Naturally Derived Malabaricone B as a Promising Bactericidal Candidate Targeting Multidrug-Resistant Staphylococcus aureus also Possess Synergistic Interactions with Clinical Antibiotics
Abstract
:1. Introduction
2. Results
2.1. Isolation of Malabaricones from the Fruit Rinds of M. malabarica Lam
2.2. Antibacterial Activity against Clinically Relevant Bacteria
2.3. NS-7 Imparted No Toxicity to Eukaryotic Cells
2.4. NS-7 Is Active against MDR Clinical Strains of Staphylococci and Enterococci
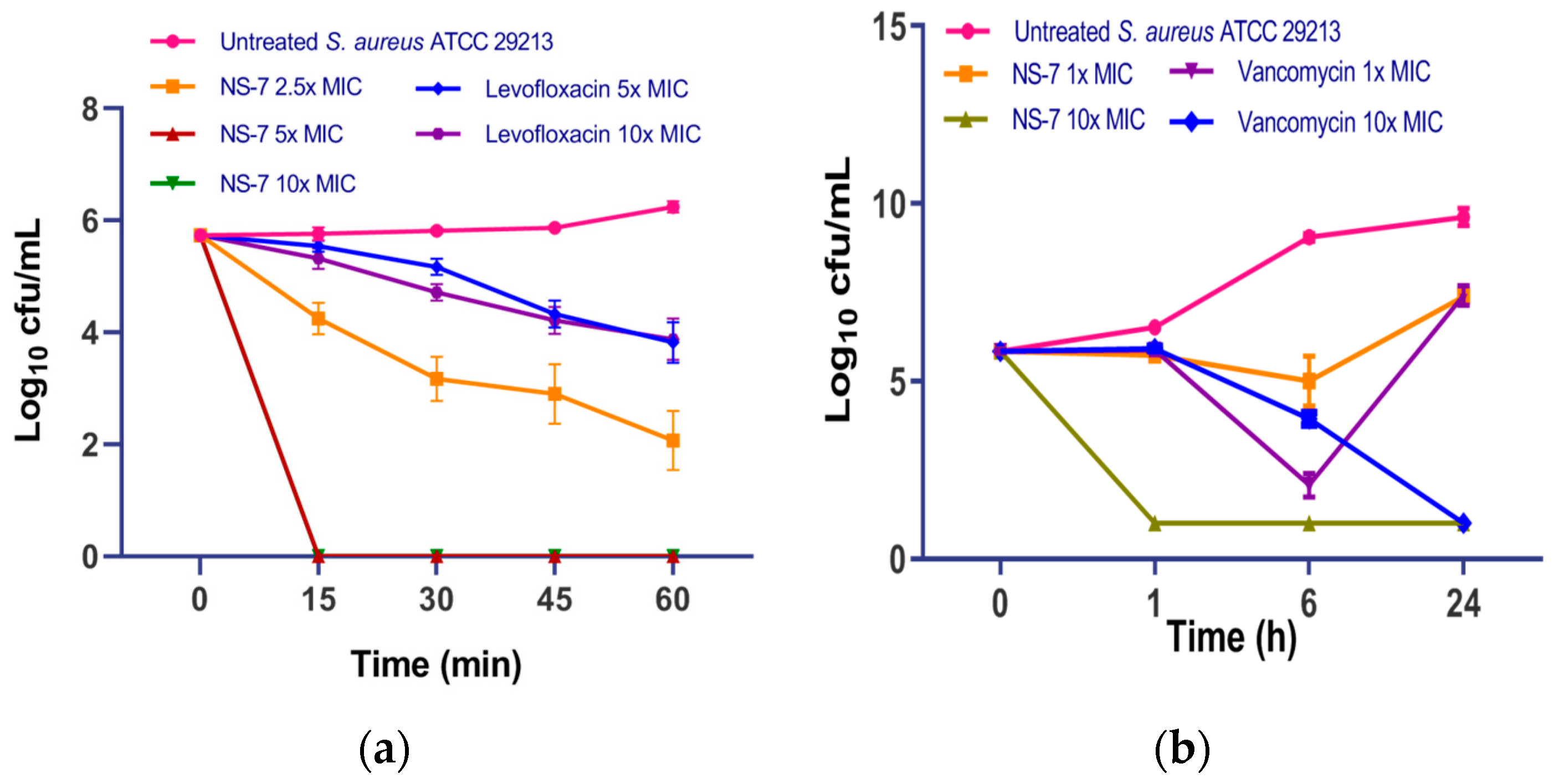
2.5. NS-7 Possesses Rapid Bactericidal Activity
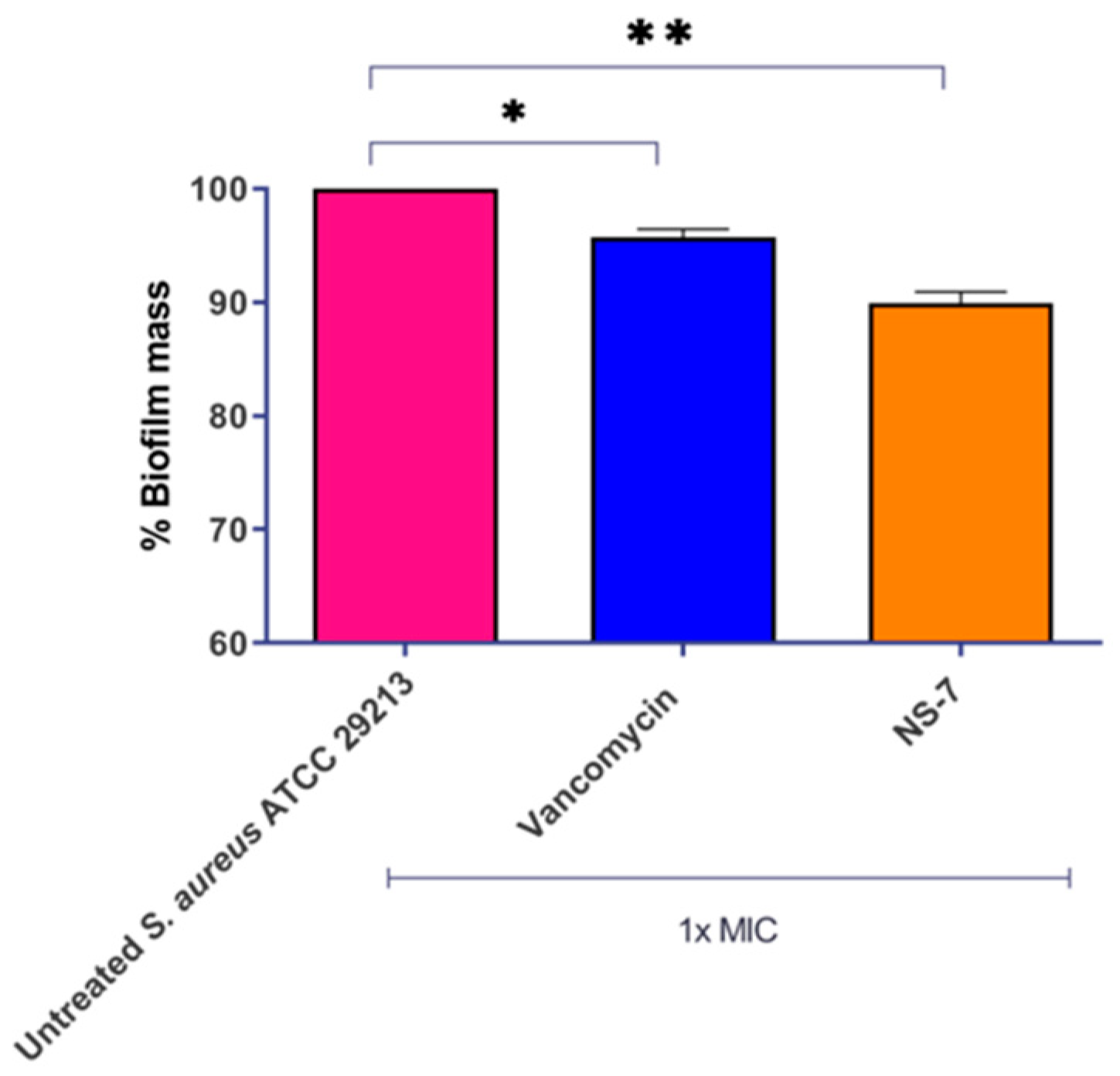
2.6. Efficient Eradication of Preformed S. aureus Biofilm by NS-7
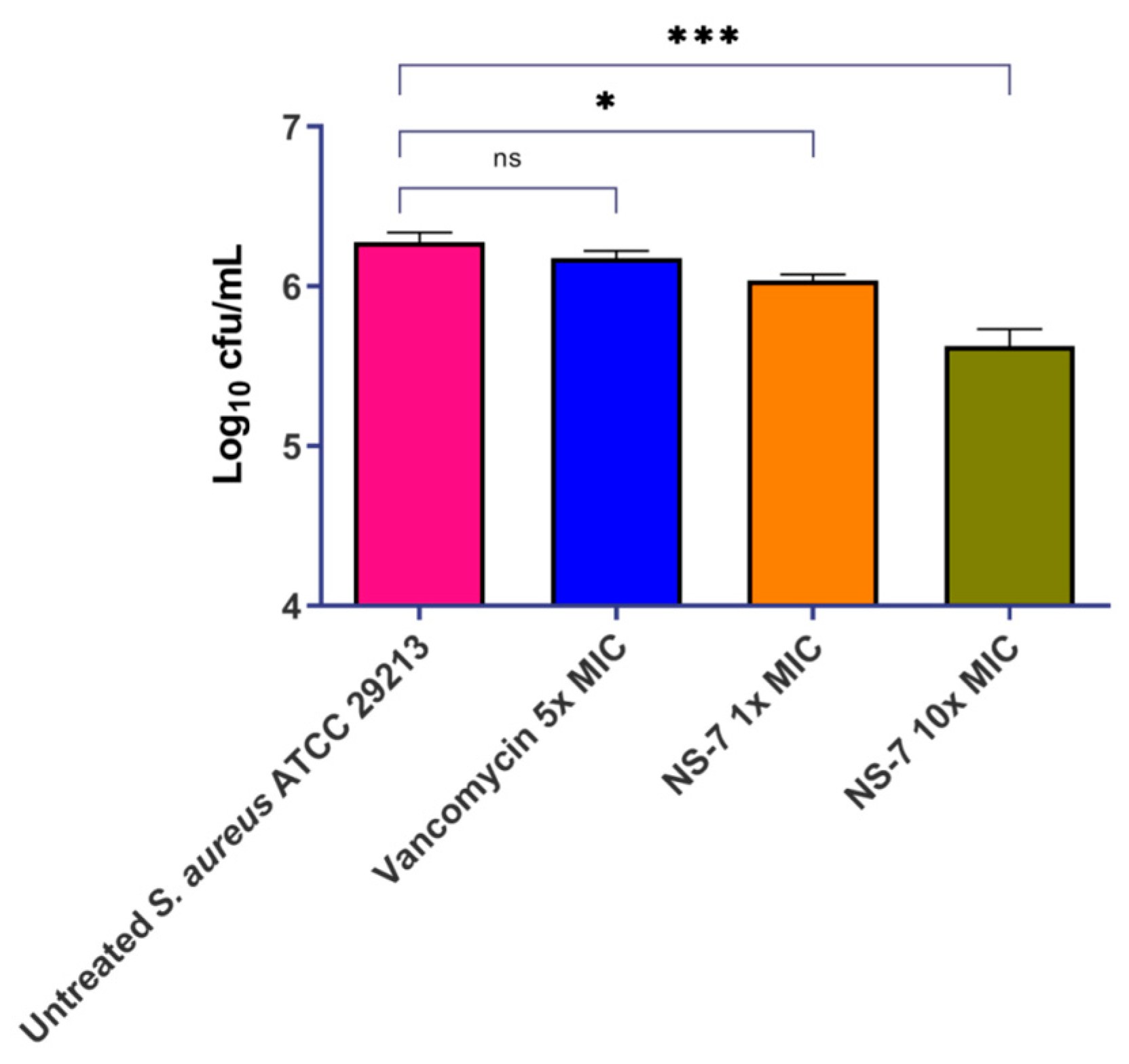
2.7. NS-7 Efficiently Cleared Intracellular S. aureus
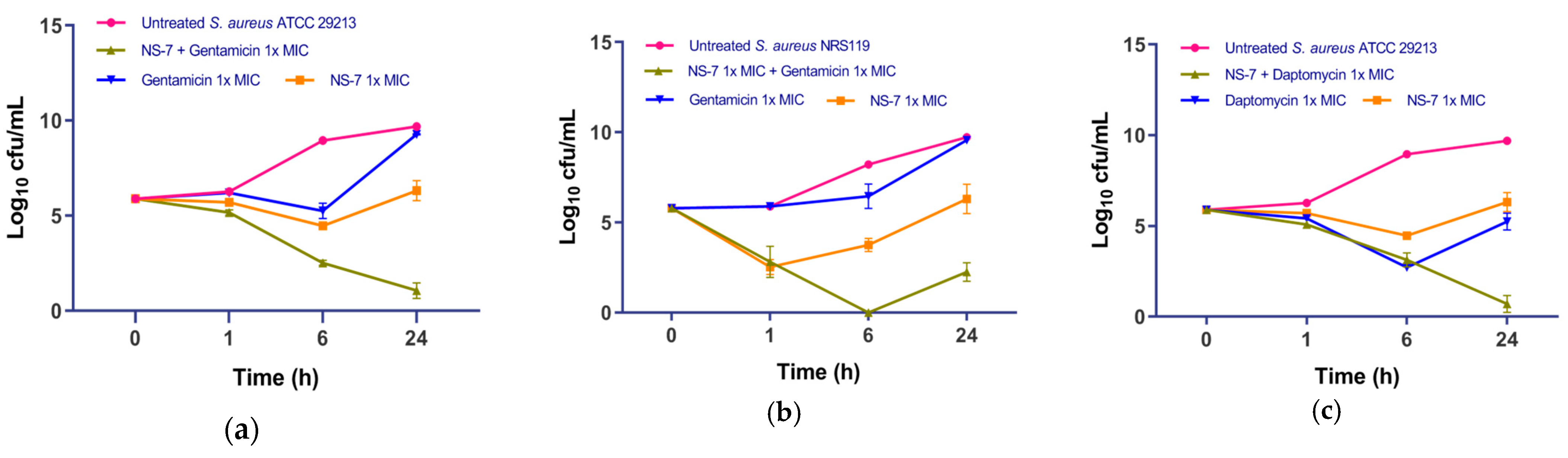
2.8. NS-7 Shows Synergism with Daptomycin and Gentamicin
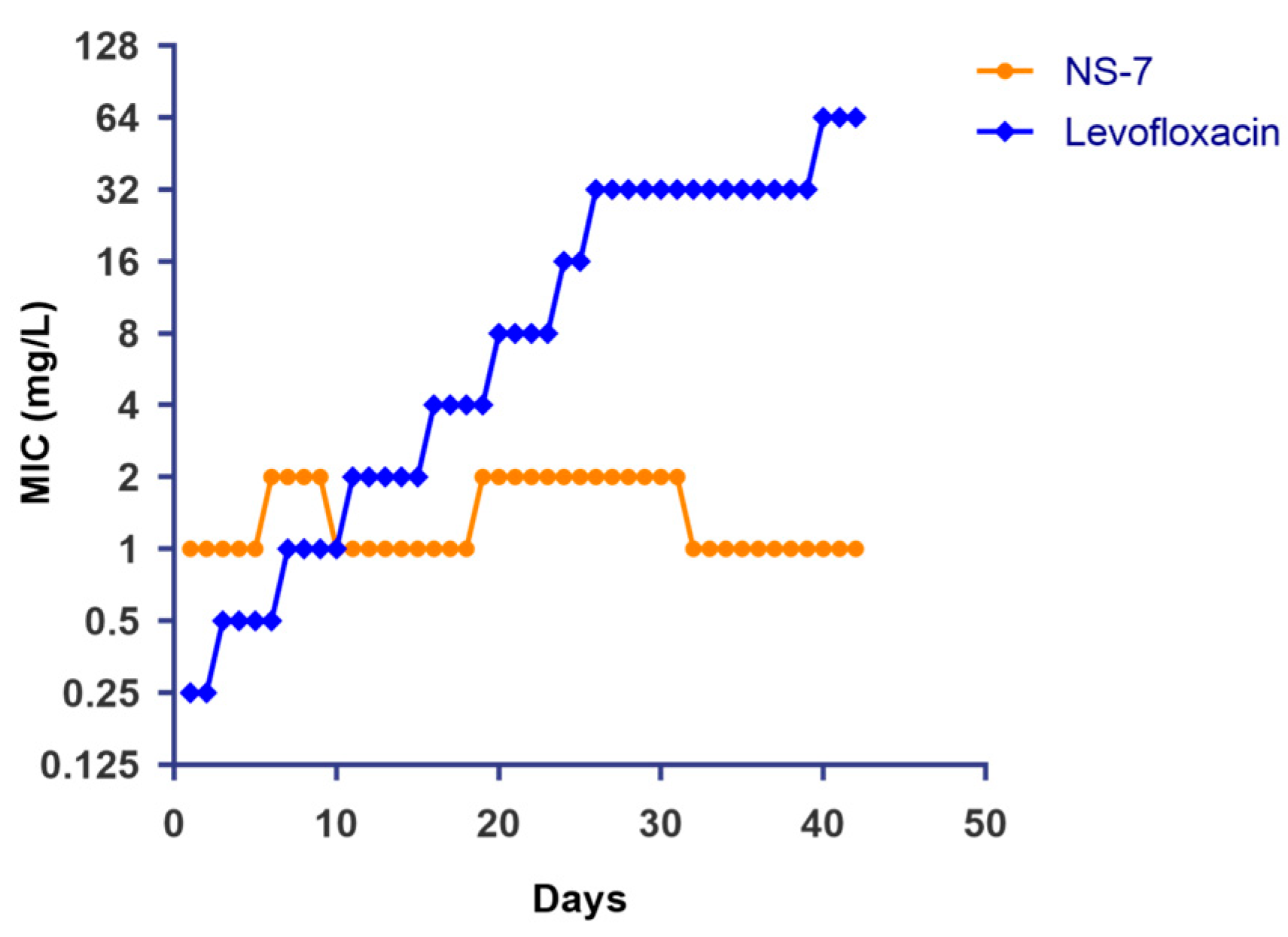
2.9. S. aureus Does Not Develop Resistance to NS-7
2.10. NS-7 Displayed Prolonged PAE against S. aureus
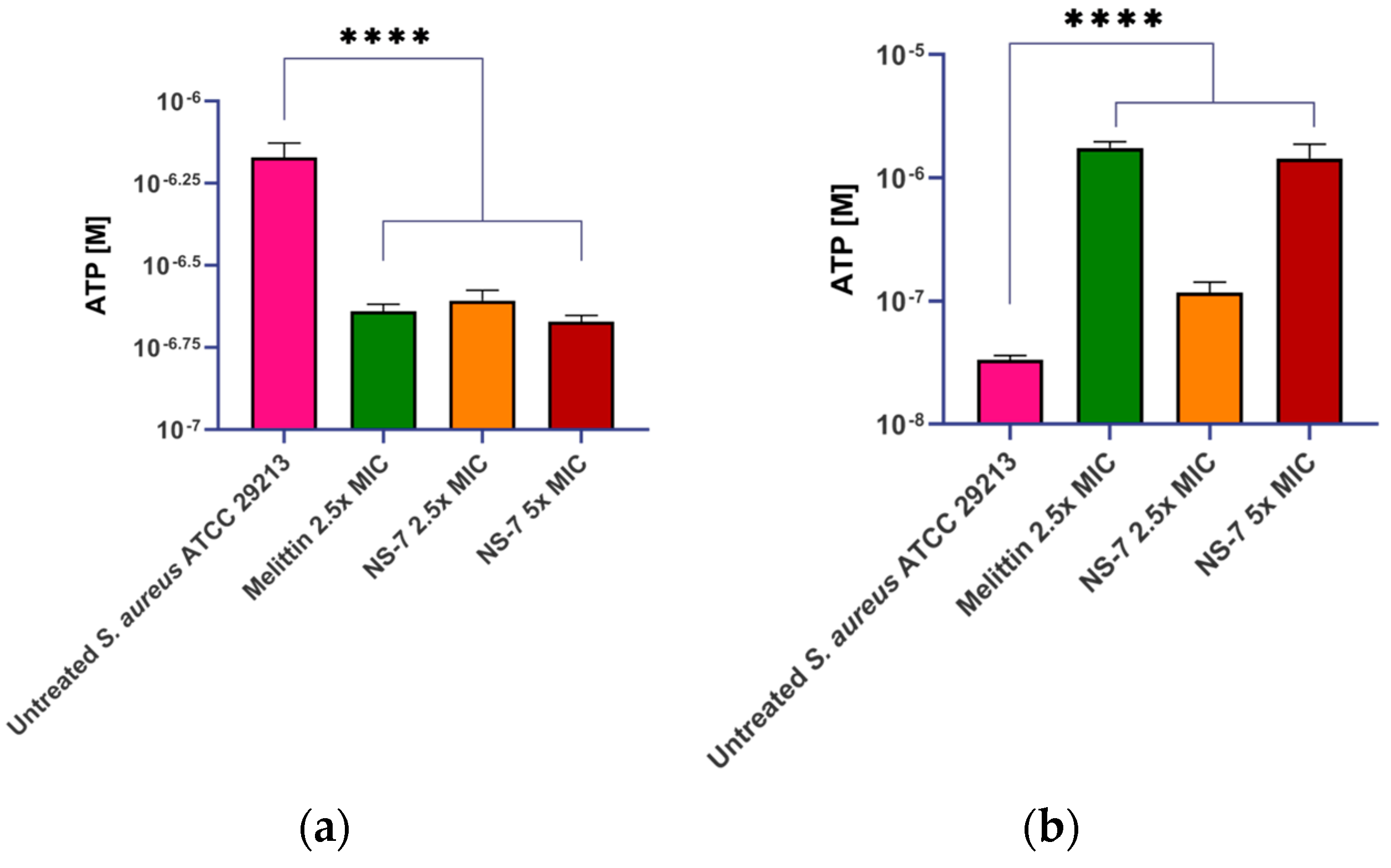
2.11. NS-7 Exerts Bactericidal Activity via Cell Membrane Lysis
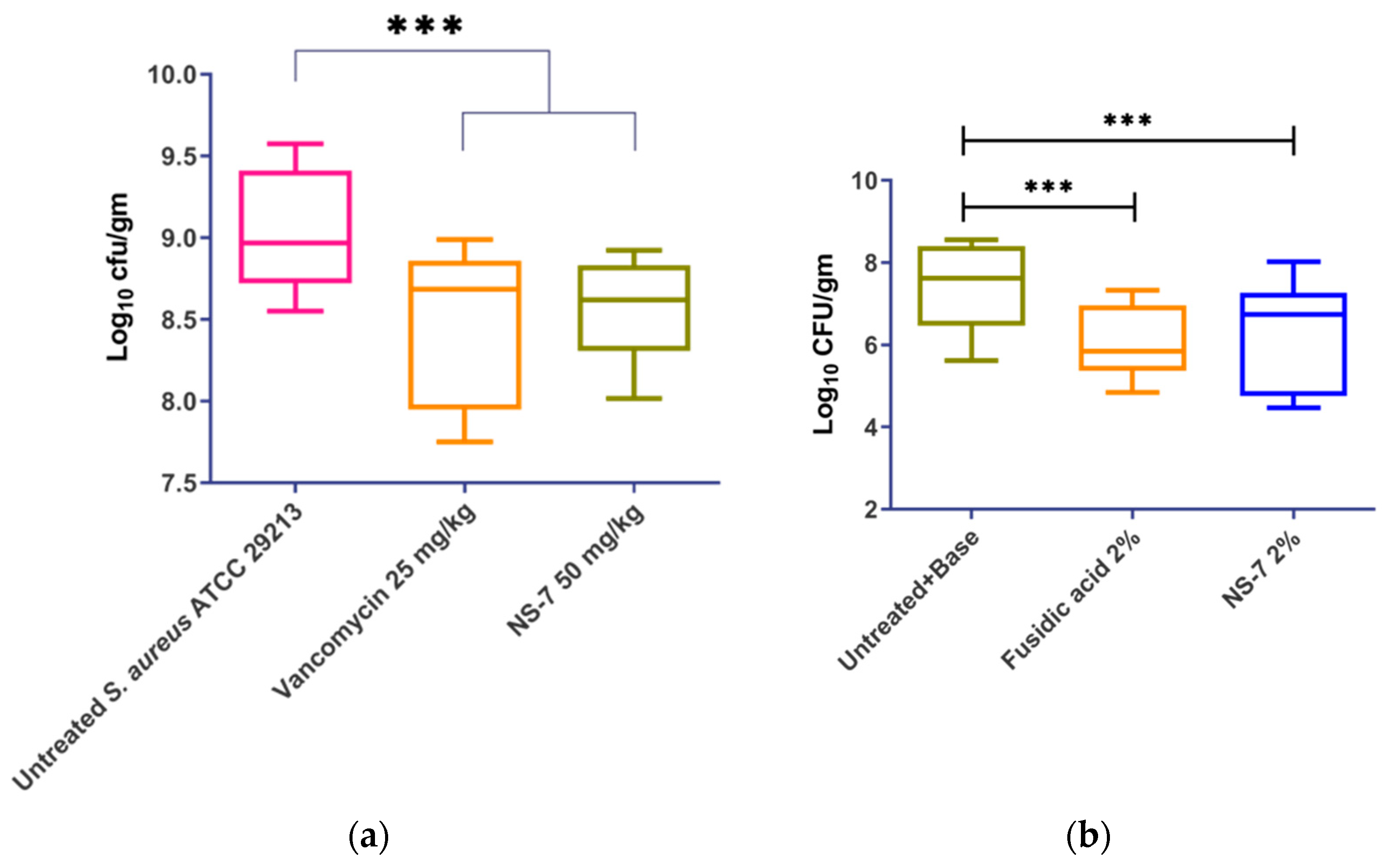
2.12. NS-7 Demonstrates Significant Efficacy in Murine Infection Models
3. Discussion
4. Materials and Methods
4.1. Plant Material
4.2. Extraction and Isolation
4.3. Reagents and Growth Media
4.4. Bacterial Strains
4.5. Antibacterial Susceptibility Testing against ESKAPE Panel of Bacteria
4.6. Outer Membrane Susceptibility Assay
4.7. Cell Viability Assay in Vero Cells
4.8. Activity against MDR Strains of S. aureus and Enterococcus Panel
4.9. Time–Kill Kinetics Study
4.10. Biofilm Eradication Assay
4.11. Intracellular Activity Assay
4.12. Synergy Screening of NS-7 with Frontline Antibiotics
4.13. Induced Resistant Mutant Generation Studies
4.14. Determination of Post-Antibiotic Effect (PAE)
4.15. Bacterial LIVE/DEAD BacLight Assay
4.16. Scanning Electron Microscopy (SEM)
4.17. Determination of Extracellular and Intracellular ATP
4.18. Murine Neutropenic Thigh Infection Model
4.19. Murine Skin Infection Model
