1. Introduction
Two-dimensional (2D) molybdenum disulfide (MoS
2), as one of the most promising candidates of 2D materials beyond graphene, has attracted a great deal of attention because of its intriguing properties leading to wide applications [
1,
2,
3]. First and foremost, scalable techniques to develop large area MoS
2 are the precondition for further research and potential applications [
4,
5,
6].
In recent years, chemical vapor deposition (CVD) has been one of the most promising methods for large area 2D MoS
2 production [
7,
8,
9]. In this method, precursors including MoO
3, MoCl
5, (NH
4)
2MoS
4 powder are used to offer molybdenum vapor source and sulfur powder to offer a sulfur vapor source, with Ar used as carrier gas to deliver the precursors onto the substrate on which the precursors react to form MoS
2 crystal [
10]. The size and shape of MoS
2 crystal, the number of MoS
2 crystal layers and the distribution of MoS
2 crystal on substrates are the key parameters concerned in the CVD growth of MoS
2. One single crystal domain can hardly spread all across a large substrate, for example, as large as millimeters or even up to centimeters. In order to achieve larger 2D MoS
2 crystal, precursors should be delivered onto the target substrate more uniformly, through which more grains could develop and spread throughout the substrate surface. Grains grow bigger with precursors added, leading to more area coated. Grains spread randomly, thus each of them develops along different crystal orientations to form different crystal shapes. A variety of MoS
2 crystal domain shapes have been synthesized, including triangles, hexagons, truncated triangles, three-point stars, and six-point stars and dendritic ones [
11]. These MoS
2 crystal with different shapes grow to meet up with each other to form crystal boundaries. The CVD process can be divided into five steps [
12]: (a) precursors sublime and are transported downstream by a carrier gas; (b) they diffuse from the bulk vapor toward the substrate; (c) they adsorb onto the surface; (d) adatoms of the precursors diffuse along the surface; (e) they react to form product structures. The first three steps are about how the precursors sublime and are transported onto different sites of the substrate; the last two steps are about how the precursor adatoms diffuse and react to form product. It is obvious that the first three steps determine the density distribution of precursors on the substrate, which further determines the 2D MoS
2 distribution throughout the substrate.
It can be noticed that the precursor transport depends on carrier gas flow [
13]. However, many researchers focused on the last two steps of the process of CVD, rather than the effect of carrier gas flow. Cheng et al. presented research on the kinetic nature of the grain boundary formation in as-grown monolayer MoS
2, revealing the mechanism of 2D MoS
2 grain boundary formation [
14]. Qingqing Ji et al. presented their work to unravel the orientation distribution and merging behavior of 2D MoS
2 crystals [
15]. A 2D MoS
2 crystal shape evaluation was also conducted, showing how the monocrystal MoS
2 crystals evolve and merge with adjacent ones to form large area sheets [
11]. These works significantly broadened and deepened our insight into the mechanism of grain development, monocrystal crystal formation and crystal merging of 2D MoS
2, namely, the last two steps of the process of CVD. Meanwhile, issues of how the carrier gas flow travels along the reaction tube in the CVD process under different conditions have been relatively much less discussed.
In this work, we demonstrated the effect of the carrier gas flow field and precursor vapor distribution in the CVD process of 2D MoS2 by simulation and experiments. The flow field and precursor vapor distribution near the substrate surface was significantly affected by the shape and size of the precursor powder carrier over which the target substrate was placed. What was more, the local flow field near the target substrate was much more complicated compared with the gas flow upstream from the target, which was almost laminar flow. Thus the carrier gas flow field and precursor vapor distribution near the target substrate were the focus in this paper.
