Occurrence of ESBL- and AmpC-Producing E. coli in French Griffon Vultures Feeding on Extensive Livestock Carcasses
Abstract
1. Introduction
2. Results
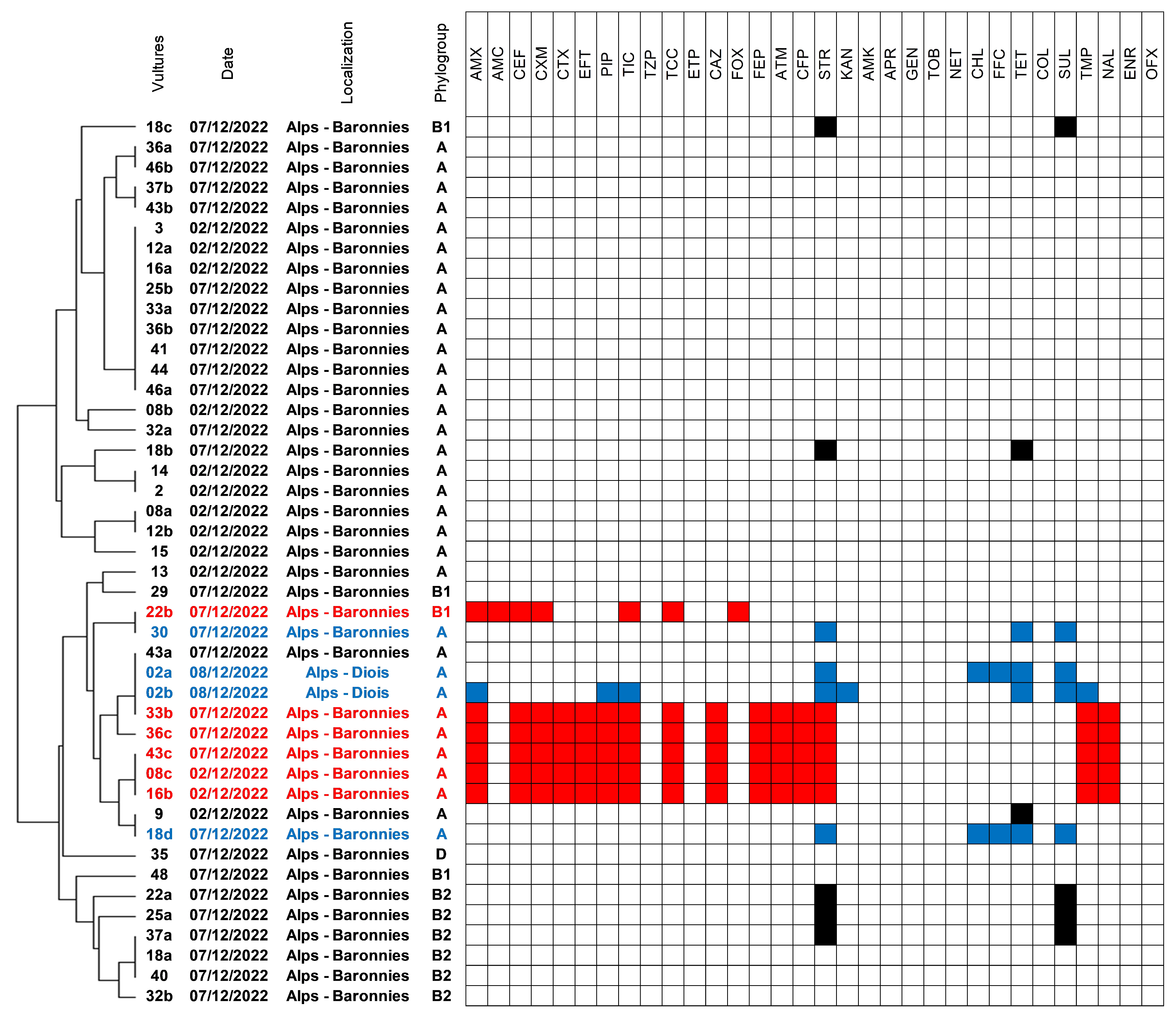
2.1. Isolation of E. coli and Genetic Diversity and Susceptibility Testing
2.2. Antimicrobial Susceptibility and Occurrence of ESBL/AmpC-Producing Isolates
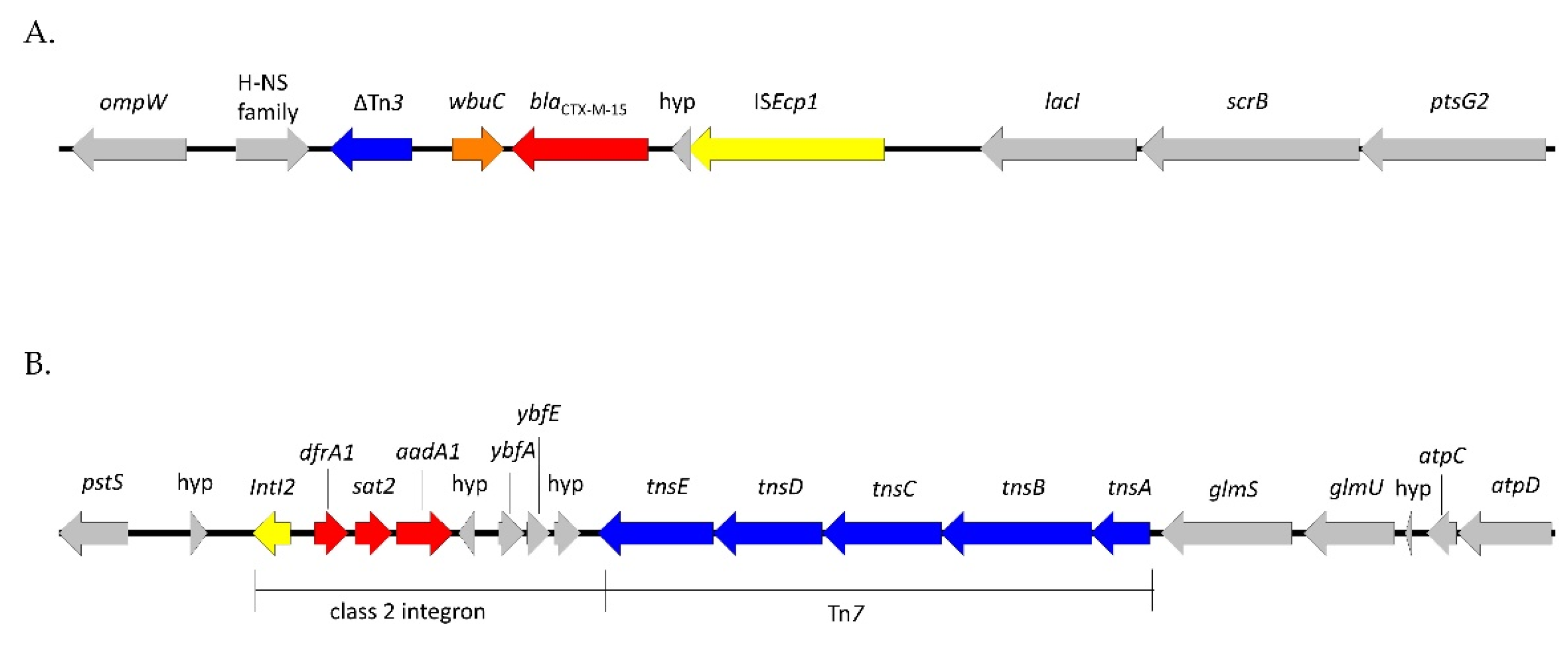
2.3. Characteristics of ESC-Resistant Isolates
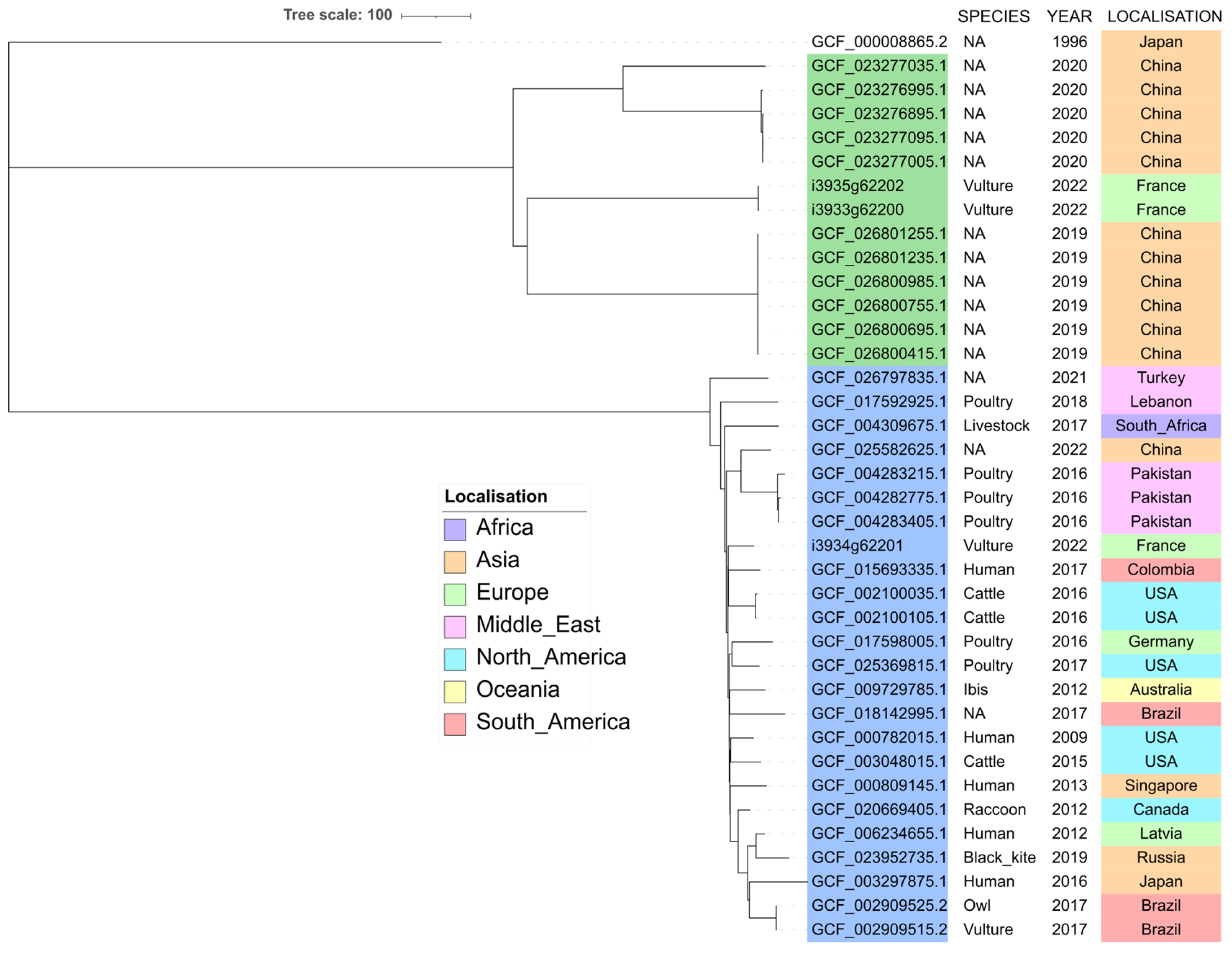
2.4. Phylogenetic Analysis of ST212 and ST3274 Isolates
3. Discussion
4. Materials and Methods
4.1. Ethical Approval
4.2. Wild Bird Sampling and Bacterial Isolation
4.3. Antimicrobial Susceptibility Testing
4.4. Illumina Short-Read Sequencing and Data Analyses
4.5. Phylogenetic Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Dolejska, M.; Literak, I. Wildlife is overlooked in the epidemiology of medically important antibiotic-resistant bacteria. Antimicrob. Agents Chemother. 2019, 63, e01167-19. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Ma, Z.B.; Zeng, Z.L.; Yang, X.W.; Huang, Y.; Liu, J.H. The role of wildlife (wild birds) in the global transmission of antimicrobial resistance genes. Zool. Res. 2017, 38, 55–80. [Google Scholar] [CrossRef] [PubMed]
- Brendecke, J.; Homeier-Bachmann, T.; Schmitz Ornés, A.; Guenther, S.; Heiden, S.E.; Schwabe, M.; Eger, E.; Schaufler, K. Multidrug-resistant high-risk Escherichia coli and Klebsiella pneumoniae clonal lineages occur in black-headed gulls from two conservation islands in Germany. Antibiotics 2022, 11, 1357. [Google Scholar] [CrossRef] [PubMed]
- Guenther, S.; Semmler, T.; Stubbe, A.; Stubbe, M.; Wieler, L.H.; Schaufler, K. Chromosomally encoded ESBL genes in Escherichia coli of ST38 from Mongolian wild birds. J. Antimicrob. Chemother. 2017, 72, 1289–1292. [Google Scholar] [CrossRef]
- Zendri, F.; Maciuca, I.E.; Moon, S.; Jones, P.H.; Wattret, A.; Jenkins, R.; Baxter, A.; Timofte, D. Occurrence of ESBL-producing Escherichia coli ST131, including the H30-Rx and C1-M27 subclones, among urban seagulls from the United Kingdom. Microb. Drug Resist. 2020, 26, 697–708. [Google Scholar] [CrossRef]
