Antibiotic Prescribing by Informal Healthcare Providers for Common Illnesses: A Repeated Cross-Sectional Study in Rural India
Abstract
1. Introduction
2. Results
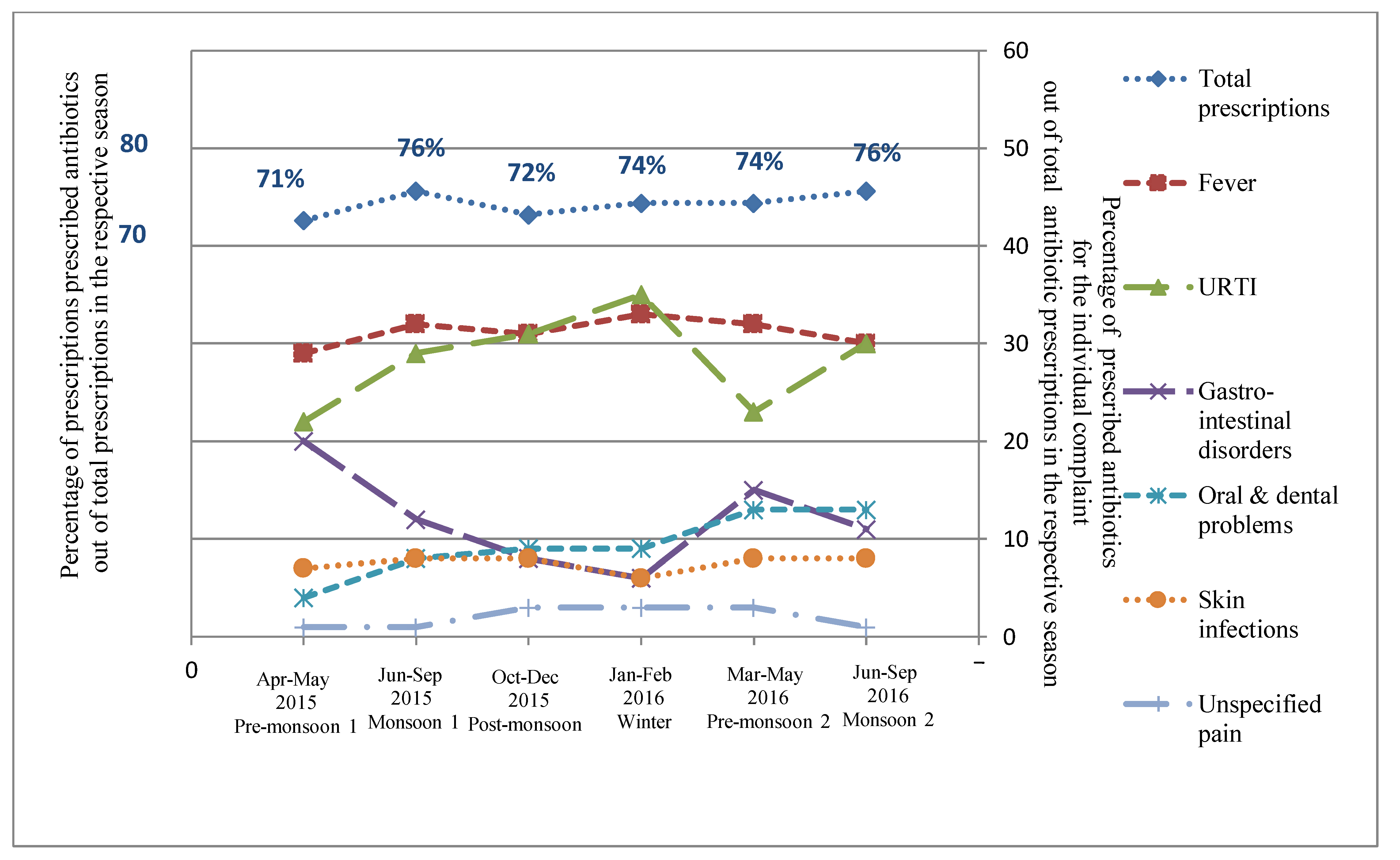
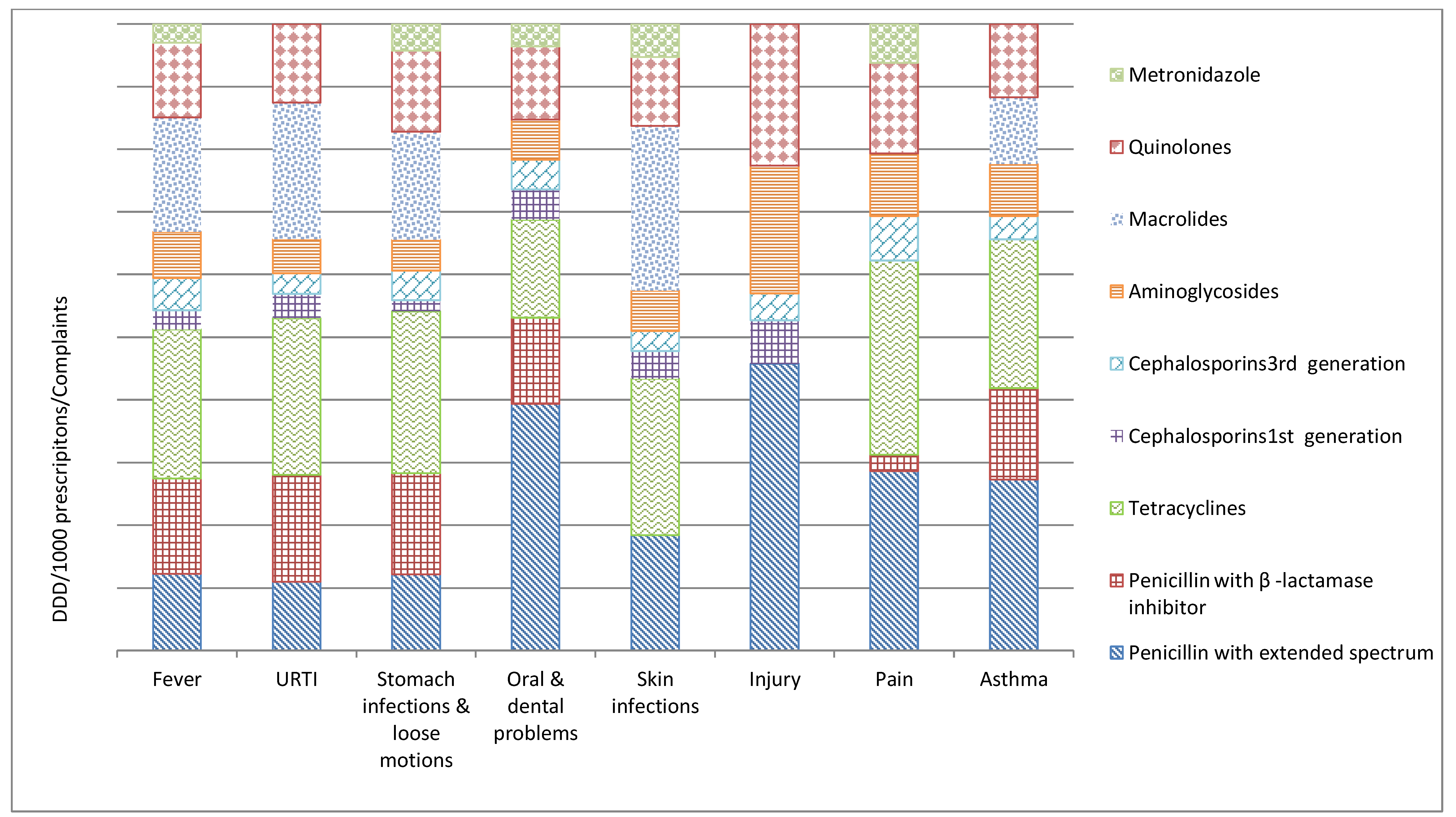
2.1. Antibiotic Prescribing for Presenting Complaints
2.1.1. Fever
2.1.2. Upper Respiratory Tract Infection (URTI)
2.1.3. Gastro-Intestinal Disorders
2.1.4. Oral and Dental Problems
2.1.5. Skin Infections
2.1.6. Unspecified Pain
2.2. Cluster Analysis
3. Discussion
3.1. Methodological Considerations
3.2. Strengths and Limitations
- This study is the first to describe and quantify the antibiotic prescription practices of IHCPs in rural India, although with some difficulty, due to the illegal status of their practice;
- We could only collect prescription data from 12 IHCPs, as many of the non-participants were afraid to reveal information regarding their treatment practices;
- Repeated data collection, including monthly recording of a considerable number of antibiotic prescribing occasions from the same group of IHCPs, made the data reliable and this study design also enabled us to minimize the percentage of missing data (1% missing data overall);
- We assessed seasonal changes in antibiotic prescription rates, which has rarely been reported in studies from India;
- The study did not evaluate the severity of the disease.
