Structure and Applications of Pectin in Food, Biomedical, and Pharmaceutical Industry: A Review
Abstract
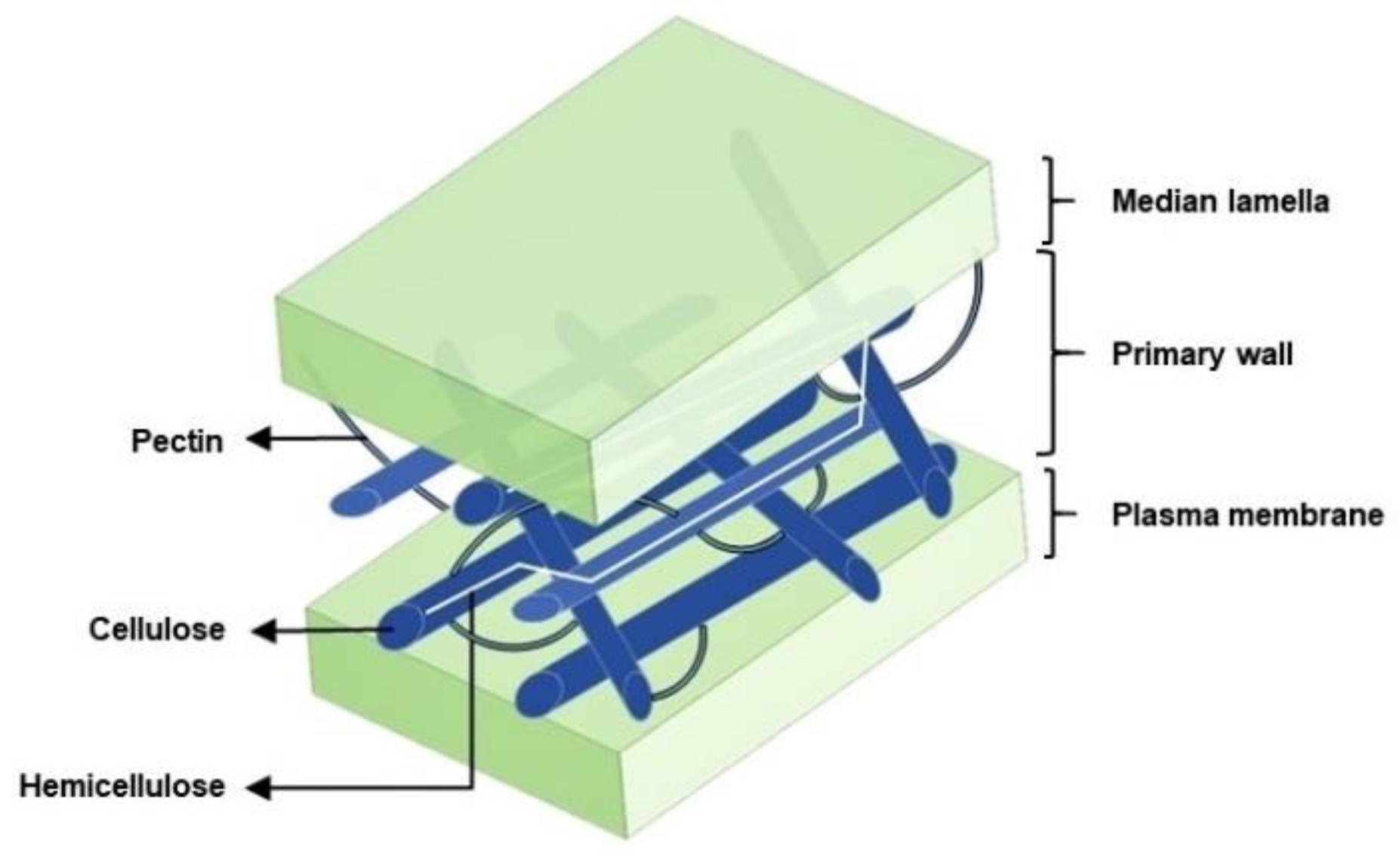
:1. Introduction
2. Pectin Structure
3. Changes in the Structure of Pectin
4. Pectin Extraction
5. Applications
5.1. Pectin in the Food Industry
5.2. Pectin as a Biomedical Product
5.3. Pectin in the Cancer Treatment
5.4. Applications in Other Segments
6. Conjugates of Pectin
7. Prospects, Future Trends and Challenges
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Setiowati, A.D.; Rwigamba, A.; Van der Meeren, P. The influence of degree of methoxylation on the emulsifying and heat stabilizing activity of whey protein-pectin conjugates. Food Hydrocoll. 2019, 96, 54–64. [Google Scholar] [CrossRef]
- González-Henríquez, C.M.; Sarabia-Vallejos, M.A.; Rodriguez-Hernandez, J. Polymers for additive manufacturing and 4D-printing: Materials, methodologies, and biomedical applications. Prog. Polym. Sci. 2019, 94, 57–116. [Google Scholar] [CrossRef]
- Gong, J.; Chen, X.; Tang, T. Recent progress in controlled carbonization of (waste) polymers. Prog. Polym. Sci. 2019, 94, 1–32. [Google Scholar] [CrossRef]
- IcrowdNewswire. Bioplastics & Biopolymer Market by Type, End-Use Industry, Region—Global Forecast for 2025. Available online: https://www.marketsandmarkets.com/Market-Reports/biopolymers-bioplastics-market-88795240.html?gclid=EAIaIQobChMIpMSBxaKH8gIVdGxvBB0kVQWqEAAYASAAEgIOhfD_BwE (accessed on 6 November 2020).
- Martau, G.A.; Mihai, M.; Vodnar, D.C. The use of chitosan, alginate, and pectin in the biomedical and food sector-biocompatibility, bioadhesiveness, and biodegradability. Polymers 2019, 11, 1837. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- MarketsandMarkets Pectin Market. Available online: https://www.marketsandmarkets.com/Market-Reports/pectin-market-139129149.html (accessed on 24 November 2020).
