Management of Penicillin Allergy in the Perioperative Setting
Abstract
1. Introduction
2. SSI Risk in Penicillin-Allergic Patients
3. Challenges with the Use of Beta-Lactam Alternatives for Perioperative Prophylaxis
3.1. Spectrum of Coverage
3.2. Administration Logistics
3.3. Adverse Drug Reactions
3.4. Healthcare-Associated Infections
3.5. Summary
4. Beta-Lactam Allergy and Cross Reactivity
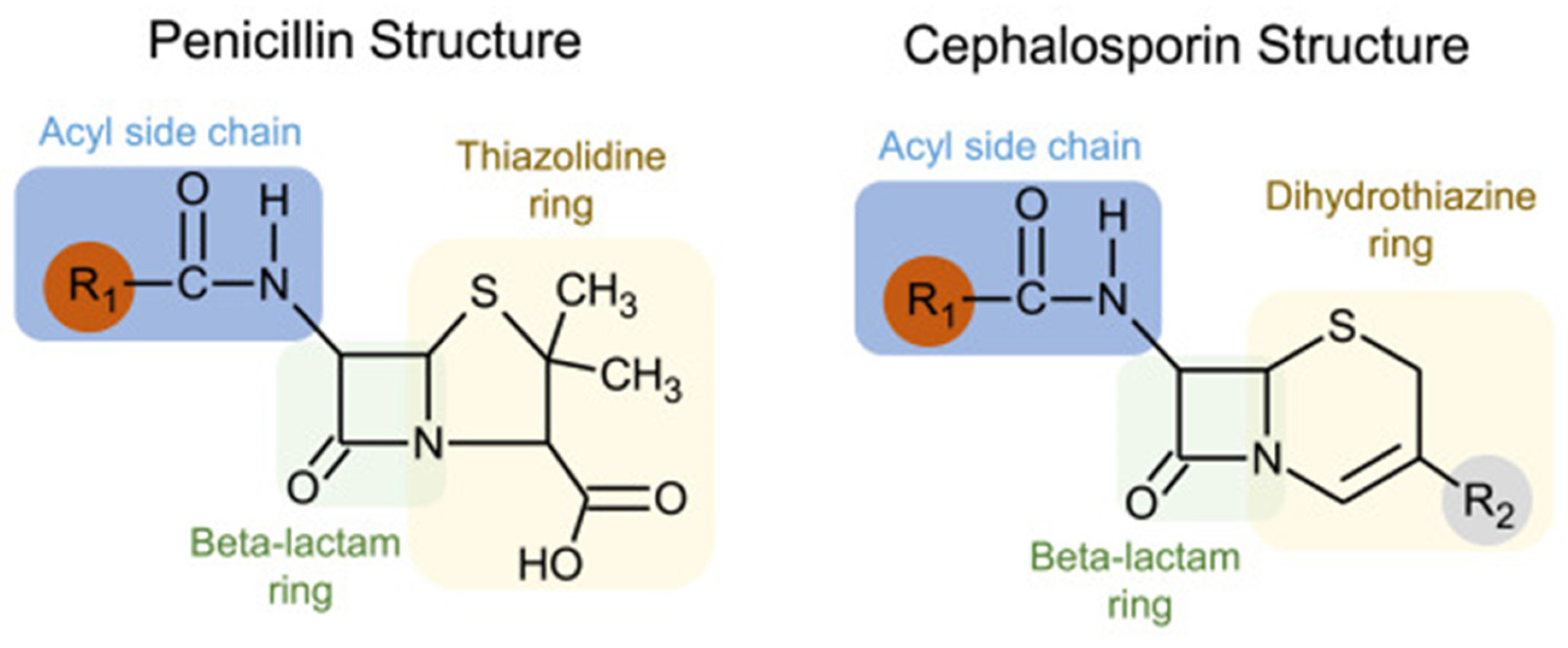
4.1. Penicillin Allergy and Cross-Reactivity with Cephalosporins
4.2. Differentiation of Cefazolin from Other Beta-Lactams
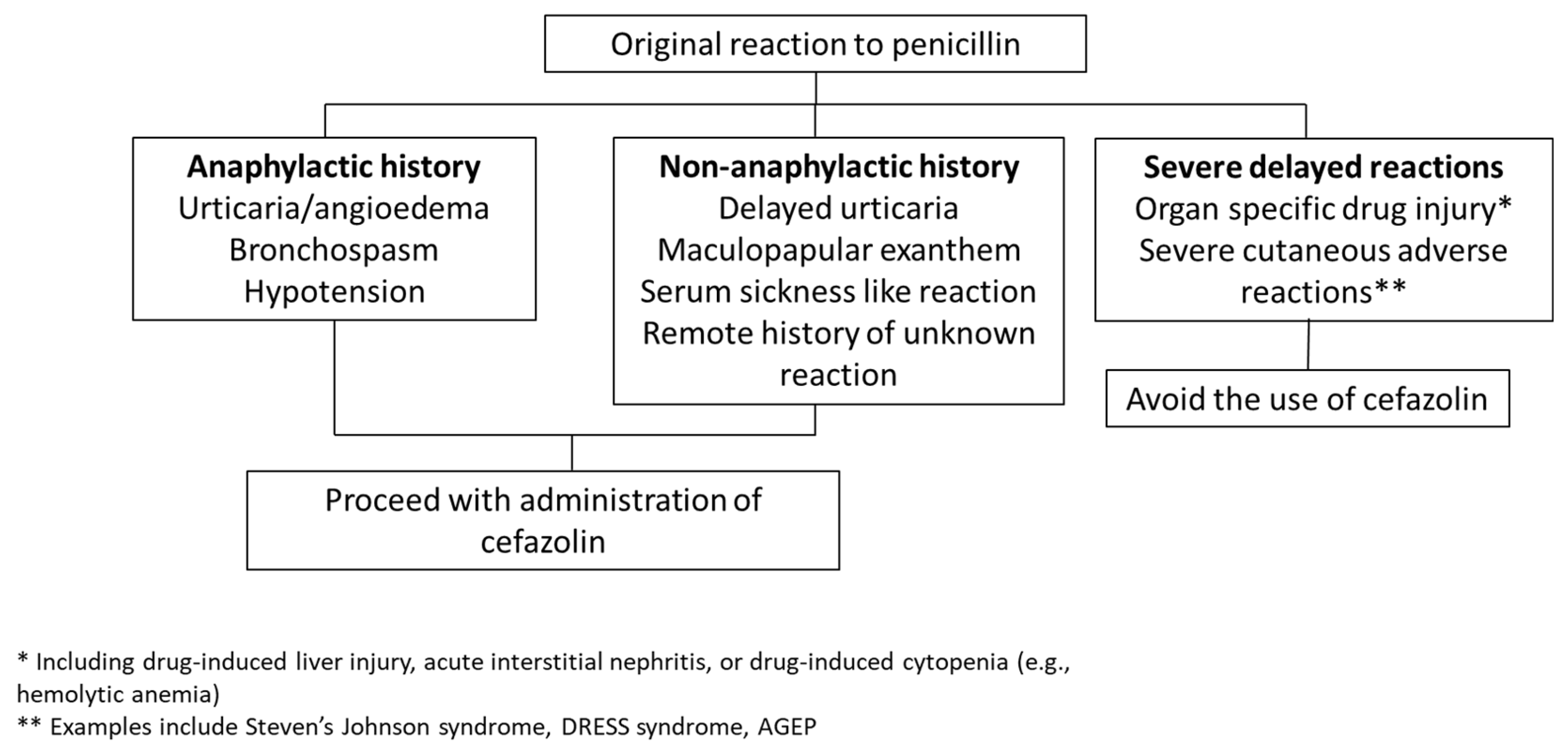
5. Evidence Supporting Safety of Cefazolin Use in Most Patients with PALs
6. Options for Management of Surgical Patients with a PAL
6.1. Allergy De-Labeling Prior to Elective Surgery
6.2. Patient Screening Questionnaires as Part of Preoperative Evaluation
6.3. Utilization of Clinical Decision Support Tools at the Time of Surgery
7. Efficacy and Cost Data
8. Implementation of Perioperative PAL Interventions: Challenges and Opportunities
8.1. Consensus Building and Provider Education
8.2. Use of the Electronic Medical Record
8.3. Advantages and Disadvantages of Potential Workflows
9. Future Directions
10. Conclusions
Funding
Conflicts of Interest
References
- Berrios-Torres, S.I.; Umscheid, C.A.; Bratzler, D.W.; Leas, B.; Stone, E.C.; Kelz, R.R.; Reinke, C.E.; Morgan, S.; Solomkin, J.S.; Mazuski, J.E.; et al. Centers for Disease Control and Prevention Guideline for the Prevention of Surgical Site Infection, 2017. JAMA Surg. 2017, 152, 784–791. [Google Scholar] [CrossRef]
- Brocard, E.; Reveiz, L.; Regnaux, J.P.; Abdala, V.; Ramon-Pardo, P.; Del Rio Bueno, A. Antibiotic prophylaxis for surgical procedures: A scoping review. Rev. Panam. Salud Publica 2021, 45, e62. [Google Scholar] [CrossRef]
- Bratzler, D.W.; Dellinger, E.P.; Olsen, K.M.; Perl, T.M.; Auwaerter, P.G.; Bolon, M.K.; Fish, D.N.; Napolitano, L.M.; Sawyer, R.G.; Slain, D.; et al. Clinical practice guidelines for antimicrobial prophylaxis in surgery. Am. J. Health Syst. Pharm. 2013, 70, 195–283. [Google Scholar] [CrossRef]
- Buchalter, D.B.; Nduaguba, A.; Teo, G.M.; Kugelman, D.; Aggarwal, V.K.; Long, W.J. Cefazolin remains the linchpin for preventing acute periprosthetic joint infection following primary total knee arthroplasty. Bone Jt. Open 2022, 3, 35–41. [Google Scholar] [CrossRef]
