Dissecting the Antimicrobial Composition of Honey
Abstract
1. Introduction
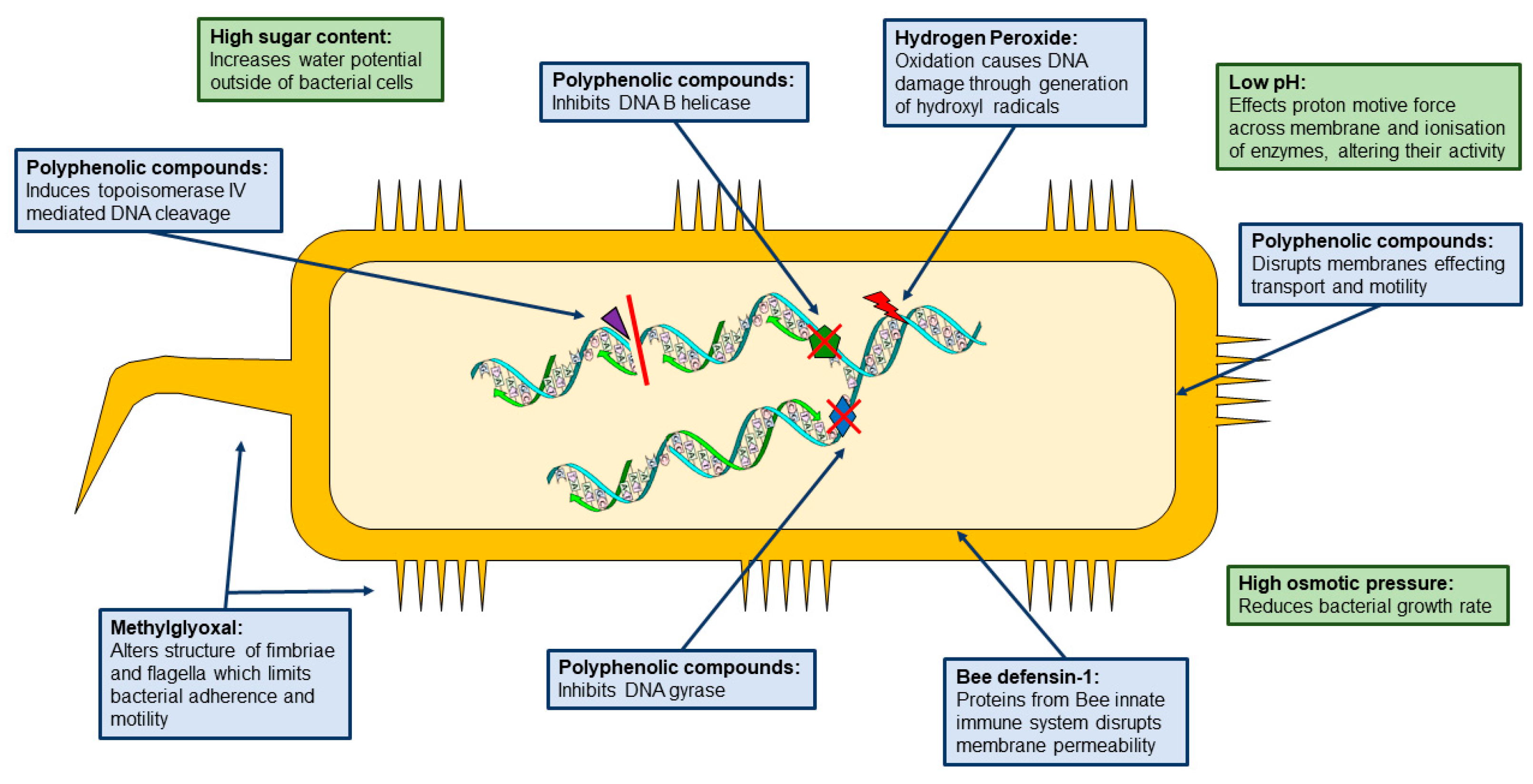
2. Composition and Classification
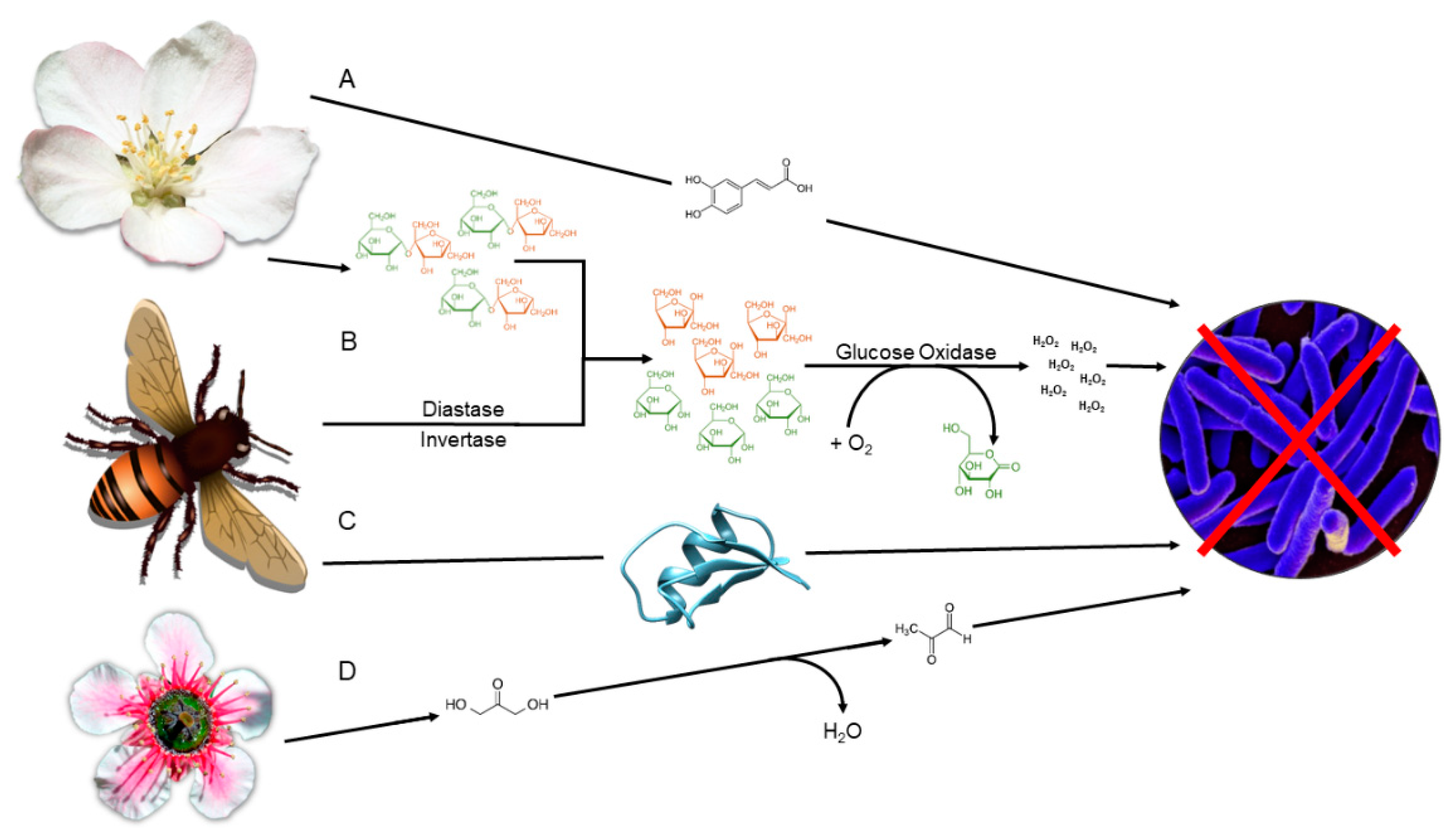
3. Carbohydrates
4. Polyphenolic Compounds:
5. Hydrogen Peroxide
6. 1,2-dicarbonyls
7. Bee defensin-1
8. Antibiotic Residue
9. Antibiofilm Properties
10. Honey and Antibiotic Synergy
11. Honey in Medical Settings
12. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Cokcetin, N.N.; Pappalardo, M.; Campbell, L.T.; Brooks, P.; Carter, D.A.; Blair, S.E.; Harry, E.J. The antibacterial activity of Australian Leptospermum honey correlates with methylglyoxal levels. PLoS ONE 2016, 11, e0167780. [Google Scholar] [CrossRef] [PubMed]
- Albaridi, N.A. Antibacterial Potency of Honey. Int. J. Microbiol. 2019, 2019, 10. [Google Scholar] [CrossRef] [PubMed]
- Saranraj, P.; Sivasakthi, S. Comprehensive Review on Honey: Biochemical and Medicinal Properties. J. Acad. Ind. Res. 2018, 6, 165–181. [Google Scholar]
- Davies, J.; Davies, D. Origins and Evolution of Antibiotic Resistance. Microbiol. Mol. Biol. Rev. 2010, 74, 417–433. [Google Scholar] [CrossRef]
- Simpkin, V.L.; Renwick, M.J.; Kelly, R.; Mossialos, E. Incentivising innovation in antibiotic drug discovery and development: Progress, challenges and next steps. J. Antibiot. 2017, 70, 1087–1096. [Google Scholar] [CrossRef]
- McLoone, P.; Warnock, M.; Fyfe, L. Honey: A realistic antimicrobial for disorders of the skin. J. Microbiol. Immunol. Infect. 2016, 49, 161–167. [Google Scholar] [CrossRef]
- Eteraf-Oskouei, T.; Najafi, M. Traditional and modern uses of natural honey in human diseases: A review. Iran. J. Basic Med. Sci. 2013, 16, 731–742. [Google Scholar]
- Dustmann, J.H. Antibacterial Effect of Honey. Apiacta 1979, 1, 1–4. [Google Scholar]
- Laallam, H.; Boughediri, L.; Bissati, S.; Menasria, T.; Mouzaoui, M.S.; Hadjadj, S.; Hammoudi, R.; Chenchouni, H. Modeling the synergistic antibacterial effects of honey characteristics of different botanical origins from the Sahara Desert of Algeria. Front. Microbiol. 2015, 6, 1–12. [Google Scholar] [CrossRef]
- El Sohaimy, S.A.; Masry, S.H.D.; Shehata, M.G. Physicochemical characteristics of honey from different origins. Ann. Agric. Sci. 2015, 60, 279–287. [Google Scholar] [CrossRef]
- Sherlock, O.; Dolan, A.; Athman, R.; Power, A.; Gethin, G.; Cowman, S.; Humphreys, H. Comparison of the antimicrobial activity of Ulmo honey from Chile and Manuka honey against methicillin-resistant Staphylococcus aureus, Escherichia coli and Pseudomonas aeruginosa. BMC Complement. Altern. Med. 2010, 10, 47. [Google Scholar] [CrossRef] [PubMed]
- Kumar, N.D.; Kalluru, R.S.; Ahmed, S.; Abhilashini, A.; Jayaprakash, T.; Garlapati, R.; Sowmya, B.; Reddy, K.N. Comparison of the antibacterial efficacy of manuka honey against E.faecalis and E.coli—An in vitro study. J. Clin. Diagn. Res. 2014, 8, 48–50. [Google Scholar] [CrossRef] [PubMed]
- Deng, J.; Liu, R.; Lu, Q.; Hao, P.; Xu, A.; Zhang, J.; Tan, J. Biochemical properties, antibacterial and cellular antioxidant activities of buckwheat honey in comparison to manuka honey. Food Chem. 2018, 252, 243–249. [Google Scholar] [CrossRef] [PubMed]
- Alvarez-suarez, J.M.; Tulipani, S.; Díaz, D.; Estevez, Y.; Romandini, S.; Giampieri, F.; Damiani, E.; Astolfi, P.; Bompadre, S.; Battino, M. Antioxidant and antimicrobial capacity of several monofloral Cuban honeys and their correlation with color, polyphenol content and other chemical compounds. Food Chem. Toxicol. 2010, 48, 2490–2499. [Google Scholar] [CrossRef] [PubMed]
