The Variability of Thymol and Carvacrol Contents Reveals the Level of Antibacterial Activity of the Essential Oils from Different Accessions of Oliveria decumbens
Abstract
1. Introduction
2. Results and Discussion
2.1. Essential Oil Content
2.2. Essential Oil Composition
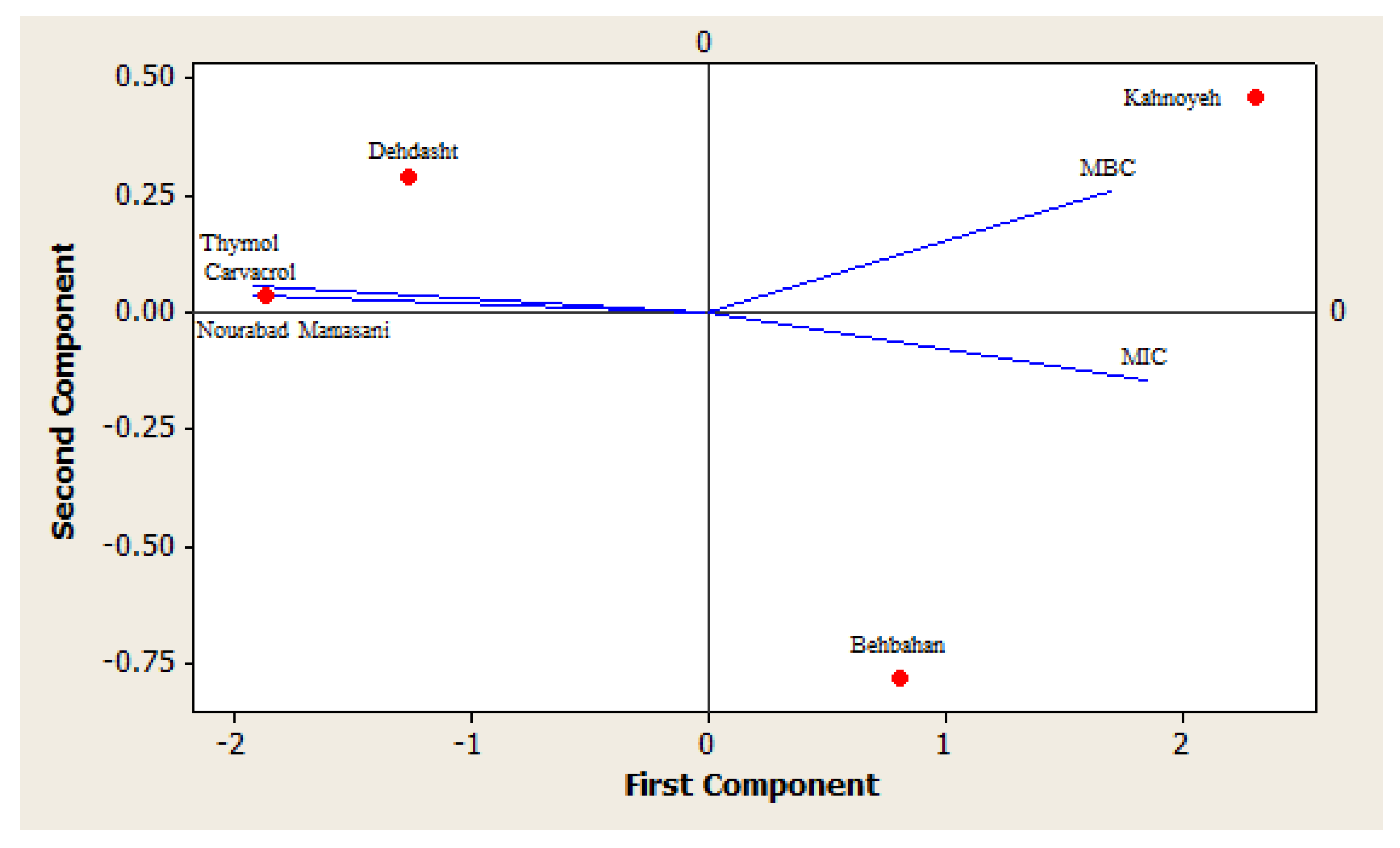
2.3. Antibacterial Activity
3. Materials and Methods
3.1. Plant Material
3.2. Essential Oil Isolation
3.3. GC-MS Analysis
3.4. Determination of Minimum Inhibitory Concentration
3.5. Determination of Minimum Bactericidal Concentration
3.6. Statistical Analysis
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Rechinger, K.H.; Hedge, I. Flora Iranica; Naturhistorisches Museum Wien: Wien, Austria, 1987. [Google Scholar]
- Amin, G.; Sourmaghi, M.S.; Zahedi, M.; Khanavi, M.; Samadi, N. Essential oil composition and antimicrobial activity of Oliveria decumbens. Fitoterapia 2005, 76, 704e707. [Google Scholar] [CrossRef] [PubMed]
- Ghahreman, A.; Okhovvat, A.R. Matching the Old Medicinal Plant Names with Scientific Terminology; University of Tehran Press: Tehran, Iran, 2010. [Google Scholar]
- Khorasani, M.A. Makhzanol-Advieh; Bavardaran Institute: Tehran, Iran, 2004. [Google Scholar]
- Tonekaboni, M.M.M. Tohfatul-Mo’menin; Nashreshahr Institute: Tehran, Iran, 2008. [Google Scholar]
- Saidi, M. Antioxidant activities and chemical composition of essential oils from Satureja khuzestanica, Oliveria decumbens and Thymus daenensis. J. Essent Oil. Bear. Plant 2014, 17, 513e521. [Google Scholar] [CrossRef]
- Karami, A.; Kavoosi, G.; Maggi, F. The emulsion made with essential oil and aromatic water from Oliveria decumbens protects murine macrophages from LPS-induced oxidation and exerts relevant radical scavenging activities. Biocatal. Agric. Biotechnol. 2019, 17, 538–544. [Google Scholar] [CrossRef]
- Eftekhari, M.; Ardekani, M.R.S.; Amin, M.; Attar, F.; Akbarzadeh, T.; Safavi, M.; Karimpour-razkenari, E.; Amini, M.; Isman, M.; Khanavi, M. Oliveria decumbens, a bioactive essential oil: Chemical composition and biological activities. Iran. J. Pharm. Res. 2019, 18, 412. [Google Scholar] [PubMed]
- Jamali, T.; Kavoosi, G.; Ardestani, S.K. In-vitro and in-vivo anti-breast cancer activity of OEO (Oliveria decumbens vent essential oil) through promoting the apoptosis and immunomodulatory effects. J. Ethnopharmacol. 2020, 248, 112313. [Google Scholar] [CrossRef] [PubMed]
- Hyldgaard, M.; Mygind, T.; Meyer, R.L. Essential oils in food preservation: Mode of action, synergies, and interactions with food matrix components. Front. Microbiol. 2012, 25, 3–12. [Google Scholar] [CrossRef]
- Khan, M.S.; Zahin, M.; Hasan, S.; Husain, F.M.; Ahmad, I. Inhibition of quorum sensing regulated bacterial functions by plant essential oils with special reference to clove oil. Lett. Appl. Microbiol. 2009, 49, 354–360. [Google Scholar] [CrossRef]