- Rodsamran, P.; Sothornvit, R. Lime peel pectin integrated with coconut water and lime peel extract as a new bioactive film sachet to retard soybean oil oxidation. Food Hydrocoll. 2019, 97, 105173. [Google Scholar] [CrossRef]
- Freitas, C.M.P.; Costa, A.R.; Rodrigues, F.Á.; Júnior, M.M.J.; Dias, M.M.; Sousa, R.C.S. Optimization of pectin extraction from passion fruit (Passiflora edulis flavicarpa) using the response surface method. Braz. J. Dev. 2020, 6, 25609–25625. [Google Scholar] [CrossRef]
- Yang, J.S.; Mu, T.H.; Ma, M.M. Extraction, structure, and emulsifying properties of pectin from potato pulp. Food Chem. 2018, 244, 197–205. [Google Scholar] [CrossRef]
- Sheldon, R.A. Green chemistry, catalysis and valorization of waste biomass. J. Mol. Catal. A Chem. 2016, 422, 3–12. [Google Scholar] [CrossRef]
- Ma, X.; Chen, W.; Yan, T.; Wang, D.; Hou, F.; Miao, S.; Liu, D. Comparison of citrus pectin and apple pectin in conjugation with soy protein isolate (SPI) under controlled dry-heating conditions. Food Chem. 2020, 309, 125501. [Google Scholar] [CrossRef]
- Wicker, L.; Kim, Y.; Kim, M.J.; Thirkield, B.; Lin, Z.; Jung, J. Pectin as a bioactive polysaccharide extracting tailored function from less. Food Hydrocoll. 2014, 42, 251–259. [Google Scholar] [CrossRef]
- Zhang, W.; Xu, P.; Zhang, H. Pectin in cancer therapy: A review. Trends Food Sci. Technol. 2015, 44, 258–271. [Google Scholar] [CrossRef]
- Wang, W.; Chen, W.; Zou, M.; Lv, R.; Wang, D.; Hou, F.; Feng, H.; Ma, X.; Zhong, J.; Ding, T.; et al. Applications of power ultrasound in oriented modification and degradation of pectin: A review. J. Food Eng. 2018, 234, 98–107. [Google Scholar] [CrossRef]
- Ngouémazong, E.D.; Christiaens, S.; Shpigelman, A.; Van Loey, A.; Hendrickx, M. The emulsifying and emulsion-stabilizing properties of pectin: A review. Compr. Rev. Food Sci. Food Saf. 2015, 14, 705–718. [Google Scholar] [CrossRef]
- Freitas, C.M.P.; Sousa, R.C.S.; Dias, M.M.; Coimbra, J.S. Extraction of pectin from passion fruit peel. Food Eng. Rev. 2020, 12, 460–472. [Google Scholar] [CrossRef]
- Munarin, F.; Tanzi, M.C.; Petrini, P. Advances in biomedical applications of pectin gels. Int. J. Biol. Macromol. 2012, 51, 681–689. [Google Scholar] [CrossRef] [PubMed]
- López-Mata, M.A.; Gastelum-Cabrera, M.; Valbuena-Gregorio, E.; Zamudio-Flores, P.B.; Burruel-Ibarra, S.E.; Morales-Figueroa, G.G.; Quihui-Cota, L.; Juárez-Onofre, J.E. Physicochemical properties of novel pectin/Aloe gel membranes. Iran. Polym. J. Engl. Ed. 2018, 27, 545–553. [Google Scholar] [CrossRef]
- Ouyang, J.; Yang, M.; Gong, T.; Ou, J.; Tan, Y.; Zhang, Z.; Li, S. Doxorubicin-loading core-shell pectin nanocell: A novel nanovehicle for anticancer agent delivery with multidrug resistance reversal. PLoS ONE 2020, 15, e235090. [Google Scholar] [CrossRef] [PubMed]
- Badaró, A.T.; Garcia-Martin, J.F.; López-Barrera, M. del C.; Barbin, D.F.; Alvarez-Mateos, P. Determination of pectin content in orange peels by near infrared hyperspectral imaging. Food Chem. 2020, 323, 126861. [Google Scholar] [CrossRef] [PubMed]
- Grassino, A.N.; Halambek, J.; Djaković, S.; Rimac Brnčić, S.; Dent, M.; Grabarić, Z. Utilization of tomato peel waste from canning factory as a potential source for pectin production and application as tin corrosion inhibitor. Food Hydrocoll. 2016, 52, 265–274. [Google Scholar] [CrossRef]
- Haas, K.T.; Wightman, R.; Meyerowitz, E.M.; Peaucelle, A. Pectin homogalacturonan nanofilament expansion drives morphogenesis in plant epidermal cells. Science 2020, 367, 1003–1007. [Google Scholar] [CrossRef]
- Raven, P.H.; Evert, R.F.; Eichhorn, S.E. Plant Biology, 8th ed.; Freeman and Co.: New York, NY, USA, 2014; ISBN 9788527723831. [Google Scholar]
- Yang, Y.; Wang, Z.; Hu, D.; Xiao, K.; Wu, J.Y. Efficient extraction of pectin from sisal waste by combined enzymatic and ultrasonic process. Food Hydrocoll. 2018, 79, 189–196. [Google Scholar] [CrossRef]
- Maxwell, E.G.; Belshaw, N.J.; Waldron, K.W.; Morris, V.J. Pectin an emerging new bioactive food polysaccharide. Trends Food Sci. Technol. 2012, 24, 64–73. [Google Scholar] [CrossRef]
- Marenda, F.R.B.; Mattioda, F.; Demiate, I.M.; de Francisco, A.; de Oliveira Petkowicz, C.L.; Canteri, M.H.G.; de Mello Castanho Amboni, R.D. Advances in studies using vegetable wastes to obtain pectic substances: A review. J. Polym. Environ. 2019, 27, 549–560. [Google Scholar] [CrossRef]
- Mohnen, D. Pectin structure and biosynthesis. Curr. Opin. Plant Biol. 2008, 11, 266–277. [Google Scholar] [CrossRef]
- Wang, D.; Yeats, T.H.; Uluisik, S.; Rose, J.K.C.; Seymour, G.B. Fruit softening: Revisiting the role of pectin. Trends Plant Sci. 2018, 23, 302–310. [Google Scholar] [CrossRef]
- Ciriminna, R.; Chavarría-Hernández, N.; Hernández, A.I.R.; Pagliaro, M. Pectin: A new perspective from the biorefinery standpoint. Biofuels Bioprod. Biorefin. 2015, 9, 368–377. [Google Scholar] [CrossRef]
- Cho, E.H.; Jung, H.T.; Lee, B.H.; Kim, H.S.; Rhee, J.K.; Yoo, S.H. Green process development for apple-peel pectin production by organic acid extraction. Carbohydr. Polym. 2019, 204, 97–103. [Google Scholar] [CrossRef]
- Han, Y.; Yu, M.; Wang, L. Preparation and characterization of antioxidant soy protein isolate films incorporating licorice residue extract. Food Hydrocoll. 2017, 75, 1–9. [Google Scholar] [CrossRef]
- Chen, H.; Qiu, S.; Gan, J.; Liu, Y.; Zhu, Q.; Yin, L. New insights into the functionality of protein to the emulsifying properties of sugar beet pectin. Food Hydrocoll. 2016, 57, 262–270. [Google Scholar] [CrossRef]
- Bichara, L.C.; Alvarez, P.E.; Fiori Bimbi, M.V.; Vaca, H.; Gervasi, C.; Brandán, S.A. Structural and spectroscopic study of a pectin isolated from citrus peel by using FTIR and FT-Raman spectra and DFT calculations. Infrared Phys. Technol. 2016, 76, 315–327. [Google Scholar] [CrossRef]
- Cardoso, S.M.; Silva, A.M.S.; Coimbra, M.A. Structural characterisation of the olive pomace pectic polysaccharide arabinan side chains. Carbohydr. Res. 2002, 337, 917–924. [Google Scholar] [CrossRef]
- Goubet, F.; Ström, A.; Dupree, P.; Williams, M.A.K. An investigation of pectin methylesterification patterns by two independent methods: Capillary electrophoresis and polysaccharide analysis using carbohydrate gel electrophoresis. Carbohydr. Res. 2005, 340, 1193–1199. [Google Scholar] [CrossRef]
- Ishii, T.; Ichita, J.; Matsue, H.; Ono, H.; Maeda, I. Fluorescent labeling of pectic oligosaccharides with 2-aminobenzamide and enzyme assay for pectin. Carbohydr. Res. 2002, 337, 1023–1032. [Google Scholar] [CrossRef]
