Cold Gas Spraying of a High-Entropy CrFeNiMn Equiatomic Alloy
Abstract
1. Introduction
2. Materials and Methods
3. Results and Discussion
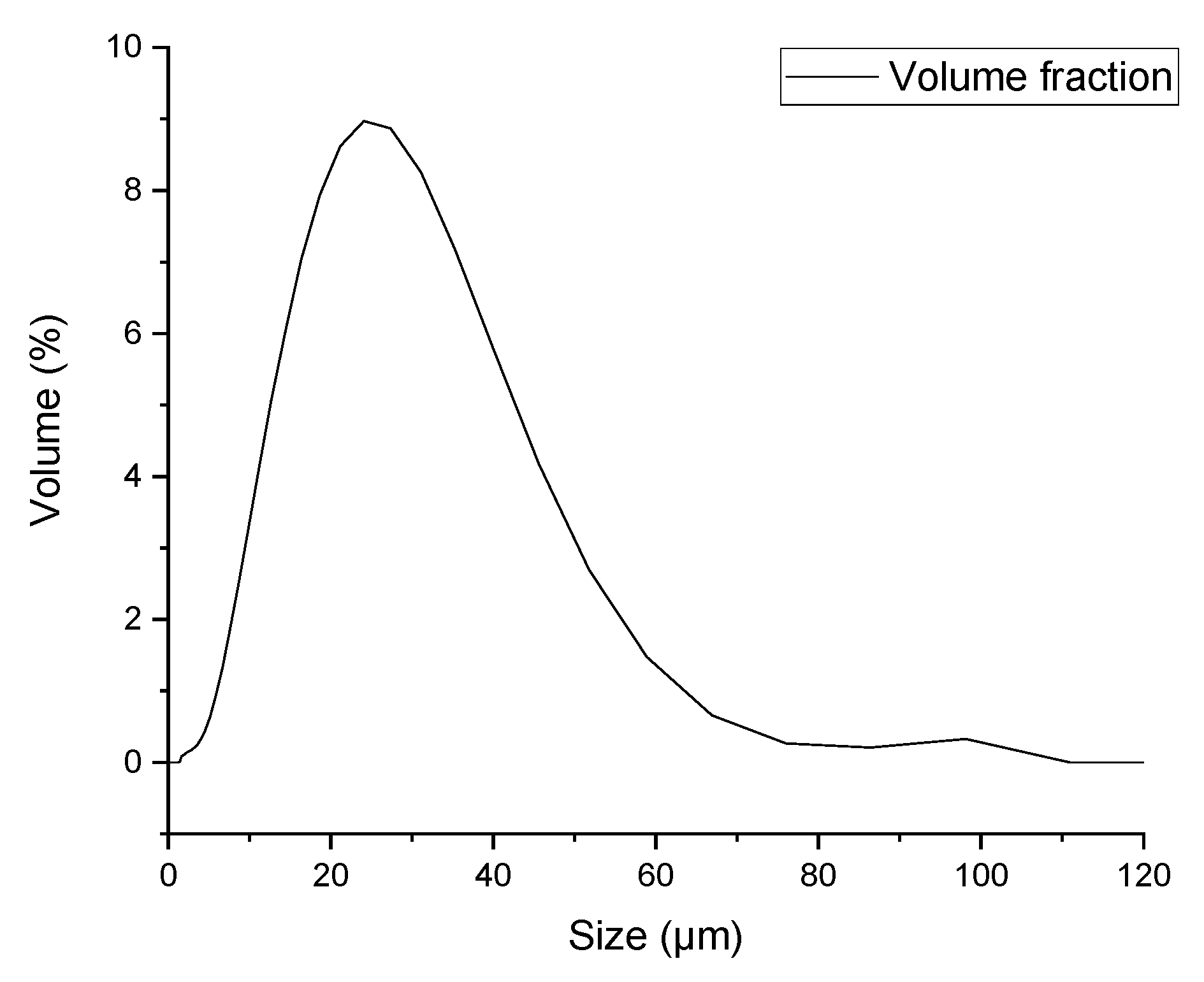
3.1. Powder Characterization
3.2. Coating Thickness, Density, and Porosity
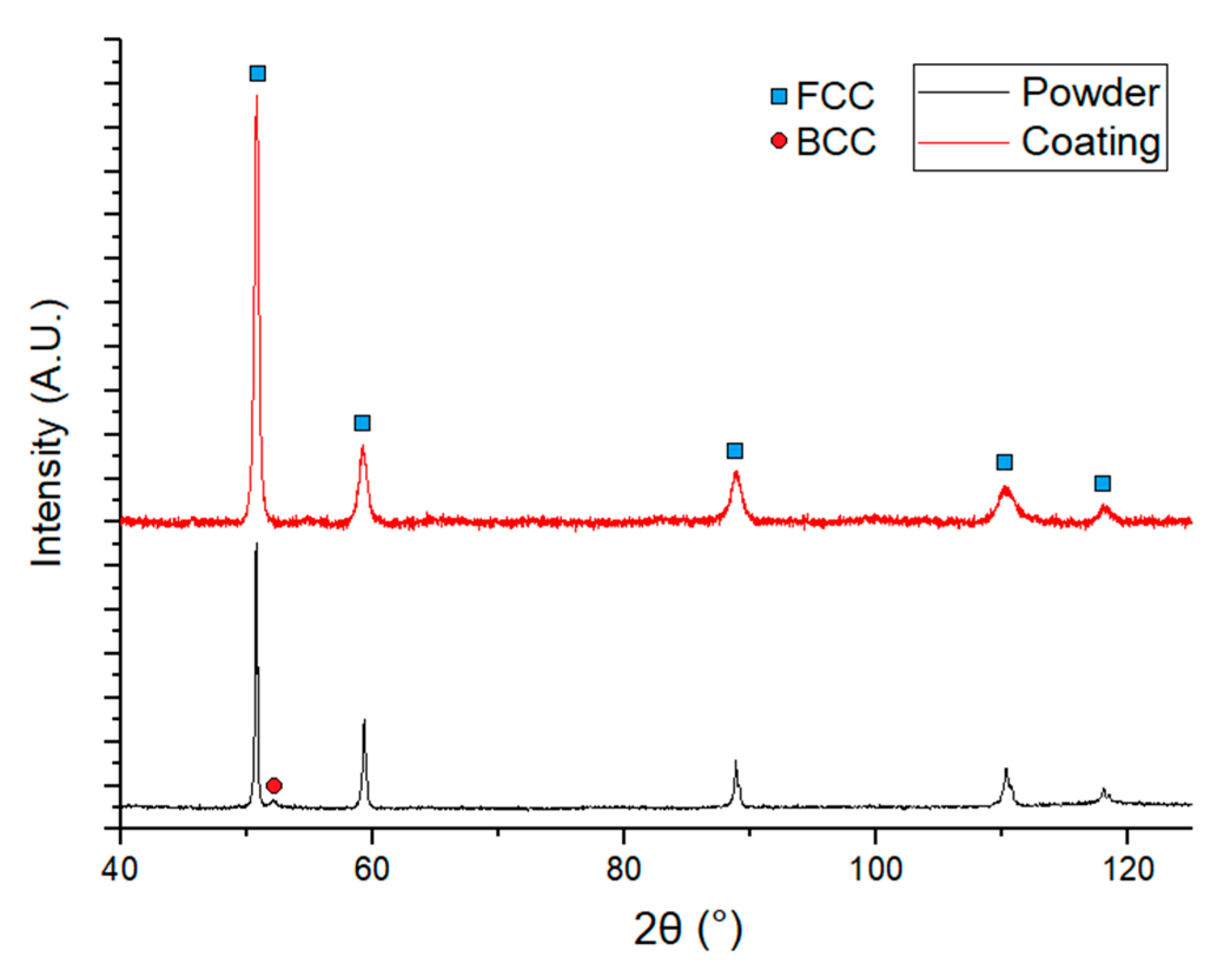
3.3. Phase Structure
3.4. Chemical Composition
3.5. Microstructure
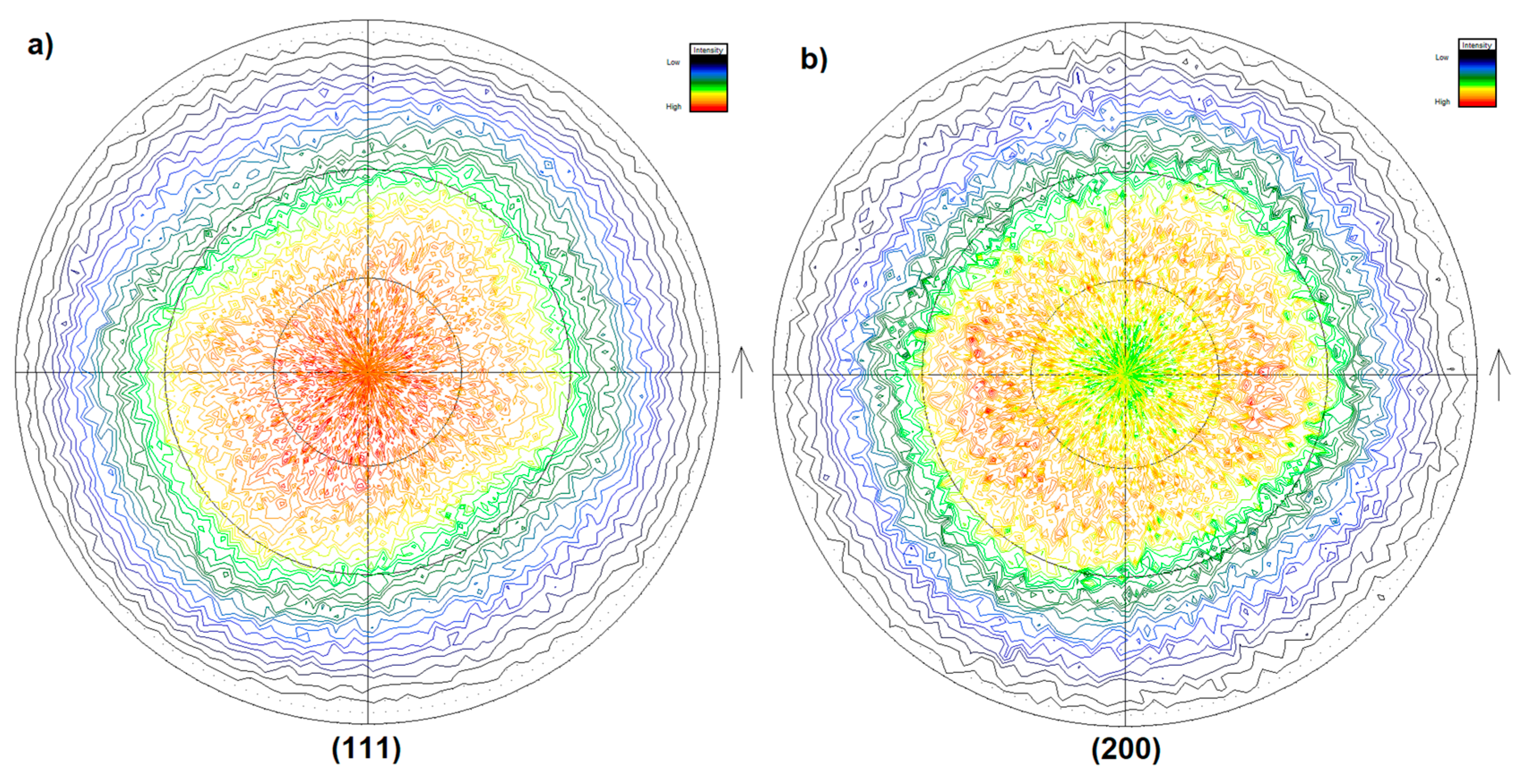
3.6. Texture
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Yeh, J.-W.; Chen, S.-K.; Lin, S.-J.; Gan, J.-Y.; Chin, T.-S.; Shun, T.-T.; Tsau, C.-H.; Chang, S.-Y. Nanostructured high-entropy alloys with multiple principal elements: Novel alloy design concepts and outcomes. Adv. Eng. Mater. 2004, 6, 299–303. [Google Scholar] [CrossRef]
- Cantor, B.; Chang, I.T.H.; Knight, P.; Vincent, A.J.B. Microstructural development in equiatomic multicomponent alloys. Mater. Sci. Eng. A 2004, 375–377, 213–218. [Google Scholar] [CrossRef]
- Kozak, R.; Sologubenko, A.; Steurer, W. Single-phase high-entropy alloys—An overview. Z. Für Krist. Cryst. Mater. 2015, 230. [Google Scholar] [CrossRef]
- Li, C.; Hu, X.; Yang, T.; Kumar, N.; Wirth, B.; Zinkle, S. Neutron irradiation response of a Co-free high entropy alloy. J. Nucl. Mater. 2019, 527. [Google Scholar] [CrossRef]
- Wu, Z.; Bei, H.; Otto, F.; Pharr, G.M.; George, E.P. Recovery, recrystallization, grain growth and phase stability of a family of FCC-structured multi-component equiatomic solid solution alloys. Intermetallics 2014, 46, 131–140. [Google Scholar] [CrossRef]
- Kumar, N.; Li, C.; Leonard, K.; Bei, H.; Zinkle, S. Microstructural stability and mechanical behavior of FeNiMnCr high entropy alloy under ion irradiation. Acta Mater. 2016, 113, 230–244. [Google Scholar] [CrossRef]