3.3. Recommendations
4. Materials and Methods
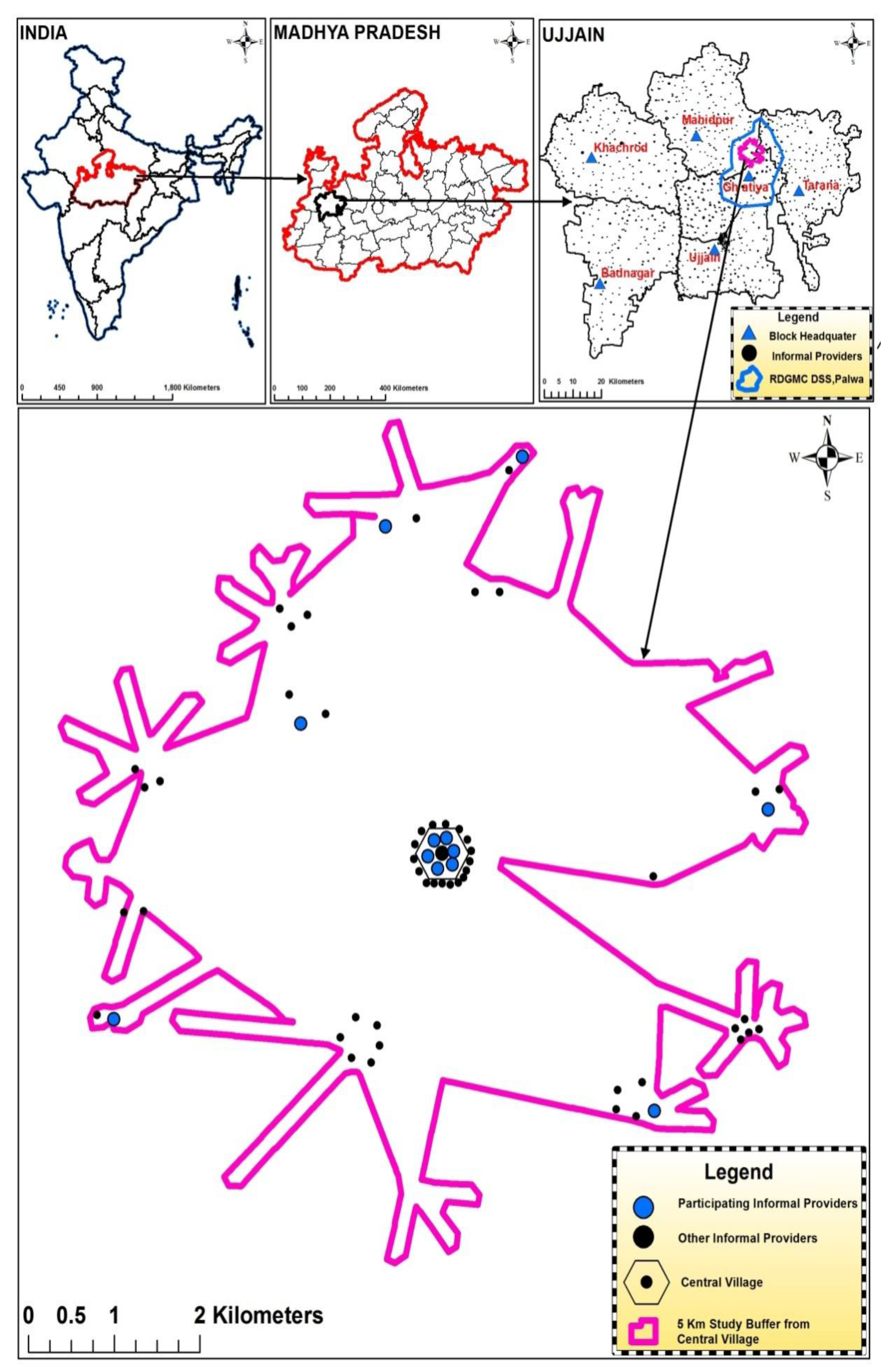
4.1. Study Design and Study Setting
4.2. Selection of Study Participants
4.3. Data Collection
4.4. Data Management
4.5. Data Analysis
4.6. Ethical Approval
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Data Availability
References
- WHO. Pre-Antibiotic Era Looming Large—The World is Almost Out of Time. 2014. Available online: http://www.searo.who.int/mediacentre/features/2014/rd-statement-amr2014/en/ (accessed on 1 July 2019).
