Effect of Micelle Encapsulation on Toxicity of CdSe/ZnS and Mn-Doped ZnSe Quantum Dots
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Preparation of Mn-Doped ZnSe QDs
2.2.1. Synthesis of MnSt2 (Manganese Stearate) Precursor
2.2.2. Synthesis of Mn-Doped ZnSe QDs
2.2.3. Purification of Mn-Doped ZnSe QDs
2.3. Preparation of Water-Soluble QDs
2.3.1. Synthesis of MPA-QDs by Ligand Exchange
2.3.2. Synthesis of Micelle-Encapsulated QDs
2.4. Toxicity Assays
2.4.1. MTT Assay
2.4.2. ROS Assay
2.4.3. TUNEL Assay
2.4.4. Live/Dead Analysis via Fl Cytometry
2.4.5. Statistical Analysis
3. Results
3.1. Characterisation of Mn-Doped ZnSe QDs following Phase Transfer
3.2. QD Effects on Cell Viability
3.3. Reactive Oxygen Species Formation
3.4. DNA Fragmentation
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Carey, G.H.; Abdelhady, A.L.; Ning, Z.; Thon, S.M.; Bakr, O.M.; Sargent, E.H. Colloidal Quantum Dot Solar Cells. Chem. Rev. 2015, 115, 12732–12763. [Google Scholar] [CrossRef] [PubMed]
- Shirasaki, Y.; Supran, G.J.; Bawendi, M.G.; Bulovic, V. Emergence of colloidal quantum-dot light-emitting technologies. Nat. Photon. 2013, 7, 13–23. [Google Scholar] [CrossRef]
- Biju, V. Chemical modifications and bioconjugate reactions of nanomaterials for sensing, imaging, drug delivery and therapy. Chem. Soc. Rev. 2014, 43, 744–764. [Google Scholar] [CrossRef]
- Hildebrandt, N.; Spillmann, C.M.; Algar, W.R.; Pons, T.; Stewart, M.H.; Oh, E.; Susumu, K.; Diaz, S.A.; Delehanty, J.B.; Medintz, I.L. Energy Transfer with Semiconductor Quantum Dot Bioconjugates: A Versatile Platform for Biosensing, Energy Harvesting, and Other Developing Applications. Chem. Rev. 2017, 117, 536–711. [Google Scholar] [CrossRef]
- Resch-Genger, U.; Grabolle, M.; Cavaliere-Jaricot, S.; Nitschke, R.; Nann, T. Quantum dots versus organic dyes as fluorescent labels. Nat. Methods 2008, 5, 763–775. [Google Scholar] [CrossRef]
- Kirchner, C.; Liedl, T.; Kudera, S.; Pellegrino, T.; Munoz Javier, A.; Gaub, H.E.; Stolzle, S.; Fertig, N.; Parak, W.J. Cytotoxicity of colloidal CdSe and CdSe/ZnS nanoparticles. Nano Lett. 2005, 5, 331–338. [Google Scholar] [CrossRef]
