Awareness of Appropriate Antibiotic Use in Primary Care for Influenza-Like Illness: Evidence of Improvement from UK Population-Based Surveys
Abstract
1. Introduction
2. Methods
2.1. Survey Design
2.2. Health State A: You Have
- A temperature,
- Aching muscles,
- A headache,
- A dry chesty cough,
- A sore throat,
- And you feel weak
2.3. Survey Participants
2.4. Statistical Analysis
3. Results
3.1. Shifts in Attitudes over Time
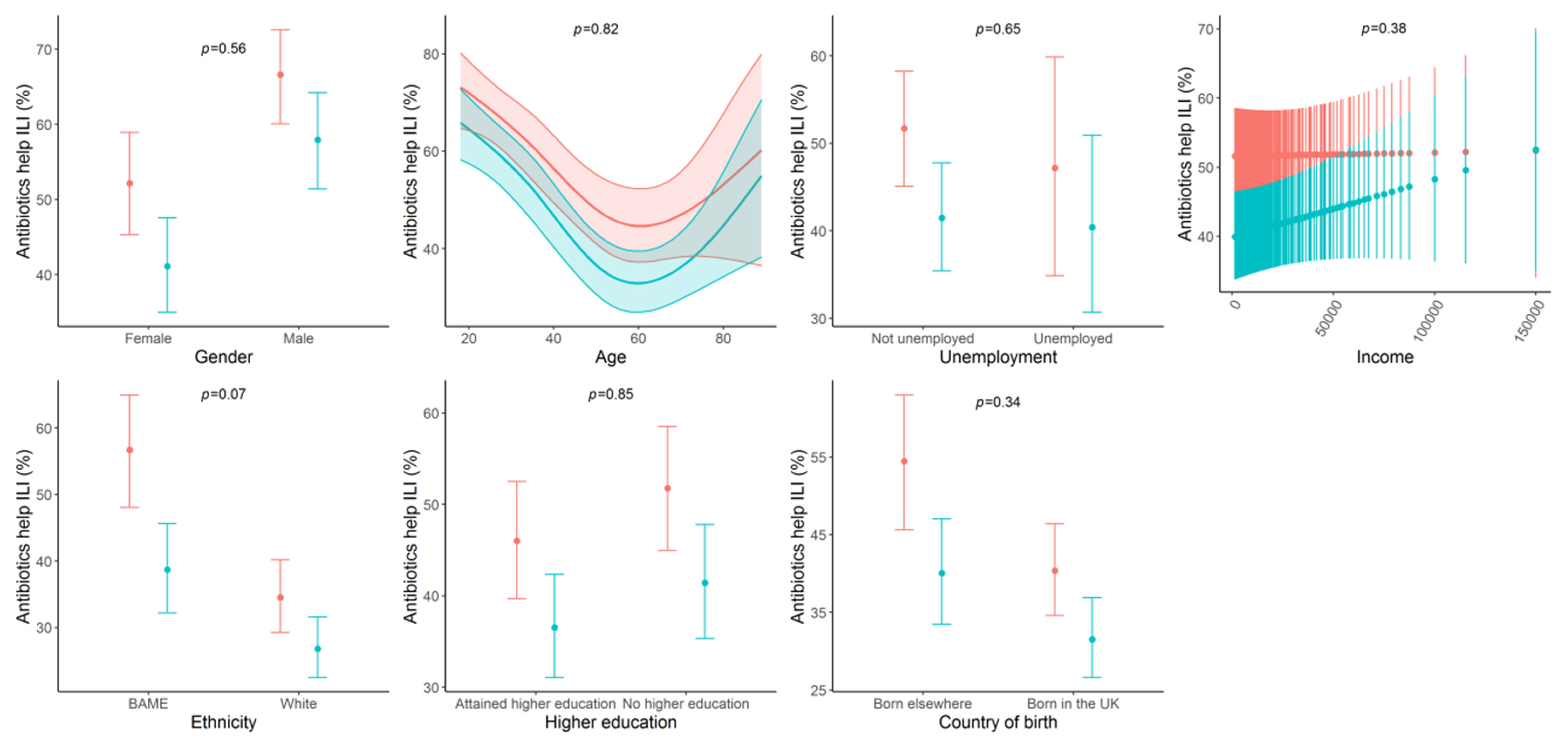
3.2. Shifts in Awareness among Subgroups
4. Discussion
4.1. Summary
4.2. Strengths and Limitations
4.3. Comparison with Existing Literature
4.4. Implications for Future Research and Clinical Practice
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Ethical approval
References
- Chatterjee, A.; Modarai, M.; Naylor, N.R.; Boyd, S.E.; Atun, R.; Barlow, J.; Holmes, A.H.; Johnson, A.; Robotham, J.V. Quantifying drivers of antibiotic resistance in humans: A systematic review. Lancet. Infect. Dis. 2018, 18, e368–e378. [Google Scholar] [CrossRef]
- Pouwels, K.B.; Freeman, R.; Muller-Pebody, B.; Rooney, G.; Henderson, K.L.; Robotham, J.V.; Smieszek, T. Association between use of different antibiotics and trimethoprim resistance: Going beyond the obvious crude association. J. Antimicrob. Chemother. 2018, 73, 1700–1707. [Google Scholar] [CrossRef] [PubMed]
- Public Health England. English Surveillance Programme for Antimicrobial Utilisation and Resistance (ESPAUR) Report—GOV.UK. Available online: https://www.gov.uk/government/publications/english-surveillance-programme-antimicrobial-utilisation-and-resistance-espaur-report (accessed on 3 December 2018).
- Pouwels, K.B.; Dolk, F.C.K.; Smith, D.R.M.; Robotham, J.V.; Smieszek, T. Actual versus ‘ideal’ antibiotic prescribing for common conditions in English primary care. J. Antimicrob. Chemother. 2018, 73, 19–26. [Google Scholar] [CrossRef] [PubMed]
- Smieszek, T.; Pouwels, K.B.; Dolk, F.C.K.; Smith, D.R.M.; Hopkins, S.; Sharland, M.; Hay, A.D.; Moore, M.V.; Robotham, J.V. Potential for reducing inappropriate antibiotic prescribing in English primary care. J. Antimicrob. Chemother. 2018, 73, ii36–ii43. [Google Scholar] [CrossRef] [PubMed]
- Pouwels, K.B.; Dolk, F.C.K.; Smith, D.R.M.; Smieszek, T.; Robotham, J.V. Explaining variation in antibiotic prescribing between general practices in the UK. J. Antimicrob. Chemother. 2018, 73, ii27–ii35. [Google Scholar] [PubMed]