2. Experiments
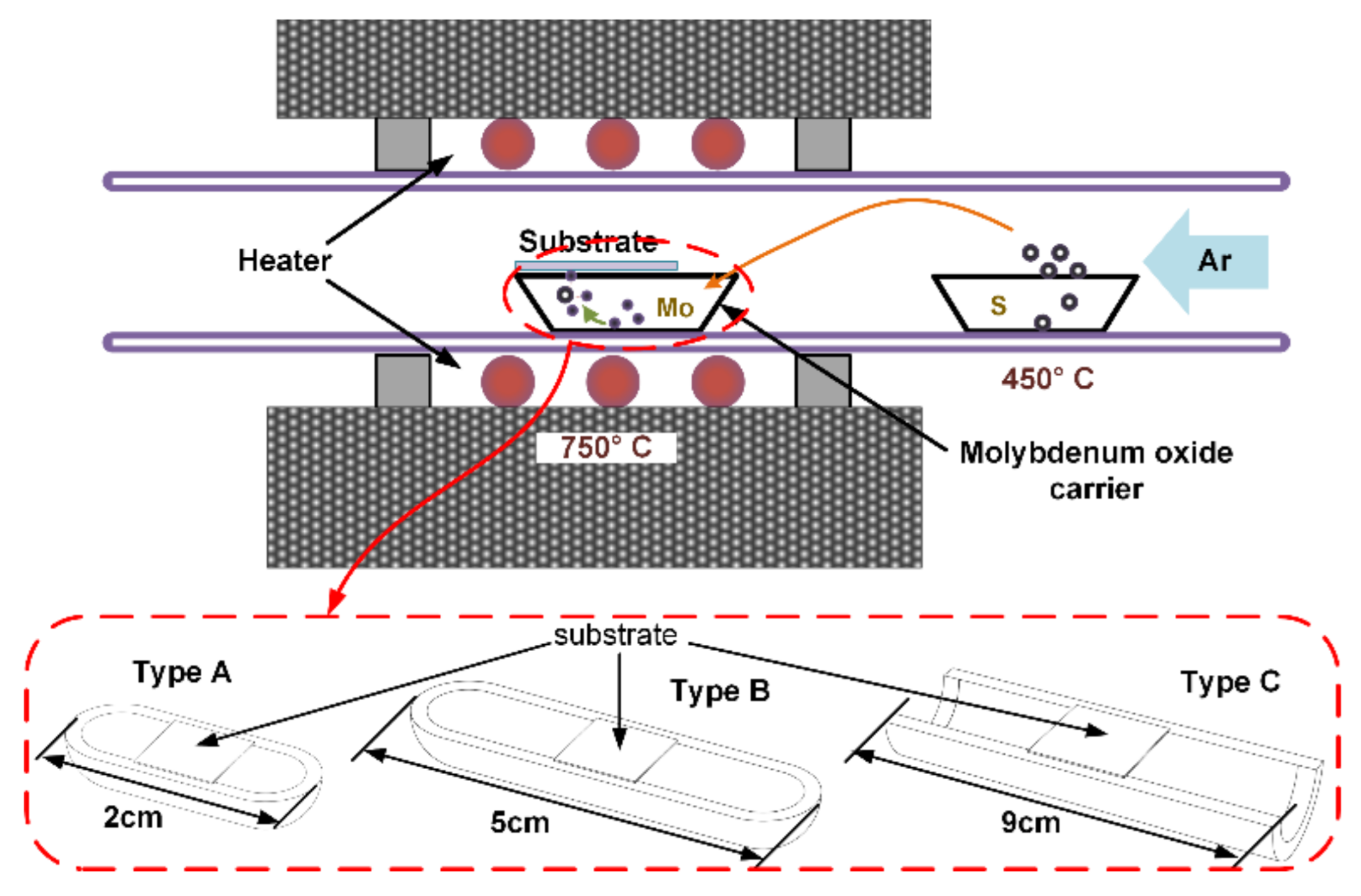
The experiment setup for the CVD process of 2D MoS
2 is presented in
Figure 1. The MoO
3 (purity 99.95%, Shanghai Aladdin Biochemical Technology Co., Ltd., Shanghai, China) powder was placed in a quartz boat to provide Mo source, almost at the center of the quartz tube and the sulfur powder (analytically pure, Damao Chemical Reagent Factory, Tianjin, China) was placed in another quartz boat, upstream from the MoO
3 powder, with a distance of 18 cm. Middle part of the quartz tube was surrounded by a heater to provide temperature control. The temperature in the zone where MoO
3 was placed was set at 750 °C. The temperature where sulfur was placed was about 450 °C, lower than that of MoO
3 due to the distance away from heater zone. The target substrate was placed over the MoO
3 powder, supported by the quartz boat of MoO
3 powder carrier. Carrier gas traveled from the inlet to take both precursors to the surface of the target substrate where they reacted to form MoS
2 crystal.