- Hardman, R. A toxicologic review of quantum dots: Toxicity depends on physicochemical and environmental factors. Environ. Health Perspect. 2006, 114, 165–172. [Google Scholar] [CrossRef]
- Nagy, A.; Steinbrück, A.; Gao, J.; Doggett, N.; Hollingsworth, J.A.; Iyer, R. Comprehensive Analysis of the Effects of CdSe Quantum Dot Size, Surface Charge, and Functionalization on Primary Human Lung Cells. ACS Nano 2012, 6, 4748–4762. [Google Scholar] [CrossRef]
- Chan, W.C.; Nie, S. Quantum dot bioconjugates for ultrasensitive nonisotopic detection. Science 1998, 281, 2016–2018. [Google Scholar] [CrossRef] [PubMed]
- Bruchez, M., Jr.; Moronne, M.; Gin, P.; Weiss, S.; Alivisatos, A.P. Semiconductor nanocrystals as fluorescent biological labels. Science 1998, 281, 2013–2016. [Google Scholar] [CrossRef] [PubMed]
- Derfus, A.M.; Chan, W.C.W.; Bhatia, S.N. Probing the cytotoxicity of semiconductor quantum dots. Nano Lett. 2004, 4, 11–18. [Google Scholar] [CrossRef] [PubMed]
- Rani, A.; Kumar, A.; Lal, A.; Pant, M. Cellular mechanisms of cadmium-induced toxicity: A review. Int. J. Environ. Health Res. 2014, 24, 378–399. [Google Scholar] [CrossRef] [PubMed]
- Smith, A.M.; Duan, H.W.; Rhyner, M.N.; Ruan, G.; Nie, S.M. A systematic examination of surface coatings on the optical and chemical properties of semiconductor quantum dots. Phys. Chem. Chem. Phys. 2006, 8, 3895–3903. [Google Scholar] [CrossRef]
- Chen, N.; He, Y.; Su, Y.Y.; Li, X.M.; Huang, Q.; Wang, H.F.; Zhang, X.Z.; Tai, R.Z.; Fan, C.H. The cytotoxicity of cadmium-based quantum dots. Biomaterials 2012, 33, 1238–1244. [Google Scholar] [CrossRef] [PubMed]
- Sukhanova, A.; Bozrova, S.; Sokolov, P.; Berestovoy, M.; Karaulov, A.; Nabiev, I. Dependence of Nanoparticle Toxicity on Their Physical and Chemical Properties. Nanoscale Res. Lett. 2018, 13, 44. [Google Scholar] [CrossRef]
- Chang, E.; Thekkek, N.; Yu, W.W.; Colvin, V.L.; Drezek, R. Evaluation of quantum dot cytotoxicity based on intracellular uptake. Small 2006, 2, 1412–1417. [Google Scholar] [CrossRef]
