The Consequences of AMR Education and Awareness Raising: Outputs, Outcomes, and Behavioural Impacts of an Antibiotic-Related Educational Activity in Lao PDR
Abstract
:1. Introduction
2. Material and Methods
2.1. Study Site
2.2. Study Population
2.3. Intervention (Educational Activity)
- “Always follow health workers’ advice when using antibiotics”,
- “Never demand antibiotics if health workers say you do not need them”,
- Only use antibiotics when prescribed by a certified health professional”,
- “Germs can become ‘stronger’ if treated inappropriately until the point that there is no medicine to treat them anymore”, and
- “Drug resistance can spread”.
2.4. Data Collection
2.5. Sample Size
2.6. Study Outcomes
2.7. Data Analysis
2.8. Ethical Considerations
3. Results
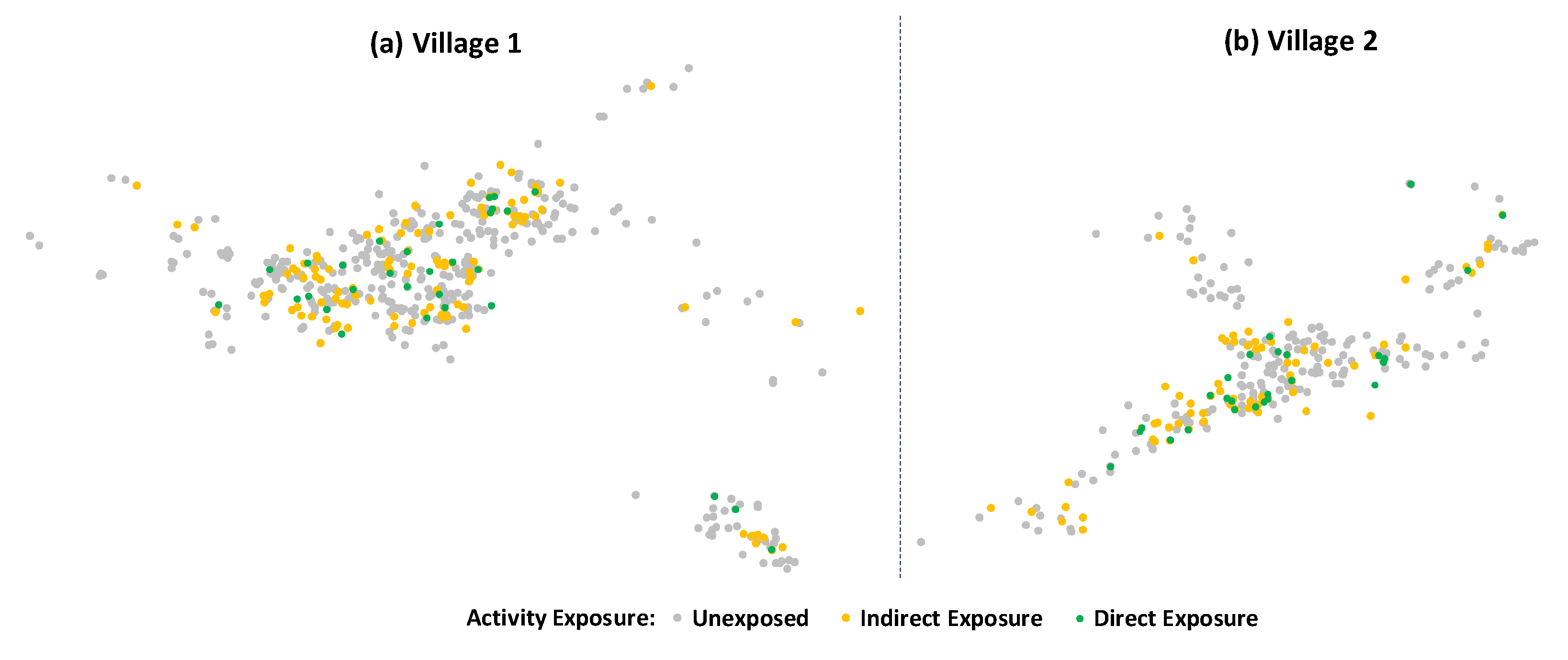
3.1. Outputs
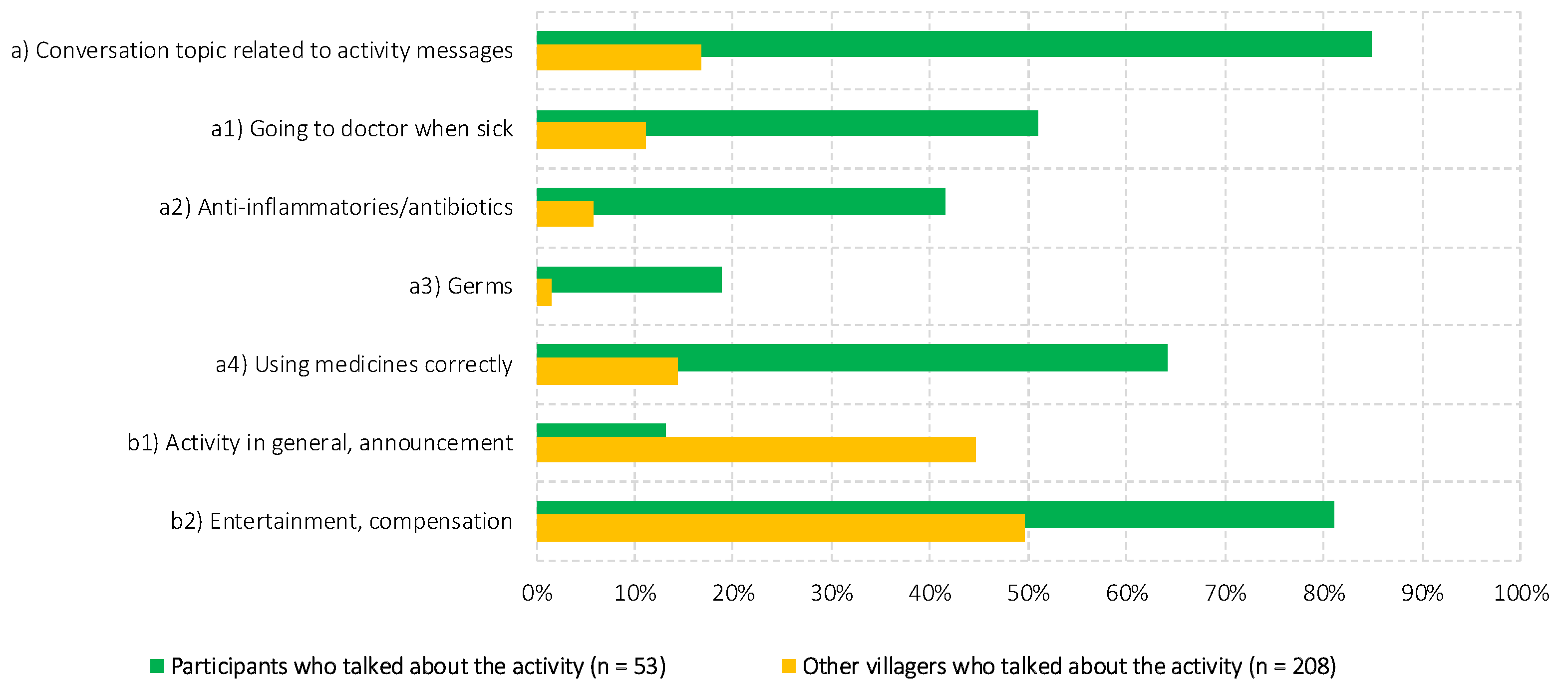
3.2. Outcomes
3.3. Impact
- The directly and indirectly exposed groups reported higher public healthcare access in the second survey round.
- Private healthcare access followed a slightly increasing trend that was absent from the indirectly exposed group.
- Informal healthcare access was generally low but followed a slightly increasing trend that was absent from the directly exposed group.
- The involvement of “other” healthcare providers (especially retired doctors running informal practices in the villages) decreased only in the directly exposed group—to a level similar to the other groups.
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- World Bank. Drug-Resistant Infections: A Threat to Our Economic Future; World Bank: Washington, DC, USA, 2017. [Google Scholar]
- The Review on Antimicrobial Resistance. Tackling Drug-Resistant Infections Globally: Final Report and Recommendations; The UK Prime Minister: London, UK, 2016. [Google Scholar]
- WHO. Global Action Plan on Antimicrobial Resistance; World Health Organization: Geneva, Switzerland, 2015. [Google Scholar]