- Norvell, M.R.; Porter, M.; Ricco, M.H.; Koonce, R.C.; Hogan, C.A.; Basler, E.; Wong, M.; Jeffres, M.N. Cefazolin vs. Second-line Antibiotics for Surgical Site Infection Prevention After Total Joint Arthroplasty Among Patients With a Beta-lactam Allergy. Open Forum Infect. Dis. 2023, 10, ofad224. [Google Scholar] [CrossRef]
- Niu, T.; Zhang, Y.; Li, Z.; Bian, Y.; Zhang, J.; Wang, Y. The association between penicillin allergy and surgical site infection after orthopedic surgeries: A retrospective cohort study. Front. Cell Infect. Microbiol. 2023, 13, 1182778. [Google Scholar] [CrossRef]
- Roistacher, D.M.; Heller, J.A.; Ferraro, N.F.; August, M. Is Penicillin Allergy a Risk Factor for Surgical Site Infection After Oral and Maxillofacial Surgery? J. Oral Maxillofac. Surg. 2022, 80, 93–100. [Google Scholar] [CrossRef] [PubMed]
- Wilhelm, N.B.; Bonsall, T.J.; Miller, C.L. The Effect of Beta-lactam Allergy Status on the Rate of Surgical Site Infections: A Retrospective Cohort Study. Ann. Surg. 2022, 275, 208–212. [Google Scholar] [CrossRef] [PubMed]
- Blumenthal, K.G.; Ryan, E.E.; Li, Y.; Lee, H.; Kuhlen, J.L.; Shenoy, E.S. The Impact of a Reported Penicillin Allergy on Surgical Site Infection Risk. Clin. Infect. Dis. 2018, 66, 329–336. [Google Scholar] [CrossRef] [PubMed]
- Decker, B.K.; Nagrebetsky, A.; Lipsett, P.A.; Wiener-Kronish, J.P.; O’Grady, N.P. Controversies in Perioperative Antimicrobial Prophylaxis. Anesthesiology 2020, 132, 586–597. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.K.; Chuang, S.K.; August, M. Antibiotic Resistance in Severe Orofacial Infections. J. Oral Maxillofac. Surg. 2017, 75, 962–968. [Google Scholar] [CrossRef] [PubMed]
- Bull, A.L.; Worth, L.J.; Richards, M.J. Impact of vancomycin surgical antibiotic prophylaxis on the development of methicillin-sensitive staphylococcus aureus surgical site infections: Report from Australian Surveillance Data (VICNISS). Ann. Surg. 2012, 256, 1089–1092. [Google Scholar] [CrossRef]
- Tan, T.L.; Springer, B.D.; Ruder, J.A.; Ruffolo, M.R.; Chen, A.F. Is Vancomycin-only Prophylaxis for Patients With Penicillin Allergy Associated With Increased Risk of Infection After Arthroplasty? Clin. Orthop. Relat. Res. 2016, 474, 1601–1606. [Google Scholar] [CrossRef]