- Muñoz-Almagro, N.; Montilla, A.; Moreno, F.J.; Villamiel, M. Modification of citrus and apple pectin by power ultrasound: Effects of acid and enzymatic treatment. Ultrason. Sonochem. 2017, 38, 807–819. [Google Scholar] [CrossRef] [Green Version]
- Ogutu, F.O.; Mu, T.H. Ultrasonic degradation of sweet potato pectin and its antioxidant activity. Ultrason. Sonochem. 2017, 38, 726–734. [Google Scholar] [CrossRef] [PubMed]
- Edashige, Y.; Murakami, N.; Tsujita, T. Inhibitory effect of pectin from the segment membrane of citrus fruits on lipase activity. J. Nutr. Sci. Vitaminol. 2008, 54, 409–415. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Celus, M.; Salvia-Trujillo, L.; Kyomugasho, C.; Maes, I.; Van Loey, A.M.; Grauwet, T.; Hendrickx, M.E. Structurally modified pectin for targeted lipid antioxidant capacity in linseed/sunflower oil-in-water emulsions. Food Chem. 2018, 241, 86–96. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Liu, W.; Liu, C.M.; Li, T.; Liang, R.H.; Luo, S.J. Pectin modifications: A review. Crit. Rev. Food Sci. Nutr. 2015, 55, 1684–1698. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Ye, X.; Ding, T.; Sun, X.; Xu, Y.; Liu, D. Ultrasound effects on the degradation kinetics, structure and rheological properties of apple pectin. Ultrason. Sonochem. 2013, 20, 222–231. [Google Scholar] [CrossRef]
- Chen, T.T.; Zhang, Z.H.; Wang, Z.W.; Chen, Z.L.; Ma, H.; Yan, J.K. Effects of ultrasound modification at different frequency modes on physicochemical, structural, functional, and biological properties of citrus pectin. Food Hydrocoll. 2021, 113, 106484. [Google Scholar] [CrossRef]
- Zhi, Z.; Chen, J.; Li, S.; Wang, W.; Huang, R.; Liu, D.; DIng, T.; Linhardt, R.J.; Chen, S.; Ye, X. Fast preparation of RG-I enriched ultra-low molecular weight pectin by an ultrasound accelerated fenton process. Sci. Rep. 2017, 7, 541. [Google Scholar] [CrossRef] [Green Version]
- Ma, X.; Zhang, L.; Wang, W.; Zou, M.; Ding, T.; Ye, X.; Liu, D. Synergistic effect and mechanisms of combining ultrasound and pectinase on pectin hydrolysis. Food Bioprocess Technol. 2016, 9, 1249–1257. [Google Scholar] [CrossRef]
- Wang, W.; Feng, Y.; Chen, W.; Wang, Y.; Wilder, G.; Liu, D.; Yin, Y. Ultrasonic modification of pectin for enhanced 2-furfurylthiol encapsulation: Process optimization and mechanisms. J. Sci. Food Agric. 2020, 100, 110–118. [Google Scholar] [CrossRef]
- Qiu, W.Y.; Cai, W.D.; Wang, M.; Yan, J.K. Effect of ultrasonic intensity on the conformational changes in citrus pectin under ultrasonic processing. Food Chem. 2019, 297, 125021. [Google Scholar] [CrossRef] [PubMed]
- Hu, W.; Chen, S.; Wu, D.; Zheng, J.; Ye, X. Ultrasonic-assisted citrus pectin modification in the bicarbonate-activated hydrogen peroxide system: Chemical and microstructural analysis. Ultrason. Sonochem. 2019, 58, 104576. [Google Scholar] [CrossRef] [PubMed]
- Fan, X.; Jiang, W.; Gong, H.; Yang, Y.; Zhang, A.; Liu, H.; Cao, J.; Guo, F.; Cui, K. Cell wall polysaccharides degradation and ultrastructure modification of apricot during storage at a near freezing temperature. Food Chem. 2019, 300, 125194. [Google Scholar] [CrossRef] [PubMed]
- Zheng, J.; Zeng, R.; Kan, J.; Zhang, F. Effects of ultrasonic treatment on gel rheological properties and gel formation of high-methoxyl pectin. J. Food Eng. 2018, 231, 83–90. [Google Scholar] [CrossRef]
- Rojas, R.; Alvarez-Pérez, O.B.; Contreras-Esquivel, J.C.; Vicente, A.; Flores, A.; Sandoval, J.; Aguilar, C.N. Valorisation of mango peels: Extraction of pectin and antioxidant and antifungal polyphenols. Waste Biomass Valoriz. 2018, 11, 89–98. [Google Scholar] [CrossRef] [Green Version]
- Colodel, C.; Vriesmann, L.C.; Teófilo, R.F.; Petkowicz, C.L.O. Optimization of acid-extraction of pectic fraction from grape (Vitis vinifera cv. Chardonnay) pomace, a Winery Waste. Int. J. Biol. Macromol. 2020, 161, 204–213. [Google Scholar] [CrossRef]
- Xu, S.Y.; Liu, J.P.; Huang, X.; Du, L.P.; Shi, F.L.; Dong, R.; Huang, X.T.; Zheng, K.; Liu, Y.; Cheong, K.L. Ultrasonic-microwave assisted extraction, characterization and biological activity of pectin from jackfruit peel. LWT Food Sci. Technol. 2018, 90, 577–582. [Google Scholar] [CrossRef]
- Karbuz, P.; Tugrul, N. Microwave and ultrasound assisted extraction of pectin from various fruits peel. J. Food Sci. Technol. 2020, 58, 641–650. [Google Scholar] [CrossRef] [PubMed]
- Raji, Z.; Khodaiyan, F.; Rezaei, K.; Kiani, H.; Hosseini, S.S. Extraction optimization and physicochemical properties of pectin from melon peel. Int. J. Biol. Macromol. 2017, 98, 709–716. [Google Scholar] [CrossRef]
- Petkowicz, C.L.O.; Vriesmann, L.C.; Williams, P.A. Pectins from food waste: Extraction, characterization and properties of watermelon rind pectin. Food Hydrocoll. 2017, 65, 57–67. [Google Scholar] [CrossRef]
- Reichembach, L.H.; Petkowicz, C.L.O. Extraction and characterization of a pectin from coffee (Coffea arabica L.) pulp with gelling properties. Carbohydr. Polym. 2020, 245, 116473. [Google Scholar] [CrossRef]
- Chan, S.Y.; Choo, W.S. Effect of extraction conditions on the yield and chemical properties of pectin from cocoa husks. Food Chem. 2013, 141, 3752–3758. [Google Scholar] [CrossRef]
- Swamy, G.J.; Muthukumarappan, K. Optimization of continuous and intermittent microwave extraction of pectin from banana peels. Food Chem. 2017, 220, 108–114. [Google Scholar] [CrossRef] [PubMed]
- Moorthy, I.G.; Maran, J.P.; Surya, S.M.; Naganyashree, S.; Shivamathi, C.S. Response surface optimization of ultrasound assisted extraction of pectin from pomegranate peel. Int. J. Biol. Macromol. 2015, 72, 1323–1328. [Google Scholar] [CrossRef]
- Maran, J.P. Statistical optimization of aqueous extraction of pectin from waste durian rinds. Int. J. Biol. Macromol. 2015, 73, 92–98. [Google Scholar] [CrossRef] [PubMed]
- Mao, Y.; Millett, R.; Lee, C.S.; Yakubov, G.; Harding, S.E.; Binner, E. Investigating the influence of pectin content and structure on its functionality in bio-flocculant extracted from okra. Carbohydr. Polym. 2020, 241, 116414. [Google Scholar] [CrossRef] [PubMed]
- Lalnunthari, C.; Devi, L.M.; Badwaik, L.S. Extraction of protein and pectin from pumpkin industry by-products and their utilization for developing edible film. J. Food Sci. Technol. 2020, 57, 1807–1816. [Google Scholar] [CrossRef]
- Koubala, B.B.; Christiaens, S.; Kansci, G.; Van Loey, A.M.; Hendrickx, M.E. Isolation and structural characterisation of papaya peel pectin. Food Res. Int. 2014, 55, 215–221. [Google Scholar] [CrossRef]
- Sun, D.; Chen, X.; Zhu, C. Physicochemical properties and antioxidant activity of pectin from hawthorn wine pomace: A comparison of different extraction methods. Int. J. Biol. Macromol. 2020, 158, 1239–1247. [Google Scholar] [CrossRef] [PubMed]
