In Vitro Efficacy of Essential Oils from Melaleuca Alternifolia and Rosmarinus Officinalis, Manuka Honey-based Gel, and Propolis as Antibacterial Agents Against Canine Staphylococcus Pseudintermedius Strains
Abstract
1. Introduction
2. Results
2.1. Antibacterial Activity of Natural Products
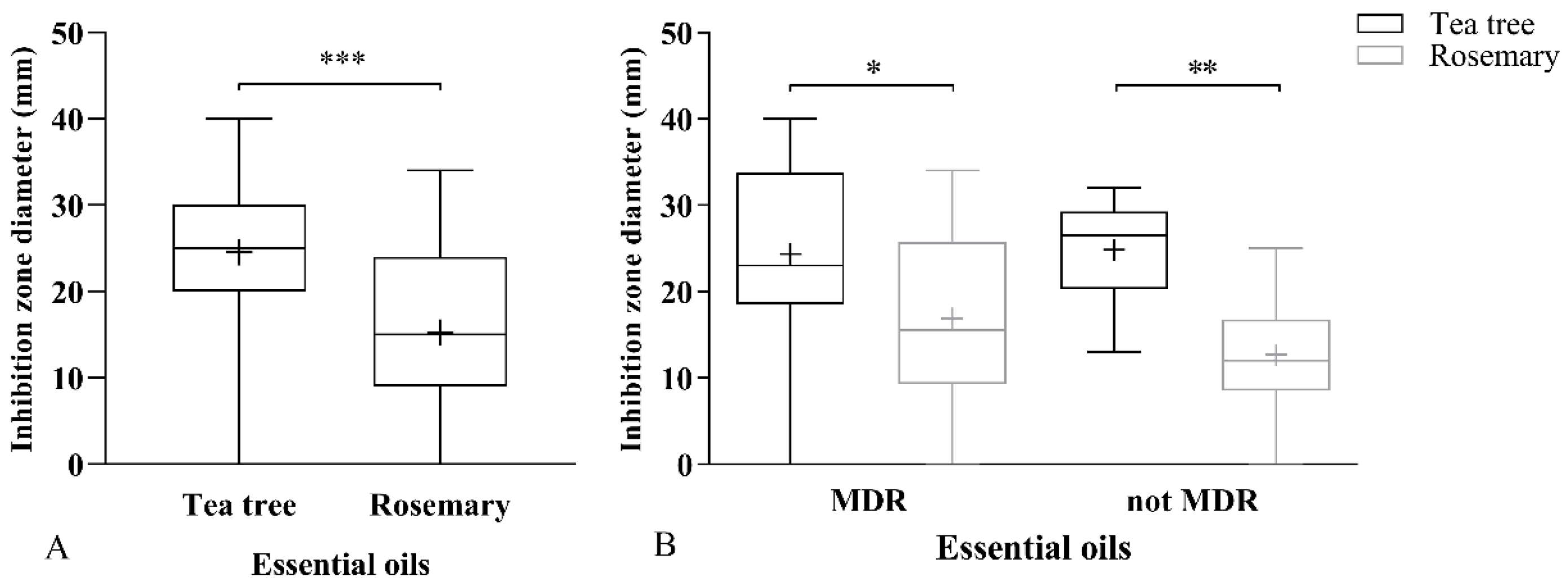
2.1.1. Aromatogram
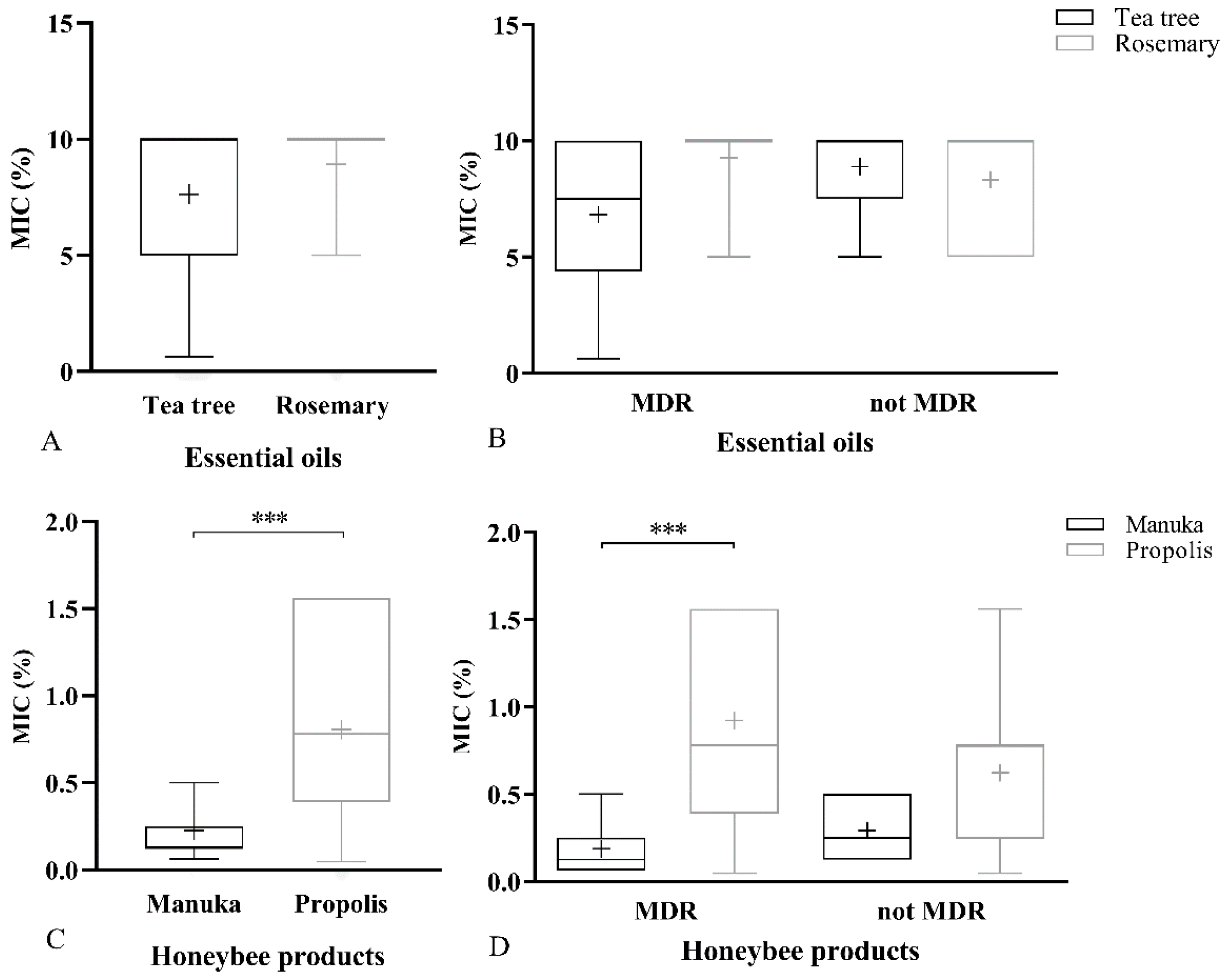
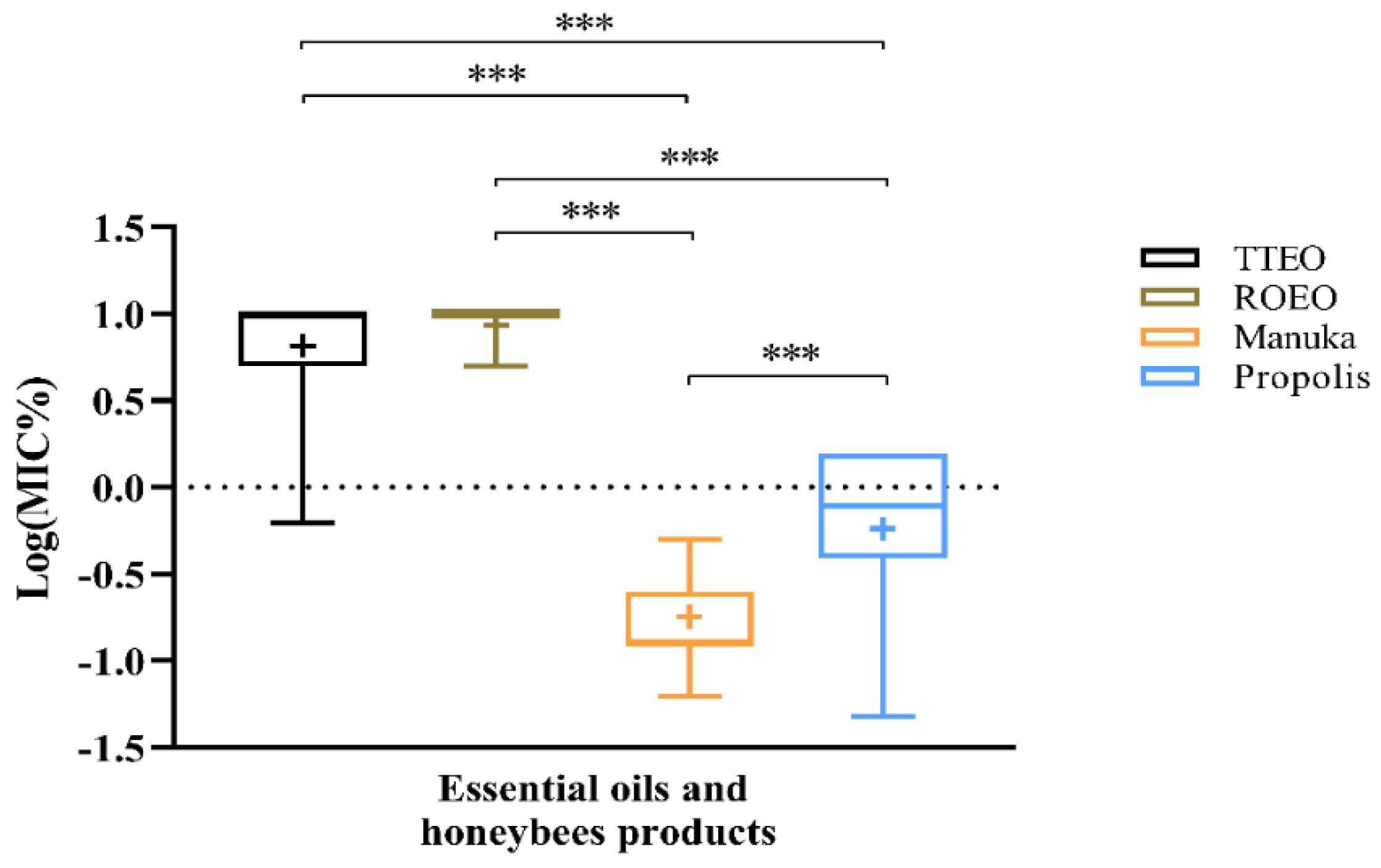
2.1.2. MIC of Natural Products
2.2. Resistance to Antimicrobial Agents
2.3. Genotyping
2.4. Virulence Factors
3. Discussion
4. Conclusions
5. Materials and Methods
5.1. Bacterial Isolation and Idenfication
5.2. DNA Extraction
5.3. Molecular Typing and Clonal Relatedness
5.4. Antimicrobial Susceptibility Testing
5.4.1. Kirby-Bauer Disk Diffusion and Detection of Antimicrobial Resistance Genes (ARG)
5.5. Aromatogram of Tea Tree and Rosmarinus Officinalis
5.6. Minimum Inhibitory Concentration (MIC)
5.6.1. Tea Tree and Rosmarinus Officinalis
5.6.2. Manuka Honey-Based Gel and Propolis
Manuka Honey-Based Gel
Propolis
5.7. Detection of Virulence Factors
5.8. Statistical Analysis
Author Contributions
Funding
Conflicts of Interest
References
- Larsen, R.F.; Boysen, L.; Jessen, L.R.; Guardabassi, L.; Damborg, P. Diversity of Staphylococcus pseudintermedius in carriage sites and skin lesions of dogs with superficial bacterial folliculitis: Potential implications for diagnostic testing and therapy. Vet. Dermatol. 2018, 29, 291-e100. [Google Scholar] [CrossRef]
- Tabatabaei, S.; Najafifar, A.; Askari Badouei, M.; Zahraei Salehi, T.; Ashrafi Tamai, I.; Khaksar, E.; Salah Abbassi, M.; Ghazisaeedi, F. Genetic characterisation of methicillin-resistant Staphylococcus aureus and Staphylococcus pseudintermedius in pets and veterinary personnel in Iran: New insights into emerging methicillin-resistant S. pseudintermedius (MRSP). J. Glob. Antimicrob. Resist. 2019, 16, 6–10. [Google Scholar] [CrossRef]
- Guardabassi, L.; Loeber, M.E.; Jacobson, A. Transmission of multiple antimicrobial-resistant Staphylococcus intermedius between dogs affected by deep pyoderma and their owners. Vet. Microbiol. 2004, 98, 23–27. [Google Scholar] [CrossRef]
- Bartlett, S.J.; Rosenkrantz, W.S.; Sanchez, S. Bacterial contamination of commercial ear cleaners following routine home use. Vet. Dermatol. 2011, 22, 546–553. [Google Scholar] [CrossRef]
- Bannoehr, J.; Guardabassi, L. Staphylococcus pseudintermedius in the dog: Taxonomy, diagnostics, ecology, epidemiology and pathogenicity. Vet. Dermatol. 2012, 23, 253–e52. [Google Scholar] [CrossRef]
- Devriese, L.A.; Vancanneyt, M.; Baele, M.; Vaneechoutte, M.; De Graef, E.; Snauwaert, C.; Cleenwerck, I.; Dawyndt, P.; Swings, J.; Decostere, A.; et al. Staphylococcus pseudintermedius sp. nov., a coagulase-positive species from animals. Int. J. Syst. Evol. Microbiol. 2005, 55, 1569–1573. [Google Scholar] [CrossRef]