- Lange, S.M.; Sharpe, E.E.; Hertzfeldt, D.N.; Schroeder, D.R.; Sviggum, H.P. Effect of penicillin allergy on prophylactic antibiotic administration in the parturient undergoing cesarean delivery. Acta Anaesthesiol. Scand. 2021, 65, 329–334. [Google Scholar] [CrossRef] [PubMed]
- Kheir, M.M.; Tan, T.L.; Azboy, I.; Tan, D.D.; Parvizi, J. Vancomycin Prophylaxis for Total Joint Arthroplasty: Incorrectly Dosed and Has a Higher Rate of Periprosthetic Infection Than Cefazolin. Clin. Orthop. Relat. Res. 2017, 475, 1767–1774. [Google Scholar] [CrossRef]
- Catanzano, A.; Phillips, M.; Dubrovskaya, Y.; Hutzler, L.; Bosco, J., 3rd. The standard one gram dose of vancomycin is not adequate prophylaxis for MRSA. Iowa Orthop. J. 2014, 34, 111–117. [Google Scholar] [PubMed]
- Burzynska, J.; Jaworski, R.; Maruszewski, B.; Kansy, A.; Dzierzanowska-Fangrat, K. Perioperative Antibiotic Prophylaxis in Pediatric Cardiac Surgery-Simple Is Better. Antibiotics 2022, 12, 66. [Google Scholar] [CrossRef]
- Strymish, J.M.; O’Brien, W.; Itani, K.; Gupta, K.; Branch-Elliman, W. What Is the Primary Driver of Preoperative Vancomycin Use? It’s Not Methicillin-resistant Staphylococcus aureus-or Allergy. Clin. Infect. Dis. 2020, 71, 2732–2735. [Google Scholar] [CrossRef]
- Srisung, W.; Teerakanok, J.; Tantrachoti, P.; Karukote, A.; Nugent, K. Surgical prophylaxis with gentamicin and acute kidney injury: A systematic review and meta-analysis. Ann. Transl. Med. 2017, 5, 100. [Google Scholar] [CrossRef]
- Kalmanson, O.A.; McLoughlin, K.C.; Kiser, T.H.; Gubbels, S.P. Debilitating Gentamicin Ototoxicity: Case Report and Recommendations Against Routine Use in Surgical Prophylaxis. Ann. Otol. Rhinol. Laryngol. 2023, 132, 1686–1689. [Google Scholar] [CrossRef]
- Butler, D.F.; Lee, B.R.; Suppes, S.; Sandritter, T.; Newland, J.G.; Harte, L.; Goldman, J.L. Variability of surgical prophylaxis in penicillin-allergic children. Infect. Control Hosp. Epidemiol. 2018, 39, 1480–1483. [Google Scholar] [CrossRef]
- Blumenthal, K.G.; Lu, N.; Zhang, Y.; Li, Y.; Walensky, R.P.; Choi, H.K. Risk of meticillin resistant Staphylococcus aureus and Clostridium difficile in patients with a documented penicillin allergy: Population based matched cohort study. BMJ 2018, 361, k2400. [Google Scholar] [CrossRef]
- Zaura, E.; Brandt, B.W.; Teixeira de Mattos, M.J.; Buijs, M.J.; Caspers, M.P.; Rashid, M.U.; Weintraub, A.; Nord, C.E.; Savell, A.; Hu, Y.; et al. Same Exposure but Two Radically Different Responses to Antibiotics: Resilience of the Salivary Microbiome versus Long-Term Microbial Shifts in Feces. mBio 2015, 6, e01693-15. [Google Scholar] [CrossRef] [PubMed]