- Lefsih, K.; Giacomazza, D.; Dahmoune, F.; Mangione, M.R.; Bulone, D.; San Biagio, P.L.; Passantino, R.; Costa, M.A.; Guarrasi, V.; Madani, K. Pectin from Opuntia ficus indica: Optimization of microwave-assisted extraction and preliminary characterization. Food Chem. 2017, 221, 91–99. [Google Scholar] [CrossRef]
- Arrutia, F.; Adam, M.; Calvo-Carrascal, M.Á.; Mao, Y.; Binner, E. Development of a continuous-flow system for microwave-assisted extraction of pectin-derived oligosaccharides from food waste. Chem. Eng. J. 2020, 395, 125056. [Google Scholar] [CrossRef]
- Souza, C.G.; Rodrigues, T.H.S.; Silva, L.M.A.; Ribeiro, P.R.V.; Brito, E.S. Sequential extraction of flavonoids and pectin from yellow passion fruit rind using pressurized solvent or ultrasound. J. Sci. Food Agric. 2018, 98, 1362–1368. [Google Scholar] [CrossRef]
- Jafarzadeh-Moghaddam, M.; Shaddel, R.; Peighambardoust, S.H. Sugar beet pectin extracted by ultrasound or conventional heating: A comparison. J. Food Sci. Technol. 2020, 58, 2567–2578. [Google Scholar] [CrossRef] [PubMed]
- Muñoz-Almagro, N.; Valadez-Carmona, L.; Mendiola, J.A.; Ibáñez, E.; Villamiel, M. Structural characterisation of pectin obtained from cacao pod husk. Comparison of conventional and subcritical water extraction. Carbohydr. Polym. 2019, 217, 69–78. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liew, S.Q.; Teoh, W.H.; Tan, C.K.; Yusoff, R.; Ngoh, G.C. Subcritical water extraction of low methoxyl pectin from pomelo (Citrus grandis (L.) Osbeck) peels. Int. J. Biol. Macromol. 2018, 116, 128–135. [Google Scholar] [CrossRef] [PubMed]
- Li, W.J.; Fan, Z.G.; Wu, Y.Y.; Jiang, Z.G.; Shi, R.C. Eco-friendly extraction and physicochemical properties of pectin from jackfruit peel waste with subcritical water. J. Sci. Food Agric. 2019, 99, 5283–5292. [Google Scholar] [CrossRef] [PubMed]
- Shafie, M.H.; Yusof, R.; Gan, C.Y. Deep eutectic solvents (DES) mediated extraction of pectin from Averrhoa bilimbi: Optimization and characterization studies. Carbohydr. Polym. 2019, 216, 303–311. [Google Scholar] [CrossRef]
- Liew, S.Q.; Ngoh, G.C.; Yusoff, R.; Teoh, W.H. Acid and deep eutectic solvent (DES) extraction of pectin from pomelo (Citrus grandis (L.) Osbeck) peels. Biocatal. Agric. Biotechnol. 2018, 13, 1–11. [Google Scholar] [CrossRef]
- Elgharbawy, A.A.M.; Hayyan, A.; Hayyan, M.; Mirghani, M.E.S.; Salleh, H.M.; Rashid, S.N.; Ngoh, G.C.; Liew, S.Q.; Nor, M.R.M.; Yusoff, M.Y.Z.B.M.; et al. Natural deep eutectic solvent-assisted pectin extraction from pomelo peel using sonoreactor. Processes 2019, 7, 416. [Google Scholar] [CrossRef] [Green Version]
- Vorobiex, E.; Lebovka, N. Pulse Eletric Field-Assisted Extraction. In Enhancing Extraction Processes in the Food Industry; CRC Press: Boca Raton, FL, USA, 2016; ISBN 9781439845950. [Google Scholar]
- Su, D.L.; Li, P.J.; Quek, S.Y.; Huang, Z.Q.; Yuan, Y.J.; Li, G.Y.; Shan, Y. Efficient extraction and characterization of pectin from orange peel by a combined surfactant and microwave assisted process. Food Chem. 2019, 286, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.S.; Mu, T.H.; Ma, M.M. Optimization of ultrasound-microwave assisted acid extraction of pectin from potato pulp by response surface methodology and its characterization. Food Chem. 2019, 289, 351–359. [Google Scholar] [CrossRef] [PubMed]
- Gharibzahedi, S.M.T.; Smith, B.; Guo, Y. Ultrasound-microwave assisted extraction of pectin from fig (Ficus carica L.) skin: Optimization, characterization and bioactivity. Carbohydr. Polym. 2019, 222, 114992. [Google Scholar] [CrossRef] [PubMed]
- Adetunji, L.R.; Adekunle, A.; Orsat, V.; Raghavan, V. Advances in the pectin production process using novel extraction techniques: A review. Food Hydrocoll. 2017, 62, 239–250. [Google Scholar] [CrossRef]
- Naqash, F.; Masoodi, F.A.; Rather, S.A.; Wani, S.M.; Gani, A. Emerging concepts in the nutraceutical and functional properties of pectin—A Review. Carbohydr. Polym. 2017, 168, 227–239. [Google Scholar] [CrossRef] [PubMed]
- Espitia, P.J.P.; Du, W.X.; Avena-Bustillos, R.d.J.; Soares, N.d.F.F.; McHugh, T.H. Edible films from pectin: Physical-mechanical and antimicrobial properties: A review. Food Hydrocoll. 2014, 35, 287–296. [Google Scholar] [CrossRef]
- Douglas, T.E.L.; Hempel, U.; Żydek, J.; Vladescu, A.; Pietryga, K.; Kaeswurm, J.A.H.; Buchweitz, M.; Surmenev, R.A.; Surmeneva, M.A.; Cotrut, C.M.; et al. Pectin coatings on titanium alloy scaffolds produced by additive manufacturing: Promotion of human bone marrow stromal cell proliferation. Mater. Lett. 2018, 227, 225–228. [Google Scholar] [CrossRef] [Green Version]
- Jindal, M.; Kumar, V.; Rana, V.; Tiwary, A.K. Aegle marmelos fruit pectin for food and pharmaceuticals: Physico-chemical, rheological and functional performance. Carbohydr. Polym. 2013, 93, 386–394. [Google Scholar] [CrossRef]
- Masuelli, M.A. Viscometric study of pectin. Effect of temperature on the hydrodynamic properties. Int. J. Biol. Macromol. 2011, 48, 286–291. [Google Scholar] [CrossRef]
- Kim, H.W.; Lee, Y.J.; Kim, Y.H.B. Effects of membrane-filtered soy hull pectin and pre-emulsified fiber/oil on chemical and technological properties of low fat and low salt meat emulsions. J. Food Sci. Technol. 2016, 53, 2580–2588. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sakooei-Vayghan, R.; Peighambardoust, S.H.; Hesari, J.; Peressini, D. Effects of osmotic dehydration (with and without sonication) and pectin-based coating pretreatments on functional properties and color of hot-air dried apricot cubes. Food Chem. 2020, 311, 125978. [Google Scholar] [CrossRef]
- Gaona-Sánchez, V.A.; Calderón-Domínguez, G.; Morales-Sánchez, E.; Moreno-Ruiz, L.A.; Terrés-Rojas, E.; de la Salgado-Cruz, M.P.; Escamilla-García, M.; Barrios-Francisco, R. Physicochemical and superficial characterization of a bilayer film of zein and pectin obtained by electrospraying. J. Appl. Polym. Sci. 2021, 138, 1–15. [Google Scholar] [CrossRef]
- Sucheta; Chaturvedi, K.; Sharma, N.; Yadav, S.K. Composite edible coatings from commercial pectin, corn flour and beetroot powder minimize post-harvest decay, reduces ripening and improves sensory liking of tomatoes. Int. J. Biol. Macromol. 2019, 133, 284–293. [Google Scholar] [CrossRef]
- Anastas, P.; Eghbali, N. Green chemistry: Principles and practice. Chem. Soc. Rev. 2010, 39, 301–312. [Google Scholar] [CrossRef] [PubMed]
- Souza, V.G.L.; Pires, J.R.A.; Rodrigues, C.; Coelhoso, I.M.; Fernando, A.L. Chitosan composites in packaging industry-current trends and future challenges. Polymers 2020, 12, 417. [Google Scholar] [CrossRef] [Green Version]