In our experiment, the MoO3 powder was placed beneath the target substrate and the sulfur powder was placed upstream from the target substrate at a distance of about 18 cm. It can be inferred that the precursor sulfur was adequately evaporated and diffused before reaching the target, meaning that the sulfur vapor concentration could be regarded as uniform in the carrier gas around the target substrate. However, the space between MoO3 powder and the target substrate was only a few millimeters that the molybdenum vapor concentration could hardly attain a stable value before reaching the target substrate. Thus the diffusion and transportation of molybdenum were focused on the target substrate in our research.
As was stated, the sulfur vapor concentration could be regarded uniform in the carrier gas around the target substrate. At the same time, the inlet velocity was usually set rather small that the gas would keep almost stable as the laminar flow in the zone upstream from the target substrate. As a result, the carrier gas flow field around the substrate was the main focus. The quartz boat carrier of MoO
3 powder and target substrate were obstacles that caused perturbation within the carrier gas. Thus, three types of typical quartz boat carrier of MoO
3 powder were selected in our experiment, including a short boat-shaped carrier (type A), a long boat-shaped carrier (type B) and a U-slot carrier (type C), as are illustrated in
Figure 1. The lengths of the quartz boat carriers were 2 cm, 5 cm and 9 cm of type A, B and C, respectively; the width and height were all 1 cm and 0.5 cm, the same for each type.
After deposition of MoS2, several methods were utilized to characterize it. First and foremost, optical imaging was applied to have an overall glance at the 2D MoS2 crystal covering on the target substrate and obtain a general estimation of the 2D MoS2 crystal deposition results. X-ray photoelectron spectroscopy (XPS) measurement was operated to investigate the components of the obtained materials from the CVD process. Raman spectra were utilized to determine whether the deposited crystal sheets were of a few layers or more, which was validated by AFM test.