- Van Hoovels, L.; Vankeerberghen, A.; Boel, A.; Van Vaerenbergh, K.; De Beenhouwer, H. First case of Staphylococcus pseudintermedius infection in a human. J. Clin. Microbiol. 2006, 44, 4609–4612. [Google Scholar] [CrossRef]
- Duim, B.; Verstappen, K.M.; Broens, E.M.; Laarhoven, L.M.; Van Duijkeren, E.; Hordijk, J.; De Heus, P.; Spaninks, F.; Timmerman, J.A.; Wagenaar, A.J. Changes in the population of methicillin-resistant Staphylococcus pseudintermedius and dissemination of antimicrobial-resistant phenotypes in the Netherlands. J. Clin. Microbiol. 2016, 54, 283–288. [Google Scholar] [CrossRef] [PubMed]
- Guardabassi, L.; Larsen, J.; Weese, J.S.; Butaye, P.; Battisti, A.; Kluytmans, J.; Lloyd, H.D.; Skov, L.R. Public health impact and antimicrobial selection of meticillin-resistant staphylococci in animals. J. Glob. Antimicrob. Resist. 2013, 1, 55–62. [Google Scholar] [CrossRef] [PubMed]
- Duijkeren, E.V.; Catry, B.; Greko, C.; Moreno, M.A.; Constanc, M.; Ruz, M. Review on methicillin-resistant Staphylococcus pseudintermedius. J. Antimicrob. Chemother. 2011, 66, 2705–2714. [Google Scholar] [CrossRef] [PubMed]
- Paul, N.C.; Moodley, A.; Ghibaudo, G.; Guardabassi, L. Carriage of Methicillin-Resistant Staphylococcus pseudintermedius in Small Animal Veterinarians: Indirect Evidence of Zoonotic Transmission. Zoonoses Public Health 2011, 58, 533–539. [Google Scholar] [CrossRef] [PubMed]
- Moodley, A.; Damborg, P.; Nielsen, S.S. Antimicrobial resistance in methicillin susceptible and methicillin resistant Staphylococcus pseudintermedius of canine origin: Literature review from 1980 to 2013. Vet. Microbiol. 2014, 171, 337–341. [Google Scholar] [CrossRef] [PubMed]
- Meroni, G.; Filipe, J.F.S.; Drago, L.; Martino, P.A. Investigation on antibiotic-resistance, biofilm formation and virulence factors in multi drug resistant and non multi drug resistant Staphylococcus pseudintermedius. Microorganisms 2019, 7, 702. [Google Scholar] [CrossRef] [PubMed]
- Wielders, C.L.C.; Vriens, M.R.; Brisse, S.; De Graaf-Miltenburg, L.A.M.; Troelstra, A.; Fleer, A.; Schmitz, F.J.; Verhoef, J.; Fluit, A.C. Evidence for in-vivo transfer of mecA DNA between strains of Staphylococcus aureus. Lancet 2001, 357, 1674–1675. [Google Scholar] [CrossRef]
- Saber, H.; Jasni, A.S.; Jamaluddin, T.Z.M.T.; Ibrahim, R. A Review of Staphylococcal Cassette Chromosome mec (SCC mec) Types in Coagulase-Negative Staphylococci (CoNS) Species. Malays. J. Med. Sci. 2017, 24, 7–18. [Google Scholar] [PubMed]
- Rolo, J.; Worning, P.; Boye Nielsen, J.; Bowden, R.; Bouchami, O.; Damborg, P.; Guardabassi, L.; Perreten, V.; Tomasz, A.; Westh, H.; et al. Evolutionary Origin of the Staphylococcal Cassette Chromosome mec (SCCmec). Antimicrob. Agents Chemother. 2017, 61, e02302-16. [Google Scholar] [CrossRef]
