Impact of a Diagnosis-Centered Antibiotic Stewardship on Incident Clostridioides difficile Infections in Older Inpatients: An Observational Study
Abstract
1. Introduction
2. Methods
2.1. Patients
2.2. Antibiotics Stewardship Program
3. Results
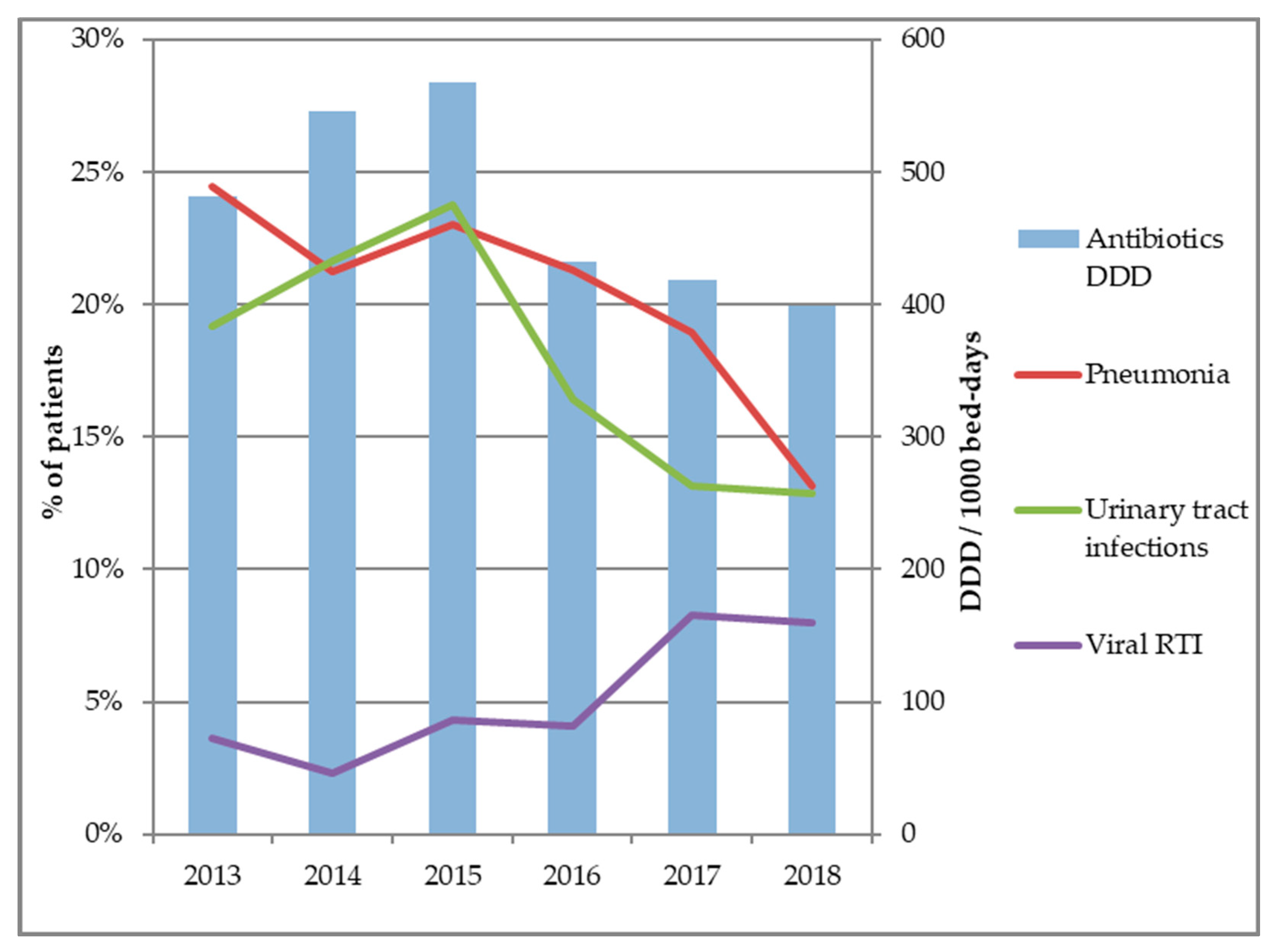
3.1. Infection Diagnoses and Antibiotics Consumption
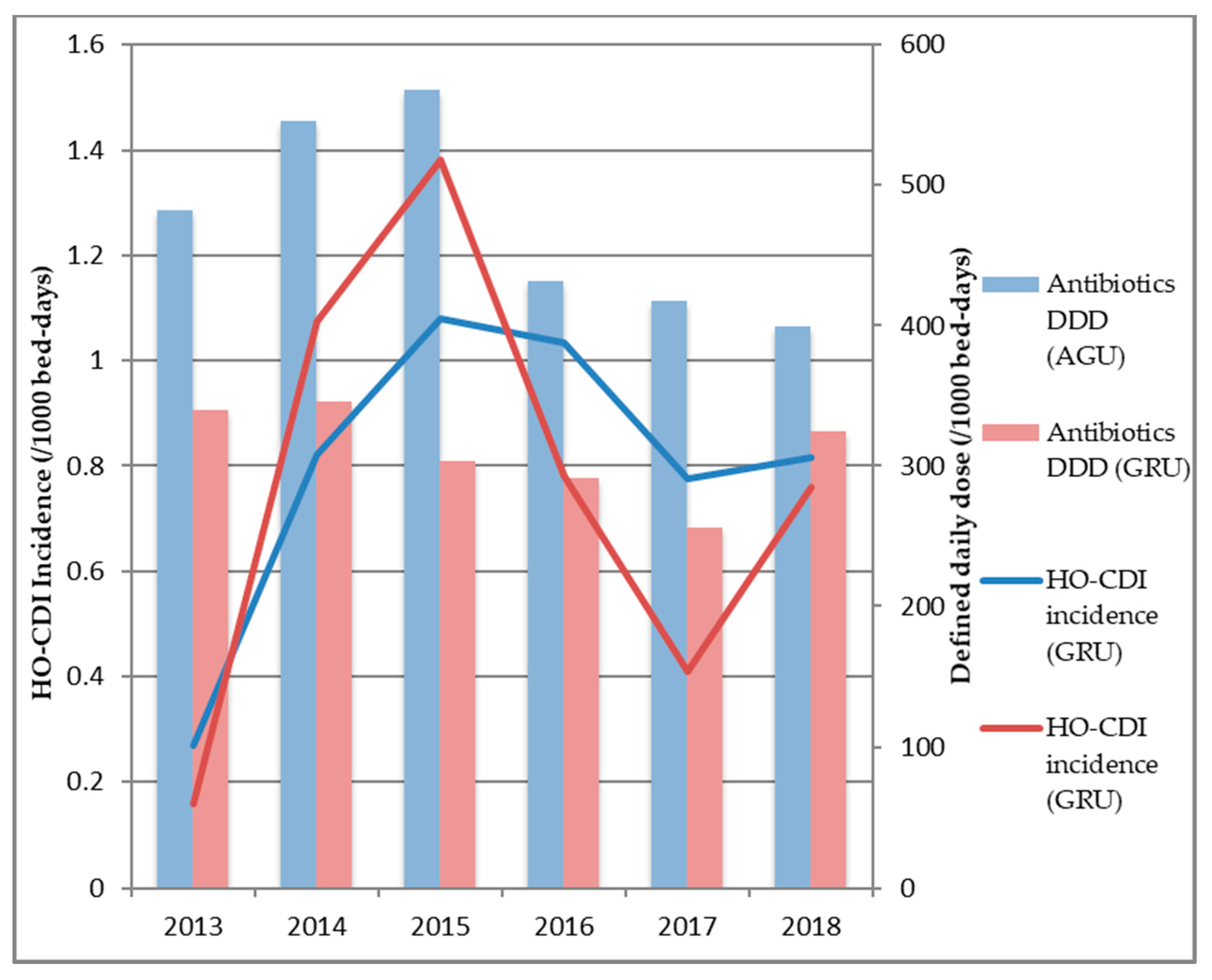
3.2. Antibiotic Consumption and HO-CDI Incidence
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Barbut, F.; Bouée, S.; Longepierre, L.; Goldberg, M.; Bensoussan, C.; Levy-Bachelot, L. Excess mortality between 2007 and 2014 among patients with Clostridium difficile infection: A French health insurance database analysis. J. Hosp. Infect. 2018, 98, 21–28. [Google Scholar] [CrossRef] [PubMed]
- Crew, P.E.; Rhodes, N.J.; O’Donnell, J.N.; Miglis, C.; Gilbert, E.M.; Zembower, T.R.; Qi, C.; Silkaitis, C.; Sutton, S.H.; Scheetz, M.H. Correlation between hospital-level antibiotic consumption and incident health care facility-onset Clostridium difficile infection. Am. J. Infect. Control 2018, 46, 270–275. [Google Scholar] [CrossRef] [PubMed]
- Feazel, L.M.; Malhotra, A.; Perencevich, E.N.; Kaboli, P.; Diekema, D.J.; Schweizer, M.L. Effect of antibiotic stewardship programmes on Clostridium difficile incidence: a systematic review and meta-analysis. J. Antimicrob. Chemother. 2014, 69, 1748–1754. [Google Scholar] [CrossRef]
- Aldeyab, M.A.; Kearney, M.P.; Scott, M.G.; Aldiab, M.A.; Alahmadi, Y.M.; Darwish Elhajji, F.W.; Magee, F.A.; McElnay, J.C. An evaluation of the impact of antibiotic stewardship on reducing the use of high-risk antibiotics and its effect on the incidence of Clostridium difficile infection in hospital settings. J. Antimicrob. Chemother. 2012, 67, 2988–2996. [Google Scholar] [CrossRef] [PubMed]
