Phytochemical Composition and In Vitro Antimicrobial Activity of Essential Oils from the Lamiaceae Family against Streptococcus agalactiae and Candida albicans Biofilms
Abstract
1. Introduction
2. Results
2.1. Agar Disk Diffusion Assay
2.2. Qualitative and Semi-Quantitative Analysis of EOs
2.3. Minimum Inhibitory Concentration (MIC)
2.4. Determination of the Fractional Inhibitory Concentration Index (FICI)
2.5. Time–Kill Studies
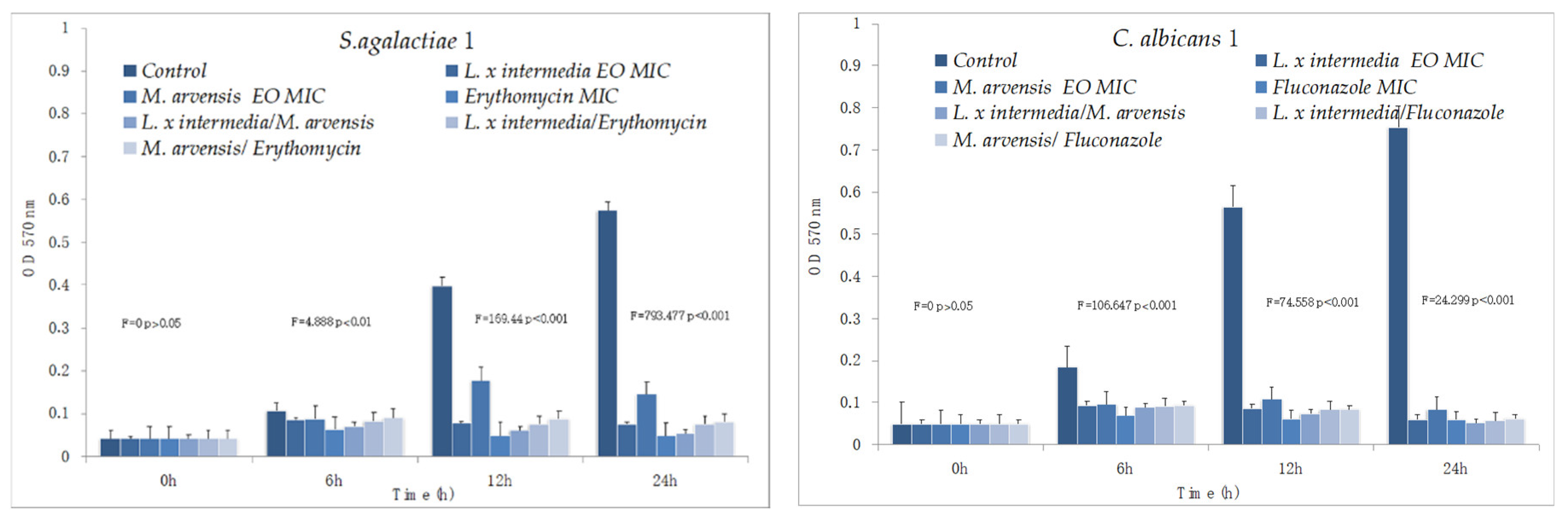
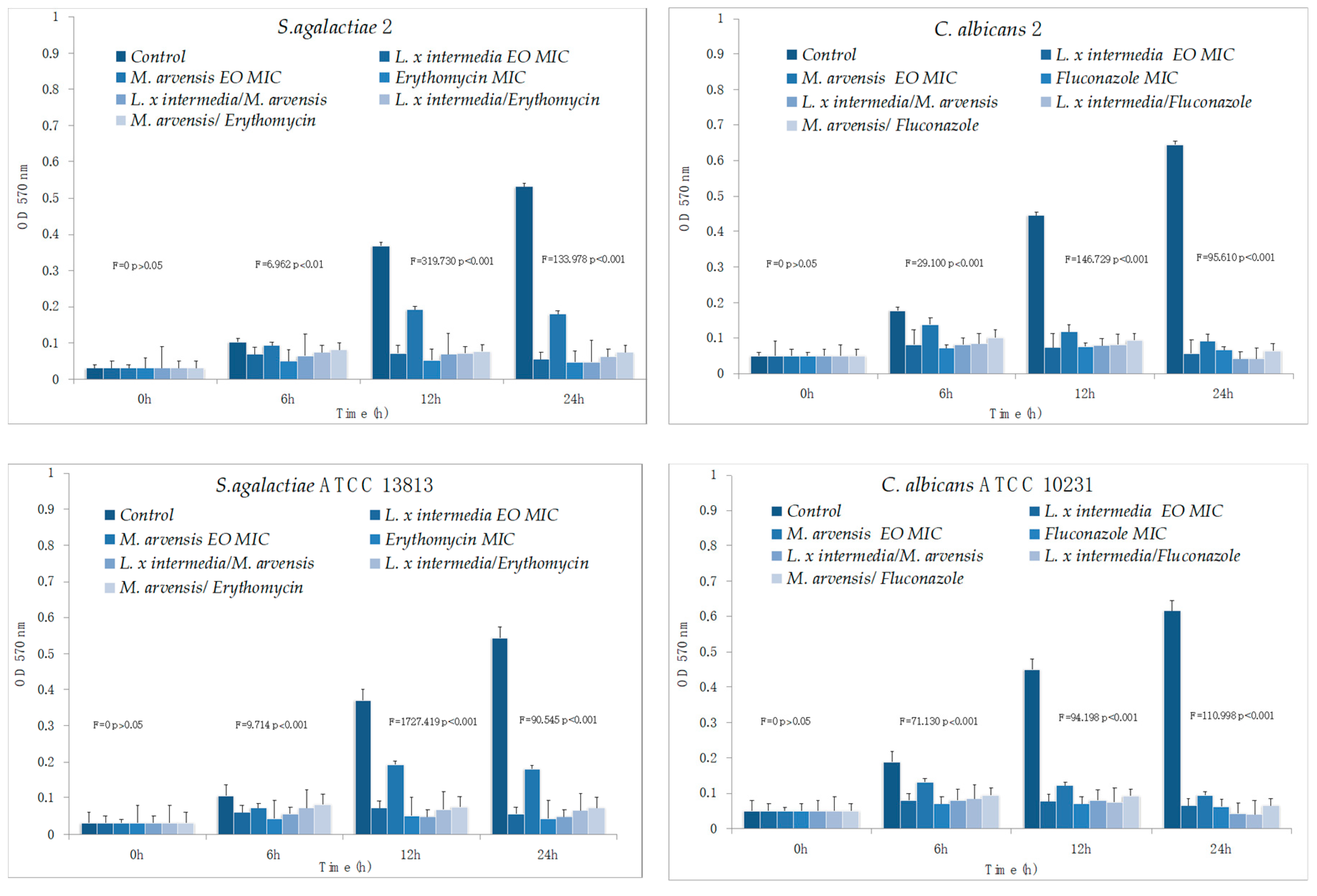
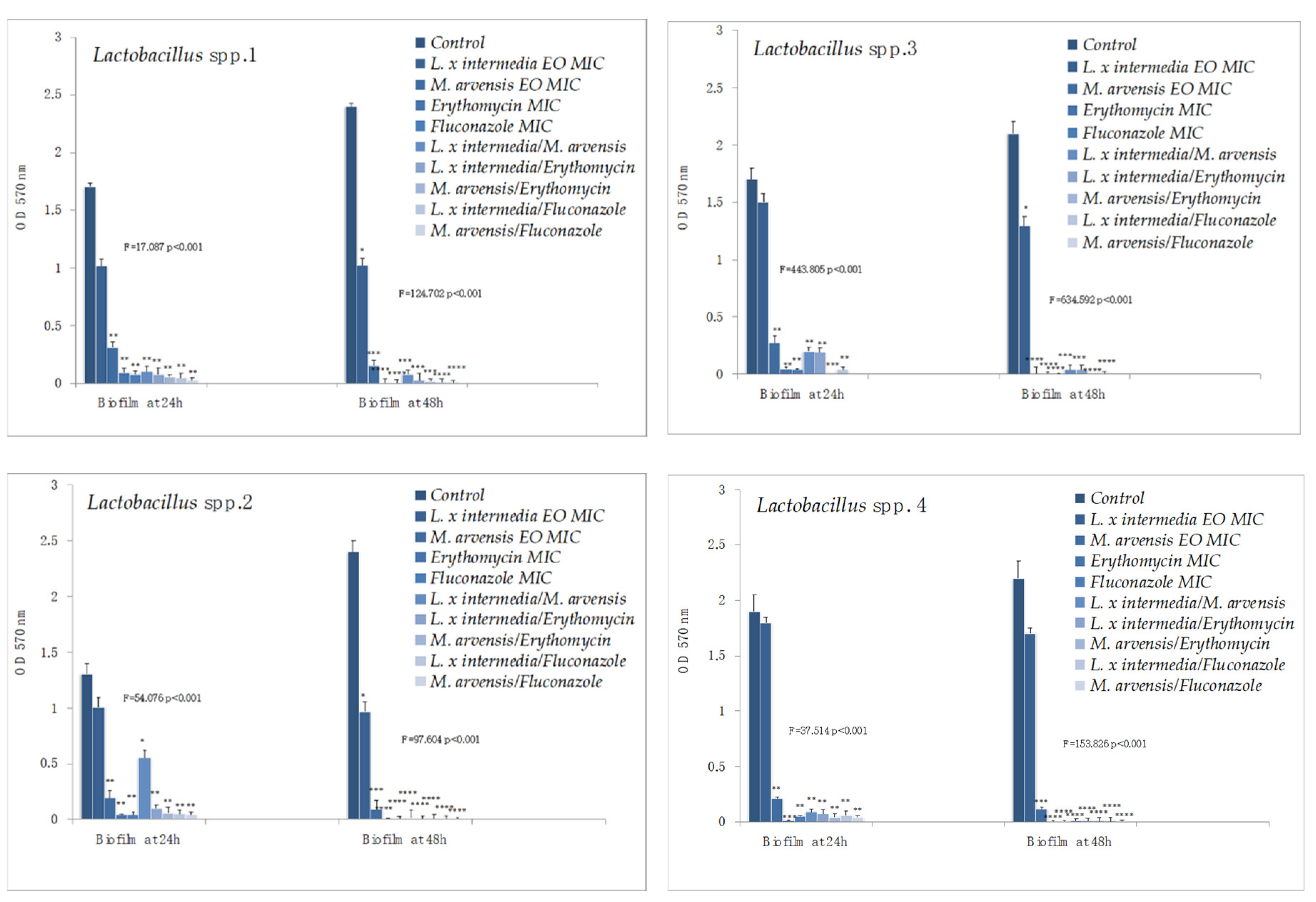
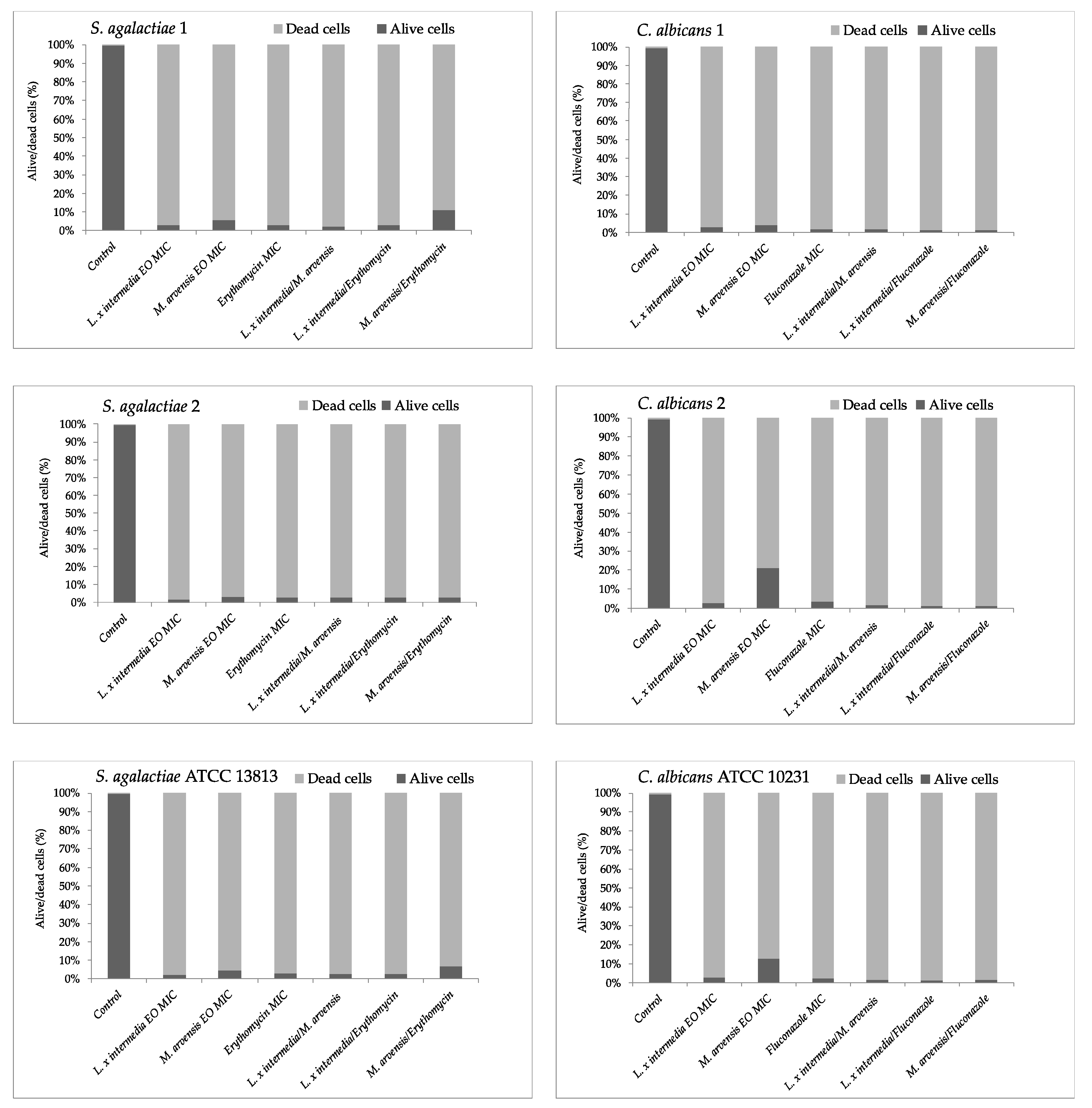
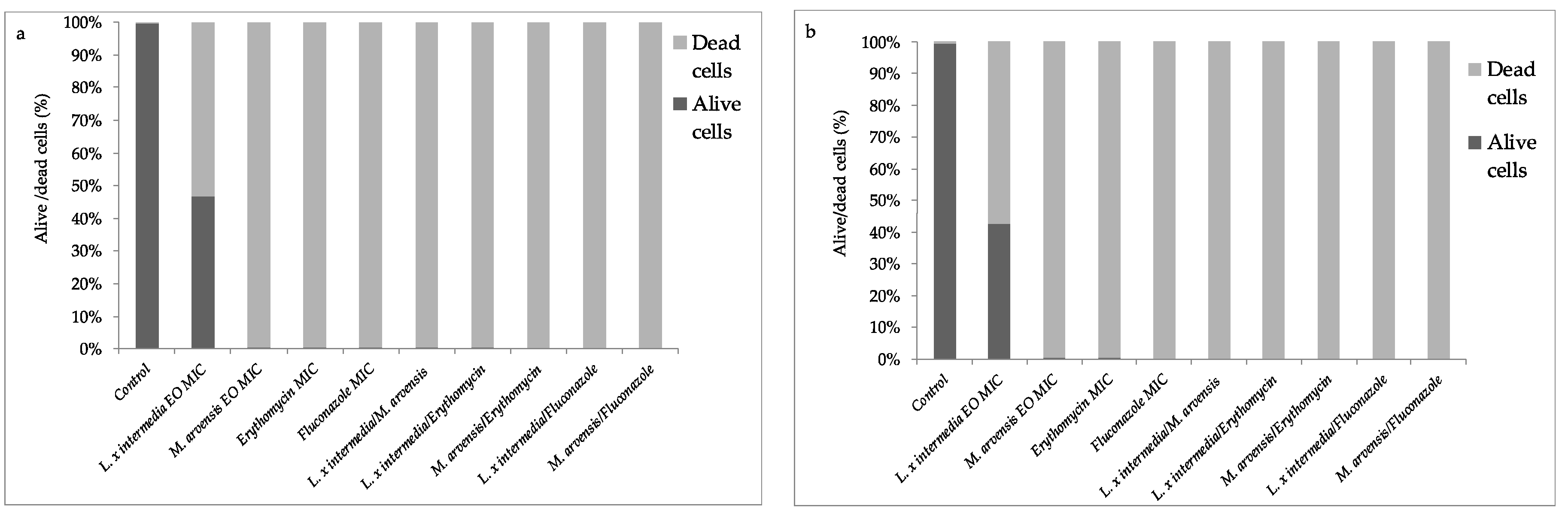
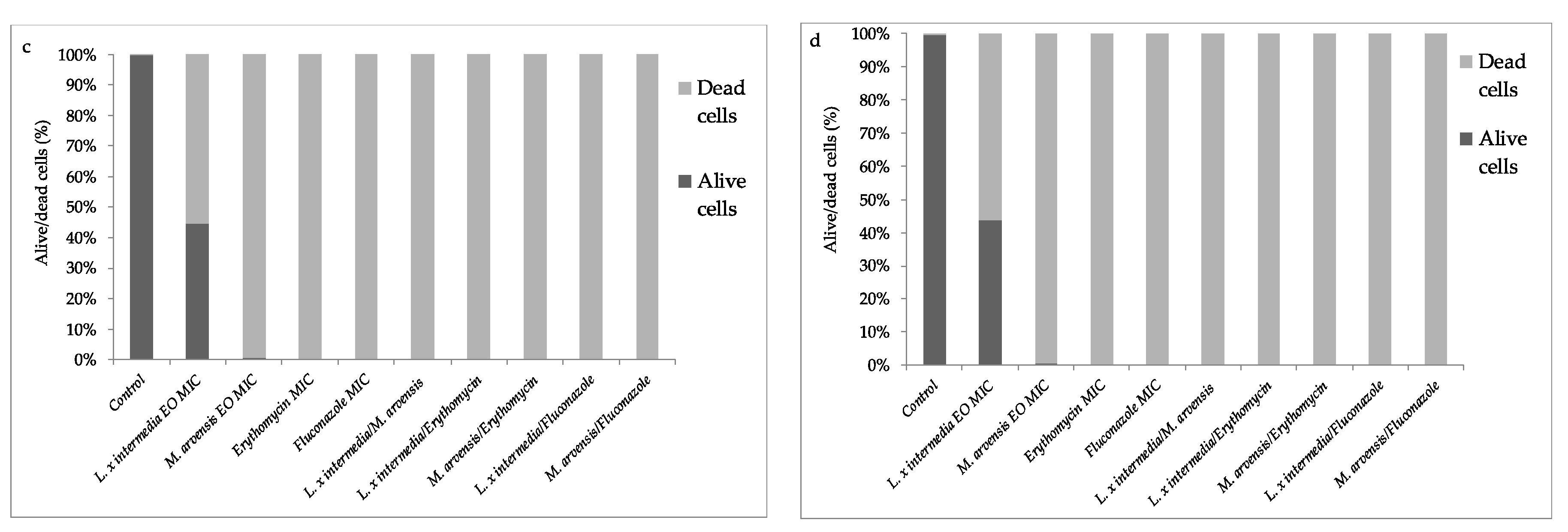
2.6. Anti-Biofilm Activity Determination
2.7. Quantification of EOs Activity on Mature Biofilm by Fluorescence Assay Study
2.8. Effectiveness of EOs Activity on Mature Biofilm by Fluorescence Microscopy Study
3. Discussion
4. Materials and Methods
4.1. Chemicals and Reagents
4.2. Essential Oils
4.3. Microbial Strains
4.4. Agar Disk Diffusion Assay
4.5. GC Analysis
4.6. Qualitative and Semi-Quantitative Analysis
4.7. Minimum Inhibitory Concentration (MIC)
4.8. Determination of the Fractional Inhibitory Concentration Index (FICI)
4.9. Time–Kill Studies
4.10. Anti-Biofilm Activity Determination
4.11. Quantification of EOs Activity on Mature Biofilm by Fluorescence Assay Study
4.12. Effectiveness of EOs Activity on Mature Biofilm by Fluorescence Microscopy Study
4.13. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Sabia, C.; Anacarso, I.; Bergonzini, A.; Gargiulo, R.; Sarti, M.; Condò, C.; Messi, P.; de Niederhausern, S.; Iseppi, R.; Bondi, M. Detection and partial characterization of a bacteriocin-like substance produced by Lactobacillus fermentum CS57 isolated from human vaginal secretions. Anaerobe 2014, 26, 41–45. [Google Scholar] [CrossRef] [PubMed]
- Pendharkar, S.; Brandsborg, E.; Hammarström, L.; Marcotte, H.; Larsson, P.G. Vaginal colonisation by probiotic lactobacilli and clinical outcome in women conventionally treated for bacterial vaginosis and yeast infection. BMC Infect. Dis. 2015, 15, 255. [Google Scholar] [CrossRef] [PubMed]
- Kumherová, M.; Veselá, K.; Kosová, M.; Mašata, J.; Horáčková, Š.; Šmidrkal, J. Novel Potential Probiotic Lactobacilli for Prevention and Treatment of Vulvovaginal Infections. Probiotics Antimicrob. Proteins 2020. [Google Scholar] [CrossRef]