- Matzen, R.D.; Leth-Espensen, J.Z.; Jansson, T.; Nielsen, D.S.; Lund, M.N.; Matzen, S. The Antibacterial Effect in Vitro of Honey Derived from Various Danish Flora. Dermatol. Res. Pract. 2018, 2018, 10. [Google Scholar] [CrossRef]
- Grecka, K.; Kús, P.M.; Worobo, R.W.; Szweda, P. Study of the anti-staphylococcal potential of honeys produced in Northern Poland. Molecules 2018, 23, 260. [Google Scholar] [CrossRef]
- Escuredo, O.; Silva, L.R.; Valentão, P.; Seijo, M.C.; Andrade, P.B. Assessing Rubus honey value: Pollen and phenolic compounds content and antibacterial capacity. Food Chem. 2012, 130, 671–678. [Google Scholar] [CrossRef]
- Isla, M.I.; Craig, A.; Ordoñez, R.; Zampini, C.; Sayago, J.; Bedascarrasbure, E.; Alvarez, A.; Salomón, V.; Maldonado, L. Physico chemical and bioactive properties of honeys from Northwestern Argentina. LWT Food Sci. Technol. 2011, 44, 1922–1930. [Google Scholar] [CrossRef]
- Fyfe, L.; Okoro, P.; Paterson, E.; Coyle, S.; Mcdougall, G.J. Compositional analysis of Scottish honeys with antimicrobial activity against antibiotic-resistant bacteria reveals novel antimicrobial components. LWT Food Sci. Technol. 2017, 79, 52–59. [Google Scholar] [CrossRef]
- Mohapatra, D.P.; Thakur, V.; Brar, S.K. Antibacterial Efficacy of Raw and Processed Honey. Biotechnol. Res. Int. 2011, 2011, 1–6. [Google Scholar] [CrossRef]
- El-Borai, A.; Yousasef, G.A.; Ghareeb, D.A.; Abdel-Tawab, M.M. Antibacterial and Antioxidant Activities of Different Varieties of Locally Produced Egyptian Honey. Egypt. J. Bot. 2018, 58, 97–107. [Google Scholar]
- Girma, A.; Seo, W.; She, R.C. Antibacterial activity of varying UMF-graded Manuka honeys. PLoS ONE 2019, 14, e0224495. [Google Scholar] [CrossRef] [PubMed]
- Cokcetin, N.; Williams, S.; Blair, S.; Carter, D.; Brooks, P.; Harry, L. Active Australian Leptospermum Honey: New Sources and Their Bioactivity; Agrifutures Australia: Canberra, Australia, 2019. [Google Scholar]
- Brudzynski, K. Effect of hydrogen peroxide on antibacterial activities of Canadian honeys. Can. J. Microbiol. 2006, 52, 1228–1237. [Google Scholar] [CrossRef]
- Bucekova, M.; Jardekova, L.; Juricova, V.; Bugarova, V.; Di Marco, G.; Gismondi, A.; Leonardi, D.; Farkasovska, J.; Godocikova, J.; Laho, M.; et al. Antibacertial Activity of Different Blossom Honeys: New Findings. Molecules 2019, 24, 1573. [Google Scholar] [CrossRef]
- Hegazi, A.G. Antimicrobial Activity of Different Egyptian honeys as comparison of Saudi Arabia honey. Res. J. Microbiol. 2011, 6, 488–495. [Google Scholar] [CrossRef][Green Version]
- John-isa, J.F.; Adebolu, T.T.; Oyetayo, V.O. Antibacterial Effects of Honey in Nigeria on Selected Diarrhoeagenic Bacteria. South Asian J. Res. Microbiol. 2019, 3, 1–11. [Google Scholar] [CrossRef]
- Maddocks, S.E.; Jenkins, R.E. Honey: A sweet solution to the growing problem of antimicrobial resistance? Future Microbiol. 2013, 8, 1419–1429. [Google Scholar] [CrossRef]
- Jenkins, R.E.; Cooper, R. Synergy between oxacillin and manuka honey Sensitizes methicillin-resistant Staphylococcus aureus to oxacillin. J. Antimicrob. Chemother. 2012, 67, 1405–1407. [Google Scholar] [CrossRef]
- Molan, P.C. The Antibacterial Activity of Honey. Bee World 1992, 73, 5–27. [Google Scholar] [CrossRef]
- Mavric, E.; Wittmann, S.; Barth, G.; Henle, T. Identification and quantification of methylglyoxal as the dominant antibacterial constituent of Manuka (Leptospermum scoparium) honeys from New Zealand. Mol. Nutr. Food Res. 2008, 52, 483–489. [Google Scholar] [CrossRef]
