function of concentration and temperature. Food Res. Int. 2001, 34, 695–703. [Google Scholar] [CrossRef]
- Kester, J.J.; Fennema, O. Edible films and coatings: A review. Food Technol. 1986, 40, 47–59. [Google Scholar]
- Bunjes, H. Structural properties of solid lipid based colloidal drug delivery systems. Curr. Opin. Colloid Interface Sci. 2011, 16, 405–411. [Google Scholar] [CrossRef]
- Gordillo-Galeano, A.; Ponce, A.; Mora-Huertas, C.E. Surface structural characteristics of some colloidal lipid systems used in pharmaceutics. J. Drug Deliv. Sci. Technol. 2021, 62, 102345. [Google Scholar] [CrossRef]
- Heurtault, B.; Saulnier, P.; Pech, B.; Proust, J.-E.; Benoit, J.-P. Physico-chemical stability of colloidal lipid particles. Biomaterials 2003, 24, 4283–4300. [Google Scholar] [CrossRef]
- Patel, A.R.; Velikov, K.P. Colloidal delivery systems in foods: A general comparison with oral drug delivery. LWT—Food Sci. Technol. 2011, 44, 1958–1964. [Google Scholar] [CrossRef]
- Wilde, P.J.; Chu, B.S. Interfacial & colloidal aspects of lipid digestion. Adv. Colloid Interface Sci. 2011, 165, 14–22. [Google Scholar]
- Bourtoom, T.; Chinnan, M. Improvement of water barrier property of rice starch-chitosan composite film incorporated with lipids. Food Sci. Technol. Int. 2009, 15, 149–158. [Google Scholar] [CrossRef]
- Ajesh, K.V.; Hasan, M.; Shukadev, M.; Pravitha, M. Deepak Kumar Verma, Prem Prakash Srivastav, Trends in Edible Packaging Films and its Prospective Future in Food: A Review. Appl. Food Res. 2022, 2, 100118. [Google Scholar]
- Baranwal, J.; Barse, B.; Fais, A.; Delogu, G.L.; Kumar, A. Biopolymer: A Sustainable Material for Food and Medical Applications. Polymers 2022, 14, 983. [Google Scholar] [CrossRef] [PubMed]
- Gutiérrez, T.J. Surface and nutraceutical properties of edible films made from starchy sources with and without added blackberry pulp. Carbohydr. Polym. 2017, 165, 169–179. [Google Scholar] [CrossRef]
- Dhumal, C.V.; Sarkar, P. Composite edible films and coatings from food-grade biopolymers. J. Food Sci. Technol. 2018, 55, 4369–4383. [Google Scholar] [CrossRef]
- Fu, X.; Chang, X.; Ding, Z.; Xu, H.; Kong, H.; Chen, F.; Wang, R.; Shan, Y.; Ding, S. Fabrication and Characterization of Eco-Friendly Polyelectrolyte Bilayer Films Based on Chitosan and Different Types of Edible Citrus Pectin. Foods 2022, 11, 3536. [Google Scholar] [CrossRef]
- Zhao, K.; Wang, W.; Teng, A.; Zhang, K.; Ma, Y.; Duan, S.; Li, S.; Guo, Y. Using cellulose nanofibers to reinforce polysaccharide films: Blending vs layer-by-layer casting. Carbohydr. Polym. 2020, 227, 115264. [Google Scholar] [CrossRef]
- Gamage, A.; Thiviya, P.; Mani, S.; Ponnusamy, P.G.; Manamperi, A.; Evon, P.; Merah, O.; Madhujith, T. Environmental Properties and Applications of Biodegradable Starch-Based Nanocomposites. Polymers 2022, 14, 4578. [Google Scholar] [CrossRef]
- Wang, B.; Yan, L.; Guo, S.; Wen, L.; Yu, M.; Feng, L.; Jia, X. Structural Elucidation, Modification, and Structure-Activity Relationship of Polysaccharides in Chinese Herbs: A Review. Front. Nutr. 2022, 9, 08175. [Google Scholar] [CrossRef]
- Guo, M.Q.; Hu, X.; Wang, C.; Ai, L. Polysaccharides: Structure and solubility. Solubility Polysacch. 2017, 2, 8–21. [Google Scholar]
- Ross-Murphy, S.; Shatwell, K. Polysaccharide strong and weak gels. Biorheology 1993, 30, 217–227. [Google Scholar] [CrossRef]
- Burchard, W. Structure formation by polysaccharides in concentrated solution. Biomacromolecules 2001, 2, 342–353. [Google Scholar] [CrossRef]
- Wettstein, S.G.; Alonso, D.M.; Gürbüz, E.I.; Dumesic, J.A. A roadmap for conversion of lignocellulosic biomass to chemicals and fuels. Curr. Opin. Chem. Eng. 2012, 1, 218–224. [Google Scholar] [CrossRef]
- Knauf, M.; Moniruzzaman, M. Lignocellulosic biomass processing: A perspective. Int. Sugar J. 2004, 106, 147–150. [Google Scholar]
- Tang, X.; Zuo, M.; Li, Z.; Liu, H.; Xiong, C.; Zeng, X.; Sun, Y.; Hu, L.; Liu, S.; Lei, T. Green processing of lignocellulosic biomass and its derivatives in deep eutectic solvents. Chem. Sus. Chem. 2017, 10, 2696–2706. [Google Scholar] [CrossRef]
- Xue, H.; Sun, M.; Zhao, X.; Wang, Y.; Yan, J.; Zhang, W. Preparation and Characterization of Polysaccharide-Based Hydrogels for Cutaneous Wound Healing. Polymers 2022, 14, 1716. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Li, B.; Li, C.; Xu, Y.; Luo, Y.; Liang, D.; Huang, C. Comprehensive Review of Polysaccharide-Based Materials in Edible Packaging: A Sustainable Approach. Foods 2021, 10, 1845. [Google Scholar] [CrossRef] [PubMed]
- Dhaka, R.; Upadhyay, A. Edible films and coatings: A brief overview. Pharma Innov. J. 2018, 7, 331–333. [Google Scholar]
- Lacroix, M.; Le Tien, C. 20—Edible films and coatings from nonstarch polysaccharides. In Innovations in Food Packaging; Han, J.H., Ed.; Academic Press: London, UK, 2005; pp. 338–361. [Google Scholar]
- Vazquez, A.; Foresti, M.L.; Moran, J.I.; Cyras, V.P. Extraction and production of cellulose nanofibers. In Handbook of Polymer Nanocomposites. Processing, Performance and Application; Springer: Berlin/Heidelberg, Germany, 2015; pp. 81–118. [Google Scholar]
- Dufresne, A. Nanocellulose: From Nature to High Performance Tailored Materials; Walter de Gruyter GmbH & Co KG: Berlin, Germany, 2017. [Google Scholar]
- Kargarzadeh, H.; Ahmad, I.; Thomas, S.; Dufresne, A. Handbook of Nanocellulose and Cellulose Nanocomposites; John Wiley & Sons: Hoboken, NJ, USA, 2017. [Google Scholar]
- Phanthong, P.; Reubroycharoen, P.; Hao, X.; Xu, G.; Abudula, A.; Guan, G. Nanocellulose: Extraction and application. Carbon Resour. Convers. 2018, 1, 32–43. [Google Scholar] [CrossRef]
- Burchard, W.; Schulz, L. Functionality of the β (1, 4) glycosidic linkage in polysaccharides. In Proceedings of the Macromolecular Symposia; Hüthig & Wepf Verlag: Basel, Switzerland, 1995; Volume 99, pp. 57–69. [Google Scholar]
- Trache, D.; Tarchoun, A.F.; Derradji, M.; Hamidon, T.S.; Masruchin, N.; Brosse, N.; Hussin, M.H. Nanocellulose: From Fundamentals to Advanced Applications. Front. Chem. 2020, 8, 392. [Google Scholar] [CrossRef]
- Habibi, Y.; Lucia, L.A.; Rojas, O.J. Cellulose nanocrystals: Chemistry, self-assembly, and applications. Chem. Rev. 2010, 110, 3479–3500. [Google Scholar] [CrossRef] [PubMed]
- Gu, H.; Zhou, X.; Lyu, S.; Pan, D.; Dong, M.; Wu, S.; Ding, T.; Wei, X.; Seok, I.; Wei, S. Magnetic nanocellulose-magnetite aerogel for easy oil adsorption. J. Colloid Interface Sci. 2020, 560, 849–856. [Google Scholar] [CrossRef] [PubMed]
- Hudson, S.M.; Cuculo, J.A. The solubility of unmodified cellulose: A critique of the literature. J. Macromol. Sci. Rev. Macromol. Chem. 1980, 18, 1–82. [Google Scholar] [CrossRef]
- Roy, S.; Rhim, J.-W. Advances and Challenges in Biopolymer-Based Films. Polymers 2022, 14, 3920. [Google Scholar] [CrossRef]
- Tajeddin, B. Cellulose-based polymers for packaging applications. In Lignocellulosic Polymer Composites; Thakur, V.K., Ed.; Scrivener Publishing LLC: Beverly, MA, USA, 2014; pp. 477–498. [Google Scholar]
- Osorio, F.A.; Molina, P.; Matiacevich, S.; Enrione, J.; Skurtys, O. Characteristics of hydroxy propyl methyl cellulose (HPMC) based edible film developed for blueberry coatings. Procedia Food Sci. 2011, 1, 287–293. [Google Scholar] [CrossRef]
- Singh, P.; Magalhães, S.; Alves, L.; Antunes, F.; Miguel, M.; Lindman, B.; Medronho, B. Cellulose-based edible films for probiotic entrapment. Food Hydrocoll. 2019, 88, 68–74. [Google Scholar] [CrossRef]
- Bastos, M.d.S.R.; Laurentino, L.d.S.; Canuto, K.M.; Mendes, L.G.; Martins, C.M.; Silva, S.M.F.; Furtado, R.F.; Kim, S.; Biswas, A.; Cheng, H.N. Physical and mechanical testing of essential oil-embedded cellulose ester films. Polym. Test. 2016, 49, 156–161. [Google Scholar] [CrossRef]