- Burt, S. Essential oils: Their antibacterial properties and potential applications in foods—a review. Int. J. Food. Microbiol. 2004, 94, 223–253. [Google Scholar] [CrossRef]
- Bahraminejad, S.; Seifolahpour, B.; Amiri, R. Antifungal effects of some medicinal and aromatic plant essential oils against Alternaria solani. J. Crop Prot. 2016, 5, 603–616. [Google Scholar] [CrossRef][Green Version]
- Alizadeh Behbahani, B.; Tabatabaei Yazdi, F.; Vasiee, A.; Mortazavi, S.A. Oliveria decumbens essential oil: Chemical compositions and antimicrobial activity against the growth of some clinical and standard strains causing infection. Microb. Pathog. 2018, 114, 449–452. [Google Scholar] [CrossRef]
- Hajimehdipoor, H.; Samadi, N.; Mozaffarian, V.; Rahimifard, N.; Shoeibi, S.; Pirali, H.M. Chemical composition and antimicrobial activity of Oliveria decumbens volatile oil from west of Iran. J. Med. Plants 2010, 1, 39e44. [Google Scholar]
- Khajehie, N.; Golmakani, M.T.; Eblaghi, M.; Eskandari, M.H. Evaluating the Effects of Microwave-assisted hydrodistillation on antifungal and radical scavenging activities of Oliveria decumbens and Chaerophyllum macropodum essential oils. J. Food. Prot. 2017, 80, 783–791. [Google Scholar] [CrossRef]
- Masoum, S.; Samadi, N.; Mehrara, B.; Mahboubi, M. Potentiality of independent component regression in assessment of the peaks responsible for antimicrobial activity of Satureja hortensis L. and Oliveria decumbens Vent. Using GC-MS. J. Iran. Chem. Soc. 2018, 15, 2007–2016. [Google Scholar] [CrossRef]
- Vazirzadeh, A.; Jalali, S.; Farhadi, A. Antibacterial activity of Oliveria decumbens against Streptococcus iniae in Nile tilapia (Oreochromis niloticus) and its effects on serum and mucosal immunity and antioxidant status. Fish. Shellfish Immunol. 2019, 94, 407–416. [Google Scholar] [CrossRef]
- Bachir, R.G.; Benali, M. Antibacterial activity of the essential oils from the leaves of Eucalyptus globulus against Escherichia coli and Staphylococcus aureus. Asian. Pac. J. Trop Biomed. 2012, 2, 739–742. [Google Scholar] [CrossRef]
- Earl, A.M.; Losick, R.; Kolter, R. Ecology and genomics of Bacillus subtilis. Trends Microbiol. 2008, 16, 269–275. [Google Scholar] [CrossRef]
- Sundin, G.W.; Castiblanco, L.F.; Yuan, X.; Zeng, Q.; Yang, C.H. Bacterial disease management: Challenges, experience, innovation and future prospects: Challenges in bacterial molecular plant pathology. Mol. Plant Pathol. 2016, 17, 1506–1518. [Google Scholar] [CrossRef] [PubMed]
- Nandi, M.; Macdonald, J.; Liu, P.; Weselowski, B.; Yuan, Z.C. Clavibacter michiganensis ssp. michiganensis: Bacterial canker of tomato, molecular interactions and disease management. Mol. Plant Pathol. 2018, 19, 2036–2050. [Google Scholar] [CrossRef] [PubMed]
