Plasma-Activated Polyvinyl Alcohol Foils for Cell Growth
Abstract
1. Introduction
2. Materials and Methods
2.1. Material
2.2. Preparation of PVA Foil
2.3. Surface Modification
2.4. Analytical Methods
2.5. Cell–Material Interaction—A Study of the Adhesion and Proliferation of the Cells
3. Results and Discussion
3.1. Optimizing the Preparation Conditions of the PVA Foil
3.2. Characterization of Pristine and Modified PVA Foils
3.2.1. Surface Wettability
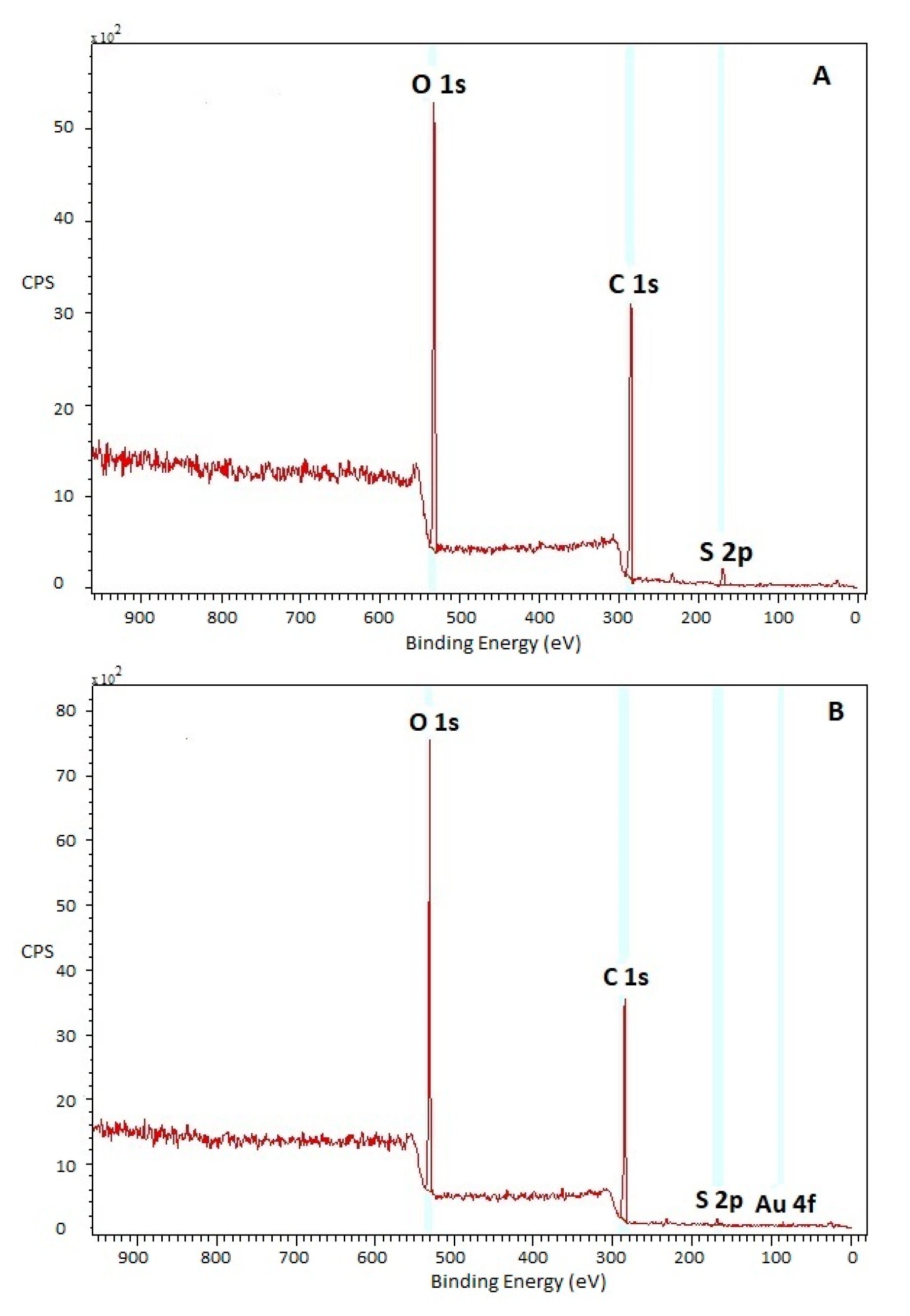
3.2.2. Chemical Composition
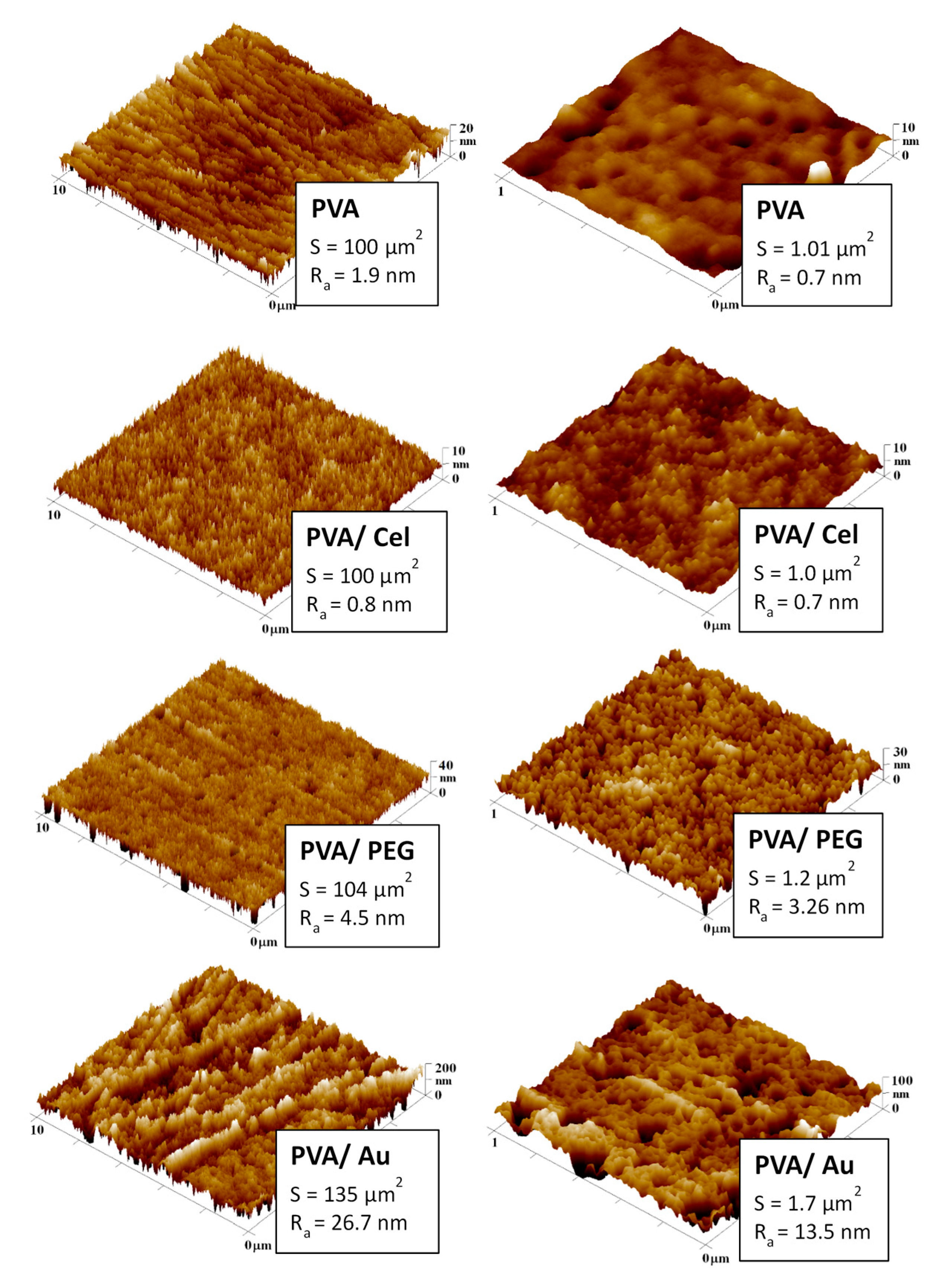
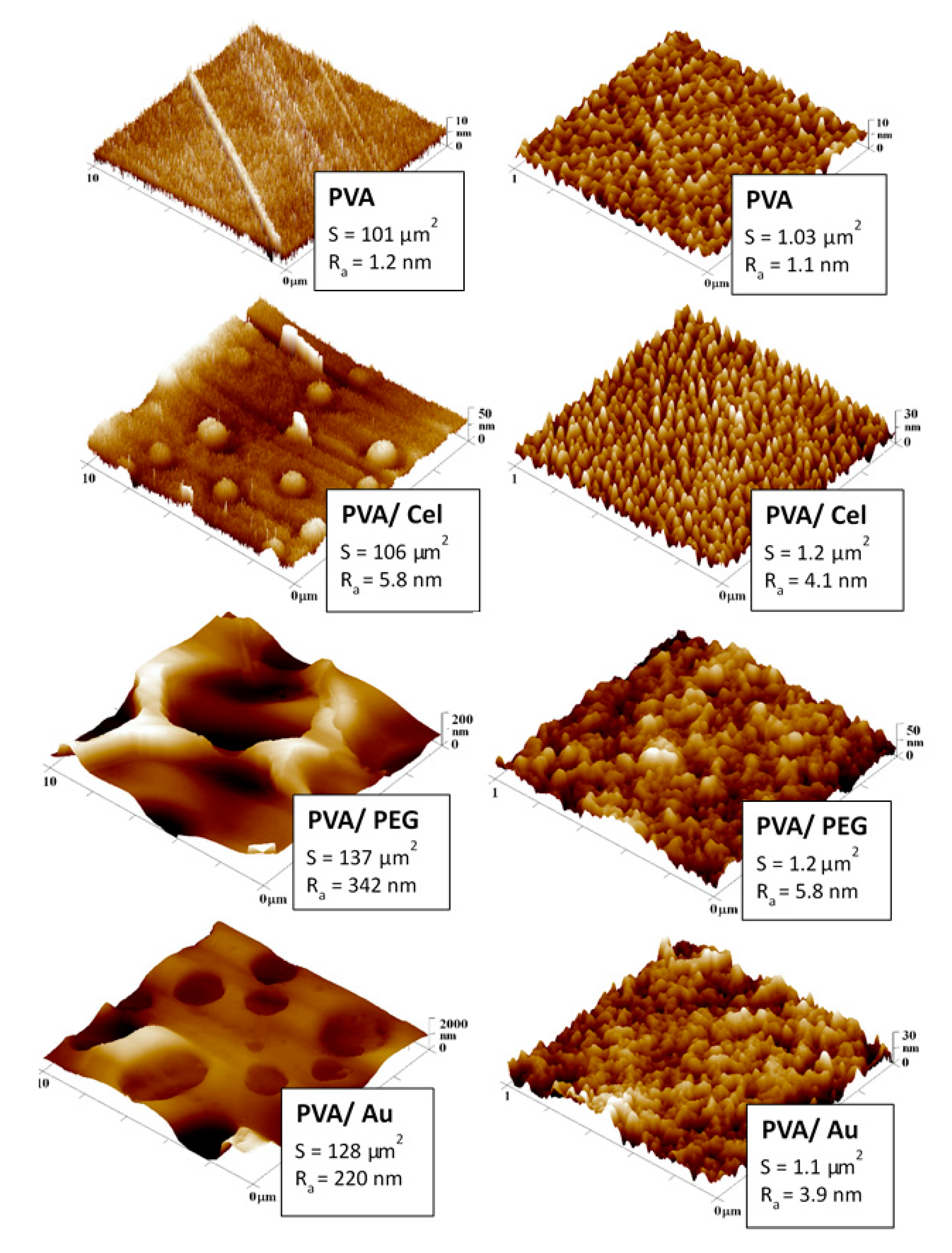
3.2.3. Surface Morphology and Roughness, Thickness of Samples
3.2.4. Swelling
3.2.5. Mechanical Properties
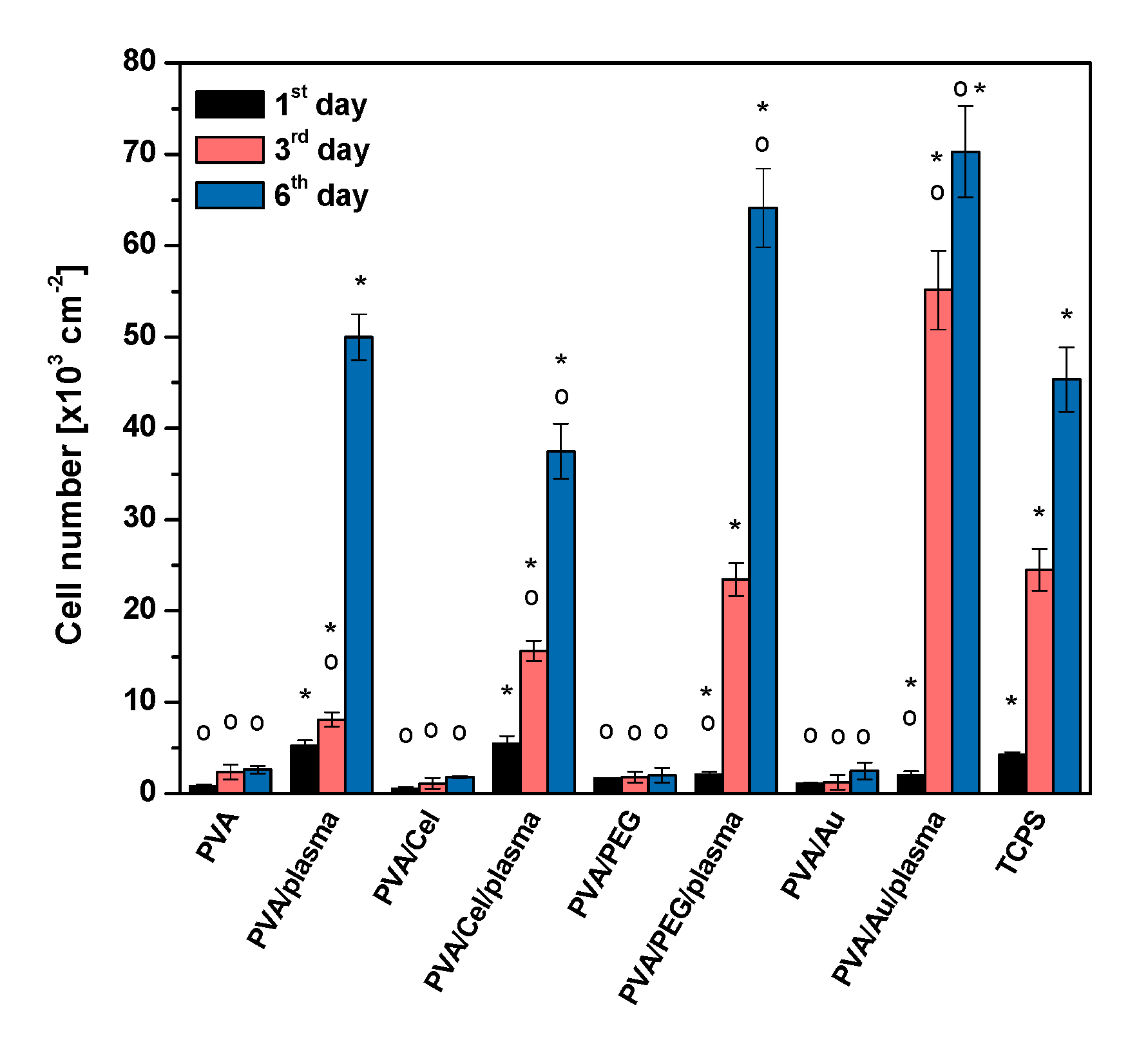
3.2.6. Biocompatibility
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Menazea, A.A.; Mostafa, A.M.; Al-Ashkar, E.A. Effect of nanostructured metal oxides (CdO, Al2O3, Cu2O) embedded in PVA via Nd:YAG pulsed laser ablation on their optical and structural properties. J. Mol. Struct. 2020, 1203, 1–7. [Google Scholar] [CrossRef]
- Ratner, B.D.; Bryant, S.J. Biomaterials: Where we have been and where we are going. Annu. Rev. Biomed. Eng. 2004, 6, 41–78. [Google Scholar] [CrossRef] [PubMed]
- O’Brien, F.J. Biomaterials & scaffolds for tissue engineering. Mater. Today 2011, 14, 88–95. [Google Scholar]
- Deb, P.; Deoghare, A.B.; Borah, A.; Barua, E.; Lala, S.D. Scaffold development using biomaterials: A review. Mater. Today Proc. 2018, 5, 12909–12919. [Google Scholar] [CrossRef]
- Caló, E.; Khutoryanskiy, V.V. Biomedical applications of hydrogels: A review of patents and commercial products. Eur. Polym. J. 2015, 65, 252–267. [Google Scholar] [CrossRef]
- Hoffman, A.S. Hydrogels for biomedical applications. Adv. Drug Deliv. Rev. 2012, 64, 18–23. [Google Scholar] [CrossRef]
- Coviello, T.; Matricardi, P.; Marianecci, C.; Alhaique, F. Polysaccharide hydrogels for modified release formulations. J. Control. Release 2007, 119, 5–24. [Google Scholar] [CrossRef]