- Rivera-Hernández, L.; Chavarría-Hernández, N.; del López Cuellar, M.R.; Martínez-Juárez, V.M.; Rodríguez-Hernández, A.I. Pectin-gellan films intended for active food packaging: Release kinetics of nisin and physico-mechanical characterization. J. Food Sci. Technol. 2020, 58, 2973–2981. [Google Scholar] [CrossRef] [PubMed]
- Norcino, L.B.; Mendes, J.F.; Natarelli, C.V.L.; Manrich, A.; Oliveira, J.E.; Mattoso, L.H.C. Pectin films loaded with copaiba oil nanoemulsions for potential use as bio-based active packaging. Food Hydrocoll. 2020, 106, 105862. [Google Scholar] [CrossRef]
- Muñoz-Almagro, N.; Herrero-Herranz, M.; Guri, S.; Nieves, C.; Antonia, M.; Mar, V. Application of sunflower pectin gels with low glycemic index in the coating of fresh strawberries stored in modified atmospheres running title: Strawberries coated with sunflower pectin and stored in modified atmospheres. J. Sci. Food Agric. 2021. [Google Scholar] [CrossRef] [PubMed]
- Xiong, Y.; Li, S.; Warner, R.D.; Fang, Z. Effect of oregano essential oil and resveratrol nanoemulsion loaded pectin edible coating on the preservation of pork loin in modified atmosphere packaging. Food Control 2020, 114, 107226. [Google Scholar] [CrossRef]
- Eça, K.S.; Machado, M.T.C.; Hubinger, M.D.; Menegalli, F.C. Development of active films from pectin and fruit extracts: Light protection, antioxidant capacity, and compounds stability. J. Food Sci. 2015, 80, C2389–C2396. [Google Scholar] [CrossRef]
- Jridi, M.; Abdelhedi, O.; Salem, A.; Kechaou, H.; Nasri, M.; Menchari, Y. Physicochemical, antioxidant and antibacterial properties of fish gelatin-based edible films enriched with orange peel pectin: Wrapping application. Food Hydrocoll. 2020, 103, 105688. [Google Scholar] [CrossRef]
- Go, E.J.; Song, K.B. Development and characterization of citrus junos pomace pectin films incorporated with rambutan (nephelium lappaceum) peel extract. Coatings 2020, 10, 714. [Google Scholar] [CrossRef]
- Porta, R.; Di Pierro, P.; Sabbah, M.; Regalado-Gonzales, C.; Mariniello, L.; Kadivar, M.; Arabestani, A. Blend films of pectin and bitter vetch (Vicia ervilia) proteins: Properties and effect of transglutaminase. Innov. Food Sci. Emerg. Technol. 2016, 36, 245–251. [Google Scholar] [CrossRef]
- Leo, R.D.; Quartieri, A.; Haghighi, H.; Gigliano, S.; Bedin, E.; Pulvirenti, A. Application of pectin-alginate and pectin-alginate-laurolyl arginate ethyl coatings to eliminate Salmonella enteritidis cross contamination in egg shells. J. Food Saf. 2018, 38, 1–9. [Google Scholar] [CrossRef]
- Muñoz-Labrador, A.; Moreno, R.; Villamiel, M.; Montilla, A. Preparation of citrus pectin gels by power ultrasound and its application as an edible coating in strawberries. J. Sci. Food Agric. 2018, 98, 4866–4875. [Google Scholar] [CrossRef] [Green Version]
- Estrada-Girón, Y.; Cabrera-Díaz, E.; Esparza-Merino, R.M.; Martín-del-Campo, A.; Valencia-Botín, A.J. Innovative edible films and coatings based on red color pectin obtained from the byproducts of Hibiscus sabdariffa L. for strawberry preservation. J. Food Meas. Charact. 2020, 14, 3371–3380. [Google Scholar] [CrossRef]
- Guerreiro, A.C.; Gago, C.M.L.; Faleiro, M.L.; Miguel, M.G.C.; Antunes, M.D.C. The effect of edible coatings on the nutritional quality of ‘Bravo de Esmolfe’ fresh-cut apple through shelf-life. LWT Food Sci. Technol. 2017, 75, 210–219. [Google Scholar] [CrossRef]
- Mendes, J.F.; Norcino, L.B.; Manrich, A.; Pinheiro, A.C.M.; Oliveira, J.E.; Mattoso, L.H.C. Characterization of pectin films integrated with cocoa butter by continuous casting: Physical, thermal and barrier properties. J. Polym. Environ. 2020, 28, 2905–2917. [Google Scholar] [CrossRef]
- Ullah, K.; Sohail, M.; Buabeid, M.A.; Murtaza, G.; Ullah, A.; Rashid, H.; Khan, M.A.; Khan, S.A. Pectin-based (LA-co-MAA) semi-IPNS as a potential biomaterial for colonic delivery of oxaliplatin. Int. J. Pharm. 2019, 569, 118557. [Google Scholar] [CrossRef]
- Sriamornsak, P.; Wattanakorn, N.; Takeuchi, H. Study on the mucoadhesion mechanism of pectin by atomic force microscopy and mucin-particle method. Carbohydr. Polym. 2010, 79, 54–59. [Google Scholar] [CrossRef]
- Tian, L.; Singh, A.; Singh, A.V. Synthesis and characterization of pectin-chitosan conjugate for biomedical application. Int. J. Biol. Macromol. 2020, 153, 533–538. [Google Scholar] [CrossRef]
- Hwang, S.W.; Shin, J.S. Pectin-coated curcumin-chitosan microparticles crosslinked with Mg2+ for delayed drug release in the digestive system. Int. J. Polym. Sci. 2018, 2018, 2071071. [Google Scholar] [CrossRef] [Green Version]
- Bai, F.; Diao, J.; Wang, Y.; Sun, S.; Zhang, H.; Liu, Y.; Wang, Y.; Cao, J. A new water-soluble nanomicelle formed through self-assembly of pectin-curcumin conjugates: Preparation, characterization, and anticancer activity evaluation. J. Agric. Food Chem. 2017, 65, 6840–6847. [Google Scholar] [CrossRef] [PubMed]
- Majzoob, S.; Atyabi, F.; Dorkoosh, F.; Kafedjiiski, K.; Loretz, B.; Bernkop-Schnürch, A. Pectin-cysteine conjugate: Synthesis and in-vitro evaluation of its potential for drug delivery. J. Pharm. Pharmacol. 2006, 58, 1601–1610. [Google Scholar] [CrossRef]
- Zhu, Y.; Yao, Z.; Liu, Y.; Zhang, W.; Geng, L.; Ni, T. Incorporation of ROS-responsive substance P-loaded zeolite imidazolate framework-8 nanoparticles into a Ca2+-cross-linked alginate/pectin hydrogel for wound dressing applications. Int. J. Nanomedicine 2020, 15, 333–346. [Google Scholar] [CrossRef] [Green Version]
- Sarioglu, E.; Arabacioglu Kocaaga, B.; Turan, D.; Batirel, S.; Guner, F.S. Theophylline-loaded pectin-based hydrogels. II. Effect of concentration of initial pectin solution, crosslinker type and cation concentration of external solution on drug release profile. J. Appl. Polym. Sci. 2019, 136, 1–15. [Google Scholar] [CrossRef]
- Shishir, M.R.I.; Karim, N.; Gowd, V.; Xie, J.; Zheng, X.; Chen, W. Pectin-chitosan conjugated nanoliposome as a promising delivery system for neohesperidin: Characterization, release behavior, cellular uptake, and antioxidant property. Food Hydrocoll. 2019, 95, 432–444. [Google Scholar] [CrossRef]
- Tummalapalli, M.; Berthet, M.; Verrier, B.; Deopura, B.L.; Alam, M.S.; Gupta, B. Composite wound dressings of pectin and gelatin with aloe vera and curcumin as bioactive agents. Int. J. Biol. Macromol. 2016, 82, 104–113. [Google Scholar] [CrossRef]
- Cancer Burden Keeps Growing. UN News. Available online: https://canceratlas.cancer.org/the-burden/the-burden-of-cancer/ (accessed on 18 April 2021).