- Osdaghi, E.; Young, A.J.; Harveson, R.M. Bacterial wilt of dry beans caused by Curtobacterium flaccumfaciens pv. flaccumfaciens: A new threat from an old enemy. Mol. Plant Pathol. 2020, 21, 605–621. [Google Scholar] [PubMed]
- Bansal, K.; Midha, S.; Kumar, S.; Patil, P.B. Ecological and evolutionary insights into Xanthomonas citri pathovar diversity. Appl. Environ. Microbiol. 2017, 83, e02993-16. [Google Scholar] [CrossRef]
- Escobar, M.A.; Dandekar, A.M. Agrobacterium tumefaciens as an agent of disease. Trends. Plant Sci. 2003, 8, 380–386. [Google Scholar] [CrossRef]
- Altundag, S.; Aslim, B. Effect of some endemic plants essential oils on bacterial spot of tomato. J. Plant Pathol. 2011, 93, 37–41. [Google Scholar]
- Wińska, K.; Mączka, W.; Łyczko, J.; Grabarczyk, M.; Czubaszek, A.; Szumny, A. Essential oils as antimicrobial agents–myth or real alternative? Molecules 2019, 24, 2130. [Google Scholar] [CrossRef] [PubMed]
- Aghraz, A.; Benameur, Q.; Gervasi, T.; Ait Dra, L.; Ben-Mahdi, M.H.; Larhsini, M.; Markouk, M.; Cicero, N. Antibacterial activity of Cladanthus arabicus and Bubonium imbricatum essential oils alone and in combination with conventional antibiotics against Enterobacteriaceae isolates. Lett. Appl. Microbiol. 2018, 67, 175–182. [Google Scholar] [CrossRef] [PubMed]
- Benameur, Q.; Gervasi, T.; Pellizzeri, V.; Pľuchtová, M.; Tali-Maama, H.; Assaous, F.; Guettou, B.; Rahal, K.; Gruľová, D.; Dugo, G.; et al. Antibacterial activity of Thymus vulgaris essential oil alone and in combination with cefotaxime against bla ESBL producing multidrug resistant Enterobacteriaceae isolates. Nat. Prod. Res. 2019, 33, 2647–2654. [Google Scholar] [CrossRef]
- Esmaeili, H.; Karami, A.; Maggi, F. Essential oil composition, total phenolic and flavonoids contents, and antioxidant activity of Oliveria decumbens Vent. (Apiaceae) at different phenological stages. J. Clean. Prod. 2018, 198, 91–95. [Google Scholar] [CrossRef]
- Tahmasebi, A.; Hosseini, S.M.; Karami, A.; Afsharifar, A.; Sharifi Olounabadi, A.R. Variation in essential oil composition of Rydingia michauxii at the three developmental stages. Nat. Prod. Res. 2019, 33, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Eyres, G.; Dufour, J.P.; Hallifax, G.; Sotheeswaran, S.; Marriott, P.J. Identification of character-impact odorants in coriander and wild coriander leaves using gas chromatography-olfactometry (GCO) and comprehensive two-dimensional gas chromatographyetime-of-flight mass spectrometry (GC_ GCeTOFMS). J. Sep. Sci. 2005, 28, 1061e1074. [Google Scholar] [CrossRef]
- Karami, A.; Khoushbakht, T.; Esmaeili, H.; Maggi, F. Essential oil chemical variability in Oliveria decumbens (Apiaceae) from different regions of Iran and its relationship with environmental factors. Plants 2020, 9, 680. [Google Scholar] [CrossRef] [PubMed]