3. Results and Analysis
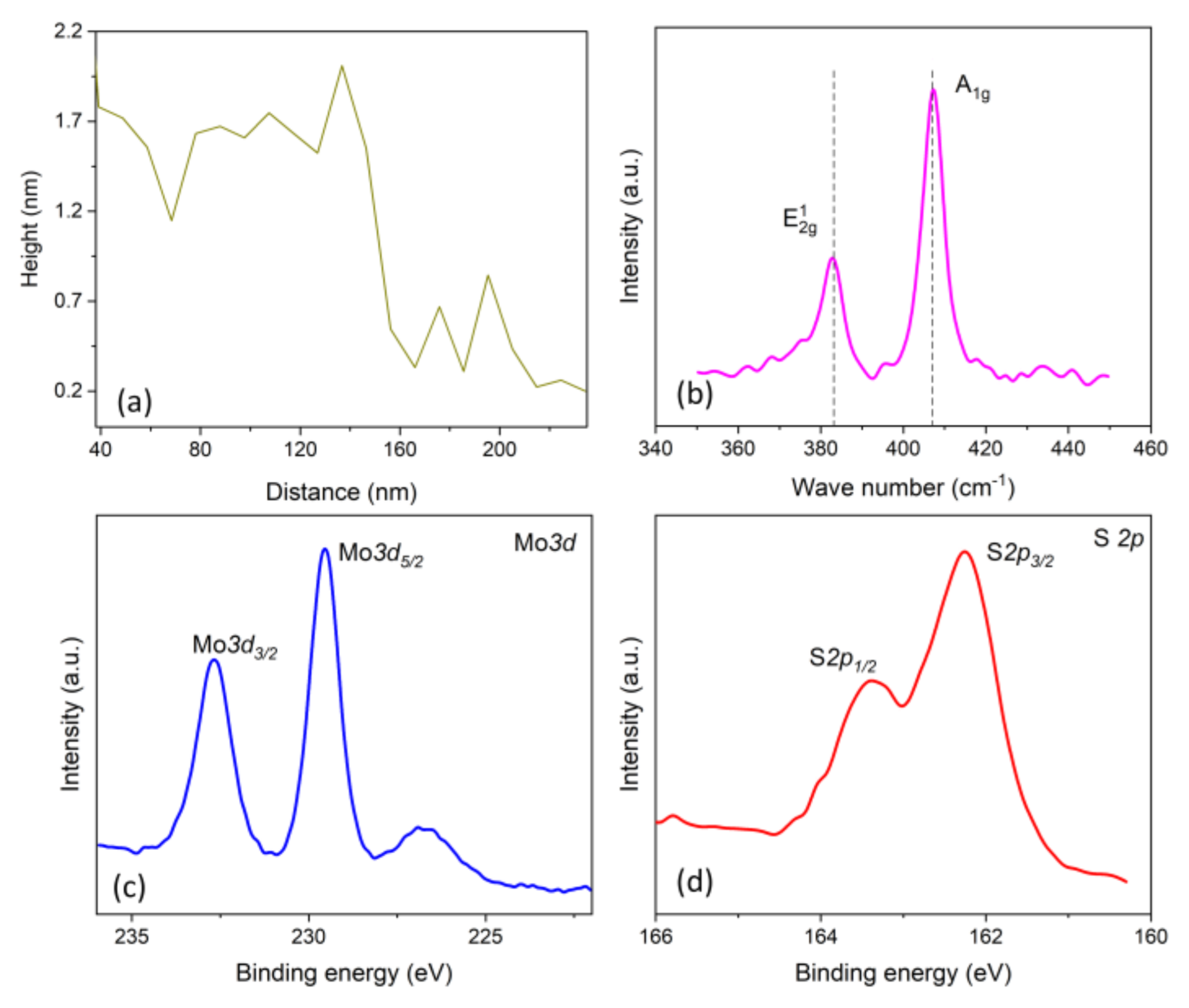
The MoS
2 crystal pieces deposited upon the target surface are shown in
Figure 2. It can be seen that most of the MoS
2 crystal pieces here were triangles, depending on the kinetics of crystal formation, which has been clearly presented [
14]. The single layer MoS
2 crystal was about 0.6 nm in thickness [
16]; as the AFM result shows in
Figure 3a, the MoS
2 crystal pieces obtained in our experiment were a few layers of crystals. The few layers of MoS
2 could also be validated by the Raman result, as shown in
Figure 3b, the Raman data interpretation can be found elsewhere [
17]. Two characteristic peaks relating to the Raman vibration modes can be found in
Figure 3b, in which the E
2g peak is associated with the in-plane vibration mode of molybdenum and sulfur atoms and A
1g is associated with the out-of-plane vibration mode of sulfur atoms [
18]. The number of layers of 2D MoS
2 determined the frequency variation between the E
2g and A
1g modes [
16]. As is presented in
Figure 3b, the frequency variation between these two modes was around 24.8 cm
−1, from 382.3 (E
2g location) to 407.1 (A
1g location), corresponding to four layers of MoS
2 crystal. The results were in accordance with the frequency variation of a few layer MoS
2 demonstrated before, meaning that a few layers of MoS
2 crystal were obtained in our experiment.
Figure 3c,d show the XPS data of the obtained MoS
2, from which the existence of the elements sulfur and molybdenum and the atomic composition ratio for the two elements were confirmed [
19]. The intensity peaks at 229.5 and 232.7 were attributed to the doublet Mo 3d
5/2 and 3d
3/2, while the peaks at 162.3 and 163.4 were attributed to S 2p
1/2 and 2p
3/2, respectively, which can be found in previous works [
18,
19,
20]. The peak intensities ratio of Mo and S were 2.0, revealing the components of MoS
2.