- Helmen, Z.M.; Helm, M.C.; Helm, J.H.; Nielsen, A.; Kindel, T.; Higgins, R.; Gould, J.C. Predictors of Postoperative Urinary Tract Infection After Bariatric Surgery. Obes. Surg. 2018, 28, 1950–1954. [Google Scholar] [CrossRef] [PubMed]
- Pichichero, M.E.; Zagursky, R. Penicillin and cephalosporin allergy. Ann. Allergy Asthma Immunol. 2014, 112, 404–412. [Google Scholar] [CrossRef] [PubMed]
- Macy, E.; Contreras, R. Adverse reactions associated with oral and parenteral use of cephalosporins: A retrospective population-based analysis. J. Allergy Clin. Immunol. 2015, 135, 745–752.e5. [Google Scholar] [CrossRef]
- Shenoy, E.S.; Macy, E.; Rowe, T.; Blumenthal, K.G. Evaluation and Management of Penicillin Allergy: A Review. JAMA 2019, 321, 188–199. [Google Scholar] [CrossRef]
- Picard, M.; Robitaille, G.; Karam, F.; Daigle, J.M.; Bedard, F.; Biron, E.; Tardif, M.R.; Lacombe-Barrios, J.; Begin, P. Cross-Reactivity to Cephalosporins and Carbapenems in Penicillin-Allergic Patients: Two Systematic Reviews and Meta-Analyses. J. Allergy Clin. Immunol. Pract. 2019, 7, 2722–2738.e5. [Google Scholar] [CrossRef]
- Khan, D.A.; Banerji, A.; Bernstein, J.A.; Bilgicer, B.; Blumenthal, K.; Castells, M.; Ein, D.; Lang, D.M.; Phillips, E. Cephalosporin Allergy: Current Understanding and Future Challenges. J. Allergy Clin. Immunol. Pract. 2019, 7, 2105–2114. [Google Scholar] [CrossRef]
- Khan, D.A.; Banerji, A.; Blumenthal, K.G.; Phillips, E.J.; Solensky, R.; White, A.A.; Bernstein, J.A.; Chu, D.K.; Ellis, A.K.; Golden, D.B.K.; et al. Drug allergy: A 2022 practice parameter update. J. Allergy Clin. Immunol. 2022, 150, 1333–1393. [Google Scholar] [CrossRef]
- Uyttebroek, A.P.; Decuyper, I.I.; Bridts, C.H.; Romano, A.; Hagendorens, M.M.; Ebo, D.G.; Sabato, V. Cefazolin Hypersensitivity: Toward Optimized Diagnosis. J. Allergy Clin. Immunol. Pract. 2016, 4, 1232–1236. [Google Scholar] [CrossRef] [PubMed]
- Sousa-Pinto, B.; Blumenthal, K.G.; Courtney, L.; Mancini, C.M.; Jeffres, M.N. Assessment of the Frequency of Dual Allergy to Penicillins and Cefazolin: A Systematic Review and Meta-analysis. JAMA Surg. 2021, 156, e210021. [Google Scholar] [CrossRef] [PubMed]
