Molecular Epidemiology of Carbapenem-Resistant Acinetobacter baumannii Isolated from War-Injured Patients from the Eastern Ukraine
Abstract
:1. Introduction
2. Results
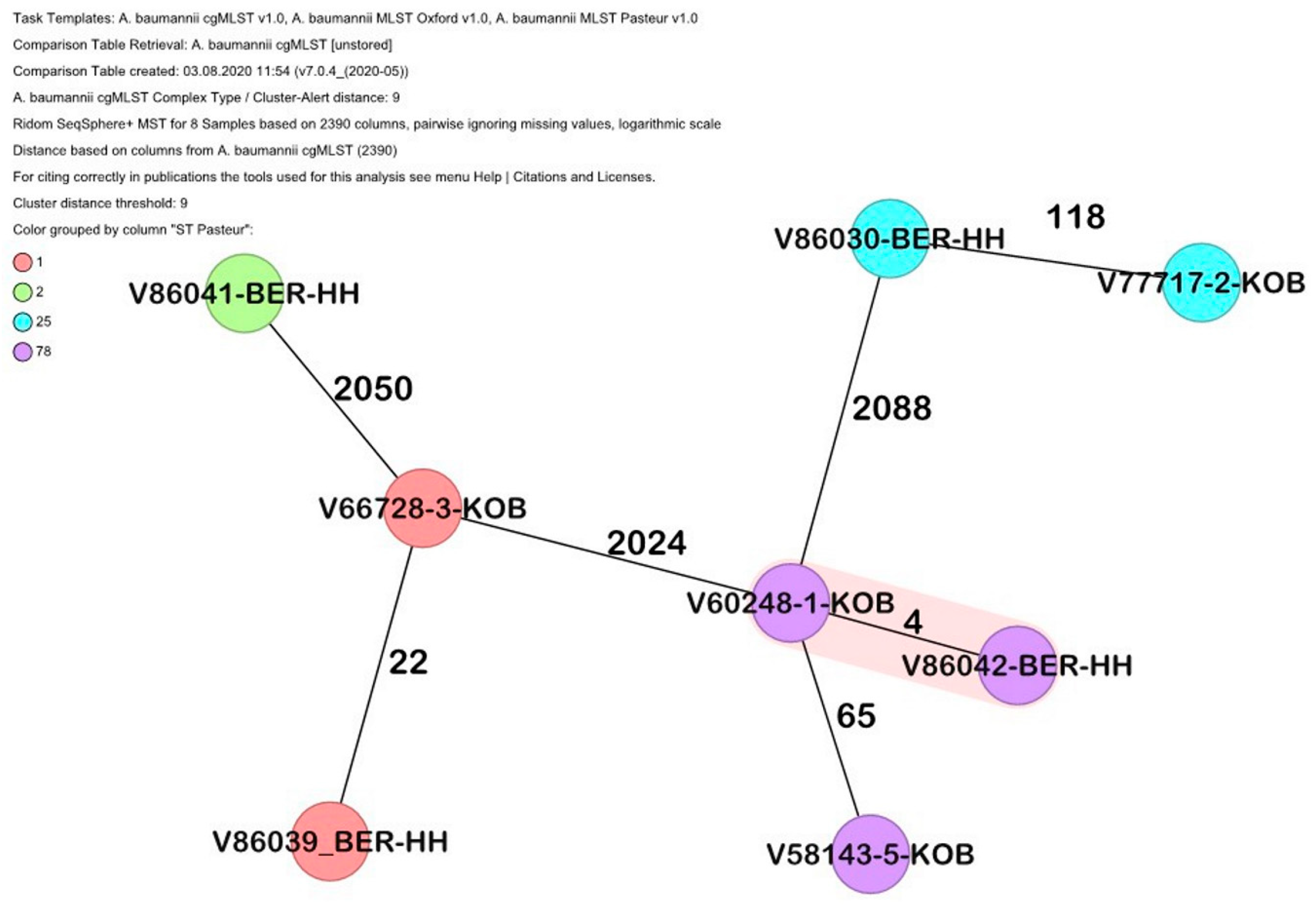
2.1. Core Genome-Based Confirmation on Species Level and Clustering with International Outbreak Strains
2.2. Identified Molecular Resistance Mechanisms
3. Discussion
4. Materials and Methods
4.1. Patient Isolates
4.2. DNA Extraction and Whole Genome Sequencing
4.3. Molecular Epidemiology and Determination of Antibiotic Resistance Genes
4.4. Ethical Clearance
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Davis, K.A.; Moran, K.A.; McAllister, C.K.; Gray, P.J. Multidrug-resistant Acinetobacter extremity infections in soldiers. Emerg. Infect. Dis. 2005, 11, 1218–1224. [Google Scholar] [CrossRef]
- Zapor, M.J.; Moran, K.A. Infectious diseases during wartime. Curr. Opin. Infect. Dis. 2005, 18, 395–399. [Google Scholar] [CrossRef]
- Dallo, S.F.; Weitao, T. Insights into acinetobacter war-wound infections, biofilms, and control. Adv. Skin. Wound Care 2010, 23, 169–174. [Google Scholar] [CrossRef]
- O’Shea, M.K. Acinetobacter in modern warfare. Int. J. Antimicrob. Agents. 2012, 39, 363–375. [Google Scholar] [CrossRef] [PubMed]
- Arivett, B.A.; Ream, D.C.; Fiester, S.E.; Kidane, D.; Actis, L.A. Draft Genome Sequences of Acinetobacter baumannii Isolates from Wounded Military Personnel. Genome Announc. 2016, 4. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schaal, J.V.; Donat, N.; Ragot, C.; Soler, C.; Leclerc, T. Carbapenem-resistant Acinetobacter baumannii in Military Burn Centre. Burns 2020, 46, 747–748. [Google Scholar] [CrossRef]
- Velasco, J.M.; Valderama, M.T.; Margulieux, K.; Diones, P.C.; Peacock, T.; Navarro, F.C.; Liao, C.; Chua, D.; Macareo, L.; Crawford, J.; et al. Comparison of Carbapenem-Resistant Microbial Pathogens in Combat and Non-combat Wounds of Military and Civilian Patients Seen at a Tertiary Military Hospital, Philippines (2013–2017). Mil. Med. 2020, 185, e197–e202. [Google Scholar] [CrossRef] [PubMed]
- Griffith, M.E.; Gonzalez, R.S.; Holcomb, J.B.; Hospenthal, D.R.; Wortmann, G.W.; Murray, C.K. Factors associated with recovery of Acinetobacter baumannii in a combat support hospital. Infect. Control. Hosp. Epidemiol. 2008, 29, 664–666. [Google Scholar] [CrossRef] [Green Version]
- Moultrie, D.; Hawker, J.; Cole, S. Factors associated with multidrug-resistant Acinetobacter transmission: An integrative review of the literature. AORN J. 2011, 94, 27–36. [Google Scholar] [CrossRef] [PubMed]
- Petersen, K.; Cannegieter, S.C.; van der Reijden, T.J.; van Strijen, B.; You, D.M.; Babel, B.S.; Philip, A.I.; Dijkshoorn, L. Diversity and clinical impact of Acinetobacter baumannii colonization and infection at a military medical center. J. Clin. Microbiol. 2011, 49, 159–166. [Google Scholar] [CrossRef] [Green Version]