- Hope, E.C.; Crump, R.E.; Hollingsworth, T.D.; Smieszek, T.; Robotham, J.V.; Pouwels, K.B. Identifying English Practices that Are High Antibiotic Prescribers Accounting for Comorbidities and Other Legitimate Medical Reasons for Variation. EClinicalMedicine 2018, 6, 36–41. [Google Scholar] [CrossRef]
- Dolk, F.C.K.; Pouwels, K.B.; Smith, D.R.; Robotham, J.V.; Smieszek, T. Antibiotics in primary care in England: Which antibiotics are prescribed and for which conditions? J. Antimicrob. Chemother. 2018, 73, ii2–ii10. [Google Scholar] [CrossRef]
- Coenen, S.; Francis, N.; Kelly, M.; Hood, K.; Nuttall, J.; Little, P.; Verheij, T.J.M.; Melbye, H.; Goossens, H.; Butler, C.C. Are patient views about antibiotics related to clinician perceptions, management and outcome? A multi-country study in outpatients with acute cough. PLoS ONE 2013, 8, e76691. [Google Scholar] [CrossRef]
- Coenen, S.; Michiels, B.; Renard, D.; Denekens, J.; Royen, P.V. Antibiotic prescribing for acute cough: The effect of perceived patient demand. Br. J. Gen. Pract. 2006, 56, 183–190. [Google Scholar]
- Tonkin-Crine, S.; Yardley, L.; Little, P. Antibiotic prescribing for acute respiratory tract infections in primary care: A systematic review and meta-ethnography. J. Antimicrob. Chemother. 2011, 66, 2215–2223. [Google Scholar] [CrossRef]
- McNulty, C.A.; Nichols, T.; French, D.P.; Joshi, P.; Butler, C.C. Expectations for consultations and antibiotics for respiratory tract infection in primary care: The RTI clinical iceberg. Br. J. Gen. Pract. 2013, 63, e429–e436. [Google Scholar] [CrossRef] [PubMed]
- Roope, L.S.J.; Tonkin-Crine, S.; Butler, C.C.; Crook, D.; Peto, T.; Peters, M.; Walker, A.S.; Wordsworth, S. Reducing demand for antibiotic prescriptions: Evidence from an online survey of the general public on the interaction between preferences, beliefs and information, United Kingdom, 2015. Euro. Surveill. 2018, 23. [Google Scholar] [CrossRef] [PubMed]
- Roope, L.S.J.; Tonkin-Crine, S.; Herd, N.; Michie, S.; Pouwels, K.B.; Castro-Sanchez, E.; Sallis, A.; Hopkins, S.; Robotham, J.V.; Crook, D.W.; et al. Reducing expectations for antibiotics in primary care: A randomised experiment to test the response to fear based messages about antimicrobial resistance. BMC Med. 2020, 18, 110. [Google Scholar] [CrossRef] [PubMed]