- Jamali, T.; Kavoosi, G.; Safavi, M.; Ardestani, S.K. In-vitro evaluation of apoptotic effect of OEO and thymol in 2D and 3D cell cultures and the study of their interaction mode with DNA. Sci. Rep. 2018, 8, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Mahboubi, M.; Taghizadeh, M.; Khamechian, T.; Tamtaji, O.R.; Mokhtari, R.; Talaei, S.A. The wound healing effects of herbal cream containing Oliveria decumbens and Pelargonium graveolens essential oils in diabetic foot ulcer model. World. J. Plast. Surg. 2018, 7, 45–50. [Google Scholar]
- Sajjadi, S.E.; Hoseini, S.A. Essential oil constituents of Oliveria decumbens Vent. J. Essent. Oil. Res. 2002, 14, 220–221. [Google Scholar] [CrossRef]
- Chorianopoulos, N.; Kalpoutzakis, E.; Aligiannis, N.; Mitaku, S.; Nychas, G.J.; Haroutounian, S.A. Essential oils of Satureja, Origanum, and Thymus species: Chemical composition and antibacterial activities against foodborne pathogens. J. Agric. Food Chem. 2004, 52, 8261e8267. [Google Scholar] [CrossRef]
- Sereshti, H.; Izadmanesh, Y.; Samadi, S. Optimized ultrasonic assisted extraction–dispersive liquid–liquid microextraction coupled with gas chromatography for determination of essential oil of Oliveria decumbens Vent. J. Chromatogr. A 2011, 1218, 4593–4598. [Google Scholar] [CrossRef] [PubMed]
- Najafpour Navaei, M.; Mirza, M. Essential oil of Oliveria decumbens Vent. Iran. J. Med. Aromat Plant. 2003, 15, 23–31. [Google Scholar]
- Zouari, S.; Ayadi, I.; Fakhfakh, N.; Jdir, H.; Aloui, L.; Kossentini, M.; Rebai, A.; Zouari, N. Essential oil variation in wild populations of Artemisia saharae (Asteraceae) from Tunisia: Chemical composition, antibacterial and antioxidant properties. Bot. Stud. 2014, 55, 76. [Google Scholar] [CrossRef] [PubMed]
- Nejad, S.; Badi, H.N.; Mehrafarin, A.; Abdossi, V.; Khalighi-Sigaroodi, F. The impact of macro environmental factors on essential oils of Oliveria decumbens Vent. from different regions of Iran. Jundishapur. J. Nat. Pharm. Prod. 2019, 14, e59456. [Google Scholar]
- Morshedloo, M.R.; Craker, L.E.; Salami, A.; Nazeri, V.; Sang, H.; Maggi, F. Effect of prolonged water stress on essential oil content, compositions and gene expression patterns of mono-and sesquiterpene synthesis in two oregano (Origanum vulgare L.) subspecies. Plant. Physiol. Biochem. 2017, 111, 119–128. [Google Scholar] [CrossRef]
- Mahboubi, M.; Feizabadi, M.M.; Haghi, G.; Hosseini, H. Antimicrobial activity and chemical composition of essential oil from Oliveria decumbens Vent. Iran. J. Med. Aromat. Plants. 2008, 24, 56–65. [Google Scholar]