- Wirth, T.; Falush, D.; Lan, R.; Colles, F.; Mensa, P.; Wieler, L.H.; Karch, H.; Reeves, P.R.; Maiden, M.C.; Ochman, H.; et al. Sex and virulence in Escherichia coli: An evolutionary perspective. Mol. Microbiol. 2006, 60, 1136–1151. [Google Scholar] [CrossRef]
- Atterby, C.; Borjesson, S.; Ny, S.; Jarhult, J.D.; Byfors, S.; Bonnedahl, J. ESBL-producing Escherichia coli in Swedish gulls-A case of environmental pollution from humans? PLoS ONE 2017, 12, e0190380. [Google Scholar] [CrossRef]
- Bonnedahl, J.; Stedt, J.; Waldenstrom, J.; Svensson, L.; Drobni, M.; Olsen, B. Comparison of Extended-Spectrum beta-Lactamase (ESBL) CTX-M genotypes in Franklin gulls from Canada and Chile. PLoS ONE 2015, 10, e0141315. [Google Scholar] [CrossRef]
- Yang, H.; Rehman, M.U.; Zhang, S.; Yang, J.; Li, Y.; Gao, J.; Gu, L.; Wang, M.; Jia, R.; Chen, S.; et al. High prevalence of CTX-M belonging to ST410 and ST889 among ESBL producing E. coli isolates from waterfowl birds in China’s tropical island, Hainan. Acta Trop. 2019, 194, 30–35. [Google Scholar] [CrossRef]
- Bonnedahl, J.; Drobni, M.; Gauthier-Clerc, M.; Hernandez, J.; Granholm, S.; Kayser, Y.; Melhus, A.; Kahlmeter, G.; Waldenstrom, J.; Johansson, A.; et al. Dissemination of Escherichia coli with CTX-M type ESBL between humans and yellow-legged gulls in the south of France. PLoS ONE 2009, 4, e5958. [Google Scholar] [CrossRef]
- Dolejska, M.; Masarikova, M.; Dobiasova, H.; Jamborova, I.; Karpiskova, R.; Havlicek, M.; Carlile, N.; Priddel, D.; Cizek, A.; Literak, I. High prevalence of Salmonella and IMP-4-producing Enterobacteriaceae in the silver gull on Five Islands, Australia. J. Antimicrob. Chemother. 2016, 71, 63–70. [Google Scholar] [CrossRef][Green Version]
- Vittecoq, M.; Laurens, C.; Brazier, L.; Durand, P.; Elguero, E.; Arnal, A.; Thomas, F.; Aberkane, S.; Renaud, N.; Prugnolle, F.; et al. VIM-1 carbapenemase-producing Escherichia coli in gulls from southern France. Ecol. Evol. 2017, 7, 1224–1232. [Google Scholar] [CrossRef]