The MoS
2 crystals formed on the substrate over the different types of quartz boat carrier of MoO
3 are shown in
Figure 4. It can be noted that the MoS
2 crystal pieces did not spread over the target surface randomly and uniformly. Instead, they occurred intensively in certain areas of the target surface, leaving other areas sparsely coated by MoS
2, even barely. More MoS
2 crystal pieces were achieved by using quartz boat carrier type A of MoO
3. The downstream zone of the target substrate above MoO
3 carrier type A showed a greater intensity of MoS
2 crystal pieces. Substantial crystal grains appeared in the same zone on the substrate above the MoO
3 carrier type B, most of which, however, did not grow as big as those on the substrate above carrier type A. As for the substrate above the MoO
3 carrier type C, few crystal grains of monocrystal MoS
2 were found and they were not of a large size.
It is known that the surface energy state differs from the substrate edge to the inner substrate surface but the energy state should be roughly the same all around the substrate surface beside those areas near the edge [
21,
22,
23]. On the basis of this conclusion, the MoS
2 crystal domains should have spread over the target surface uniformly in the CVD process, only taking into consideration the energy state of the target surface. Unfortunately, the experiments results presented a rather different MoS
2 crystal domain distribution. It is obvious that crystal formation depends not only on the energy state of the target, but also on the intensity of the precursor vapor which is significantly affected by the local carrier gas flow field.
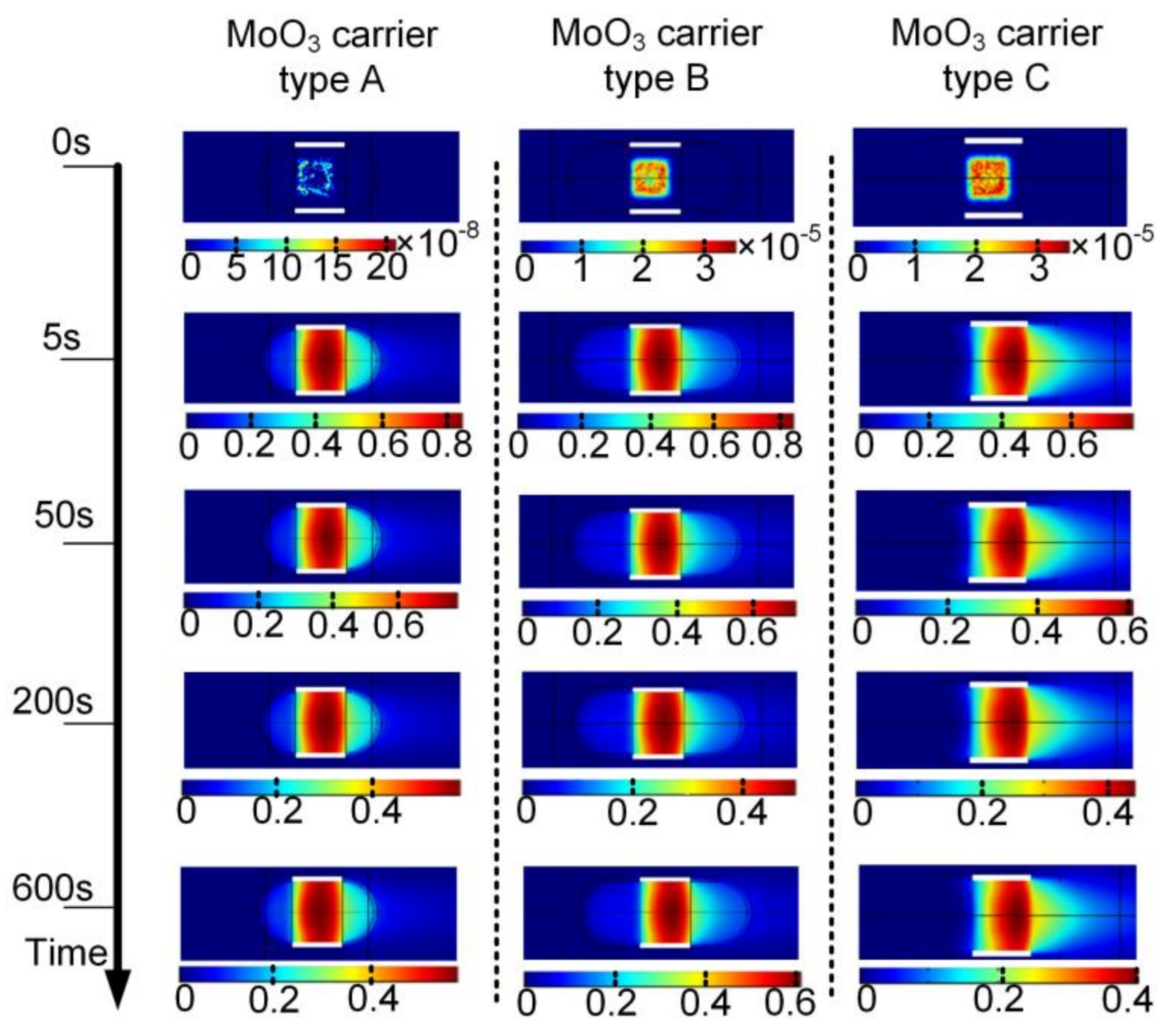