- Kong, I.; Degraeve, P.; Pui, L.P. Polysaccharide-Based Edible Films Incorporated with Essential Oil Nanoemulsions: Physico-Chemical, Mechanical Properties and Its Application in Food Preservation—A Review. Foods 2022, 11, 555. [Google Scholar] [CrossRef]
- Ghiasi, F.; Golmakani, M.-T.; Eskandari, M.H.; Hosseini, S.M.H. A new approach in the hydrophobic modification of polysaccharide-based edible films using structured oil nanoparticles. Ind. Crops Prod. 2020, 154, 112679. [Google Scholar] [CrossRef]
- Zhuang, C.; Tao, F.; Cui, Y. Eco-friendly biorefractory films of gelatin and TEMPO-oxidized cellulose ester for food packaging application. J. Sci. Food Agric. 2017, 97, 3384–3395. [Google Scholar] [CrossRef]
- Bras, J.; Vaca-Garcia, C.; Borredon, M.-E.; Glasser, W. Oxygen and water vapor permeability of fully substituted long chain cellulose esters (LCCE). Cellulose 2007, 14, 367–374. [Google Scholar] [CrossRef]
- Bilbao-Sáinz, C.; Avena-Bustillos, R.J.; Wood, D.F.; Williams, T.G.; McHugh, T.H. Composite Edible Films Based on Hydroxypropyl Methylcellulose Reinforced with Microcrystalline Cellulose Nanoparticles. J. Agric. Food Chem. 2010, 58, 3753–3760. [Google Scholar] [CrossRef]
- Fakhouri, F.; Tanada-Palmu, P.; Grosso, C. Characterization of composite biofilms of wheat gluten and cellulose acetate phthalate. Braz. J. Chem. Eng. 2004, 21, 261–264. [Google Scholar] [CrossRef]
- Debeaufort, F.; Quezada-Gallo, J.-A.; Delporte, B.; Voilley, A. Lipid hydrophobicity and physical state effects on the properties of bilayer edible films. J. Membr. Sci. 2000, 180, 47–55. [Google Scholar] [CrossRef]
- Kester, J.J.; Fennema, O. An Edible Film of Lipids and Cellulose Ethers: Barrier Properties to Moisture Vapor Transmission and Structural Evaluation. J. Food Sci. 1989, 54, 1383–1389. [Google Scholar] [CrossRef]
- Koh, H.-Y.; MS, C. Characteristics of corn zein and methyl cellulose bilayer edible films according to preparation protocol. Food Sci. Biotechnol. 2002, 11, 310–315. [Google Scholar]
- Park, J.W.; Testin, R.F.; Park, H.J.; Vergano, P.J.; Weller, C.L. Fatty Acid Concentration Effect on Tensile Strength, Elongation, and Water Vapor Permeability of Laminated Edible Films. J. Food Sci. 1994, 59, 916–919. [Google Scholar] [CrossRef]
- Shao, P.; Wu, W.; Chen, H.; Sun, P.; Gao, H. Bilayer edible films with tunable humidity regulating property for inhibiting browning of Agaricus bisporus. Food Res. Int. 2020, 138, 109795. [Google Scholar] [CrossRef]
- Liu, Z.; Lin, D.; Lopez-Sanchez, P.; Yang, X. Characterizations of bacterial cellulose nanofibers reinforced edible films based on konjac glucomannan. Int. J. Biol. Macromol. 2020, 145, 634–645. [Google Scholar] [CrossRef] [PubMed]
- Tabari, M. Characterization of a new biodegradable edible film based on Sago Starch loaded with Carboxymethyl Cellulose nanoparticles. Nanomed. Res. J. 2018, 3, 25–30. [Google Scholar]
- Tabari, M. Investigation of Carboxymethyl Cellulose (CMC) on Mechanical Properties of Cold Water Fish Gelatin Biodegradable Edible Films. Foods 2017, 6, 41. [Google Scholar] [CrossRef] [PubMed]
- Indumathi, M.; Sarojini, K.S.; Rajarajeswari, G. Antimicrobial and biodegradable chitosan/cellulose acetate phthalate/ZnO nano composite films with optimal oxygen permeability and hydrophobicity for extending the shelf life of black grape fruits. Int. J. Biol. Macromol. 2019, 132, 1112–1120. [Google Scholar] [CrossRef] [PubMed]
- Bhopal, R. Investigation of water vapour permeation and antibacterial properties of nano silver loaded cellulose acetate film. Int. Food Res. J. 2010, 17, 623–639. [Google Scholar]
- Atta, O.M.; Manan, S.; Ul-Islam, M.; Ahmed, A.A.Q.; Ullah, M.W.; Yang, G. Development and characterization of plant oil-incorporated carboxymethyl cellulose/bacterial cellulose/glycerol-based antimicrobial edible films for food packaging applications. Adv. Compos. Hybrid Mater. 2022, 5, 973–990. [Google Scholar] [CrossRef]
- Necas, J.; Bartosikova, L. Carrageenan: A review. Vet. Med. 2013, 58, 187–205. [Google Scholar] [CrossRef]
- Park, H.-J.; Rhim, J.-W.; Jung, S.-T.; Kang, S.-G.; Hwang, K.-T.; Park, Y.-K. Mechanical properties of carrageenan-based biopolymer films. Korean J. Packag. Sci. Technol. 1995, 1, 38–50. [Google Scholar]
- Balqis, A.I.; Khaizura, M.N.; Russly, A.; Hanani, Z.N. Effects of plasticizers on the physicochemical properties of kappa-carrageenan films extracted from Eucheuma cottonii. Int. J. Biol. Macromol. 2017, 103, 721–732. [Google Scholar] [CrossRef] [PubMed]
- Thomas, W. Carrageenan. In Thickening and Gelling Agents for Food; Springer: Berlin/Heidelberg, Germany, 1997; pp. 45–59. [Google Scholar]
- Baldwin, E.A.; Nisperos-Carriedo, M.O.; Baker, R.A. Use of edible coatings to preserve quality of lightly (and slightly) processed products. Crit. Rev. Food Sci. Nutr. 1995, 35, 509–524. [Google Scholar] [CrossRef]
- Lin, M.G.; Lasekan, O.; Saari, N.; Khairunniza-Bejo, S. The Effect of the Application of Edible Coatings on or before Ultraviolet Treatment on Postharvested Longan Fruits. J. Food Qual. 2017, 2017, 5454263. [Google Scholar] [CrossRef]
- Abdou, E.; Sorour, M. Preparation and characterization of starch/carrageenan edible films. Int. Food Res. J. 2014, 21, 189. [Google Scholar]
- Thakur, R.; Saberi, B.; Pristijono, P.; Golding, J.; Stathopoulos, C.; Scarlett, C.; Bowyer, M.; Vuong, Q. Characterization of rice starch-ι-carrageenan biodegradable edible film. Effect of stearic acid on the film properties. Int. J. Biol. Macromol. 2016, 93, 952–960. [Google Scholar] [CrossRef] [PubMed]
- Sandhu, K.S.; Sharma, L.; Kaur, M.; Kaur, R. Physical, structural and thermal properties of composite edible films prepared from pearl millet starch and carrageenan gum: Process optimization using response surface methodology. Int. J. Biol. Macromol. 2020, 143, 704–713. [Google Scholar] [CrossRef]
- Larotonda, F.D.S.; Torres, M.D.; Gonçalves, M.P.; Sereno, A.M.; Hilliou, L. Hybrid carrageenan-based formulations for edible film preparation: Benchmarking with kappa carrageenan. J. Appl. Polym. Sci. 2016, 133, 42263. [Google Scholar] [CrossRef]
- Campos, C.A.; Gerschenson, L.N.; Flores, S.K. Development of Edible Films and Coatings with Antimicrobial Activity. Food Bioprocess Technol. 2011, 4, 849–875. [Google Scholar] [CrossRef]
- Paula, G.A.; Benevides, N.M.B.; Cunha, A.P.; de Oliveira, A.V.; Pinto, A.M.B.; Morais, J.P.S.; Azeredo, H.M.C. Development and characterization of edible films from mixtures of κ-carrageenan, ι-carrageenan, and alginate. Food Hydrocoll. 2015, 47, 140–145. [Google Scholar] [CrossRef]
- Hambleton, A.; Voilley, A.; Debeaufort, F. Transport parameters for aroma compounds through i-carrageenan and sodium alginate-based edible films. Food Hydrocoll. 2011, 25, 1128–1133. [Google Scholar] [CrossRef]
- Thiviya, P.; Gamage, A.; Liyanapathiranage, A.; Makehelwala, M.; Dassanayake, R.S.; Manamperi, A.; Merah, O.; Mani, S.; Koduru, J.R.; Terrence Madhujith, T. Algal polysaccharides: Structure, preparation and applications in food packaging. Food Chem. 2022, 405, 134903. [Google Scholar] [CrossRef]
- Jayakody, M.M.; Vanniarachchy, M.P.G.; Wijesekara, I. Seaweed derived alginate, agar, and carrageenan based edible coatings and films for the food industry: A review. J. Food Meas. Charact. 2022, 16, 1195–1227. [Google Scholar] [CrossRef]
- Cebrián-Lloret, V.; Göksen, G.; Martínez-Abad, A.; López-Rubio, A.; Martínez-Sanz, M. Agar-based packaging films produced by melt mixing: Study of their retrogradation upon storage. Algal Res. 2022, 66, 102802. [Google Scholar] [CrossRef]
- Mostafavi, F.S.; Zaeim, D. Agar-based edible films for food packaging applications—A review. Int. J. Biol. Macromol. 2020, 159, 1165–1176. [Google Scholar] [CrossRef]