- Sun, F.; Chen, J.; Liu, K.; Tang, M.; Yang, Y. The avian gut microbiota: Diversity, influencing factors, and future directions. Front. Microbiol. 2022, 13, 934272. [Google Scholar] [CrossRef]
- Woksepp, H.; Camara, F.; Bonnedahl, J. High prevalence of blaCTX-M-15 type extended-spectrum beta-lactamases in Gambian hooded vultures (Necrosyrtes monachus): A threatened species with substantial human interaction. Microbiologyopen 2023, 12, e1349. [Google Scholar] [CrossRef]
- Carvalho, I.; Tejedor-Junco, M.T.; González-Martín, M.; Corbera, J.A.; Suárez-Pérez, A.; Silva, V.; Igrejas, G.; Torres, C.; Poeta, P. Molecular diversity of Extended-spectrum β-lactamase-producing Escherichia coli from vultures in Canary Islands. Environ. Microbiol. Rep. 2020, 12, 540–547. [Google Scholar] [CrossRef]
- Guenther, S.; Aschenbrenner, K.; Stamm, I.; Bethe, A.; Semmler, T.; Stubbe, A.; Stubbe, M.; Batsajkhan, N.; Glupczynski, Y.; Wieler, L.H.; et al. Comparable high rates of extended-spectrum-beta-lactamase-producing Escherichia coli in birds of prey from Germany and Mongolia. PLoS ONE 2012, 7, e53039. [Google Scholar] [CrossRef]
- Alcalá, L.; Alonso, C.A.; Simón, C.; González-Esteban, C.; Orós, J.; Rezusta, A.; Ortega, C.; Torres, C. Wild birds, frequent carriers of extended-spectrum β-lactamase (ESBL) producing Escherichia coli of CTX-M and SHV-12 types. Microb. Ecol. 2016, 72, 861–869. [Google Scholar] [CrossRef]
- Sevilla, E.; Marín, C.; Delgado-Blas, J.F.; González-Zorn, B.; Vega, S.; Kuijper, E.; Bolea, R.; Mainar-Jaime, R.C. Wild griffon vultures (Gyps fulvus) fed at supplementary feeding stations: Potential carriers of pig pathogens and pig-derived antimicrobial resistance? Transbound. Emerg. Dis. 2020, 67, 1295–1305. [Google Scholar] [CrossRef]
- Blanco, G.; López-Hernández, I.; Morinha, F.; López-Cerero, L. Intensive farming as a source of bacterial resistance to antimicrobial agents in sedentary and migratory vultures: Implications for local and transboundary spread. Sci. Total Environ. 2020, 739, 140356. [Google Scholar] [CrossRef]