- Gavaric, N.; Mozina, S.S.; Kladar, N.; Bozin, B. Chemical profile, antioxidant and antibacterial activity of thyme and oregano essential oils, thymol and carvacrol and their possible synergism. J. Essent Oil. Bear. Plant 2015, 18, 1013–1021. [Google Scholar] [CrossRef]
- Pirbalouti, A.; Rahimmalek, M.; Malekpoor, F.; Karimi, A. Variation in antibacterial activity, thymol and carvacrol contents of wild populations of’ Thymus daenensis subsp. daenensis’ Celak. Plant Omics. 2011, 4, 209–214. [Google Scholar]
- Zakaria Nabti, L.; Sahli, F.; Laouar, H.; Olowo-okere, A.; Wandjou, N.; Guileine, J.; Maggi, F. Chemical composition and antibacterial activity of essential oils from the Algerian endemic Origanum glandulosum Desf. Against Multidrug-Resistant Uropathogenic E. coli isolates. Antibiotics 2020, 9, 29. [Google Scholar] [CrossRef] [PubMed]
- Memar, M.Y.; Raei, P.; Alizadeh, N.; Aghdam, M.A.; Kafil, H.S. Carvacrol and thymol: Strong antimicrobial agents against resistant isolates. Rev. Med. Microbiol. 2017, 28, 63–68. [Google Scholar] [CrossRef]
- Lambert, R.; Skandamis, P.N.; Coote, P.J.; Nychas, G.J. A study of the minimum inhibitory concentration and mode of action of oregano essential oil, thymol and carvacrol. J. Appl. Microbiol. 2001, 91, 453–462. [Google Scholar] [CrossRef]
- Eloff, J.N. A sensitive and quick microplate method to determine the minimal inhibitory concentration of plant extracts for bacteria. Planta Med. 1998, 64, 711–713. [Google Scholar] [CrossRef] [PubMed]
- Valgas, C.; Souza, S.M.D.; Smânia, E.F.; Smânia, J.A. Screening methods to determine antibacterial activity of natural products. Braz. J. Microbiol. 2007, 38, 369–380. [Google Scholar] [CrossRef]
- Ghasemi Pirbalouti, A.; Nourafcan, H.; Solyamani-Babadi, E. Variation in chemical composition and antibacterial activity of essential oils from Bakhtiari Savory (Satureja bachtiarica Bunge.). J. Essent Oil. Bear. Plant 2017, 20, 474–484. [Google Scholar] [CrossRef]
| Bacteria | Mean | Essential Oils | Chloramphenicol (mg/ml) | ||||
|---|---|---|---|---|---|---|---|
| Nourabad Mamasani | Dehdasht | Behbahan | Kahnoyeh | ||||
| Gram-positive | S. aureus | 0.63 ± 0.25 a* | 0.5 | 0.5 | 1 | 0.5 | 0.2 |
| B. subtilis | 0.45 ± 0.41 a | 0.0625 | 0.25 | 0.5 | 1 | 0.8 | |
| C. michiganensis | 0.44 ± 0.13 a | 0.5 | 0.25 | 0.5 | 0.5 | 0.025 | |
| C. flaccumfaciens | 0.47 ± 0.39 a | 0.125 | 0.25 | 0.5 | 1 | 0.025 | |
| Average MIC against Gram-positive bacteria | 0.49 | 0.30 ± 0.24 a | 0.31 ± 0.13 a | 0.63 ± 0.25 ab | 0.75 ± 0.29 b | 0.26 ± 0.37 a | |
| Gram-negative | X. citri | 1.25 ± 0.5 b | 1 | 1 | 2 | 1 | 0.4 |
| A. tumefaciens | 0.44 ± 0.38 a | 0.25 | 0.25 | 0.25 | 1 | 0.025 | |
| E. coli | 0.69 ± 0.38 ab | 0.25 | 0.5 | 1 | 1 | 0.4 | |
| Average MIC against Gram-negative bacteria | 0.78 | 0.5 ± 0.43 a | 0.58 ± 0.38 a | 1.08 ± 0.88 a | 1 ± 0 a | 0.28 ± 0.22 a | |