- Penicillin, A.F. Nobel Lecture Dec 11. 1945. Available online: http://www.nobelprize.org/nobel_prizes/medicine/laureates/1945/fleming-lecture.pdf (accessed on 1 July 2019).
- WHO. Global Action Plan on Antimicrobial Ressitance. 2015. Available online: http://apps.who.int/iris/bitstream/handle/10665/193736/9789241509763_eng.pdf?sequence=1 (accessed on 1 July 2019).
- CDC. Report on “Antibiotic Resistance Threats in the United States”. 2013. Available online: http://www.cdc.gov/drugresistance/threat-report-2013/ (accessed on 1 July 2019).
- Jacob, J.T.; Muliyil, J. Public health is infrastructure for human development. Indian J. Med. Res. 2009, 130, 9–11. [Google Scholar]
- National Treatment Guidelines for Antimicrobial Use in Infectious Diseases. National Center for Disease Control, Directorate General of Health Services, Ministry of Health & Family Welfare, Government of India 2016. Available online: http://pbhealth.gov.in/AMR_guideline7001495889.pdf (accessed on 1 July 2019).
- UNICEF. 2013 Statistical Snapshot- Child Mortality. Available online: http://www.childinfo.org/files/Child_Mortality_Stat_Snapshot_e-version_Sep_17.pdf (accessed on 1 July 2019).
- Chokshi, M.; Patil, B.; Khanna, R.; Neogi, S.B.; Sharma, J.; Paul, V.K.; Zodpey, S. Health systems in India. J. Perinatol. 2016, 36, S9–S12. [Google Scholar] [CrossRef]
- de Costa, A.; Diwan, V. Where is the public health sector? Public and private sector healthcare provision in Madhya Pradesh, India. Health Policy 2007, 84, 269–276. [Google Scholar] [CrossRef]
- Sharma, A.; Ladd, E.; Unnikrishnan, M.K. Healthcare inequity and physician scarcity. Econ. Political Wkly. 2013, 48, 112–117. [Google Scholar]
- Swedish Agency for Growth Policy Analysis. Report on India’s Healthcare System—Overview and Quality Improvements, Direct Response 2013. Available online: http://www.tillvaxtanalys.se/download/18.5d9caa4d14d0347533bcf42e/1430909773826/direct_response_2013_04.pdf (accessed on 1 July 2019).