- Espunyes, J.; Illera, L.; Dias-Alves, A.; Lobato, L.; Ribas, M.P.; Manzanares, A.; Ayats, T.; Marco, I.; Cerdà-Cuéllar, M. Eurasian griffon vultures carry widespread antimicrobial resistant Salmonella and Campylobacter of public health concern. Sci. Total Environ. 2022, 844, 157189. [Google Scholar] [CrossRef]
- Fluhr, J.; Benhamou, S.; Peyrusque, D.; Duriez, O. Space use and time budget in two populations of Griffon vultures in contrasting landscapes. J. Raptor Res. 2021, 55, 425–437. [Google Scholar] [CrossRef]
- Olivier, D. Commentary: Not all vulture feeding stations are supplementary—Proposed terminology for carcass provisioning with reference to management goals and food sources. J. Raptor Res. 2022, 56, 131–137. [Google Scholar] [CrossRef]
- Mahmud, B.; Wallace, M.A.; Reske, K.A.; Alvarado, K.; Muenks, C.E.; Rasmussen, D.A.; Burnham, C.-A.D.; Lanzas, C.; Dubberke, E.R.; Dantas, G. Epidemiology of plasmid lineages mediating the spread of extended-spectrum beta-lactamases among clinical Escherichia coli. Msystems 2022, 7, e00519-22. [Google Scholar] [CrossRef] [PubMed]
- Ben Yahia, H.; Ben Sallem, R.; Tayh, G.; Klibi, N.; Ben Amor, I.; Gharsa, H.; Boudabbous, A.; Ben Slama, K. Detection of CTX-M-15 harboring Escherichia coli isolated from wild birds in Tunisia. BMC Microbiol. 2018, 18, 26. [Google Scholar] [CrossRef]
- He, J.; Li, C.; Cui, P.; Wang, H. Detection of Tn7-like transposons and antibiotic resistance in Enterobacterales from animals used for food production with identification of three novel transposons Tn6813, Tn6814, and Tn6765. Front. Microbiol. 2020, 11, 2049. [Google Scholar] [CrossRef]
- de Carvalho, M.P.N.; Fernandes, M.R.; Sellera, F.P.; Lopes, R.; Monte, D.F.; Hippólito, A.G.; Milanelo, L.; Raso, T.F.; Lincopan, N. International clones of extended-spectrum β-lactamase (CTX-M)-producing Escherichia coli in peri-urban wild animals, Brazil. Transbound. Emerg. Dis. 2020, 67, 1804–1815. [Google Scholar] [CrossRef]
- Guenther, S.; Wuttke, J.; Bethe, A.; Vojtech, J.; Schaufler, K.; Semmler, T.; Ulrich, R.G.; Wieler, L.H.; Ewers, C. Is fecal carriage of extended-spectrum-beta-lactamase-producing Escherichia coli in urban rats a risk for public health? Antimicrob. Agents Chemother. 2013, 57, 2424–2425. [Google Scholar] [CrossRef]