5. Conclusions
6. Patents
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Turner, N.A.; Sharma-Kuinkel, B.K.; Maskarinec, S.A.; Eichenberger, E.M.; Shah, P.P.; Carugati, M.; Holland, T.L.; Fowler, V.G., Jr. Methicillin-Resistant Staphylococcus aureus: An Overview of Basic and Clinical Research. Nat. Rev. Microbiol. 2019, 17, 203–218. [Google Scholar] [CrossRef] [PubMed]
- Hiramatsu, K.; Katayama, Y.; Matsuo, M.; Sasaki, T.; Morimoto, Y.; Sekiguchi, A.; Baba, T. Multi-Drug-Resistant Staphylococcus aureus and Future Chemotherapy. J. Infect. Chemother. 2014, 20, 593–601. [Google Scholar] [CrossRef]
- Blair, J.M.A.; Webber, M.A.; Baylay, A.J.; Ogbolu, D.O.; Piddock, L.J.V. Molecular Mechanisms of Antibiotic Resistance. Nat. Rev. Microbiol. 2015, 13, 42–51. [Google Scholar] [CrossRef]
- Wright, G.D. Molecular Mechanisms of Antibiotic Resistance. Chem. Commun. 2011, 47, 4055–4061. [Google Scholar] [CrossRef]
- WHO. WHO Publishes List of Bacteria for Which New Antibiotics Are Urgently Needed; WHO: Geneva, Switzerland, 2017. [Google Scholar]
- Chopra, B.; Dhingra, A.K. Natural Products: A Lead for Drug Discovery and Development. Phytother. Res. 2021, 35, 4660–4702. [Google Scholar] [CrossRef] [PubMed]
- Rossiter, S.E.; Fletcher, M.H.; Wuest, W.M. Natural Products as Platforms to Overcome Antibiotic Resistance. Chem. Rev. 2017, 117, 12415–12474. [Google Scholar] [CrossRef] [PubMed]
- Wu, S.-C.; Liu, F.; Zhu, K.; Shen, J.-Z. Natural Products That Target Virulence Factors in Antibiotic-Resistant Staphylococcus aureus. J. Agric. Food Chem. 2019, 67, 13195–13211. [Google Scholar] [CrossRef]
- Porras, G.; Chassagne, F.; Lyles, J.T.; Marquez, L.; Dettweiler, M.; Salam, A.M.; Samarakoon, T.; Shabih, S.; Farrokhi, D.R.; Quave, C.L. Ethnobotany and the Role of Plant Natural Products in Antibiotic Drug Discovery. Chem. Rev. 2020, 121, 3495–3560. [Google Scholar] [CrossRef]
- Liu, K.; Huigens, R.W., III. Instructive Advances in Chemical Microbiology Inspired by Nature’s Diverse Inventory of Molecules. ACS Infect. Dis. 2019, 6, 541–562. [Google Scholar] [CrossRef]