- Wegener, A.; Broens, E.M.; Zomer, A.; Spaninks, M.; Wagenaar, J.A.; Duim, B. Comparative genomics of phenotypic antimicrobial resistances in methicillin-resistant Staphylococcus pseudintermedius of canine origin. Vet. Microbiol. 2018, 225, 125–131. [Google Scholar] [CrossRef]
- Descloux, S.; Rossano, A.; Perreten, V. Characterization of new staphylococcal cassette chromosome mec (SCCmec) and topoisomerase genes in fluoroquinolone- and methicillin-resistant Staphylococcus pseudintermedius. J. Clin. Microbiol. 2008, 46, 1818–1823. [Google Scholar] [CrossRef]
- Ference, E.H.; Danielian, A.; Kim, H.W.; Yoo, F.; Kuan, E.C.; Suh, J.D. Zoonotic Staphylococcus pseudintermedius sinonasal infections: Risk factors and resistance patterns. Int. Forum Allergy Rhinol. 2019, 9, 724–729. [Google Scholar] [CrossRef]
- Harvey, A.L. Medicines from nature: Are natural products still relevant to drug discovery? Trends Pharmacol. Sci. 1999, 20, 196–198. [Google Scholar] [CrossRef]
- Silva-carvalho, R.; Baltazar, F.; Almeida-aguiar, C. Propolis: A Complex Natural Product with a Plethora of Biological Activities That Can Be Explored for Drug Development. Evid. Based Complement. Altern. Med. 2015, 2015, 1–29. [Google Scholar] [CrossRef] [PubMed]
- Yuan, H.; Ma, Q.; Ye, L.; Piao, G. The Traditional Medicine and Modern Medicine from Natural Products. Molecules 2016, 21, 559. [Google Scholar] [CrossRef] [PubMed]
- Yap, P.S.X.; Yiap, B.C.; Ping, H.C.; Lim, S.H.E. Essential Oils, A New Horizon in Combating Bacterial Antibiotic Resistance. Open Microbiol. J. 2014, 8, 6–14. [Google Scholar] [CrossRef] [PubMed]
- Stojiljkovic, J. Antibacterial activities of rosemary essential oils and their components against pathogenic bacteria. Adv. Cytol. Pathol. 2018, 3, 93–96. [Google Scholar] [CrossRef]
- Wińska, K.; Mączka, W.; Łyczko, J.; Grabarczyk, M.; Czubaszek, A.; Szumny, A. Essential oils as antimicrobial agents—Myth or real alternative? Molecules 2019, 24, 2130. [Google Scholar] [CrossRef] [PubMed]
- Cox, S.D.; Mann, C.M.; Markham, J.L.; Bell, H.C.; Gustafson, J.E.; Warmington, J.R.; Wyllie, S.G. The mode of antimicrobial action of the essential oil of Melaleuca alternifolia (Tea tree oil). J. Appl. Microbiol. 2000, 88, 170–175. [Google Scholar] [CrossRef]
- Ait-Ouazzou, A.; Lorán, S.; Bakkali, M.; Laglaoui, A.; Rota, C.; Herrera, A.; Pagan, R.; Conchello, P. Chemical composition and antimicrobial activity of essential oils of Thymus algeriensis, Eucalyptus globulus and Rosmarinus officinalis from Morocco. J. Sci. Food Agric. 2011, 91, 2643–2651. [Google Scholar] [CrossRef]
- Alvarez-Suarez, J.; Gasparrini, M.; Forbes-Hernández, T.; Mazzoni, L.; Giampieri, F. The Composition and Biological Activity of Honey: A Focus on Manuka Honey. Foods 2014, 3, 420–432. [Google Scholar] [CrossRef]
- Plant, R.M.; Dinh, L.; Argo, S.; Shah, M. The Essentials of Essential Oils. Adv. Pediatr. 2019, 66, 111–122. [Google Scholar] [CrossRef]