- Clift, M.J.D.; Rothen-Rutishauser, B.; Brown, D.M.; Duffin, R.; Donaldson, K.; Proudfoot, L.; Guy, K.; Stone, V. The impact of different nanoparticle surface chemistry and size on uptake and toxicity in a murine macrophage cell line. Toxicol. Appl. Pharmacol. 2008, 232, 418–427. [Google Scholar] [CrossRef]
- Peng, L.; He, M.; Chen, B.B.; Wu, Q.M.; Zhang, Z.L.; Pang, D.W.; Zhu, Y.; Hu, B. Cellular uptake, elimination and toxicity of CdSe/ZnS quantum dots in HepG2 cells. Biomaterials 2013, 34, 9545–9558. [Google Scholar] [CrossRef]
- Lovric, J.; Bazzi, H.S.; Cuie, Y.; Fortin, G.R.; Winnik, F.M.; Maysinger, D. Differences in subcellular distribution and toxicity of green and red emitting CdTe quantum dots. J. Mol. Med. 2005, 83, 377–385. [Google Scholar] [CrossRef]
- Pelley, J.L.; Daar, A.S.; Saner, M.A. State of Academic Knowledge on Toxicity and Biological Fate of Quantum Dots. Toxicol. Sci. 2009, 112, 276–296. [Google Scholar] [CrossRef]
- Rocha, T.L.; Mestre, N.C.; Saboia-Morais, S.M.T.; Bebianno, M.J. Environmental behaviour and ecotoxicity of quantum dots at various trophic levels: A review. Environ. Int. 2017, 98, 1–17. [Google Scholar] [CrossRef]
- Brunetti, V.; Chibli, H.; Fiammengo, R.; Galeone, A.; Malvindi, M.A.; Vecchio, G.; Cingolani, R.; Nadeau, J.L.; Pompa, P.P. InP/ZnS as a safer alternative to CdSe/ZnS core/shell quantum dots: In vitro and in vivo toxicity assessment. Nanoscale 2013, 5, 307–317. [Google Scholar] [CrossRef]
- Lin, G.M.; Ouyang, Q.L.; Hu, R.; Ding, Z.C.; Tian, J.L.; Yin, F.; Xu, G.X.; Chen, Q.; Wang, X.M.; Yong, K.T. In vivo toxicity assessment of non-cadmium quantum dots in BALB/c mice. Nanomed. Nanotechnol. Biol. Med. 2015, 11, 341–350. [Google Scholar] [CrossRef]
- Liu, J.A.; Wei, X.L.; Qu, Y.; Cao, J.H.; Chen, C.S.; Jiang, H. Aqueous synthesis and bio-imaging application of highly luminescent and low cytotoxicity Mn2+-doped ZnSe nanocrystals. Mater. Lett. 2011, 65, 2139–2141. [Google Scholar] [CrossRef]
- Zhou, R.H.; Li, M.; Wang, S.L.; Wu, P.; Wu, L.; Hou, X.D. Low-toxic Mn-doped ZnSe@ZnS quantum dots conjugated with nano-hydroxyapatite for cell imaging. Nanoscale 2014, 6, 14319–14325. [Google Scholar] [CrossRef]