- Goodman, E.J.; Morgan, M.J.; Johnson, P.A.; Nichols, B.A.; Denk, N.; Gold, B.B. Cephalosporins can be given to penicillin-allergic patients who do not exhibit an anaphylactic response. J. Clin. Anesth. 2001, 13, 561–564. [Google Scholar] [CrossRef] [PubMed]
- Cooper, J.J.; Atluri, V.L.; Jain, R.; Pottinger, P.S.; Coleman, D.T. Safety of cefazolin for perioperative prophylaxis in patients with penicillin allergy labels. Ann. Allergy Asthma Immunol. 2022, 129, 115–117. [Google Scholar] [CrossRef] [PubMed]
- Kurcz, B.P.; Allan, D.G.; Nestler, A.J.; Lister, E.L.; Yarnell, W.C.; Delfino, K.R.; Thuppal, S.; Horberg, J.V. Documented Penicillin Allergies Should Not Preclude Use of Preoperative Cefazolin in Hip and Knee Arthroplasty. J. Am. Acad. Orthop. Surg. 2023, 31, e107–e117. [Google Scholar] [CrossRef] [PubMed]
- Beltran, R.J.; Kako, H.; Chovanec, T.; Ramesh, A.; Bissonnette, B.; Tobias, J.D. Penicillin allergy and surgical prophylaxis: Cephalosporin cross-reactivity risk in a pediatric tertiary care center. J. Pediatr. Surg. 2015, 50, 856–859. [Google Scholar] [CrossRef] [PubMed]
- McDanel, D.L.; Azar, A.E.; Dowden, A.M.; Murray-Bainer, S.; Noiseux, N.O.; Willenborg, M.; Clark, C.R.; Callaghan, J.J.; Haleem, A. Screening for Beta-Lactam Allergy in Joint Arthroplasty Patients to Improve Surgical Prophylaxis Practice. J. Arthroplast. 2017, 32, S101–S108. [Google Scholar] [CrossRef]
- Wyles, C.C.; Hevesi, M.; Osmon, D.R.; Park, M.A.; Habermann, E.B.; Lewallen, D.G.; Berry, D.J.; Sierra, R.J. 2019 John Charnley Award: Increased risk of prosthetic joint infection following primary total knee and hip arthroplasty with the use of alternative antibiotics to cefazolin: The value of allergy testing for antibiotic prophylaxis. Bone Jt. J. 2019, 101-B, 9–15. [Google Scholar] [CrossRef]
- Plager, J.H.; Mancini, C.M.; Fu, X.; Melnitchouk, S.; Shenoy, E.S.; Banerji, A.; Collier, L.; Chaudhary, N.; Yerneni, S.; Zhang, Y.; et al. Preoperative penicillin allergy testing in patients undergoing cardiac surgery. Ann. Allergy Asthma Immunol. 2020, 124, 583–588. [Google Scholar] [CrossRef]
- Moussa, Y.; Shuster, J.; Matte, G.; Sullivan, A.; Goldstein, R.H.; Cunningham, D.; Ben-Shoshan, M.; Baldini, G.; Carli, F.; Tsoukas, C. De-labeling of beta-lactam allergy reduces intraoperative time and optimizes choice in antibiotic prophylaxis. Surgery 2018, 164, 117–123. [Google Scholar] [CrossRef]