- Graziano, T.S.; Calil, C.M.; Sartoratto, A.; César, G.; Franco, N. In vitro effects of Melaleuca alternifolia essential oil on growth and production of volatile sulphur compounds by oral bacteria. J. Appl. Oral Sci. 2016, 24, 582–589. [Google Scholar] [CrossRef]
- Johnston, M.; McBride, M.; Dahiya, D.; Owusu-Apenten, R.; Singh Nigam, P. Antibacterial activity of Manuka honey and its components: An overview. AIMS Microbiol. 2018, 4, 655–664. [Google Scholar] [CrossRef] [PubMed]
- Nakajima, Y.; Mukai, K.; Komatsu, E.; Iuchi, T.; Kitayama, Y.; Sugama, J.; Nakatani, T. Evaluation of the Effects of Honey on Acute-Phase Deep Burn Wounds. Evid. Based Complement. Altern. Med. 2013, 2013, 784959. [Google Scholar] [CrossRef] [PubMed]
- Litwin, A.; Ophth, F.R.C.; Sagili, S.; Ophth, F.R.C. Effect of Manuka Honey on Eyelid Wound Healing: A Randomized Controlled Trial. Ophthal. Plast. Reconstr. Surg. 2016, 20, 268–272. [Google Scholar]
- Almasaudi, S.B.; Al-nahari, A.A.M.; Sayed, E.; El-ghany, M.A.; Barbour, E.; Al Muhayawi, S.M.; Al-Jaouni, S.; Azhar, E.; Qari, M.; Qari, Y.A.; et al. Antimicrobial effect of different types of honey on Staphylococcus aureus. Saudi J. Biol. Sci. 2017, 24, 1255–1261. [Google Scholar] [CrossRef]
- Bonifacio, M.A.; Cometa, S.; Cochis, A.; Gentile, P.; Ferreira, A.M.; Azzimonti, B.; Procino, G.; Ceci, E.; Rimondini, E.; De Giglio, E. Antibacterial effectiveness meets improved mechanical properties: Manuka honey/gellan gum composite hydrogels for cartilage repair. Carbohydr. Polym. 2018, 198, 462–472. [Google Scholar] [CrossRef] [PubMed]
- AL-Ani, I.; Zimmermann, S.; Reichling, J.; Wink, M. Antimicrobial Activities of European Propolis Collected from Various Geographic Origins Alone and in Combination with Antibiotics. Medicines 2018, 5, 2. [Google Scholar] [CrossRef]
- Santos-Buelga, C.; González-Paramás, A.M. Phenolic Composition of Propolis. In Bee Products—Chemical and Biological Properties; Alvarez-Suarez, J.M., Ed.; Springer International Publishing: Cham, Switzerland, 2017; pp. 99–111. [Google Scholar] [CrossRef]
- Sunde, M.; Osland, A.M.; Vestby, L.K.; Fanuelsen, H.; Slettemea, J.S. Clonal diversity and biofilm-forming ability of methicillin-resistant Staphylococcus pseudintermedius. J. Antimicrob. Chemother. 2012, 67, 841–848. [Google Scholar]
- Bardiau, M.; Yamazaki, K.; Ote, I.; Misawa, N.; Mainil, J.G. Characterization of methicillin-resistant Staphylococcus pseudintermedius isolated from dogs and cats. Microbiol. Immunol. 2013, 57, 496–501. [Google Scholar]
- Gharsa, H.; Slama, K.B.; Gómez-sanz, E.; Lozano, C.; Klibi, N.; Jouini, A.; Messadi, L.; Boudabous, A.; Torres, C. Antimicrobial Resistance, Virulence Genes, and Genetic Lineages of Staphylococcus pseudintermedius in Healthy Dogs in Tunisia. Microb. Ecol. 2013, 66, 363–368. [Google Scholar] [CrossRef]