- Antibiotic Resistance Challenge (ARC). Available online: http://www.nhs.uk/NHSEngland/ARC/Pages/AboutARC.aspx (accessed on 15 January 2019).
- Antibiotic Guardian Campaign. Available online: https://antibioticguardian.com/ (accessed on 15 January 2019).
- Alliance for the Prudent Use of Antibiotics. Available online: http://emerald.tufts.edu/med/apua/about_issue/antibiotic_res.shtml (accessed on 15 January 2019).
- World Health Organization. Antimicrobial Resistance. Available online: https://www.who.int/en/news-room/fact-sheets/detail/antimicrobial-resistance (accessed on 15 January 2019).
- RCGP Research and Surveillance Centre. Available online: https://www.rcgp.org.uk/clinical-and-research/our-programmes/research-and-surveillance-centre/public-health-data.aspx (accessed on 24 September 2020).
- Bartlett, J.W.; Carpenter, J.R.; Tilling, K.; Vansteelandt, S. Improving upon the efficiency of complete case analysis when covariates are MNAR. Biostatistics 2014, 15, 719–730. [Google Scholar]
- White, I.R.; Carlin, J.B. Bias and efficiency of multiple imputation compared with complete-case analysis for missing covariate values. Stat. Med. 2010, 29, 2920–2931. [Google Scholar] [CrossRef]
- Singer, M.; Bulled, N.; Ostrach, B.; Mendenhall, E. Syndemics and the biosocial conception of health. Lancet 2017, 389, 941–950. [Google Scholar] [CrossRef]
- Thomson, K.; Berry, R.; Robinson, T.; Brown, H.; Bambra, C.; Todd, A. An examination of trends in antibiotic prescribing in primary care and the association with area-level deprivation in England. BMC Public Health 2020, 20, 1148. [Google Scholar] [CrossRef]
- Adekanmbi, V.; Jones, H.; Farewell, D.; Francis, N.A. Antibiotic use and deprivation: An analysis of Welsh primary care antibiotic prescribing data by socioeconomic status. J. Antimicrob. Chemother. 2020, 75, 2363–2371. [Google Scholar] [CrossRef]
- The Review on Antimicrobial Resistance. Antimicrobial Resistance: Tackling a Crisis for the Health and Wealth of Nations. Available online: https://amr-review.org/sites/default/files/AMR%20Review%20Paper%20-%20Tackling%20a%20crisis%20for%20the%20health%20and%20wealth%20of%20nations_1.pdf (accessed on 24 September 2020).