- Valiquette, L.; Cossette, B.; Garant, M.-P.; Diab, H.; Pépin, J. Impact of a reduction in the use of high-risk antibiotics on the course of an epidemic of Clostridium difficile-associated disease caused by the hypervirulent NAP1/027 strain. Clin. Infect. Dis. 2007, 45 (Suppl. 2), S112–S121. [Google Scholar] [CrossRef]
- Muto, C.A.; Blank, M.K.; Marsh, J.W.; Vergis, E.N.; O’Leary, M.M.; Shutt, K.A.; Pasculle, A.W.; Pokrywka, M.; Garcia, J.G.; Posey, K.; et al. Control of an outbreak of infection with the hypervirulent Clostridium difficile BI strain in a university hospital using a comprehensive “bundle” approach. Clin. Infect. Dis. 2007, 45, 1266–1273. [Google Scholar] [CrossRef] [PubMed]
- Fowler, S.; Webber, A.; Cooper, B.S.; Phimister, A.; Price, K.; Carter, Y.; Kibbler, C.C.; Simpson, A.J.H.; Stone, S.P. Successful use of feedback to improve antibiotic prescribing and reduce Clostridium difficile infection: a controlled interrupted time series. J. Antimicrob. Chemother. 2007, 59, 990–995. [Google Scholar] [CrossRef] [PubMed]
- Carling, P.; Fung, T.; Killion, A.; Terrin, N.; Barza, M. Favorable impact of a multidisciplinary antibiotic management program conducted during 7 years. Infect. Control Hosp. Epidemiol. 2003, 24, 699–706. [Google Scholar] [CrossRef]
- Morgan, F.; Belal, M.; Lisa, B.; Ford, F.; LeMaitre, B.; Psevdos, G. Antimicrobial stewardship program achieved marked decrease in Clostridium difficile infections in a Veterans Hospital. Am. J. Infect. Control 2020. [Google Scholar] [CrossRef] [PubMed]
- Bui, C.; Zhu, E.; Donnelley, M.A.; Wilson, M.D.; Morita, M.; Cohen, S.H.; Brown, J. Antimicrobial stewardship programs that target only high-cost, broad-spectrum antimicrobials miss opportunities to reduce Clostridium difficile infections. Am. J. Infect. Control 2016, 44, 1684–1686. [Google Scholar] [CrossRef] [PubMed]
- Debast, S.B.; Bauer, M.P.; Kuijper, E.J. European society of clinical microbiology and infectious diseases: update of the treatment guidance document for Clostridium difficile infection. Clin. Microbiol. Infect. 2014, 20, 1–26. [Google Scholar] [CrossRef]