- Wongphan, P.; Harnkarnsujarit, N. Characterization of starch, agar and maltodextrin blends for controlled dissolution of edible films. Int. J. Biol. Macromol. 2020, 156, 80–93. [Google Scholar] [CrossRef]
- Phan The, D.; Debeaufort, F.; Voilley, A.; Luu, D. Biopolymer interactions affect the functional properties of edible films based on agar, cassava starch and arabinoxylan blends. J. Food Eng. 2009, 90, 548–558. [Google Scholar] [CrossRef]
- Arham, R.; Mulyati, M.; Metusalach, M.; Salengke, S. Physical and mechanical properties of agar based edible film with glycerol plasticizer. Int. Food Res. J. 2016, 23, 1669–1675. [Google Scholar]
- Sousa, A.M.; Sereno, A.M.; Hilliou, L.; Gonçalves, M.P. Biodegradable Agar Extracted from Gracilaria vermiculophylla: Film Properties and Application to Edible Coating. In Proceedings of the Materials Science Forum; Trans Tech Publications Ltd: Stafa-Zurich, Switzerland, 2010; Volume 636, pp. 739–744. [Google Scholar]
- Shahidi, F.; Hossain, A. Preservation of aquatic food using edible films and coatings containing essential oils: A review. Crit. Rev. Food Sci. Nutr. 2022, 62, 66–105. [Google Scholar] [CrossRef] [PubMed]
- Perera, K.Y.; Sharma, S.; Pradhan, D.; Jaiswal, A.K.; Jaiswal, S. Seaweed Polysaccharide in Food Contact Materials (Active Packaging, Intelligent Packaging, Edible Films, and Coatings). Foods 2021, 10, 2088. [Google Scholar] [CrossRef]
- Thakur, B.R.; Singh, R.K.; Handa, A.K.; Rao, M. Chemistry and uses of pectin—A review. Crit. Rev. Food Sci. Nutr. 1997, 37, 47–73. [Google Scholar] [CrossRef] [PubMed]
- Maftoonazad, N.; Ramaswamy, H.S.; Marcotte, M. Evaluation of factors affecting barrier, mechanical and optical properties of pectin-based films using response surface methodology. J. Food Process Eng. 2007, 30, 539–563. [Google Scholar] [CrossRef]
- Rodsamran, P.; Sothornvit, R. Preparation and characterization of pectin fraction from pineapple peel as a natural plasticizer and material for biopolymer film. Food Bioprod. Process. 2019, 118, 198–206. [Google Scholar] [CrossRef]
- Lazaridou, A.; Biliaderis, C.G. Edible Films and Coatings with Pectin. In Pectin: Technological and Physiological Properties; Kontogiorgos, V., Ed.; Springer International Publishing: Cham, Switzerland, 2020; pp. 99–123. [Google Scholar]
- Shahrampour, D.; Khomeiri, M.; Razavi, S.M.A.; Kashiri, M. Development and characterization of alginate/pectin edible films containing Lactobacillus plantarum KMC 45. LWT 2020, 118, 108758. [Google Scholar] [CrossRef]
- Alvarez, M.V.; Ortega-Ramirez, L.A.; Gutierrez-Pacheco, M.M.; Bernal-Mercado, A.T.; Rodriguez-Garcia, I.; Gonzalez-Aguilar, G.A.; Ponce, A.; Moreira, M.d.R.; Roura, S.I.; Ayala-Zavala, J.F. Oregano essential oil-pectin edible films as anti-quorum sensing and food antimicrobial agents. Front. Microbiol. 2014, 5, 699. [Google Scholar] [CrossRef]
- Estrada-Girón, Y.; Cabrera-Díaz, E.; Esparza-Merino, R.M.; Martín-del-Campo, A.; Valencia-Botín, A.J. Innovative edible films and coatings based on red color pectin obtained from the byproducts of Hibiscus sabdariffa L. for strawberry preservation. J. Food Meas. Charact. 2020, 14, 3371–3380. [Google Scholar] [CrossRef]
- Rossi Marquez, G.; Di Pierro, P.; Mariniello, L.; Esposito, M.; Giosafatto, C.V.L.; Porta, R. Fresh-cut fruit and vegetable coatings by transglutaminase-crosslinked whey protein/pectin edible films. LWT 2017, 75, 124–130. [Google Scholar] [CrossRef]
- Alvarez-Pérez, O.B.; Montañez, J.; Aguilar, C.N.; Rojas, R. Pectin–Candelilla wax: An alternative mixture for edible films. J. Microbiol. Biotechnol. Food Sci. 2021, 2021, 167–171. [Google Scholar] [CrossRef]
- Piekarska, K.; Sikora, M.; Owczarek, M.; Jó ’zwik-Pruska, J.; Wiśniewska-Wrona, M. Chitin and Chitosan as Polymers of the Future—Obtaining, Modification, Life Cycle Assessment and Main Directions of Application. Polymers 2023, 15, 793. [Google Scholar] [CrossRef]
- Roberts, G.A.; Roberts, G.A. Chitin Chemistry; Springer: Berlin/Heidelberg, Germany, 1992. [Google Scholar]
- Hudson, S.; Smith, C. Polysaccharides: Chitin and chitosan: Chemistry and technology of their use as structural materials. In Biopolymers from Renewable Resources; Springer: Berlin/Heidelberg, Germany, 1998; pp. 96–118. [Google Scholar]
- Sivakanthan, S.; Rajendran, S.; Gamage, A.; Madhujith, T.; Mani, S. Antioxidant and antimicrobial applications of biopolymers: A review. Food Res. Int. 2020, 136, 109327. [Google Scholar] [CrossRef] [PubMed]
- Hernández-Téllez, C.N.; Luque-Alcaraz, A.G.; Núñez-Mexía, S.A.; Cortez-Rocha, M.O.; Lizardi-Mendoza, J.; Rosas-Burgos, E.C.; Rosas-Durazo, A.d.J.; Parra-Vergara, N.V.; Plascencia-Jatomea, M. Relationship between the Antifungal Activity of Chitosan–Capsaicin Nanoparticles and the Oxidative Stress Response on Aspergillus parasiticus. Polymers 2022, 14, 2774. [Google Scholar] [CrossRef] [PubMed]
- Ardean, C.; Davidescu, C.M.; Neme¸s, N.S.; Negrea, A.; Ciopec, M.; Duteanu, N.; Negrea, P.; Duda-Seiman, D.; Musta, V. Factors Influencing the Antibacterial Activity of Chitosan and Chitosan Modified by Functionalization. Int. J. Mol. Sci. 2021, 22, 7449. [Google Scholar] [CrossRef] [PubMed]
- Pavinatto, A.; de Almeida Mattos, A.V.; Malpass, A.C.G.; Okura, M.H.; Balogh, D.T.; Sanfelice, R.C. Coating with chitosan-based edible films for mechanical/biological protection of strawberries. Int. J. Biol. Macromol. 2020, 151, 1004–1011. [Google Scholar] [CrossRef]
- da Mata Cunha, O.; Lima, A.M.F.; Assis, O.B.G.; Tiera, M.J.; de Oliveira Tiera, V.A. Amphiphilic diethylaminoethyl chitosan of high molecular weight as an edible film. Int. J. Biol. Macromol. 2020, 164, 3411–3420. [Google Scholar] [CrossRef]
- Salama, H.E.; Abdel Aziz, M.S.; Sabaa, M.W. Novel biodegradable and antibacterial edible films based on alginate and chitosan biguanidine hydrochloride. Int. J. Biol. Macromol. 2018, 116, 443–450. [Google Scholar] [CrossRef]
- Velickova, E.; Winkelhausen, E.; Kuzmanova, S.; Moldão-Martins, M.; Alves, V.D. Characterization of multilayered and composite edible films from chitosan and beeswax. Food Sci. Technol. Int. 2015, 21, 83–93. [Google Scholar] [CrossRef]
- Tokatlı, K.; Demirdöven, A. Effects of chitosan edible film coatings on the physicochemical and microbiological qualities of sweet cherry (Prunus avium L.). Sci. Hortic. 2020, 259, 108656. [Google Scholar] [CrossRef]
- Doublier, J.-L.; Garnier, C.; Cuvelier, G. Gums and hydrocolloids: Functional aspects. In Carbohydrates in Food; CRC Press: Boca Raton, FL, USA, 2017; pp. 307–354. [Google Scholar]
- Lim, W.; Shin, S.Y.; Cha, J.M.; Bae, H. Optimization of Polysaccharide Hydrocolloid for the Development of Bioink with High Printability/Biocompatibility for Coextrusion 3D Bioprinting. Polymers 2021, 13, 1773. [Google Scholar] [CrossRef] [PubMed]
- Khezerlou, A.; Zolfaghari, H.; Banihashemi, S.A.; Forghani, S.; Ehsani, A. Plant gums as the functional compounds for edible films and coatings in the food industry: A review. Polym. Adv. Technol. 2021, 32, 2306–2326. [Google Scholar] [CrossRef]
- Islam, A.; Phillips, G.; Sljivo, A.; Snowden, M.; Williams, P. A review of recent developments on the regulatory, structural and functional aspects of gum arabic. Food Hydrocoll. 1997, 11, 493–505. [Google Scholar] [CrossRef]
- Musa, H.H.; Ahmed, A.A.; Musa, T.H. Chemistry, biological, and pharmacological properties of gum Arabic. Bioactive Molecules in Food; Springer International Publishing AG: Cham, Switzerland, 2018; pp. 1–18. [Google Scholar]
- Xu, T.; Gao, C.; Feng, X.; Yang, Y.; Shen, X.; Tang, X. Structure, physical and antioxidant properties of chitosan-gum arabic edible films incorporated with cinnamon essential oil. Int. J. Biol. Macromol. 2019, 134, 230–236. [Google Scholar] [CrossRef] [PubMed]