- The Review on Antimicrobial Resistance. Tackling Drug-Resistant Infections Globally: Final Report and Recommendations. Available online: https://amr-review.org/sites/default/files/160525_Final%20paper_with%20cover.pdf (accessed on 24 September 2020).
- Newitt, S.; Oloyede, O.; Puleston, R.; Hopkins, S.; Ashiru-Oredope, D. Demographic, knowledge and impact analysis of 57,627 antibiotic guardians who have pledged to contribute to tackling antimicrobial resistance. Antibiotics. 2019, 8, 21. [Google Scholar] [CrossRef]
- McNulty, C.A.M. European Antibiotic Awareness Day 2012: General practitioners encouraged to TARGET antibiotics through guidance, education and tools. J. Antimicrob. Chemother. 2012, 67, 2543–2546. [Google Scholar] [CrossRef]
- Royal College of General Practitioners. TARGET Antibiotic Toolkit. Available online: https://www.rcgp.org.uk/clinical-and-research/resources/toolkits/target-antibiotic-toolkit.aspx (accessed on 20 January 2019).
- Johnson, A.; Ashiru-Oredope, D.; Beech, E.; Johnson, A.P.; Ashiru-Oredope, D.; Beech, E. Antibiotic Stewardship Initiatives as Part of the UK 5-Year Antimicrobial Resistance Strategy. Antibiotics 2015, 4, 467–479. [Google Scholar] [CrossRef] [PubMed]
- McNulty, C.; Hawking, M.; Lecky, D.; Jones, L.; Owens, R.; Charlett, A.; Butler, C.; Moore, P.; Francis, N. Effects of primary care antimicrobial stewardship outreach on antibiotic use by general practice staff: Pragmatic randomized controlled trial of the TARGET antibiotics workshop. J. Antimicrob. Chemother. 2018, 73, 1423–1432. [Google Scholar] [CrossRef] [PubMed]
- Hood, G.; Toleikyte, L.; Ashiru-Oredope, D. Assessing National Antimicrobial Resistance Campaigns Using a Health Equity Assessment Tool (HEAT). Antibiotics 2019, 8, 121. [Google Scholar] [CrossRef] [PubMed]
- Public Health England. Surveillance of Influenza and Other Respiratory Viruses in the UK: Winter 2018 to 2019. Available online: https://assets.publishing.service.gov.uk/government/uploads/system/uploads/attachment_data/file/839350/Surveillance_of_influenza_and_other_respiratory_viruses_in_the_UK_2018_to_2019-FINAL.pdf (accessed on 24 September 2020).
- Gualano, M.R.; Gili, R.; Scaioli, G.; Bert, F.; Siliquini, R. General population’s knowledge and attitudes about antibiotics: A systematic review and meta-analysis. Pharmacoepidemiol. Drug Saf. 2015, 24, 2–10. [Google Scholar] [CrossRef] [PubMed]
- Public Health England. Fingertips. AMR Local Indicators. Antibiotic Prescribing. Trends. Available online: https://fingertips.phe.org.uk/profile/amr-local-indicators/data#page/4/gid/1938132909/pat/152/par/E38000001/ati/7/are/B83620 (accessed on 18 January 2019).