- Vasić, V.; Đurđić, S.; Tosti, T.; Radoičić, A.; Lušić, D.; Milojković-Opsenica, D.; Tešić, Ž.; Trifković, J. Two aspects of honeydew honey authenticity: Application of advance analytical methods and chemometrics. Food Chem. 2020, 305, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Manivanan, P.; Rajagopalan, S.M.; Subbarayalu, M. Studies on authentication of true source of honey using pollen DNA barcoding. J. Entomol. Zool. Stud. 2018, 6, 255–261. [Google Scholar]
- Silici, S.; Sagdic, O.; Ekici, L. Total phenolic content, antiradical, antioxidant and antimicrobial activities of Rhododendron honeys. Food Chem. 2010, 121, 238–243. [Google Scholar] [CrossRef]
- Pita-Calvo, C.; Vázquez, M. Differences between honeydew and blossom honeys: A review. Trends Food Sci. Technol. 2017, 59, 79–87. [Google Scholar] [CrossRef]
- Almasaudi, S.B.; Al-Nahari, A.A.; El Sayed, M.; Barbour, E.; Al Muhayawi, S.M.; Al-Jaouni, S.; Azhar, E.; Qari, M.; Qari, Y.A.; Harakeh, S. Antimicrobial effect of different types of honey on Staphylococcus aureus. Saudi J. Biol. Sci. 2017, 24, 1255–1261. [Google Scholar] [CrossRef]
- Chirife, J.; Zamora, M.C.; Motto, A. The correlation between water activity and % moisture in honey: Fundamental aspects and application to Argentine honeys. J. Food Eng. 2006, 72, 287–292. [Google Scholar] [CrossRef]
- Wahdan, H.A.L. Causes of the antimicrobial activity of honey. Infection 1998, 26, 26–31. [Google Scholar] [CrossRef]
- Brady, N.F.; Molan, P.C.; Harfoot, C.G. The sensitivity of dermatophytes to the antimicrobial activity of manuka honey and other honey. Pharm. Sci. 1996, 2, 471–473. [Google Scholar]
- Cianciosi, D.; Forbes-Hernández, T.Y.; Afrin, S.; Gasparrini, M.; Reboredo-Rodriguez, P.; Manna, P.P.; Zhang, J.; Bravo Lamas, L.; Martinez Florez, S.; Agudo Toyos, P.; et al. Phenolic compounds in honey and their associated health benefits: A review. Molecules 2018, 23, 2322. [Google Scholar] [CrossRef]
- Güneş, M.E.; Şahin, S.; Demir, C.; Borum, E.; Tosunoğlu, A. Determination of phenolic compounds profile in chestnut and floral honeys and their antioxidant and antimicrobial activities. J. Food Biochem. 2017, 41, 1–12. [Google Scholar] [CrossRef]
- Bucekova, M.; Buriova, M.; Pekarik, L.; Majtan, V.; Majtan, J. Phytochemicals-mediated production of hydrogen peroxide is crucial for high antibacterial activity of honeydew honey. Sci. Rep. 2018, 8, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Jibril, F.I.; Hilmi, A.B.M.; Manivannan, L. Isolation and characterization of polyphenols in natural honey for the treatment of human diseases. Bull. Natl. Res. Cent. 2019, 43, 4. [Google Scholar] [CrossRef]
- Estevinho, L.; Pereira, A.P.; Moreira, L.; Dias, L.G.; Pereira, E. Antioxidant and antimicrobial effects of phenolic compounds extracts of Northeast Portugal honey. Food Chem. Toxicol. 2008, 46, 3774–3779. [Google Scholar] [CrossRef] [PubMed]
- Das, A.; Datta, S.; Mukherjee, S.; Bose, S.; Ghosh, S.; Dhar, P. Evaluation of antioxidative, antibacterial and probiotic growth stimulatory activities of Sesamum indicum honey containing phenolic compounds and lignans. LWT Food Sci. Technol. 2015, 61, 244–250. [Google Scholar] [CrossRef]
- Arakawa, H.A.; Meeda, M.M.; Okubo, S.O.; Shimamura, T.S. Role of Hydrogen Peroxide in Bactericidal Action of Catechin. Biol. Pharm. Bull. 2004, 27, 277–281. [Google Scholar] [CrossRef]