- Wu, Z.; Bei, H. Microstructures and mechanical properties of compositionally complex Co-free FeNiMnCr 18 FCC solid solution alloy. Mater. Sci. Eng. A 2015, 640, 217–224. [Google Scholar] [CrossRef]
- Stepanov, N.; Shaysultanov, D.; Tikhonovsky, M.; Salishchev, G. Tensile properties of the Cr–Fe–Ni–Mn non-equiatomic multicomponent alloys with different Cr contents. Mater. Des. 2015, 87, 60–65. [Google Scholar] [CrossRef]
- Lehtonen, J.; Ge, Y.; Ciftci, N.; Heczko, O.; Uhlenwinkel, V.; Hannula, S.-P. Phase structures of gas atomized equiatomic CrFeNiMn high entropy alloy powder. J. Alloys Compd. 2019. submitted. [Google Scholar]
- Papyrin, A.; Kosarev, V.; Klinkov, S.; Alkhimov, A.; Fomin, V.M. Cold Spray Technology; Papyrin, A., Kosarev, V., Klinkov, S., Alkhimov, A., Fomin, V.M., Eds.; Elsevier: Amsterdam, The Netherlands, 2006. [Google Scholar]
- Alkhimov, A. Gas-Dynamic Spraying Method for Applying a Coating. U.S. Patent US5302414A, 12 April 1994. [Google Scholar]
- Rokni, M.R.; Nutt, S.R.; Widener, C.A.; Crawford, G.A.; Champagne, V.K. Structure-properties relations in high-pressure cold-sprayed deposits. In Cold-Spray Coatings, Recent Trends and Future Perspectives; Cavaliere, P., Ed.; Springer International Publishing AG: Cham, Switzerland, 2018; Chapter 5; pp. 143–192. [Google Scholar]
- Oksa, M.; Turunen, E.; Suhonen, T.; Varis, T.; Hannula, S.-P. Optimization and characterization of high velocity oxy-fuel sprayed coatings: Techniques, materials, and applications. Coatings 2011, 1, 17–52. [Google Scholar] [CrossRef]
- Cavaliere, P. Cold-Spray Coatings, Recent Trends and Future Perspectives; Cavaliere, P., Ed.; Springer International Publishing AG: Cham, Switzerland, 2018. [Google Scholar] [CrossRef]
- Villafuerte, J. Modern Cold Spray, Materials, Process, and Applications; Villafuerte, J., Ed.; Springer International Publishing AG: Cham, Switzerland, 2015. [Google Scholar]