- McCullough, A.R.; Parekh, S.; Rathbone, J.; Del Mar, C.B.; Hoffmann, T.C. A systematic review of the public’s knowledge and beliefs about antibiotic resistance. J. Antimicrob. Chemother. 2016, 71, 27–33. [Google Scholar] [CrossRef] [PubMed]
- WHO. Antibiotic Resistance: Multi-Country Public Awareness Survey; World Health Organization: Geneva, Switzerland, 2015. [Google Scholar]
- Bloom, G.; Merrett, G.B.; Wilkinson, A.; Lin, V.; Paulin, S. Antimicrobial resistance and universal health coverage. BMJ Glob. Health 2017, 2. [Google Scholar] [CrossRef] [PubMed]
- Ringold, D.J. Boomerang effects in response to public health interventions: Some unintended consequences in the alcoholic beverage market. J. Consum. Policy 2002, 25, 27–63. [Google Scholar] [CrossRef]
- Cho, H.; Salmon, C.T. Unintended effects of health communication campaigns. J. Commun. 2007, 57, 293–317. [Google Scholar] [CrossRef]
- Davis, M.; Whittaker, A.; Lindgren, M.; Djerf-Pierre, M.; Manderson, L.; Flowers, P. Understanding media publics and the antimicrobial resistance crisis. Glob. Public Health 2017, 13, 1158–1168. [Google Scholar] [CrossRef] [PubMed]
- Fynbo, L.; Jensen, C.S. Antimicrobial stigmatization: Public health concerns about conventional pig farming and pig farmers’ experiences with stigmatization. Soc. Sci. Med. 2018, 201, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Gelband, H.; Miller-Petrie, M.; Pant, S.; Gandra, S.; Levinson, J.; Barter, D.; White, A.; Laxminarayan, R. State of the World’s Antibiotics, 2015; Center for Disease Dynamics, Economics & Policy: Washington, DC, USA, 2015. [Google Scholar]
- ReAct. Antibiotic Smart Use, Thailand: Involving Community to Curb Antibiotic Resistance; ReAct: Uppsala, Sweden, 2016. [Google Scholar]
- WHO. Antimicrobial Resistance Behaviour Change First Informal Technical Consultation, 6–7 November, 2017 Château de Penthes, Geneva: Meeting Report; World Health Organization: Geneva, Switzerland, 2017. [Google Scholar]
- Elsevier, B.V. Scopus. Available online: http://www.scopus.com/ (accessed on 30 August 2018).