- Sudhinaraset, M.; Ingram, M.; Lofthouse, H.K.; Montagu, D. What is the role of informal healthcare providers in developing countries? A systematic review. PLoS ONE 2013, 8, 54978. [Google Scholar] [CrossRef]
- Deshpande, K.; Diwan, V.; Lönnroth, K.; Mahadik, V.K.; Chandorkar, R.K. Spatial pattern of private health care provision in Ujjain, India: A provider survey processed and analysed with a Geographical Information System. Health Policy 2004, 68, 211–222. [Google Scholar] [CrossRef]
- Gautham, M.; Binnendijk, E.; Koren, R.; Dror, D.M. ‘First we go to the small doctor’: First contact for curative health care sought by rural communities in Andhra Pradesh & Orissa, India. Indian J. Med. Res. 2011, 134, 627–638. [Google Scholar]
- Rao, M.; Rao, K.D.; Kumar, A.S.; Chatterjee, M.; Sundararaman, T. Human resources for health in India. Lancet 2011, 377, 587–598. [Google Scholar] [CrossRef]
- Singh, P.; Yadav, R.J.; Pandey, A. Utilization of indigenous systems of medicine & homoeopathy in India. Indian J. Med. Res. 2005, 122, 137–142. [Google Scholar]
- Caucci, S.; Karkman, A.; Cacace, D.; Rybicki, M.; Timpel, P.; Voolaid, V.; Gurke, R.; Virta, M.; Berendonk, T.U. Seasonality of antibiotic prescriptions for outpatients and resistance genes in sewers and wastewater treatment plant outflow. FEMS Microbiol. Ecol. 2016, 92, 060. [Google Scholar] [CrossRef]
- Sun, L.; Klein, E.Y.; Laxminarayan, R. Seasonality and temporal correlation between community antibiotic use and resistance in the United States. Clin. Infect. Dis. 2012, 55, 687–694. [Google Scholar] [CrossRef]
- Alvarez-Uria, G.; Zachariah, S.; Thomas, D. High prescription of antimicrobials in a rural district hospital in India. Pharm. Pract. (Granada) 2014, 12, 384. [Google Scholar] [CrossRef]
- Dimri, S.; Tiwari, P.; Basu, S.; Parmar, V.R. Drug use pattern in children at a teaching hospital. Indian Pediatr. 2009, 46, 165–167. [Google Scholar]
- Kumar, R.; Indira, K.; Rizvi, A.; Rizvi, T.; Jeyaseelan, L. Antibiotic prescribing practices in primary and secondary health care facilities in Uttar Pradesh, India. J. Clin. Pharm. Ther. 2008, 33, 625–634. [Google Scholar] [CrossRef]
- Kotwani, A.; Holloway, K. Trends in antibiotic use among outpatients in New Delhi, India. BMC Infect. Dis. 2011, 11, 99. [Google Scholar] [CrossRef]
- Pathak, A.; Mahadik, K.; Dhaneria, S.P.; Sharma, A.; Eriksson, B.; Lundborg, C.S. Antibiotic prescribing in outpatients: Hospital and seasonal variations in Ujjain, India. Scand. J. Infect. Dis. 2011, 43, 479–488. [Google Scholar] [CrossRef]
- Pathak, D.; Pathak, A.; Marrone, G.; Diwan, V.; Lundborg, C.S. Adherence to treatment guidelines for acute diarrhoea in children up to 12 years in Ujjain, India—A cross-sectional prescription analysis. BMC Infect. Dis. 2011, 11, 32. [Google Scholar] [CrossRef]
- Kotwani, A.; Joshi, P.C.; Jhamb, U.; Holloway, K. Prescriber and dispenser perceptions about antibiotic use in acute uncomplicated childhood diarrhea and upper respiratory tract infection in New Delhi: Qualitative study. Indian J. Pharmacol. 2017, 49, 419–431. [Google Scholar] [CrossRef]
- Costelloe, C.; Metcalfe, C.; Lovering, A.; Mant, D.; Hay, A.D. Effect of antibiotic prescribing in primary care on antimicrobial resistance in individual patients: Systematic review and meta-analysis. BMJ 2010, 340, c2096. [Google Scholar] [CrossRef]
- Shakya, P.; Barrett, P.; Diwan, V.; Marothi, Y.; Shah, H.; Chhari, N.; Tamhankar, A.J.; Pathak, A.; Lundborg, C.S. Antibiotic resistance among Escherichia coli isolates from stool samples of children aged 3 to 14 years from Ujjain, India. BMC Infect. Dis. 2013, 13, 477. [Google Scholar] [CrossRef]
- Purohit, M.; Chandran, S.; Shah, H.; Diwan, V.; Tamhankar, A.; Stålsby Lundborg, C. Antibiotic Resistance in an Indian Rural Community: A‘One-Health’ Observational Study on Commensal Coliform from Humans, Animals, and Water. Int. J. Environ. Res. Public Health 2017, 14, 386. [Google Scholar] [CrossRef]
- Das, J.; Chowdhury, A.; Hussam, R.; Banerjee, A.V. The impact of training informal health care providers in India: A randomized controlled trial. Science 2016, 354, 7384. [Google Scholar] [CrossRef]
- United Nations General Assembly—Political Declaration of the High-Level Meeting of the General Assembly on Antimicrobial Resistance. 2016. Available online: https://digitallibrary.un.org/record/842813/files/A_71_L-2-EN.pdf (accessed on 1 July 2019).