- Plumley, J.B.; Akins, B.A.; Alas, G.J.; Fetrow, M.E.; Nguyen, J.; Jain, P.; Yang, S.; Brandt, Y.I.; Smolyakov, G.A.; Ornatowski, W.; et al. Non-cytotoxic Mn-doped ZnSe/ZnS quantum dots for biomedical applications. In Proceedings of the Conference on Colloidal Nanoparticles for Biomedical Applications IX, San Francisco, CA, USA, 1–4 February 2014. [Google Scholar]
- Drobintseva, A.O.; Matyushkin, L.B.; Aleksandrova, O.A.; Drobintsev, P.D.; Kvetnoy, I.M.; Mazing, D.S.; Moshnikov, V.A.; Polyakova, V.O.; Musikhin, S.F. Colloidal CdSe and ZnSe/Mn quantum dots: Their cytotoxicity and effects on cell morphology. St. Petersburg Polytech. Univ. J. Phys. Math. 2015, 1. [Google Scholar] [CrossRef]
- Swift, B.J.F.; Baneyx, F. Microbial Uptake, Toxicity, and Fate of Biofabricated ZnS:Mn Nanocrystals. PLoS ONE 2015, 10, e0124916. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Lv, S.-Y.; Yu, B.; Xu, S.; Shen, J.; Zhao, T.; Zhang, H. Hepatotoxicity assessment of Mn-doped ZnS quantum dots after repeated administration in mice. Int. J. Nanomed. 2015, 10, 5787–5796. [Google Scholar] [CrossRef] [PubMed]
- Mohammad, F.; Al-Lohedan, H.A. Toxicity assessment of engineered Mn-ZnS quantum dots in vitro. J. Mater. Sci. 2016, 51, 9207–9216. [Google Scholar] [CrossRef]
- Li, Y.; Zhang, H.; Guo, C.; Hu, G.; Wang, L. Multiresponsive Nanoprobes for Turn-On Fluorescence/19F MRI Dual-Modal Imaging. Anal. Chem. 2020, 92, 11739–11746. [Google Scholar] [CrossRef] [PubMed]
- Pradhan, N.; Peng, X.G. Efficient and color-tunable Mn-doped ZnSe nanocrystal emitters: Control of optical performance via greener synthetic chemistry. J. Am. Chem. Soc. 2007, 129, 3339–3347. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Fan, Q.; Mahajan, K.D.; Ruan, G.; Herrington, A.; Tehrani, K.F.; Kner, P.; Winter, J.O. Micelle-templated composite quantum dots for super-resolution imaging. Nanotechnology 2014, 25, 195601. [Google Scholar] [CrossRef] [PubMed]