- Abouelhassan, Y.; Garrison, A.T.; Yang, H.; Chávez-Riveros, A.; Burch, G.M.; Huigens, R.W., III. Recent Progress in Natural-Product-Inspired Programs Aimed to Address Antibiotic Resistance and Tolerance. J. Med. Chem. 2019, 62, 7618–7642. [Google Scholar] [CrossRef]
- Tyers, M.; Wright, G.D. Drug Combinations: A Strategy to Extend the Life of Antibiotics in the 21st Century. Nat. Rev. Microbiol. 2019, 17, 141–155. [Google Scholar] [CrossRef] [PubMed]
- Kaul, G.; Shukla, M.; Dasgupta, A.; Chopra, S. Update on Drug-Repurposing: Is It Useful for Tackling Antimicrobial Resistance? Future Microbiol. 2019, 14, 829–831. [Google Scholar] [CrossRef] [PubMed]
- Shin, J.; Prabhakaran, V.-S.; Kim, K. The Multi-Faceted Potential of Plant-Derived Metabolites as Antimicrobial Agents against Multidrug-Resistant Pathogens. MicrobPathog 2018, 116, 209–214. [Google Scholar] [CrossRef]
- Silva, L.N.; Zimmer, K.R.; Macedo, A.J.; Trentin, D.S. Plant Natural Products Targeting Bacterial Virulence Factors. Chem. Rev. 2016, 116, 9162–9236. [Google Scholar] [CrossRef] [PubMed]
- Davison, E.K.; Brimble, M.A. Natural Product Derived Privileged Scaffolds in Drug Discovery. Curr. Opin. Chem. Biol. 2019, 52, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Purushothaman, K.K.; Sarada, A.; Connolly, J.D. Malabaricones A–D, Novel Diarylnonanoids from Myristica malabarica Lam (Myristicaceae). J. Chem. Soc. Perkin Trans. 1 1977, 587–588. [Google Scholar] [CrossRef]
- Chelladurai, P.K.; Ramalingam, R. Myristica malabarica: A Comprehensive Review. J. Pharmacogn. Phytochem. 2017, 6, 255–258. [Google Scholar]
- Yin, W.; Xu, S.; Wang, Y.; Zhang, Y.; Chou, S.H.; Galperin, M.Y.; He, J. Ways to Control Harmful Biofilms: Prevention, Inhibition, and Eradication. Crit. Rev. Microbiol. 2021, 47, 57. [Google Scholar] [CrossRef]
- Craft, K.M.; Nguyen, J.M.; Berg, L.J.; Townsend, S.D. Methicillin-Resistant Staphylococcus aureus (MRSA): Antibiotic-Resistance and the Biofilm Phenotype. Medchemcomm 2019, 10, 1231–1241. [Google Scholar] [CrossRef]