- Razak, A.S.; Lazim, A.M. Starch-based edible film with gum arabic for fruits coating. AIP Conf. Proc. 2015, 1678, 050020. [Google Scholar]
- Xue, F.; Gu, Y.; Wang, Y.; Li, C.; Adhikari, B. Encapsulation of essential oil in emulsion based edible films prepared by soy protein isolate-gum acacia conjugates. Food Hydrocoll. 2019, 96, 178–189. [Google Scholar] [CrossRef]
- Li, C.; Pei, J.; Xiong, X.; Xue, F. Encapsulation of Grapefruit Essential Oil in Emulsion-Based Edible Film Prepared by Plum (Pruni Domesticae Semen) Seed Protein Isolate and Gum Acacia Conjugates. Coatings 2020, 10, 784. [Google Scholar] [CrossRef]
- Mathur, N. Industrial Galactomannan Polysaccharides; CRC Press Taylor & Francis Group: Boca Raton, FL, USA, 2012. [Google Scholar]
- Prajapati, V.D.; Jani, G.K.; Moradiya, N.G.; Randeria, N.P.; Nagar, B.J.; Naikwadi, N.N.; Variya, B.C. Galactomannan: A versatile biodegradable seed polysaccharide. Int. J. Biol. Macromol. 2013, 60, 83–92. [Google Scholar] [CrossRef]
- Antoniou, J.; Liu, F.; Majeed, H.; Qazi, H.J.; Zhong, F. Physicochemical and thermomechanical characterization of tara gum edible films: Effect of polyols as plasticizers. Carbohydr. Polym. 2014, 111, 359–365. [Google Scholar] [CrossRef] [PubMed]
- Mudgil, D.; Barak, S.; Khatkar, B.S. Guar gum: Processing, properties and food applications—A review. J. Food Sci. Technol. 2014, 51, 409–418. [Google Scholar] [CrossRef] [PubMed]
- Patel, J.; Maji, B.; Moorthy, N.H.N.; Maiti, S. Xanthan gum derivatives: Review of synthesis, properties and diverse applications. RSC Adv. 2020, 10, 27103–27136. [Google Scholar] [CrossRef]
- Hashemi Gahruie, H.; Safdarianghomsheh, R.; Zamanifar, P.; Salehi, S.; Niakousari, M.; Hosseini, S.M.H. Characterization of novel edible films and coatings for food preservation based on gum cordia. J. Food Qual. 2020, 2020, 8883916. [Google Scholar] [CrossRef]
- Radev, R.; Pashova, S. Application of edible films and coatings for fresh fruit and vegetables. Qual. Access Success 2020, 21, 108–112. [Google Scholar]
- Dubey, N.K.; Dubey, R. Edible films and coatings: An update on recent advances. In Biopolymer-Based Formulations; Elsevier: Amsterdam, The Netherlands, 2020; pp. 675–695. [Google Scholar]
- Dhumal, C.V.; Ahmed, J.; Bandara, N.; Sarkar, P. Improvement of antimicrobial activity of sago starch/guar gum bi-phasic edible films by incorporating carvacrol and citral. Food Packag. Shelf Life 2019, 21, 100380. [Google Scholar] [CrossRef]
- Saberi, B.; Chockchaisawasdee, S.; Golding, J.B.; Scarlett, C.J.; Stathopoulos, C.E. Physical and mechanical properties of a new edible film made of pea starch and guar gum as affected by glycols, sugars and polyols. Int. J. Biol. Macromol. 2017, 104, 345–359. [Google Scholar] [CrossRef]
- Saberi, B.; Chockchaisawasdee, S.; Golding, J.B.; Scarlett, C.J.; Stathopoulos, C.E. Characterization of pea starch-guar gum biocomposite edible films enriched by natural antimicrobial agents for active food packaging. Food Bioprod. Process. 2017, 105, 51–63. [Google Scholar] [CrossRef]
- Saberi, B.; Vuong, Q.V.; Chockchaisawasdee, S.; Golding, J.B.; Scarlett, C.J.; Stathopoulos, C.E. Physical, Barrier, and Antioxidant Properties of Pea Starch-Guar Gum Biocomposite Edible Films by Incorporation of Natural Plant Extracts. Food Bioprocess Technol. 2017, 10, 2240–2250. [Google Scholar] [CrossRef]
- Naji-Tabasi, S.; Razavi, S.M.A. Functional properties and applications of basil seed gum: An overview. Food Hydrocoll. 2017, 73, 313–325. [Google Scholar] [CrossRef]
- Hashemi, S.M.B.; Mousavi Khaneghah, A. Characterization of novel basil-seed gum active edible films and coatings containing oregano essential oil. Prog. Org. Coat. 2017, 110, 35–41. [Google Scholar] [CrossRef]
- Salehi, F. Characterization of New Biodegradable Edible Films and Coatings Based on Seeds Gum: A Review. J. Packag. Technol. Res. 2019, 3, 193–201. [Google Scholar] [CrossRef]
- Hashemi Gahruie, H.; Mostaghimi, M.; Ghiasi, F.; Tavakoli, S.; Naseri, M.; Hosseini, S.M.H. The effects of fatty acids chain length on the techno-functional properties of basil seed gum-based edible films. Int. J. Biol. Macromol. 2020, 160, 245–251. [Google Scholar] [CrossRef]
- Prajapati, V.D.; Jani, G.K.; Zala, B.S.; Khutliwala, T.A. An insight into the emerging exopolysaccharide gellan gum as a novel polymer. Carbohydr. Polym. 2013, 93, 670–678. [Google Scholar] [CrossRef] [PubMed]
- Giavasis, I.; Harvey, L.M.; McNeil, B. Gellan gum. Crit. Rev. Biotechnol. 2000, 20, 177–211. [Google Scholar] [CrossRef] [PubMed]
- Falguera, V.; Quintero, J.P.; Jiménez, A.; Muñoz, J.A.; Ibarz, A. Edible films and coatings: Structures, active functions and trends in their use. Trends Food Sci. Technol. 2011, 22, 292–303. [Google Scholar] [CrossRef]
- Alvarado-González, J.; Chanona-Pérez, J.; Welti-Chanes, J.; Calderón-Domínguez, G.; Arzate-Vázquez, I.; Pacheco-Alcalá, S.; Garibay-Febles, V.; Gutiérrez-López, G. Optical, microstructural, functional and nanomechanical properties of Aloe vera gel/gellan gum edible films. Rev. Mex. Ing. Quím. 2012, 11, 193–210. [Google Scholar]
- Maan, A.A.; Reiad Ahmed, Z.F.; Iqbal Khan, M.K.; Riaz, A.; Nazir, A. Aloe vera gel, an excellent base material for edible films and coatings. Trends Food Sci. Technol. 2021, 116, 329–341. [Google Scholar] [CrossRef]
- Téllez-Rangel, E.C.; Rodríguez-Huezo, E.; Totosaus, A. Effect of gellan, xanthan or locust bean gum and/or emulsified maize oil on proteins edible films properties. Emir. J. Food Agric. 2018, 30, 404–412. [Google Scholar]
- Tester, R.F.; Karkalas, J.; Qi, X. Starch—Composition, fine structure and architecture. J. Cereal Sci. 2004, 39, 151–165. [Google Scholar] [CrossRef]
- van Soest, J.J.G.; Vliegenthart, J.F.G. Crystallinity in starch plastics: Consequences for material properties. Trends Biotechnol. 1997, 15, 208–213. [Google Scholar] [CrossRef] [PubMed]
- Rindlav-Westling, Å.; Stading, M.; Gatenholm, P. Crystallinity and morphology in films of starch, amylose and amylopectin blends. Biomacromolecules 2002, 3, 84–91. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Rempel, C.; Liu, Q. Thermoplastic Starch Processing and Characteristics—A Review. Crit. Rev. Food Sci. Nutr. 2014, 54, 1353–1370. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z. 19—Edible films and coatings from starches. In Innovations in Food Packaging; Han, J.H., Ed.; Academic Press: London, UK, 2005; pp. 318–337. [Google Scholar]
- Shah, U.; Naqash, F.; Gani, A.; Masoodi, F.A. Art and Science behind Modified Starch Edible Films and Coatings: A Review. Compr. Rev. Food Sci. Food Saf. 2016, 15, 568–580. [Google Scholar] [CrossRef]
- Chiumarelli, M.; Hubinger, M.D. Evaluation of edible films and coatings formulated with cassava starch, glycerol, carnauba wax and stearic acid. Food Hydrocoll. 2014, 38, 20–27. [Google Scholar] [CrossRef]
- Pedreiro, S.; Figueirinha, A.; Silva, A.S.; Ramos, F. Bioactive Edible Films and Coatings Based in Gums and Starch: Phenolic Enrichment and Foods Application. Coatings 2021, 11, 1393. [Google Scholar] [CrossRef]
- Basiak, E.; Lenart, A.; Debeaufort, F. Effect of starch type on the physico-chemical properties of edible films. Int. J. Biol. Macromol. 2017, 98, 348–356. [Google Scholar] [CrossRef]
- García, M.A.; Martino, M.N.; Zaritzky, N.E. Lipid Addition to Improve Barrier Properties of Edible Starch-based Films and Coatings. J. Food Sci. 2000, 65, 941–944. [Google Scholar] [CrossRef]
- Rodríguez, M.; Osés, J.; Ziani, K.; Maté, J.I. Combined effect of plasticizers and surfactants on the physical properties of starch based edible films. Food Res. Int. 2006, 39, 840–846. [Google Scholar] [CrossRef]