- Johri, A.K.; Lata, H.; Yadav, P.; Dua, M.; Yang, Y.; Xu, X.; Homma, A.; Barocchi, M.A.; Bottomley, M.J.; Saul, A.; et al. Epidemiology of Group B Streptococcus in developing countries. Vaccine 2013, 31S, D43–D45. [Google Scholar] [CrossRef]
- Clifford, V.; Garland, S.M.; Grimwood, K. Prevention of neonatal group B Streptococcus disease in the 21st century. J. Paediatr. Child Health 2012, 48, 808–815. [Google Scholar] [CrossRef] [PubMed]
- Madrid, L.; Seale, A.C.; Kohli-Lynch, M.; Edmond, K.M.; Lawn, J.E.; Heath, P.T.; Madhi, S.A.; Baker, C.J.; Bartlett, L.; Cutland, C.; et al. Infant GBS Disease Investigator Group. Infant Group B Streptococcal Disease Incidence and Serotypes Worldwide: Systematic Review and Meta-analyses. Clin. Infect. Dis. 2017, 65, S160–S172. [Google Scholar] [CrossRef]
- Nan, C.; Dangor, Z.; Cutland, C.L.; Edwards, M.S.; Madhi, S.A.; Cunnington, M.C. Maternal group B Streptococcus-related stillbirth: A systematic review. BJOG 2015, 122, 1437–1445. [Google Scholar] [CrossRef]
- Mukesi, M.; Iweriebor, B.; Obi, L.C.; Nwodo, U.U.; Moyo, S.R.; Okoh, A. The activity of commercial antimicrobials, and essential oils and ethanolic extracts of Olea europaea on Streptococcus agalactiae isolated from pregnant women. BMC Complement Altern. Med. 2019, 19, 34. [Google Scholar] [CrossRef]
- Sobel, J.D. Vulvovaginal candidosis. Lancet 2007, 369, 1961–1971. [Google Scholar] [CrossRef]
- Abd Ellaha, N.H.; Abdel-Aleem, J.A.; Abdo, M.N.; Abou-Ghadir, O.F.; Zahran, K.M.; Hetta, H.F. Efficacy of ketoconazole gel-flakes in treatment of vaginal candidiasis: Formulation, in vitro and clinical evaluation. Int. J. Pharm. 2019, 567, 118472. [Google Scholar] [CrossRef]
- Giordani, B.; Basnet, P.; Mishchenko, E.; Luppi, B.; Škalko-Basnet, N. Utilizing liposomal quercetin and gallic acid in localized treatment of vaginal Candida infections. Pharmaceutics 2020, 12, 9. [Google Scholar] [CrossRef] [PubMed]
- Willems, H.M.E.; Ahmed, S.S.; Liu, J.; Xu, Z.; Brian, M.; Peters, B.M. Vulvovaginal candidiasis: A current understanding and burning questions. J. Fungi 2020, 6, 27. [Google Scholar] [CrossRef] [PubMed]
- Hawser, S.P.; Douglas, L.J. Resistance of Candida albicans biofilms to antifungal agents in vitro. Antimicrob. Agents Chemother. 1995, 39, 2128–2131. [Google Scholar] [CrossRef] [PubMed]