- Maev, R.G.; Leshchynsky, V. Introduction to Low Pressure Gas Dynamic Spray: Physics and Technology; John Wiley & Sons: Heppenheim, Germany, 2009. [Google Scholar]
- Champagne, V.K. The Cold Spray Materials Deposition Process: Fundamentals and Applications; Champagne, V.K., Ed.; Elsevier: Amsterdam, The Netherlands, 2007. [Google Scholar]
- Koivuluoto, H.; Honkanen, M.; Vuoristo, P. Cold-sprayed copper and tantalum coatings—Detailed FESEM and TEM analysis. Surf. Coat. Technol. 2010, 204, 2353–2361. [Google Scholar] [CrossRef]
- Koivuluoto, H.; Milanti, A.; Bolelli, G.; Lusvarghi, L.; Vuoristo, P. High-pressure cold-sprayed Ni and Ni–Cu coatings: Improved structures and corrosion properties. J. Therm. Spray Technol. 2014, 23, 98–103. [Google Scholar] [CrossRef]
- Koivuluoto, H.; Vuoristo, P. Structural analysis of cold-sprayed nickel-based metallic and metallic-ceramic coatings. J. Therm. Spray Coat. 2010, 19, 975–989. [Google Scholar] [CrossRef]
- Yeom, H.; Maier, B.; Johnson, G.; Dabney, T.; Lenling, M.; Sridharan, K. High temperature oxidation and microstructural evolution of cold spray chromium coatings on Zircaloy-4 in steam environments. J. Nucl. Mater. 2019, 526, 151737. [Google Scholar] [CrossRef]
- Magaro, P.; Marino, A.L.; di Schino, A.; Furgiuele, F.; Maletta, C.; Pileggi, R.; Sgambitterra, E.; Testani, C.; Tului, M. Effect of process parameters on the properties of Stellite-6 coatings deposited by cold gas dynamic spray. Surf. Coat. Technol. 2019, 377, 124934. [Google Scholar] [CrossRef]
- Tului, M.; Bartuli, C.; Bezzon, A.; Marino, A.L.; Marra, F.; Matera, S.; Bulci, G. Amorphous steel coatings deposited by cold-gas spraying. Metals 2019, 9, 678. [Google Scholar] [CrossRef]