- Blaskovic, M.; Rosenkrantz, W.; Neuber, A.; Sauter-Louis, C.; Mueller, R.S. The effect of a spot-on formulation containing polyunsaturated fatty acids and essential oils on dogs with atopic dermatitis. Vet. J. 2014, 199, 39–43. [Google Scholar] [CrossRef][Green Version]
- Oliveira, A.M.P.; Devesa, J.S.P.; Hill, P.B. In vitro efficacy of a honey-based gel against canine clinical isolates of Staphylococcus pseudintermedius and Malassezia pachydermatis. Vet. Dermatol. 2018, 29, 180-e65. [Google Scholar] [CrossRef] [PubMed]
- Piotr, S.; Magdalena, Z.; Joanna, P.; Barbara, K.; Sławomir, M. Essential oils as potential anti-staphylococcal agents. Acta Vet. (Beogr.) 2018, 68, 95–107. [Google Scholar] [CrossRef]
- Song, S.-Y.; Hyun, J.-E.; Kang, J.-H.; Hwang, C.-Y. In vitro antibacterial activity of the manuka essential oil from Leptospermum scoparium combined with Tris-EDTA against Gram-negative bacterial isolates from dogs with otitis externa. Vet. Dermatol. 2020, 31, 81-e6. [Google Scholar] [CrossRef] [PubMed]
- De Medeiros Barbosa, I.; Da Costa Medeiros, J.A.; De Oliveira, K.A.R.; Gomes-Neto, N.J.; Fechine Tavares, J.; Magnani, M.; De Souza, E.L. Efficacy of the combined application of oregano and rosemary essential oils for the control of Escherichia coli, Listeria monocytogenes and Salmonella Enteritidis in leafy vegetables. Food Control 2016, 59, 468–477. [Google Scholar] [CrossRef]
- Takarada, K.; Kimizuka, R.; Takahashi, N.; Honma, K.; Okuda, K.; Kato, T.A. A comparison of the antibacterial efficacies of essential oils against oral pathogens. Oral Microbiol. Immunol. 2004, 19, 61–64. [Google Scholar] [CrossRef]
- Sabo, V.A.; Knezevic, P. Antimicrobial activity of Eucalyptus camaldulensis Dehn. plant extracts and essential oils: A review. Ind. Crops Prod. 2019, 132, 413–429. [Google Scholar] [CrossRef] [PubMed]
- Lima, C.O.; Barreto, H.M.; De Oliveira Lima, E.; Dde Leite Souza, E.; De Siqueira Júnior, J.P. Antimicrobial effect of the essential oil from Rosmarinus officinalis, L.; against Staphylococcus pseudintermedius isolated from dogs. Rev. Bras. Biociênc. 2013, 11, 280–283. [Google Scholar]
- Hussain, A.I.; Anwar, F.; Chatha, S.A.S.; Jabbar, A.; Mahboob, S.; Nigam, P.S. Rosmarinus officinalis essential oil: Antiproliferative, antioxidant and antibacterial activities. Braz. J. Microbiol. 2010, 41, 1070–1078. [Google Scholar] [CrossRef]
- Song, C.Y.; Nam, E.H.; Park, S.H.; Hwang, C.Y. In vitro efficacy of the essential oil from Leptospermum scoparium (manuka) on antimicrobial susceptibility and biofilm formation in Staphylococcus pseudintermedius isolates from dogs. Vet. Dermatol. 2013, 24, 404–409. [Google Scholar] [CrossRef]
- Yildirim, Z.; Hacievliyagil, S.; Onur, N.; Engin, N.; Kurkcuoglu, M.; Iraz, M.; Durmaz, L. Effect of water extract of Turkish propolis on tuberculosis infection in guinea-pigs. Pharmacol. Res. 2004, 49, 287–292. [Google Scholar] [CrossRef]