- Taff, H.T.; Mitchell, K.F.; Edward, J.A.; Andes, D.R. Mechanisms of Candida biofilm drug resistance. Future Microbiol. 2013, 8, 1325–1337. [Google Scholar] [CrossRef] [PubMed]
- Zarnowski, R.; Sanchez, H.; Covelli, A.S.; Dominguez, E.; Jaromin, A.; Bernhardt, J.; Mitchell, K.F.; Heiss, C.; Azadi, P.; Mitchell, A.; et al. Candida albicans biofilm–induced vesicles confer drug resistance through matrix biogenesis. PLoS Biol. 2018, 16, e2006872. [Google Scholar] [CrossRef]
- Ben-Ami, R. Treatment of Invasive Candidiasis: A Narrative Review. J. Fungi 2018, 4, 97. [Google Scholar] [CrossRef]
- Harriott, M.M.; Lilly, E.A.; Rodriguez, T.E.; Fidel, P.L.; Noverr, M.C. Candida albicans forms biofilms on the vaginal mucosa. Microbiology 2010, 156, 3635–3644. [Google Scholar] [CrossRef]
- Cai, Y.M.; Chen, X.S. Concerns regarding bias in estimates of recurrent vulvovaginal candidiasis. Lancet Infect. Dis. 2018, 18, 1180. [Google Scholar] [CrossRef]
- Denning, D.W.; Kneale, M.; Sobel, J.D.; Rautemaa-Richardson, R. Global burden of recurrent vulvovaginal candidiasis: A systematic review. Lancet Infect. Dis. 2018, 18, e339–e347. [Google Scholar] [CrossRef]
- Russo, R.; Superti, F.; Karadja, E.; De Seta, F. Randomised clinical trial in women with recurrent vulvovaginal candidiasis: Efficacy of probiotics and lactoferrin as maintenance treatment. Mycoses 2019, 62, 328–335. [Google Scholar] [CrossRef] [PubMed]
- Donders, G.G.; Grienceviciene, S.; Ruban, K.; Bellen, G. Vaginal pH and microbiota during fluconazole maintenance treatment for recurrent vulvovaginal candidosis (RVVC). Diagn. Microbiol. Infect. Dis. 2020, 97, 115024. [Google Scholar] [CrossRef] [PubMed]
- Hemaiswaryaa, S.; Kruthiventib, A.K.; Doblea, M. Synergism between natural products and antibiotics against infectious diseases. Phytomedicine 2008, 15, 639–652. [Google Scholar] [CrossRef] [PubMed]
- Kon, K.V.; Rai, M.K. Plant essential oils and their constituents in coping with multidrug-resistant bacteria. Expert Rev. Anti-Infect. Ther. 2012, 10, 775–790. [Google Scholar] [CrossRef] [PubMed]
- Raut, J.S.; Shinde, R.B.; Chauhan, N.M.; Karuppayil, S.M. Phenylpropanoids of plant origin as inhibitors of biofilm formation by Candida albicans. J. Microbiol. Biotechnol. 2014, 24, 1216–1225. [Google Scholar] [CrossRef]