- OECD Development Assistance Committee. Principles for Evaluation of Development Assistance; Organisation for Economic Co-operation and Development: Paris, France, 1991. [Google Scholar]
- Haenssgen, M.J.; Charoenboon, N.; Zanello, G.; Mayxay, M.; Reed-Tsochas, F.; Jones, C.O.H.; Kosaikanont, R.; Praphattong, P.; Manohan, P.; Lubell, Y.; et al. Antibiotics and activity spaces: Protocol of an exploratory study of behaviour, marginalisation, and knowledge diffusion. BMJ Glob. Health 2018, 3, e000621. [Google Scholar] [CrossRef] [PubMed]
- Coulombe, H.; Epprecht, M.; Pimhidzai, O.; Sisoulath, V. Where Are the Poor? Lao PDR 2015 Census-Based Poverty Map: Province and District Level Results; Ministry of Planning and Investment: Vientiane, Laos, 2016.
- Google Inc. Google Maps. Available online: http://maps.google.com (accessed on 17 April 2018).
- Lao Statistics Bureau. Results of Population and Housing Census 2015; Ministry of Planning and Investment: Vientiane, Laos, 2016.
- National Geospatial-Intelligence Agency. Country Files (GNS). Available online: http://geonames.nga.mil/gns/html/namefiles.html (accessed on 17 July 2017).
- Carter, E.; Bryce, J.; Perin, J.; Newby, H. Harmful practices in the management of childhood diarrhea in low- and middle-income countries: A systematic review. BMC Public Health 2015, 15, 788. [Google Scholar] [CrossRef] [PubMed]
- WHO. Antibiotic Resistance. Available online: http://www.who.int/news-room/fact-sheets/detail/antibiotic-resistance (accessed on 15 September 2017).
- Charoenboon, N.; Haenssgen, M.J.; Warapikuptanun, P.; Xayavong, T.; Khine Zaw, Y. Translating AMR: A case study of three Thai villages. In Proceedings of the International Health Conference, St Hugh’s College, Oxford, UK, 28–30 June 2018. [Google Scholar]
- Chuang, Y.; Schechter, L. Social networks in developing countries. Annu. Rev. Resour. Econ. 2015, 7, 451–472. [Google Scholar] [CrossRef]
- Valente, T.W. Social Networks and Health: Models, Methods, and Applications; Oxford University Press: Oxford, UK, 2010. [Google Scholar]
- Dobility Inc. SurveyCTO; Dobility Inc.: Cambridge, MA, USA, 2017. [Google Scholar]
- Haenssgen, M.J. Satellite-aided survey sampling and implementation in low- and middle-income contexts: A low-cost/low-tech alternative. Emerg. Themes Epidemiol. 2015, 12, 20. [Google Scholar] [CrossRef] [PubMed]
- Mann, H.B.; Whitney, D.R. On a test of whether one of two random variables is stochastically larger than the other. Ann. Math. Stat. 1947, 18, 50–60. [Google Scholar] [CrossRef]
- Wilcoxon, F. Individual comparisons by ranking methods. Biom. Bull. 1945, 1, 80–83. [Google Scholar] [CrossRef]
- StataCorp. Stata Statistical Software: Release 15; StataCorp LP: College Station, TX, USA, 2017. [Google Scholar]
- Haenssgen, M.J. The patient—Health system interface: A case study of antibiotic-related health behaviour in rural Thailand and Lao PDR. In Proceedings of the International Health Conference, St Hugh’s College, Oxford, UK, 28–30 June 2018. [Google Scholar]