- Sensenig, R.A.; Murray, C.K.; Mende, K.; Wolf, S.E.; Chung, K.K.; Hospenthal, D.R.; Yun, H.C. Longitudinal characterization of Acinetobacter baumannii-calcoaceticus complex, Klebsiella pneumoniae, and methicillin-resistant Staphylococcus aureus colonizing and infecting combat casualties. Am. J. Infect. Control. 2012, 40, 183–185. [Google Scholar] [CrossRef] [PubMed]
- Keen, E.F., III; Mende, K.; Yun, H.C.; Aldous, W.K.; Wallum, T.E.; Guymon, C.H.; Cole, D.W.; Crouch, H.K.; Griffith, M.E.; Thompson, B.L.; et al. Evaluation of potential environmental contamination sources for the presence of multidrug-resistant bacteria linked to wound infections in combat casualties. Infect. Control Hosp. Epidemiol. 2012, 33, 905–911. [Google Scholar] [CrossRef] [PubMed]
- Murray, C.K.; Hospenthal, D.R.; Kotwal, R.S.; Butler, F.K. Efficacy of point-of-injury combat antimicrobials. J. Trauma Acute Care Surg. 2011, 71 (Suppl. S2), S307–S313. [Google Scholar] [CrossRef]
- Scott, P.; Deye, G.; Srinivasan, A.; Murray, C.; Moran, K.; Hulten, E.; Fishbain, J.; Craft, D.; Riddell, S.; Lindler, L.; et al. An outbreak of multidrug-resistant Acinetobacter baumannii-calcoaceticus complex infection in the US military health care system associated with military operations in Iraq. Clin. Infect. Dis. 2007, 44, 1577–1584. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Johnson, E.N.; Marconi, V.C.; Murray, C.K. Hospital-acquired device-associated infections at a deployed military hospital in Iraq. J. Trauma Acute Care Surg. 2009, 66 (Suppl. S4), S157–S163. [Google Scholar] [CrossRef] [PubMed]
- Recio, A.C.; Bohart, Z.W.; Havens, S.R.; Stiens, S.A. Acute spinal cord injury and infection with multidrug-resistant Acinetobacter calcoaceticus-baumannii complex among returning Operation Iraqi Freedom soldiers: Successful innovations in rehabilitation during isolation. Am. J. Phys. Med. Rehabil. 2010, 89, 331–335. [Google Scholar] [CrossRef] [PubMed]
- Murphy, R.A.; Ronat, J.B.; Fakhri, R.M.; Herard, P.; Blackwell, N.; Abgrall, S.; Anderson, D.J. Multidrug-resistant chronic osteomyelitis complicating war injury in Iraqi civilians. J. Trauma Acute Care Surg. 2011, 71, 252–254. [Google Scholar] [CrossRef]
- Huang, X.Z.; Chahine, M.A.; Frye, J.G.; Cash, D.M.; Lesho, E.P.; Craft, D.W.; Lindler, L.E.; Nikolich, M.P. Molecular analysis of imipenem-resistant Acinetobacter baumannii isolated from US service members wounded in Iraq, 2003–2008. Epidemiol. Infect. 2012, 140, 2302–2307. [Google Scholar] [CrossRef] [Green Version]
- Yun, H.C.; Murray, C.K. Infection Prevention in the Deployed Environment. US Army Med. Dep. J. 2016, 2–16, 114–118. [Google Scholar]