- Karapandzova, M.; Stefkova, G.; Cvetkovikj, I.; Trajkovska-Dokik, E.; Kaftandzieva, A.; Kulevanova, S. Chemical composition and antimicrobial activity of the essential oils of Pinus peuce (Pinaceae) growing wild in R. Macedonia. Nat. Prod. Commun. 2014, 9, 1623–1628. [Google Scholar] [CrossRef]
- Ennigrou, A.; Casabianca, H.; Vulliet, E.; Hanchi, B.; Hosni, K. Assessing the fatty acid, essential oil composition, their radical scavenging and antibacterial activities of Schinus terebinthifolius Raddi leaves and twigs. J. Food Sci. Technol. 2018, 55, 1582–1590. [Google Scholar] [CrossRef]
- Carbone, C.; Teixeira, M.D.C.; Sousa, M.D.C.; Martins-Gomes, C.; Silva, A.M.; Souto, E.M.B.; Musumeci, T. Clotrimazole-Loaded Mediterranean Essential Oils NLC: A Synergic Treatment of Candida Skin Infections. Pharmaceutics 2019, 11, 231. [Google Scholar] [CrossRef]
- Maietti, S.; Rossi, D.; Guerrini, A.; Useli, C.; Romagnoli, C.; Poli, F.; Bruni, R.; Sacchetti, G. A multivariate analysis approach to the study of chemical and functional properties of chemo-diverse plant derivatives: Lavender essential oils. Flavour Fragr. J. 2013, 28, 144–154. [Google Scholar] [CrossRef]
- Tardugno, R.; Serio, A.; Pellati, F.; D’Amato, S.; López, C.C.; Bellardi, M.G.; Di Vito, M.; Savini, V.; Paparella, A.; Benvenuti, S. Lavandula x intermedia and Lavandula angustifolia essential oils: Phytochemical composition and antimicrobial activity against foodborne pathogens. Nat. Prod. Res. 2019, 33, 3330–3335. [Google Scholar] [CrossRef]
- Tardugno, R.; Pellati, F.; Iseppi, R.; Bondi, M.; Bruzzesi, G.; Benvenuti, S. Phytochemical composition and in vitro screening of the antimicrobial activity of essential oils on oral pathogenic bacteria. Nat. Prod. Res. 2018, 32, 544–551. [Google Scholar] [CrossRef]
- Bogavac, M.; Karaman, M.; Janjušević, L.J.; Sudji, J.; Radovanović, B.; Novaković, Z.; Simeunović, J.; Božin, B. Alternative treatment of vaginal infections—In vitro antimicrobial and toxic effects of Coriandrum sativum L. and Thymus vulgaris L. essential oils. J. Appl. Microbiol. 2015, 119, 697–710. [Google Scholar] [CrossRef]
- Bogavac, M.A.; Karaman, M.A.; Sudi, J.J.; Radovanović, B.B.; Janjušević, L.N.; Ćetković, N.B.; Tešanović, K.D. Antimicrobial potential of Rosmarinus officinalis commercial essential oil in the treatment of vaginal infections in pregnant women. Nat. Prod. Commun. 2017, 12, 127–130. [Google Scholar] [CrossRef] [PubMed]