- Ottenbrite, R.M. Biomedical Applications of Hydrogels Handbook; Ottenbrite, R.M., Park, K., Okano, T., Eds.; Springer Science: New York, NY, USA, 2010. [Google Scholar]
- Premkumar, P.S. Preparation and electrical studies on pure and oxygen plasma treated polyvinyl alcohol films. J. Mater. Res. Technol. 2019, 8, 2232–2237. [Google Scholar] [CrossRef]
- Ahmed, E.M. Hydrogel: Preparation, characterization, and applications: A review. J. Adv. Res. 2015, 6, 105–121. [Google Scholar] [CrossRef]
- Discher, D.E.; Mooney, D.J.; Zandstra, P.W. Growth factors, matrices, and forces combine and control stem cells. Science 2009, 324, 1673–1677. [Google Scholar] [CrossRef]
- Farida, O.; Mansour, F.; Habib, M.; Robinson, J.; Tarleton, S. Investigating the sorption influence of poly(vinyl alcohol) (PVA) at different crosslinking content. J. Environ. Chem. Eng. 2016, 4, 293–298. [Google Scholar] [CrossRef]
- Raca, V.; Lević, S.; Balanč, B.; Graells, B.O.; Bijelić, G. PVA Cryogel as model hydrogel for iontophoretic transdermal drug delivery investigations. Comparison with PAA/PVA and PAA/PVP interpenetrating networks. Colloid. Surf. B 2019, 180, 441–448. [Google Scholar] [CrossRef] [PubMed]
- Hoare, T.R.; Kohane, D.S. Hydrogels in drug delivery: Progress and challenges. Polymer 2008, 49, 1993–2007. [Google Scholar] [CrossRef]
- Boateng, J.S.; Matthews, K.H.; Stevens, H.N.E.; Eccleston, G.M. Wound healing dressings and drug delivery systems: A review. J. Pharm. Sci. 2008, 9, 2892–2923. [Google Scholar] [CrossRef] [PubMed]
- Kima, M.-S.; Oh, G.-W.; Jang, Y.-M.; Ko, S.-C.; Park, W.-S.; Choi, I.-W.; Kim, Y.-M.; Jung, W.-K. Antimicrobial hydrogels based on PVA and diphlorethohydroxycarmalol (DPHC) derived from brown algaIshige okamurae: An in vitro and in vivo study for wound dressing application. Mat. Sci. Eng. C Mater. 2020, 107, 1–12. [Google Scholar]
- Fang, H.; Wang, J.; Li, L.; Xu, L.; Wu, Y.; Wang, Y.; Fei, X.; Tian, J.; Li, Y. A novel high-strength poly(ionic liquid)/PVA hydrogel dressing for antibacterial applications. Chem. Eng. J. 2019, 365, 153–164. [Google Scholar] [CrossRef]