- Lozo, S.; Wagner, D.; Shah, N.; Goldberg, R.; Gafni-Kane, A.; Solomonides, A. Should Penicillin Allergy Testing Be Included as Part of Preoperative Testing? J. Healthc. Qual. 2023, 45, 255–260. [Google Scholar] [CrossRef]
- Wade, S.; Marshall, E. A pharmacist-led penicillin allergy de-labelling project within a preoperative assessment clinic: The low-hanging fruit is within reach. J. Hosp. Infect. 2023, 139, 1–5. [Google Scholar] [CrossRef]
- Vaisman, A.; McCready, J.; Hicks, S.; Powis, J. Optimizing preoperative prophylaxis in patients with reported beta-lactam allergy: A novel extension of antimicrobial stewardship. J. Antimicrob. Chemother. 2017, 72, 2657–2660. [Google Scholar] [CrossRef]
- Jones, R.; Quartuccio, K.S.; Stern, J.L.; Heintz, E.V.; Pillinger, K.E.; Myers, T.G. Antibiotic Stewardship Interventions Improve Choice of Antibiotic Prophylaxis in Total Joint Arthroplasty in Patients with Reported Penicillin Allergies. Clin. Orthop. Relat. Res. 2021, 479, 1484–1494. [Google Scholar] [CrossRef]
- Kuruvilla, M.; Sexton, M.; Wiley, Z.; Langfitt, T.; Lynde, G.C.; Wolf, F. A Streamlined Approach to Optimize Perioperative Antibiotic Prophylaxis in the Setting of Penicillin Allergy Labels. J. Allergy Clin. Immunol. Pract. 2020, 8, 1316–1322. [Google Scholar] [CrossRef]
- Lam, P.W.; Tarighi, P.; Elligsen, M.; Nathens, A.B.; Riegert, D.; Tarshis, J.; Leis, J.A. Impact of the Allergy Clarification for Cefazolin Evidence-based Prescribing Tool on Receipt of Preferred Perioperative Prophylaxis: An Interrupted Time Series Study. Clin. Infect. Dis. 2020, 71, 2955–2957. [Google Scholar] [CrossRef] [PubMed]
- Grant, J.M.; Song, W.H.C.; Shajari, S.; Mak, R.; Meikle, A.T.; Partovi, N.; Masri, B.A.; Lau, T.T.Y. Safety of administering cefazolin versus other antibiotics in penicillin-allergic patients for surgical prophylaxis at a major Canadian teaching hospital. Surgery 2021, 170, 783–789. [Google Scholar] [CrossRef] [PubMed]
- Collins, C.D.; Scheidel, C.; Anam, K.; Polega, S.; Malani, A.N.; Hayward, A.; Leo, H.L.; Shankar, T.; Morrin, C.; Brockhaus, K. Impact of an Antibiotic Side-Chain-Based Cross-reactivity Chart Combined With Enhanced Allergy Assessment Processes for Surgical Prophylaxis Antimicrobials in Patients With beta-Lactam Allergies. Clin. Infect. Dis. 2021, 72, 1404–1412. [Google Scholar] [CrossRef] [PubMed]