- Basiak, E.; Lenart, A.; Debeaufort, F. How Glycerol and Water Contents Affect the Structural and Functional Properties of Starch-Based Edible Films. Polymers 2018, 10, 412. [Google Scholar] [CrossRef]
- Farahnaky, A.; Saberi, B.; Majzoobi, M. Effect of Glycerol on Physical and Mechanical Properties of Wheat Starch Edible Films. J. Texture Stud. 2013, 44, 176–186. [Google Scholar] [CrossRef]
- Kim, K.W.; Ko, C.J.; Park, H.J. Mechanical Properties, Water Vapor Permeabilities and Solubilities of Highly Carboxymethylated Starch-Based Edible Films. J. Food Sci. 2002, 67, 218–222. [Google Scholar] [CrossRef]
- Charles, A.L.; Motsa, N.; Abdillah, A.A. A Comprehensive Characterization of Biodegradable Edible Films Based on Potato Peel Starch Plasticized with Glycerol. Polymers 2022, 14, 3462. [Google Scholar] [CrossRef] [PubMed]
- Wilpiszewska, K.; Adrian Krzysztof Antosik, A.K.; Schmidt, B.; Janik, J.; Rokicka, J. Hydrophilic Films Based on Carboxymethylated Derivatives of Starch and Cellulose. Polymers 2020, 12, 2447. [Google Scholar] [CrossRef] [PubMed]
- Nogueira, G.F.; Leme, B.d.O.; Santos, G.R.S.d.; Silva, J.V.d.; Nascimento, P.B.; Soares, C.T.; Fakhouri, F.M.; de Oliveira, R.A. Edible Films and Coatings Formulated with Arrowroot Starch as a Non-Conventional Starch Source for Plums Packaging. Polysaccharides 2021, 2, 373–386. [Google Scholar] [CrossRef]
- Peressini, D.; Bravin, B.; Lapasin, R.; Rizzotti, C.; Sensidoni, A. Starch–methylcellulose based edible films: Rheological properties of film-forming dispersions. J. Food Eng. 2003, 59, 25–32. [Google Scholar] [CrossRef]
- Cuq, B.; Gontard, N.; Guilbert, S. Proteins as Agricultural Polymers for Packaging Production. Cereal Chem. 1998, 75, 1–9. [Google Scholar] [CrossRef]
- Mchugh, T.H.; Aujard, J.F.; Krochta, J. Plasticized whey protein edible films: Water vapor permeability properties. J. Food Sci. 1994, 59, 416–419. [Google Scholar] [CrossRef]
- Martucci, J.F.; Ruseckaite, R.A. Biodegradation of three-layer laminate films based on gelatin under indoor soil conditions. Polym. Degrad. Stab. 2009, 94, 1307–1313. [Google Scholar] [CrossRef]
- Jauregi, P. Bioactive peptides from food proteins: New opportunities and challenges. Food Sci. Technol. Bull. Funct. Foods 2009, 5, 11–25. [Google Scholar] [CrossRef]
- Alkan, D.; Yemenicioğlu, A. Potential application of natural phenolic antimicrobials and edible film technology against bacterial plant pathogens. Food Hydrocoll. 2016, 55, 1–10. [Google Scholar] [CrossRef]
- Banerjee, R.; Chen, H.; Wu, J. Milk protein-based edible film mechanical strength changes due to ultrasound process. J. Food Sci. 1996, 61, 824–828. [Google Scholar] [CrossRef]
- Panyam, D.; Kilara, A. Enhancing the functionality of food proteins by enzymatic modification. Trends Food Sci. Technol. 1996, 7, 120–125. [Google Scholar] [CrossRef]
- Yada, R.Y. Proteins in Food Processing; Woodhead Publishing: Sawston, UK, 2017. [Google Scholar]
- Audic, J.-L.; Chaufer, B.; Daufin, G. Non-food applications of milk components and dairy co-products: A review. Le Lait 2003, 83, 417–438. [Google Scholar] [CrossRef]
- Hinz, K.; O’Connor, P.M.; Huppertz, T.; Ross, R.P.; Kelly, A.L. Comparison of the principal proteins in bovine, caprine, buffalo, equine and camel milk. J. Dairy Res. 2012, 79, 185–191. [Google Scholar] [CrossRef]
- Petrova, S.Y.; Khlgatian, S.; Emel’yanova, O.Y.; Pishulina, L.; Berzhets, V. Current Data about Milk Caseins. Russ. J. Bioorg. Chem. 2022, 48, 273–280. [Google Scholar] [CrossRef]
- Swaisgood, H.E. Review and Update of Casein Chemistry. J. Dairy Sci. 1993, 76, 3054–3061. [Google Scholar] [CrossRef]
- Lacroix, M.; Cooksey, K. 18—Edible films and coatings from animal origin proteins. In Innovations in Food Packaging; Han, J.H., Ed.; Academic Press: London, UK, 2005; pp. 301–317. [Google Scholar]
- Shendurse, A.; Gopikrishna, G.; Patel, A.; Pandya, A. Milk protein based edible films and coatings–preparation, properties and food applications. J. Nutr. Health Food Eng. 2018, 8, 219–226. [Google Scholar] [CrossRef]
- Chen, H. Functional Properties and Applications of Edible Films Made of Milk Proteins. J. Dairy Sci. 1995, 78, 2563–2583. [Google Scholar] [CrossRef]
- Phillips, L.G.; Whitehead, D.M.; Kinsella, J. Introduction to Functional Properties of Proteins. In Structure–Function Properties of Food Proteins; Phillips, L.G., Whitehead, D.M., Kinsella, J., Eds.; Academic Press: Cambridge, MA, USA, 1994; pp. 107–109. [Google Scholar]
- Avena-Bustillos, R.J.; Cisneros-Zevallos, L.A.; Krochta, J.M.; Saltveit, M.E. Application of casein-lipid edible film emulsions to reduce white blush on minimally processed carrots. Postharvest Biol. Technol. 1994, 4, 319–329. [Google Scholar] [CrossRef]
- Yangılar, F.; Oğuzhan Yıldız, P. Casein/natamycin edible films efficiency for controlling mould growth and on microbiological, chemical and sensory properties during the ripening of Kashar cheese. J. Sci. Food Agric. 2016, 96, 2328–2336. [Google Scholar] [CrossRef] [PubMed]
- Avena-Bustillos, R.J.; Krochta, J.M. Water Vapor Permeability of Caseinate-Based Edible Films as Affected by pH, Calcium Crosslinking and Lipid Content. J. Food Sci. 1993, 58, 904–907. [Google Scholar] [CrossRef]
- Chick, J.; Hernandez, R.J. Physical, Thermal, and Barrier Characterization of Casein-Wax-Based Edible Films. J. Food Sci. 2002, 67, 1073–1079. [Google Scholar] [CrossRef]
- Chevalier, E.; Chaabani, A.; Assezat, G.; Prochazka, F.; Oulahal, N. Casein/wax blend extrusion for production of edible films as carriers of potassium sorbate—A comparative study of waxes and potassium sorbate effect. Food Packag. Shelf Life 2018, 16, 41–50. [Google Scholar] [CrossRef]
- Yadav, J.S.S.; Yan, S.; Pilli, S.; Kumar, L.; Tyagi, R.D.; Surampalli, R.Y. Cheese whey: A potential resource to transform into bioprotein, functional/nutritional proteins and bioactive peptides. Biotechnol. Adv. 2015, 33, 756–774. [Google Scholar] [CrossRef]
- Schmid, M.; Müller, K. Whey protein-based packaging films and coatings. In Whey Proteins; Elsevier: Amsterdam, The Netherlands, 2019; pp. 407–437. [Google Scholar]
- de Castro, R.J.S.; Domingues, M.A.F.; Ohara, A.; Okuro, P.K.; dos Santos, J.G.; Brexó, R.P.; Sato, H.H. Whey protein as a key component in food systems: Physicochemical properties, production technologies and applications. Food Struct. 2017, 14, 17–29. [Google Scholar] [CrossRef]
- Kandasamy, S.; Yoo, J.; Yun, J.; Kang, H.-B.; Seol, K.-H.; Kim, H.-W.; Ham, J.-S. Application of Whey Protein-Based Edible Films and Coatings in Food Industries: An Updated Overview. Coatings 2021, 11, 1056. [Google Scholar] [CrossRef]
- Di Pierro, P.; Mariniello, L.; Giosafatto, V.L.; Esposito, M.; Sabbah, M.; Porta, R. Chapter 13—Dairy Whey Protein-Based Edible Films and Coatings for Food Preservation. In Food Packaging and Preservation; Grumezescu, A.M., Holban, A.M., Eds.; Academic Press: Cambridge, MA, USA, 2018; pp. 439–456. [Google Scholar]
- Ramos, Ó.L.; Fernandes, J.C.; Silva, S.I.; Pintado, M.E.; Malcata, F.X. Edible Films and Coatings from Whey Proteins: A Review on Formulation, and on Mechanical and Bioactive Properties. Crit. Rev. Food Sci. Nutr. 2012, 52, 533–552. [Google Scholar] [CrossRef]
- Galus, S.; Lenart, A. Optical, mechanical, and moisture sorption properties of whey protein edible films. J. Food Process Eng. 2019, 42, e13245. [Google Scholar] [CrossRef]
- Galus, S.; Kadzińska, J. Whey protein edible films modified with almond and walnut oils. Food Hydrocoll. 2016, 52, 78–86. [Google Scholar] [CrossRef]
- Gounga, M.E.; Xu, S.-Y.; Wang, Z. Whey protein isolate-based edible films as affected by protein concentration, glycerol ratio and pullulan addition in film formation. J. Food Eng. 2007, 83, 521–530. [Google Scholar] [CrossRef]