- Denner, S.S. Lavandula angustifolia Miller: English lavender. Holist. Nurs. Pract. 2009, 23, 57–64. [Google Scholar] [CrossRef]
- Bajalan, I.; Rouzbahani, R.; Pirbalouti, A.G.; Maggi, F. Chemical composition and Antibacterial activity of iranian Lavandula x hybrida. Chem. Biodivers. 2017, 14, e1700064. [Google Scholar] [CrossRef] [PubMed]
- Haba, E.; Bouhdid, S.; Torrego-Solana, N.; Marqués, A.M.; Espuny, M.J.; García-Celma, M.J.; Manresa, A. Rhamnolipids as emulsifying agents for essential oil formulations: Antimicrobial effect against Candida albicans and methicillin-resistant Staphylococcus aureus. Int. J. Pharm. 2014, 476, 134–141. [Google Scholar] [CrossRef]
- Sienkiewicz, M.; Głowacka, A.; Kowalczyk, E.; Wiktorowska-Owczarek, A.; Jóźwiak-Bębenista, M.; Łysakowska, M. The biological activities of cinnamon, geranium and lavender essential oils. Molecules 2014, 19, 20929–20940. [Google Scholar] [CrossRef]
- Ait Said, L.; Zahlane, K.; Ghalbane, I.; El Messoussi, S.; Romane, A.; Cavaleiro, C.; Salgueiro, L. Chemical composition and antibacterial activity of Lavandula coronopifolia essential oil against antibiotic-resistant bacteria. Nat. Prod. Res. 2015, 29, 582–585. [Google Scholar] [CrossRef] [PubMed]
- Karpiński, T.M. Essential oils of Lamiaceae family plants as antifungals. Biomolecules 2020, 10, 103. [Google Scholar] [CrossRef]
- De Rapper, S.; Viljoen, A.; van Vuuren, S. The in vitro antimicrobial effects of Lavandula angustifolia essential oil in combination with conventional antimicrobial agents. Evid. -Based Complement. Alternat. Med. 2016, 2016, 2752739. [Google Scholar] [CrossRef] [PubMed]
- Garzoli, S.; Turchett, I.G.; Giacomello, P.; Tiezzi, A.; Laghezza Masci, V.; Ovidi, E. Liquid and vapour phase of Lavandin (Lavandula x intermedia) essential oil: Chemical composition and antimicrobial activity. Molecules 2019, 24, 2701. [Google Scholar] [CrossRef] [PubMed]
- Imai, H.; Osawa, K.; Yasuda, H.; Hamashima, H.; Arai, T.; Sasatsu, M. Inhibition by the essential oils of peppermint and spearmint of the growth of pathogenic bacteria. Microbios 2001, 106, 31–39. [Google Scholar]
- Ludwiczuk, A.; Kieltyka-Dadasiewiczb, A.; Sawicki, R.; Golusd, J.; Ginalskad, G. Essential Oils of some Mentha species and cultivars, their chemistry and bacteriostatic activity. Nat. Prod. Commun. 2016, 11, 1015–1018. [Google Scholar] [CrossRef] [PubMed]