- WHO. Task Shifting-Global Recommendations and Guidelines. 2008. Available online: http://www.who.int/healthsystems/TTR-TaskShifting.pdf (accessed on 1 July 2019).
- Ministry of Skill Development and Entrepreneurship, National Skill Development Mission-Pradhan Mantri Kaushal Vikas Yojana (PMKVY) 2016–2020. 2015. Available online: https://www.msde.gov.in/nationalskillmission.html (accessed on 1 July 2019).
- UNDP. 2007–2008, MADHYA PRADESH-Economic and Human Development Indicators. Available online: https://www.undp.org/content/dam/india/docs/madhyapradesh_factsheet.pdf (accessed on 1 July 2019).
- State profile Madhya Pradesh 2011. Available online: http://www.census2011.co.in/census/state/madhya+pradesh.html (accessed on 1 July 2019).
- Lundborg, C.S.; Diwan, V.; Pathak, A.; Purohit, M.R.; Shah, H.; Sharma, M.; Mahadik, V.K.; Tamhankar, A.J. Protocol: A ‘One health’ two year follow-up, mixed methods study on antibiotic resistance, focusing children under 5 and their environment in rural India. BMC Public Health 2015, 15, 1321. [Google Scholar]
- WHO. Anatomical Therapeutic Chemical (ATC) Classification System: Guidelines for ATC Classification and DDD Assignment 2018, WHO Collaborating Centre for Drug Statistics Methodology, ATC Classification Index with DDDs, 2018. Oslo, Norway 2018. Available online: https://www.whocc.no/ddd/list_of_ddds_combined_products/ (accessed on 1 July 2019).
- Landstedt, K.; Sharma, A.; Johansson, F.; Lundborg, C.S.; Sharma, M. Antibiotic prescriptions for inpatients having non-bacterial diagnosis at medicine departments of two private sector hospitals in Madhya Pradesh, India: A cross-sectional study. BMJ Open 2017, 7, e012974. [Google Scholar] [CrossRef]
- Pathak, A.; Mahadik, K.; Dhaneria, S.P.; Sharma, A.; Eriksson, B.; Lundborg, C.S. Surveillance of antibiotic consumption using the “focus of infection” approach in 2 hospitals in Ujjain, India. PLoS ONE 2012, 7, e38641. [Google Scholar] [CrossRef]
- Sharma, M.; Damlin, A.; Pathak, A.; Lundborg, C.S. Antibiotic Prescribing among Pediatric Inpatients with Potential Infections in Two Private Sector Hospitals in Central India. PLoS ONE 2015, 10, e0142317. [Google Scholar] [CrossRef]
- Sharma, M.; Damlin, A.L.; Sharma, A.; Stålsby Lundborg, C. Antibiotic prescribing in medical intensive care units—A comparison between two private sector hospitals in Central India. Infect. Dis. 2015, 47, 302–309. [Google Scholar] [CrossRef]
- Sharma, M.; Eriksson, B.; Marrone, G.; Dhaneria, S.; Lundborg, C.S. Antibiotic prescribing in two private sector hospitals; one teaching and one non-teaching: A cross-sectional study in Ujjain, India. BMC Infect. Dis. 2012, 12, 155. [Google Scholar] [CrossRef]
- Arora, S.K.; Jha, B. Indian Meteorological Department 2018. Available online: http://imd.gov.in/section/nhac/wxfaq.pdf (accessed on 1 July 2019).