- Kamoun, E.A.; Kenawy, E.-R.S.; Chen, X. A review on polymeric hydrogel membranes for wound dressing applications: PVA-based hydrogel dressings. J. Adv. Res. 2017, 8, 217–233. [Google Scholar] [CrossRef]
- Goenka, S.; Sant, V.; Sant, S. Graphene based nanomaterials for drug delivery and tissue engineering. J. Control. Release 2014, 173, 75–88. [Google Scholar] [CrossRef]
- Žáková, P.; Slepičková Kasálková, N.; Kolská, Z.; Leitner, J.; Karpíšková, J.; Stibor, I.; Slepička, P.; Švorčík, V. Cytocompatibility of amine functionalized carbon nanoparticles grafted on polyethylene. Mat. Sci. Eng. C Mater. 2016, 60, 394–401. [Google Scholar] [CrossRef]
- Slepička, P.; Malá, Z.; Rimpelová, S.; Švorčík, V. Antibacterial properties of modified biodegradable PHB non-woven fabric. Mat. Sci. Eng. C Mater. 2016, 65, 364–368. [Google Scholar] [CrossRef]
- Motealleh, A.; Kehr, N.S. Nanocomposite hydrogels and their applications in tissue engineering. Adv. Healthc. Mater. 2017, 6, 1–19. [Google Scholar]
- DeMerlis, C.C.; Schoneker, D.R. Review of the oral toxicity of polyvinyl alcohol (PVA). Food Chem. Toxicol. 2003, 41, 319–326. [Google Scholar] [CrossRef]
- Thong, C.C.; Teo, D.C.L.; Ng, C.K. Application of polyvinyl alcohol (PVA) in cement-based composite materials: A review of its engineering properties and microstructure behavior. Constr. Build. Mater. 2016, 107, 172–180. [Google Scholar] [CrossRef]
- Tong, R.; Zhao, Y.; Chen, M.; Peng, Y. Multimode interferometer based on no-core fiber with GQDs-PVA composite coating for relative humidity sensing. Opt. Fiber Technol. 2019, 48, 242–247. [Google Scholar] [CrossRef]
- Baker, M.I.; Walsh, S.P.; Schwartz, Z.; Boyan, B.D. A review of polyvinyl alcohol and its uses in cartilage and orthopedic applications. J. Biomed. Mater. Res. B 2012, 100B, 1451–1457. [Google Scholar] [CrossRef] [PubMed]