- Saeed, F.; Ahmad, R.S.; Arshad, M.U.; Niaz, B.; Batool, R.; Naz, R.; Suleris, H.A.R. Propolis to Curb Lifestyle Related Disorders: An Overview. Int. J. Food Prop. 2016, 19, 420–437. [Google Scholar] [CrossRef]
- Veiga, R.S.; Mendonca, S.D.; Mendes, P.B.; Paulino, N.; Mimica, M.J.; Lagareiro Netto, A.A.; Lira, I.S.; Lopez, B.G.-C.; Negrao, V.; Marcucci, M.C. Artepillin C and phenolic compounds responsible for antimicrobial and antioxidant activity of green propolis and Baccharis dracunculifolia. J. Appl. Microbiol. 2017, 122, 911–920. [Google Scholar] [CrossRef]
- Bannoehr, J.; Franco, A.; Iurescia, M.; Battisti, A.; Fitzgerald, J.R. Molecular diagnostic identification of Staphylococcus pseudintermedius. J. Clin. Microbiol. 2009, 47, 469–471. [Google Scholar] [CrossRef]
- Sasaki, T.; Tsubakishita, S.; Tanaka, Y.; Sakusabe, A.; Ohtsuka, M.; Hirotaki, S.; Kawakami, T.; Fukata, T.; Hiramatsu, K. Multiplex-PCR method for species identification of coagulase-positive staphylococci. J. Clin. Microbiol. 2010, 48, 765–769. [Google Scholar] [CrossRef]
- Adwan, K. Fast DNA isolation and PCR protocols for detection of methicillin-resistant staphylococci. Folia Microbiol. 2014, 59, 5–8. [Google Scholar] [CrossRef]
- Solyman, S.M.; Black, C.C.; Duim, B.; Perreten, V.; Duijkeren, E.V.; Wagenaar, J.A.; Eberlein, L.C.; Sadeghji, L.N.; Videla, R.; Bemis, D.A.; et al. Multilocus Sequence Typing for Characterization of Staphylococcus pseudintermedius. J. Clin. Microbiol. 2013, 51, 306–310. [Google Scholar] [CrossRef]
- Bannoehr, J.; Zakour, N.L.B.; Waller, A.S.; Guardabassi, L.; Thoday, K.L.; Van der Broek, A.H.M.; Fitzgerald, J.R. Population Genetic Structure of the Staphylococcus intermedius Group: Insights into agr Diversification and the Emergence of Methicillin-Resistant Strains. J. Bacteriol. 2007, 189, 8685–8692. [Google Scholar] [CrossRef]
- Kadlec, K.; Schwarz, S.; Perreten, V.; Grö Nlund Andersson, U.; Finn, M.; Greko, C.; Moodley, A.; Kania, S.A.; Frank, L.A.; Bemis, D.A.; et al. Molecular analysis of methicillin-resistant Staphylococcus pseudintermedius of feline origin from different European countries and North America. J. Antimicrob. Chemother. 2010, 65, 1826–1837. [Google Scholar] [CrossRef]
- Perreten, V.; Kadlec, K.; Schwarz, S.; Andersson, U.G.; Finn, M.; Greko, C.; Moodley, A.; Kania, S.A.; Frank, L.A.; Bemis, D.A.; et al. Clonal spread of methicillin-resistant Staphylococcus pseudintermedius in Europe and North America: An international multicentre study. J. Antimicrob. Chemother. 2010, 65, 1145–1154. [Google Scholar] [CrossRef]
- Dolinsky, A.L.; Ohiro, R.K.; Fan, W.; Xiao, C.; Wu, F. National Committee for Clinical Laboratory Standards. 2000. Performance standard for antimicrobial susceptibility testing. Document M100–S10. J. Int. Med. Res. 2017, 46, 18. [Google Scholar]