- Turton, J.F.; Kaufmann, M.E.; Gill, M.J.; Pike, R.; Scott, P.T.; Fishbain, J.; Craft, D.; Deye, G.; Riddell, S.; Lindler, L.E.; et al. Comparison of Acinetobacter baumannii isolates from the United Kingdom and the United States that were associated with repatriated casualties of the Iraq conflict. J. Clin. Microbiol. 2006, 44, 2630–2634. [Google Scholar] [CrossRef] [Green Version]
- Stuart, T.L.; Mulvey, M.; Simor, A.E.; Tien, H.C.; Battad, A.; Taylor, G.; Vayalumkal, J.V.; Weir, C.; Ofner, M.; Gravel, D.; et al. Acinetobacter baumannii in casualties returning from Afghanistan. Can. J. Infect. Control. 2007, 22, 152–154. [Google Scholar] [PubMed]
- Tien, H.C.; Battad, A.; Bryce, E.A.; Fuller, J.; Mulvey, M.; Bernard, K.; Brisebois, R.; Doucet, J.J.; Rizoli, S.B.; Fowler, R.; et al. Multi-drug resistant Acinetobacter infections in critically injured Canadian forces soldiers. BMC Infect. Dis. 2007, 7, 95. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Calhoun, J.H.; Murray, C.K.; Manring, M.M. Multidrug-resistant organisms in military wounds from Iraq and Afghanistan. Clin. Orthop. Relat. Res. 2008, 466, 1356–1362. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Murray, C.K.; Yun, H.C.; Griffith, M.E.; Thompson, B.; Crouch, H.K.; Monson, L.S.; Aldous, W.K.; Mende, K.; Hospenthal, D.R. Recovery of multidrug-resistant bacteria from combat personnel evacuated from Iraq and Afghanistan at a single military treatment facility. Mil. Med. 2009, 174, 598–604. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hospenthal, D.R.; Crouch, H.K.; English, J.F.; Leach, F.; Pool, J.; Conger, N.G.; Whitman, T.J.; Wortmann, G.W.; Robertson, J.L.; Murray, C.K. Multidrug-resistant bacterial colonization of combat-injured personnel at admission to medical centers after evacuation from Afghanistan and Iraq. J. Trauma Acute Care Surg. 2011, 71 (Suppl. S1), S52–S57. [Google Scholar] [CrossRef] [Green Version]
- Sutter, D.E.; Bradshaw, L.U.; Simkins, L.H.; Summers, A.M.; Atha, M.; Elwood, R.L.; Robertson, J.L.; Murray, C.K.; Wortmann, G.W.; Hospenthal, D.R. High incidence of multidrug-resistant gram-negative bacteria recovered from Afghan patients at a deployed US military hospital. Infect. Control. Hosp. Epidemiol. 2011, 32, 854–860. [Google Scholar] [CrossRef]
- Valentine, K.P.; Viacheslav, K.M. Bacterial flora of combat wounds from eastern Ukraine and time-specified changes of bacterial recovery during treatment in Ukrainian military hospital. BMC Res. Notes 2017, 10, 152. [Google Scholar] [CrossRef] [Green Version]
- Salmanov, A.G.; Voronenko, Y.V.; Vozianov, S.O.; Shunko, Y.Y.; Mamenko, M.Y.; Verner, O.M.; Mykhalchuk, V.M.; Vydyborets, S.V.; Shkorbotun, V.O.; Beketova, H.V.; et al. Bloodstream infections and antimicrobial resistance of responsible pathogens in Ukraine: Results of a multicenter study (2013–2015). Wiad. Lek. 2019, 72, 2069–2075. [Google Scholar] [CrossRef]
- Salmanov, A.; Litus, V.; Vdovychenko, S.; Litus, O.; Davtian, L.; Ubogov, S.; Bisyuk, Y.; Drozdova, A.; Vlasenko, I. Healthcare-associated infections in intensive care units. Wiad. Lek. 2019, 72, 963–969. [Google Scholar]