- Polgar, S. Health action in cross-cultural perspective. In Handbook of Medical Sociology; Freeman, H.E., Levine, S., Reeder, L.G., Eds.; Prentice-Hall: Englewood Cliffs, NJ, USA, 1963; Volume 1, pp. 397–419. [Google Scholar]
- Rogers, E.M. Diffusion of Innovations, 5th ed.; Free Press: New York, NY, USA, 2003. [Google Scholar]
- Broom, A.; Broom, J.; Kirby, E.; Gibson, A.; Davis, M. Antibiotic optimisation in ‘the bush’: Local know-how and core-periphery relations. Health Place 2017, 48, 56–62. [Google Scholar] [CrossRef] [PubMed]
- Hawkings, N.J.; Wood, F.; Butler, C.C. Public attitudes towards bacterial resistance: A qualitative study. J. Antimicrob. Chemother. 2007, 59, 1155–1160. [Google Scholar] [CrossRef] [PubMed]
- Nerlich, B.; James, R. “The post-antibiotic apocalypse” and the “war on superbugs”: Catastrophe discourse in microbiology, its rhetorical form and political function. Public Underst. Sci. 2009, 18, 574–590. [Google Scholar] [CrossRef] [PubMed]
- Tangcharoensathien, V.; Sommanustweechai, A.; Chanvatik, S.; Kosiyaporn, H.; Tisocki, K. Addressing the threat of antibiotic resistance in Thailand: Monitoring population knowledge and awareness. WHO South-East Asia J. Public Health 2018, 7, 73–78. [Google Scholar] [CrossRef] [PubMed]
- Peglidou, A. Therapeutic itineraries of ‘depressed’ women in Greece: Power relationships and agency in therapeutic pluralism. Anthropol. Med. 2010, 17, 41–57. [Google Scholar] [CrossRef] [PubMed]
- Bloom, G.; Wilkinson, A.; Tomson, G.; Awor, P.; Zhang, X.; Ahmed, S.M.; Khan, W.A.; Blessing, V.; Wang, L.; Liang, X.; et al. Addressing Resistance to Antibiotics in Pluralistic Health Systems; University of Sussex: Brighton, UK, 2015. [Google Scholar]
- Leventhal, H.; Weinman, J.; Leventhal, E.A.; Phillips, L.A. Health psychology: The search for pathways between behavior and health. Annu. Rev. Psychol. 2008, 59, 477–505. [Google Scholar] [CrossRef] [PubMed]
- Ribera, J.M.; Hausmann-Muela, S. The straw that breaks the camel’s back: Redirecting health-seeking behavior studies on malaria and vulnerability. Med. Anthropol. Q. 2011, 25, 103–121. [Google Scholar] [CrossRef] [PubMed]
- Ocan, M.; Obuku, E.A.; Bwanga, F.; Akena, D.; Richard, S.; Ogwal-Okeng, J.; Obua, C. Household antimicrobial self-medication: A systematic review and meta-analysis of the burden, risk factors and outcomes in developing countries. BMC Public Health 2015, 15, 742. [Google Scholar] [CrossRef] [PubMed]
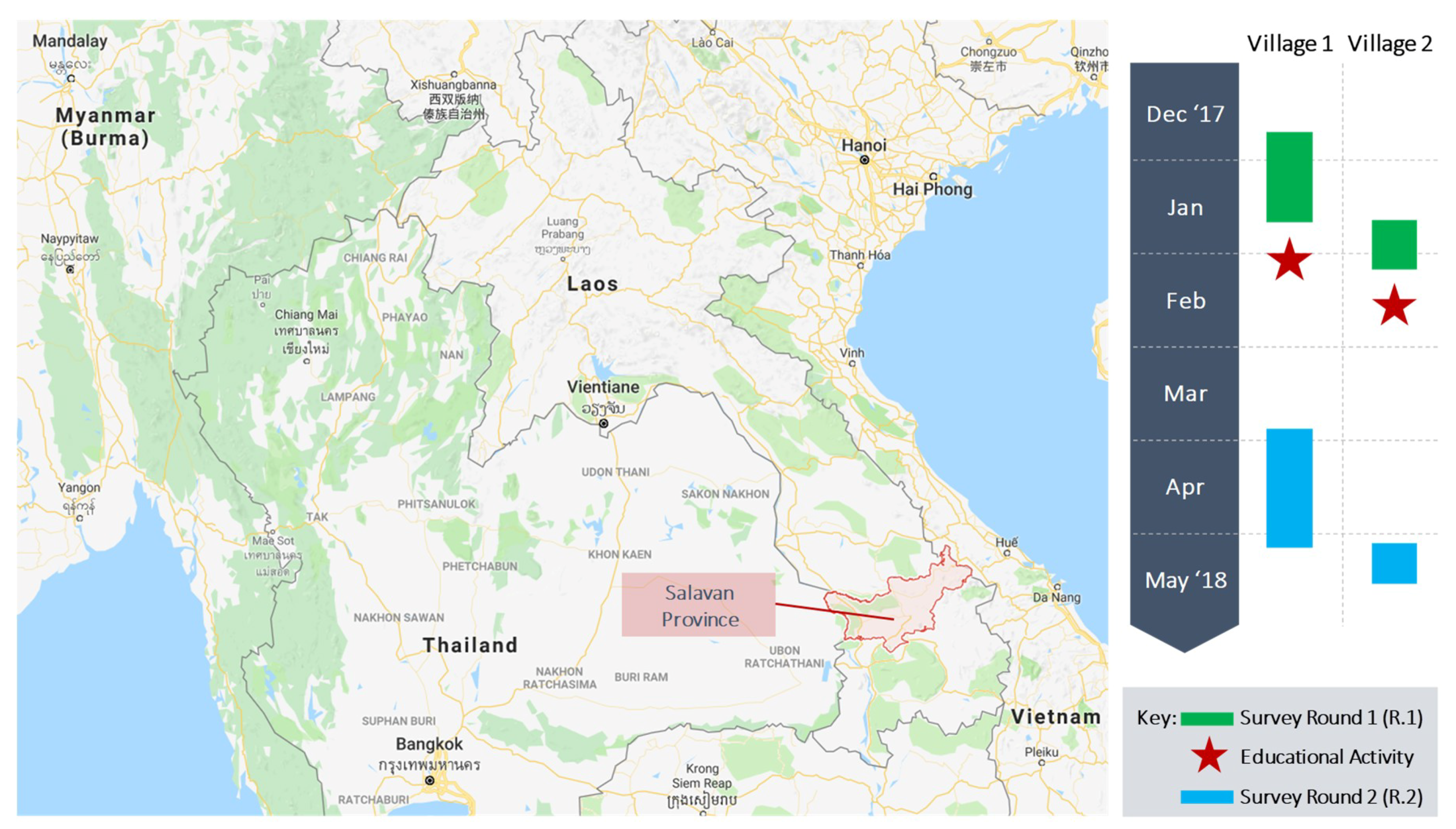

| Village Attributes | Village 1 | Village 2 | Salavan Average |
|---|---|---|---|
| Village Size | 1462 | 744 | 369 a |
| Household Size | 5.0 | 4.5 | 5.9 |
| Female Population Share | 48.8% | 53.1% | 50.1% |
| Dependency Ratio c | 0.68 | 0.63 | 0.64 b |
| Households Owning Mobile Phones | 96.0% | 89.9% | 81.6% |
| Session & Duration | Description | Expected Outcomes | Main Message a | Direction of Communication |
|---|---|---|---|---|
| Ice Breaking (15 min) | Ice breaking. | |||