- Rohlf, F.; Sokal, R. Statistical Tables, 3rd ed.; Freeman: New York, NY, USA, 1995. [Google Scholar]
| Co-variates | Total Prescriptions N | Antibiotic Prescriptions n | Proportions of Antibiotic Prescriptions (n/N) | 95% Confidence Interval |
|---|---|---|---|---|
| 15,322 | 11,336 | 0.74 | 0.73–0.75 | |
| Age groups | ||||
| 18 years and above | 11,223 | 7996 | 0.71 | 0.70–0.72 |
| 6–17 years | 1852 | 1441 | 0.78 | 0.76–0.80 |
| 0–5 years | 2247 | 1899 | 0.85 | 0.83–0.86 |
| Gender | ||||
| Female | 6643 | 4829 | 0.73 | 0.72–0.74 |
| Male | 8679 | 6507 | 0.75 | 0.74–0.76 |
| Presenting complaints | ||||
| Fever | 4118 | 3569 | 0.87 | 0.86–0.88 |
| URTI ** | 4047 | 3273 | 0.81 | 0.79–0.82 |
| Gastro-intestinal disorders | 2107 | 1271 | 0.60 | 0.58–0.62 |
| Oral & dental problems | 1273 | 1126 | 0.88 | 0.87–0.90 |
| Skin infections | 1068 | 844 | 0.79 | 0.76–0.81 |
| Seasons | ||||
| Pre-monsoon 1 | 1329 | 938 | 0.71 | 0.68–0.73 |
| Pre-monsoon 2 | 2432 | 1811 | 0.74 | 0.73–0.76 |
| Monsoon 1 | 2869 | 2166 | 0.75 | 0.74–0.77 |
| Monsoon 2 | 3100 | 2350 | 0.76 | 0.74–0.77 |
| Post-monsoon | 3423 | 2476 | 0.72 | 0.71–0.74 |
| Winter | 2169 | 1595 | 0.74 | 0.72–0.75 |
| Variable | Number of Prescriptions | % of Total |
|---|---|---|
| Number of antibiotics per prescription | N = 11336 | |
| 1 | 7072 | 62 |
| 2 | 4046 | 36 |
| 3 and more | 218 | 2 |
| Number of fixed dose combinations present | N = 2522 | |
| Ampicillin, Cloxacillin | 1552 | 62 |
| Sulfamethoxazole, Trimethoprim (Co-trimoxazole) | 362 | 14 |
| Norfloxacin, Tinidazole | 320 | 13 |
| Others | 288 | 11 |
| Route of drug administration | N = 15472 | |
| Oral (tablets/capsules/syrup) | 10128 | 65 |
| Parenteral | 5007 | 32 |
| Others | 337 | 3 |
| Age Groups & Complaints | Antibiotics | ||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Penicillin with Extended Spectrum; Amoxicillin and Ampicillin with Cloxacillin; J01CA n = 4120 | Penicillin with β -Lactamase Inhibitor, Amoxicillin with Clavulanic acid; J01CR n = 20 | Tetracyclines; J01A n = 323 | Cephalosporins; J01D | Aminoglycosides; J01GB n = 1932 | Macrolides; J01FA n = 157 | Fluoroquinolones; J01MA n = 4307 | Metronidazole; P01AB n = 147 | ||||||||||||
| 1st Generation; J01DB n = 90 | 3rd Generation; J01DD n = 3062 | ||||||||||||||||||
| O | P | O | P | O | P | O | P | O | P | O | P | O | P | O | P | O | P | ||
| Fever | 0–5yr | 315 | 9 | - | - | - | - | 10 | - | 25 | 148 | - | 39 | 5 | - | 91 | - | - | - |
| 6–17 yr | 191 | 20 | - | - | 2 | - | 3 | - | 30 | 143 | - | 141 | 2 | - | 140 | 2 | - | - | |
| 18 yr and above | 357 | 59 | 5 | - | 95 | - | 5 | 1 | 226 | 623 | - | 644 | 13 | - | 1657 | 53 | - | 1 | |
| URTI* | 0–5yr | 747 | 39 | 2 | 1 | 1 | - | 10 | - | 118 | 315 | - | 48 | 10 | - | 86 | - | - | - |
| 6–17 yr | 326 | 39 | - | - | 10 | - | 12 | - | 42 | 222 | - | 92 | 6 | - | 114 | 1 | - | - | |
| 18 yr and above | 463 | 121 | 3 | - | 76 | - | 9 | - | 96 | 577 | 1 | 222 | 42 | - | 843 | 11 | - | - | |