- Ayaz, M.; Ullah, F.; Sadiq, A.; Ullah, F.; Ovais, M.; Ahmed, J.; Devkota, H.P. Synergistic Interactions of Phytochemicals with Antimicrobial Agents: Potential Strategy to Counteract Drug Resistance. Chem. Biol. Interact. 2019, 308, 294–303. [Google Scholar] [CrossRef]
- Odds, F.C. Synergy, Antagonism, and What the Chequerboard Puts between Them. J. Antimicrob. Chemother. 2003, 52, 1. [Google Scholar] [CrossRef] [PubMed]
- Beyer, P.; Paulin, S. The Antibacterial Research and Development Pipeline Needs Urgent Solutions. ACS Infect. Dis. 2020, 6, 1289–1291. [Google Scholar] [CrossRef]
- Patro, B.S.; Bauri, A.K.; Mishra, S.; Chattopadhyay, S. Antioxidant Activity of Myristica malabarica Extracts and Their Constituents. J. Agric. Food Chem. 2005, 53, 6912–6918. [Google Scholar] [CrossRef]
- Tyagi, M.; Patro, B.S.; Chattopadhyay, S. Mechanism of the Malabaricone C-Induced Toxicity to the MCF-7 Cell Line. Free Radic. Res. 2014, 48, 466–477. [Google Scholar] [CrossRef] [PubMed]
- Patro, B.S.; Tyagi, M.; Saha, J.; Chattopadhyay, S. Comparative Nuclease and Anti-Cancer Properties of the Naturally Occurring Malabaricones. Bioorg. Med. Chem. 2010, 18, 7043–7051. [Google Scholar] [CrossRef] [PubMed]
- Tyagi, M.; Bhattacharyya, R.; Bauri, A.K.; Patro, B.S.; Chattopadhyay, S. DNA Damage Dependent Activation of Checkpoint Kinase-1 and Mitogen-Activated Protein Kinase-P38 Are Required in Malabaricone C-Induced Mitochondrial Cell Death. Biochim. Et. Biophys. Acta BBA-Gen. Subj. 2014, 1840, 1014–1027. [Google Scholar] [CrossRef]
- Manna, A.; De Sarkar, S.; De, S.; Bauri, A.K.; Chattopadhyay, S.; Chatterjee, M. Impact of MAPK and PI3K/AKT Signaling Pathways on Malabaricone-A Induced Cytotoxicity in U937, a Histiocytic Lymphoma Cell Line. Int. Immunopharmacol. 2016, 39, 34–40. [Google Scholar] [CrossRef]
- Kang, J.; Tae, N.; Min, B.S.; Choe, J.; Lee, J.-H. Malabaricone C Suppresses Lipopolysaccharide-Induced Inflammatory Responses via Inhibiting ROS-Mediated Akt/IKK/NF-ΚB Signaling in Murine Macrophages. Int. Immunopharmacol. 2012, 14, 302–310. [Google Scholar] [CrossRef]