- Galus, S.; Kadzińska, J. Moisture Sensitivity, Optical, Mechanical and Structural Properties of Whey Protein-Based Edible Films Incorporated with Rapeseed Oil. Food Technol. Biotechnol. 2016, 54, 78–89. [Google Scholar] [CrossRef] [PubMed]
- Dangaran, K.; Tomasula, P.M.; Qi, P. Structure and Function of Protein-Based Edible Films and Coatings. In Edible Films and Coatings for Food Applications; Huber, K.C., Embuscado, M.E., Eds.; Springer New York: New York, NY, USA, 2009; pp. 25–56. [Google Scholar]
- Linsenmayer, T.F. Collagen. In Cell Biology of Extracellular Matrix: Second Edition; Hay, E.D., Ed.; Springer US: Boston, MA, USA, 1991; pp. 7–44. [Google Scholar]
- Brodsky, B.; Ramshaw, J.A. The collagen triple-helix structure. Matrix Biol. 1997, 15, 545–554. [Google Scholar] [CrossRef]
- Deiber, J.A.; Peirotti, M.B.; Ottone, M.L. Rheological characterization of edible films made from collagen colloidal particle suspensions. Food Hydrocoll. 2011, 25, 1382–1392. [Google Scholar] [CrossRef]
- O’Sullivan, A.; Shaw, N.B.; Murphy, S.C.; van de Vis, J.W.; van Pelt-Heerschap, H.; Kerry, J.P. Extraction of Collagen from Fish Skins and Its Use in the Manufacture of Biopolymer Films. J. Aquat. Food Prod. Technol. 2006, 15, 21–32. [Google Scholar] [CrossRef]
- Zhao, R.; Guan, W.; Zhou, X.; Lao, M.; Cai, L. The physiochemical and preservation properties of anthocyanidin/chitosan nanocomposite-based edible films containing cinnamon-perilla essential oil pickering nanoemulsions. LWT 2022, 153, 112506. [Google Scholar] [CrossRef]
- Song, D.-H.; Hoa, V.B.; Kim, H.W.; Khang, S.M.; Cho, S.-H.; Ham, J.-S.; Seol, K.-H. Edible Films on Meat and Meat Products. Coatings 2021, 11, 1344. [Google Scholar] [CrossRef]
- Fadini, A.L.; Rocha, F.S.; Alvim, I.D.; Sadahira, M.S.; Queiroz, M.B.; Alves, R.M.V.; Silva, L.B. Mechanical properties and water vapour permeability of hydrolysed collagen–cocoa butter edible films plasticised with sucrose. Food Hydrocoll. 2013, 30, 625–631. [Google Scholar] [CrossRef]
- Wang, Z.; Hu, S.; Wang, H. Scale-Up Preparation and Characterization of Collagen/Sodium Alginate Blend Films. J. Food Qual. 2017, 2017, 4954259. [Google Scholar] [CrossRef]
- Ma, D.; Jiang, Y.; Ahmed, S.; Qin, W.; Liu, Y. Antilisterial and physical properties of polysaccharide-collagen films embedded with cell-free supernatant of Lactococcus lactis. Int. J. Biol. Macromol. 2020, 145, 1031–1038. [Google Scholar] [CrossRef]
- Marangoni Júnior, L.; Rodrigues, P.R.; da Silva, R.G.; Vieira, R.P.; Alves, R.M.V. Sustainable Packaging Films Composed of Sodium Alginate and Hydrolyzed Collagen: Preparation and Characterization. Food Bioprocess Technol. 2021, 14, 2336–2346. [Google Scholar] [CrossRef]
- Wu, X.; Luo, Y.; Liu, Q.; Jiang, S.; Mu, G. Improved structure-stability and packaging characters of crosslinked collagen fiber-based film with casein, keratin and SPI. J. Sci. Food Agric. 2019, 99, 4942–4951. [Google Scholar] [CrossRef]
- Jiang, Y.; Lan, W.; Sameen, D.E.; Ahmed, S.; Qin, W.; Zhang, Q.; Chen, H.; Dai, J.; He, L.; Liu, Y. Preparation and characterization of grass carp collagen-chitosan-lemon essential oil composite films for application as food packaging. Int. J. Biol. Macromol. 2020, 160, 340–351. [Google Scholar] [CrossRef]
- Poppe, J. Gelatin. In Thickening and Gelling Agents for Food; Springer: Berlin/Heidelberg, Germany, 1992; pp. 98–123. [Google Scholar]
- Keenan, T.R. Gelatin. Kirk-Othmer Encyclopedia of Chemical Technology; J. Wiley & Sons: Hoboken, NJ, USA, 1998; Volume 26, pp. 517–541. [Google Scholar]
- Arvanitoyannis, I.; Psomiadou, E.; Nakayama, A.; Aiba, S.; Yamamoto, N. Edible films made from gelatin, soluble starch and polyols, Part 3. Food Chem. 1997, 60, 593–604. [Google Scholar] [CrossRef]
- Fakhouri, F.M.; Martelli, S.M.; Caon, T.; Velasco, J.I.; Mei, L.H.I. Edible films and coatings based on starch/gelatin: Film properties and effect of coatings on quality of refrigerated Red Crimson grapes. Postharvest Biol. Technol. 2015, 109, 57–64. [Google Scholar] [CrossRef]
- Fakhouri, F.M.; Maria Martelli, S.; Canhadas Bertan, L.; Yamashita, F.; Innocentini Mei, L.H.; Collares Queiroz, F.P. Edible films made from blends of manioc starch and gelatin—Influence of different types of plasticizer and different levels of macromolecules on their properties. LWT 2012, 49, 149–154. [Google Scholar] [CrossRef]
- Sobral, P.J.A.; Menegalli, F.C.; Hubinger, M.D.; Roques, M.A. Mechanical, water vapor barrier and thermal properties of gelatin based edible films. Food Hydrocoll. 2001, 15, 423–432. [Google Scholar] [CrossRef]
- Bonilla, J.; Sobral, P.J.A. Investigation of the physicochemical, antimicrobial and antioxidant properties of gelatin-chitosan edible film mixed with plant ethanolic extracts. Food Biosci. 2016, 16, 17–25. [Google Scholar] [CrossRef]
- Hosseini, S.F.; Rezaei, M.; Zandi, M.; Ghavi, F.F. Preparation and functional properties of fish gelatin–chitosan blend edible films. Food Chem. 2013, 136, 1490–1495. [Google Scholar] [CrossRef] [PubMed]
- Ma, W.; Tang, C.-H.; Yin, S.-W.; Yang, X.-Q.; Wang, Q.; Liu, F.; Wei, Z.-H. Characterization of gelatin-based edible films incorporated with olive oil. Food Res. Int. 2012, 49, 572–579. [Google Scholar] [CrossRef]
- Chambi, H.; Grosso, C. Edible films produced with gelatin and casein cross-linked with transglutaminase. Food Res. Int. 2006, 39, 458–466. [Google Scholar] [CrossRef]
- Buffo, R.A.; Han, J.H. 17—Edible films and coatings from plant origin proteins. In Innovations in Food Packaging; Han, J.H., Ed.; Academic Press: London, UK, 2005; pp. 277–300. [Google Scholar]
- Chiralt, A.; González-Martínez, C.; Vargas, M.; Atarés, L. 18—Edible films and coatings from proteins. In Proteins in Food Processing, 2nd ed.; Yada, R.Y., Ed.; Woodhead Publishing: Sawston, UK, 2018; pp. 477–500. [Google Scholar]
- Brandenburg, A.H.; Weller, C.L.; Testin, R.F. Edible Films and Coatings from Soy Protein. J. Food Sci. 1993, 58, 1086–1089. [Google Scholar] [CrossRef]
- Hill, K.; Höfer, R. Natural fats and oils. Sustainable Solutions for Modern Economies; The Royal Society of Chemistry: London, UK, 2009; pp. 167–237. [Google Scholar]
- Rhim, J.W.; Shellhammer, T.H. 21—Lipid-based edible films and coatings. In Innovations in Food Packaging; Han, J.H., Ed.; Academic Press: London, UK, 2005; pp. 362–383. [Google Scholar]
- Hernandez, E. Edible coatings from lipids and resins. In Edible Coatings and Films to Improve Food Quality; Krochta, J., Nisperos-Carriedo, M., Eds.; CRC Press: Boca Raton, FL, USA, 1994; pp. 279–303. [Google Scholar]
- Debeaufort, F.; Voilley, A. Lipid-Based Edible Films and Coatings. In Edible Films and Coatings for Food Applications; Huber, K.C., Embuscado, M.E., Eds.; Springer: New York, NY, USA, 2009; pp. 135–168. [Google Scholar]
- Gordon, M. Fats and Fatty Foods. In Food Industries Manual; Ranken, M.D., Kill, R.C., Eds.; Springer: Boston, MA, USA, 1993; pp. 288–327. [Google Scholar]
- Duarte, M.; Duarte, R.; Rodrigues, R.; Rodrigues, M. Essential oils and their characteristics. Essential Oils in Food Processing: Chemistry, Safety and Applications; IFT Press: Chicago, IL, USA, 2018; pp. 1–19. [Google Scholar]
- Antunes, M.; Gago, C.; Cavaco, A.; Miguel, M. Edible coatings enriched with essential oils and their compounds for fresh and fresh-cut fruit. Recent Pat. Food Nutr. Agric. 2012, 4, 114–122. [Google Scholar] [CrossRef] [PubMed]
- Bassolé, I.H.N.; Juliani, H.R. Essential oils in combination and their antimicrobial properties. Molecules 2012, 17, 3989–4006. [Google Scholar] [CrossRef] [PubMed]