- Arrondo, E.; Sanz-Aguilar, A.; Pérez-García, J.M.; Cortés-Avizanda, A.; Sánchez-Zapata, J.A.; Donázar, J.A. Landscape anthropization shapes the survival of a top avian scavenger. Biodivers. Conserv. 2020, 29, 1411–1425. [Google Scholar] [CrossRef]
- FernÁNdez-GÓMez, L.; CortÉS-Avizanda, A.; Arrondo, E.; GarcÍA-Alfonso, M.; Ceballos, O.; MontelÍO, E.; DonÁZar, J.A. Vultures feeding on the dark side: Current sanitary regulations may not be enough. Bird Conserv. Int. 2022, 32, 590–608. [Google Scholar] [CrossRef]
- Gangoso, L.; Cortés-Avizanda, A.; Sergiel, A.; Pudifoot, B.; Miranda, F.; Muñoz, J.; Delgado-González, A.; Moleón, M.; Sánchez-Zapata, J.A.; Arrondo, E.; et al. Avian scavengers living in anthropized landscapes have shorter telomeres and higher levels of glucocorticoid hormones. Sci. Total Environ. 2021, 782, 146920. [Google Scholar] [CrossRef]
- Jamborova, I.; Janecko, N.; Halova, D.; Sedmik, J.; Mezerova, K.; Papousek, I.; Kutilova, I.; Dolejska, M.; Cizek, A.; Literak, I. Molecular characterization of plasmid-mediated AmpC beta-lactamase- and extended-spectrum beta-lactamase-producing Escherichia coli and Klebsiella pneumoniae among corvids (Corvus brachyrhynchos and Corvus corax) roosting in Canada. FEMS Microbiol. Ecol. 2018, 94, fiy166. [Google Scholar] [CrossRef]
- Foster-Nyarko, E.; Alikhan, N.F.; Ravi, A.; Thomson, N.M.; Jarju, S.; Kwambana-Adams, B.A.; Secka, A.; O’Grady, J.; Antonio, M.; Pallen, M.J. Genomic diversity of Escherichia coli isolates from backyard chickens and guinea fowl in the Gambia. Microb. Genom. 2021, 7, mgen000484. [Google Scholar] [CrossRef]
- Wyrsch, E.R.; Chowdhury, P.R.; Wallis, L.; Cummins, M.L.; Zingali, T.; Brandis, K.J.; Djordjevic, S.P. Whole-genome sequence analysis of environmental Escherichia coli from the faeces of straw-necked ibis (Threskiornis spinicollis) nesting on inland wetlands. Microb. Genom. 2020, 6, e000385. [Google Scholar] [CrossRef]