- Sun, W.; Bhowmik, A.; Tan, A.W.; Li, R.; Xue, F.; Marinescu, I.; Liu, E. Improving microstructural and mechanical characteristics of cold-sprayed Inconel 718 deposits via local induction heat treatment. J. Alloys Compd. 2019, 797, 1268–1279. [Google Scholar] [CrossRef]
- Ma, W.; Xie, Y.; Chen, C.; Fukanuma, H.; Wang, J.; Ren, Z.; Huang, R. Microstructural and mechanical properties of high-performance Inconel 718 alloy by cold spraying. J. Alloys Compd. 2019, 792, 456–467. [Google Scholar] [CrossRef]
- Huang, J.; Ma, W.; Xie, Y.; Fukanuma, H.; Zhang, K.; Wang, G.; Huang, R. Influence of cold gas spray processing conditions on the properties of 316L stainless steel coatings. Surf. Eng. 2019. [Google Scholar] [CrossRef]
- Assadi, H.; Kreye, H.; Gärtner, F.; Klassen, T. Cold spraying—A materials perspective. Acta Mater. 2016, 116, 382–407. [Google Scholar] [CrossRef]
- Couto, M.; Dosta, S.; Guilemany, J.M. Comparison of the mechanical and electrochemical properties of WC-17 and 12Co coatings onto Al7075-T6 obtained by high velocity oxy-fuel and cold gas spraying. Surf. Coat. Technol. 2015, 268, 180–189. [Google Scholar] [CrossRef]
- Yue, T.; Xie, H.; Lin, X.; Yang, H.; Meng, G. Microstructure of laser re-melted AlCoCrCuFeNi high entropy alloy coatings produced by plasma spraying. Entropy 2013, 15, 2833–2845. [Google Scholar] [CrossRef]
- Löbel, M.; Lindner, T.; Kohrt, C.; Lampke, T. Processing of AlCoCrFeNiTi high entropy alloy by atmospheric plasma spraying. In IOP Conference Series: Materials Science and Engineering; IOP Publishing: Bristol, UK, 2017; Volume 181. [Google Scholar] [CrossRef]
- Ang, A.; Berndt, C.; Sesso, M.L.; Anupam, A.; Kottada, R.; Murty, B. Plasma-sprayed high entropy alloys: Microstructure and properties of AlCoCrFeNi and MnCoCrFeNi. Metall. Mater. Trans. A 2015, 46, 791–800. [Google Scholar] [CrossRef]