- Heydari, M.; Zanfardino, A.; Taleei, A.; Hadian, J.; Maresca, V.; Sorbo, S.; Di Napoli, M.; Varcamonti, M.; Basile, A.; Rigano, D.; et al. Effect of heat stress on yield, monoterpene content and antibacterial activity of essential oils of Mentha x piperita var. Mitcham and Mentha arvensis var. piperascens. Molecules 2018, 23, 1903. [Google Scholar] [CrossRef] [PubMed]
- Bose, S.K.; Yadav, R.K.; Mishra, S.; Sangwan, R.S.; Singh, A.K.; Mishra, B.; Srivastava, A.K.; Sangwan, N.S. Effect of gibberellic acid and calliterpenone on plant growth attributes, trichomes, essential oil biosynthesis and pathway gene expression in differential manner in Mentha arvensis L. Plant Physiol. Biochem. 2013, 66, 150–158. [Google Scholar] [CrossRef] [PubMed]
- Kubota, T.; Nojima, M.; Itoh, S. Vaginal bacterial flora of pregnant women colonized with group B Streptococcus. J. Infect. Chemother. 2002, 8, 326–330. [Google Scholar] [CrossRef] [PubMed]
- Ronnqvist, P.D.; Forsgren-Brusk, U.B.; Grahn-Hakansson, E.E. Lactobacilli in the female genital tract in relation to other genital microbes and vaginal pH. Acta Obstet. Gynecol Scand. 2006, 85, 726–735. [Google Scholar] [CrossRef]
- Parolin, C.; Marangoni, A.; Laghi, L.; Foschi, C.; Ñahui Palomino, R.A.; Calonghi, N.; Cevenini, R.; Vitali, B. Isolation of vaginal Lactobacilli and characterization of anti-Candida activity. PLoS ONE 2015, 10, e0131220. [Google Scholar] [CrossRef]
- Benzaid, C.; Belmadani, A.; Djeribi, R.; Rouabhia, M. The effects of Mentha piperita essential oil on C. albicans growth, transition, biofilm formation, and the expression of secreted aspartyl proteinases genes. Antibiotics 2019, 8, 10. [Google Scholar] [CrossRef]
- Minooeianhaghighi, M.H.; Sepehrian, L.; Shokri, H. Antifungal effects of Lavandula binaludensis and Cuminum cyminum essential oils against Candida albicans strains isolated from patients with recurrent vulvovaginal candidiasis. J. Mycol. Med. 2017, 27, 65–71. [Google Scholar] [CrossRef]
- Pietrella, D.; Angiolella, L.; Vavala, E.; Rachini, A.; Mondello, F.; Ragno, R.; Bistoni, F.; Vecchiarelli, A. Beneficial effect of Mentha suaveolens essential oil in the treatment of vaginal candidiasis assessed by real-time monitoring of infection. BMC Complement Altern. Med. 2011, 11, 18. [Google Scholar] [CrossRef]