- Teodorescu, M.; Bercea, M.; Morariu, S. Biomaterials of PVA and PVP in medical and pharmaceutical applications: Perspectives and challenge. Biotechnol. Adv. 2019, 37, 109–131. [Google Scholar] [CrossRef] [PubMed]
- Siegel, J.; Polívková, M.; Slepičková Kasálková, N.; Kolská, Z.; Švorčík, V. Properties of silver nanostructure-coated PTFE and its biocompatibility. Nanoscale Res. Lett. 2013, 8, 1–10. [Google Scholar] [CrossRef]
- Figueiredo, K.C.S.; Alves, T.L.M.; Borges, C.P. Poly(vinyl alcohol) films crosslinked by glutaraldehyde under mild conditions. J. Appl. Polym. Sci. 2009, 111, 3074–3080. [Google Scholar] [CrossRef]
- Kim, K.-J.; Lee, S.-B.; Han, N.-W. Kinetics of crosslinking reaction of PVA membrane with glutaraldehyde. Korean J. Chem. Eng. 1994, 11, 41–47. [Google Scholar] [CrossRef]
- Liu, Z.; Dong, Y.; Men, H.; Jiang, M.; Tong, J.; Zhou, J. Post-crosslinking modification of thermoplastic starch/PVA blend films by using sodium hexametaphosphate. Carbohydr. Polym. 2012, 89, 473–474. [Google Scholar] [CrossRef]
- Han, B.; Li, J.; Chen, C.; Xu, C.; Wickramasinghe, S.R. Effects of degree of formaldehyde acetal treatment and maleic acid crosslinking on solubility and diffusivity of water in PVA membranes. Chem. Eng. Res. Des. 2003, 81, 1385–1392. [Google Scholar] [CrossRef]
- Guo, R.; Hu, C.; Li, B.; Jiang, Z. Pervaporation separation of ethylene glycol/water mixtures through surface crosslinked PVA membranes: Coupling effect and separation performance analysis. J. Membr. Sci. 2007, 289, 191–198. [Google Scholar] [CrossRef]
- Reznickova, A.; Novotna, Z.; Kolska, Z.; Slepickova Kasalkova, N.; Rimpelova, S.; Svorcik, V. Enhanced adherence of mouse fibroblast and vascular cells to plasma modified polyethylene. Mat. Sci. Eng. C Mater. 2015, 52, 259–266. [Google Scholar] [CrossRef] [PubMed]
- Slepičková Kasálková, N.; Slepička, P.; Bačáková, L.; Sajdl, P.; Švorčík, V. Biocompatibility of plasma nanostructured biopolymers. Nucl. Instrum. Meth. B 2013, 307, 642–646. [Google Scholar] [CrossRef]