- Collins, W.; Lowen, N.; Blake, D.J. Caffeic Acid Esters Are Effective Bactericidal Compounds Against Paenibacillus larvae by Altering Intracellular Oxidant and Antioxidant Levels. Biomolecules 2019, 9, 312. [Google Scholar] [CrossRef]
- Górniak, I.; Bartoszewski, R.; Króliczewski, J. Comprehensive review of antimicrobial activities of plant flavonoids. Phytochem. Rev. 2019, 18, 241–272. [Google Scholar] [CrossRef]
- Lou, Z.; Wang, H.; Zhu, S.; Ma, C.; Wang, Z. Antibacterial activity and mechanism of action of chlorogenic acid. J. Food Sci. 2011, 76, M398–M403. [Google Scholar] [CrossRef]
- Eumkeb, G.; Siriwong, S.; Phitaktim, S.; Rojtinnakorn, N.; Sakdarat, S. Synergistic activity and mode of action of flavonoids isolated from smaller galangal and amoxicillin combinations against amoxicillin-resistant Escherichia coli. J. Appl. Microbiol. 2012, 112, 55–64. [Google Scholar] [CrossRef]
- Verdrengh, M.; Collins, L.V.; Bergin, P.; Tarkowski, A. Phytoestrogen genistein as an anti-staphylococcal agent. Microbes Infect. 2004, 6, 86–92. [Google Scholar] [CrossRef]
- Shi, C.; Zhang, X.; Sun, Y.; Yang, M.; Song, K.; Zheng, Z.; Chen, Y.; Liu, X.; Jia, Z.; Dong, R. Antimicrobial Activity of Ferulic Acid Against Cronobacter sakazakii and Possible Mechanism of Action. Foodborne Pathog. Dis. 2016, 13, 196–204. [Google Scholar] [CrossRef] [PubMed]
- Borges, A.; Ferreira, C.; Saavedra, M.J.; Simões, M. Antibacterial activity and mode of action of ferulic and gallic acids against pathogenic bacteria. Microb. Drug Resist. 2013, 19, 256–265. [Google Scholar] [CrossRef] [PubMed]
- Lou, Z.; Wang, H.; Rao, S.; Sun, J.; Ma, C.; Li, J. P-Coumaric acid kills bacteria through dual damage mechanisms. Food Control 2012, 25, 550–554. [Google Scholar] [CrossRef]
- Griep, M.A.; Blood, S.; Larson, M.A.; Koepsell, S.A.; Hinrichs, S.H. Myricetin inhibits Escherichia coli DnaB helicase but not primase. Bioorganic Med. Chem. 2007, 15, 7203–7208. [Google Scholar] [CrossRef]
- Shi, C.; Sun, Y.; Zheng, Z.; Zhang, X.; Song, K.; Jia, Z.; Chen, Y.; Yang, M.; Liu, X.; Dong, R.; et al. Antimicrobial activity of syringic acid against Cronobacter sakazakii and its effect on cell membrane. Food Chem. 2016, 197, 100–106. [Google Scholar] [CrossRef]
- Mirzoeva, O.K.; Grishanin, R.N.; Calder, P.C. Antimicrobial action of propolis and some of its components: The effects on growth, membrane potential and motility of bacteria. Microbiol. Res. 1997, 152, 239–246. [Google Scholar] [CrossRef]
- Bernard, F.X.; Sable, S.; Cameron, B.; Provost, J.; Desnottes, J.F.; Crouzet, J.; Blanche, F. Glycosylated flavones as selective inhibitors of topoisomerase IV. Antimicrob. Agents Chemother. 1997, 41, 992–998. [Google Scholar] [CrossRef]
- Ball, D.W. The chemical composition of honey. J. Chem. Educ. 2007, 84, 1643–1646. [Google Scholar] [CrossRef]
- Brudzynski, K.; Abubaker, K.; St-Martin, L.; Castle, A. Re-examining the role of hydrogen peroxide in bacteriostatic and bactericidal activities of honey. Front. Microbiol. 2011, 2, 1–9. [Google Scholar] [CrossRef]
- Brudzynski, K.; Miotto, D.; Kim, L.; Sjaarda, C.; Maldonado-Alvarez, L.; Fukś, H. Active macromolecules of honey form colloidal particles essential for honey antibacterial activity and hydrogen peroxide production. Sci. Rep. 2017, 7, 1–15. [Google Scholar] [CrossRef]
- Taormina, P.J.; Niemira, B.A.; Beuchat, L.R. Inhibitory activity of honey against foodborne pathogens as influenced by the presence of hydrogen peroxide and level of antioxidant power. Int. J. Food Microbiol. 2001, 69, 217–225. [Google Scholar] [CrossRef]