- Abdallah, R.; Kuete Yimagou, E.; Hadjadj, L.; Mediannikov, O.; Ibrahim, A.; Davoust, B.; Barciela, A.; Hernandez-Aguilar, R.A.; Diatta, G.; Sokhna, C.; et al. Population diversity of antibiotic resistant Enterobacterales in samples from wildlife origin in Senegal: Identification of a multidrug resistance transposon carrying blaCTX–M–15 in Escherichia coli. Front. Microbiol. 2022, 13, 838392. [Google Scholar] [CrossRef]
- Savin, M.; Bierbaum, G.; Hammerl, J.A.; Heinemann, C.; Parcina, M.; Sib, E.; Voigt, A.; Kreyenschmidt, J. ESKAPE bacteria and extended-spectrum-beta-lactamase-producing Escherichia coli isolated from wastewater and process water from German poultry slaughterhouses. Appl. Environ. Microbiol. 2020, 86, e02748-19. [Google Scholar] [CrossRef]
- Le Gouar, P.; Robert, A.; Choisy, J.-P.; Henriquet, S.; Lecuyer, P.; Tessier, C.; Sarrazin, F. Roles of survival and dispersal in reintroduction success of Griffon vulture (Gyps fulvus). Ecol. Appl. 2008, 18, 859–872. [Google Scholar] [CrossRef]
- Duriez, O., (CEFE, Montpellier, France). GPS Telemetry Data. 2021; (Unpublished work). [Google Scholar]
- Duriez, O., (CEFE, Montpellier, France); Gregory, K., (CEFE, Montpellier, France); Bharadwaj, A., (CEFE, Montpellier, France). Quantification of food resources of griffon vultures in Causses and French Alps. 2022; manuscript in preparation. [Google Scholar]
- Duriez, O.; Herman, S.; Sarrazin, F. Intra-specific competition in foraging Griffon Vultures Gyps fulvus: 2. The influence of supplementary feeding management. Bird Study 2012, 59, 193–206. [Google Scholar] [CrossRef]
- Duriez, O.; Harel, R.; Hatzofe, O. Studying movement of avian scavengers to understand carrion ecology. In Carrion Ecology and Management; Olea, P.P., Mateo-Tomás, P., Sánchez-Zapata, J.A., Eds.; Springer International Publishing: Cham, Switzerland, 2019; pp. 255–274. [Google Scholar]
- Doumith, M.; Day, M.J.; Hope, R.; Wain, J.; Woodford, N. Improved multiplex PCR strategy for rapid assignment of the four major Escherichia coli phylogenetic groups. J. Clin. Microbiol. 2012, 50, 3108–3110. [Google Scholar] [CrossRef]