- Mishra, S.; Bajpai, R.; Katare, R.; Bajpai, A.K. Radiation induced crosslinking effect on semiinterpenetrating polymer networks of poly(vinyl alcohol). Expess Polym. Lett. 2007, 1, 407–415. [Google Scholar] [CrossRef]
- Simpson, R.L.; Nazhat, S.N.; Blaker, J.J.; Bismarck, A.; Hille, R.; Boccaccini, R.; Hansen, U.N.; Amisa, A.A. A comparative study of the effects of different bioactive fillers in PLGA matrix composites and their suitability as bone substitute materials: A thermo-mechanical and in vitro investigation. J. Mech. Behav. Biomed. Mater. 2015, 50, 277–289. [Google Scholar] [CrossRef]
- Nawawi, W.M.F.W.; Lee, K.Y.; Kontturi, E.; Murphy, R.J.; Bismarck, A. Chitin nanopaper from mushroom extract: Natural composite of nanofibers and glucan from a single biobased source. ACS Sustain. Chem. Eng. 2019, 7, 6492–6496. [Google Scholar] [CrossRef]
- Woodward, R.T.; Markoulidis, F.; De Luca, F.; Anthony, D.; Malko, D.; McDonald, T.; Shaffer, M.; Bismarck, A. Carbon foams from emulsion-templated reduced graphene oxide polymer composites: Electrodes for supercapacitor devices. J. Mater. Chem. A 2018, 6, 1840–1849. [Google Scholar] [CrossRef]
- Kamoun, E.A.; Chen, X.; Eldin, M.S.M.; Kenawy, E.-R.S. Crosslinked poly(vinyl alcohol) hydrogels for wound dressing applications: A review of remarkably blended polymers. Arab. J. Chem. 2015, 8, 1–14. [Google Scholar] [CrossRef]
- Benabbas, R.; Sanchez-Ballester, N.M.; Bataille, B.; Sharkawi, T.; Soulairol, I. Development and pharmaceutical performance of a novel co-processed excipient of alginic acid and microcrystalline cellulose. Powder Technol. 2021, 378, 576–584. [Google Scholar] [CrossRef]
- Mohanapriya, S.; Raj, V. Tuning biological properties of poly (vinyl alcohol) with amino acids and studying its influence on osteoblastic cell adhesion. Mat. Sci. Eng. C Mater. 2018, 86, 70–82. [Google Scholar] [CrossRef] [PubMed]
- Faxälv, L.; Ekblad, T.; Liedberg, B.; Lindahl, T. Blood compatibility of photografted hydrogel coatings. Acta Biomater. 2010, 6, 2599–2608. [Google Scholar] [CrossRef] [PubMed]