- Strommenger, B.; Kettlitz, C.; Werner, G. Multiplex PCR assay for simultaneous detection of nine clinically relevant antibiotic resistance genes in Staphylococcus aureus. J. Clin. Microbiol. 2003, 41, 4089–4094. [Google Scholar] [CrossRef]
- Kang, M.H.; Chae, M.J.; Yoon, J.W.; Kim, S.G.; Lee, S.Y.; Yoo, J.H.; Park, H.-E. Antibiotic resistance and molecular characterization of ophthalmic Staphylococcus pseudintermedius isolates from dogs. J. Vet. Sci. 2014, 15, 409–415. [Google Scholar] [CrossRef]
- Mith, H.; Duré, R.; Delcenserie, V.; Zhiri, A.; Daube, G.; Clinquart, A. Antimicrobial activities of commercial essential oils and their components against food-borne pathogens and food spoilage bacteria. Food Sci. Nutr. 2014, 2, 403–416. [Google Scholar] [CrossRef]
- Elshikh, M.; Ahmed, S.; Funston, S.; Dunlop, P.; McGaw, M.; Marchant, R.; Banat, I.M. Resazurin-based 96-well plate microdilution method for the determination of minimum inhibitory concentration of biosurfactants. Biotechnol. Lett. 2016, 38, 1015–1019. [Google Scholar] [CrossRef]
- Futagawa-Saito, K.; Sugiyama, T.; Karube, S.; Sakurai, N.; Ba-Thein, W.; Fukuyasu, T. Prevalence and characterization of leukotoxin-producing Staphylococcus intermedius in isolates from dogs and pigeons. J. Clin. Microbiol. 2004, 42, 5324–5326. [Google Scholar] [CrossRef] [PubMed]
- Futagawa-Saito, K.; Suzuki, M.; Ohsawa, M.; Ohshima, S.; Sakurai, N.; Ba-Thein, W.; Fukuyasu, T. Identification and prevalence of an enterotoxin-ralated gene, se-int, in Staphylococcus intermedius isolates from dogs and pigeons. J. Appl. Microbiol. 2004, 96, 1361–1366. [Google Scholar] [CrossRef]
- Lautz, S.; Kanbar, T.; Alber, J.; Lämmler, C.; Weiss, R.; Prenger-Berninghoff, E.; Zschöck, M. Dissemination of the gene encoding exfoliative toxin of Staphylococcus intermedius among strains isolated from dogs during routine microbiological diagnostics. J. Vet. Med. Ser. B Infect. Dis. Vet. Public Health 2006, 53, 434–438. [Google Scholar] [CrossRef]
- Yoon, J.W.; Lee, S.; Park, H. High Prevalence of Fluoroquinolone- and Methicillin-Resistant Staphylococcus pseudintermedius Isolates from Canine Pyoderma and Otitis Externa in Veterinary Teaching Hospital. J. Microbiol. Biotechnol. 2010, 20, 798–802. [Google Scholar]
- Casagrande Proietti, P.; Stefanetti, V.; Hyatt, D.R.; Marenzoni, M.L.; Capomaccio, S.; Coletti, M.; Bietta, A.; Franciosini, M.P.; Passamonti, F. Phenotypic and genotypic characterization of canine pyoderma isolates of Staphylococcus pseudintermedius for biofilm formation. J. Vet. Med. Sci. 2015, 77, 945–951. [Google Scholar] [CrossRef]
| No. of Isolates | Antimicrobial Resistance Profile | Resistance Genes Detected | Enterotoxin Genes Detected a | Other Toxin Genes Detected a | Biofilm Genes Detected a | MLST a | SCCmec Type a |