- Kwiatkowski, S.; Mulugeta, S.; Davis, S.; Kenney, R.; Kalus, J.; Walton, L.; Patel, N. Optimizing preoperative antibiotics in patients with beta-lactam allergies: A role for pharmacy. Am. J. Health Syst. Pharm. 2021, 78, S76–S82. [Google Scholar] [CrossRef] [PubMed]
- Stonerock, D.; Hallo-Carrasco, A.; Edwards, M.; Porter, S.B.; Epps, K.L.; Gonzalez-Estrada, A. Pharmacist-led improvement in perioperative antibiotic selection for patients with a penicillin allergy label. Am. J. Health Syst. Pharm. 2023, 80, e111–e118. [Google Scholar] [CrossRef] [PubMed]
- Sexton, M.E.; Kuruvilla, M.E.; Wolf, F.A.; Lynde, G.C.; Wiley, Z. Anatomy of a successful stewardship intervention: Improving perioperative prescribing in penicillin-allergic patients. Infect. Control Hosp. Epidemiol. 2022, 43, 1101–1107. [Google Scholar] [CrossRef] [PubMed]
- Ramsey, A. Penicillin Allergy and Perioperative Anaphylaxis. Front. Allergy 2022, 3, 903161. [Google Scholar] [CrossRef] [PubMed]
- Savic, L.; Thomas, C.; Fallaha, D.; Wilson, M.; Hopkins, P.M.; Savic, S.; Clark, S.H.; The RAFT Group. DALES, Drug Allergy Labels in Elective Surgical patients: A prospective multicentre cross-sectional study of incidence, risks, and attitudes in penicillin de-labelling strategies. Br. J. Anaesth. 2020, 125, 962–969. [Google Scholar] [CrossRef] [PubMed]
- VanderVelde, K.A.; Suppes, S.L.; Gibbs, K.A.; Latz, K.H.; Vanderpool, A.C.; El Feghaly, R.E.; Goldman, J.L. Increasing cefazolin use for surgical prophylaxis in penicillin-allergy-labeled patients. Antimicrob. Steward. Healthc. Epidemiol. 2023, 3, e11. [Google Scholar] [CrossRef] [PubMed]
- Isserman, R.S.; Cheung, J.; Varallo, D.; Cafone, J.; Lee, J.; Chiotos, K.; Muhly, W.T.; Metjian, T.A.; Swami, S.; Baldwin, K.; et al. Increasing Cefazolin Use for Perioperative Antibiotic Prophylaxis in Penicillin-Allergic Children. Pediatrics 2022, 149, e2021050694. [Google Scholar] [CrossRef] [PubMed]
- Macy, E.; McCormick, T.A.; Adams, J.L.; Crawford, W.W.; Nguyen, M.T.; Hoang, L.; Eng, V.; Davis, A.C.; McGlynn, E.A. Association Between Removal of a Warning Against Cephalosporin Use in Patients with Penicillin Allergy and Antibiotic Prescribing. JAMA Netw. Open 2021, 4, e218367. [Google Scholar] [CrossRef] [PubMed]
- Romano, A.; Gaeta, F.; Valluzzi, R.L.; Maggioletti, M.; Zaffiro, A.; Caruso, C.; Quaratino, D. IgE-mediated hypersensitivity to cephalosporins: Cross-reactivity and tolerability of alternative cephalosporins. J. Allergy Clin. Immunol. 2015, 136, 685–691.e3. [Google Scholar] [CrossRef] [PubMed]