| (1) Community Mapping (30 min) | In groups, participants sketch a village map and mark down their own houses, important locations in the village, and draw lines to connect themselves with places as well as people they go to when sick. | Team learns about places, existing health networks, and health facilities within and nearby the village. | Participants ↓ Team | |
| (2) Medicine Matching (30 min) | Part (I): Participants sort medicines into 2 groups—those that they know and do not know. | Team gains overview of medicines and their purposes from participants’ perspective. | Participants ↓ Team | |
| Part (II): Participants free-sort pictures of common medicines into their own categories. | Team understands participants’ general conceptions around medicines and treatments. | Participants ↓ Team | ||
| Part (III): Participants sort medicines into two groups—over-the-counter medicines and prescription medicines. | Participants reflect on the ways to access medicines. | Only use antibiotics when prescribed by a certified health professional. | Team ↓ Participants | |
| (3) Resistance (30 min) | Participants pass a germ around in a circle. When the music stops, the person with the germ answers a right-or-wrong question about taking medicines. If incorrect, she/he are out of the game, the germ evolves, and the game continues. The last remaining person wins a prize. | Participants become familiar with the idea of bacteria evolving and resisting medicines. | (1) Germs can become “stronger” if treated inappropriately until the point that there is no medicine to treat them anymore. (2) Drug resistance can spread. | Team ↓ Participants |
| Break (15 min) | Break. | |||
| (4) Role-Play (20 min) | A short skit performed by the team with a simple storyline around antibiotics and antibiotic resistance. | Participants gain a deeper understanding about drug resistance and their own part in the issue. | (1) Always follow health workers’ advice when using antibiotics. (2) Never demand antibiotics if health workers say you don’t need them. (3) Drug resistance can spread. | Team ↓ Participants |
| (5) Healthy-Wealthy Game (30 min) | Participants simulate running a family business that produces goods and sells to the market. Each group (family) has different tools to make as much money as they can. Throughout the activity, family members are diagnosed randomly with a disease, provided with different treatment scenarios, and the rest of the family have to pay hospital fees to bring that sick member back. | Participants reflect on common illnesses and the various ways of treating them; the team gains an understanding of health decisions in the local context. | Only use antibiotics when prescribed by a certified health professional. | Team ↓ ↑ Participants |
| (6) Feedback and Reflection (15 min) | Participants provide their reflections on the activities and lessons learned. | Participants understand the key messages from the activities, and express these to the group. | Participants ↓ Participants/Team | |
| Variable | Survey Round I | Survey Round II | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Mean | Std. Dev. | Min | Max | n | Mean | Std. Dev. | Min | Max | n | ||
| Activity | % participated in educational activity: Throughout | n/a g | 0.05 | 0.21 | 0 | 1 | 1216 | ||||