- Dubberke, E.R.; Carling, P.; Carrico, R.; Donskey, C.J.; Loo, V.G.; McDonald, L.C.; Maragakis, L.L.; Sandora, T.J.; Weber, D.J.; Yokoe, D.S.; et al. Strategies to prevent Clostridium difficile infections in acute care hospitals: 2014 update. Infect. Control Hosp. Epidemiol. 2014, 35 (Suppl. 2), S48–S65. [Google Scholar] [CrossRef]
- WHO Collaborating Centre for Drug Statistics Methodology, Guidelines for ATC classification and DDD Assignment 2020. 2019. Available online: https://www.whocc.no/atc_ddd_index_and_guidelines/guidelines/ (accessed on 12 December 2019).
- Loeb, M.; Bentley, D.W.; Bradley, S.; Crossley, K.; Garibaldi, R.; Gantz, N.; McGeer, A.; Muder, R.R.; Mylotte, J.; Nicolle, L.E. Development of minimum criteria for the initiation of antibiotics in residents of long-term–care facilities: results of a consensus conference. Infect. Control Hosp. Epidemiol. 2001, 22, 120–124. [Google Scholar] [CrossRef] [PubMed]
- Brendish, N.J.; Malachira, A.K.; Armstrong, L.; Houghton, R.; Aitken, S.; Nyimbili, E.; Ewings, S.; Lillie, P.J.; Clark, T.W. Routine molecular point-of-care testing for respiratory viruses in adults presenting to hospital with acute respiratory illness (ResPOC): a pragmatic, open-label, randomised controlled trial. Lancet Respir. Med. 2017, 5, 401–411. [Google Scholar] [CrossRef]
- Schuetz, P.; Müller, B.; Christ-Crain, M.; Stolz, D.; Tamm, M.; Bouadma, L.; Luyt, C.E.; Wolff, M.; Chastre, J.; Tubach, F.; et al. Procalcitonin to initiate or discontinue antibiotics in acute respiratory tract infections. Cochrane Database Syst. Rev. 2012, CD007498. [Google Scholar] [CrossRef]
- Prendki, V.; Scheffler, M.; Huttner, B.; Garin, N.; Herrmann, F.; Janssens, J.-P.; Marti, C.; Carballo, S.; Roux, X.; Serratrice, C.; et al. Low-dose computed tomography for the diagnosis of pneumonia in elderly patients: A prospective, interventional cohort study. Eur. Respir. J. 2018, 51, 1702375. [Google Scholar] [CrossRef]