- Pedersen, K.; van Schalkwyk, J.; Brewerton, M.; Jordan, A.; Cooke, P. Penicillin cross-sensitivity in patients with confirmed peri-operative allergic hypersensitivity reactions to cefazolin: A retrospective observational study. Anaesthesia 2023, 79, 208–210. [Google Scholar] [CrossRef]
- Liu, M.Y.; Challa, M.; McCoul, E.D.; Chen, P.G. Economic Viability of Penicillin Allergy Testing to Avoid Improper Clindamycin Surgical Prophylaxis. Laryngoscope 2023, 133, 1086–1091. [Google Scholar] [CrossRef]
- Mancini, C.M.; Fu, X.; Zhang, Y.; Kuper, K.; Schulz, L.T.; Bhowmick, T.; Postelnick, M.; Lee, F.; Walensky, R.P.; Blumenthal, K.G. Penicillin Allergy Evaluation Access: A National Survey. Clin. Infect. Dis. 2020, 71, 2972–2975. [Google Scholar] [CrossRef]
- Trubiano, J.A.; Chua, K.Y.L.; Holmes, N.E.; Douglas, A.P.; Mouhtouris, E.; Goh, M.; Phillips, E.J. Safety of cephalosporins in penicillin class severe delayed hypersensitivity reactions. J. Allergy Clin. Immunol. Pract. 2020, 8, 1142–1146.e4. [Google Scholar] [CrossRef] [PubMed]
- Blumenthal, K.G.; Youngster, I.; Shenoy, E.S.; Banerji, A.; Nelson, S.B. Tolerability of cefazolin after immune-mediated hypersensitivity reactions to nafcillin in the outpatient setting. Antimicrob. Agents Chemother. 2014, 58, 3137–3143. [Google Scholar] [CrossRef] [PubMed]
| Perioperative Prophylaxis Option | Advantages of Utilization | Challenges with Utilization |
|---|---|---|
| Cefazolin |
|
|
| Vancomycin |
|
|
| Clindamycin |
|
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sexton, M.E.; Kuruvilla, M.E. Management of Penicillin Allergy in the Perioperative Setting. Antibiotics 2024, 13, 157. https://doi.org/10.3390/antibiotics13020157
Sexton ME, Kuruvilla ME. Management of Penicillin Allergy in the Perioperative Setting. Antibiotics. 2024; 13(2):157. https://doi.org/10.3390/antibiotics13020157
Chicago/Turabian StyleSexton, Mary Elizabeth, and Merin Elizabeth Kuruvilla. 2024. "Management of Penicillin Allergy in the Perioperative Setting" Antibiotics 13, no. 2: 157. https://doi.org/10.3390/antibiotics13020157
APA StyleSexton, M. E., & Kuruvilla, M. E. (2024). Management of Penicillin Allergy in the Perioperative Setting. Antibiotics, 13(2), 157. https://doi.org/10.3390/antibiotics13020157