- Moore, M.; Little, P.; Rumsby, K.; Kelly, J.; Watson, L.; Warner, G.; Fahey, T.; Williamson, I. Effect of antibiotic prescribing strategies and an information leaflet on longer-term reconsultation for acute lower respiratory tract infection. Br. J. Gen. Pract. 2009, 59, 728–734. [Google Scholar] [CrossRef]
- Little, P.; Gould, C.; Williamson, I.; Warner, G.; Gantley, M.; Kinmonth, A.L. Reattendance and complications in a randomised trial of prescribing strategies for sore throat: The medicalising effect of prescribing antibiotics. BMJ 1997, 315, 350–352. [Google Scholar] [CrossRef] [PubMed]
- McNulty, C.A.M.; Nichols, T.; Boyle, P.J.; Woodhead, M.; Davey, P. The English antibiotic awareness campaigns: Did they change the public’s knowledge of and attitudes to antibiotic use? J. Antimicrob. Chemother. 2010, 65, 1526–1533. [Google Scholar] [CrossRef]
- Cals, J.W.L.; Boumans, D.; Lardinois, R.J.M.; Gonzales, R.; Hopstaken, R.M.; Butler, C.C.; Dinant, G.-J. Public beliefs on antibiotics and respiratory tract infections: An internet-based questionnaire study. Br. J. Gen. Pract. 2007, 57, 942–947. [Google Scholar] [CrossRef]
- Pechère, J.C. Patients’ Interviews and Misuse of Antibiotics. Clin. Infect. Dis. 2001, 33, S170–S173. [Google Scholar] [CrossRef]
- Shallcross, L.; Beckley, N.; Rait, G.; Hayward, A.; Petersen, I. Antibiotic prescribing frequency amongst patients in primary care: A cohort study using electronic health records. J. Antimicrob. Chemother. 2017, 72, 1818–1824. [Google Scholar] [CrossRef]
| Survey 1 (May–June 2015) (n = 2064) a | Survey 2 (Oct–Nov 2016) (n = 4000) a | Survey 3 (Mar 2017) (n = 4000) a | |
|---|---|---|---|
| Gender | |||
| Male | 994 (48.2%) | 1941 (48.5%) | 1941 (48.5%) |
| Female | 1067 (51.7%) | 2055 (51.4%) | 2055 (51.4%) |
| Missing | 3 (0.1%) | 4 (0.1%) | 4 (0.1%) |
| Age (years) | |||
| Mean (SD) | 44.2 (15.7) | 46.6 (16.9) | 46.5 (16.8) |
| Ethnicity | |||
| White | 1821 (88.2%) | 3624 (90.6%) | 3606 (90.2%) |
| Black, and minority ethnic (BAME) | 221 (10.7%) | 352 (8.8%) | 372 (9.3%) |
| Missing | 22 (1.1%) | 24 (0.6%) | 22 (0.6%) |
| Religion | |||
| Christian | 1019 (49.4%) | 1986 (49.6%) | 1858 (46.5%) |
| No or other religion | 988 (47.9%) | 1913 (47.8%) | 2049 (51.2%) |
| Missing | 57 (2.8%) | 101 (2.5%) | 93 (2.3%) |
| Married/civil partnership/live with a partner | |||
| Yes | 1351 (65.5%) | 2720 (68.0%) | 2668 (66.7%) |
| No | 713 (34.5%) | 1280 (32.0%) | 1332 (33.3%) |
| Have dependent children | |||
| Yes | 816 (39.5%) | 1600 (40%) | 1600 (40%) |
| No | 1248 (60.5%) | 2400 (60%) | 2400 (60%) |
| Higher education | |||
| Attained higher education | 954 (46.2%) | 1875 (46.9%) | 1756 (43.9%) |
| Did not attain higher education | 1093 (53.0%) | 2125 (53.1%) | 2244 (56.1%) |
| Missing | 17 (0.8%) | 0 (0%) | 0 (0%) |
| Unemployed | |||
| Yes | 105 (5.1%) | 202 (5.0%) | 184 (4.6%) |
| No | 1959 (94.9%) | 3798 (95.0%) | 3816 (95.4%) |
| Household equivalent income £ | |||
| Mean (SD) | 22,405 (18,343) | 22,109 (17,123) | 20,477 (15,012) |
| Missing | 186 (9.0%) | 390 (9.8%) | 357 (8.9%) |
| Region | |||
| England | 1780 (86.2%) | 3381 (84.5%) | 8542 (84.9%) |
| Northern Ireland | 32 (1.6%) | 104 (2.6%) | 240 (2.4%) |
| Scotland | 161 (7.8%) | 318 (8.0%) | 797 (7.9%) |