- Dubertret, B.; Skourides, P.; Norris, D.J.; Noireaux, V.; Brivanlou, A.H.; Libchaber, A. In vivo imaging of quantum dots encapsulated in phospholipid micelles. Science 2002, 298, 1759–1762. [Google Scholar] [CrossRef]
- Smith, W.E.; Brownell, J.; White, C.C.; Afsharinejad, Z.; Tsai, J.; Hu, X.; Polyak, S.J.; Gao, X.; Kavanagh, T.J.; Eaton, D.L. In Vitro Toxicity Assessment of Amphiphillic Polymer-Coated CdSe/ZnS Quantum Dots in Two Human Liver Cell Models. ACS Nano 2012, 6, 9475–9484. [Google Scholar] [CrossRef]
- Liu, J.; Hu, R.; Liu, J.; Zhang, B.; Wang, Y.; Liu, X.; Law, W.C.; Liu, L.; Ye, L.; Yong, K.T. Cytotoxicity assessment of functionalized CdSe, CdTe and InP quantum dots in two human cancer cell models. Mater. Sci. Eng. C Mater. Biol. Appl. 2015, 57, 222–231. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Tang, M. Review of in vitro toxicological research of quantum dot and potentially involved mechanisms. Sci. Total Environ. 2018, 625, 940–962. [Google Scholar] [CrossRef] [PubMed]
- Pandey, V.; Pandey, G.; Tripathi, V.K.; Yadav, S.; Mudiam, M.K.R. Nucleation temperature-controlled synthesis and in vitro toxicity evaluation of l-cysteine-capped Mn:ZnS quantum dots for intracellular imaging. Luminescence 2016, 31, 341–347. [Google Scholar] [CrossRef]
- Yang, Y.; Lv, S.; Wang, F.; An, Y.; Fang, N.; Zhang, W.; Zhao, W.; Guo, X.; Ji, S. Toxicity and serum metabolomics investigation of Mn-doped ZnS quantum dots in mice. Int. J. Nanomed. 2019, 14, 6297–6311. [Google Scholar] [CrossRef] [PubMed]
- Labiadh, H.; Sellami, B.; Khazri, A.; Saidani, W.; Khemais, S. Optical properties and toxicity of undoped and Mn-doped ZnS semiconductor nanoparticles synthesized through the aqueous route. Opt. Mater. 2017, 64, 179–186. [Google Scholar] [CrossRef]
- Popescu, T.; Matei, C.O.; Vlaicu, I.D.; Tivig, I.; Kuncser, A.C.; Stefan, M.; Ghica, D.; Miclea, L.C.; Savopol, T.; Culita, D.C.; et al. Influence of surfactant-tailored Mn-doped ZnO nanoparticles on ROS production and DNA damage induced in murine fibroblast cells. Sci. Rep. 2020, 10, 18062. [Google Scholar] [CrossRef]