- Maity, B.; Yadav, S.K.; Patro, B.S.; Tyagi, M.; Bandyopadhyay, S.K.; Chattopadhyay, S. Molecular Mechanism of the Anti-Inflammatory Activity of a Natural Diarylnonanoid, Malabaricone C. Free Radic. Biol. Med. 2012, 52, 1680–1691. [Google Scholar] [CrossRef]
- Rathee, J.S.; Patro, B.S.; Brown, L.; Chattopadhyay, S. Mechanism of the Anti-Hypertensive Property of the Naturally Occurring Phenolic, Malabaricone C in DOCA-Salt Rats. Free Radic. Res. 2016, 50, 111–121. [Google Scholar] [CrossRef]
- Choi, N.H.; Choi, G.J.; Jang, K.S.; Choi, Y.H.; Lee, S.O.; Choi, J.E.; Kim, J.C. Antifungal Activity of the Methanol Extract of Myristica malabarica Fruit Rinds and the Active Ingredients Malabaricones against Phytopathogenic Fungi. Plant Pathol. J. 2008, 24, 317–321. [Google Scholar] [CrossRef]
- Orabi, K.Y.; Mossa, J.S.; El-Feraly, F.S. Isolation and Characterization of Two Antimicrobial Agents from Mace (Myristica fragrans). J. Nat. Prod. 1991, 54, 856–859. [Google Scholar] [CrossRef] [PubMed]
- Shinohara, C.; Mori, S.; Ando, T.; Tsuji, T. Arg-Gingipain Inhibition and Anti-Bacterial Activity Selective for Porphyromonas Gingivalis by Malabaricone C. Biosci. Biotechnol. Biochem. 1999, 63, 1475–1477. [Google Scholar]
- Chong, Y.M.; Yin, W.F.; Ho, C.Y.; Mustafa, M.R.; Hadi, A.H.A.; Awang, K.; Narrima, P.; Koh, C.-L.; Appleton, D.R.; Chan, K.-G. Malabaricone C from Myristica Cinnamomea Exhibits Anti-Quorum Sensing Activity. J. Nat. Prod. 2011, 74, 2261–2264. [Google Scholar] [CrossRef]
- Nakamura, N.; Kiuchi, F.; Tsuda, Y.; Kondo, K. Studies on Crude Drugs Effective on Visceral Larva Migrans. V.: The Larvicidal Principle in Mace (Aril of Myristica fragrans). Chem. Pharm. Bull. 1988, 36, 2685–2688. [Google Scholar] [CrossRef]
- Craig, W.A. The Postantibiotic Effect. Clin. Microbiol. Newsl. 1991, 13, 121–124. [Google Scholar] [CrossRef]
- CLSI. Performance Standards for Antimicrobial Susceptibility Testing—30th Edition: M100; Clinical and Laboratory Standards Institute: Wayne, PA, USA, 2000. [Google Scholar]