- Nahaichuk, V.I.; Nazarchuk, O.A.; Osadchuk, N.I.; Dmytriiev, D.V.; Nazarchuk, H.H. The analytical prognosis of the susceptibility to aminoglycosides and doxycycline in Acinetobacter baumannii isoolated from burns of intensive care unit patients. Wiad. Lek. 2018, 71, 705–709. [Google Scholar]
- Hrabák, J.; Študentová, V.; Adámková, V.; Šemberová, L.; Kabelíková, P.; Hedlová, D.; Čurdová, M.; Zemlickova, H.; Papagiannitsis, C.C. Report on a transborder spread of carbapenemase-producing bacteria by a patient injured during Euromaidan, Ukraine. New Microbes New Infect. 2015, 8, 28–30. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- López-Hernández, I.; García Barrionuevo, A.; Díaz de Alba, P.; Clavijo, E.; Pascual, A. Characterization of NDM-1- and CMH-3-producing Enterobacter cloacae complex ST932 in a patient transferred from Ukraine to Spain. Enferm. Infecc. Microbiol. Clin. 2019, 38, 327–330. [Google Scholar] [CrossRef] [PubMed]
- Bilozor, A.; Balode, A.; Chakhunashvili, G.; Chumachenko, T.; Egorova, S.; Ivanova, M.; Kaftyreva, L.; Kõljalg, S.; Kõressaar, T.; Lysenko, O.; et al. Application of Molecular Methods for Carbapenemase Detection. Front. Microbiol. 2019, 10, 1755. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Granzer, H.; Hagen, R.M.; Warnke, P.; Bock, W.; Baumann, T.; Schwarz, N.G.; Podbielski, A.; Frickmann, H.; Koeller, T. Molecular epidemiology of carbapenem-resistant Acinetobacter baumannii complex isolates from patients that were injured during the Eastern Ukrainian conflict. Eur. J. Microbiol. Immunol. 2016, 6, 109–117. [Google Scholar] [CrossRef] [PubMed]
- Poirel, L.; Walsh, T.R.; Cuvillier, V.; Nordmann, P. Multiplex PCR for detection of acquired carbapenemase genes. Diagn. Microbiol. Infect. Dis. 2011, 70, 119–123. [Google Scholar] [CrossRef]
- Sun, C.; Hao, J.; Dou, M.; Gong, Y. Mutant prevention concentrations of levofloxacin, pazufloxacin and ciprofloxacin for A. baumannii and mutations in gyrA and parC genes. J. Antibiot. 2015, 68, 313–317. [Google Scholar] [CrossRef] [Green Version]
- Higgins, P.G.; Prior, K.; Harmsen, D.; Seifert, H. Development and evaluation of a core genome multilocus typing scheme for whole-genome sequence-based typing of Acinetobacter baumannii. PLoS ONE 2017, 12, e0179228. [Google Scholar] [CrossRef] [Green Version]
- Zarrilli, R.; Pournaras, S.; Giannouli, M.; Tsakris, A. Global evolution of multidrug-resistant Acinetobacter baumannii clonal lineages. Int. J. Antimicrob. Agents. 2013, 41, 11–19. [Google Scholar] [CrossRef]
- Gaiarsa, S.; Batisti Biffignandi, G.; Esposito, E.P.; Castelli, M.; Jolley, K.A.; Brisse, S.; Sassera, D.; Zarrilli, R. Comparative Analysis of the Two Acinetobacter baumannii Multilocus Sequence Typing (MLST) Schemes. Front. Microbiol. 2019, 10, 930. [Google Scholar] [CrossRef] [Green Version]