- Gaceur, M.; Giraud, M.; Hemadi, M.; Nowak, S.; Menguy, N.; Quisefit, J.P.; David, K.; Jahanbin, T.; Benderbous, S.; Boissière, M.; et al. Polyol-synthesized Zn0.9Mn0.1S nanoparticles as potential luminescent and magnetic bimodal imaging probes: Synthesis, characterization, and toxicity study. J. Nanopart. Res. 2012, 14, 932. [Google Scholar] [CrossRef]
- Kuznetsova, V.A.; Visheratina, A.K.; Ryan, A.; Martynenko, I.V.; Loudon, A.; Maguire, C.M.; Purcell-Milton, F.; Orlova, A.O.; Baranov, A.V.; Fedorov, A.V.; et al. Enantioselective cytotoxicity of ZnS:Mn quantum dots in A549 cells. Chirality 2017, 29, 403–408. [Google Scholar] [CrossRef] [PubMed]
- Selvaraj, J.; Mahesh, A.; Asokan, V.; Baskaralingam, V.; Dhayalan, A.; Paramasivam, T. Phosphine-Free, Highly Emissive, Water-Soluble Mn:ZnSe/ZnS Core–Shell Nanorods: Synthesis, Characterization, and in Vitro Bioimaging of HEK293 and HeLa Cells. ACS Appl. Nano Mater. 2018, 1, 371–383. [Google Scholar] [CrossRef]
- Bae, J.; Lawrence, J.; Miesch, C.; Ribbe, A.; Li, W.; Emrick, T.; Zhu, J.; Hayward, R.C. Multifunctional Nanoparticle-Loaded Spherical and Wormlike Micelles Formed by Interfacial Instabilities. Adv. Mater. 2012, 24, 2735–2741. [Google Scholar] [CrossRef]
- Duong, A.D.; Ruan, G.; Mahajan, K.; Winter, J.O.; Wyslouzil, B.E. Scalable, Semicontinuous Production of Micelles Encapsulating Nanoparticles via Electrospray. Langmuir 2014, 30, 3939–3948. [Google Scholar] [CrossRef] [PubMed]
- Eugene, M. Polyethyleneglycols and immunocamouflage of the cells tissues and organs for transplantation. Cell. Mol. Biol. (Noisy-Le-Grand) 2004, 50, 209–215. [Google Scholar]
- Hoshino, A.; Fujioka, K.; Oku, T.; Suga, M.; Sasaki, Y.F.; Ohta, T.; Yasuhara, M.; Suzuki, K.; Yamamoto, K. Physicochemical properties and cellular toxicity of nanocrystal quantum dots depend on their surface modification. Nano Lett. 2004, 4, 2163–2169. [Google Scholar] [CrossRef]
- Tsoi, K.M.; Dai, Q.; Alman, B.A.; Chan, W.C.W. Are Quantum Dots Toxic? Exploring the Discrepancy Between Cell Culture and Animal Studies. Acc. Chem. Res. 2013, 46, 662–671. [Google Scholar] [CrossRef] [PubMed]