- Nisar, T.; Wang, Z.-C.; Yang, X.; Tian, Y.; Iqbal, M.; Guo, Y. Characterization of citrus pectin films integrated with clove bud essential oil: Physical, thermal, barrier, antioxidant and antibacterial properties. Int. J. Biol. Macromol. 2018, 106, 670–680. [Google Scholar] [CrossRef]
- Kavoosi, G.; Rahmatollahi, A.; Dadfar, S.M.M.; Purfard, A.M. Effects of essential oil on the water binding capacity, physico-mechanical properties, antioxidant and antibacterial activity of gelatin films. LWT-Food Sci. Technol. 2014, 57, 556–561. [Google Scholar] [CrossRef]
- Aitboulahsen, M.; El Galiou, O.; Laglaoui, A.; Bakkali, M.; Hassani Zerrouk, M. Effect of plasticizer type and essential oils on mechanical, physicochemical, and antimicrobial characteristics of gelatin, starch, and pectin-based films. J. Food Process. Preserv. 2020, 44, e14480. [Google Scholar] [CrossRef]
- Benbettaïeb, N.; Karbowiak, T.; Debeaufort, F. Bioactive edible films for food applications: Influence of the bioactive compounds on film structure and properties. Crit. Rev. Food Sci. Nutr. 2019, 59, 1137–1153. [Google Scholar] [CrossRef]
- Roy, S.; Rhim, J.-W. Genipin-Crosslinked Gelatin/Chitosan-Based Functional Films Incorporated with Rosemary Essential Oil and Quercetin. Materials 2022, 15, 3769. [Google Scholar] [CrossRef]
- Shellhammer, T.; Rumsey, T.; Krochta, J. Viscoelastic properties of edible lipids. J. Food Eng. 1997, 33, 305–320. [Google Scholar] [CrossRef]
- Shit, S.C.; Shah, P.M. Edible polymers: Challenges and opportunities. J. Polym. 2014, 2014, 427529. [Google Scholar] [CrossRef]
- Baldwin, E.A. Surface treatments and edible coatings in food preservation. In Handbook of Food Preservation; CRC Press: Boca Raton, FL, USA, 2020; pp. 507–528. [Google Scholar]
- Wang, D.; Huang, J.; Guo, Z.; Liu, W. Durable mixed edible wax coating with stretching superhydrophobicity. J. Mater. Chem. A 2021, 9, 1495–1499. [Google Scholar] [CrossRef]
- Dhall, R.K. Advances in Edible Coatings for Fresh Fruits and Vegetables: A Review. Crit. Rev. Food Sci. Nutr. 2013, 53, 435–450. [Google Scholar] [CrossRef] [PubMed]
- Hagenmaier, R.D. Wax microemulsion formulations used as fruit coatings. Proc. Fla. State Hortic. Soc. 1998, 111, 251–254. [Google Scholar]
- Hagenmaier, R.D.; Baker, R.A. Edible Coatings from Morpholine-Free Wax Microemulsions. J. Agric. Food Chem. 1997, 45, 349–352. [Google Scholar] [CrossRef]
- Oregel-Zamudio, E.; Angoa-Pérez, M.V.; Oyoque-Salcedo, G.; Aguilar-González, C.N.; Mena-Violante, H.G. Effect of candelilla wax edible coatings combined with biocontrol bacteria on strawberry quality during the shelf-life. Sci. Hortic. 2017, 214, 273–279. [Google Scholar] [CrossRef]
- Baldwin, E.; Burns, J.; Kazokas, W.; Brecht, J.; Hagenmaier, R.; Bender, R.; Pesis, E. Effect of two edible coatings with different permeability characteristics on mango (Mangifera indica L.) ripening during storage. Postharvest Biol. Technol. 1999, 17, 215–226. [Google Scholar] [CrossRef]
- Ruiz-Martínez, J.; Aguirre-Joya, J.A.; Rojas, R.; Vicente, A.; Aguilar-González, M.A.; Rodríguez-Herrera, R.; Alvarez-Perez, O.B.; Torres-León, C.; Aguilar, C.N. Candelilla Wax Edible Coating with Flourensia cernua Bioactives to Prolong the Quality of Tomato Fruits. Foods 2020, 9, 1303. [Google Scholar] [CrossRef]
- De León-Zapata, M.A.; Sáenz-Galindo, A.; Rojas-Molina, R.; Rodríguez-Herrera, R.; Jasso-Cantú, D.; Aguilar, C.N. Edible candelilla wax coating with fermented extract of tarbush improves the shelf life and quality of apples. Food Packag. Shelf Life 2015, 3, 70–75. [Google Scholar] [CrossRef]
- Cruz, R.M.; Krauter, V.; Krauter, S.; Agriopoulou, S.; Weinrich, R.; Herbes, C.; Scholten, P.B.; Uysal-Unalan, I.; Sogut, E.; Kopacic, S.; et al. Bioplastics for Food Packaging: Environmental Impact, Trends and Regulatory Aspects. Foods 2022, 11, 3087. [Google Scholar] [CrossRef]
- Pham, T.T.; Nguyen, L.L.P.; Dam, M.S.; Baranyai, L. Application of Edible Coating in Extension of Fruit Shelf Life: Review. AgriEngineering 2023, 5, 520–536. [Google Scholar] [CrossRef]
- Gupta, I.; Cherwoo, L.; Bhatia, R.; Setia, H. Biopolymers: Implications and application in the food industry. Biocatal. Agric. Biotechnol. 2022, 46, 102534. [Google Scholar] [CrossRef]
- Ding, D. A review about edible food coatings and films. Master’s Thesis, Cornell University, Ithaca, NY, USA, 2021. [Google Scholar]
- Pellicer, E.; Nikolic, D.; Sort, J.; Baró, M.; Zivic, F.; Grujovic, N.; Grujic, R.; Pelemis, S. Advances in Applications of Industrial Biomaterials; Springer International Publishing: Berlin/Heidelberg, Germany, 2017. [Google Scholar]
- Ragitha, V.M.; Edison, L.K. Safety Issues, Environmental Impacts, and Health Effects of Biopolymers. In Handbook of Biopolymers; Springer Nature: Singapore, 2022; pp. 1–27. [Google Scholar]

| Polysaccharides Based Edible Films and Coatings | |||
|---|---|---|---|
| Films/Coatings Material | Uses | Properties | References |
| Cellulose and cellulose derivatives | Edible films/coatings | Provide strong adhesion between fiber and cellulosic interface | [94] |
| Plant-based essential oils encapsulated cellulose acetate, cellulose acetate propionate, and cellulose acetate butyrate | Edible films/coatings | Fragrance and air freshening effects | [97] |
| Oils embedded in cellulose esters | Edible films | Enhance the physicomechanical properties and plasticizing effects; minimize moisture loss and improve the water and gas barrier | [98,99] |
| Cellulose esters/acyl groups with C2 to C18 | Edible films | Enhance the water and oxygen barrier properties and hydrophobicity | [101] |
| CMC/Steric acid | Edible films | Improve the water barrier. The water vapor transmittance rate (WVTR) significantly dropped with loading more steric acid to CMC | [102] |
| Cellulose esters/chitosan or silver nanoparticles | edible films and coatings | Antibacterial properties | [112,113] |
| Ginger and olive plant oils/bacterial cellulose/CMC composites | Edible coatings | Antibacterial properties | [114] |
| Rice starch/ι-carrageenan/stearic acids | Edible films | Improved mechanical and WVTR properties | [121,122] |
| Pearl millet starch/carrageenan gum/glycerol | Edible films | Reduced the water vapor permeability and mechanical properties | [123] |
| Carrageenan/rice starch hybrids | Transparent, stable films and coatings | Enhanced UV protection, oxygen barrier, and hydrophobic characteristics | [124] |
| iota carrageenan/glycerol plasticizer/glycerol monostearate surfactant/fat | Edible films | Improved surface properties | [125] |
| κ-carrageenan/ι-carrageenan/alginate blends | Edible films | Improved optical, barrier, and tensile performances | [126] |
| I-carrageenan/sodium alginate blends | Edible films | Good mechanical characteristics with emulsion stabilizers. | [127] |
| Starch/agar-maltodextrin blends | Edible films | improved barrier properties due to extensive hydrogen bonding and hydrophobic aggregations | [132] |
| Agar/essential oils | Edible films/coatings | Improved mechanical and water barrier properties with antimicrobial and antioxidant activities | [136] |
| Agar-based composites/nanoparticles/natural active ingredients | Edible films/coatings | Improve physico-mechanical, thermal, and antioxidant characteristics | [137] |
| Pectin/oregano essential oils | Edible films/coatings | Improve antimicrobial properties against food-related micro-organisms | [143] |
| Pectin extracted as a byproduct of Hibiscus sabdariffa L. | Edible films- strawberry preservation | Excellent film-forming properties | [144] |
| Pectin/whey protein blends cross-linked by transglutaminase | Edible films/coatings for freshly cut fruits and vegetables such as apples, potatoes, and carrots | Improve preservation quality | [145] |