|---|---|---|---|---|---|---|---|
| 6 | Susceptible | - | se-int; sec2 | siet | icaA; icaD | ST 44 | - |
| 5 | OX-AMC-AML-CL-ENR-MAR | mecA; blaZ; tetM;aacA-aphD | se-int; sec | siet; lukS/F-I | icaA2; icaD5 | ST 1062; ST 713 | IV1; II-III4 |
| 4 | OX-AMC-AML-CL-CVN-TE-ENR-MAR | mecA; blaZ; tetM;tetK; aacA-aphD | se-int3; sec | siet; lukS/F-I3 | icaA; icaD | ST 1062; ST 712 | IV1; II-III3 |
| 2 | TE | tetM | se-int; sec1 | siet | icaA; icaD | ST 108 | - |
| 2 | TE-ENR-MAR-CVN | tetK | sec1 | siet | icaA1; icaD | ST 127; ST 100 | - |
| 1 | CVN | blaZ | se-int | siet | icaA; icaD | ST 28 | - |
| 1 | OX-AMC-AML-CL-CVN-TE-ENR | mecA; blaZ; tetM | sec | siet; lukS/F-I | icaA; icaD | ST 71 | - |
| 1 | AMC-AML-CVN-ENR | blaZ | se-int | siet | icaD | ST 71 | IV |
| 1 | OX-AMC-AML-CVN- TE-ENR-MAR | mecA; blaZ; tetM | siet | icaA; icaD | ST 26 | IV |
| Virulence Genes | MDR (N = 14) | Non-MDR (N = 9) | p |
|---|---|---|---|
| lukS/F-I | 10/14; (71.4%) | 0/9; (0%) | - |
| seccanine | 11/14; (78.6%) | 3/9; (33.3%) | 0.03 |
| se-int | 9/14; (64.3%) | 9/9; (100%) | 0.0427 |
| siet | 14/14; (100%) | 9/9; (100%) | - |
| icaA | 7/14; (42.8%) | 9/9; (100%) | 0.011 |
| icaD | 14/14; (100%) | 9/9; (100%) | - |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Meroni, G.; Cardin, E.; Rendina, C.; Herrera Millar, V.R.; Soares Filipe, J.F.; Martino, P.A. In Vitro Efficacy of Essential Oils from Melaleuca Alternifolia and Rosmarinus Officinalis, Manuka Honey-based Gel, and Propolis as Antibacterial Agents Against Canine Staphylococcus Pseudintermedius Strains. Antibiotics 2020, 9, 344. https://doi.org/10.3390/antibiotics9060344
Meroni G, Cardin E, Rendina C, Herrera Millar VR, Soares Filipe JF, Martino PA. In Vitro Efficacy of Essential Oils from Melaleuca Alternifolia and Rosmarinus Officinalis, Manuka Honey-based Gel, and Propolis as Antibacterial Agents Against Canine Staphylococcus Pseudintermedius Strains. Antibiotics. 2020; 9(6):344. https://doi.org/10.3390/antibiotics9060344
Chicago/Turabian StyleMeroni, Gabriele, Elena Cardin, Charlotte Rendina, Valentina Rafaela Herrera Millar, Joel Fernando Soares Filipe, and Piera Anna Martino. 2020. "In Vitro Efficacy of Essential Oils from Melaleuca Alternifolia and Rosmarinus Officinalis, Manuka Honey-based Gel, and Propolis as Antibacterial Agents Against Canine Staphylococcus Pseudintermedius Strains" Antibiotics 9, no. 6: 344. https://doi.org/10.3390/antibiotics9060344
APA StyleMeroni, G., Cardin, E., Rendina, C., Herrera Millar, V. R., Soares Filipe, J. F., & Martino, P. A. (2020). In Vitro Efficacy of Essential Oils from Melaleuca Alternifolia and Rosmarinus Officinalis, Manuka Honey-based Gel, and Propolis as Antibacterial Agents Against Canine Staphylococcus Pseudintermedius Strains. Antibiotics, 9(6), 344. https://doi.org/10.3390/antibiotics9060344