- Porter, T. Development of Quantum Dots as Biosensing Probes. Bachelor’s Thesis, The Ohio State University, Columbus, OH, USA, 2020. [Google Scholar]
- Pradhan, N.; Battaglia, D.M.; Liu, Y.C.; Peng, X.G. Efficient, stable, small, and water-soluble doped ZnSe nanocrystal emitters as non-cadmium biomedical labels. Nano Lett. 2007, 7, 312–317. [Google Scholar] [CrossRef]
- Zhu, J.; Hayward, R.C. Spontaneous generation of amphiphilic block copolymer micelles with multiple morphologies through interfacial instabilities. J. Am. Chem. Soc. 2008, 130, 7496–7502. [Google Scholar] [CrossRef]
- Fan, Q. Designing Photo-Switchable Quantum Dots for Super Resolution Imaging. Ph.D. Thesis, OhioLINK Electronic Theses and Dissertations Center, The Ohio State University, Columbus, OH, USA, 2015. [Google Scholar]
- Zhang, Y.; Clapp, A. Overview of Stabilizing Ligands for Biocompatible Quantum Dot Nanocrystals. Sensors 2011, 11, 11036–11055. [Google Scholar] [CrossRef] [PubMed]
- Zhao, C.Z.; Bai, Z.L.; Liu, X.Y.; Zhang, Y.J.; Zou, B.S.; Zhong, H.Z. Small GSH-Capped CuInS2 Quantum Dots: MPA-Assisted Aqueous Phase Transfer and Bioimaging Applications. ACS Appl. Mater. Interfaces 2015, 7, 17623–17629. [Google Scholar] [CrossRef]
- Yong, K.T.; Law, W.C.; Hu, R.; Ye, L.; Liu, L.W.; Swihart, M.T.; Prasad, P.N. Nanotoxicity assessment of quantum dots: From cellular to primate studies. Chem. Soc. Rev. 2013, 42, 1236–1250. [Google Scholar] [CrossRef] [PubMed]
- Salin, K.; Auer, S.K.; Rudolf, A.M.; Anderson, G.J.; Cairns, A.G.; Mullen, W.; Hartley, R.C.; Selman, C.; Metcalfe, N.B. Individuals with higher metabolic rates have lower levels of reactive oxygen species in vivo. Biol. Lett. 2015, 11. [Google Scholar] [CrossRef] [PubMed]
- Lovric, J.; Cho, S.J.; Winnik, F.M.; Maysinger, D. Unmodified cadmium telluride quantum dots induce reactive oxygen species formation leading to multiple organelle damage and cell death. Chem. Biol. 2005, 12, 1227–1234. [Google Scholar] [CrossRef] [PubMed]