- Ruppitsch, W.; Pietzka, A.; Prior, K.; Bletz, S.; Fernandez, H.L.; Allerberger, F.; Harmsen, D.; Mellmann, A. Defining and Evaluating a Core Genome Multilocus Sequence Typing Scheme for Whole-Genome Sequence-Based Typing of Listeria monocytogenes. J. Clin. Microbiol. 2015, 53, 2869–2876. [Google Scholar] [CrossRef] [Green Version]
- Nowak, J.; Zander, E.; Stefanik, D.; Higgins, P.G.; Roca, I.; Vila, J.; McConnell, M.J.; Cisneros, J.M.; Seifert, H.; MagicBullet Working Group WP4. High incidence of pandrug-resistant Acinetobacter baumannii isolates collected from patients with ventilator-associated pneumonia in Greece, Italy and Spain as part of the MagicBullet clinical trial. J. Antimicrob. Chemother. 2017, 72, 3277–3282. [Google Scholar] [CrossRef] [PubMed]
- Pfeifer, Y.; Hunfeld, K.P.; Borgmann, S.; Maneg, D.; Blobner, W.; Werner, G.; Higgins, P.G. Carbapenem-resistant Acinetobacter baumannii ST78 with OXA-72 carbapenemase and ESBL gene blaCTX-M-115. J. Antimicrob. Chemother. 2016, 71, 1426–1428. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Delbrück, H.; Bogaerts, P.; Kupper, M.B.; Rezende de Castro, R.; Bennink, S.; Glupczynski, Y.; Galleni, M.; Hoffmann, K.M.; Bebrone, C. Kinetic and crystallographic studies of extended-spectrum GES-11, GES-12, and GES-14 β-lactamases. Antimicrob. Agents Chemother. 2012, 56, 5618–5625. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bogaerts, P.; Naas, T.; El Garch, F.; Cuzon, G.; Deplano, A.; Delaire, T.; Huang, T.D.; Lissoir, B.; Nordmann, P.; Glupczynski, Y. GES extended-spectrum β-lactamases in Acinetobacter baumannii isolates in Belgium. Antimicrob. Agents Chemother. 2010, 54, 4872–4878. [Google Scholar] [CrossRef] [Green Version]
- Bartual, S.G.; Seifert, H.; Hippler, C.; Luzon, M.A.; Wisplinghoff, H.; Rodríguez-Valera, F. Development of a multilocus sequence typing scheme for characterization of clinical isolates of Acinetobacter baumannii. J. Clin. Microbiol. 2005, 43, 4382–4390. [Google Scholar] [CrossRef] [Green Version]
- Wisplinghoff, H.; Hippler, C.; Bartual, S.G.; Haefs, C.; Stefanik, D.; Higgins, P.G.; Seifert, H. Molecular epidemiology of clinical Acinetobacter baumannii and Acinetobacter genomic species 13TU isolates using a multilocus sequencing typing scheme. Clin. Microbiol. Infect. 2008, 14, 708–715. [Google Scholar] [CrossRef] [Green Version]
- Diancourt, L.; Passet, V.; Nemec, A.; Dijkshoorn, L.; Brisse, S. The population structure of Acinetobacter baumannii: Expanding multiresistant clones from an ancestral susceptible genetic pool. PLoS ONE 2010, 5, e10034. [Google Scholar] [CrossRef] [Green Version]
- Kleinheinz, K.A.; Joensen, K.G.; Larsen, M.V. Applying the ResFinder and VirulenceFinder web-services for easy identification of acquired antibiotic resistance and E. coli virulence genes in bacteriophage and prophage nucleotide sequences. Bacteriophage 2014, 4, e27943. [Google Scholar] [CrossRef] [Green Version]