| % participated in educational activity: Partly | n/a g | 0.00 | 0.06 | 0 | 1 | 1216 | |||||
| % heard about educational activity | n/a g | 0.22 | 0.41 | 0 | 1 | 1216 | |||||
| Demographic Attributes | Sex (% female) | 0.55 | 0.50 | 0 | 1 | 1264 | 0.56 | 0.50 | 0 | 1 | 1216 |
| Age | 39.91 | 17.14 | 18 | 100 | 1264 | 40.04 | 17.02 | 18 | 100 | 1216 | |
| Completed years of formal education | 6.28 | 4.59 | 0 | 21 | 1264 | 6.22 | 4.56 | 0 | 21 | 1216 | |
| Wealth index (range: 0 to 1) a,b | 0.49 | 0.13 | 0.11 | 0.78 | 454 | 0.50 | 0.13 | 0.11 | 0.78 | 446 | |
| % speaking Lao | 1.00 | 0.00 | 1 | 1 | 1264 | 1.00 | 0.00 | 1 | 1 | 1216 | |
| Ethnic group: Lao Loum | 0.97 | 0.18 | 0 | 1 | 1264 | 0.96 | 0.19 | 0 | 1 | 1216 | |
| Ethnic group: Other | 0.01 | 0.10 | 0 | 1 | 1264 | 0.01 | 0.10 | 0 | 1 | 1216 | |
| Ethnic group: Don’t know/prefer not to say | 0.02 | 0.15 | 0 | 1 | 1264 | 0.03 | 0.16 | 0 | 1 | 1216 | |
| Antibiotic Knowledge/Attitudes | % have seen antibiotic capsules | 0.97 | 0.18 | 0 | 1 | 1264 | 0.96 | 0.19 | 0 | 1 | 1216 |
| % have heard of drug resistance (“due yah”) c | 0.39 | 0.49 | 0 | 1 | 1264 | 0.63 | 0.48 | 0 | 1 | 1216 | |
| % have heard of drug resistance (“lueng yah”) c | 0.79 | 0.40 | 0 | 1 | 1264 | 0.82 | 0.39 | 0 | 1 | 1216 | |
| % buy antibiotics over the counter (attitude) | 0.30 | 0.46 | 0 | 1 | 1264 | 0.27 | 0.44 | 0 | 1 | 1216 | |
| % prefer antibiotics over alternatives (attitude) | 0.29 | 0.45 | 0 | 1 | 1264 | 0.24 | 0.43 | 0 | 1 | 1216 | |
| % do not keep antibiotics for future use (knowledge) | 0.22 | 0.41 | 0 | 1 | 1264 | 0.27 | 0.44 | 0 | 1 | 1216 | |
| % antibiotic resistance can spread (knowledge) | 0.12 | 0.32 | 0 | 1 | 1264 | 0.02 | 0.15 | 0 | 1 | 1216 | |
| No. of desirable knowledge/attitude answers (0–4) | 0.93 | 0.93 | 0 | 4 | 1264 | 0.81 | 0.87 | 0 | 4 | 1216 | |
| Treatment-Seeking Behaviour d,e | % of illness episodes involving children | 0.39 | 0.49 | 0 | 1 | 512 | 0.35 | 0.48 | 0 | 1 | 284 |
| Self-rated severity (1 = mild, 2 = medium, 3 = severe) | 1.79 | 0.70 | 1 | 3 | 512 | 1.88 | 0.67 | 1 | 3 | 284 | |
| Average duration of illness episode (days) | 7.29 | 9.25 | 1 | 130 | 512 | 7.36 | 14.42 | 1 | 219 | 284 | |
| Average no. of medicines and treatments received f | 2.74 | 1.71 | 0 | 13 | 512 | 2.46 | 1.39 | 0 | 8 | 284 | |
| Average no. of antibiotics | 0.50 | 0.70 | 0 | 4 | 512 | 0.42 | 0.59 | 0 | 3 | 284 | |
| Average no. of antibiotics (incl. “uncertain” medicine) | 1.31 | 1.45 | 0 | 10 | 512 | 1.15 | 1.25 | 0 | 6 | 284 | |
| % public providers (health centres, hospitals) | 0.27 | 0.44 | 0 | 1 | 512 | 0.27 | 0.44 | 0 | 1 | 284 | |
| % private providers (clinics, hospitals, pharmacies) | 0.53 | 0.50 | 0 | 1 | 512 | 0.59 | 0.49 | 0 | 1 | 284 | |
| % informal providers (grocery stores, healers) | 0.02 | 0.14 | 0 | 1 | 512 | 0.05 | 0.22 | 0 | 1 | 284 | |
| % family and self-care | 0.97 | 0.18 | 0 | 1 | 512 | 1.00 | 0.06 | 0 | 1 | 284 | |
| % others | 0.06 | 0.25 | 0 | 1 | 512 | 0.07 | 0.26 | 0 | 1 | 284 | |
| Framework Element | Outputs | Outcomes | Impacts |