- Ticinesi, A.; Lauretani, F.; Nouvenne, A.; Mori, G.; Chiussi, G.; Maggio, M.; Meschi, T. Lung ultrasound and chest x-ray for detecting pneumonia in an acute geriatric ward. Medicine 2016, 95, e4153. [Google Scholar] [CrossRef] [PubMed]
- Caterino, J.M.; Stevenson, K.B. Disagreement between emergency physician and inpatient physician diagnosis of infection in older adults admitted from the emergency department. Acad. Emerg. Med. 2012, 19, 908–915. [Google Scholar] [CrossRef]
- Schoevaerdts, D.; Glupczynski, Y.; Delaere, B.; Hecq, J.-D.; Swine, C. Effect of a 5-year multidisciplinary collaborative program on antibiotic consumption in an acute geriatric ward. J. Am. Geriatr. Soc. 2007, 55, 145–147. [Google Scholar] [CrossRef]
- Slimings, C.; Riley, T.V. Antibiotics and hospital-acquired Clostridium difficile infection: update of systematic review and meta-analysis. J. Antimicrob. Chemother. 2014, 69, 881–891. [Google Scholar] [CrossRef]
- Bonnassot, P.; Barben, J.; Tetu, J.; Bador, J.; Bonniaud, P.; Manckoundia, P.; Putot, A. Clostridioides difficile infection after pneumonia in older patients: Which antibiotic is at lower risk? J. Hosp. Infect. 2020, S0195-6701, 30240-1. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.C.; Kim, E.J.; Heo, J.Y.; Choi, Y.H.; Ahn, J.Y.; Jeong, S.J.; Ku, N.S.; Choi, J.Y.; Yeom, J.-S.; Kim, H.Y. Impact of an infectious disease specialist on an antimicrobial stewardship program at a resource-limited, non-academic community hospital in Korea. J. Clin. Med. 2019, 8, 1293. [Google Scholar] [CrossRef] [PubMed]
- Zafar, A.B.; Gaydos, L.A.; Furlong, W.B.; Nguyen, M.H.; Mennonna, P.A. Effectiveness of infection control program in controlling nosocomial Clostridium difficile. Am. J. Infect. Control. 1998, 26, 588–593. [Google Scholar] [CrossRef] [PubMed]
- Mills, J.P.; Rao, K.; Young, V.B. Probiotics for prevention of Clostridium difficile infection. Curr. Opin. Gastroenterol. 2018, 34, 3–10. [Google Scholar] [CrossRef] [PubMed]