- Sabra, R.; Billa, N.; Roberts, C.J. An augmented delivery of the anticancer agent, curcumin, to the colon. React. Funct. Polym. 2018, 123, 54–60. [Google Scholar] [CrossRef]
- Glinsky, V.V.; Raz, A. Modified citrus pectin anti-metastatic properties: One bullet, multiple targets. Carbohydr. Res. 2009, 344, 1788–1791. [Google Scholar] [CrossRef] [Green Version]
- Maxwell, E.G.; Colquhoun, I.J.; Chau, H.K.; Hotchkiss, A.T.; Waldron, K.W.; Morris, V.J.; Belshaw, N.J. Modified sugar beet pectin induces apoptosis of colon cancer cells via an interaction with the neutral sugar side-chains. Carbohydr. Polym. 2016, 136, 923–929. [Google Scholar] [CrossRef] [PubMed]
- Paharia, A.; Yadav, A.K.; Rai, G.; Jain, S.K.; Pancholi, S.S.; Agrawal, G.P. Eudragit-coated pectin microspheres of 5-Fluorouracil for colon targeting. AAPS PharmSciTech 2007, 8, E87–E93. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Moideen, M.M.J.; Karuppaiyan, K.; Kandhasamy, R.; Seetharaman, S. Skimmed milk powder and pectin decorated solid lipid nanoparticle containing soluble curcumin used for the treatment of colorectal cancer. J. Food Process Eng. 2019, 43, 1–15. [Google Scholar] [CrossRef]
- Cheewatanakornkool, K.; Niratisai, S.; Manchun, S.; Dass, C.R.; Sriamornsak, P. Characterization and in vitro release studies of oral microbeads containing thiolated pectin–doxorubicin conjugates for colorectal cancer treatment. Asian J. Pharm. Sci. 2017, 12, 509–520. [Google Scholar] [CrossRef] [PubMed]
- Katas, H.; Amin, M.C.I.M.; Moideen, N.; Ng, L.Y.; Baharudin, P.A.A.M. Cell growth inhibition effect of DsiRNA vectorised by pectin-coated chitosan-graphene oxide nanocomposites as potential therapy for colon cancer. J. Nanomater. 2017, 2017, 298218. [Google Scholar] [CrossRef] [Green Version]
- Yan, J.K.; Qiu, W.Y.; Wang, Y.Y.; Wu, J.Y. Biocompatible polyelectrolyte complex nanoparticles from lactoferrin and pectin as potential vehicles for antioxidative curcumin. J. Agric. Food Chem. 2017, 65, 5720–5730. [Google Scholar] [CrossRef]
- Ye, P.J.; Huang, C.; Yang, S.; Gao, P.; Li, Z.P.; Tang, S.Y.; Xiang, Y.; Liu, Y.F.; Chen, Y.P.; He, D.X.; et al. Facile fabrication of a novel hybrid nanoparticles by self-assembling based on pectin-doxorubicin conjugates for hepatocellular carcinoma therapy. Artif. Cells Nanomed. Biotechnol. 2018, 46, S661–S670. [Google Scholar] [CrossRef]
- Li, Z.P.; Jiang, M.C.; Chen, B.; Gao, P.; Yang, S.; Liu, Y.F.; Ye, P.J.; He, D.X.; Huang, H.L.; Yu, C.Y. Fabrication and characterization of a novel self-assembling micelle based on chitosan cross-linked pectin-doxorubicin conjugates macromolecular pro-drug for targeted cancer therapy. RSC Adv. 2018, 8, 12004–12016. [Google Scholar] [CrossRef] [Green Version]
- Diao, J.; Bai, F.; Wang, Y.; Han, Q.; Xu, X.; Zhang, H.; Luo, Q.; Wang, Y. Engineering of pectin-dopamine nano-conjugates for carrying ruthenium complex: A potential tool for biomedical applications. J. Inorg. Biochem. 2019, 191, 135–142. [Google Scholar] [CrossRef]
- Sabra, R.; Billa, N.; Roberts, C.J. Cetuximab-conjugated chitosan-pectinate (modified) composite nanoparticles for targeting colon cancer. Int. J. Pharm. 2019, 572, 118775. [Google Scholar] [CrossRef] [PubMed]
- Conti, S.; Vexler, A.; Hagoel, L.; Kalich-Philosoph, L.; Corn, B.W.; Honig, N.; Shtraus, N.; Meir, Y.; Ron, I.; Eliaz, I.; et al. Modified citrus pectin as a potential sensitizer for radiotherapy in prostate cancer. Integr. Cancer Ther. 2018, 17, 1225–1234. [Google Scholar] [CrossRef] [Green Version]
- Ueberall, M.A.; Lorenzl, S.; Lux, E.A.; Voltz, R.; Perelman, M. Efficacy, safety, and tolerability of fentanyl pectin nasal spray in patients with breakthrough cancer pain. J. Pain Res. 2016, 9, 571–585. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Izadi, Z.; Divsalar, A.; Saboury, A.A.; Sawyer, L. β-lactoglobulin–pectin nanoparticle-based oral drug delivery system for potential treatment of colon cancer. Chem. Biol. Drug Des. 2016, 88, 209–216. [Google Scholar] [CrossRef] [PubMed]
- Delphi, L.; Sepehri, H. Apple pectin: A natural source for cancer suppression in 4T1 breast cancer cells in vitro and express p53 in mouse bearing 4T1 cancer tumors, in vivo. Biomed. Pharmacother. 2016, 84, 637–644. [Google Scholar] [CrossRef]
- Nsom, M.V.; Etape, E.P.; Tendo, J.F.; Namond, B.V.; Chongwain, P.T.; Yufanyi, M.D.; William, N. A green and facile approach for synthesis of starch-pectin magnetite nanoparticles and application by removal of methylene blue from textile effluent. J. Nanomater. 2019, 2019, 576135. [Google Scholar] [CrossRef]
- Raghav, S.; Nehra, S.; Kumar, D. Biopolymer scaffold of pectin and alginate for the application of health hazardous fluoride removal studies by equilibrium adsorption, kinetics and thermodynamics. J. Mol. Liq. 2019, 284, 203–214. [Google Scholar] [CrossRef]
- Fiori-Bimbi, M.V.; Alvarez, P.E.; Vaca, H.; Gervasi, C.A. Corrosion inhibition of mild steel in HCL solution by pectin. Corros. Sci. 2015, 92, 192–199. [Google Scholar] [CrossRef]
- Qi, P.X.; Xiao, Y.; Wickham, E.D. Changes in physical, chemical and functional properties of whey protein isolate (WPI) and sugar beet pectin (SBP) conjugates formed by controlled dry-heating. Food Hydrocoll. 2017, 69, 86–96. [Google Scholar] [CrossRef] [Green Version]
- Koch, L.; Emin, M.A.; Schuchmann, H.P. Influence of processing conditions on the formation of whey protein-citrus pectin conjugates in extrusion. J. Food Eng. 2017, 193, 1–9. [Google Scholar] [CrossRef]
- Wefers, D.; Bindereif, B.; Karbstein, H.P.; van der Schaaf, U.S. Whey protein-pectin conjugates: Linking the improved emulsifying properties to molecular and physico-chemical characteristics. Food Hydrocoll. 2018, 85, 257–266. [Google Scholar] [CrossRef]
- Iviglia, G.; Cassinelli, C.; Bollati, D.; Baino, F.; Torre, E.; Morra, M.; Vitale-Brovarone, C. Engineered porous scaffolds for periprosthetic infection prevention. Mater. Sci. Eng. C 2016, 68, 701–715. [Google Scholar] [CrossRef] [PubMed]
- Iviglia, G.; Cassinelli, C.; Torre, E.; Baino, F.; Morra, M.; Vitale-Brovarone, C. Novel bioceramic-reinforced hydrogel for alveolar bone regeneration. Acta Biomater. 2016, 44, 97–109. [Google Scholar] [CrossRef]
- Li, L.; Gao, X.; Liu, J.; Chitrakar, B.; Wang, B.; Wang, Y. Hawthorn pectin: Extraction, function and utilization. Curr. Res. Food Sci. 2021, 4, 429–435. [Google Scholar] [CrossRef] [PubMed]
- Freitas, C.M.P.; Junior, D.B.S.; Martins, R.D.; Dias, M.M.S.; Coimbra, J.S.R.; Sousa, R.C.S. Simulation of ethanol recovery and economic analysis of pectin production on an industrial scale. Bioprocess Biosyst. Eng. 2021. [Google Scholar] [CrossRef]
- Li, D.; Li, J.; Dong, H.; Li, X.; Zhang, J.; Ramaswamy, S.; Xu, F. Pectin in biomedical and drug delivery applications: A review. Int. J. Biol. Macromol. 2021, 185, 49–65. [Google Scholar] [CrossRef]

| Sub-Domains Title | Amount (%) | Structural Composition | Ref |
|---|---|---|---|
| Homogalacturonan (HG) | 65 | Linear homopolymer of GalA partially esterified with methyl esters (α1-4 bonded) at the C-6 position and acetyl esters at the O-2 and/or O-3 positions. | [9,12,13,15,25,27,28] |
| Rhamnogalacturonan I (RGI) | 20–35 | -Repeated disaccharides composed of GalA residues and rhamnose (Rha); -Rha residues (20% to 80%) can be replaced by neutral sugar side chains (galactose, arabinose, xylose, and apiosis). | [9,12,13,15,25,27,28] |
| Rhamnogalacturonan (RG II) | <10 | -HG backbone composed of GalA (7–9 units) where complex branches made up of 12 types of monosaccharides (including monomers such as apiose, fucose, acetic acid, DHA, or KDO) can exist. | [13,15,25,27,28] |
| Xilogalacturonan (XG) | <10 | Highly complex branched structure linked through a β-glycoside bond with GalA’s O-3 in HG. | [13,25] |
| Activity of Pectin | Features and Conditions | Results | Applications | Ref |
|---|---|---|---|---|