- Liu, F.; Mishbak, H.; Bartolo, P. Hybrid polycaprolactone/hydrogel scaffold fabrication and in-process plasma treatment using PABS. Int. J. Bioprint. 2019, 5, 1–9. [Google Scholar]
- Nitschke, M.; Zschoche, S.; Baier, A.; Simon, F.; Werner, C. Low pressure plasma immobilization of thin hydrogel films onpolymer surfaces. Surf. Coat. Technol. 2004, 185, 120–125. [Google Scholar] [CrossRef]
- Liang, C.; Wang, H.; Yang, J.; Cai, Y.; Hu, X.; Yang, Y.; Li, B.; Li, H.; Li, C.; Yang, X. Femtosecond laser-induced micropattern and Ca/P deposition on Ti implant surface and its acceleration on early osseointegration. Appl. Mater. Interfaces 2013, 5, 8179–8186. [Google Scholar] [CrossRef]
- Abdal-hay, A.; Hussein, K.H.; Casettari, L.; Khalil, K.A.; Hamdy, A.S. Fabrication of novel high performance ductile poly(lactic acid) nanofiber scaffold coated with poly(vinyl alcohol) for tissue engineering applications. Mat. Sci. Eng. C Mater. 2016, 60, 143–150. [Google Scholar] [CrossRef]
- Lee, P.-J.; Ho, C.-C.; Hwang, C.-S.; Ding, S.-J. Improved physicochemical properties and biocompatibility of stainless steel implants by PVA/ZrO2-based composite coatings. Surf. Coat. Technol. 2014, 258, 374–380. [Google Scholar] [CrossRef]
- Borzenkov, M.; Määttänen, A.; Ihalainen, P.; Collini, M.; Cabrini, E.; Dacarro, G.; Pallavicini, P.; Chirico, G. Photothermal effect of gold nanostar patterns inkjet-printed on coated paper substrates with different permeability. Beilstein J. Nanotechnol. 2016, 7, 1480–1485. [Google Scholar] [CrossRef]
- Thoniyot, P.; Tan, M.J.; Karim, A.A.; Young, D.J.; Loh, X.J. Nanoparticle–hydrogel composites: Concept, design, and applications of these promising, multi-functional materials. Adv. Sci. 2015, 2, 1–13. [Google Scholar] [CrossRef]
- Borzenkov, M.; Moros, M.; Tortiglione, C.; Bertoldi, S.; Contessi, N.; Faré, S.; Taglietti, A.; D’Agostino, A.; Pallavicini, P.; Collini, M.; et al. Fabrication of photothermally active poly(vinyl alcohol) films with gold nanostars for antibacterial applications. Beilstein J. Nanotechnol. 2018, 9, 2040–2048. [Google Scholar] [CrossRef]