- Camele, I.; Elshafie, H.S.; Caputo, L.; De Feo, V. Anti-quorum sensing and antimicrobial effect of mediterranean plant essential oils against phytopathogenic bacteria. Front. Microbiol. 2019, 10, 2619. [Google Scholar] [CrossRef] [PubMed]
- CLSI. Performance Standards for Antimicrobial Disk Susceptibility Tests: Approved Standard, 11th ed.; CLSI document M02eA11; Clinical and Laboratory Standards Institute: Wayne, PA, USA, 2012. [Google Scholar]
- Klancnik, A.; Piskernik, S.; Jersek, B.; Mozina, S.S. Evaluation of diffusion and dilution methods to determine the antibacterial activity of plant extracts. J. Microbiol. Methods 2010, 81, 121–126. [Google Scholar] [CrossRef] [PubMed]
- Adams, R.P. Identification of Essential Oil Components by Gas Chromatography/Mass Spectrometry, 4th ed.; Allured Publishing: Carol Stream, IL, USA, 2007. [Google Scholar]
- Şahin, F.; Güllüce, M.; Daferera, D.; Sökmen, A.; Sökmen, M.; Polissiou, M.; Agar, G.; Özer, H. Biological activities of the essential oils and methanol extract of Origanum vulgare ssp. vulgare in the Eastern Anatolia region of Turkey. Food Control. 2004, 15, 549–557. [Google Scholar]
- Stepanovic, S.; Vukovic, D.; Hola, V.; Di Bona Ventura, G.; Djukic, S.; Irkovic, I.C.; Ruzicka, F. Quantification of biofilm in microtiter plates: Overview of testing conditions and practical recommendations for assessment of biofilm production by staphylococci. APMIS 2007, 115, 891–899. [Google Scholar] [CrossRef]



| Compound a | LRIb | Lavandula x intermedia | Mentha arvensis |
|---|---|---|---|
| α-Thujene | 926 | 0.1 c | - |
| α-Pinene | 931 | 0.7 c | 0.7 c |
| Camphene | 946 | 0.5 c | - |
| Sabinene | 971 | 0.2 c | 0.3 c |
| β-Pinene | 974 | 0.5 c | 0.8 c |
| β-Myrcene | 990 | 1.0 ± 0.1 | 0.7 c |
| p-Cymene | 1024 | 0.3 c | - |
| Limonene | 1027 | 1.4 c | 3.6 ± 0.1 |
| 1,8-Cineole | 1029 | 5.0 c | 0.2 c |
| cis-Ocimene | 1037 | 0.8 c | - |
| trans-Ocimene | 1047 | 0.9 c | - |
| γ- Terpinene | 1058 | 0.1 c | - |
| Terpinolene | 1087 | 0.4 c | - |
| Linalool | 1100 | 36.0 ± 0.1 | 0.1 c |
| Camphor | 1143 | 5.9 ± 0.1 | - |
| Isopulegol | 1146 | - | 0.8 c |
| Menthone | 1156 | - | 7.8 ± 0.1 |
| Borneol | 1165 | 4.0 c | - |
| Isomenthone | 1167 | - | 5.4 c |
| Terpinen-4-ol | 1177 | 2.7 c | - |
| Menthol | 1180 | - | 73.8 ± 0.2 |
| α- Terpineol | 1191 | 0.9 c | 0.1 c |
| Pulegone | 1245 | - | 0.5 c |