- Shen, N.T.; Maw, A.; Tmanova, L.L.; Pino, A.; Ancy, K.; Crawford, C.V.; Simon, M.S.; Evans, A.T. Timely use of probiotics in hospitalized adults prevents Clostridium difficile infection: a systematic review with meta-regression analysis. Gastroenterology 2017, 152, 1889–1900.e9. [Google Scholar] [CrossRef]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Putot, A.; Astruc, K.; Barben, J.; Mihai, A.M.; Nuss, V.; Bador, J.; Putot, S.; Dipanda, M.; Laborde, C.; Vovelle, J.; et al. Impact of a Diagnosis-Centered Antibiotic Stewardship on Incident Clostridioides difficile Infections in Older Inpatients: An Observational Study. Antibiotics 2020, 9, 303. https://doi.org/10.3390/antibiotics9060303
Putot A, Astruc K, Barben J, Mihai AM, Nuss V, Bador J, Putot S, Dipanda M, Laborde C, Vovelle J, et al. Impact of a Diagnosis-Centered Antibiotic Stewardship on Incident Clostridioides difficile Infections in Older Inpatients: An Observational Study. Antibiotics. 2020; 9(6):303. https://doi.org/10.3390/antibiotics9060303
Chicago/Turabian StylePutot, Alain, Karine Astruc, Jeremy Barben, Anca Maria Mihai, Valentine Nuss, Julien Bador, Sophie Putot, Mélanie Dipanda, Caroline Laborde, Jeremie Vovelle, and et al. 2020. "Impact of a Diagnosis-Centered Antibiotic Stewardship on Incident Clostridioides difficile Infections in Older Inpatients: An Observational Study" Antibiotics 9, no. 6: 303. https://doi.org/10.3390/antibiotics9060303
APA StylePutot, A., Astruc, K., Barben, J., Mihai, A. M., Nuss, V., Bador, J., Putot, S., Dipanda, M., Laborde, C., Vovelle, J., Da Silva, S., Mazen, E., Aho Glélé, L. S., & Manckoundia, P. (2020). Impact of a Diagnosis-Centered Antibiotic Stewardship on Incident Clostridioides difficile Infections in Older Inpatients: An Observational Study. Antibiotics, 9(6), 303. https://doi.org/10.3390/antibiotics9060303