In order to have deeper insight into the CVD process, a simulation was conducted. The ambient temperature and ambient pressure in the simulation were set at 293.15 K and 101.33 kPa, respectively, trying to be close to the real parameters in our lab. The temperature was set at 750 °C to heat the precursor. The inlet velocity of the carrier gas was set at 0.1 m/s. The molybdenum vapor concentration development with the target substrate above the three types of MoO
3 carrier was simulated and is presented in
Figure 5.
From
Figure 5, it can be seen that the molybdenum vapor concentration increased rapidly in the very beginning of the deposition process, namely the first 5 s presented simulation results. After 50 s, the molybdenum vapor concentration changed at a rather small rate. What should be noted is that the shape of the MoO
3 carrier played an important role in influencing the molybdenum vapor concentration. For the molybdenum vapor concentration with MoO
3 carrier type C, the highest value was nearly 33% lower than that with MoO
3 carrier type A and B. The molybdenum vapor concentration distributions near the surface of the target substrate over MoO
3 carriers type A and B were very close to each other; however, taking the experiment results and simulation results in
Figure 5 into consideration together, it can be found that molybdenum vapor concentration was not the only factor that determined the MoS
2 deposition. In other words, a higher concentration of molybdenum vapor alone (when sulfur vapor was kept unchanged) may not lead to a larger size of MoS
2 crystal pieces or more of the target area being coated with MoS
2. It was clear that the molybdenum vapor concentration was higher with MoO
3 carrier type B, while the experiment results of MoS
2 coating rate on the target surface were the opposite.