| Piperitone | 1259 | - | 0.5 c |
| Linalyl acetate | 1263 | 27.3 ± 0.2 | - |
| Lavandulyl acetate | 1293 | 1.8 c | - |
| Menthyl Acetate | 1297 | - | 2.1 c |
| β-Bourbonene | 1390 | - | 0.1 c |
| β-Caryophyllene | 1424 | 1.5 c | 0.3 c |
| Germacrene D | 1487 | - | 0.4 c |
| Caryophyllene oxide | 1592 | 0.2 c | - |
| Total | 92.1 | 98.2 |
| Strains | Lavandula x intermedia | Mentha arvensis | Erythromycin | Fluconazole |
|---|---|---|---|---|
| Streptococcus agalactiae 1 | 18 | 36 | 2 | - |
| Streptococcus agalactiae 2 | 9 | 18 | 1 | - |
| Streptococcus agalactiae ATCC 13813 | 18 | 18 | 0.125 | - |
| Candida albicans 1 | 18 | 18 | - | 4 |
| Candida albicans 2 | 9 | 144 | - | 4 |
| Candida albicans ATCC 10231 | 18 | 72 | - | 0.25 |
| Lactobacillus spp.1 | 144 | 9 | 0.5 | 1 |
| Lactobacillus spp.2 | 144 | 18 | 0.25 | 1 |
| Lactobacillus spp.3 | 72 | 18 | 0.25 | 1 |
| Lactobacillus spp.4 | 72 | 18 | 1 | 0.5 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Iseppi, R.; Tardugno, R.; Brighenti, V.; Benvenuti, S.; Sabia, C.; Pellati, F.; Messi, P. Phytochemical Composition and In Vitro Antimicrobial Activity of Essential Oils from the Lamiaceae Family against Streptococcus agalactiae and Candida albicans Biofilms. Antibiotics 2020, 9, 592. https://doi.org/10.3390/antibiotics9090592
Iseppi R, Tardugno R, Brighenti V, Benvenuti S, Sabia C, Pellati F, Messi P. Phytochemical Composition and In Vitro Antimicrobial Activity of Essential Oils from the Lamiaceae Family against Streptococcus agalactiae and Candida albicans Biofilms. Antibiotics. 2020; 9(9):592. https://doi.org/10.3390/antibiotics9090592
Chicago/Turabian StyleIseppi, Ramona, Roberta Tardugno, Virginia Brighenti, Stefania Benvenuti, Carla Sabia, Federica Pellati, and Patrizia Messi. 2020. "Phytochemical Composition and In Vitro Antimicrobial Activity of Essential Oils from the Lamiaceae Family against Streptococcus agalactiae and Candida albicans Biofilms" Antibiotics 9, no. 9: 592. https://doi.org/10.3390/antibiotics9090592
APA StyleIseppi, R., Tardugno, R., Brighenti, V., Benvenuti, S., Sabia, C., Pellati, F., & Messi, P. (2020). Phytochemical Composition and In Vitro Antimicrobial Activity of Essential Oils from the Lamiaceae Family against Streptococcus agalactiae and Candida albicans Biofilms. Antibiotics, 9(9), 592. https://doi.org/10.3390/antibiotics9090592