| Emulsifying | The use of pectin as an emulsifying agent is favored by its molecular characteristics (protein portion, acetyl group, acetylation position, ferulic acid content, degree of esterification, neutral sugar side chain, and average molecular weight) and environmental conditions (pectin concentration and pH of the solution). Proteins play the main role to confer emulsifying capacity: the droplet size of pectin-stabilized emulsions decreased (4.12 to 1.5 mm) as the protein content of pectin from beetroot pulp increased (0.5 wt.% to 3 wt.%) [32]. Pectin with the highest protein content exhibited good emulsifying capacity. | - Anchor formation by the protein portion: allows the adsorption of pectin on the oil drop surface, decreasing the interfacial tension at the oil/water interface, enhancement and improvements on the emulsion stability due to the protein–oil/water interface binds, allowing the pectin to form a thicker stabilizing layer protecting droplets against aggregation; - Increase the viscosity of the continuous aqueous phase of emulsions, stimulating the restriction of the mobility of the droplets dispersed in the oil, inhibiting or minimizing its tendency to migrate and coalesce; - Use of a low amount of pectin (about 1.5%) as an emulsifier compared to other polysaccharides (4% of soy-soluble polysaccharides and up to 10% of gum arabic). | - Emulsified oils and emulsion-based foods to use in low-fat mayonnaise, fatty dairy products, ice cream, and emulsified meat products; - Emulsified pectin oils as a fat substitute to develop products with low fat and/or low salt content, improving the nutritional quality of food. | [9,12,13,15,16,32,81,86] |
| Use of Pectin | Goal | Results | Ref |
|---|---|---|---|
| Films | Films of apple pectin (2%), low-acyl gellan (0.5%) and glycerol (2.2%) incorporated with nisin were produced and the kinetics release was studied (at 5 and 30 °C) from brain heart infusion (BHI) broth and nutrient agar (food simulant) used for cultivation of L. monocytogenes food model to determine the period of film bioactivity and its potential use as an antimicrobial film | - At equilibrium time (72 h), 83% of initial nisin was released at 30 °C while it was 81% at 5 °C; - Release patterns of nisin from the pectin-gellan films indicate that these materials can be used as anti-listerial films for prolonging shelf-life of packed food systems; - The incorporation of nisin formulation into the films led to more plasticized films. | [92] |
| Films | Citrus high-methoxyl pectin (PEC) films activated by nanoemulsions (NE) of copaiba oil (CP) at 1%, 3%, and 6% (wt. of water) and Tween 80 (1 wt.% of CP) to ultrapure water were evaluated of chemical, morphological, thermal, mechanical and microbial properties | - The addition of CP-NE into the films improved their physico-chemical and antimicrobial properties, and increased the film’s biodegradation profile; - The active pectin film showed great potential to be used as food packaging. | [93] |
| Coating | Edible coating of HM sunflower pectin (1% w/v), with sweeteners: sucrose (10%), stevia or saccharin (concentration 400× lower than sucrose) and their possible combination with two modified atmosphere packs (MAP) to extend the shelf life of strawberries |
- The coatings, formed only with calcium and stevia or saccharin, prolonged the shelf life of strawberries up to 12 days compared to uncoated fruits by reducing microbial growth, maintaining the fruit firmness, and constant mass loss; - When combined with MAP (10% CO2, 85% N2, and 5% O2), the same edible coatings have extended the shelf life of strawberries up to 23 days. | [94] |
| Coating | Edible coating of HM pectin from citrus peel (3%) loaded with oregano essential oil (OEO) and resveratrol (RES) nanoemulsion (2% OEO, 5% Tween 80%, 1.6% ethanol and 800 mg/L RES) applied in fresh pork tenderloin and packaged under high oxygen MAP (HOMAP) | - The coatings extended the shelf life of the meat, minimizing the change in color and pH, delaying lipid and protein oxidations, maintaining its tenderness, and inhibiting microbial growth during the 20 days of HOMAP packaging at 4 °C; - Incorporation of OEO or resveratrol further increased the preservative effects due to their antioxidant and antimicrobial properties. | [95] |
| Films | Films were prepared with LM pectin (2% w/v), glycerol (1.5 g/g of pectin), calcium chloride (0.005 g/g of pectin) and incorporated with acerola alcoholic extract, cashew apple alcoholic extract, strawberry alcoholic extract or the combination of the 3 extracts (0.17 g of total solids/g of pectin). | - The films with acerola alcoholic extract exhibits the highest antioxidant capacity retention (DPPH = 98.3, FRAP = 969, and ABTS = 114.24 mg of Trolox/g of dry extract); - The films with acerola alcoholic extract exhibits the highest phenolic compounds (236 mg of GAE/g of dry extract) and vitamin C (8.8 mg of AA/of dry extract). | [96] |
| Coating | Edible coatings of fish gelatin (3% w/v), orange peel HM pectin (3% w/v) and glycerol (15%) applied in packaged ricotta cheese stored under refrigeration for 7 days | - The coating provided an improvement in the physical-chemical and textural properties of the cheese during storage; - The coating increases the microbial stability of the cheese during refrigerated storage and offers health-promoting benefits to consumers. | [97] |
| Films | Packaging material from Citrus junos pomace (CJP) pectin (3% w/v) and a selected plasticizer (fructose, sorbitol, and glycerol) (0.3 g/g CJP) dissolved in water were elaborated in addition of rambutan peel extract (RPE) (0.25%, 0.5%, and 1.0% w/v) | - Glycerol was selected as the optimal plasticizer for CJP films; - The CJP without RPE had lower Total Phenolic Content (TPC) (5.08 mg GAE/g film) than the CJP film with 1% of RPE (53 mg GAE/g film); - The CJP prepared in this study can be used as a low-cost active biodegradable film material. | [98] |
| Coating | Combinations of HM pectin and corn flour (relation 1:0, 1:1, 4:1, 3:2 v/v) pectin, corn flour and beet powder (with addition of beet powder at concentration 0.4%) to protect tomatoes | - All treatments led to a reduction in weight loss compared to untreated tomatoes; - The coating of pectin and corn flour (1:1 v/v) was the best treatment, resulting in less weight loss and a percentage of deterioration. The coated fruits retained their maximum brightness and showed minimal shrinkage of the pericarp at the end of 30 days of storage; - The incorporation of beet powder resulted in greater permeability to water vapor. However, due to the higher solids, coatings with the addition of beet powder were less effective in reducing evapotranspiration. | [89] |
| Films | Films from blending of citrus peel low-methylated (7%) PEC and Vicia ervilia proteins concentrate (BVPC) were elaborated by dispersion of 5g of BVPC in 100 mL of distilled water, addition of 50% (w/v protein) glycerol | - BVPC/PEC exhibited a tensile strength double (2.90 MPa) than the one observed with films containing only BVPC (1.52 MPa); - The elongation at break resulted higher in the films containing PEC (41.17%) than films containing only BVPC (30.15%), leading to conclude that films are more extensible when PEC occur in the films forming solutions. | [99] |
| Coating | Pectin-alginate (PA) coatings (pectin 15 g·L−1, sodium alginate 10 g·L−1, glycerol 6.75 g·L−1, sodium bicarbonate 2 g·L−1) and a mixture of PA and ethyl lauroyl arginate (PAL) to eliminate cross-contamination of enteritidis by Salmonella spp. on fresh eggs | - Eggshells treated with PA and PAL coatings had a significantly smaller microbial population compared to uncoated eggshells; - PA and PAL coatings effectively inhibited the growth of Salmonella spp. after 1 and 7 days of storage, respectively. | [100] |
| Coating | Solutions of industrial citrus pectin (1%, 3%, 5%, and 8%) used as an edible coating for fresh strawberries | - Pectin coatings samples showed better behavior in terms of color variation than control; which indicates the positive effect of the coatings as a selective barrier, preventing the exposure of the fruit to environmental oxygen and inhibiting possible oxidation reactions; - All samples had a considerable loss of moisture during storage. | [101] |
| Films | Films were produced using pectin (0.8%–1.8% w/v) extracted from byproduct of Hibiscus sabdariffa L. (HsL) and glycerol (0.5%–1.5% w/w of pectin), as plasticizer, at different casting volumes (10–20 mL). The films were used to preserve fresh strawberries | - At the end of the 21 days storage, strawberries protected with the films presented lower weight loss (39.1%) in comparison to unprotected fruits (43.8%); - Fruits packaged with the active films presented reduction of one Log cycle in the aerobic plate counts at 21 days of storage. | [102] |
| Coating | Combination of pectin with anti-browning agents: ascorbic acid 1% (w/v), citric acid 1% (w/v) and sodium chlorite 0.05% (w/v); for apple coating | - Coatings were considered to be a safe and effective treatment, improving the fruit’s shelf-life; - There was a reduction in microbial deterioration without significantly affecting the nutritional value of the apple. | [103] |
| Use of Pectin | Goal | Results | Ref |
|---|---|---|---|