|---|---|---|---|
| Level of Analysis | Individual | Individual | Illness |
| Indicators | Direct and indirect exposure to educational activity | Awareness and understanding of drug resistance a | Patterns of healthcare utilisation during acute illnesses c |
| Lessons and feedback from educational activity | “Desirability” of antibiotic-related attitudes and knowledge b | Sources of antibiotics during acute illnesses d |
| Variable | Direct Exposure (i.e., Participated in Activity) (n = 58) | Indirect Exposure (i.e., Talked About Activity) (n = 208) | Unexposed (n = 864) |
|---|---|---|---|
| Mean (Std. Dev) | Mean (Std. Dev) | Mean (Std. Dev) | |
| Sex (% female) | 0.71 (0.46) | 0.63 (0.49) | 0.54 (0.50) |
| Age | 44.76 (11.36) | 38.18 (14.91) | 40.85 (17.75) |
| Education | 7.10 (4.06) | 8.18 (4.99) | 5.67 (4.36) |
| Wealth index (range: 0 to 1) a | 0.51 (0.12) | 0.54 (0.11) | 0.50 (0.12) |
| Ethnic group: Lao Loum | 1.00 (0.00) | 0.99 (0.12) | 0.96 (0.20) |
| Ethnic group: Other | 0.00 (0.00) | 0.01 (0.10) | 0.01 (0.11) |
| Ethnic group: Don’t know/prefer not to say | 0.00 (0.00) | 0.00 (0.07) | 0.03 (0.16) |
| Variable | Direct Exposure (n = 58) | Indirect Exposure (n = 208) | Unexposed (n = 864) | Difference-in-Difference | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Survey Round 1 | Survey Round 2 | Diffe-rence | Survey Round 1 | Survey Round 2 | Diffe-rence | Survey Round 1 | Survey Round 2 | Diffe-rence | Direct vs. Unexposed | Indirect vs. Unexposed | ||
| (a1) Awareness of Drug Resistance (Due Yah) | Heard of drug resistance (“due yah”) | 27.6% | 91.4% | +63.8% | 49.0% | 75.0% | +26.0% | 36.2% | 58.8% | +22.6% | +41.2% | +3.4% |
| Interpretations: Reference to antibiotics/drug-resistant germs | 1.7% | 3.5% | +1.7% | 5.3% | 1.0% | −4.3% | 3.7% | 2.4% | −1.3% | +3.0% | −3.1% | |
| Medicine does not work | 6.9% | 17.2% | +10.3% | 15.9% | 15.9% | 0.0% | 12.6% | 10.0% | −2.7% | +13.0% | +2.7% | |
| Taking medicine wrongly (e.g., wrong type, too much) | 1.7% | 6.9% | +5.2% | 2.9% | 1.9% | −1.0% | 2.3% | 2.2% | −0.1% | +5.3% | −0.9% | |
| Stubborn patient, medicine restrictions/dislikes | 5.2% | 1.7% | −3.5% | 11.1% | 4.8% | −6.3% | 10.1% | 7.5% | −2.6% | −0.9% | −3.7% | |
| Addicted to or strong preference for medicine | 3.5% | 0.0% | −3.5% | 3.4% | 4.3% | +1.0% | 2.7% | 2.7% | 0.0% | −3.5% | +1.0% | |
| Side-effects, drug allergy, or a specific illness | 13.8% | 6.9% | −6.9% | 7.2% | 10.1% | +2.9% | 8.2% | 6.0% | −2.2% | −4.7% | +5.1% | |
| “Lueng yah” | 25.9% | 55.2% | +29.3% | 17.3% | 41.4% | +24.0% | 14.7% | 30.6% | +15.9% | +13.5% | +8.2% | |
| Other interpretation | 3.5% | 1.7% | −1.7% | 1.9% | 1.4% | −0.5% | 2.1% | 1.9% | −0.2% | −1.5% | −0.3% | |
| Don’t know / cannot or prefer not to answer | 37.9% | 6.9% | −31.0% | 35.1% | 19.2% | −15.9% | 43.6% | 36.8% | −6.8% | −24.2% | −9.1% | |
| (a2) Awareness of Drug Resistance (Lueng Yah) | Heard of drug resistance (“lueng yah”) | 93.1% | 96.6% | +3.4% | 84.6% | 90.4% | +5.8% | 77.5% | 80.2% | +2.7% | +0.8% | +3.1% |