- Thangamani, S.; Mohammad, H.; Abushahba, M.F.N.; Sobreira, T.J.P.; Hedrick, V.E.; Paul, L.N.; Seleem, M.N. Antibacterial Activity and Mechanism of Action of Auranofin against Multi-Drug Resistant Bacterial Pathogens. Sci. Rep. 2016, 6, 22571. [Google Scholar] [CrossRef]
- Meenu, M.T.; Kaul, G.; Akhir, A.; Shukla, M.; Radhakrishnan, K.V.; Chopra, S. Developing the Natural Prenylflavone Artocarpin from Artocarpus Hirsutus as a Potential Lead Targeting Pathogenic, Multidrug-Resistant Staphylococcus aureus, Persisters and Biofilms with No Detectable Resistance. J. Nat. Prod. 2022, 85, 2413–2423. [Google Scholar] [CrossRef]
- Grace, K.; Abdul, A.; Manjulika, S.; Hasham, S.; Ravikumar, A.; Gaurav, P.; Mahammad, G.; Nanduri, S.; Sidharth, C. Oxiconazole Potentiates Gentamicin against Gentamicin-Resistant Staphylococcus aureus In Vitro and In Vivo. Microbiol. Spectr. 2023, 11, e05031-22. [Google Scholar] [CrossRef]
- Seral, C.; Van Bambeke, F.; Tulkens, P.M. Quantitative Analysis of Gentamicin, Azithromycin, Telithromycin, Ciprofloxacin, Moxifloxacin, and Oritavancin (LY333328) Activities against Intracellular Staphylococcus aureus in Mouse J774 Macrophages. Antimicrob. Agents Chemother. 2003, 47, 2283–2292. [Google Scholar] [CrossRef]
- Suller, M.T.E.; Lloyd, D. The Antibacterial Activity of Vancomycin towards Staphylococcus aureus under Aerobic and Anaerobic Conditions. J. Appl. Microbiol. 2002, 92, 866–872. [Google Scholar] [CrossRef] [PubMed]
- Kaul, G.; Akhir, A.; Shukla, M.; Rawat, K.S.; Sharma, C.P.; Sangu, K.G.; Rode, H.B.; Goel, A.; Chopra, S. Nitazoxanide Potentiates Linezolid against Linezolid-Resistant Staphylococcus aureus In Vitro and In Vivo. J. Antimicrob. Chemother. 2022, 77, 2456–2460. [Google Scholar] [CrossRef] [PubMed]
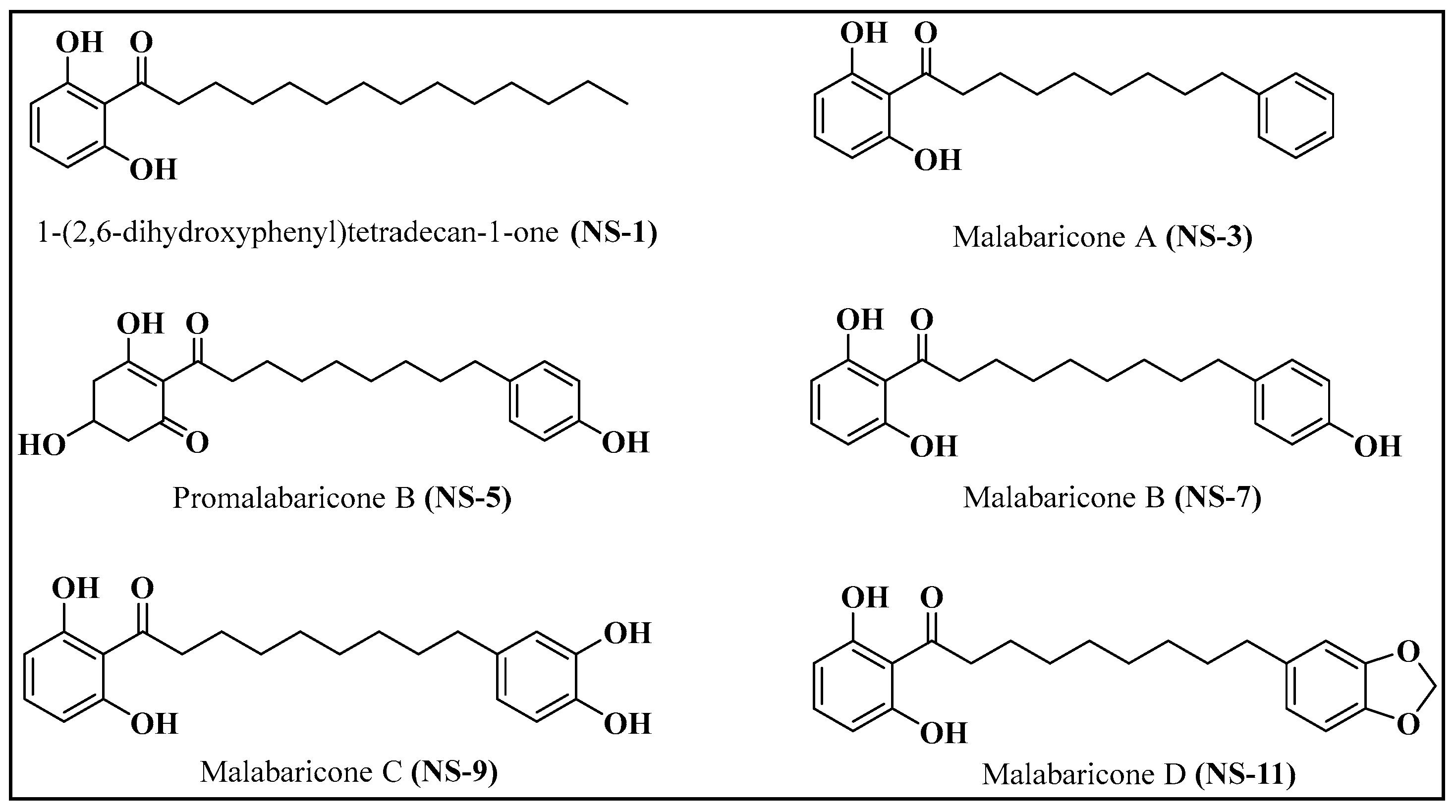

| Bacterial Strains | NS-1 | NS-3 | NS-5 | NS-7 | NS-9 | NS-11 | Levofloxacin |
|---|---|---|---|---|---|---|---|
| S. aureus ATCC 29213 | >128 | >128 | 16 | 0.5 | 4 | 1 | 0.125 |
| Enterococcus NR 31903 | NT | NT | NT | 2 | NT | NT | >64 |
| K. pneumoniae BAA 1705 | >128 | >128 | >128 | >128 | >128 | >128 | 64 |
| A. baumannii BAA 1605 | >128 | >128 | >128 | >128 | >128 | >128 | 8 |