- Zankari, E. Comparison of the web tools ARG-ANNOT and ResFinder for detection of resistance genes in bacteria. Antimicrob. Agents Chemother. 2014, 58, 4986. [Google Scholar] [CrossRef] [Green Version]
| Sample (rep-PCR Cluster) | MLST | Clonal Lineage | Antibiotic Resistance Determinants | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| STox | STpas | Sulphonamide | Phenicol | Beta-Lactam | Aminoglycoside | Macrolide | Tetracycline | Trimethoprim | Fluoroquinolone and Aminoglycoside | ||
| V86041-BER-HH (rep-PCR 1) | 1102 | 2 | IC2 | blaADC-11, blaOXA-23, blaOXA-66 | aac(3)-Ia-like, aadA1-like, aph(3′)-Ic, aph(3′)-VIa-like | ||||||
| V86039-BER-HH (rep-PCR 2) | 231 | 1 | IC1 | sul1 | catA1-like, cmlA1-like | blaADC-158, blaGES-12, blaOXA-23, blaOXA-69 | aac(3)-Ia-like, aadA1, aadA2, aadB, aph(3′)-VIa-like, strA-like, strB-like | tet(A) | dfrA7 | aac(6′)Ib-cr-like | |
| V66728-3-KOB (rep-PCR 3) | 231 | 1 | IC1 | sul1 | cmlA1-like | blaADC-158, blaGES-12, blaOXA-69 | aadA2, aadB, aph(3′)-VIa, strA-like, strB-like | dfrA7 | aac(6′)Ib-cr-like | ||
| V77717-2-KOB (rep-PCR 3 outlier) | 440 | 25 | IC7 | sul2 | blaADC-26-like, blaOXA-64 | aadB-like, aph(3′)-Ic, strA, strB-like | |||||
| V60248_1_KOB (rep-PCR 4) | 944 | 78 | IC6 | sul1 | blaADC-152, blaOXA-90, blaOXA-72 | aadA5, armA | mph(E) | ||||
| V86042_BER_HH (rep-PCR 4 outlier) | 944 | 78 | IC6 | sul1, sul2-like | catA1-like, floR-like | blaADC-152, blaCARB-16, blaCTX-M-115, blaOXA-90, blaOXA-72 | aadA5, armA | mph(E) | |||
| V58143_5_KOB (singleton) | 2144 | 78 | IC6 | sul2-like | catA1-like, floR-like | blaADC-107, blaCTX-M-115, blaOXA-90, blaOXA-72 | |||||
| V86030_BER_HH (singleton) | 690 | 25 | IC7 | sul2 | blaADC-26-like*, blaOXA-23, blaOXA-64 | aph(3′)-Ic, aph(3′)-VIa-like, strA-like, strB | |||||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Higgins, P.G.; Hagen, R.M.; Podbielski, A.; Frickmann, H.; Warnke, P. Molecular Epidemiology of Carbapenem-Resistant Acinetobacter baumannii Isolated from War-Injured Patients from the Eastern Ukraine. Antibiotics 2020, 9, 579. https://doi.org/10.3390/antibiotics9090579
Higgins PG, Hagen RM, Podbielski A, Frickmann H, Warnke P. Molecular Epidemiology of Carbapenem-Resistant Acinetobacter baumannii Isolated from War-Injured Patients from the Eastern Ukraine. Antibiotics. 2020; 9(9):579. https://doi.org/10.3390/antibiotics9090579
Chicago/Turabian StyleHiggins, Paul G., Ralf Matthias Hagen, Andreas Podbielski, Hagen Frickmann, and Philipp Warnke. 2020. "Molecular Epidemiology of Carbapenem-Resistant Acinetobacter baumannii Isolated from War-Injured Patients from the Eastern Ukraine" Antibiotics 9, no. 9: 579. https://doi.org/10.3390/antibiotics9090579