- Caméléna, F.; Birgy, A.; Smail, Y.; Courroux, C.; Mariani-Kurkdjian, P.; Le Hello, S.; Bonacorsi, S.; Bidet, P. Rapid and simple universal Escherichia coli genotyping method based on multiple-locus variable-number tandem-repeat analysis using single-tube multiplex PCR and standard gel electrophoresis. Appl. Environ. Microbiol. 2019, 85, e02812-18. [Google Scholar] [CrossRef]
| ID | Species | ST | ESBL Resistance Genes | AmpC Mutations | Additional Resistance Genes | Plasmid Content | Genetic Support of the ESBL/AmpC Genes | Total Length | No. Contigs | N50 | Coverage |
|---|---|---|---|---|---|---|---|---|---|---|---|
| 33b | E.coli | 3274 | CTX-M-15 | − | aadA1, dfrA1 | ColpVC, IncX1 | chromosome | 4762230 | 117 | 107,779 | 377 |
| 22b | E.coli | 212 | − | AmpC-promoter (g.-42C>T) | none | Col156, IncFIB | chromosome | 4900458 | 99 | 192,556 | 399 |
| 16b | E.coli | 3274 | CTX-M-15 | − | aadA1, dfrA1 | ColpVC, IncX1 | chromosome | 4762328 | 117 | 107,976 | 420 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Haenni, M.; Du Fraysseix, L.; François, P.; Drapeau, A.; Bralet, T.; Madec, J.-Y.; Boulinier, T.; Duriez, O. Occurrence of ESBL- and AmpC-Producing E. coli in French Griffon Vultures Feeding on Extensive Livestock Carcasses. Antibiotics 2023, 12, 1160. https://doi.org/10.3390/antibiotics12071160
Haenni M, Du Fraysseix L, François P, Drapeau A, Bralet T, Madec J-Y, Boulinier T, Duriez O. Occurrence of ESBL- and AmpC-Producing E. coli in French Griffon Vultures Feeding on Extensive Livestock Carcasses. Antibiotics. 2023; 12(7):1160. https://doi.org/10.3390/antibiotics12071160
Chicago/Turabian StyleHaenni, Marisa, Laetitia Du Fraysseix, Pauline François, Antoine Drapeau, Tristan Bralet, Jean-Yves Madec, Thierry Boulinier, and Olivier Duriez. 2023. "Occurrence of ESBL- and AmpC-Producing E. coli in French Griffon Vultures Feeding on Extensive Livestock Carcasses" Antibiotics 12, no. 7: 1160. https://doi.org/10.3390/antibiotics12071160
APA StyleHaenni, M., Du Fraysseix, L., François, P., Drapeau, A., Bralet, T., Madec, J.-Y., Boulinier, T., & Duriez, O. (2023). Occurrence of ESBL- and AmpC-Producing E. coli in French Griffon Vultures Feeding on Extensive Livestock Carcasses. Antibiotics, 12(7), 1160. https://doi.org/10.3390/antibiotics12071160