- Weston, R.J. The contribution of catalase and other natural products to the antibacterial activity of honey: A review. Food Chem. 2000, 71, 235–239. [Google Scholar] [CrossRef]
- Finnegan, M.; Linley, E.; Denyer, S.P.; McDonnell, G.; Simons, C.; Maillard, J.Y. Mode of action of hydrogen peroxide and other oxidizing agents: Differences between liquid and gas forms. J. Antimicrob. Chemother. 2010, 65, 2108–2115. [Google Scholar] [CrossRef]
- Brudzynski, K.; Abubaker, K.; Wang, T. Powerful bacterial killing by buckwheat honeys is concentration-dependent, involves complete DNA degradation and requires hydrogen peroxide. Front. Microbiol. 2012, 3, 1–9. [Google Scholar] [CrossRef]
- Brudzynski, K.; Lannigan, R. Mechanism of honey bacteriostatic action against MRSA and VRE involves hydroxyl radicals generated from honey’s hydrogen peroxide. Front. Microbiol. 2012, 3, 1–8. [Google Scholar] [CrossRef]
- Brudzynski, K.; Abubaker, K.; Miotto, D. Unraveling a mechanism of honey antibacterial action: Polyphenol/H 2O 2-induced oxidative effect on bacterial cell growth and on DNA degradation. Food Chem. 2012, 133, 329–336. [Google Scholar] [CrossRef]
- Degen, J.; Hellwig, M.; Henle, T. 1,2-Dicarbonyl compounds in commonly consumed foods. J. Agric. Food Chem. 2012, 60, 7071–7079. [Google Scholar] [CrossRef]
- Arena, E.; Ballistreri, G.; Tomaselli, F.; Fallico, B. Survey of 1,2-Dicarbonyl Compounds in Commercial Honey of Different Floral Origin. J. Food Sci. 2011, 76, C1203–C1210. [Google Scholar] [CrossRef]
- Schalkwijk, C.G.; Posthuma, N.; Ten Brink, H.J.; Ter Wee, P.M.; Teerlink, T. Induction of 1,2-dicarbonyl compounds, intermediates in the formation of advanced glycation end-products, during heat-sterilization of glucose-based peritoneal dialysis fluids. Perit. Dial. Int. 1999, 19, 325–333. [Google Scholar]
- Roberts, A.; Jenkins, R.; Brown, H.L. On the antibacterial effects of manuka honey: Mechanistic insights. Res. Rep. Biol. 2015, 6, 215. [Google Scholar]
- Johnston, M.; Mcbride, M.; Dahiya, D.; Owusu-apenten, R. Antibacterial activity of Manuka honey and its components: An overview. AIMS Microbiol. 2018, 4, 655–664. [Google Scholar] [CrossRef] [PubMed]
- Adams, C.J.; Manley-Harris, M.; Molan, P.C. The origin of methylglyoxal in New Zealand manuka (Leptospermum scoparium) honey. Carbohydr. Res. 2009, 344, 1050–1053. [Google Scholar] [CrossRef] [PubMed]
- Majtan, J.; Klaudiny, J.; Bohova, J.; Kohutova, L.; Dzurova, M.; Sediva, M.; Bartosova, M.; Majtan, V. Methylglyoxal-induced modifications of significant honeybee proteinous components in manuka honey: Possible therapeutic implications. Fitoterapia 2012, 83, 671–677. [Google Scholar] [CrossRef] [PubMed]
- Grainger, M.N.C.; Manley-harris, M.; Lane, J.R.; Field, R.J. Kinetics of the conversion of dihydroxyacetone to methylglyoxal in New Zealand manuka honey: Part II—Model systems. Food Chem. 2016, 202, 492–499. [Google Scholar] [CrossRef] [PubMed]
- Rabie, E.; Serem, J.C.; Oberholzer, H.M.; Gaspar, A.R.M.; Bester, M.J. How methylgyloxal kills bacteria: An ultrastructural study. Ultrastruct. Pathol. 2016, 40, 107–111. [Google Scholar] [CrossRef]
- Ilyasov, R.A.; Gaifullina, L.R.; Saltykova, E.S.; Poskryakov, A.V.; Nikolenko, A.G. Review of the expression of antimicrobial peptide defensin in honey bees Apis mellifera L. J. Apic. Sci. 2012, 56, 115–124. [Google Scholar] [CrossRef]
- Oryan, A.; Alemzadeh, E.; Moshiri, A. Biological properties and therapeutic activities of honey in wound healing: A narrative review and meta-analysis. J. Tissue Viability 2016, 25, 98–118. [Google Scholar] [CrossRef]