- Hsu, W.; Murakami, H.; Yeh, J.; Yeh, A.; Shimoda, K. On the study of thermal-sprayed Ni0.2Co0.6Fe0.2CrSi0.2AlTi0.2 HEA overlay coating. Surf. Coat. Technol. 2017, 316, 71–74. [Google Scholar] [CrossRef]
- Yin, S.; Li, W.; Song, B.; Yan, X.; Kuang, M.; Xu, Y.; Wen, K.; Lupoi, R. Deposition of FeCoNiCrMn high entropy alloy (HEA) coating via cold spraying. J. Mater. Sci. Technol. 2019, 35, 1003–1007. [Google Scholar] [CrossRef]
- Schmidt, T.; Assadi, H.; Gärtner, F.; Richter, H.; Stoltenhoff, T.; Kreye, H.; Klassen, T. From particle acceleration to impact and bonding in cold spraying. J. Therm. Spray Tech. 2009, 18, 794–808. [Google Scholar] [CrossRef]
- Schmidt, T.; Gärtner, F.; Assadi, H.; Kreye, H. Development of a generalized parameter window for cold spray deposition. Acta Materialia 2006, 54, 729–742. [Google Scholar] [CrossRef]
- Henao, J.; Concustell, A.; Cano, I.; Cinca, N.; Dosta, S.; Guilemany, J.M. Influence of cold gas spray process conditions on the microstructure of Fe-based amorphous coatings. J. Alloys Compd. 2005, 622, 995–999. [Google Scholar] [CrossRef]
- Klinkov, S.V.; Kosarev, V.F.; Shikalov, V.S. Influence of nozzle velocity and powder feed rate on the coating mass and deposition efficiency in cold spraying. Surf. Coat. Tech. 2019, 367, 231–243. [Google Scholar] [CrossRef]
- Buchheit, T.E.; Carroll, J.D.; Clark, B.G.; Boyce, B.L. Evaluating deformation-induced grain orientation change in a polycrystal during in situ tensile deformation using EBSD. Microscopy Microanal. 2015, 21, 969–984. [Google Scholar] [CrossRef]
- Chen, C.; Yan, X.; Xie, Y.; Huang, R.; Kuang, M.; Ma, W.; Zhao, R.; Wang, J.; Liu, M.; Ren, Z.; et al. Microstructure evolution and mechanical properties of maraging steel 300 fabricated by cold spraying. Mater. Sci. Eng. A 2019, 743, 482–493. [Google Scholar] [CrossRef]