| Hydrogel manufacturing | Preparation of a substance using zeolite imidazolate framework-8 (ZIF-8) nanoparticles, coated with polyethylene glycol-thioketal (PEG-TK) to manufacture SP@ZIF-8-PEG-TK nanoparticles, encapsulated with injectable hydrogel composed of sodium alginate and pectin | - The SP@ZIF-8-PEG-TK nanoparticles promoted the proliferation of human dermal fibroblasts, increased the expression levels of inflammation-related genes in macrophages, and exhibited favorable compatibility in vitro; - Models of full-thickness excision wounds in vivo confirmed that the dressings had excellent efficacy in wound healing. | [111] |
| Hydrogel manufacturing | Preparation of pectin hydrogels loaded with theophylline for application of wound dressings and their performance of controlled drug release | - The concentration of the initial pectin solution is a critical parameter to obtain an effective drug release; - Dressings can be synthesized as a controlled drug delivery system. | [112] |
| Development of nano-liposomal systems | Development of nano-liposomal pectin and chitosan systems for controlled delivery of neohesperidin to the gastrointestinal tract | - The in vitro study revealed that the systems significantly controlled the delivery of neohesperidin; - The system is considered efficient for the controlled release profile. | [113] |
| Hydrogel manufacturing | Preparation of hydrogels based on pectin, lactic acid (LA), and methacrylic acid (MAA) as a potential biomaterial for colonic delivery of oxaliplatin | - The drug release increased with the increase in the concentration of pectin and LA, while the MAA showed the opposite behavior; - The developed hydrogels can be explored in-vivo for the colonic distribution of anticancer and other drugs. | [105] |
| Coating | Production of titanium alloy discs (Ti6Al4V) coated with citrus (C) and apple (A) pectins, containing alkaline phosphatase (ALP) | - Pectin coatings containing ALP promoted the adhesion and proliferation of bone marrow stromal cells; - A-ALP coatings were more hydrophilic than C-ALP. | [83] |
| Membrane production | Production of membrane from pectin and Aloe gel for use as a biomaterial | - All membranes showed high solubility (100%) and low permeability to water vapor; - The membranes produced have high potential as a biomaterial that can be used for practical applications in human health. | [18] |
| Biocomposite dressings | Development of wounding dressings composed of pectin and gelatin loaded with Aloe vera and curcumin to treat wounds |
- Biocomposite dressings are considered viable materials with strong potential for the effective treatment of wounds; - Wounds treated with pectin and gelatin matrices loaded with Aloe vera showed very fast healing, with 80% of the healing in just 8 days. | [114] |
| Use of Pectin | Goal | Results | Ref |
|---|---|---|---|
| Nanocell manufacturing | Development of a pectin nanocell containing doxorubicin (DOX) for the administration of anticancer agent with reversal of resistance to multiple drugs | - The developed nanocell showed the expected potency against tumor growth in vitro and in vivo, significantly increasing the intracellular accumulation of doxorubicin and the prolonged release of the drug; - The nanocell reversed the drug resistance of tumor cells to some degree. | [19] |
| Conjugates manufacturing | Pectin and dopamine conjugates to coordinate the ruthenium complex, as a metal model based on anticancer drugs | - The modification of pectin by dopamine and ruthenium complex changed the amorphous network, viscosity, and viscoelastic behavior of pectin; - Conjugates increase the prospects for the development and medical applications of new metal-based anticancer drugs. | [126] |
| Coating | Development of solid double layered lipid nanoparticles coated with pectin loaded with soluble curcumin (CMN) to increase the cytotoxicity of the drug used in the treatment of colon cancer | - The efficiency of the nanoparticles indicated their potential application for the treatment of cancer with an increase in the bioavailability of soluble curcumin orally; - The nanoparticles showed a significantly high drug load, better stability, and a slower release profile. | [120] |
| Nanoparticle manufacturing | Evaluation of the specificity and efficiency of cetuximab (Cet) conjugated to the modified citrus pectin nanoparticle and chitosan containing curcumin (MCPCNPs) | - The in vitro release of curcumin in a simulator medium supports the adequacy of this formulation for delivery to the colon; - The propensity to mucoadhesion of MCPCNPs was not altered after Cet conjugation; - There was superior uptake of curcumin when encapsulated in Cet-MCPCNPs. | [127] |
| Nanoparticle manufacturing | Development of pectin and doxorubicin nanoparticles for the treatment of hepatocellular carcinoma | - The nanoparticles have achieved sustained and prolonged release capacity; - In vivo studies have shown that nanoparticles have significantly reduced the side effect of doxorubicin. | [124] |
| Nanoparticle manufacturing | Development of a delivery system for modified citrus pectin and chitosan nanoparticles in the safe delivery of curcumin (MCPCNPs) for the treatment of colon cancer | - MCPCNPs were highly prone to mucoadhesion in the region/medium of the colon and minimal at pH 1.2 (stomach); - The data obtained suggest that MCPCNPs can be applied as a colon cancer formulation for alternative oral administration. | [116] |
| Conjugates manufacturing | Preparation of a drug delivery system composed of chitosan, pectin, and doxorubicin for the treatment of liver cancer | - The antitumor efficiency was significantly high, demonstrated by the in vitro test; - The system effectively supplied tumor growth, according to the in vivo test; - The system is non-toxic, highly biocompatible, and safe. | [125] |
| Therapeutical compounds | Evaluation of the effectiveness of combining ionizing radiation with modified citrus pectin in prostate cancer cells | - This combination increased radiosensitivity associated with a decrease in Gal3; - Pectin significantly decreased the invasive and migratory potential of cancer cells. | [128] |
| Coating | Development of graphene-chitosan oxide nanocomposite coated with pectin for delivery of drugs directed to the colon | - Nanocomposites have selectively eliminated cancer cells, which indicates that it is a promising therapeutic agent for cancer treatment. | [122] |
| Conjugates manufacturing | Development of oral microspheres containing conjugates of pectin and doxorubicin for the treatment of colon cancer | - The conjugate of pectin and doxorubicin can be decoupled in reducing environments, resulting in cleavage of the disulfide ligands and releasing DOX; - Microspheres proved to be a promising platform for the distribution of cancer-directed doxorubicin. | [121] |
| Nasal spray manufacturing | Evaluation of analgesic efficacy, safety, and tolerability of fentanyl pectin nasal spray for the treatment of disruptive cancer pain | - The spray provided quick and effective pain relief, with substantial improvements in the patient life quality. | [129] |
| Nanoparticle manufacturing | Development of β-lactoglobulin and pectin nanoparticles for the treatment of colon cancer | - The developed nanoparticles were able to provide anticancer drugs and increase antitumor efficacy with low systemic toxicity. | [130] |
| Therapeutical compounds | Evaluation of the anticancer properties of pectin in vitro in breast cancer cells and in vivo using an animal model | - In vitro studies have shown that pectin can induce apoptosis, inhibit cell growth and reduce cell fixation; - In vivo studies have shown that pectin can inhibit tumor progression and increase apoptotic cells. | [131] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Freitas, C.M.P.; Coimbra, J.S.R.; Souza, V.G.L.; Sousa, R.C.S. Structure and Applications of Pectin in Food, Biomedical, and Pharmaceutical Industry: A Review. Coatings 2021, 11, 922. https://doi.org/10.3390/coatings11080922
Freitas CMP, Coimbra JSR, Souza VGL, Sousa RCS. Structure and Applications of Pectin in Food, Biomedical, and Pharmaceutical Industry: A Review. Coatings. 2021; 11(8):922. https://doi.org/10.3390/coatings11080922
Chicago/Turabian StyleFreitas, Cariny Maria Polesca, Jane Sélia Reis Coimbra, Victor Gomes Lauriano Souza, and Rita Cássia Superbi Sousa. 2021. "Structure and Applications of Pectin in Food, Biomedical, and Pharmaceutical Industry: A Review" Coatings 11, no. 8: 922. https://doi.org/10.3390/coatings11080922
APA StyleFreitas, C. M. P., Coimbra, J. S. R., Souza, V. G. L., & Sousa, R. C. S. (2021). Structure and Applications of Pectin in Food, Biomedical, and Pharmaceutical Industry: A Review. Coatings, 11(8), 922. https://doi.org/10.3390/coatings11080922