| Gastro- intestinal disorders | 0–5yr | 52 | 3 | - | - | - | - | 2 | - | 1 | 21 | - | 79 | - | - | 20 | - | 3 | - |
| 6–17 yr | 16 | 4 | - | - | - | - | - | - | 2 | 18 | - | 54 | 1 | - | 28 | 1 | 9 | 2 | |
| 18 yr and above | 47 | 9 | 3 | - | 38 | - | - | 1 | 12 | 169 | - | 381 | 1 | - | 311 | 13 | 80 | 48 | |
| Oral & dental problems | 0–5yr | 3 | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | |
| 6–17 yr | 5 | 1 | - | - | - | - | - | - | - | - | - | 1 | - | - | 8 | - | - | - | |
| 18 yr and above | 872 | 55 | 4 | - | 7 | - | 4 | - | 3 | 19 | - | 39 | - | - | 185 | 1 | 1 | 1 | |
| Skin infections | 0–5yr | 33 | 12 | - | - | 1 | - | 6 | - | 5 | 10 | - | 7 | - | - | 10 | - | - | - |
| 6–17 yr | 42 | 18 | - | - | 5 | - | 7 | - | 4 | 26 | - | 35 | 12 | - | 42 | - | - | - | |
| 18 yr and above | 62 | 31 | - | - | 54 | - | 10 | - | 12 | 48 | - | 72 | 64 | 1 | 289 | - | 1 | - | |
| Injury | 0–5yr | 9 | 1 | - | - | - | - | 5 | - | 1 | 2 | - | 3 | - | - | 3 | - | - | - |
| 6–17 yr | 10 | 9 | - | - | - | - | 2 | - | 1 | 15 | - | 5 | - | - | 23 | - | - | - | |
| 18 yr and above | 33 | 18 | - | - | - | - | 4 | 1 | 43 | 37 | - | - | 163 | 1 | - | - | |||
| Pain | 0–5yr | - | 1 | - | - | - | - | - | - | - | - | - | 2 | - | - | 1 | - | - | - |
| 6–17 yr | - | - | - | - | - | - | - | - | - | - | - | 3 | - | - | 1 | - | - | - | |
| 18 yr and above | 26 | 2 | - | 1 | 6 | - | - | - | 7 | 13 | - | 12 | - | - | 172 | - | 1 | - | |
| Asthma | 0–5yr | 3 | 1 | - | - | - | - | - | - | 1 | 2 | - | - | - | - | - | - | - | - |
| 6–17 yr | 1 | 1 | - | - | - | - | - | - | - | 2 | - | - | - | - | 1 | - | - | - | |
| 18 yr and above | 41 | 13 | 1 | - | 28 | - | - | - | 10 | 49 | - | 15 | 4 | - | 30 | 6 | - | - | |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Khare, S.; Purohit, M.; Sharma, M.; Tamhankar, A.J.; Lundborg, C.S.; Diwan, V.; Pathak, A. Antibiotic Prescribing by Informal Healthcare Providers for Common Illnesses: A Repeated Cross-Sectional Study in Rural India. Antibiotics 2019, 8, 139. https://doi.org/10.3390/antibiotics8030139
Khare S, Purohit M, Sharma M, Tamhankar AJ, Lundborg CS, Diwan V, Pathak A. Antibiotic Prescribing by Informal Healthcare Providers for Common Illnesses: A Repeated Cross-Sectional Study in Rural India. Antibiotics. 2019; 8(3):139. https://doi.org/10.3390/antibiotics8030139
Chicago/Turabian StyleKhare, Shweta, Manju Purohit, Megha Sharma, Ashok J. Tamhankar, Cecilia Stalsby Lundborg, Vishal Diwan, and Ashish Pathak. 2019. "Antibiotic Prescribing by Informal Healthcare Providers for Common Illnesses: A Repeated Cross-Sectional Study in Rural India" Antibiotics 8, no. 3: 139. https://doi.org/10.3390/antibiotics8030139
APA StyleKhare, S., Purohit, M., Sharma, M., Tamhankar, A. J., Lundborg, C. S., Diwan, V., & Pathak, A. (2019). Antibiotic Prescribing by Informal Healthcare Providers for Common Illnesses: A Repeated Cross-Sectional Study in Rural India. Antibiotics, 8(3), 139. https://doi.org/10.3390/antibiotics8030139