- Jakupi, P.; Keech, P.G.; Barker, I.; Ramamurthy, S.; Jacklin, R.L.; Shoesmith, D.W.; Moser, D.E. Characterization of commercially cold sprayed copper coatings and determination of the effects of impacting copper powder velocities. J. Nucl. Mater. 2015, 466, 1–11. [Google Scholar] [CrossRef]




| Powder Feed Rate | Feed Rate 1 (rpm) | Feed Rate 2 (rpm) | |||
|---|---|---|---|---|---|
| Coating Number (#) | 1 | 2 | 3 | 4 | 5 |
| Gas feed pressure | 50 | 55 | 60 | 55 | 60 |
| Thickness (µm) | 311 ± 24 | 234 ± 23 | 348 ± 15 | 489 ± 31 | 380 ± 31 |
| Porosity (%) | 5.7 ± 1 | 7.4 ± 1.9 | 3.3 ± 0.8 | 6.4 ± 0.9 | 8.9 ± 1.7 |
| Surface hardness (HV0.3) | 306 ± 5 | 307 ± 14 | 314 ± 10 | 303 ± 6 | 242 ± 12 |
| Cross section hardness | 255 ± 9 | 265 ± 9 | 304 ± 10 | 303 ± 6 | 242 ± 12 |
| Particle roundness | 0.48 ± 0.1 | 0.47 ± 0.1 | 0.45 ± 0.1 | 0.47 ± 0.1 | 0.48 ± 0.1 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lehtonen, J.; Koivuluoto, H.; Ge, Y.; Juselius, A.; Hannula, S.-P. Cold Gas Spraying of a High-Entropy CrFeNiMn Equiatomic Alloy. Coatings 2020, 10, 53. https://doi.org/10.3390/coatings10010053
Lehtonen J, Koivuluoto H, Ge Y, Juselius A, Hannula S-P. Cold Gas Spraying of a High-Entropy CrFeNiMn Equiatomic Alloy. Coatings. 2020; 10(1):53. https://doi.org/10.3390/coatings10010053
Chicago/Turabian StyleLehtonen, Joonas, Heli Koivuluoto, Yanling Ge, Aapo Juselius, and Simo-Pekka Hannula. 2020. "Cold Gas Spraying of a High-Entropy CrFeNiMn Equiatomic Alloy" Coatings 10, no. 1: 53. https://doi.org/10.3390/coatings10010053
APA StyleLehtonen, J., Koivuluoto, H., Ge, Y., Juselius, A., & Hannula, S.-P. (2020). Cold Gas Spraying of a High-Entropy CrFeNiMn Equiatomic Alloy. Coatings, 10(1), 53. https://doi.org/10.3390/coatings10010053