| Average MIC against both groups of bacteria | 0.61 | 0.38 ± 0.32 a | 0.43 ± 0.28 ab | 0.82 ± 0.59 bc | 0.86 ± 0.24 c | 0.27 ± 0.29 a | |
| Bacteria | Mean | Essential Oils | Chloramphenicol (mg/ml) | ||||
|---|---|---|---|---|---|---|---|
| Nourabad Mamasani | Dehdasht | Behbahan | Kahnoyeh | ||||
| Gram-positive | S. aureus | 8 ± 5.66 b* | 4 | 8 | 4 | 16 | 5 |
| B.subtilis | 7 ± 2 ab | 4 | 8 | 8 | 8 | 1.6 | |
| C. michiganensis | 4.25 ± 2.87 ab | 1 | 4 | 4 | 8 | 1.6 | |
| C. flaccumfaciens | 2 ± 1.41 a | 1 | 1 | 2 | 4 | 3.2 | |
| Average MBC against Gram-positive bacteria | 5.31 | 2.5 ± 1.73 a | 5.25 ± 3.4 ab | 4.5 ± 2.52 ab | 9 ± 5.03 b | 2.85 ± 1.62 a | |
| Gram-negative | X. citri | 4.5 ± 2.52 a | 2 | 4 | 4 | 8 | 5 |
| A. tumefaciens | 6 ± 6.73 a | 2 | 2 | 4 | 16 | 0.1 | |
| E. coli | 6 ± 6.73 a | 2 | 2 | 4 | 16 | 5 | |
| Average MBC against Gram-negative bacteria | 5.5 | 2 ± 0 a | 2.67 ± 1.15 a | 4 ± 0 a | 13.33 ± 4.62 b | 3.37 ± 2.83 a | |
| Average MBC against both groups of bacteria | 5.39 | 2.29 ± 1.25 a | 4.14 ± 2.85 a | 4.29 ± 1.8 a | 10.86 ± 5.01 b | 3.07 ± 2.01 a | |
| No. | Population | Province | Latitude (N) | Longitude (E) | Altitude (m) | Voucher Specimen |
|---|---|---|---|---|---|---|
| 1 | Nourabad Mamasani | Fars | 30°08′21.6” | 51°33′16.3” | 1268 | 55079 |
| 2 | Kahnoyeh, Lar | Fars | 27°57′59.6” | 53°24′11.8” | 758 | 55076 |
| 3 | Behbahan | Khuzestan | 30°32′09.2” | 50°23′36.5” | 394 | 55075 |
| 4 | Dehdasht | Kohgiluyeh and Boyer-Ahmad | 30°51′07.3” | 50°35′14.5” | 971 | 55080 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Khoshbakht, T.; Karami, A.; Tahmasebi, A.; Maggi, F. The Variability of Thymol and Carvacrol Contents Reveals the Level of Antibacterial Activity of the Essential Oils from Different Accessions of Oliveria decumbens. Antibiotics 2020, 9, 409. https://doi.org/10.3390/antibiotics9070409
Khoshbakht T, Karami A, Tahmasebi A, Maggi F. The Variability of Thymol and Carvacrol Contents Reveals the Level of Antibacterial Activity of the Essential Oils from Different Accessions of Oliveria decumbens. Antibiotics. 2020; 9(7):409. https://doi.org/10.3390/antibiotics9070409
Chicago/Turabian StyleKhoshbakht, Tahereh, Akbar Karami, Aminallah Tahmasebi, and Filippo Maggi. 2020. "The Variability of Thymol and Carvacrol Contents Reveals the Level of Antibacterial Activity of the Essential Oils from Different Accessions of Oliveria decumbens" Antibiotics 9, no. 7: 409. https://doi.org/10.3390/antibiotics9070409
APA StyleKhoshbakht, T., Karami, A., Tahmasebi, A., & Maggi, F. (2020). The Variability of Thymol and Carvacrol Contents Reveals the Level of Antibacterial Activity of the Essential Oils from Different Accessions of Oliveria decumbens. Antibiotics, 9(7), 409. https://doi.org/10.3390/antibiotics9070409