| P. aeruginosa ATCC 27853 | >128 | >128 | >128 | >128 | >128 | >128 | 0.5 |
| E. coli ATCC 25922 | >128 | >128 | >128 | >128 | >128 | >128 | 0.03 |
| Bacterial Strains | MIC (µg/mL) | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| PMBN | NS-7 | Rifampicin | Vancomycin | Levofloxacin | |||||
| PMBN | PMBN | PMBN | PMBN | ||||||
| (−) | (+) | (−) | (+) | (−) | (+) | (−) | (+) | ||
| E. coli (ATCC 25922) | >64 | >512 | 2 | 8 | 0.06 | 512 | 128 | 0.03 | 0.015 |
| A. baumannii (ATCC BAA-1605) | >64 | >512 | 4 | 2 | 0.03 | 256 | 64 | 8 | 8 |
| Compound | MIC (µg/mL) against S. aureus ATCC 29213 | CC50 (µg/mL) | Selectivity Index (CC50/MIC) |
|---|---|---|---|
| NS-7 | 0.5 | >40 | >80 |
| Strains | MIC (µg/mL) | ||||
|---|---|---|---|---|---|
| NS-7 | Levofloxacin | Methicillin | Vancomycin | ||
| MSSA | S. aureus ATCC 29213 | 1 | 0.125 | 1 | 1 |
| MRSA | NRS 100 | 1 | 0.125 | >64 | 2 |
| NRS 119 | 1 | 16 | >64 | 2 | |
| NRS 129 | 1 | 0.125 | 64 | 1 | |
| NRS198 | 1 | 32 | >64 | 2 | |
| NRS192 | 1 | 8 | >64 | 1 | |
| NRS191 | 1 | 32 | >64 | 2 | |
| NRS193 | 2 | 32 | >64 | 2 | |
| NRS186 | 2 | 8 | 64 | 2 | |
| NRS194 | 1 | 0.125 | 16 | 1 | |
| VRSA | VRS 1 | 1 | 64 | >64 | >64 |
| VRS 4 | 2 | >64 | >64 | >64 | |
| VRS 12 | 2 | 64 | 64 | >64 | |
| VSE | NR 31884 | 2 | 0.5 | 64 | 2 |
| NR 31885 | 2 | 0.5 | >64 | 2 | |
| NR 31886 | 2 | 0.5 | >64 | 2 | |
| NR 31887 | 2 | 0.5 | 16 | 1 | |
| NR 31888 | 2 | 0.5 | >64 | 1 | |
| VRE | NR 31903 | 2 | >64 | >64 | >64 |
| NR 31909 | 2 | >64 | >64 | >64 | |
| NR 31912 | 1 | >64 | >64 | >64 | |
| Compound/Drug | MIC (µg/mL) | MIC (µg/mL) of NS-7 “A” in Combination with Antibiotic | MIC (µg/mL) of Antibiotic “B” in Combination with NS-7 | FIC A | FIC B | ΣFIC (FIC A + FIC B) | Inference |
|---|---|---|---|---|---|---|---|
| NS-7 | 2 | ||||||
| Gentamicin | 0.5 | 0.06 | 0.125 | 0.03 | 0.25 | 0.28 | Synergy |
| Daptomycin | 1 | 0.06 | 0.25 | 0.03 | 0.25 | 0.28 | Synergy |
| Ceftazidime | 16 | 2 | 16 | 1 | 1 | 2 | No interaction |
| Levofloxacin | 0.25 | 2 | 0.25 | 1 | 1 | 2 | No interaction |
| Linezolid | 4 | 1 | 2 | 0.5 | 0.5 | 1 | No interaction |