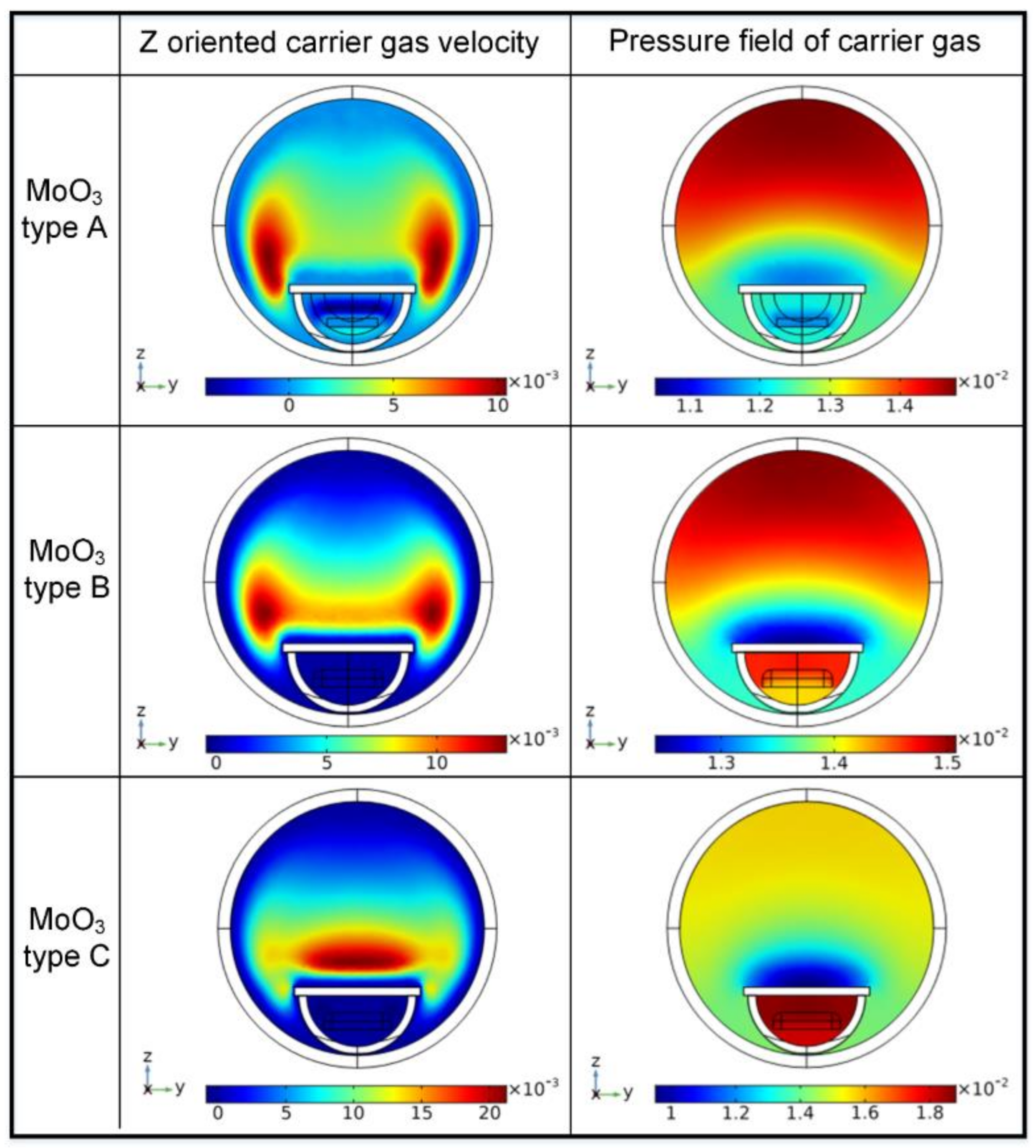
In order to find out the influence of the local carrier gas flow field, the gas velocity and pressure were simulated as is presented in
Figure 6. As was stated, the focus concerned the local carrier gas flow field near the surface of target substrate. It can be noted from
Figure 6 that the z oriented carrier gas flow velocity was about 4.0 × 10
−3 m/s, −2.0 × 10
−4 m/s and −5.6 × 10
−4 m/s, with the substrate over MoO
3 carrier type A, B and C, respectively. Here, plus z oriented gas flow velocity represented the carrier gas flowing towards the surface of target substrate; on the other hand, minus z oriented gas flow velocity represented the carrier gas flowing backwards from the surface of target substrate. It can be inferred that carrier gas flow towards the target surface brought precursor adatoms to the surface where reaction and deposition occurred; while carrier gas flow backwards from the target surface took precursor adatoms away from the target surface. The former process should contribute to the reaction and deposition because it increases the adatoms required by the reaction process and makes the adatoms stay longer where the reaction takes place. In general, higher pressure results in a more efficient chemical reaction process [
24,
25]; however, this deduction was not supported by the CVD process of MoS
2. It can be seen from
Figure 6 that the pressure near the target surface with substrate on MoO
3 carrier C was the highest, but resulted in the least deposition of MoS
2 crystal. It can be inferred that pressure in this CVD process affected the results very little.