| Candelilla wax/pectin blends | Edible films/coatings | Improve hydrophobicity and barrier performance | [146] |
| Chitosan/30% glycerol | Edible films/coatings-strawberries | Enhanced biological and mechanical protection | [153] |
| Modified diethylaminoethylchitosan | Coatings for Strawberries and bananas | Improve antimicrobial protection against fungi; extended the shelf life of selected fruits | [154] |
| Biguanide modified chitosan/alginate blends | Edible films/coatings | Improved thermal and mechanical properties and antibacterial activity against gram (+) and gram (−) bacteria; reduced WVTR | [155] |
| Multilayered emulsion composites of chitosan/beeswax crosslinked with tripolyphosphate | Edible films | Reduced WVTR and adequate mechanical properties | [156] |
| Different ratios of chitosan | Coatings-Sweet cherries | Effectively prevented moisture losses at 20 °C and improved shelf life by reducing microbial growth | [157] |
| Gum arabic/chitosan/cinnamon essential oil | Edible films | Improved WVTR performance and low mechanical properties | [163] |
| Gum arabic/starch/glycerol and sorbitol | Edible coatings for fruits | Effectiveness against moisture loss by 30%, preserving firmness, facilitating respiration, and delaying the ripening process | [165] |
| Guar gum and tara gum | Edible films and coatings | Tara gums have poor mechanical and barrier performance and require improvements using plasticizers, including glycerol | [169] |
| Guar gum and pea starch incorporated with natural antimicrobial agents | Edible films | Changes in mechanical and antimicrobial properties following a concentration dependent trend | [177] |
| Basil seed gum/oregano essential oils | Active edible films and coatings | Mechanical, antioxidant, and antimicrobial properties; enhanced physicomechanical properties | [180] |
| Basil seed gum hydrocolloids/glycerol | Improved physical, mechanical, microstructural, and thermal characteristics | [181] | |
| Gellan gum/aloe vera gel | Edible films; for active food packaging | Improved mechanical properties | [186,187] |
| Gellan gum/proteins | Edible films and coatings | Excellent mechanical properties and barrier performance. | [188] |
| Starch | Edible films and coatings | Mechanical, oil, and oxygen barrier properties | [193] |
| Modified starch | Edible films | Enhances the physicomechanical characteristics. However, due to its high hydrophilicity, antioxidant, antimicrobial, and other food additive agents are incorporated into the starch matrix. | [194] |
| Cassava starch/glycerol, carnauba wax/stearic acid | Edible coatings-fresh-cut fruits and vegetables | Prolonging shelf life | [195] |
| Starch/chitosan blends | Edible films | Reduced aerobic mesophilic and psychrophilic cell counts while maintaining pH and weight loss in refrigerated storage, extending product life | [153] |
| Corn starch/Sunflower oil | Edible films and coatings | Improvements in mechanical and water barrier properties | [198] |
| Starch/glycerol/Tween 20/Spam 80/soy lecithin | Edible films and coatings | High mechanical and water barrier properties. | [199] |
| Highly carboxymethylated starch (HCMS)/sorbitol, xylitol/mannitol/glycerol | Edible films | Reduced WVTR and decreased solubility with increasing plasticizer concentration | [202,203,204] |
| Non-conventional arrowroot starch | Edible films and coatings-Plum packaging | Increase in WVP from 2.20 to 3.68 g mm/m2 day kPa, moisture content from 3.22% to 7.95%, and a decrease in solubility in water from 22.45% to 13.89%, delivering extended post-harvesting preservation up to 35 days | [205] |
| Corn starch methylcellulose/Glycerol | Edible films and coating | of corn starch methylcellulose and glycerol edible films and coating | [206] |
| Protein-Based Edible Films and Coatings | |||
| Films/Coatings material | Uses | Properties | References |
| Casein | Edible films and coatings | Processing convenience of emulsion preparation in amphipathic dispersion systems. | [215,216,217] |
| Provide good barrier to gas permeation and nutritional value. | [219,220,221,222] | ||
| Casein and beeswax blends | Edible films and coatings from blends | Exhibit good bacteriostatic properties, inhibiting E. coli growth. | [224,225,226,227] |
| Controlled efficiently mold growth and microbial activity | |||
| Whey protein | Edible films and coatings Edible blends | For emulsifying, thickening agent, gelling agent, foaming, and water-binding. | [229] |
| Inherit good mechanical, odorless, flexible, and transparent characteristics with moderate water vapor permeability and excellent oxygen gas barrier. | [230] | ||
| For microbial growth reduction, extending shelf life, and minimizing moisture loss and spoilage | [231,232] | ||
| Collagen | Edible films and coatings Blends for edible films and coatings | For excellent mechanical and oxygen-carbon dioxide barrier properties. | [238] |
| Have proven acceptable films with mechanical, optical properties tensile strength (Young’s modulus, elongation) and WVR | [242,243] | ||
| Gelatin | Edible films and coatings | For improved transparency, mechanical properties, and satisfactory WVTR | [253,254] |
| Gelatin/Chitosan blends | Edible films and coatings | Antioxidant and antimicrobial edible films and coatings | [257] |
| Soy and wheat proteins | Edible films and coatings | For appearance, good tensile strength, and low WVTR of soy hydrocolloid films. Can be enhanced with alkaline treatments | [262,263] |
| Lipids | Sources | Reference |
|---|---|---|
| Fats, oils, shortening and margarine | (a) Various native fats and oil from animals, vegetables, and seeds Such as butter from dairy, lard, sunflower, mustered, olive, almond, peanut, coconut, palm, cocoa, etc. (b) Fractionated, concentrated, or reconstituted oils and fats, and mono, di and tri-glycerides (c) Hydrogenated or trans-esterified oils: Margarine and shortening | [264] |
| Waxes | (a) Waxes from natural animals, insects and vegetables: Beeswax, carnauba wax, candelilla wax, genuine rice bran wax, and laurel wax (b) Waxes from synthetic sources; paraffin, mineral, microcrystalline, oxidized-non-oxidized polyethylene wax | [265] |
| Natural resins | Resins from natural sources: Asafoetida, Benjoin, Chicle, Guarana, Myrrhe, oblibanum, Opponax, Sandaraque, etc. | [266] |
| Essential oils and liquorices | Various essential oils from an extract from flowers, vegetables, animals and fruits; Citrus, rose, ginger, mint, etc. | [267] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liyanapathiranage, A.; Dassanayake, R.S.; Gamage, A.; Karri, R.R.; Manamperi, A.; Evon, P.; Jayakodi, Y.; Madhujith, T.; Merah, O. Recent Developments in Edible Films and Coatings for Fruits and Vegetables. Coatings 2023, 13, 1177. https://doi.org/10.3390/coatings13071177
Liyanapathiranage A, Dassanayake RS, Gamage A, Karri RR, Manamperi A, Evon P, Jayakodi Y, Madhujith T, Merah O. Recent Developments in Edible Films and Coatings for Fruits and Vegetables. Coatings. 2023; 13(7):1177. https://doi.org/10.3390/coatings13071177
Chicago/Turabian StyleLiyanapathiranage, Anuradhi, Rohan S. Dassanayake, Ashoka Gamage, Rama Rao Karri, Asanga Manamperi, Philippe Evon, Yasasvi Jayakodi, Terrence Madhujith, and Othmane Merah. 2023. "Recent Developments in Edible Films and Coatings for Fruits and Vegetables" Coatings 13, no. 7: 1177. https://doi.org/10.3390/coatings13071177
APA StyleLiyanapathiranage, A., Dassanayake, R. S., Gamage, A., Karri, R. R., Manamperi, A., Evon, P., Jayakodi, Y., Madhujith, T., & Merah, O. (2023). Recent Developments in Edible Films and Coatings for Fruits and Vegetables. Coatings, 13(7), 1177. https://doi.org/10.3390/coatings13071177