- Kwakman, P.H.S.; Zaat, S.A.J. Antibacterial components of honey. IUBMB Life 2012, 64, 48–55. [Google Scholar] [CrossRef]
- Kwakman, P.H.S.; te Velde, A.A.; de Boer, L.; Vandenbroucke-Grauls, C.M.J.E.; Zaat, S.A.J. Two major medicinal honeys have different mechanisms of bactericidal activity. PLoS ONE 2011, 6, e17709. [Google Scholar] [CrossRef]
- Ganz, T. Defensins: Antimicrobial peptides of innate immunity. Nat. Rev. Immunol. 2003, 3, 710–720. [Google Scholar] [CrossRef]
- Bucekova, M.; Sojka, M.; Valachova, I.; Martinotti, S.; Ranzato, E.; Szep, Z.; Majtan, V.; Klaudiny, J.; Majtan, J. Bee-derived antibacterial peptide, defensin-1, promotes wound re-epithelialisation in vitro and in vivo. Sci. Rep. 2017, 7, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Bargańska, Ż.; Ślebioda, M.; Namieśnik, J. Determination of antibiotic residues in honey. Trends Anal. Chem. 2011, 30, 1035–1041. [Google Scholar] [CrossRef]
- Al-waili, N.; Salom, K.; Al-ghamdi, A.; Ansari, M.J. Antibiotic, Pesticide, and Microbial Contaminants of Honey: Human Health Hazards. Sci. World J. 2012, 2012, 9. [Google Scholar] [CrossRef] [PubMed]
- Mcewen, S.A.; Fedorka, P.J. Antimicrobial Use and Resistance in Animals. Clin. Infect. Dis. 2002, 34, 93–106. [Google Scholar] [CrossRef] [PubMed]
- Chowdhury, S.; Hassan, M.M.; Alam, M.; Sattar, S.; Bari, M.S.; Saifuddin, A.K.M.; Hoque, M.A. Antibiotic residues in milk and eggs of commercial and local farms at Chittagong, Bangladesh. Vet. World 2015, 8, 467–471. [Google Scholar] [CrossRef]
- Ramatla, T.; Ngoma, L.; Adetunji, M.; Mwanza, M. Evaluation of antibiotic residues in raw meat using different analytical methods. Antibiotics 2017, 6, 34. [Google Scholar] [CrossRef]
- Adams, S.J.; Fussell, R.J.; Dickinson, M.; Wilkins, S.; Sharman, M. Study of the depletion of lincomycin residues in honey extracted from treated honeybee (Apis mellifera L.) colonies and the effect of the shook swarm procedure. Anal. Chim. Acta 2009, 637, 315–320. [Google Scholar] [CrossRef]
- López, D.; Vlamakis, H.; Kolter, R. Biofilms. Cold Spring Harb. Lab. Prospect. Biol. 2010, 2, 1–12. [Google Scholar] [CrossRef]
- Stewart, P.S.; Costerton, J.W. Antibiotic resistance of bacterial biofilms. Lancet 2001, 358, 135–138. [Google Scholar] [CrossRef]
- Minden-Birkenmaier, B.A.; Bowlin, G.L. Honey-based templates in wound healing and tissue engineering. Bioengineering 2018, 5, 46. [Google Scholar] [CrossRef]
- Alandejani, T.; Marsan, J.; Ferris, W.; Slinger, R.; Chan, F. Effectiveness of honey on Staphylococcus aureus and Pseudomonas aeruginosa biofilms. Otolaryngol. Head Neck Surg. 2009, 141, 114–118. [Google Scholar] [CrossRef] [PubMed]
- Maddocks, S.E.; Lopez, M.S.; Rowlands, R.S.; Cooper, R.A. Manuka honey inhibits the development of Streptococcus pyogenes biofilms and causes reduced expression of two fibronectin binding proteins. Microbiology 2012, 158, 781–790. [Google Scholar] [CrossRef] [PubMed]
- Sojka, M.; Valachova, I.; Bucekova, M.; Majtan, J. Antibiofilm efficacy of honey and bee-derived defensin-1 on multispecies wound biofilm. J. Med. Microbiol. 2016, 65, 337–344. [Google Scholar] [CrossRef] [PubMed]
- Lu, J.; Turnbull, L.; Burke, C.M.; Liu, M.; Carter, D.A.; Schlothauer, R.C.; Whitchurch, C.B.; Harry, E.J. Manuka-type honeys can eradicate biofilms produced by Staphylococcus aureus strains with different biofilm-forming abilities. PeerJ 2014, 2014, 1–25. [Google Scholar]