| Wales | 91 (4.4%) | 197 (4.9%) | 485 (4.8%) |
| Antibiotic use | |||
| Taken antibiotics for any condition in last 12 months | 815 (39%) | 1367 (34%) | 1422 (36%) |
| Survey 1 (May–June 2015) n = 2064 n (%; 95% CI) | Survey 2 (Oct–Nov 2016) n = 4000 n (%; 95% CI) | Survey 3 (March 2017) n = 4000 n (%; 95% CI) | p-Value d | |
|---|---|---|---|---|
| Definitely/Probably go to GP for ILI | 988 (48%; 46–50%) | 1587 (40%; 38-–41%) | 1438 (36%; 34–37%) | <0.0001 |
| Would ask GP for antibiotics if went | 706/1816 a (39%; 37–41%) | 1084/3298 a (33%; 31–24%) | 975/3236 a (30%; 29–32%) | <0.0001 |
| Think antibiotics would definitely/probably help ILI | 762 (37%; 35–39%) | 1,153 (29%; 27–30%) | 1,112 (28%; 26–29%) | <0.0001 |
| Have taken antibiotics for ILI in last 12 months | 426 (21%; 19–22%) | 595 (15%; 14–16%) | 580 (14%; 13–16%) | <0.0001 |
| Definitely/Probably go to GP for child with ILI | 673/816 b (82%, 80–85%) | 1232/1600 b (77%; 75–79%) | 1191/1600 b (74%; 72–77%) | <0.0001 |
| Would ask GP for antibiotics for child with ILI if went | 419/797 c (53%; 49–56%) | 734/1519 c (48%; 46–51%) | 671/1526 c (44%; 41–46%) | <0.0001 |
| Think antibiotics would definitely/probably help child with ILI | 430/816 b (53%; 49–56%) | 740/1600 b (46%; 44–49%) | 688/1600 b (43%; 41–45%) | <0.0001 |
| Child has taken antibiotics for ILI in last 12 months | 200/816 (25%; 22–28%) | 338/1600 (21%; 19–23%) | 319/1600 (20%; 18–22%) | 0.0097 |
| AMR information (Box 1) is very/somewhat new (very/somewhat surprising in Survey 1) | 705 (34%; 32–36%) | 303/1000 (30%; 28–33%) | 285/1000 (28%; 26–31%) | 0.0017 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pouwels, K.B.; Roope, L.S.J.; Buchanan, J.; Morrell, L.; Tonkin-Crine, S.; Peters, M.; Jones, L.F.; Castro-Sánchez, E.; Crook, D.W.; Peto, T.; et al. Awareness of Appropriate Antibiotic Use in Primary Care for Influenza-Like Illness: Evidence of Improvement from UK Population-Based Surveys. Antibiotics 2020, 9, 690. https://doi.org/10.3390/antibiotics9100690
Pouwels KB, Roope LSJ, Buchanan J, Morrell L, Tonkin-Crine S, Peters M, Jones LF, Castro-Sánchez E, Crook DW, Peto T, et al. Awareness of Appropriate Antibiotic Use in Primary Care for Influenza-Like Illness: Evidence of Improvement from UK Population-Based Surveys. Antibiotics. 2020; 9(10):690. https://doi.org/10.3390/antibiotics9100690
Chicago/Turabian StylePouwels, Koen B., Laurence S. J. Roope, James Buchanan, Liz Morrell, Sarah Tonkin-Crine, Michele Peters, Leah F. Jones, Enrique Castro-Sánchez, Derrick W. Crook, Tim Peto, and et al. 2020. "Awareness of Appropriate Antibiotic Use in Primary Care for Influenza-Like Illness: Evidence of Improvement from UK Population-Based Surveys" Antibiotics 9, no. 10: 690. https://doi.org/10.3390/antibiotics9100690
APA StylePouwels, K. B., Roope, L. S. J., Buchanan, J., Morrell, L., Tonkin-Crine, S., Peters, M., Jones, L. F., Castro-Sánchez, E., Crook, D. W., Peto, T., Butler, C. C., Robotham, J. V., Walker, A. S., & Wordsworth, S. (2020). Awareness of Appropriate Antibiotic Use in Primary Care for Influenza-Like Illness: Evidence of Improvement from UK Population-Based Surveys. Antibiotics, 9(10), 690. https://doi.org/10.3390/antibiotics9100690