| Interpretations: Reference to antibiotics / drug-resistant germs | 19.0% | 22.4% | +3.4% | 17.8% | 6.7% | −11.1% | 9.6% | 4.9% | −4.8% | +8.2% | −6.3% | |
| Medicine does not work | 48.3% | 44.8% | −3.5% | 44.7% | 53.4% | +8.7% | 43.1% | 41.4% | −1.6% | −1.8% | +10.3% | |
| Taking medicine wrongly (e.g., wrong type, too much) | 3.5% | 6.9% | +3.5% | 2.9% | 1.9% | −1.0% | 2.2% | 5.8% | +3.6% | −0.1% | −4.6% | |
| Stubborn patient, medicine restrictions/dislikes | 0.0% | 0.0% | 0.0% | 1.0% | 1.0% | 0.0% | 1.3% | 1.0% | −0.2% | +0.2% | +0.2% | |
| Addicted to or strong preference for medicine | 20.7% | 10.3% | −10.4% | 21.6% | 24.0% | +2.4% | 20.8% | 23.2% | +2.3% | −12.7% | +0.1% | |
| Side-effects, drug allergy, or a specific illness | 1.7% | 3.5% | +1.7% | 0.5% | 0.0% | −0.5% | 1.4% | 1.6% | +0.2% | +1.5% | −0.7% | |
| Other interpretation | 3.5% | 6.9% | +3.5% | 2.4% | 3.4% | +1.0% | 4.3% | 3.7% | −0.6% | +4.0% | +1.6% | |
| Don’t know / cannot or prefer not to answer | 3.5% | 5.2% | +1.7% | 9.1% | 9.6% | +0.5% | 17.4% | 18.4% | +1.0% | +0.7% | −0.6% | |
| (b) Attitudes and Knowledge | No. of desirable knowledge/attitude answers (0–4) | 1.22 | 1.33 | +0.10 | 0.96 | 0.82 | −0.14 | 0.90 | 0.79 | −0.11 | +0.22 | −0.03 |
| Would buy antibiotics over the counter | 48.3% | 55.2% | +6.9% | 32.2% | 32.7% | +0.5% | 28.5% | 24.7% | −3.8% | +10.7% | +4.3% | |
| Prefers antibiotics over alternatives | 32.8% | 31.0% | −1.7% | 29.8% | 20.2% | −9.6% | 28.0% | 25.5% | −2.5% | +0.8% | −7.1% | |
| Would not keep antibiotics for future use | 27.6% | 32.8% | +5.2% | 19.7% | 28.4% | +8.7% | 22.2% | 26.6% | +4.4% | +0.8% | +4.3% | |
| Thinks that antibiotic resistance can spread | 13.8% | 13.8% | 0.0% | 14.4% | 0.5% | −13.9% | 11.6% | 2.1% | −9.5% | +9.5% | −4.5% | |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Haenssgen, M.J.; Xayavong, T.; Charoenboon, N.; Warapikuptanun, P.; Khine Zaw, Y. The Consequences of AMR Education and Awareness Raising: Outputs, Outcomes, and Behavioural Impacts of an Antibiotic-Related Educational Activity in Lao PDR. Antibiotics 2018, 7, 95. https://doi.org/10.3390/antibiotics7040095
Haenssgen MJ, Xayavong T, Charoenboon N, Warapikuptanun P, Khine Zaw Y. The Consequences of AMR Education and Awareness Raising: Outputs, Outcomes, and Behavioural Impacts of an Antibiotic-Related Educational Activity in Lao PDR. Antibiotics. 2018; 7(4):95. https://doi.org/10.3390/antibiotics7040095
Chicago/Turabian StyleHaenssgen, Marco J., Thipphaphone Xayavong, Nutcha Charoenboon, Penporn Warapikuptanun, and Yuzana Khine Zaw. 2018. "The Consequences of AMR Education and Awareness Raising: Outputs, Outcomes, and Behavioural Impacts of an Antibiotic-Related Educational Activity in Lao PDR" Antibiotics 7, no. 4: 95. https://doi.org/10.3390/antibiotics7040095
APA StyleHaenssgen, M. J., Xayavong, T., Charoenboon, N., Warapikuptanun, P., & Khine Zaw, Y. (2018). The Consequences of AMR Education and Awareness Raising: Outputs, Outcomes, and Behavioural Impacts of an Antibiotic-Related Educational Activity in Lao PDR. Antibiotics, 7(4), 95. https://doi.org/10.3390/antibiotics7040095