- Majtan, J.; Bohova, J.; Horniackova, M.; Klaudiny, J.; Majtan, V. Anti-biofilm effects of honey against wound pathogens proteus mirabilis and enterobacter cloacae. Phyther. Res. 2014, 28, 69–75. [Google Scholar] [CrossRef]
- Steinberg, N.; Kolodkin-Gal, I. The matrix reloaded: How sensing the extracellular matrix synchronizes bacterial communities. J. Bacteriol. 2015, 197, 2092–2103. [Google Scholar] [CrossRef]
- Jenkins, R.; Cooper, R. Improving Antibiotic Activity against Wound Pathogens with Manuka Honey In Vitro. PLoS ONE 2012, 7, e45600. [Google Scholar] [CrossRef]
- Müller, P.; Alber, D.G.; Turnbull, L.; Schlothauer, R.C.; Carter, D.A.; Whitchurch, C.B.; Harry, E.J. Synergism between Medihoney and Rifampicin against Methicillin-Resistant Staphylococcus aureus (MRSA). PLoS ONE 2013, 8, e57679. [Google Scholar] [CrossRef]
- Campeau, M.E.M.; Patel, R. Antibiofilm Activity of Manuka Honey in Combination with Antibiotics. Int. J. Bacteriol. 2014, 2014, 1–7. [Google Scholar] [CrossRef]
- Oliveira, A.; Ribeiro, H.G.; Silva, A.C.; Silva, M.D.; Sousa, J.C.; Rodrigues, C.F.; Melo, L.D.; Henriques, A.F.; Sillankorva, S. Synergistic antimicrobial interaction between honey and phage against Escherichia coli biofilms. Front. Microbiol. 2017, 8, 1–18. [Google Scholar] [CrossRef]
- Kwakman, P.H.S.; Velde, A.A.; Boer, L.; Speijer, D.; Vandenbrouke-Grauls, C.M.J.E.; Zaat, S.A.J. How honey kills bacteria. FASEB J. 2010, 24, 2576–2582. [Google Scholar] [CrossRef] [PubMed]
- Carter, D.A.; Blair, S.E.; Cokcetin, N.N.; Bouzo, D.; Brooks, P.; Schothauer, R.; Harry, E.J. Therapeutic manuka honey: No longer so alternative. Front. Microbiol. 2016, 7, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Molan, P.C. Re-introducing honey in the management of wounds and ulcers—Theory and practice. Ostomy Wound. Manag. 2002, 48, 28–40. [Google Scholar] [PubMed]
- Samarghandian, S.; Farkhondeh, T.; Samini, F. Honey and health: A review of recent clinical research. Pharmacogn. Res. 2017, 9, 121–127. [Google Scholar]
- Molan, P.C. The evidence supporting the use of honey as a wound dressing. Int. J. Low. Extrem. Wounds 2006, 5, 40–54. [Google Scholar] [CrossRef] [PubMed]
- Dunford, C.; Cooper, R.; Molan, P.; White, R. The use of honey in wound treatment. Nurs. Stand. 2000, 15, 63–68. [Google Scholar] [CrossRef] [PubMed]
- Subrahmanyam, M. Honey Dressing Accelerates Split-Thickness Skin Graft Donor Site Healing. Indian J. Surg. 2015, 77, 261–263. [Google Scholar] [CrossRef]
- Mashhood, A.A.; Khan, T.A.; Sami, A.N. Honey compared with 1% silver sulfadiazine cream in the treatment of superficial and partial thickness burns. J. Pak. Assoc. Dermatol. 2006, 16, 14–19. [Google Scholar]
| Geographical Variation in Honeys Antimicrobial Activity | ||
|---|---|---|
| Country of Origin | Honey Sample | Organisms |
| Australia | ||
| New Zealand [13] | Manuka | Staphylococcus aureus, Pseudomonas aeruginosa |
| New Zealand [22] | Manuka | S. aureus, MRSA, MSSA, coagulase-negative Staphylococcus epidermidis, Klebsiella pneumonia, ESBL E. coli |
| Australia [23] | Leptospermum based honey | S. aureus |
| North America | ||
| Canada [24] | Canadian Honey | E. coli, Bacillus subtilis |
| Cuba [14] | Christmas vine, Morning glory, Black mangrove, Linen vine, Singing bean | S. aureus, P. aeruginosa, E. coli and B. subtilis |
| South America | ||
| Chile [11] | Ulmo Honey | MRSA, E. coli and P. aeruginosa |
| Argentina [18] | Algarrobo, citrus and multifloral honey | S. aureus, Enterococcus faecalis, E. coli, Morganella morganii and P. aeruginosa |
| Europe | ||