| Meropenem | 1 | 2 | 1 | 1 | 1 | 2 | No interaction |
| Minocycline | 0.25 | 2 | 0.25 | 1 | 1 | 2 | No interaction |
| Rifampicin | 0.0078 | 2 | 0.0078 | 1 | 1 | 2 | No interaction |
| Vancomycin | 1 | 1 | 0.5 | 0.5 | 0.5 | 1 | No interaction |
| Untreated | Levofloxacin 1X | Levofloxacin 10X | Vancomycin 1X | Vancomycin 10X | NS-7 1X | NS-7 10X | |
|---|---|---|---|---|---|---|---|
| Time for 1 log10 (h) | ~2 | ~2.5 | ~3.5 | ~3 | ~3.5 | ~3 | >24 |
| PAE (h) | 0 | ~0.5 | ~1.5 | ~1 | ~1.5 | ~1 | >22 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sivadas, N.; Kaul, G.; Akhir, A.; Shukla, M.; Govind, M.G.; Dan, M.; Radhakrishnan, K.V.; Chopra, S. Naturally Derived Malabaricone B as a Promising Bactericidal Candidate Targeting Multidrug-Resistant Staphylococcus aureus also Possess Synergistic Interactions with Clinical Antibiotics. Antibiotics 2023, 12, 1483. https://doi.org/10.3390/antibiotics12101483
Sivadas N, Kaul G, Akhir A, Shukla M, Govind MG, Dan M, Radhakrishnan KV, Chopra S. Naturally Derived Malabaricone B as a Promising Bactericidal Candidate Targeting Multidrug-Resistant Staphylococcus aureus also Possess Synergistic Interactions with Clinical Antibiotics. Antibiotics. 2023; 12(10):1483. https://doi.org/10.3390/antibiotics12101483
Chicago/Turabian StyleSivadas, Neethu, Grace Kaul, Abdul Akhir, Manjulika Shukla, Murugan Govindakurup Govind, Mathew Dan, Kokkuvayil Vasu Radhakrishnan, and Sidharth Chopra. 2023. "Naturally Derived Malabaricone B as a Promising Bactericidal Candidate Targeting Multidrug-Resistant Staphylococcus aureus also Possess Synergistic Interactions with Clinical Antibiotics" Antibiotics 12, no. 10: 1483. https://doi.org/10.3390/antibiotics12101483
APA StyleSivadas, N., Kaul, G., Akhir, A., Shukla, M., Govind, M. G., Dan, M., Radhakrishnan, K. V., & Chopra, S. (2023). Naturally Derived Malabaricone B as a Promising Bactericidal Candidate Targeting Multidrug-Resistant Staphylococcus aureus also Possess Synergistic Interactions with Clinical Antibiotics. Antibiotics, 12(10), 1483. https://doi.org/10.3390/antibiotics12101483