- Ipe, B.I.; Lehnig, M.; Niemeyer, C.M. On the generation of free radical species from quantum dots. Small 2005, 1, 706–709. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Gao, X.; Su, X. Study the damage of DNA molecules induced by three kinds of aqueous nanoparticles. Talanta 2010, 80, 1228–1233. [Google Scholar] [CrossRef]
- Anderson, N.C.; Hendricks, M.P.; Choi, J.J.; Owen, J.S. Ligand Exchange and the Stoichiometry of Metal Chalcogenide Nanocrystals: Spectroscopic Observation of Facile Metal-Carboxylate Displacement and Binding. J. Am. Chem. Soc. 2013, 135, 18536–18548. [Google Scholar] [CrossRef]
- Shen, Y.; Tan, R.; Gee, M.Y.; Greytak, A.B. Quantum Yield Regeneration: Influence of Neutral Ligand Binding on Photophysical Properties in Colloidal Core/Shell Quantum Dots. ACS Nano 2015, 9, 3345–3359. [Google Scholar] [CrossRef]
- Zhao, J.; Stenzel, M.H. Entry of nanoparticles into cells: The importance of nanoparticle properties. Polym. Chem. 2018, 9, 259–272. [Google Scholar] [CrossRef]
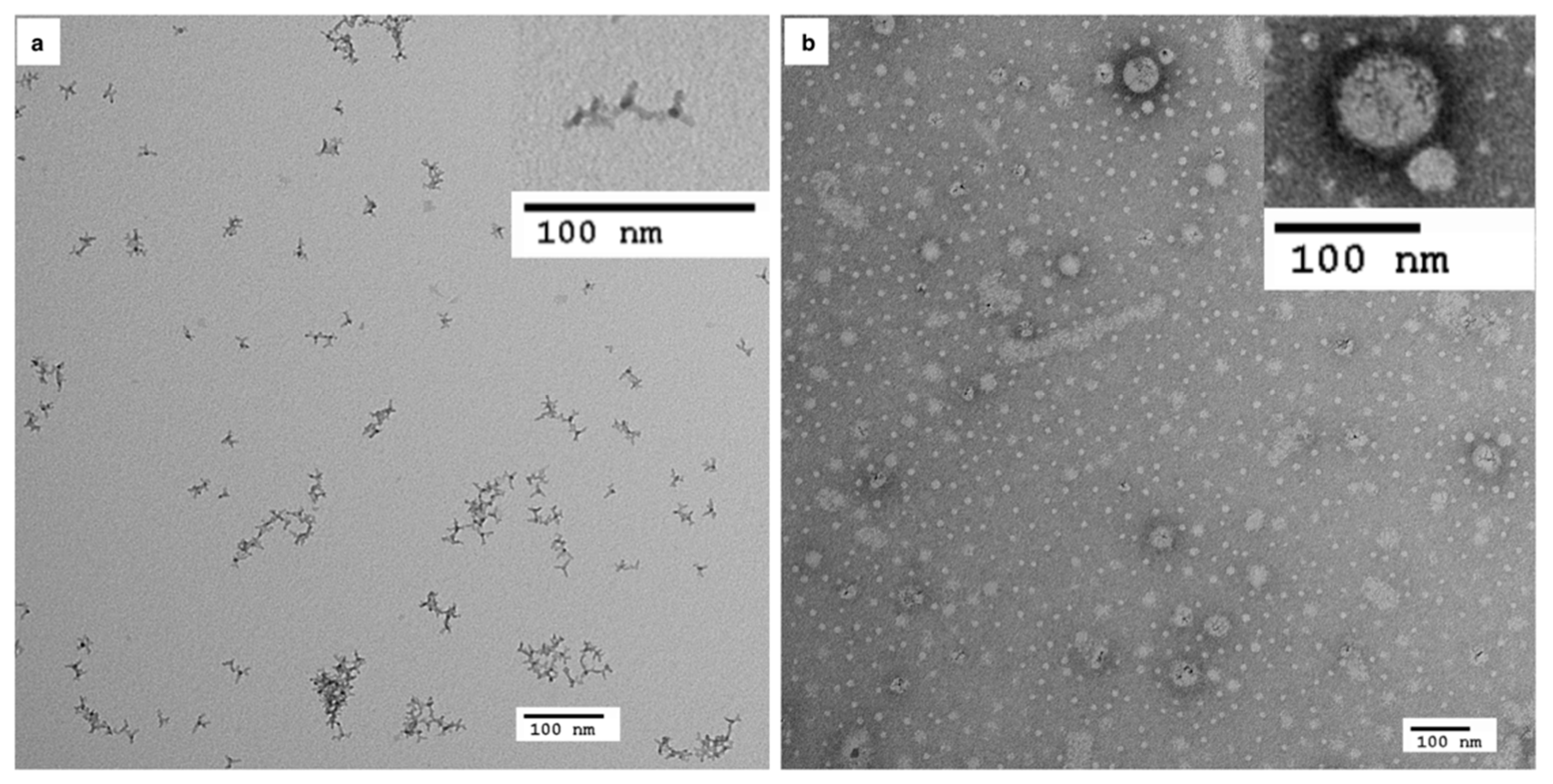
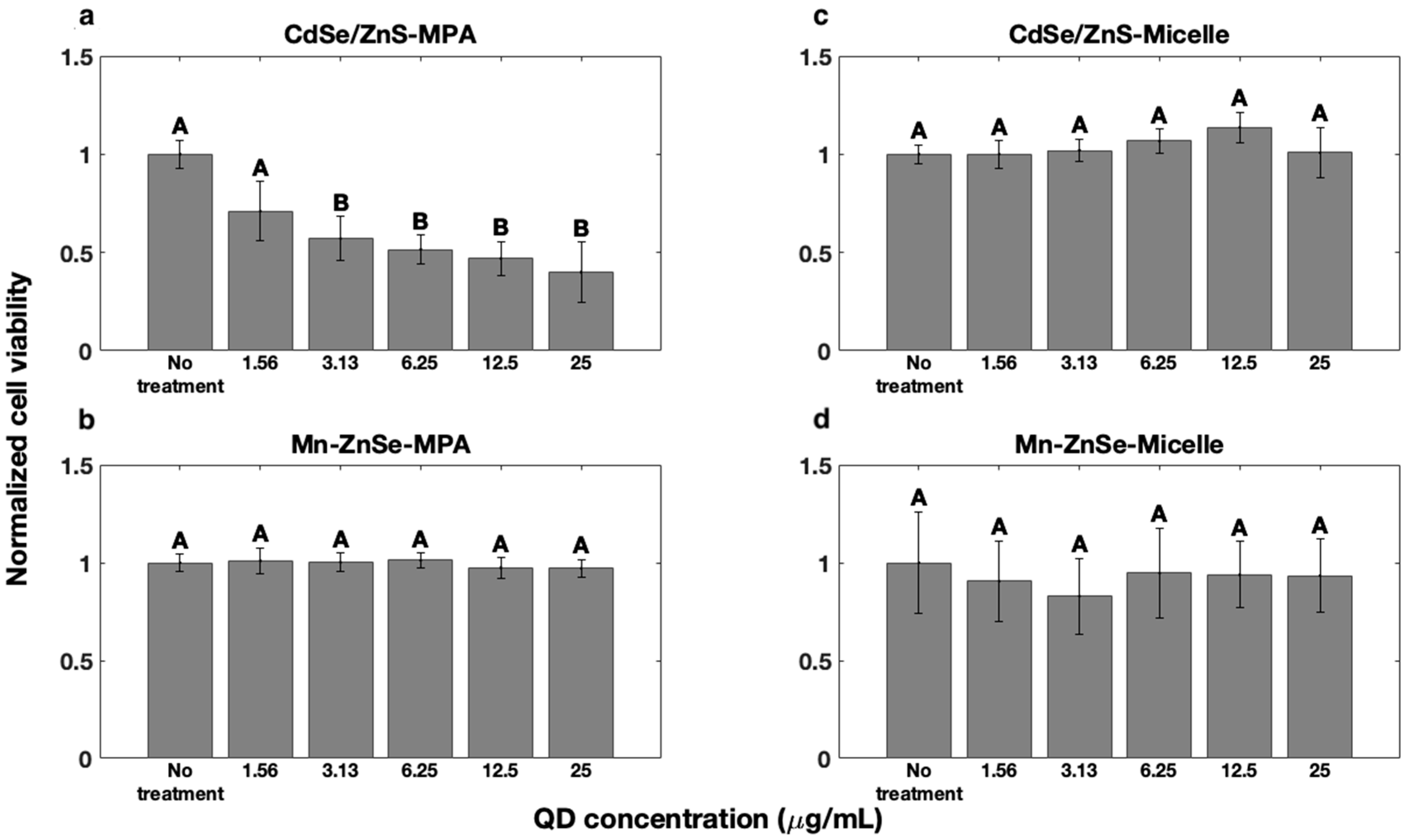
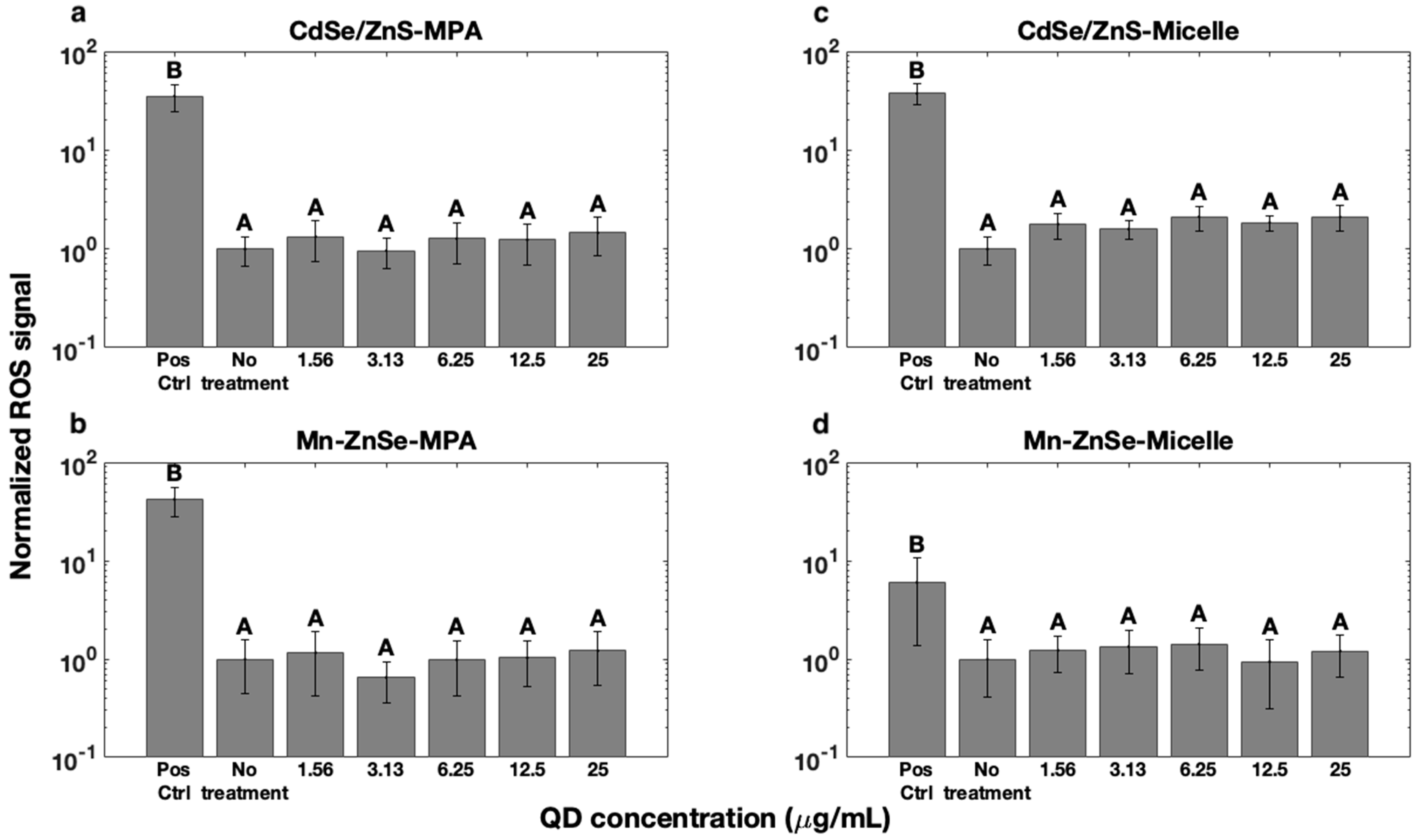
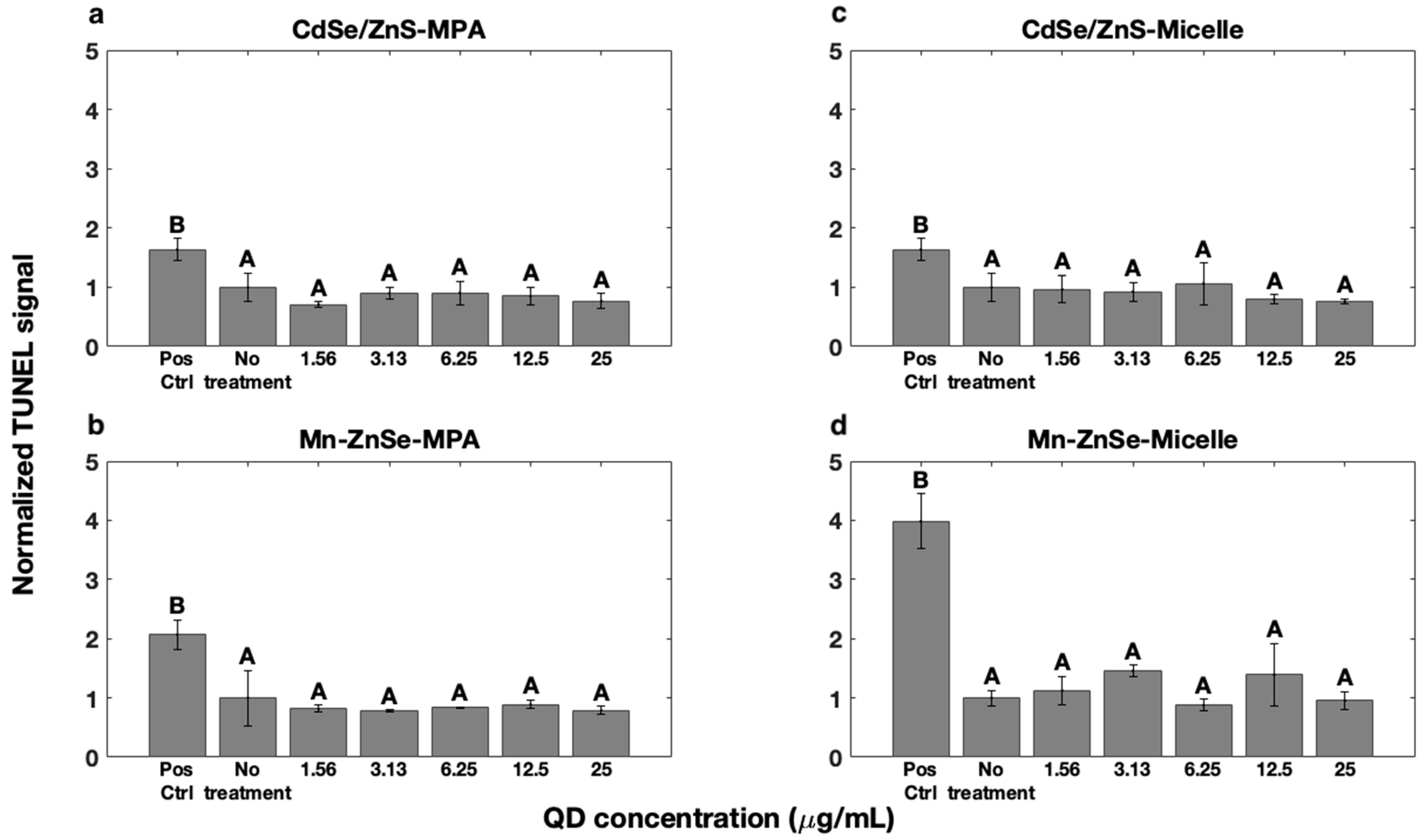
| Concentration (µg/mL) | Live % | Dead % | Injured % |
|---|---|---|---|
| 6.25 | 82.66 | 0.19 | 16.99 |
| 25 | 78.16 | 0.16 | 21.64 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fan, Q.; Dehankar, A.; Porter, T.K.; Winter, J.O. Effect of Micelle Encapsulation on Toxicity of CdSe/ZnS and Mn-Doped ZnSe Quantum Dots. Coatings 2021, 11, 895. https://doi.org/10.3390/coatings11080895
Fan Q, Dehankar A, Porter TK, Winter JO. Effect of Micelle Encapsulation on Toxicity of CdSe/ZnS and Mn-Doped ZnSe Quantum Dots. Coatings. 2021; 11(8):895. https://doi.org/10.3390/coatings11080895
Chicago/Turabian StyleFan, Qirui, Abhilasha Dehankar, Thomas K. Porter, and Jessica O. Winter. 2021. "Effect of Micelle Encapsulation on Toxicity of CdSe/ZnS and Mn-Doped ZnSe Quantum Dots" Coatings 11, no. 8: 895. https://doi.org/10.3390/coatings11080895
APA StyleFan, Q., Dehankar, A., Porter, T. K., & Winter, J. O. (2021). Effect of Micelle Encapsulation on Toxicity of CdSe/ZnS and Mn-Doped ZnSe Quantum Dots. Coatings, 11(8), 895. https://doi.org/10.3390/coatings11080895