| Scotland [19] | Blossom, heather, Highland, Portobello Orchard | Acinetobactor calcoaceticus, S. aureus, P. aeruginosa and E. coli |
| Northwest Spain [17] | Rubus Honey | S. aureus, S. epidermidis, Micrococcus luteus, E. faecalis, B. cereus, Proteus mirabilis, E. coli, P. aeruginosa and Salmonella. typhimurium |
| Denmark [15] | Heather, Rasberry, Rapeseed, Hawthorn and White Clover | S. aureus, P. aeruginosa and E. coli |
| Slovakia [25] | Honeydew Honey | P. aeruginosa and S. aureus |
| Asia | ||
| China [13] | Buckwheat Honey | S. aureus and P. aeruginosa |
| Saudi Arabia [26] | Sider Honey | S. aureus, Streptococcus pyogenes, Corynebacteria pseudotuberculosis, K. pneumonia, P. aeruginosa and E. coli |
| Africa | ||
| Algeria [9] | Astragalus, Wall-rocket, Eucalyptus, Legume, Peach, Juniper, Buckthorn and multifloral | Clostridium perfringens, S. aureus, E. coli and B. subtilis. |
| Nigeria [27] | Wildflower and Bitter leaf Honey | S. typhimurium, Shigella dysenteriae, E. coli, B. cereus and S. aureus |
| Egypt [21] | Cotton, Blackseed, Orange, Eucalyptus, Sidr and Clover Honey | E. coli, S. aureus, Streptococcus mutans, P. mirabilis, P. aeruginosa and K. pnuemoniae |
| Egypt [26] | Acacia, Citrus, Clover, Coriander, Cotton and Palm Honey | S. aureus, S. pyogenes, Corynebacteria pseudotuberculosis, K. pneumonia, P. aeruginosa and E. coli |
| Phenolic Acids | Mechanism | Flavonoids | Mechanism |
|---|---|---|---|
| 2-cis,A-trans Abscisic acid | Unknown | Apigenin | Inhibits DNA gyrase [44] |
| 2-Hydroxycinnamic acid | Unknown | Catechin | Hydrogen peroxide generation [45] |
| Caffeic acid | Oxidative Stress [46] | Chrysin | Inhibits DNA gyrase [47] |
| Chlorogenic acid | Increase in membrane permeability resulting in cytoplasmic and nucleotide leakage [48] | Galangin | Inhibition of peptidoglycan and ribosome synthesis [49] |
| Cinnamic acid | Unknown | Genistein | Disruption to topoisomerase-II DNA cleavage complex [50] |
| Ellagic acid | Unknown | Isorhamnetin | Unknown |
| Ferulic acid | Cell membrane dysfunction and changes in cell morphology [51] | Kaempferol | Inhibits DNA gyrase [47] |
| Gallic acid | Cell membrane disruption resulting in pore formation and intracellular leakage [52] | Luteolin | Inhibits FAS-I in Mycobacteria and inhibits DNA helicase DnaB and RecBCD [47] |
| p-Coumaric acid | Cell membrane disruption and binding to bacterial DNA [53] | Myricetin | Inhibits DNA B helicase [54] |
| p-Hydroxybenzoic acid | Unknown | Naringenin | Unknown |
| Protocatechuic acid | Unknown | Pinobanksin | Unknown |
| Sinapic acid | Unknown | Pinocembrin | Induces cell lysis [47] |
| Syringic acid | Cell membrane dysfunction [55] | Quercetin | Disrupts membranes, transport and motility [56] |
| Vannilic acid | Unknown | Rutin | Induces topoisomerase IV mediated DNA cleavage [57] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nolan, V.C.; Harrison, J.; Cox, J.A.G. Dissecting the Antimicrobial Composition of Honey. Antibiotics 2019, 8, 251. https://doi.org/10.3390/antibiotics8040251
Nolan VC, Harrison J, Cox JAG. Dissecting the Antimicrobial Composition of Honey. Antibiotics. 2019; 8(4):251. https://doi.org/10.3390/antibiotics8040251
Chicago/Turabian StyleNolan, Victoria C., James Harrison, and Jonathan A. G. Cox. 2019. "Dissecting the Antimicrobial Composition of Honey" Antibiotics 8, no. 4: 251. https://doi.org/10.3390/antibiotics8040251
APA StyleNolan, V. C., Harrison, J., & Cox, J. A. G. (2019). Dissecting the Antimicrobial Composition of Honey. Antibiotics, 8(4), 251. https://doi.org/10.3390/antibiotics8040251