- Mahmoud, N.N.; Hikmat, S.; Ghith, D.A.; Hajeer, M.; Hamadneh, L.; Qattan, D.; Khalil, E.A. Gold nanoparticles loaded into polymeric hydrogel for wound healing inrats: Effect of nanoparticles’ shape and surface modification. Int. J. Pharm. 2019, 565, 174–186. [Google Scholar] [CrossRef] [PubMed]
- Denoziere, G.; Ku, D.N.; Carticept Medical, Inc. (Assignee) Methods of Producing PVA Hydrogel Implants and Related Devices. U.S. Patent No. 8,038,920 B2, 18 October 2011. [Google Scholar]
- Slepička, P.; Přibyl, M.; Fajstavr, D.; Ulbrich, P.; Siegel, J.; Řezníčková, A.; Švorčík, V. Grafting of platinum nanostructures on biopolymer at elevated temperature. Colloids Surf. A 2018, 546, 316–325. [Google Scholar] [CrossRef]
- Slepička, P.; Elashnikov, R.; Ulbrich, P.; Staszek, M.; Kolská, Z.; Švorčík, V. Stabilization of sputtered gold and silver nanoparticles in PEG colloid solution. J. Nanopart. Res. 2015, 17, 1–15. [Google Scholar] [CrossRef]
- Travnickova, M.; Pajorova, J.; Zarubova, J.; Krocilova, N.; Molitor, M.; Bacakova, L. The Influence of Negative Pressure and of the Harvesting site on the characteristics of human adipose tissue-derived stromal cells from lipoaspirates. Stem Cells Int. 2020, 2020, 1–13. [Google Scholar] [CrossRef]
- Slepičková Kasálková, N.; Slepička, P.; Sajdl, P.; Švorčík, V. Surface changes of biopolymers PHB and PLLA induced by Ar+ plasma treatment and wet etching. Nucl. Instrum. Meth. B 2014, 332, 63–67. [Google Scholar] [CrossRef]
- Yang, T. Mechanical and Swelling Properties of Hydrogels. Ph.D. Thesis, KTH Royal Institute of Technology, Stockholm, Sweden, 2012. [Google Scholar]
- Anushree, D. Characterization of Polyethylene Glycol Hydrogels for Biomedical Applications. Master’s Theses, Louisiana State University, Baton Rouge, LA, USA, 2007. Available online: https://digitalcommons.lsu.edu/gradschool_theses/3502 (accessed on 4 November 2020).
- Kask, A.M. Degradable Poly(ethylene glycol) Hydrogels for 2D and 3D Cell Culture, Technical-Documents/Articles; SigmaAldrich.com. Available online: https://www.sigmaaldrich.com/technical-documents/articles/material-matters/degradable-polyethylene-glycol-hydrogels.html?gclid=EAIaIQobChMI1KCmkPnp7AIVQ-R3Ch3p1w-tEAMYASAAEgKLNvD_BwE (accessed on 4 November 2020).
- Takakura, K.; Takayama, G.; Ukida, J. Ultraviolet-induced crosslinking of poly(viny alcohol) in the presence of sensitizers. J. Appl. Polym. Sci. 1965, 9, 3217–3224. [Google Scholar] [CrossRef]


| Sample | Contact Angle (°) | |||
|---|---|---|---|---|
| PVA | PVA/Cel | PVA/PEG | PVA/Au | |
| Plasma-untreated | 71.7 ± 1.3 | 73.1 ± 1.7 | 28.0 ± 1.4 | 24.3 ± 1.6 |
| Plasma-treated/aged | 62.3 ± 1.2 | 60.4 ± 0.8 | 22.5 ± 0.9 | 24.7 ± 0.6 |
| Sample | Atomic Concentrations (at.%) | ||||
|---|---|---|---|---|---|
| C(1s) | O(1s) | N(1s) | S(2p) | Au(4f) | |
| PVA | 75.6 | 22.4 | – | 2.0 | – |
| PVA/plasma | 69.6 | 28.0 | 0.2 | 2.2 | – |
| PVA/Cel | 73.3 | 24.3 | – | 2.4 | – |
| PVA/Cel/plasma | 69.0 | 27.1 | 1.7 | 2.2 | – |
| PVA/PEG | 68.6 | 29.1 | – | 2.3 | – |
| PVA/PEG/plasma | 65.2 | 32.3 | 0.6 | 1.9 | – |
| PVA/Au | 67.9 | 29.9 | – | 2.2 | 0.1 |
| PVA/Au/plasma | 66.3 | 31.5 | 0.4 | 1.7 | 0.1 |
| Sample | Degree of Swelling | |
|---|---|---|
| Q 24 | Q 48 | |
| PVA | 1 | 1.1 |
| PVA/plasma | 0.5 | 0.5 |
| PVA/Cel | 1 | 1 |
| PVA/Cel/plasma | 0.33 | 0.35 |
| PVA/PEG | 3 | 3 |
| PVA/PEG/plasma | 2 | 2 |
| PVA/Au | 3 | 3 |
| PVA/Au/plasma | 2.33 | 2.31 |
| Sample | Mechanical Properties | |||
|---|---|---|---|---|
| Young’s Modulus (MPa) | Ultimate Strength (MPa) | Elongation at Yield Strength (%) | Elongation at Break (%) | |
| PVA | 25.4 ± 3.7 | 12.4 ± 2.2 | 55.0 ± 10.0 | 63.0 ± 100. |
| PVA/Cel | 26.1 ± 12.1 | 24.1 ± 3.9 | 135.0 ± 9.0 | 181.0 ± 22.0 |
| PVA/PEG | 197.9 ± 43.5 | 21.0 ± 1.9 | 162.0 ± 34.0 | 22 ± 22.0 |
| PVA/Au | 65.6 ± 18.4 | 17.2 ± 3.6 | 215.0 ± 47.0 | 302.0 ± 55 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Slepičková Kasálková, N.; Slepička, P.; Ivanovská, B.; Trávníčková, M.; Malinský, P.; Macková, A.; Bačáková, L.; Švorčík, V. Plasma-Activated Polyvinyl Alcohol Foils for Cell Growth. Coatings 2020, 10, 1083. https://doi.org/10.3390/coatings10111083
Slepičková Kasálková N, Slepička P, Ivanovská B, Trávníčková M, Malinský P, Macková A, Bačáková L, Švorčík V. Plasma-Activated Polyvinyl Alcohol Foils for Cell Growth. Coatings. 2020; 10(11):1083. https://doi.org/10.3390/coatings10111083
Chicago/Turabian StyleSlepičková Kasálková, Nikola, Petr Slepička, Barbora Ivanovská, Martina Trávníčková, Petr Malinský, Anna Macková, Lucie Bačáková, and Václav Švorčík. 2020. "Plasma-Activated Polyvinyl Alcohol Foils for Cell Growth" Coatings 10, no. 11: 1083. https://doi.org/10.3390/coatings10111083
APA StyleSlepičková Kasálková, N., Slepička, P., Ivanovská, B., Trávníčková, M., Malinský, P., Macková, A., Bačáková, L., & Švorčík, V. (2020). Plasma-Activated Polyvinyl Alcohol Foils for Cell Growth. Coatings, 10(11), 1083. https://doi.org/10.3390/coatings10111083







